Histamine Intolerance—The More We Know the Less We Know. A Review
Abstract
:1. Introduction
1.1. Histamine Intolerance (HIT)
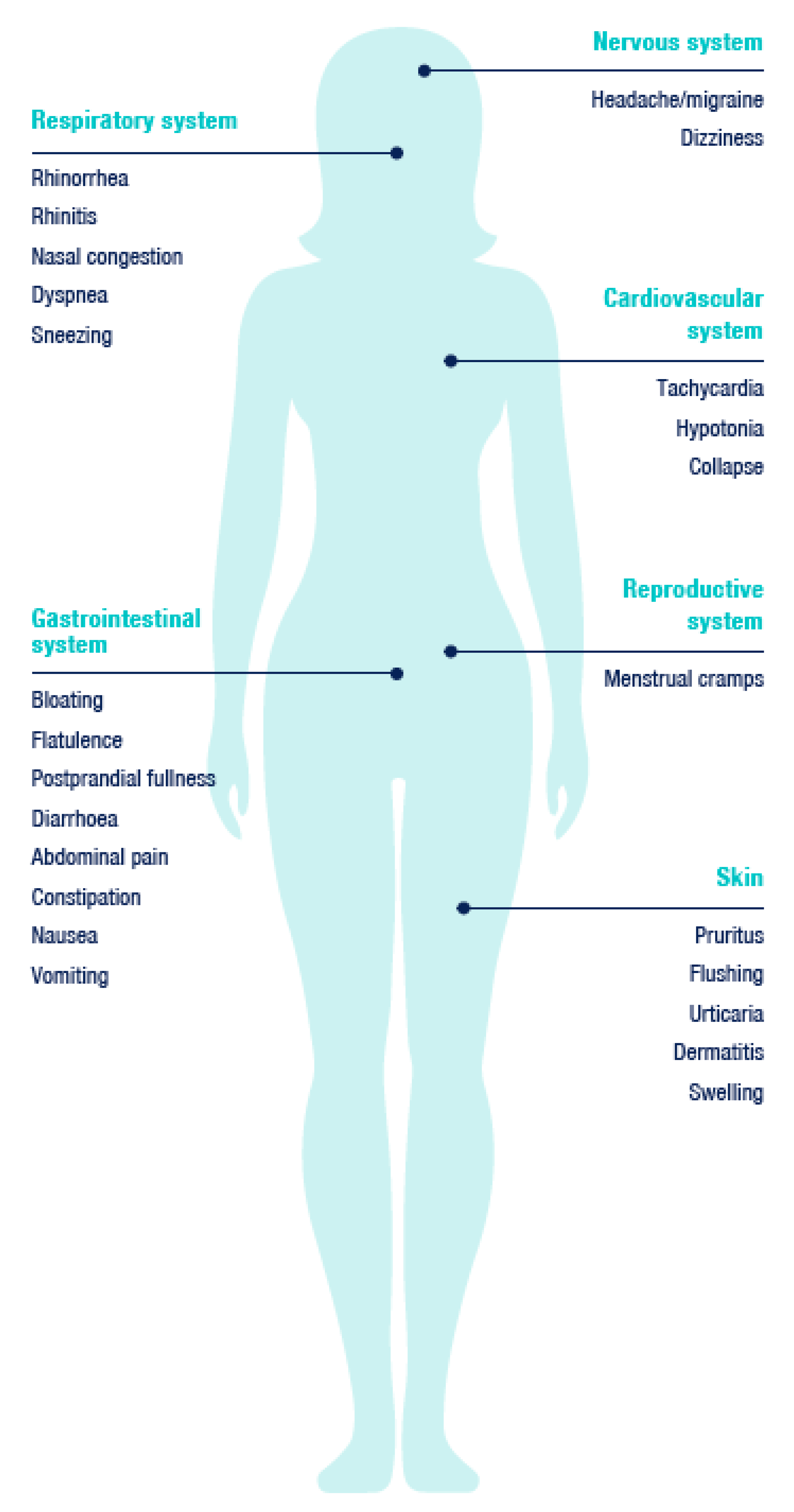
Manifestations of HIT
1.2. Histamine
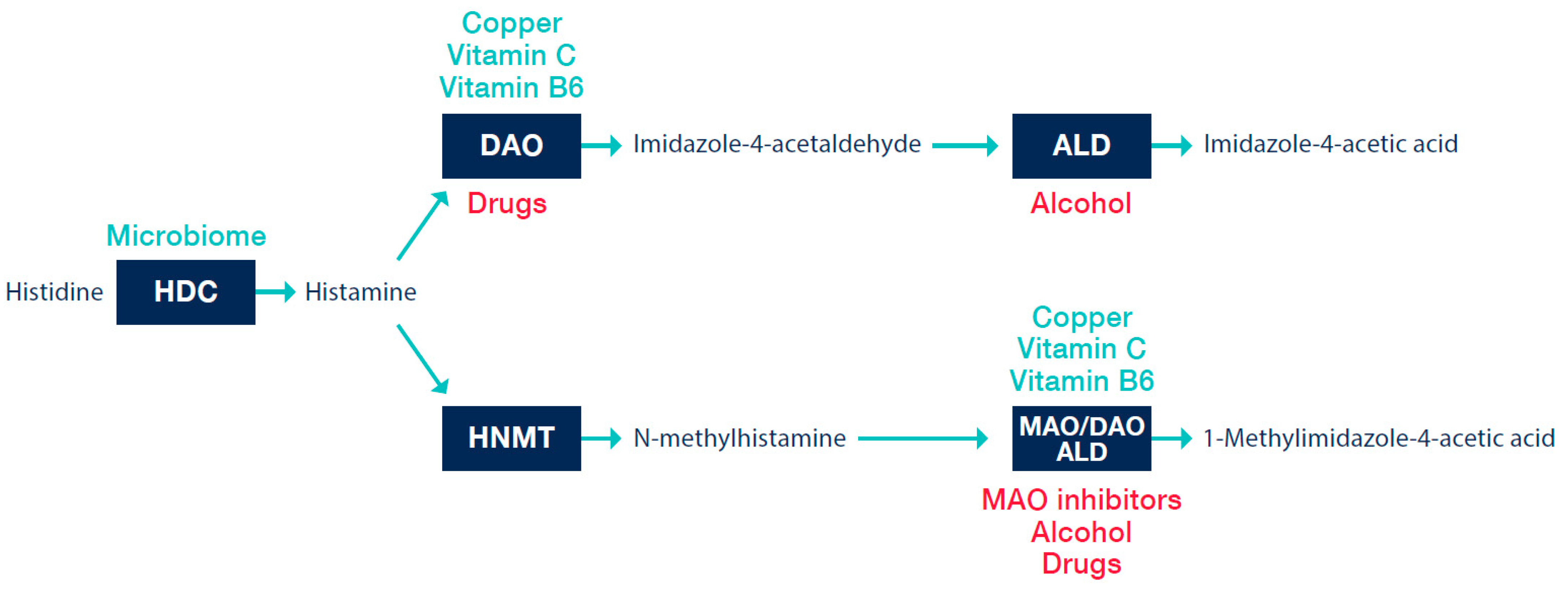
Metabolism of Histamine
1.3. Biogenic Amines in Food
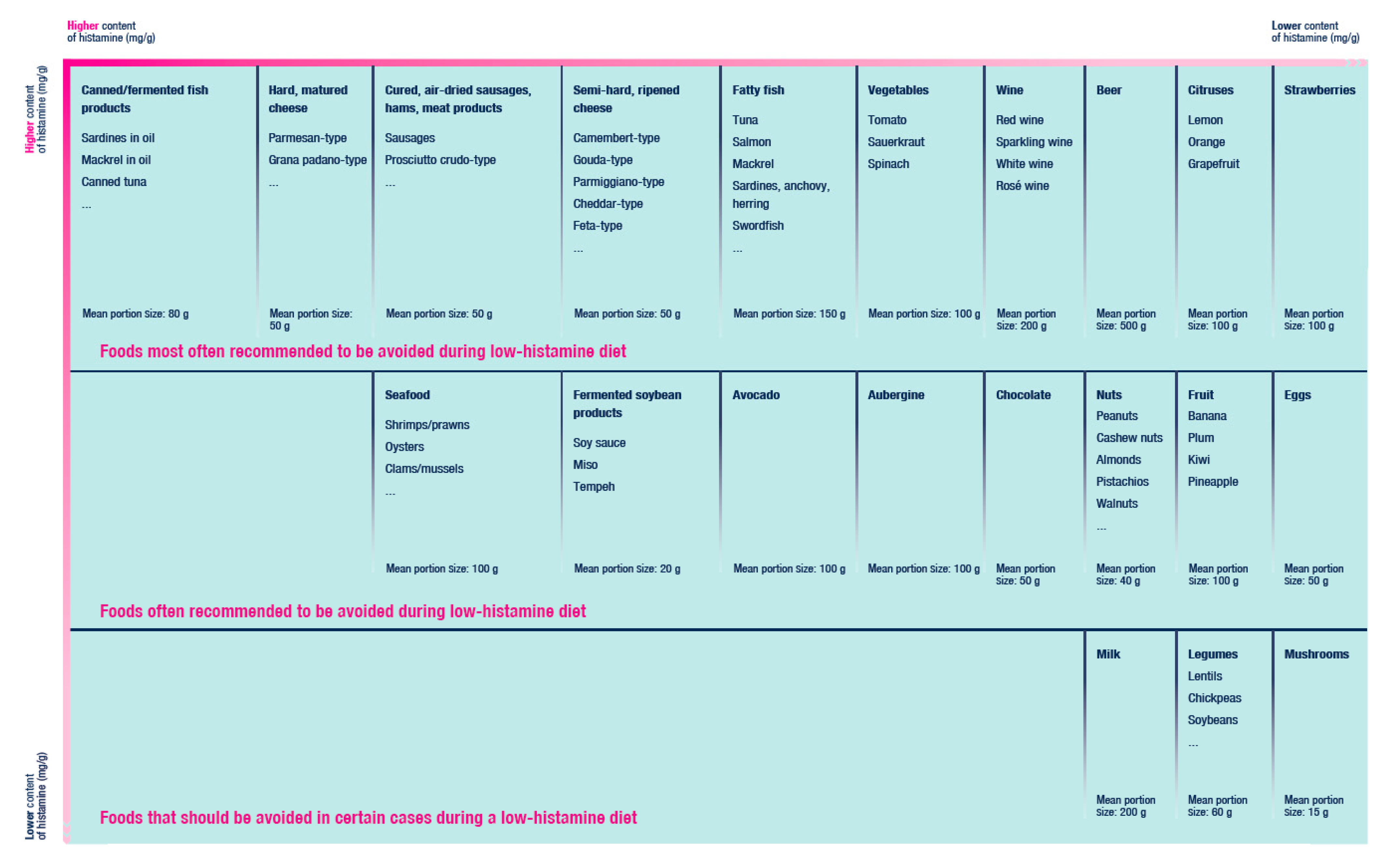
Values of Biogenic Amines and Histamine in Foods
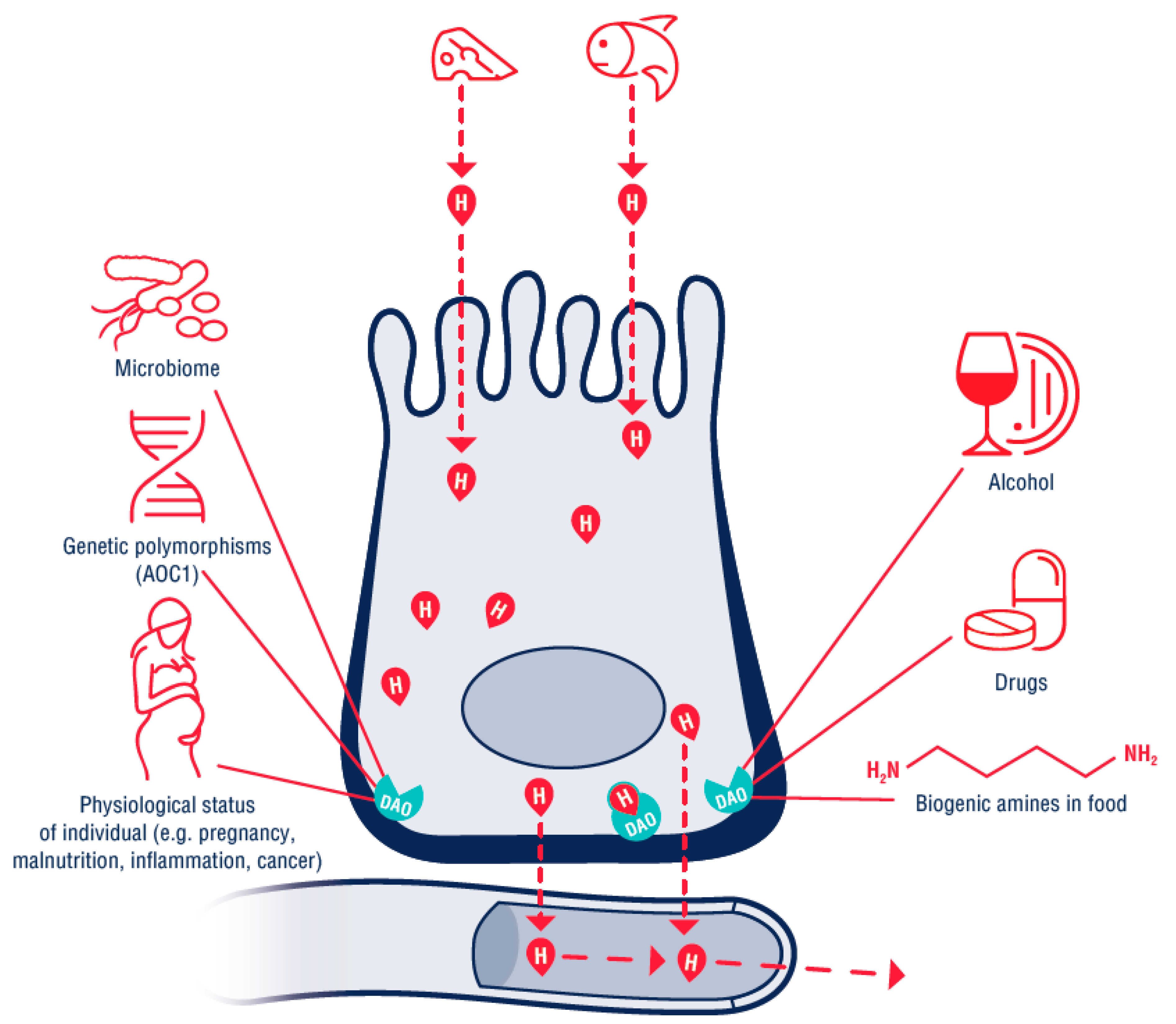
1.4. Factors Contributing to Increased Sensitivity to Histamine
1.5. Microbiome and HIT
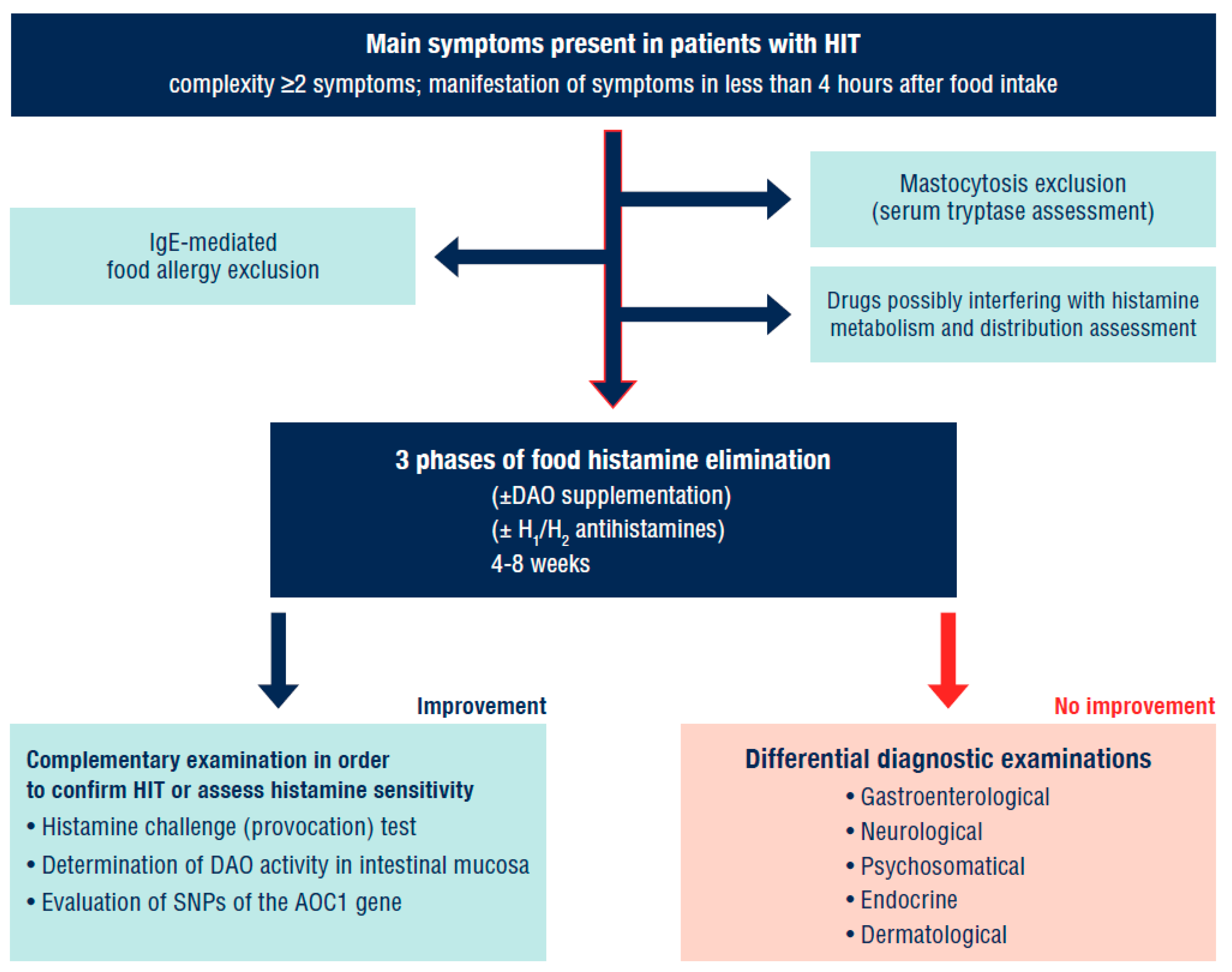
2. Diagnostic Approaches
2.1. DAO Enzyme Activity in Serum
2.2. DAO Activity in the Intestinal Mucosa
2.3. Faecal Histamine Levels
2.4. Skin Prick Test
2.5. Histamine Levels in Plasma
2.6. The Histamine Challenge Test
2.7. Single Nucleotide Polymorphisms (SNPs) of AOC1 Gene Evaluation
2.8. Determination of Histamine and Its Metabolite 1-Methylhistamine from Urine
2.9. Differential Diagnostic Exclusion of Other Diseases
3. Therapeutic Approaches with HIT
3.1. Low-Histamine Diet
3.2. Exogenous Supply of DAO
Other Potential Sources of DAO
3.3. Antihistamines
3.4. Complementary Strategies in HIT Management
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Reese, I.; Ballmer-Weber, B.; Beyer, K.; Fuchs, T.; Kleine-Tebbe, J.; Klimek, L.; Lepp, U.; Niggemann, B.; Saloga, J.; Schäfer, C.; et al. German guideline for the management of adverse reactions to ingested histamine. Allergo J. Int. 2017, 26, 72–79. [Google Scholar] [CrossRef] [Green Version]
- Sicherer, S.H. Epidemiology of food allergy. J. Allergy Clin. Immunol. 2011, 127, 594–602. [Google Scholar] [CrossRef]
- Stapel, S.O.; Asero, R.; Ballmer-Weber, B.K.; Knol, E.F.; Strobel, S.; Vieths, S.; Kleine-Tebbe, J. Testing for IgG4 against foods is not recommended as a diagnostic tool: EAACI task force report. Allergy 2008, 63, 793–796. [Google Scholar] [CrossRef] [Green Version]
- Burks, A.W.; Tang, M.; Sicherer, S.; Muraro, A.; Eigenmann, P.A.; Ebisawa, M.; Fiocchi, A.; Chiang, W.; Beyer, K.; Wood, R.; et al. ICON: Food allergy. J. Allergy Clin. Immunol. 2012, 129, 906–920. [Google Scholar] [CrossRef]
- Matricon, J.; Meleine, M.; Gelot, A.; Piche, T.; Dapoigny, M.; Müller, E.; Ardid, D. Associations between immune activation, intestinal permeability and the irritable bowel syndrome. Aliment. Pharmacol. Ther. 2012, 36, 1009–1031. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuck, C.J.; Biesiekierski, J.R.; Schmid-Grendelmeier, P.; Pohl, D. Food intolerances. Nutrients 2019, 11, 1684. [Google Scholar] [CrossRef] [Green Version]
- Johansson, S.G.O.; Bieber, T.; Dahl, R.; Friedmann, P.S.; Lanier, B.Q.; Lockey, R.F.; Motala, C. Revised nomenclature for allergy for global use: Report of the nomenclature review committee of the World Allergy Organization, October 2003. J. Allergy Clin. Immunol. 2004, 113, 832–836. [Google Scholar] [CrossRef] [PubMed]
- Lomer, M.C.E. The aetiology, diagnosis, mechanisms and clinical evidence for food intolerance. Aliment. Pharmacol. Ther. 2015, 41, 262–275. [Google Scholar] [CrossRef] [PubMed]
- Comas-Basté, O.; Sánchez-Pérez, S.; Veciana-Nogués, M.T.; Latorre-Moratalla, M.; del Carmen Vidal-Carou, M. Histamine intolerance: The current state of the art. Biomolecules 2020, 10, 1181. [Google Scholar] [CrossRef]
- Colombo, F.M.; Cattaneo, P.; Confalonieri, E.; Bernardi, C. Histamine food poisonings: A systematic review and meta-analysis. Crit. Rev. Food Sci. Nutr. 2018, 58, 1131–1151. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Wang, P.; Bian, L.; Hong, S. Rare death via histamine poisoning following crab consumption: A case report. J. Forensic Sci. 2017, 63, 980–982. [Google Scholar] [CrossRef] [PubMed]
- Afify, S.M.; Pali-Schöll, I. Adverse reactions to food: The female dominance—A secondary publication and update. World Allergy Organ. J. 2017, 10, 43. [Google Scholar] [CrossRef] [Green Version]
- Maintz, L.; Novak, N. Histamine and histamine intolerance. Am. J. Clin. Nutr. 2007, 85, 1185–1196. [Google Scholar] [CrossRef]
- Hamada, Y.; Shinohara, Y.; Yano, M.; Yamamoto, M.; Yoshio, M.; Satake, K.; Toda, A.; Hirai, M.; Usami, M. Effect of the menstrual cycle on serum diamine oxidase levels in healthy women. Clin. Biochem. 2013, 46, 99–102. [Google Scholar] [CrossRef]
- Jarisch, R. Histamine intolerance in women. In Histamine Intolerance; Springer: Berlin, Germany, 2014; pp. 109–115. [Google Scholar]
- Komericki, P.; Klein, G.; Reider, N.; Hawranek, T.; Strimitzer, T.; Lang, R.; Kranzelbinder, B.; Aberer, W. Histamine intolerance: Lack of reproducibility of single symptoms by oral provocation with histamine: A randomised, double-blind, placebo-controlled cross-over study. Wien. Klin. Wochenschr. 2010, 123, 15–20. [Google Scholar] [CrossRef]
- Alnouri, G.; Cha, N.; Sataloff, R.T. Histamine sensitivity: An uncommon recognized cause of living laryngopharyngeal reflux symptoms and signs—A case report. Ear Nose Throat J. 2020. [Google Scholar] [CrossRef]
- Ansaranta, M.; Kauppi, P.; Malmberg, L.P.; Vilkman, E.; Geneid, A. Inspiratory and expiratory flow changes, voice symptoms and laryngeal findings during Histamine Challenge tests. Folia Phoniatr. Logop. 2019, 72, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Waldum, H.L.; Sørdal, Ø.F.; Mjønes, P.G. The enterochromaffin-like [ECL] cell—Central in gastric physiology and pathology. Int. J. Mol. Sci. 2019, 20, 2444. [Google Scholar] [CrossRef] [Green Version]
- Yoshikawa, T.; Nakamura, T.; Yanai, K. Histaminergic neurons in the tuberomammillary nucleus as a control centre for wakefulness. Br. J. Pharmacol. 2021, 178, 750–769. [Google Scholar] [CrossRef]
- Kucher, A.N. Association of polymorphic variants of key histamine metabolism genes and histamine receptor genes with multifactorial diseases. Russ. J. Genet. 2019, 55, 794–814. [Google Scholar] [CrossRef]
- Scammell, T.E.; Jackson, A.C.; Franks, N.P.; Wisden, W.; Dauvilliers, Y. Histamine: Neural circuits and new medications. Sleep 2019, 42, 183. [Google Scholar] [CrossRef]
- Sánchez-Pérez, S.; Comas-Basté, O.; Rabell-González, J.; Veciana-Nogués, M.T.; Latorre-Moratalla, M.L.; Vidal-Carou, M.C. Biogenic amines in plant-origin foods: Are they frequently underestimated in low-histamine diets? Foods 2018, 7, 205. [Google Scholar] [CrossRef] [Green Version]
- Schnedl, W.J.; Enko, D. Considering histamine in functional gastrointestinal disorders. Crit. Rev. Food Sci. Nutr. 2020, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Laroche, D.; Vergnaud, M.-C.; Sillard, B.; Soufarapis, H.; Bricard, H. Biochemical markers of anaphylactoid reactions to drugs comparison of plasma histamine and tryptase. J. Am. Soc. Anesthesiol. 1991, 75, 945–949. [Google Scholar] [CrossRef]
- Vakal, S.; Jalkanen, S.; Dahlström, K.M.; Salminen, T.A. Human copper-containing amine oxidases in drug design and development. Molecules 2020, 25, 1293. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boehm, T.; Pils, S.; Gludovacz, E.; Szoelloesi, H.; Petroczi, K.; Majdic, O.; Quaroni, A.; Borth, N.; Valent, P.; Jilma, B. Quantification of human diamine oxidase. Clin. Biochem. 2017, 50, 444–451. [Google Scholar] [CrossRef]
- Schwelberger, H.G.; Feurle, J.; Ahrens, F. Characterization of diamine oxidase from human seminal plasma. J. Neural Transm. 2013, 120, 983–986. [Google Scholar] [CrossRef]
- McGrath, A.P.; Hilmer, K.M.; Collyer, C.A.; Shepard, E.M.; Elmore, B.O.; Brown, D.E.; Dooley, D.M.; Guss, M. Structure and inhibition of human diamine oxidase. Biochemistry 2009, 48, 9810–9822. [Google Scholar] [CrossRef] [Green Version]
- Ji, Y.; Sakata, Y.; Li, X.; Zhang, C.; Yang, Q.; Xu, M.; Wollin, A.; Langhans, W.; Tso, P. Lymphatic diamine oxidase secretion stimulated by fat absorption is linked with histamine release. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G732–G740. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leitner, R.; Zoernpfenning, E.; Missbichler, A. Evaluation of the inhibitory effect of various drugs/active ingredients on the activity of human diamine oxidase in vitro. Clin. Transl. Allergy 2014, 4, P23. [Google Scholar] [CrossRef] [Green Version]
- Kovacova-Hanuskova, E.; Buday, T.; Gavliakova, S.; Plevkova, J. Histamine, histamine intoxication and intolerance. Allergol. Immunopathol. 2015, 43, 498–506. [Google Scholar] [CrossRef]
- Rosell-Camps, A.; Zibetti, S.; Pérez-Esteban, G.; Vila-Vidal, M.; Ferrés-Ramis, L.; García-Teresa-García, E. Histamine intolerance as a cause of chronic digestive complaints in pediatric patients. Rev. Esp. Enferm. Dig. 2013, 105, 201–207. [Google Scholar] [CrossRef]
- Kacik, J.; Wróblewska, B.; Lewicki, S.; Zdanowski, R.; Kalicki, B. Serum diamine oxidase in pseudoallergy in the pediatric population. In Advances in Experimental Medicine and Biology; Springer: Cham, Switzerland, 2017; Volume 1039, pp. 35–44. [Google Scholar]
- Nei, D. Evaluation of non-bacterial factors contributing to histamine accumulation in fish fillets. Food Control. 2014, 35, 142–145. [Google Scholar] [CrossRef]
- Verma, N.; Hooda, V.; Gahlaut, A.; Gothwal, A.; Hooda, V. Enzymatic biosensors for the quantification of biogenic amines: A literature update. Crit. Rev. Biotechnol. 2019, 40, 1–14. [Google Scholar] [CrossRef]
- D’Andrea, G.; D’Arrigo, A.; Carbonare, M.D.; Leon, A. Pathogenesis of migraine: Role of neuromodulators. Headache 2012, 52, 1155–1163. [Google Scholar] [CrossRef] [PubMed]
- Kohn, J.B. Is there a diet for histamine intolerance? J. Acad. Nutr. Diet. 2014, 114, 1860. [Google Scholar] [CrossRef] [PubMed]
- Moniente, M.; García-Gonzalo, D.; Ontañón, I.; Pagán, R.; Botello-Morte, L. Histamine accumulation in dairy products: Microbial causes, techniques for the detection of histamine-producing microbiota, and potential solutions. Compr. Rev. Food Sci. Food Saf. 2021, 20, 1481–1523. [Google Scholar] [CrossRef] [PubMed]
- Martin, I.S.M.; Brachero, S.; Vilar, E.G. Histamine intolerance and dietary management: A complete review. Allergol. Immunopathol. 2016, 44, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Scalzo, R.L.; Fibiani, M.; Francese, G.; D’Alessandro, A.; Rotino, G.L.; Conte, P.; Mennella, G. Cooking influence on physico-chemical fruit characteristics of eggplant (Solanum melongena L.). Food Chem. 2016, 194, 835–842. [Google Scholar] [CrossRef] [Green Version]
- Preti, R.; Rapa, M.; Vinci, G. Effect of steaming and boiling on the antioxidant properties and biogenic amines content in green bean (Phaseolus vulgaris) varieties of different colours. J. Food Qual. 2017, 2017, 5329070. [Google Scholar] [CrossRef] [Green Version]
- Chung, B.Y.; Park, S.Y.; Byun, Y.S.; Son, J.H.; Choi, Y.W.; Cho, Y.S.; Kim, H.O.; Park, C.W. Effect of different cooking methods on histamine levels in selected foods. Ann. Dermatol. 2017, 29, 706–714. [Google Scholar] [CrossRef] [Green Version]
- Schink, M.; Konturek, P.C.; Tietz, E.; Dieterich, W.; Pinzer, T.C.; Wirtz, S.; Neurath, M.F.; Zopf, Y. Microbial patterns in patients with histamine intolerance. J. Physiol. Pharmacol. 2018, 69, 579–593. [Google Scholar]
- O’Callaghan, A.; van Sinderen, D. Bifidobacteria and their role as members of the human gut microbiota. Front. Microbiol. 2016, 7, 925. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mokhtar, S.; Mostafa, G.; Taha, R.; Eldeep, G.S.S. Effect of different starter cultures on the biogenic amines production as a critical control point in fresh fermented sausages. Eur. Food Res. Technol. 2012, 235, 527–535. [Google Scholar] [CrossRef]
- Priyadarshani, D.; Mesthri, W.; Rakshit, S.K. Screening selected strains of probiotic lactic acid bacteria for their ability to produce biogenic amines (histamine and tyramine). Int. J. Food Sci. Technol. 2011, 46, 2062–2069. [Google Scholar] [CrossRef]
- Coton, M.; Romano, A.; Spano, G.; Ziegler, K.; Vetrana, C.; Desmarais, C.; Lonvaud-Funel, A.; Lucas, P.; Coton, E. Occurrence of biogenic amine-forming lactic acid bacteria in wine and cider. Food Microbiol. 2010, 27, 1078–1085. [Google Scholar] [CrossRef]
- Poveda, J.M.; Ruiz, P.; Sesena, S.; Palop, M.L. Occurrence of biogenic amine-forming lactic acid bacteria during a craft brewing process. LWT Food Sci. Technol. 2017, 85, 129–136. [Google Scholar] [CrossRef]
- Ferstl, R.; Frei, R.; Schiavi, E.; Konieczna, P.; Barcik, W.; Ziegler, M.; Lauener, R.P.; Chassard, C.; Lacroix, C.; Akdis, C.A.; et al. Histamine receptor 2 is a key influence in immune responses to intestinal histamine-secreting microbes. J. Allergy Clin. Immunol. 2014, 134, 744–746.e3. [Google Scholar] [CrossRef]
- Spinler, J.K.; Sontakke, A.; Hollister, E.B.; Venable, S.F.; Oh, P.L.; Balderas, M.A.; Saulnier, D.M.; Mistretta, T.-A.; Devaraj, S.; Walter, J.; et al. From prediction to function using evolutionary genomics: Human-specific ecotypes of lactobacillus reuteri have diverse probiotic functions. Genome Biol. Evol. 2014, 6, 1772–1789. [Google Scholar] [CrossRef] [Green Version]
- Wechsler, D.; Irmler, S.; Berthoud, H.; Portmann, R.; Badertscher, R.; Bisig, W.; Schafroth, K.; Fröhlich-Wyder, M.-T. Influence of the inoculum level of Lactobacillus parabuchneri in vat milk and of the cheese-making conditions on histamine formation during ripening. Int. Dairy J. 2021, 113, 104883. [Google Scholar] [CrossRef]
- Frei, R.; Ferstl, R.; Konieczna, P.; Ziegler, M.; Simon, T.; Rugeles, T.M.; Mailand, S.; Watanabe, T.; Lauener, R.; Akdis, C.A.; et al. Histamine receptor 2 modifies dendritic cell responses to microbial ligands. J. Allergy Clin. Immunol. 2013, 132, 194–204. [Google Scholar] [CrossRef]
- Oksaharju, A.; Kankainen, M.; Kekkonen, R.A.; Lindstedt, K.A.; Kovanen, P.T.; Korpela, R.; Miettinen, M. Probiotic lactobacillus rhamnosus downregulates FCER1 and HRH4 expression in human mast cells. World J. Gastroenterol. 2011, 17, 750. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, M.A.; Moreno-Arribas, M.V. The problem of biogenic amines in fermented foods and the use of potential biogenic amine-degrading microorganisms as a solution. Trends Food Sci. Technol. 2014, 39, 146–155. [Google Scholar] [CrossRef] [Green Version]
- Tittarelli, F.; Perpetuini, G.; Di Gianvito, P.; Tofalo, R. Biogenic amines producing and degrading bacteria: A snapshot from raw ewes’ cheese. Lebensm. Wiss. Technol. 2019, 101, 1–9. [Google Scholar] [CrossRef]
- Kung, H.-F.; Lee, Y.-C.; Huang, Y.-L.; Huang, Y.R.; Su, Y.-C.; Tsai, Y.-H. Degradation of histamine by lactobacillus plantarum isolated from miso products. J. Food Prot. 2017, 80, 1682–1688. [Google Scholar] [CrossRef] [PubMed]
- Schnedl, W.J.; Schenk, M.; Lackner, S.; Enko, D.; Mangge, H.; Forster, F. Diamine oxidase supplementation improves symptoms in patients with histamine intolerance. Food Sci. Biotechnol. 2019, 28, 1779–1784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Manzotti, G.; Breda, D.; Di Gioacchino, M.; Burastero, S. Serum diamine oxidase activity in patients with histamine intolerance. Int. J. Immunopathol. Pharmacol. 2016, 29, 105–111. [Google Scholar] [CrossRef] [Green Version]
- Mušič, E.; Korošec, P.; Šilar, M.; Adamič, K.; Košnik, M.; Rijavec, M. Serum diamine oxidase activity as a diagnostic test for histamine intolerance. Wien. Klin. Wochenschr. 2013, 125, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Peralta, T.; Carla, B.; Beltran-Ortiz, C.; Magdalena, D.; Verónica, R.; Antonieta, G.M.; Pablo, F. Histamine intolerance: Clinical characterization and determination of serum diamine oxidase (Dao) in a series of cases and controls. Res. Sq. 2020. [Google Scholar] [CrossRef]
- Schwelberger, H.G.; Feurle, J.; Houen, G. New tools for studying old questions: Antibodies for human diamine oxidase. J. Neural Transm. 2012, 120, 1019–1026. [Google Scholar] [CrossRef]
- Pinzer, T.C.; Tietz, E.; Waldmann, E.; Schink, M.; Neurath, M.F.; Zopf, Y. Circadian profiling reveals higher histamine plasma levels and lower diamine oxidase serum activities in 24% of patients with suspected histamine intolerance compared to food allergy and controls. Allergy 2017, 73, 949–957. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lessof, M.H.; Gant, V.; Hinuma, K.; Murphy, G.M.; Dowling, R.H. Recurrent urticaria and reduced diamine oxidase activity. Clin. Exp. Allergy 1990, 20, 373–376. [Google Scholar] [CrossRef] [PubMed]
- Schwelberger, H.G.; Weidenhiller, M.; Hahn, E.G.; Raithel, M.; Kuefner, M.A. Both catabolic pathways of histamine via histamine-N-methyltransferase and diamine oxidase are diminished in the colonic mucosa of patients with food allergy. Inflamm. Res. 2004, 53, S31–S32. [Google Scholar] [CrossRef] [PubMed]
- Wagner, A.; Buczyłko, K.; Zielińska-Bliźniewska, H.; Wagner, W. Impaired resolution of wheals in the skin prick test and low diamine oxidase blood level in allergic patients. Adv. Dermatol. Allergol. Postȩp. Dermatol. Alergol. 2019, 36, 538–543. [Google Scholar] [CrossRef] [Green Version]
- Giera, B.; Straube, S.; Konturek, P.; Hahn, E.G.; Raithel, M. Plasma histamine levels and symptoms in double blind placebo controlled histamine provocation. Inflamm. Res. 2008, 57, 73–74. [Google Scholar] [CrossRef] [PubMed]
- Wöhrl, S.; Hemmer, W.; Focke, M.; Rappersberger, K.; Jarisch, R. Histamine intolerance-like symptoms in healthy volunteers after oral provocation with liquid histamine. Allergy Asthma Proc. 2004, 25, 305–311. [Google Scholar]
- Comas-Basté, O.; Latorre-Moratalla, M.; Bernacchia, R.; Veciana-Nogués, M.; Vidal-Carou, M. New approach for the diagnosis of histamine intolerance based on the determination of histamine and methylhistamine in urine. J. Pharm. Biomed. Anal. 2017, 145, 379–385. [Google Scholar] [CrossRef]
- Mayo-Yáñez, M.; Díaz-Díaz, A.; Calvo-Henríquez, C.; Chiesa-Estomba, C.; Figueroa, A.; Martín-Martín, C. Usefulness of the histamine intolerance assessment questionnaire for diagnosis. Rev. Française d’Allergol. 2021, 61, 87–91. [Google Scholar] [CrossRef]
- Honzawa, Y.; Nakase, H.; Matsuura, M.; Chiba, T. Clinical significance of serum diamine oxidase activity in inflammatory bowel disease: Importance of evaluation of small intestinal permeability. Inflamm. Bowel Dis. 2011, 17, E23–E25. [Google Scholar] [CrossRef]
- Mennigen, R.; Kusche, J.; Streffer, C.; Krakamp, B. Diamine oxidase activities in the large bowel mucosa of ulcerative colitis patients. Inflamm. Res. 1990, 30, 264–266. [Google Scholar] [CrossRef] [PubMed]
- García-Martín, E.; Mendoza, J.L.; Martínez, C.; Taxonera, C.; Urcelay, E.; Ladero, J.M.; de la Concha, E.G.; Díaz-Rubio, M.; Agúndez, J.A.G. Severity of ulcerative colitis is associated with a polymorphism at diamine oxidase gene but not at histamine N-methyltransferase gene. World J. Gastroenterol. 2006, 12, 615. [Google Scholar] [CrossRef] [PubMed]
- Palacios, N.L.; Agúndez, J.A.G.; Mendoza, J.L.; Garcia-Martin, E.; Martínez, C.; Ferrer, M.E.F.; Ladero, J.M.; Taxonera, C.; Díaz-Rubio, M. Analysis of a non-synonymous single nucleotide polymorphism of the human diamine oxidase gene (ref. SNP ID: rs1049793) in patients with Crohn’s disease. Scand. J. Gastroenterol. 2009, 44, 1207–1212. [Google Scholar] [CrossRef] [PubMed]
- Schnedl, W.J.; Lackner, S.; Enko, D.; Schenk, M.; Mangge, H.; Holasek, S.J. Non-celiac gluten sensitivity: People without celiac disease avoiding gluten—Is it due to histamine intolerance? Inflamm. Res. 2017, 67, 279–284. [Google Scholar] [CrossRef]
- Schnedl, W.J.; Mangge, H.; Schenk, M.; Enko, D. Non-responsive celiac disease may coincide with additional food intolerance/malabsorption, including histamine intolerance. Med. Hypotheses 2021, 146, 110404. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Pérez, S.; Comas-Basté, O.; Veciana-Nogués, M.; Latorre-Moratalla, M.; Vidal-Carou, M. Low-histamine diets: Is the exclusion of foods justified by their histamine content? Nutrients 2021, 13, 1395. [Google Scholar] [CrossRef]
- Wagner, N.; Dirk, D.; Reese, I.; Peveling-Oberhag, A.; Rady-Pizarro, U.; Mitzel, H.; Staubach, P. A popular myth—Low-histamine diet improves chronic spontaneous urticaria—Fact or fiction? J. Eur. Acad. Dermatol. Venereol. 2016, 31, 650–655. [Google Scholar] [CrossRef] [Green Version]
- Lackner, S.; Malcher, V.; Enko, D.; Mangge, H.; Holasek, S.J.; Schnedl, W.J. Histamine-reduced diet and increase of serum diamine oxidase correlating to diet compliance in histamine intolerance. Eur. J. Clin. Nutr. 2018, 73, 102–104. [Google Scholar] [CrossRef]
- European Commission. Commission Implementing Regulation (EU) 2020/973 of 6 July 2020: Authorising a Change of the Conditions of Use of the Novel Food ‘Protein Extract from Pig Kidneys’ and Amending Implementing Regulation (EU) 2017/2470 (Text with EEA Relevance). Available online: https://eur-lex.europa.eu/legal-content/SK/TXT/PDF/?uri=CELEX:32020R0973&from=EN. (accessed on 23 March 2021). (In Slovak).
- Yacoub, M.-R.; Ramirez, G.A.; Berti, A.; Mercurio, G.; Breda, D.; Saporiti, N.; Burastero, S.; Dagna, L.; Colombo, G. Diamine oxidase supplementation in chronic spontaneous urticaria: A randomized, double-blind placebo-controlled study. Int. Arch. Allergy Immunol. 2018, 176, 268–271. [Google Scholar] [CrossRef]
- Izquierdo-Casas, J.; Comas-Basté, O.; Latorre-Moratalla, M.; Lorente-Gascón, M.; Duelo, A.; Soler-Singla, L.; Vidal-Carou, M.C. Diamine oxidase (DAO) supplement reduces headache in episodic migraine patients with DAO deficiency: A randomized double-blind trial. Clin. Nutr. 2019, 38, 152–158. [Google Scholar] [CrossRef] [PubMed]
- Comas-Basté, O.; Latorre-Moratalla, M.L.; Rabell-González, J.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. Lyophilised legume sprouts as a functional ingredient for diamine oxidase enzyme supplementation in histamine intolerance. Lebensm. Wiss. Technol. 2020, 125, 109201. [Google Scholar] [CrossRef]
- Maintz, L.; Benfadal, S.; Allam, J.-P.; Hagemann, T.; Fimmers, R.; Novak, N. Evidence for a reduced histamine degradation capacity in a subgroup of patients with atopic eczema. J. Allergy Clin. Immunol. 2006, 117, 1106–1112. [Google Scholar] [CrossRef] [PubMed]
| Symptoms | Differential Diagnosis |
|---|---|
| Flushing | Neuroendocrine tumors |
| Itching | Urticaria, pruritus sine materia, prurigo |
| Nausea/vomiting/abdominal pain | Peptic ulcer disease, hiatal hernia, gastroesophageal reflux disease |
| Diarrhea and abdominal pain | Chronic inflammatory bowel disorders, disorders of carbohydrate metabolism |
| (lactose intolerance, fructose malabsorption), celiac disease | |
| Rhinitis | Allergic and non-allergic rhinitis of other origin |
| Dyspnea, dysphonia | Allergic and non-allergic asthma |
| Hypotension, vertigo, tachycardia | Anaphylaxis |
| Important differential diagnostic information is achieved by the analysis of symptoms with respect to their onset time. Adverse food reactions only considered if the symptoms manifested in less than 4 h from food intake. | |
| DAO blocking foods | |
| Alcohol | wine and spirits especially |
| OTC drugs interfering with DAO activity (decreasing DAO activity) | |
| Expectorants, mucolytics | ambroxol, N-acetylcysteine |
| Nonsteroidal anti-inflammatory drugs | acetylsalicylic acid, ibuprofen |
| Rx drugs interfering with histamine metabolism or distribution | |
| Prokinetics | metoclopramide |
| Antiinfectives | clavulanic acid, isoniazid, cefuroxime, cefotiame, pentamidine, chloroquine, doxycycline, neomycin B, acriflavine, D-cycloserine |
| Bronchodilators | aminophylline, theophylline |
| Diuretics | amiloride, furosemide |
| Antidepressants | amitriptyline, monoaminooxidase 1 inhibitors |
| Anxiolytics | diazepam, barbiturates |
| Antipsychotics | haloperidol |
| Cytostatics | cyclophosphamide |
| Antihypertensives | verapamil, dihydrazine, alprenolol |
| Cardiotonics | dobutamine, dopamine |
| Opioids | pethidine, morphine, codeine |
| Analgesics | metamizole |
| Local anaesthetics | lidocaine, prilocaine, marcaine, procaine |
| General anaesthetics | thiopental |
| Muscle relaxants | pancuronium, alcuronium, D-tubocurarine |
| Antiarrhytmics | propafenone, verapamil, quinidine |
| Antihistamines decreasing DAO activity | |
| H1/H2 receptor blockers | cimetidine, promethazine |
| Other substances interfering with histamine metabolism | |
| Radiocontrast agents | iodine containing |
| Water, coffee, tea, homemade juices from allowed fruits and vegetables |
| Bread, pastry, potatoes, rice, pasta, cereals, millet, buckwheat, corn |
| Yoghurt, fresh soft cheese |
| Lettuce, cauliflower, broccoli, chicory, carrot, garlic, onion, cucumber, pumpkin, zucchini, pepper, radish, artichoke, rhubarb, asparagus |
| Apple, pear, cherry, amarelle, peach, apricot, watermelon, blueberries |
| Spices, herbs |
| Vegetable oil, vinegar |
| FRESH/IMMEDIATELY FROZEN meat: poultry, veal, beef, lamb, pork |
| FRESH/IMMEDIATELY FROZEN fish: cod/pollock, trout, zander, halibut |
| Ham (fresh, cooked and high-quality), eggs (cooked) |
| Jam made from allowed fruits, honey, butter, margarine |
| Bacteria | ||
|---|---|---|
| Acinetobacter spp. | Chryseobacterium spp. | Pediococcus pentosaceus |
| Alcalingenes faecalis | Klebsiella oxytoca | Proteus spp. |
| Klebsiella pneumoniae | ||
| Arizona spp. | Providencia spp. | |
| Kluyvera spp. | Providencia heimbachae | |
| Cedecea spp. | ||
| Lacticaseibacillus casei | Pseudomonas putida | |
| Citrobacter freundii | Lacticaseibacillus paracasei | Pseudomonas lundensis |
| Citrobacter braakii | Lacticaseibacillus rhamnosus | Pseudomonas stutzeri |
| Lactiplantibacillus plantarum | ||
| Lactobacillus curvatus | ||
| Edwardsiella spp. | Lactobacillus delbrueckii | Psychrobacter spp. |
| Lactobacillus helveticus | ||
| Enterococcus casseliflavus | Lactococcus lactis ssp. lactis | Raoultella planticola |
| Enterococcus faecalis | Latilactobacillus spp. | Raoultella ornithinolytica |
| Enterococcus faecium | Lentilactobacillus buchneri | |
| Lentilactobacillus hilgardii | Salmonella enterica ssp. arizonae | |
| Enterobacter spp. | Lentilactobacillus parabuchneri | |
| Leuconostoc spp. | ||
| Escherichia coli | Levilactobacillus brevis | Serratia spp. |
| Escherichia fergusonii | Limosilactobacillus reuteri | |
| Limosilactobacillus vaginalis | Sphingobacterium spp. | |
| Hafnia alvei | ||
| Hafnia paralvei | Microbacterium foliorum | Streptococcus thermophilus |
| Halomonas spp. | Morganella morganii | Tetragenococcus halophilus |
| Yeasts and moulds | ||
| Debaryomyces hansenii | Geotrichum candidum | |
| Phase | Objective | Recommendation | Duration |
|---|---|---|---|
| Phase 1: Elimination phase | Reduction in symptoms to a maximum possible level | • Change in diet composition - introduction of mixed diet measures with accent on fresh vegetables and reduction of biogenic amine intake, in particular histamine • Nutrient optimization | 10–14 days |
| Phase 2: Test phase | Reintroducing foods excluded in Phase 1, after taking into account individual risk factors (stress, menstruation, medication use etc.) | • Targeted gradual reintroduction of suspected foods taking into consideration patient’s individual dietary preferences • Assessment of individual sensitivity to ingested histamine | Up to 6 weeks |
| Phase 3: Long-term diet | Maintenance of high-quality of life Continual balanced diet | • Individual nutritional recommendations based on individual sensitivity to ingested histamine taking exogenous risk factors into consideration | _ |
| Study | Trial Design | Intervention | Control | Sample size | Duration of Intervention | Reported Outcomes |
|---|---|---|---|---|---|---|
| Komericki, 2011 | DBPC cross-over study | 0.5 mg DAO | Placebo | 39 | N/A | A significant improvement in symptoms after DAO administration as compared with placebo. |
| Manzotti, 2016 | Restrospective observational study | 2 × 0.3 mg DAO | N/A | 14 | 14 | 13 patients (93%) reported improvement in ≥ 1 of symptoms. |
| Yacoub, 2018 | DBPC cross-over study | 2 × 0.3 mg DAO | Placebo | 20 | 30 | A significant improvement in Urticaria Activity Score 7 (UAS7) in patients with urticaria unsatisfactorily controlled by antihistamines (p < 0.05). Mild significant reduction in antihistamines consumption (p < 0.05). |
| Izquierdo-Casas, 2019 | Randomized DBPC study | 3 × 0.6 mg DAO | Placebo | 82 | C1 without an intervention (30 days) + C2 with intervention (30 days) | A significant reduction in number (p < 0.001) and duration (p < 0.05) of migraine episodes in intervention group compared with baseline. A decrease in the percentage of patients using selective 5-HT receptor agonists (triptans). |
| Schnedl, 2019 | Open label interventional study | 3 × 0.3 mg DAO | N/A | 28 | 28 + 28 (follow-up) | Significant reduction in frequency and intensity of symptoms. 61% of patients showed mild increase in serum DAO levels (NS). During follow-up, without DAO supplementation, the symptoms sum score increased again and serum DAO levels slightly decreased. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hrubisko, M.; Danis, R.; Huorka, M.; Wawruch, M. Histamine Intolerance—The More We Know the Less We Know. A Review. Nutrients 2021, 13, 2228. https://doi.org/10.3390/nu13072228
Hrubisko M, Danis R, Huorka M, Wawruch M. Histamine Intolerance—The More We Know the Less We Know. A Review. Nutrients. 2021; 13(7):2228. https://doi.org/10.3390/nu13072228
Chicago/Turabian StyleHrubisko, Martin, Radoslav Danis, Martin Huorka, and Martin Wawruch. 2021. "Histamine Intolerance—The More We Know the Less We Know. A Review" Nutrients 13, no. 7: 2228. https://doi.org/10.3390/nu13072228
APA StyleHrubisko, M., Danis, R., Huorka, M., & Wawruch, M. (2021). Histamine Intolerance—The More We Know the Less We Know. A Review. Nutrients, 13(7), 2228. https://doi.org/10.3390/nu13072228





