Effect of Dietary or Supplemental Vitamin C Intake on Vitamin C Levels in Patients with and without Cardiovascular Disease: A Systematic Review
Abstract
:1. Introduction
2. Methods
2.1. Literature Search
2.2. Study Selection
2.3. Data Extraction and Quality Assessment
3. Results
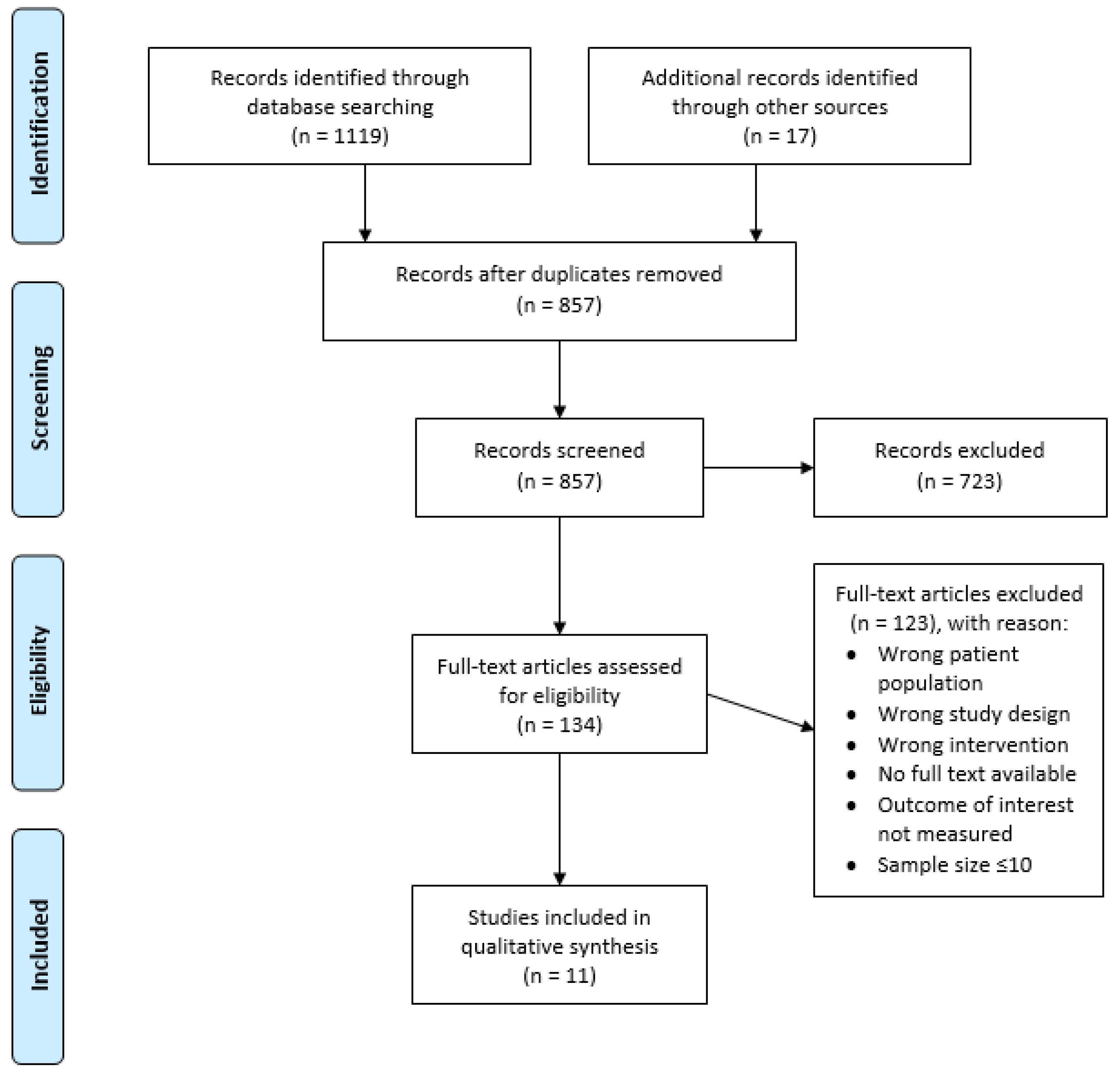
3.1. Search Results
3.2. Study Characteristics
3.2.1. Study Design and Participants
3.2.2. Vitamin C Supplementation and Measurement
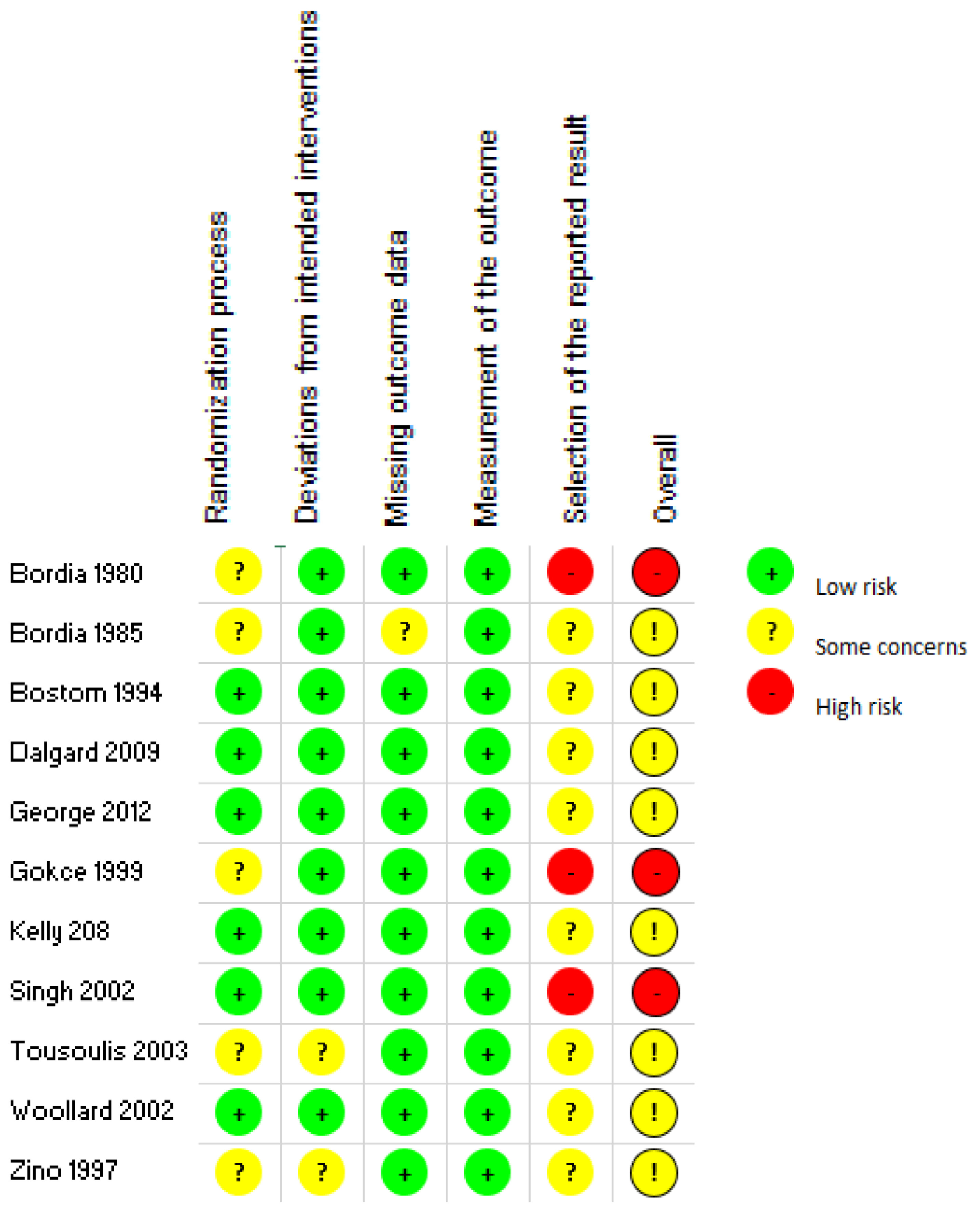
3.3. Risk of Bias and Study Quality
3.4. Change in Vitamin C Levels in Response to Increased Intake
4. Discussion
4.1. Effect of Vitamin C Supplementation on Post-Intervention Vitamin C Levels
4.2. Effect of Vitamin C Dosages on Post-Intervention Vitamin C Levels
4.3. Adequate or Optimal Intake Vitamin C Intake
4.4. Quality Assessment
4.5. Strengths and Limitations
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Australian Bureau of Statistics. National Health Survey First Results Canberra: Australian Bureau of Statistics. 2015. Available online: https://www.abs.gov.au/statistics/health/health-conditions-and-risks/national-health-survey-first-results/latest-release#data-download (accessed on 2 September 2020).
- Australian Bureau of Statistics. Australia’s Leading Causes of Death, 2018: Australian Bureau of Statistics. 2019. Available online: https://www.abs.gov.au/AUSSTATS/abs@.nsf/0/47E19CA15036B04BCA2577570014668B?Opendocument (accessed on 2 September 2020).
- Xu, H.; Jiang, J.; Chen, W.; Li, W.; Chen, Z. Vascular Macrophages in Atherosclerosis. J. Immunol. Res. 2019, 2019, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Delaney, C.; Smale, M.; Miller, M.D. Nutritional Considerations for Peripheral Arterial Disease: A Narrative Review. Nutrients 2019, 11, 1219. [Google Scholar] [CrossRef] [Green Version]
- Aguirre, R.; May, J.M. Inflammation in the vascular bed: Importance of vitamin C. Pharmacol. Ther. 2008, 119, 96–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamer, M.; Steptoe, A. Influence of specific nutrients on progression of atherosclerosis, vascular function, haemostasis and inflammation in coronary heart disease patients: A systematic review. Br. J. Nutr. 2006, 95, 849–859. [Google Scholar] [CrossRef] [Green Version]
- Torres, N.; Guevara-Cruz, M.; Velazquez-Villegas, L.; Tovar, A.R. Nutrition and Atherosclerosis. Arch. Med. Res. 2015, 46, 408–426. [Google Scholar] [CrossRef]
- Ismaeel, A.; Brumberg, R.S.; Kirk, J.S.; Papoutsi, E.; Farmer, P.J.; Bohannon, W.T.; Smith, R.S.; Eidson, J.L.; Sawicki, I.; Koutakis, P. Oxidative Stress and Arterial Dysfunction in Peripheral Artery Disease. Antioxidants 2018, 7, 145. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gimbrone, A.M.; García-Cardeña, A.G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collins, C.; Burrows, T.; Rollo, M. Dietary Patterns and Cardiovascular Disease Outcomes: An Evidence Check Rapid Review; National Heart Foundation of Australia: Sydney, Australia, 2017. [Google Scholar]
- National Heart Foundation of Australia. Dietary Position Statement-Heart Healthy Eating Patterns; National Heart Foundation of Australia: Sydney, Australia, 2019. [Google Scholar]
- Estruch, R. Anti-inflammatory effects of the Mediterranean diet: The experience of the PREDIMED study. Proc. Nutr. Soc. 2010, 69, 333–340. [Google Scholar] [CrossRef] [Green Version]
- Lykkesfeldt, J.; Tveden-Nyborg, P. The Pharmacokinetics of Vitamin C. Nutrients 2019, 11, 2412. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jansen, E.; Ruskovska, T. Serum Biomarkers of (Anti)Oxidant Status for Epidemiological Studies. Int. J. Mol. Sci. 2015, 16, 27378–27390. [Google Scholar] [CrossRef]
- Dehghan, M.; Akhtar-Danesh, N.; McMillan, C.R.; Thabane, L. Is plasma vitamin C an appropriate biomarker of vitamin C intake? A systematic review and meta-analysis. Nutr. J. 2007, 6, 41. [Google Scholar] [CrossRef] [Green Version]
- Anderson, R. SI Units for Clinical Measurement; American College of Physicians: Philadelphia, PA, USA, 1998; p. 511. [Google Scholar]
- Kratz, A.; Ferraro, M.; Sluss, P.M.; Lewandrowski, K.B. Laboratory reference values. N. Engl. J. Med. 2004, 351, 1548–1563. [Google Scholar] [CrossRef]
- Jayedi, A.; Rashidy-Pour, A.; Parohan, M.; Zargar, M.S.; Shab-Bidar, S. Dietary and circulating vitamin C, vitamin E, β-carotene and risk of total cardiovascular mortality: A systematic review and dose–response meta-analysis of prospective observational studies. Public Health Nutr. 2019, 22, 1872–1887. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: A systematic review and dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2018, 108, 1069–1091. [Google Scholar] [CrossRef]
- Ashor, A.W.; Brown, R.; Keenan, P.D.; Willis, N.D.; Siervo, M.; Mathers, J.C. Limited evidence for a beneficial effect of vitamin C supplementation on biomarkers of cardiovascular diseases: An umbrella review of systematic reviews and meta-analyses. Nutr. Res. 2019, 61, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Thomas, J.; Delaney, C.; Suen, J.; Miller, M. Nutritional status of patients admitted to a metropolitan tertiary care vascular surgery unit. Asia Pac. J. Clin. Nutr. 2019, 28, 64–71. [Google Scholar] [PubMed]
- Langlois, L.M.; Duprez, L.D.; Delanghe, L.J.; De Buyzere, L.M.; Clement, L.D. Serum Vitamin C Concentration Is Low in Peripheral Arterial Disease and Is Associated With Inflammation and Severity of Atherosclerosis. Circ. J. Am. Heart Assoc. 2001, 103, 1863–1868. [Google Scholar] [CrossRef] [Green Version]
- Bordia, A. The effect of vitamin C on blood lipids, fibrinolytic activity and platelet adhesiveness in patients with coronary artery disease. Atherosclerosis 1980, 35, 181–187. [Google Scholar] [CrossRef]
- Bordia, A.; Verma, S.K. Effect of vitamin C on platelet adhesiveness and platelet aggregation in coronary artery disease patients. Clin. Cardiol. 1985, 8, 552–554. [Google Scholar] [CrossRef] [PubMed]
- Bostom, A.G.; Yanek, L.; Hume, A.L.; Eaton, C.; McQuade, W.; Nadeau, M.; Perrone, G.; Jacques, P.F.; Selhub, J. High dose ascorbate supplementation fails to affect plasma homocyst(e)ine levels in patients with coronary heart disease. Atherosclerosis 1994, 111, 267–270. [Google Scholar] [CrossRef]
- Dalgård, C.; Nielsen, F.; Morrow, J.D.; Enghusen-Poulsen, H.; Jonung, T.; Hørder, M.; De Maat, M.P.M. Supplementation with orange and blackcurrant juice, but not vitamin E, improves inflammatory markers in patients with peripheral arterial disease. Br. J. Nutr. 2008, 101, 263–269. [Google Scholar] [CrossRef] [Green Version]
- Gokce, N.; Keaney, J.J.F.; Frei, B.; Holbrook, M.; Olesiak, M.; Zachariah, B.J.; Leeuwenburgh, C.; Heinecke, J.W.; Vita, J.A. Long-Term Ascorbic Acid Administration Reverses Endothelial Vasomotor Dysfunction in Patients With Coronary Artery Disease. Circulation 1999, 99, 3234–3240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tousoulis, D.; Antoniades, C.; Tountas, C.; Bosinakou, E.; Kotsopoulou, M.; Toutouzas, P.; Stefanadis, C. Vitamin C Affects Thrombosis/Fibrinolysis System and Reactive Hyperemia in Patients With Type 2 Diabetes and Coronary Artery Disease. Diabetes Care 2003, 26, 2749–2753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- George, T.W.; Paterson, E.; Waroonphan, S.; Gordon, M.H.; Lovegrove, J.A. Effects of chronic consumption of fruit and vegetable puree-based drinks on vasodilation, plasma oxidative stability and antioxidant status. J. Hum. Nutr. Diet. 2012, 25, 477–487. [Google Scholar] [CrossRef]
- Kelly, R.P.; Yeo, K.P.; Isaac, H.B.; Lee, C.-Y.J.; Huang, S.H.; Teng, L.; Halliwell, B.; Wise, S.D.; Lee, J.C.-Y. Lack of effect of acute oral ingestion of vitamin C on oxidative stress, arterial stiffness or blood pressure in healthy subjects. Free. Radic. Res. 2008, 42, 514–522. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Graves, J.; Taylor, P.D.; MacAllister, R.J.; Singer, D.R.J. Effects of a ’healthy’ diet and of acute and long-term vitamin C on vascular function in healthy older subjects. Cardiovasc. Res. 2002, 56, 118–125. [Google Scholar] [CrossRef] [Green Version]
- Woollard, K.; Loryman, C.J.; Meredith, E.; Bevan, R.; Shaw, J.; Lunec, J.; Griffiths, H.R. Effects of oral vitamin C on monocyte: Endothelial cell adhesion in healthy subjects. Biochem. Biophys. Res. Commun. 2002, 294, 1161–1168. [Google Scholar] [CrossRef]
- Zino, S.; Skeaff, M.; Williams, S.; Mann, J. Randomised controlled trial of effect of fruit and vegetable consumption on plasma concentrations of lipids and antioxidants. BMJ 1997, 314, 1787. [Google Scholar] [CrossRef] [Green Version]
- German Nutrition Society. New Reference Values for Vitamin C Intake. Ann. Nutr. Metab. 2015, 67, 13–20. [Google Scholar] [CrossRef]
- Michels, A.; Frei, B. Myths, Artifacts, and Fatal Flaws: Identifying Limitations and Opportunities in Vitamin C Research. Nutrients 2013, 5, 5161–5192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chambial, S.; Dwivedi, S.; Shukla, K.K.; John, P.J.; Sharma, P. Vitamin C in Disease Prevention and Cure: An Overview. Indian J. Clin. Biochem. 2013, 28, 314–328. [Google Scholar] [CrossRef] [Green Version]
- National Health and Medical Research Council. Nutrient Reference Values for Australia and New Zealand Including Recommended Dietary Intakes; National Health and Medical Research Council: Canberra, Australia, 2005. [Google Scholar]
- Miller, A.P.; Huff, C.M.; Roubin, G.S. Vascular disease in the older adult. J. Geriatr. Cardiol. 2016, 13, 727–732. [Google Scholar] [PubMed]
- Jepma, P.; Jorstad, H.; Snaterse, M.; ter Riet, G.; Kragten, H.; Lachman, S.; Minneboo, M.; Boekholdt, M.; Peters, R.J.; Reimer, W.S.O. Lifestyle modification in older versus younger patients with coronary artery disease. Heart 2020, 106, 1066–1072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koutakis, P.; Ismaeel, A.; Farmer, P.; Purcell, S.; Smith, R.S.; Eidson, J.L.; Bohannon, W.T. Oxidative stress and antioxidant treatment in patients with peripheral artery disease. Physiol. Rep. 2018, 6, e13650. [Google Scholar] [CrossRef] [PubMed]
| Concept a | Search Terms |
|---|---|
| Concept 1 | (“Vitamin C” OR Ascorb OR Antioxidant).ti,ab |
| Concept 2 | Intake OR Consumption OR “Dietary intake” OR Supplement OR Supplementation OR “Nutritional supplement” |
| Concept 3 | “Plasma vitamin C” OR “Plasma ascorb” OR “Serum vitamin C” OR “Serum ascorb” OR “Vitamin C level” OR “Ascorb level” OR “Vitamin C concentration” OR “Ascorb concentration” |
| Concept 4 | (“Cardiovascular disease” OR CVD OR “Coronary heart disease” OR CHD OR “Ischaemic heart disease” OR “Ischemic heart disease” OR IHD OR “Coronary artery disease” OR CAD OR “Peripheral artery disease” OR PAD OR “Peripheral vascular disease” OR PVD).ti,ab |
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Studies that are randomized controlled trials | Studies including participants at risk of CVD, including otherwise healthy smokers and hypertensive patients |
| Studies with a sample size greater than 10 participants | Studies that administer mixed nutrient supplementation where no group received vitamin C alone |
| Studies including participants aged 18 or above | Studies incorporating dual treatments such as exercise and supplementation |
| Studies including participants who are either healthy or have established CVD, defined as a clinically manifested disease of the blood vessels including PAD or CAD | |
| Studies that orally administer a single nutrient intervention of vitamin C through supplementation or dietary intervention resulting in vitamin C intake ≥45 mg/day | |
| Studies that record percentage change in vitamin C levels or record before and after intervention levels of vitamin C |
| Author and Year | Study Design | Disease Status | Sample Size Mean (%male) | Age Mean ± SD | Intervention Type and Dosage | Duration | Control | Baseline Vitamin C Levels ± SD (μmol/L) | Post-Intervention Vitamin C Levels ± SD (μmol/L) (p-Value) d | Percentage Change in Vitamin C Levels (%) c |
|---|---|---|---|---|---|---|---|---|---|---|
| Bordia 1980 [23] | P1: DB, RCT P2: RCT | P1: CAD P2: Acute MI | P1: 40 (100) P2: 40 (NR) | Intervention P1: G2: 47.2 G3: 50.8 Control P1: 52.7 P2: NR | Supplement P1: G2—500 mg, BD G3—1000 mg, BD P2: 1000 BD | P1: 6 months P2: 20 days | P1 and 2: Placebo vitamin C capsules | P1: NR Intervention P2: 28.39a Control P2: NR | Intervention and control for P1 and P2: NR | Intervention P1: G2: 22 (p < 0.05) d G3: 96 (p < 0.001) d Intervention P2: 94 (p < 0.001) d Control P1 and P2: NR |
| Bordia and Verma 1985 [24] (Part 2) | RCT | CAD | 20 (NR) | NR | Supplement 1000 mg, every 8 h | 10 days | Placebo vitamin C | Intervention: 28.98 ± 2.84b Control: 22.73 ± 5.68b | Intervention: 45.46 ± 1.7b (p < 0.001) Control: 24.43 ± 1.7b (NR) | Intervention: 57 Control: 7 |
| Bostom et al., 1994 [25] | DB, RCT | CAD | 44 (NR) | NR | Supplement 4500 mg | 12 weeks | Matching placebo | Intervention: 57.39b Control: 57.39b | Intervention: 126.71b (p = 0.0001) Control: 65.91b (NR) | Intervention: 121 Control: 15 |
| Dalgard et al. 2009 [26] | BR, DB, CO | PAD | 48 (46) | Average: NR Intervention:JP: 57.4 ± 6.2 Control: RP: 60.8 ± 5.8 | Dietary orange and blackcurrant juice ~210 mg | 4 weeks | Sugar containing reference beverage (0 mg vit c) | Intervention: 46.4 Control: 31.5 | Intervention: 59.4 (p < 0.005) Control: 28.6 (NR) | Intervention: 28 Control: −9 |
| Gokce et al., 1999 [27] | DB, RCT | CAD | 46 (91) | NR Intervention: 54 ± 9 Control: 56 ± 12 | Supplement 2000 mg single dose + 500 mg daily | 1 month | Matching placebo | Intervention: 41 ± 13 Control: 43 ± 19 | Intervention: Acute: 116 ± 34 (p < 0.05) 30 days: 95 ± 36 (p < 0.05) Control: Acute: 43 ± 20 (NR)30 days: 38 ± 20 (NR) | Intervention: Acute: 183 30 days: 132 Control: Acute: 0 30 days: −12 |
| Tousoulis et al., 2003 [28] | RCT | T2DM and CAD | 39 (87) | Average: NR Intervention: 63.3 ± 2.7 Control: 67.4 ± 2.1 | Supplement 2000 mg | 4 weeks | No antioxidant treatment | Intervention: 39.9 ± 2.0 Control: 39.5 ± 2.1 | Intervention: 79.9 ± 3.6 (p = 0.001) Control: 40.0 ± 2.2 (p = 0.755) | Intervention: 100 Control: 1 |
| Author and Year | Study Design | Health Status | Sample Size Mean (%male) | Age Mean ± SD | Intervention Type and Dosage | Duration | Control | Baseline Vitamin C Levels ± SD (μmol/L) | Post-Intervention Vitamin C Levels ± SD (μmol/L) (p-Value) d | Percentage Change in Vitamin C Levels (%) c |
|---|---|---|---|---|---|---|---|---|---|---|
| George et al., 2012 [29] | SB, CO, RCT | Healthy | 39 (38) | Average: 45 Intervention: NR Control: NR | Dietary-FV puree drinks 84.5 mg | 6 weeks | Diluted fruit-flavored cordial 2 × 50 mL (not identical to intervention) | Intervention: 89 ± 32 Control: 91 ± 37 | Intervention: 97 ± 44 (NS) Control: 81 ± 36 (NR) | Intervention: 9 Control: −11 |
| Kelly et al., 2008 [30] | DB, RCT, CO | Healthy | 26 (75) | Average: 23.5 ± 1.4 Intervention and Control: NR | Supplement 2000 mg | Single dose | Water flavored with lemon juice. | Intervention: 38 ± 18 Control: 40 | Intervention: 120 ± 26 (NR) Control: 40 (NR) | Intervention: 216 Control: 0 |
| Singh et al., 2002 [31] | RCT, DB and SB (diet) | Healthy | 56 (46) | Average: 67± 1 Intervention: Diet: 66 Sup: 66 Control: 69 | Dietary (Diet) extra 3 FV portions/day. Extra ~210 mg Supplement 1000 mg | 6 weeks | Diet: no control. Sup: matching placebo tablets | Intervention: Diet: 84 ± 5 Sup: 83 ± 4 Control: 83 ± 5 | Intervention: Diet: 135 ± 27 (p < 0.05) Sup: 135 ± 8 (p < 0.05) Control: 88 ± 9 (NR) | Intervention: Diet: 61 Sup: 63 Control: 6 |
| Woollard et al., 2002 [32] | DB, CO, RCT | Healthy | 40 (100) | Average: 30 Intervention and Control: NR | Supplement 250 mg | 6 weeks | Vitamin C placebo | Average: 50.4 ± 3.4 Intervention: LOC: 32.92 ± 1 3.98 HIC: 67.61 ± 6.32 | Intervention: LOC: Reported 2-fold increase = ~65.84 (p < 0.0001) HIC: 95.32 ± 25.92 (p < 0.0005) | Intervention: LOC: 100 HIC: 41 |
| Zino et al., 1997 [33] | RCT | Healthy | 87 (71) | Average: NR Intervention: 28.6 ± 7.9 Control: 31.9 ± 11.4 | Dietary—8 serve FV/day. Extra 172 mg | 8 weeks | No dietary intervention | Intervention: 33.50 ± 21 Control: 25.55 ± 21.58 | Intervention: 57.92 ± 22.14 Control: 25.55 ± 20.44 | Intervention: 73 (NR) Control: 0 (NR) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Collins, B.J.; Mukherjee, M.S.; Miller, M.D.; Delaney, C.L. Effect of Dietary or Supplemental Vitamin C Intake on Vitamin C Levels in Patients with and without Cardiovascular Disease: A Systematic Review. Nutrients 2021, 13, 2330. https://doi.org/10.3390/nu13072330
Collins BJ, Mukherjee MS, Miller MD, Delaney CL. Effect of Dietary or Supplemental Vitamin C Intake on Vitamin C Levels in Patients with and without Cardiovascular Disease: A Systematic Review. Nutrients. 2021; 13(7):2330. https://doi.org/10.3390/nu13072330
Chicago/Turabian StyleCollins, Bianca J., Mitali S. Mukherjee, Michelle D. Miller, and Christopher L. Delaney. 2021. "Effect of Dietary or Supplemental Vitamin C Intake on Vitamin C Levels in Patients with and without Cardiovascular Disease: A Systematic Review" Nutrients 13, no. 7: 2330. https://doi.org/10.3390/nu13072330
APA StyleCollins, B. J., Mukherjee, M. S., Miller, M. D., & Delaney, C. L. (2021). Effect of Dietary or Supplemental Vitamin C Intake on Vitamin C Levels in Patients with and without Cardiovascular Disease: A Systematic Review. Nutrients, 13(7), 2330. https://doi.org/10.3390/nu13072330






