A Systematic Review of Bisphenol A from Dietary and Non-Dietary Sources during Pregnancy and Its Possible Connection with Fetal Growth Restriction: Investigating Its Potential Effects and the Window of Fetal Vulnerability
Abstract
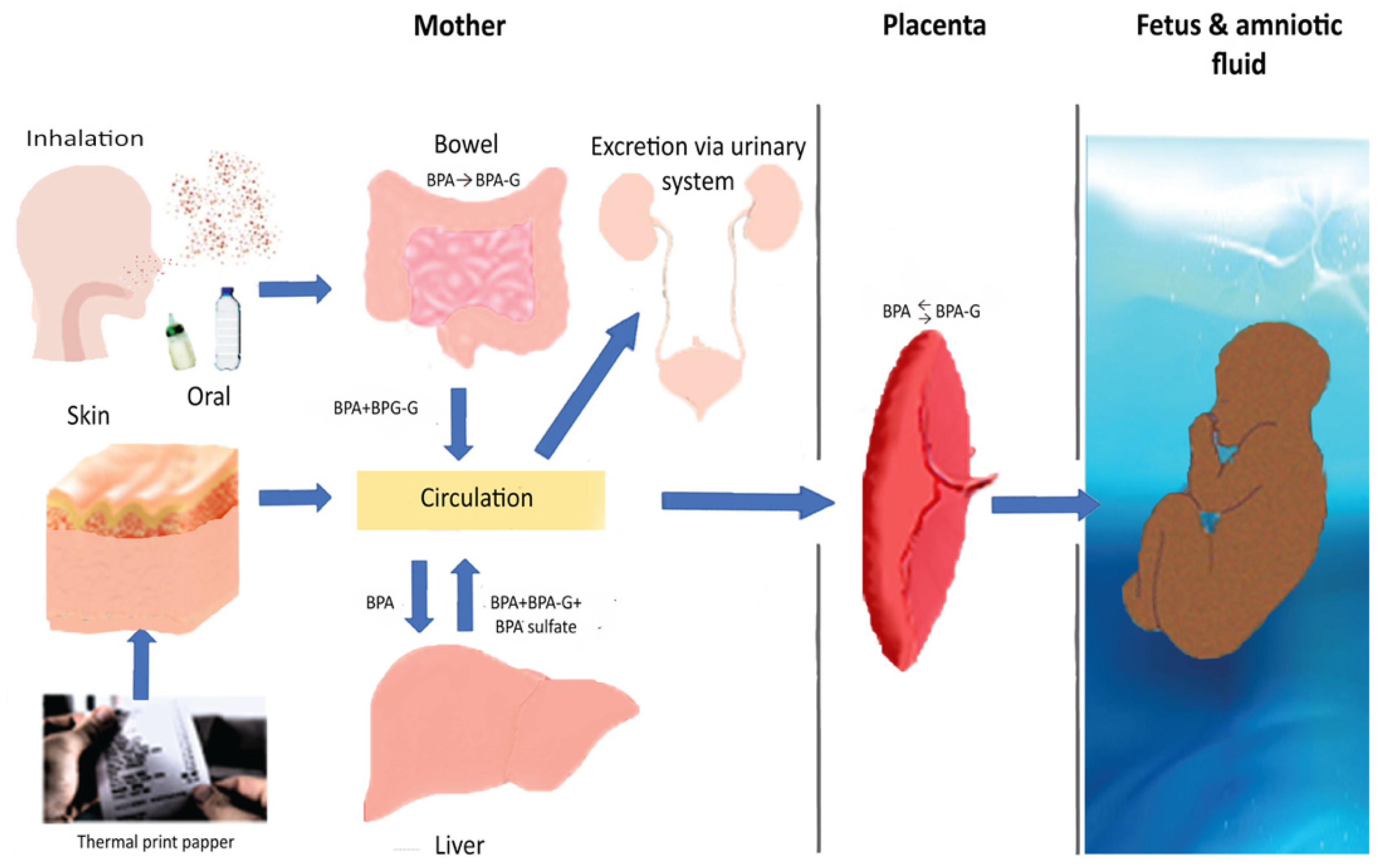
:1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Eligibility Criteria
2.3. Study Selection Progress
2.4. Quality Assessment
3. Results
3.1. Fetal Growth Studies Examining First- and Second-Trimester Exposure
3.2. Fetal Growth Studies Examining Third-Trimester Exposure and Delivery
3.3. Fetal Growth Studies with Multiple Samples and US Scan Assessments throughout Pregnancy
4. Discussion
Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Li, L.; Wang, Q.; Zhang, Y.; Niu, Y.; Yao, X.; Liu, H. The Molecular Mechanism of Bisphenol A (BPA) as an Endocrine Disruptor by Interacting with Nuclear Receptors: Insights from Molecular Dynamics (MD) Simulations. PLoS ONE 2015, 10, e0120330. [Google Scholar] [CrossRef] [PubMed]
- Stejskalova, L.; Pavek, P. The Function of Cytochrome P450 1A1 Enzyme (CYP1A1) and Aryl Hydrocarbon Receptor (AhR) in the Placenta. Curr. Pharm. Biotechnol. 2011, 12, 715–730. [Google Scholar] [CrossRef] [PubMed]
- Acconcia, F.; Pallottini, V.; Marino, M. Molecular Mechanisms of Action of BPA. Dose-Response 2015, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chu, P.W.; Yang, Z.J.; Huang, H.H.; Chang, A.A.; Cheng, Y.C.; Wu, G.J.; Lan, H.C. Low-dose bisphenol A activates the ERK signaling pathway and attenuates steroidogenic gene expression in human placental cells. Biol. Reprod. 2018, 98, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Sieppi, E.; Vähäkangas, K.; Rautio, A.; Ietta, F.; Paulesu, L.; Myllynen, P. The xenoestrogens, bisphenol A and para-nonylphenol, decrease the expression of the ABCG2 transporter protein in human term placental explant cultures. Mol. Cell. Endocrinol. 2016, 429, 41–49. [Google Scholar] [CrossRef] [PubMed]
- Ye, Y.; Tang, Y.; Xiong, Y.; Feng, L.; Li, X. Bisphenol A exposure alters placentation and causes preeclampsia-like features in pregnant mice involved in reprogramming of DNA methylation ofWNT2. FASEB J. 2019, 33, 2732–2742. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strakovsky, R.S.; Schantz, S.L. Impacts of bisphenol A (BPA) and phthalate exposures on epigenetic outcomes in the human placenta. Environ. Epigenet. 2018, 4, dvy022. [Google Scholar] [CrossRef] [Green Version]
- Basak, S.; Srinivas, V.; Duttaroy, A.K. Bisphenol-A impairs cellular function and alters DNA methylation of stress pathway genes in first trimester trophoblast cells. Reprod. Toxicol. 2018, 82, 72–79. [Google Scholar] [CrossRef]
- Xiao, X.; Zhao, Y.; Jin, R.; Chen, J.; Wang, X.; Baccarelli, A.; Zhang, Y. Fetal growth restriction and methylation of growth-related genes in the placenta. Epigenomics 2016, 8, 33–42. [Google Scholar] [CrossRef] [Green Version]
- Ferreira, J.C.; Choufani, S.; Grafodatskaya, D.; Butcher, D.T.; Zhao, C.; Chitayat, D.; Shuman, C.; Kingdom, J.; Keating, S.; Weksberg, R. WNT2 promoter methylation in human placenta is associated with low birthweight percentile in the neonate. Epigenetics 2011, 6, 440–449. [Google Scholar] [CrossRef] [Green Version]
- Chiofalo, B.; Laganà, A.S.; Vaiarelli, A.; La Rosa, V.L.; Rossetti, D.; Palmara, V.; Valenti, G.; Rapisarda, A.M.C.; Granese, R.; Sapia, F.; et al. Do miRNAs Play a Role in Fetal Growth Restriction? A Fresh Look to a Busy Corner. Biomed. Res. Int. 2017, 2017. [Google Scholar] [CrossRef]
- Mouillet, J.-F.; Ouyang, Y.; Coyne, C.B.; Sadovsky, Y. MicroRNAs in placental health and disease. Am. J. Obstet. Gynecol. 2015, 213, S163–S172. [Google Scholar] [CrossRef] [Green Version]
- Lombó, M.; Fernández-Díez, C.; González-Rojo, S.; Herráez, M.P. Genetic and epigenetic alterations induced by bisphenol A exposure during different periods of spermatogenesis: From spermatozoa to the progeny. Sci. Rep. 2019, 9, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almeida, S.; Raposo, A.; Almeida-González, M.; Carrascosa, C. Bisphenol A: Food Exposure and Impact on Human Health. Compr. Rev. Food Sci. Food Saf. 2018, 17, 1503–1517. [Google Scholar] [CrossRef] [Green Version]
- Liao, C.; Kannan, K. Concentrations and profiles of bisphenol A and other bisphenol analogues in foodstuffs from the United States and their implications for human exposure. J. Agric. Food Chem. 2013, 61, 4655–4662. [Google Scholar] [CrossRef] [PubMed]
- Vrachnis, N.; Loukas, N.; Vrachnis, D.; Antonakopoulos, N.; Christodoulaki, C.; Tsonis, O.; George, M.; Iliodromiti, Z. Phthalates and fetal growth velocity: Tracking down the suspected links. J. Matern. Neonatal Med. 2021, 1–14. [Google Scholar] [CrossRef]
- Matuszczak, E.; Komarowska, M.D.; Debek, W.; Hermanowicz, A. The Impact of Bisphenol A on Fertility, Reproductive System, and Development: A Review of the Literature. Int. J. Endocrinol. 2019, 2019, 4068717. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yacoumas, A.; Honvault, N.; Houben, D.; Fontaine, J.; Meglouli, H.; Laruelle, F.; Tisserant, B.; Faucon, M.-P.; Sahraoui, A.L.-H.; Firmin, S. Contrasting Response of Nutrient Acquisition Traits in Wheat Grown on Bisphenol A-Contaminated Soils. Water Air Soil Pollut. 2020, 231, 1–13. [Google Scholar] [CrossRef]
- Bisphenol A (BPA): Use in Food Contact Application. Available online: https://www.fda.gov/food/food-additives-petitions/bisphenol-bpa-use-food-contact-application (accessed on 29 May 2021).
- European Food Safety Authority. Available online: https://www.efsa.europa.eu/en/topics/topic/bisphenol (accessed on 4 July 2021).
- Andújar, N.; Gálvez-Ontiveros, Y.; Zafra-Gómez, A.; Rodrigo, L.; Álvarez-Cubero, M.J.; Aguilera, M.; Monteagudo, C.; Rivas, A.A. Bisphenol A Analogues in Food and Their Hormonal and Obesogenic Effects: A Review. Nutrients 2019, 11, 2136. [Google Scholar] [CrossRef] [Green Version]
- Vandenberg, L.N.; Chahoud, I.; Heindel, J.J.; Padmanabhan, V.; Paumgartten, F.J.R.; Schoenfelder, G. Urinary, Circulating, and Tissue Biomonitoring Studies Indicate Widespread Exposure to Bisphenol A. Environ. Health Perspect. 2010, 118, 1055–1070. [Google Scholar] [CrossRef] [Green Version]
- Braun, J.M.; Smith, K.W.; Williams, P.L.; Calafat, A.M.; Berry, K.; Ehrlich, S.; Hauser, R. Variability of Urinary Phthalate Metabolite and Bisphenol A Concentrations before and during Pregnancy. Environ. Health Perspect. 2012, 120, 739–745. [Google Scholar] [CrossRef]
- Pergialiotis, V.; Kotrogianni, P.; Christopoulos-Timogiannakis, E.; Koutaki, D.; Daskalakis, G.; Papantoniou, N. Bisphenol A and adverse pregnancy outcomes: A systematic review of the literature. J. Matern. Neonatal Med. 2017, 31, 3320–3327. [Google Scholar] [CrossRef]
- Ejaredar, M.; Lee, Y.; Roberts, D.J.; Sauve, R.; Dewey, D. Bisphenol A exposure and children’s behavior: A systematic review. J. Expo. Sci. Environ. Epidemiol. 2016, 27, 175–183. [Google Scholar] [CrossRef] [PubMed]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef]
- Lodge, C.; Dharmage, S.C. Breastfeeding and perinatal exposure, and the risk of asthma and allergies. Curr. Opin. Allergy Clin. Immunol. 2016, 16, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Gerona, R.R.; Woodruff, T.J.; Dickenson, C.A.; Pan, J.; Schwartz, J.M.; Sen, S.; Friesen, M.W.; Fujimoto, V.Y.; Hunt, P.A. Bisphenol-A (BPA), BPA glucuronide, and BPA sulfate in mid-gestation umbilical cord serum in a Northern and Central California population. Environ. Sci. Technol. 2013, 47, 12477–12485. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Karrer, C.; Roiss, T.; von Goetz, N.; Gramec Skledar, D.; Peterlin Mašič, L.; Hungerbühler, K. Physiologically Based Pharmacokinetic (PBPK) Modeling of the Bisphenols BPA, BPS, BPF, and BPAF with New Experimental Metabolic Parameters: Comparing the Pharmacokinetic Behavior of BPA with Its Substitutes. Environ. Health Perspect. 2018, 126, 077002. [Google Scholar] [CrossRef]
- Gluckman, P.D.; Hanson, M.; Cooper, C.; Thornburg, K. Effect of In Utero and Early-Life Conditions on Adult Health and Disease. N. Engl. J. Med. 2008, 359, 61–73. [Google Scholar] [CrossRef] [Green Version]
- Royal College of Obstetricians and Gynaecologists. Available online: https://www.rcog.org.uk/globalassets/documents/guidelines/gtg_31.pdf (accessed on 4 July 2021).
- Barker, D.J. Adult consequences of fetal growth restriction. Clin. Obstet. Gynecol. 2006, 49, 270–283. [Google Scholar] [CrossRef] [PubMed]
- Vrachnis, N.; Botsis, D.; Iliodromiti, Z. The Fetus That Is Small for Gestational Age. Ann. N. Y. Acad. Sci. 2006, 1092, 304–309. [Google Scholar] [CrossRef]
- Zbucka-Krȩtowska, M.; Łazarek, U.; Miltyk, W.; Sidorkiewicz, I.; Pierzyński, P.; Milewski, R.; Wołczyński, S.; Czerniecki, J. Simultaneous analysis of bisphenol A fractions in maternal and fetal compartments in early second trimester of pregnancy. J. Perinat. Med. 2019, 47, 765–770. [Google Scholar] [CrossRef] [PubMed]
- Pinney, S.E.; Mesaros, C.A.; Snyder, N.W.; Busch, C.M.; Xiao, R.; Aijaz, S.; Ijaz, N.; Blair, I.A.; Manson, J.M. Second trimester amniotic fluid bisphenol A concentration is associated with decreased birth weight in term infants. Reprod. Toxicol. 2017, 67, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Burstyn, I.; Martin, J.; Beesoon, S.; Bamforth, F.; Li, Q.; Yasui, Y.; Cherry, N. Maternal Exposure to Bisphenol-A and Fetal Growth Restriction: A Case-Referent Study. Int. J. Environ. Res. Public Health 2013, 10, 7001–7014. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Philippat, C.; Botton, J.; Calafat, A.M.; Ye, X.; Charles, M.-A.; Slama, R. Prenatal Exposure to Phenols and Growth in Boys. Epidemiology 2014, 25, 625–635. [Google Scholar] [CrossRef]
- Philippat, C.; Heude, B.; Botton, J.; Alfaidy, N.; Calafat, A.M.; Slama, R. Prenatal Exposure to Select Phthalates and Phenols and Associations with Fetal and Placental Weight among Male Births in the EDEN Cohort (France). Environ. Health Perspect. 2019, 127, 017002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, B.-E.; Park, H.; Hong, Y.-C.; Ha, M.; Kim, Y.; Chang, N.; Kim, B.-N.; Kim, Y.J.; Yu, S.-D.; Ha, E.-H. Prenatal bisphenol A and birth outcomes: MOCEH (Mothers and Children’s Environmental Health) study. Int. J. Hyg. Environ. Health 2014, 217, 328–334. [Google Scholar] [CrossRef]
- Lee, Y.M.; Hong, Y.-C.; Ha, M.; Kim, Y.; Park, H.; Kim, H.S.; Ha, E.-H. Prenatal Bisphenol-A exposure affects fetal length growth by maternal glutathione transferase polymorphisms, and neonatal exposure affects child volume growth by sex: From multiregional prospective birth cohort MOCEH study. Sci. Total Environ. 2018, 612, 1433–1441. [Google Scholar] [CrossRef]
- Dalkan, C.; Uncu, M.; Duran, S.; Bahçeciler, N.N. Association of cord blood bisphenol A (BPA) with cord blood adiponectin, leptin, fetal growth; adiposity and neoantal complications in a newborn cohort. J. Matern. Neonatal Med. 2019, 33, 2588–2593. [Google Scholar] [CrossRef]
- Chou, W.-C.; Chen, J.-L.; Lin, C.-F.; Chen, Y.-C.; Shih, F.-C.; Chuang, C.-Y. Biomonitoring of bisphenol A concentrations in maternal and umbilical cord blood in regard to birth outcomes and adipokine expression: A birth cohort study in Taiwan. Environ. Health 2011, 10, 94. [Google Scholar] [CrossRef] [Green Version]
- Gounden, V.; Zain Warasally, M.; Magwai, T.; Naidoo, R.; Chuturgoon, A. A pilot study: Bisphenol-A and Bisphenol-A glucuronide levels in mother and child pairs in a South African population. Reprod. Toxicol. 2019, 89, 93–99. [Google Scholar] [CrossRef]
- Xu, X.; Chiung, Y.M.; Lu, F.; Qiu, S.; Ji, M.; Huo, X. Associations of cadmium, bisphenol A and polychlorinated biphenyl co-exposure in utero with placental gene expression and neonatal outcomes. Reprod. Toxicol. 2015, 52, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, X.; Chen, Q.; Luo, Z.-C.; Zhao, S.; Wang, W.; Zhang, H.-J.; Zhang, J.; Ouyang, F. Urinary Bisphenol A Concentration and Gestational Diabetes Mellitus in Chinese Women. Epidemiology 2017, 28, S41–S47. [Google Scholar] [CrossRef]
- Troisi, J.; Mikelson, C.; Richards, S.; Symes, S.; Adair, D.; Zullo, F.; Guida, M. Placental concentrations of bisphenol A and birth weight from births in the Southeastern U.S. Placenta 2014, 35, 947–952. [Google Scholar] [CrossRef]
- Huo, W.; Xia, W.; Wan, Y.; Zhang, B.; Zhou, A.; Zhang, Y.; Huang, K.; Zhu, Y.; Wu, C.; Peng, Y.; et al. Maternal urinary bisphenol A levels and infant low birth weight: A nested case–control study of the Health Baby Cohort in China. Environ. Int. 2015, 85, 96–103. [Google Scholar] [CrossRef]
- Ding, G.; Wang, C.; Vinturache, A.; Zhao, S.; Pan, R.; Han, W.; Chen, L.; Wang, W.; Yuan, T.; Gao, Y.; et al. Prenatal low-level phenol exposures and birth outcomes in China. Sci. Total Environ. 2017, 607–608, 1400–1407. [Google Scholar] [CrossRef]
- Tang, R.; Chen, M.-J.; Ding, G.-D.; Chen, X.-J.; Han, X.-M.; Zhou, K.; Chen, L.-M.; Xia, Y.-K.; Tian, Y.; Wang, X.-R. Associations of prenatal exposure to phenols with birth outcomes. Environ. Pollut. 2013, 178, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, V.; Siefert, K.; Ransom, S.M.; Johnson, T.R.B.; Pinkerton, J.; Anderson, L.; Tao, L.; Kannan, K. Maternal bisphenol-A levels at delivery: A looming problem? J. Perinatol. 2008, 28, 258–263. [Google Scholar] [CrossRef] [Green Version]
- Veiga-Lopez, A.; Kannan, K.; Liao, C.; Ye, W.; Domino, S.E.; Padmanabhan, V. Gender-Specific Effects on Gestational Length and Birth Weight by Early Pregnancy BPA Exposure. J. Clin. Endocrinol. Metab. 2015, 100, E1394–E1403. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-F.; Pan, W.-C.; Tsai, Y.-A.; Chang, C.-H.; Chen, P.-J.; Shao, Y.-S.; Tsai, M.-S.; Hou, J.-W.; Lu, C.; Chen, M.-L. Concurrent exposures to nonylphenol, bisphenol A, phthalates, and organophosphate pesticides on birth outcomes: A cohort study in Taipei, Taiwan. Sci. Total. Environ. 2017, 607-608, 1126–1135. [Google Scholar] [CrossRef] [PubMed]
- Mustieles, V.; Williams, P.L.; Fernandez, M.F.; Mínguez-Alarcón, L.; Ford, J.B.; Calafat, A.M.; Hauser, R.; Messerlian, C. Maternal and paternal preconception exposure to bisphenols and size at birth. Hum. Reprod. 2018, 33, 1528–1537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aker, A.M.; Ferguson, K.; Rosario, Z.Y.; Mukherjee, B.; Alshawabkeh, A.N.; Cordero, J.F.; Meeker, J.D. The associations between prenatal exposure to triclocarban, phenols and parabens with gestational age and birth weight in northern Puerto Rico. Environ. Res. 2019, 169, 41–51. [Google Scholar] [CrossRef]
- Woods, M.M.; Lanphear, B.P.; Braun, J.M.; McCandless, L.C. Gestational exposure to endocrine disrupting chemicals in relation to infant birth weight: A Bayesian analysis of the HOME Study. Environ. Health 2017, 16, 1–12. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferguson, K.K.; Meeker, J.D.; Cantonwine, D.E.; Chen, Y.H.; Mukherjee, B.; McElrath, T.F. Urinary phthalate metabolite and bisphenol A associations with ultrasound and delivery indices of fetal growth. Environ. Int. 2016, 94, 531–537. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casas, M.; Valvi, D.; Ballesteros-Gómez, A.; Gascon, M.; Fernández, M.F.; Garcia-Esteban, R.; Iñiguez, C.; Martínez, D.; Murcia, M.; Monfort, N.; et al. Exposure to Bisphenol A and Phthalates during Pregnancy and Ultrasound Measures of Fetal Growth in the INMA-Sabadell Cohort. Environ. Health Perspect. 2016, 124, 521–528. [Google Scholar] [CrossRef]
- Snijder, C.A.; Heederik, D.; Pierik, F.H.; Hofman, A.; Jaddoe, V.W.; Koch, H.M.; Longnecker, M.P.; Burdorf, A. Fetal Growth and Prenatal Exposure to Bisphenol A: The Generation R Study. Environ. Health Perspect. 2013, 121, 393–398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/nosgen.pdf (accessed on 13 July 2021).
- Bell, E.M.; Yeung, E.H.; Ma, W.; Kannan, K.; Sundaram, R.; Smarr, M.M.; Louis, G.M.B. Concentrations of endocrine disrupting chemicals in newborn blood spots and infant outcomes in the upstate KIDS study. Environ. Int. 2018, 121, 232–239. [Google Scholar] [CrossRef]
- Smarr, M.M.; Grantz, K.L.; Sundaram, R.; Maisog, J.M.; Kannan, K.; Louis, G.M. Parental urinary biomarkers of preconception exposure to bisphenol A and phthalates in relation to birth outcomes. Environ. Health 2015, 14, 73. [Google Scholar] [CrossRef] [Green Version]
- Miao, M.; Yuan, W.; Zhu, G.; He, X.; Li, D.-K. In utero exposure to bisphenol-A and its effect on birth weight of offspring. Reprod. Toxicol. 2011, 32, 64–68. [Google Scholar] [CrossRef] [PubMed]
- Vrachnis, N.; Karavolos, S.; Iliodromiti, Z.; Sifakis, S.; Siristatidis, C.; Mastorakos, G.; Creatsas, G. Review: Impact of mediators present in amniotic fluid on preterm labour. In Vivo 2012, 26, 799–812. [Google Scholar] [PubMed]
- Malamitsi-Puchner, A.; Vrachnis, N.; Samoli, E.; Baka, S.; Iliodromiti, Z.; Puchner, K.-P.; Malligianis, P.; Hassiakos, D. Possible Early Prediction of Preterm Birth by Determination of Novel Proinflammatory Factors in Midtrimester Amniotic Fluid. Ann. N. Y. Acad. Sci. 2006, 1092, 440–449. [Google Scholar] [CrossRef]
- Völkel, W.; Colnot, T.; Csanády, G.A.; Filser, J.G.; Dekant, W. Metabolism and Kinetics of Bisphenol A in Humans at Low Doses Following Oral Administration. Chem. Res. Toxicol. 2002, 15, 1281–1287. [Google Scholar] [CrossRef] [PubMed]
- Gassman, N.R. Induction of oxidative stress by bisphenol A and its pleiotropic effects. Environ. Mol. Mutagen. 2017, 58, 60–71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferguson, K.K.; Cantonwine, D.E.; McElrath, T.F.; Mukherjee, B.; Meeker, J.D. Repeated measures analysis of associations between urinary bisphenol-A concentrations and biomarkers of inflammation and oxidative stress in pregnancy. Reprod. Toxicol. 2016, 66, 93–98. [Google Scholar] [CrossRef] [Green Version]
- Conde-Agudelo, A.; Papageorghiou, A.T.; Kennedy, S.H.; Villar, J. Novel biomarkers for predicting intrauterine growth restriction: A systematic review and meta-analysis. BJOG 2013, 120, 681–694. [Google Scholar] [CrossRef] [Green Version]
- Vrachnis, N.; Vitoratos, N.; Iliodromiti, Z.; Sifakis, S.; Deligeoroglou, E.; Creatsas, G. Intrauterine inflammation and preterm delivery. Ann. N. Y. Acad. Sci. 2010, 1205, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Iliodromiti, Z.; Zygouris, D.; Sifakis, S.; Pappa, K.I.; Tsikouras, P.; Salakos, N.; Daniilidis, A.; Siristatidis, C.; Vrachnis, N. Acute lung injury in preterm fetuses and neonates: Mechanisms and molecular pathways. J. Matern. Neonatal Med. 2013, 26, 1696–1704. [Google Scholar] [CrossRef]
- Bamigboye, A.A.; Morris, J. Oestrogen supplementation, mainly diethylstilbestrol, for preventing miscarriages and other adverse pregnancy outcomes. Cochrane Database Syst. Rev. 2003, CD004353. [Google Scholar] [CrossRef]
- Ferguson, K.K.; McElrath, T.F.; Cantonwine, D.E.; Mukherjee, B.; Meeker, J.D. Phthalate metabolites and bisphenol-A in association with circulating angiogenic biomarkers across pregnancy. Placenta 2015, 36, 699–703. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vrachnis, N.; Kalampokas, E.; Sifakis, S.; Vitoratos, N.; Kalampokas, T.; Botsis, D.; Iliodromiti, Z. Placental growth factor (PlGF): A key to optimizing fetal growth. J. Matern. Neonatal Med. 2013, 26, 995–1002. [Google Scholar] [CrossRef] [PubMed]
- Müller, J.E.; Meyer, N.; Santamaria, C.G.; Schumacher, A.; Luque, E.H.; Zenclussen, M.L.; Rodriguez, H.A.; Zenclussen, A.C. Bisphenol A exposure during early pregnancy impairs uterine spiral artery remodeling and provokes intrauterine growth restriction in mice. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]

| Study | Selection | Comparability | Outcome/Exposure | Total Score |
|---|---|---|---|---|
| Krętowska et al., 2019 [34] | *** | ** | *** | 8 |
| Pinney et al., 2017 [35] | *** | ** | *** | 8 |
| Burstyn et al., 2013 [36] | **** | ** | *** | 9 |
| Phillipat et al., 2014 [37] | *** | ** | *** | 8 |
| Phillipat et al., 2019 [38] | *** | ** | *** | 8 |
| Lee et al., 2014 [39] | *** | ** | *** | 8 |
| Lee et al., 2018 [40] | *** | ** | *** | 8 |
| Dalkan et al., 2019 [41] | *** | * | *** | 7 |
| Chou et al., 2011 [42] | *** | ** | *** | 8 |
| Gounden et al., 2019 [43] | *** | * | *** | 7 |
| Xu et al., 2015 [44] | ** | * | *** | 6 |
| Wang et al., 2017 [45] | *** | ** | *** | 8 |
| Troisi et al., 2014 [46] | *** | ** | *** | 8 |
| Huo et al., 2015 [47] | **** | ** | *** | 9 |
| Ding et al., 2017 [48] | *** | ** | *** | 8 |
| Tang et al., 2013 [49] | *** | ** | *** | 8 |
| Padmanabhan et al., 2008 [50] | *** | ** | *** | 8 |
| Veiga-Lopez et al., 2015 [51] | *** | * | ** | 7 |
| Huang et al., 2017 [52] | *** | ** | *** | 8 |
| Mustieles et al., 2018 [53] | ** | ** | *** | 7 |
| Aker et al., 2019 [54] | *** | ** | *** | 8 |
| Woods et al., 2017 [55] | *** | ** | *** | 8 |
| Ferguson et al., 2016 [56] | *** | ** | *** | 8 |
| Casas et al., 2016 [57] | *** | ** | *** | 8 |
| Snijder et al., 2013 [58] | *** | ** | *** | 8 |
| Author | Year | Reason for Exclusion |
|---|---|---|
| Bell et al., 2018 [60] | 2018 | BPA not measured during pregnancy or at delivery |
| Smarr et al., 2015 [61] | 2015 | BPA not measured during pregnancy or at delivery |
| Miao et al., 2011 [62] | 2011 | BPA not measured in a biological sample |
| Study | Participants—Country—Type of Study | Biological Sample—Time—Method of Detection—Detection Limit/Limit of Quantification | Median Concentration-50th Percentile/Mean Concentration/ | BW/EFW | BL/WFL | SGA/LBW | AC/FL/HC/BPD | PI | PW/PBWR |
|---|---|---|---|---|---|---|---|---|---|
| Krętowska et al., 2019 [34] | 52—Poland cohort study | Maternal blood, amniotic fluid—between 15 and 18 weeks—GC-MS | 8.69 ng/mL (plasma), 1.03 ng/mL (amniotic fluid)/- | ↓ BPA permeability factor—BW (R = −0.54, p < 0.001) | |||||
| Pinney et al., 2017 [35] | 130—USA cohort study | Amniotic fluid—between 16 and 22 weeks (mean 17.2)—LC-ECAPCI-MS/MS—0.08 ng/mL/0.25 ng/mL | 0.36 ng/mL/- | ↓ 241.8 g (group with 0.41–2.0 ng/mL BPA compared to group with ≤0.25 ng/mL | |||||
| Burstyn et al., 2013 [36] | 1100 –Canada case–control study | Maternal serum—15–16 weeks—APCI-MS/MS—0.1 ng/mL | -/0.5 ng/mL (mean difference: 0 ng/mL) | - | |||||
| Phillipat et al., 2014 [37] | 520—France cohort study | Maternal urine -between 22 and 29 weeks—0.4 ng/mL | 2.4 ng/mL/- | -/- | - | -/-/-/- | |||
| Phillipat et al., 2019 [38] | 473—France cohort study | Maternal urine—between 23 and 29 weeks—online solid phase extraction—HPLC-electrospray ionization-tandem mass spectrometry—0.4 ng/mL | 2.34 ng/mL/- | - | -/- | ||||
| Lee et al., 2014 [39] | 757—Korea cohort study | Maternal urine—28–42 weeks—HPLC-isotope dilution tandem mass spectrometry—0.12–0.28 ng/mL | 1.08 ng/mL (1.63 μg/g creatinine)/1.29 ng/mL (1.87 μg/g creatinine) | ↑ 66.9 g in ♂ (second tertile compared to first) | - | ↑ 0.12 g/cm3 × 100 in ♀ (r = 0.11) | |||
| Lee et al., 2018 [40] | 788—Korea cohort study | Maternal urine—third trimester Neonatal urine—HPLC-isotope dilution tandem mass spectrometry −0.12–0.28 ng/mL | -/1.26 μg/g Cr | ↑ in BPA by 1 log-transformed unit of BPA/Cr: ↑ z-score 0.05 and 0.06 in ♂/- | ↑ in BPA by 1 log-transformed unit of BPA/Cr: -/↑ 0.05 0.07 in ♀ | ↑ in BPA by 1 log-transformed unit of BPA/Cr: ↓ FL 0.03 cm and 0.06 cm (GSTs) | |||
| Dalkan et al., 2019 [41] | 150—Cyprus cohort study | Cord blood—delivery—sandwich enzyme-linked immunosorbent assays (ELISA)— | -/48.3 ± 2.22 ng/mL | -/ | -/ | - (HC) | |||
| Chou et al., 2011 [42] | 97—Taiwan cohort study | Maternal blood, umbilical cord blood—delivery—HPLC/UV detector—0.13 ng/mL | -/2.5 ng/mL (maternal blood) and 0.5 ng/mL (umbilical cord blood) | ↑ (OR = 2.01)/ ↑ (OR = 2.42) | |||||
| Gounden et al., 2019 [43] | 90—South Africa cohort study | Maternal blood, umbilical cord blood—third trimester –ultra HPLC–MS/MS—0.12 ng/mL | Maternal blood: BPA (0.95 ng/mL), BPA-glucuronide (4.71 ng/mL), cord blood: BPA (0.92 ng/mL), BPA-glucuronide (4.21 ng/mL)/- | ↑ (cord blood BPA)/ | |||||
| Xu et al., 2015 [44] | 200—China cohort study | Cord blood, placenta—delivery—GC/MS | 6.369 ng/mL (exposed group) 2.824 ng/mL (reference group)/- | -/ | -/ | ||||
| Wang et al., 2017 [45] | 620—China cohort study | Maternal urine—delivery -HPLC-MS/MS—0.1 ng/mL | -/1.32 ng/mL | -/ | - | ||||
| Troisi et al., 2014 [46] | 200—USA case–control study | Placenta—delivery—isotope dilution GC-MS | -/103.4 ± 61.8 ng/g | -/↓ CBWC for ↑ BPA (p = 0.0112, r = −0.179) | ↑ BPA (157.9 ng/g)/↑ BPA (125.4 ng/g) in cases | ||||
| Huo et al., 2015 [47] | 452—China case–control study | Maternal urine—delivery—UPLC–MS/MS—0.2 ng/mL | 4.70 ng/mL (cases) 2.25 ng/mL (controls) p < 0.05/- | -/↑ Risk, OR = 3.13 for the medium tertile, OR = 2.49 for the highest tertile | |||||
| Ding et al., 2017 [48] | 496—China cohort study | Maternal urine—delivery—HPLC-MS/MS—0.1 ng/mL | 0.48 ng/mL, 1.07 μg/g creatinine/- | - | 10-fold ↑ in BPA ↑ 0.63 cm in ♂/- | - (HC) | - | ||
| Tang et al., 2013 [49] | 567—China cohort study | Maternal urine—delivery—UPLC–MS/MS—0.36 ng/mL | -/0.91 ng/mL | -/ | -/ | ||||
| Padmanabhan et al., 2008 [50] | 40—USA cohort study | Maternal blood—delivery—HPLC-MS/MS—0.5 ng/mL | -/5.9 ng/mL | -/ | |||||
| Veiga-Lopez et al., 2015 [51] | 80—USA cohort study | Maternal blood— 8—14 weeks and delivery Umbilical cord blood—delivery—HPLC-MS/MS—Phase 1: 0.05 ng/mL, Phase 2: 0.02 ng/mL | -/Phase 1: Maternal blood (8–14 weeks: 1.0 ng/mL, delivery 1.7 ng/mL, umbilical cord blood 0.5 ng/mL Phase 2: Maternal blood (8–14 weeks: 4.8 ng/mL, delivery 11.9 ng/mL, umbilical cord blood 3.1 ng/mL | ↓ (−55 g and −183 g in ♀ for 2-fold increase in BPA (8–14 weeks)/ | |||||
| Huang et al., 2017 [52] | 162—Taiwan cohort study | Maternal urine—11, 26 weeks and delivery- time-of-flight mass spectrometer with an electrospray interface and UPLC—0.16 ng/mL | /11 weeks: 0.17 μg/g creatinine, 26 weeks: 0.37 μg/g creatinine, delivery: 0.34 μg/g creatinine | -/ | -/ | ↓ (HC) −0.52 cm (3rd trimester) | |||
| Mustieles et al., 2018 [53] | 346—USA cohort study | Maternal and paternal urine (preconception, 6, 21 and 35 weeks)—HPLC-MS/MS—0.4 ng/mL | /1.6 ng/mL (paternal), 1.5 ng/mL (maternal preconception) and 1.2 ng/mL (maternal prenatal) | each ln unit ↑ in BPA: ↓ 119 g (maternal preconception)/ | ↑ in BPA: ↓ HC 0.72 cm (maternal preconception) | ||||
| Aker et al., 2019 [54] | 922—Puerto Rico cohort study | Maternal urine—16–20, 20–24 and 24–28 weeks—HPLC-MS/MS—0.2 ng/mL | /2.02 ng/mL (all), 2.16 ng/mL (16–20 weeks), 2.07 ng/mL (20–24 weeks), 1.78 ng/mL (24–28 weeks) | -/ | |||||
| Woods et al., 2017 [55] | 272—USA cohort study | Maternal urine, maternal blood—16 and 26 weeks/LC -MS or GC-MS—0.4 μg/g | /2.1 μg/g | -/ | |||||
| Ferguson et al., 2016 [56] | 482—USA cohort study | Maternal urine—at median 10, 18, 26, and 35 weeks, LC-MS/MS | 1.28 ng/mL (10 weeks), 1.33 ng/mL (18 and 26 weeks), 1.32 ng/mL (35 weeks)/ | -/- | -/-/-/ | ||||
| Casas et al., 2016 [57] | 470—Spain cohort study | Maternal urine—12 and 32 weeks—LC-MS-0.1 ng/mL, 0.1 μg/g (creatinine-adjusted) | -/2.3 ng/mL, 2.6 μg/g | -/↓ (–5.74% SD) in ♂ (12 to 20 weeks), ↑ in ♀ (12 weeks)—not creatinine-adjusted | ↑ (6.41% SD) in ♀ (12 weeks)/↓ in ♂ (12 to 20 weeks)/-/- | ||||
| Snijder et al., 2013 [58] | 219—Netherlands cohort study | Maternal urine—13, 21 and 30 weeks—tandem mass spectrometry—0.26 ng/mL, 0.05 ng/mL | -/1.7 µg/g, 3.2 µg/g | -/↓ (–683 g) | -/-/↓ (–3.9 cm)/- |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vrachnis, N.; Loukas, N.; Vrachnis, D.; Antonakopoulos, N.; Zygouris, D.; Kοlialexi, A.; Pergaliotis, V.; Iavazzo, C.; Mastorakos, G.; Iliodromiti, Z. A Systematic Review of Bisphenol A from Dietary and Non-Dietary Sources during Pregnancy and Its Possible Connection with Fetal Growth Restriction: Investigating Its Potential Effects and the Window of Fetal Vulnerability. Nutrients 2021, 13, 2426. https://doi.org/10.3390/nu13072426
Vrachnis N, Loukas N, Vrachnis D, Antonakopoulos N, Zygouris D, Kοlialexi A, Pergaliotis V, Iavazzo C, Mastorakos G, Iliodromiti Z. A Systematic Review of Bisphenol A from Dietary and Non-Dietary Sources during Pregnancy and Its Possible Connection with Fetal Growth Restriction: Investigating Its Potential Effects and the Window of Fetal Vulnerability. Nutrients. 2021; 13(7):2426. https://doi.org/10.3390/nu13072426
Chicago/Turabian StyleVrachnis, Nikolaos, Nikolaos Loukas, Dionysios Vrachnis, Nikolaos Antonakopoulos, Dimitrios Zygouris, Aggeliki Kοlialexi, Vasilios Pergaliotis, Christos Iavazzo, George Mastorakos, and Zoi Iliodromiti. 2021. "A Systematic Review of Bisphenol A from Dietary and Non-Dietary Sources during Pregnancy and Its Possible Connection with Fetal Growth Restriction: Investigating Its Potential Effects and the Window of Fetal Vulnerability" Nutrients 13, no. 7: 2426. https://doi.org/10.3390/nu13072426
APA StyleVrachnis, N., Loukas, N., Vrachnis, D., Antonakopoulos, N., Zygouris, D., Kοlialexi, A., Pergaliotis, V., Iavazzo, C., Mastorakos, G., & Iliodromiti, Z. (2021). A Systematic Review of Bisphenol A from Dietary and Non-Dietary Sources during Pregnancy and Its Possible Connection with Fetal Growth Restriction: Investigating Its Potential Effects and the Window of Fetal Vulnerability. Nutrients, 13(7), 2426. https://doi.org/10.3390/nu13072426









