Vitamin D Supplementation Regulates Postoperative Serum Levels of PD-L1 in Patients with Digestive Tract Cancer and Improves Survivals in the Highest Quintile of PD-L1: A Post Hoc Analysis of the AMATERASU Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Trial Design
2.2. Participants
2.3. Measurement of Serum PD-L1 Levels
2.4. Evaluation of Other Covariates
2.5. Statistical Analysis
3. Results
3.1. Study Population
3.2. Patients’ Characteristics Stratified by Vitamin D Group and Placebo Group
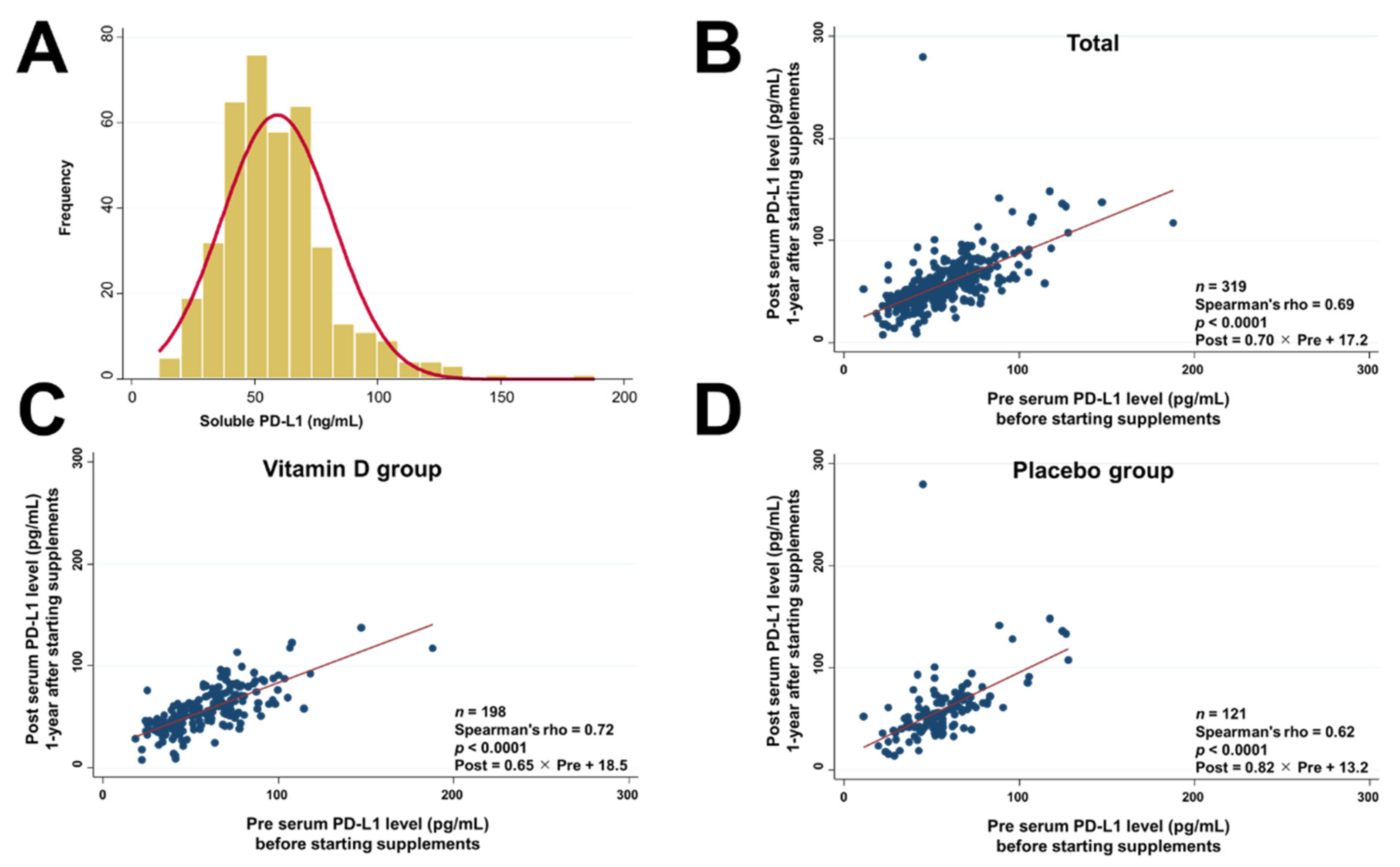
3.3. Serum PD-L1 Levels before and after Starting Supplements
3.4. Patients’ Characteristics Stratified by Quintiles of Serum PD-L1 Levels
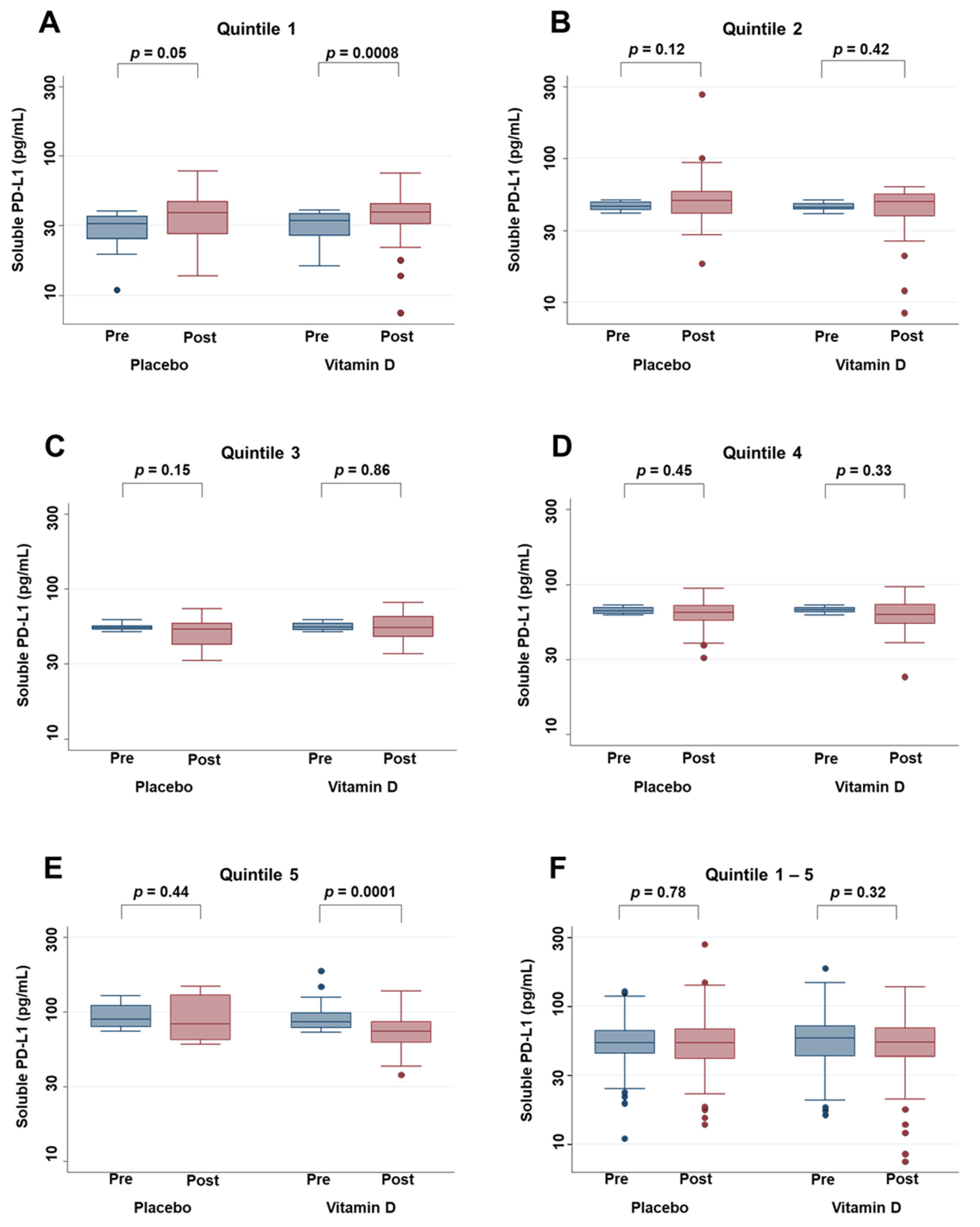
3.5. Effects of Vitamin D Supplementation on Serum PD-L1 Levels
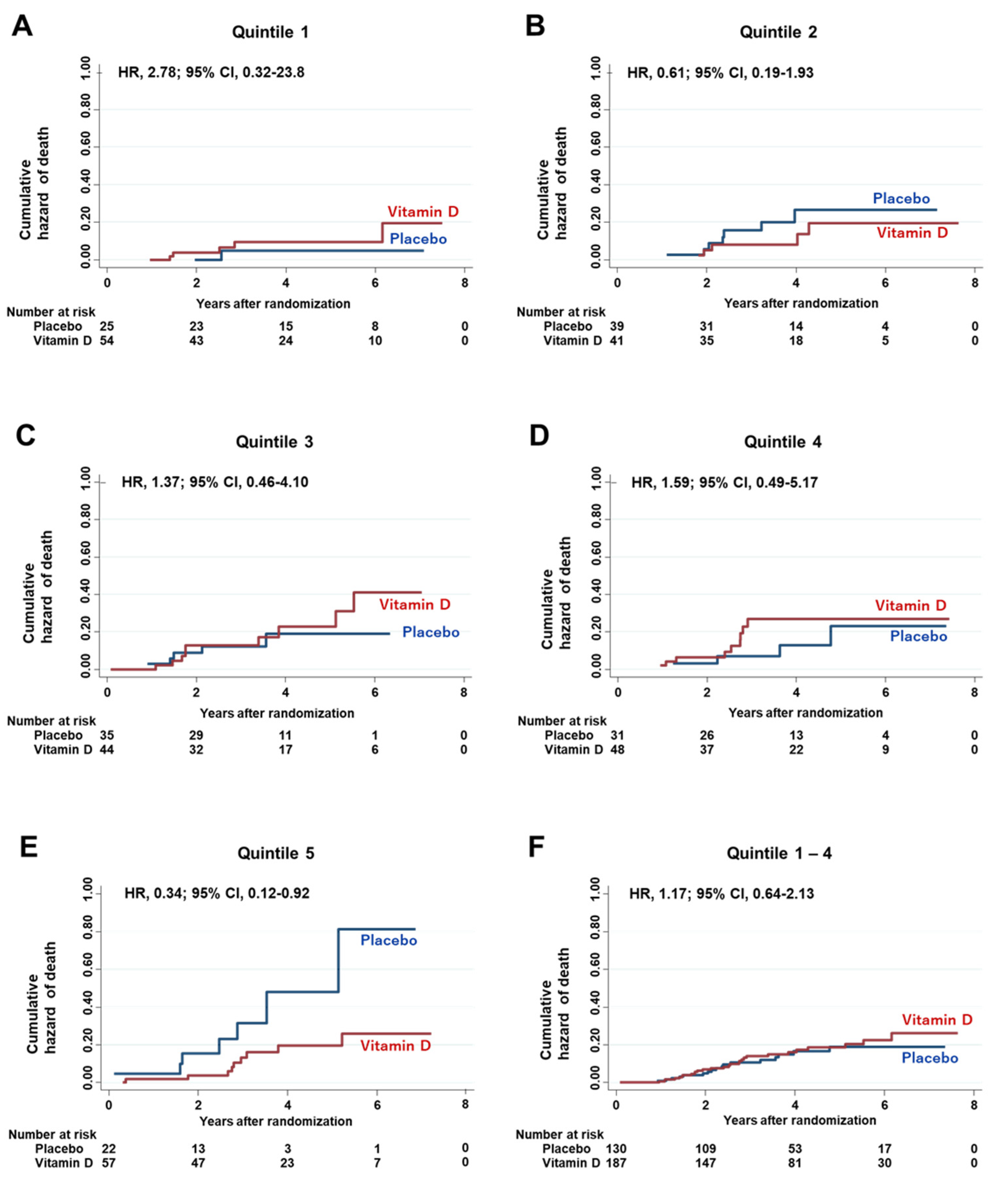
3.6. Effect of the Interaction between Vitamin D Supplementation and Serum PD-L1 Quintiles on Hazard Risk of Death
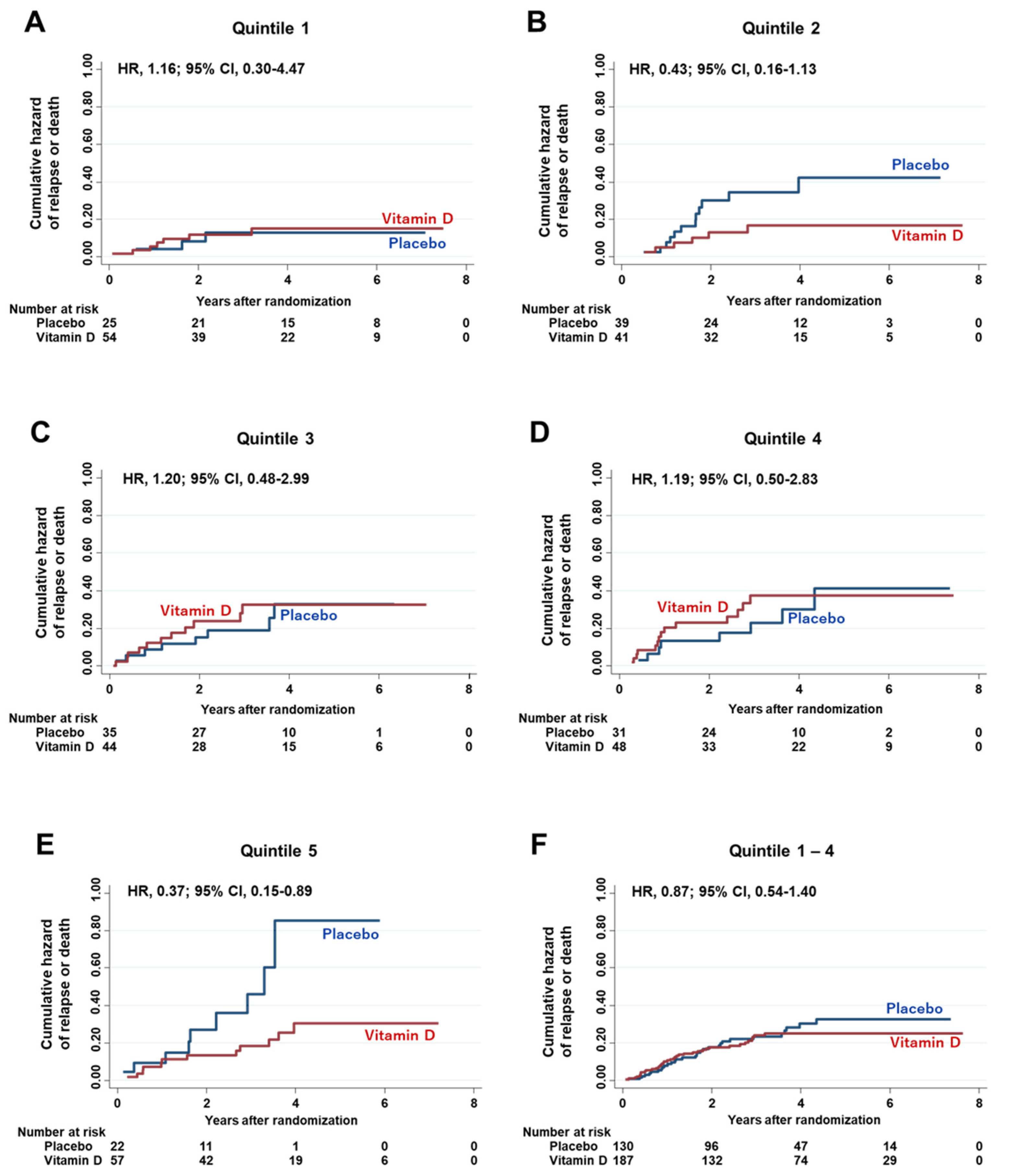
3.7. Effect of the Interaction between Vitamin D Supplementation and Serum PD-L1 Quintiles on Hazard Risk of Relapse or Death
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boussiotis, V.A. Molecular and Biochemical Aspects of the PD-1 Checkpoint Pathway. N. Engl. J. Med. 2016, 375, 767–778. [Google Scholar] [CrossRef]
- Gong, J.; Chehrazi-Raffle, A.; Reddi, S.; Salgia, R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: A comprehensive review of registration trials and future considerations. J. Immunother. Cancer 2018, 6, 8. [Google Scholar] [CrossRef]
- Zhu, X.; Lang, J. Soluble PD-1 and PD-L1: Predictive and prognostic significance in cancer. Oncotarget 2017, 8, 97671–97682. [Google Scholar] [CrossRef]
- Romero, Y.; Wise, R.; Zolkiewska, A. Proteolytic processing of PD-L1 by ADAM proteases in breast cancer cells. Cancer Immunol. Immunother. 2020, 69, 43–55. [Google Scholar] [CrossRef]
- Orme, J.J.; Jazieh, K.A.; Xie, T.; Harrington, S.; Liu, X.; Ball, M.; Madden, B.; Charlesworth, M.C.; Azam, T.; Lucien, F.; et al. ADAM10 and ADAM17 cleave PD-L1 to mediate PD-(L)1 inhibitor resistance. OncoImmunology 2020, 9, 1744980. [Google Scholar] [CrossRef] [PubMed]
- Frigola, X.; Inman, B.A.; Lohse, C.M.; Krco, C.J.; Cheville, J.C.; Thompson, R.H.; Leibovich, B.; Blute, M.L.; Dong, H.; Kwon, E.D. Identification of a Soluble Form of B7-H1 That Retains Immunosuppressive Activity and Is Associated with Aggressive Renal Cell Carcinoma. Clin. Cancer Res. 2011, 17, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Frigola, X.; Inman, B.A.; Krco, C.J.; Liu, X.; Harrington, S.M.; Bulur, P.A.; Dietz, A.B.; Dong, H.; Kwon, E.D. Soluble B7-H1: Differences in production between dendritic cells and T cells. Immunol. Lett. 2012, 142, 78–82. [Google Scholar] [CrossRef] [PubMed]
- Okuyama, M.; Mezawa, H.; Kawai, T.; Urashima, M. Elevated Soluble PD-L1 in Pregnant Women’s Serum Suppresses the Immune Reaction. Front Immunol. 2019, 10, 86. [Google Scholar] [CrossRef]
- Li, X.; Zheng, Y.; Yue, F. Prognostic Value of Soluble Programmed Cell Death Ligand-1 (sPD-L1) in Various Cancers: A Meta-analysis. Target. Oncol. 2021, 16, 13–26. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Wang, G.; Chi, F.; Jia, Y.; Wang, X.; Mu, Q.; Qin, K.; Zhu, X.; Pang, J.; Xu, B.; et al. Higher postoperative plasma EV PD-L1 predicts poor survival in patients with gastric cancer. J. Immunother. Cancer 2021, 9, e002218. [Google Scholar] [CrossRef]
- Orme, J.J.; Enninga, E.A.L.; Lucien-Matteoni, F.; Dale, H.; Burgstaler, E.; Harrington, S.M.; Ball, M.K.; Mansfield, A.S.; Park, S.S.; Block, M.S.; et al. Therapeutic plasma exchange clears circulating soluble PD-L1 and PD-L1-positive extracellular vesicles. J. Immunother. Cancer 2020, 8, e001113. [Google Scholar] [CrossRef] [PubMed]
- Feldman, D.; Krishnan, A.V.; Swami, S.; Giovannucci, E.; Feldman, B.J. The role of vitamin D in reducing cancer risk and progression. Nat. Rev. Cancer 2014, 14, 342–357. [Google Scholar] [CrossRef]
- Dimitrov, V.; Bouttier, M.; Boukhaled, G.; Salehi-Tarbar, R.; Avramescu, R.G.; Memari, B.; Hasaj, B.; Lukacs, G.L.; Krawczyk, C.M.; White, J.H. Hormonal vitamin D up-regulates tissue-specific PD-L1 and PD-L2 surface glycoprotein expression in humans but not mice. J. Biol. Chem. 2017, 292, 20657–20668. [Google Scholar] [CrossRef] [PubMed]
- Urashima, M.; Ohdaira, H.; Akutsu, T.; Okada, S.; Yoshida, M.; Kitajima, M.; Suzuki, Y. Effect of Vitamin D Supplementation on Relapse-Free Survival Among Patients With Digestive Tract Cancers. JAMA 2019, 321, 1361–1369. [Google Scholar] [CrossRef] [PubMed]
- Yonaga, H.; Okada, S.; Akutsu, T.; Ohdaira, H.; Suzuki, Y.; Urashima, M. Effect Modification of Vitamin D Supplementation by Histopathological Characteristics on Survival of Patients with Digestive Tract Cancer: Post Hoc Analysis of the AMATERASU Randomized Clinical Trial. Nutrition 2019, 11, 2547. [Google Scholar] [CrossRef] [PubMed]
- Akutsu, T.; Okada, S.; Hirooka, S.; Ikegami, M.; Ohdaira, H.; Suzuki, Y.; Urashima, M. Effect of Vitamin D on Relapse-Free Survival in a Subgroup of Patients with p53 Protein-Positive Digestive Tract Cancer: A Post Hoc Analysis of the AMATERASU Trial. Cancer Epidemiol. Biomark. Prev. 2020, 29, 406–413. [Google Scholar] [CrossRef] [PubMed]
- Urashima, M.; Okuyama, M.; Akutsu, T.; Ohdaira, H.; Kaji, M.; Suzuki, Y. Effect of Vitamin D Supplementation on Survival of Digestive Tract Cancer Patients with Low Bioavailable 25-Hydroxyvitamin D Levels: A Post Hoc Analysis of the AMATERASU Randomized Clinical Trial. Cancers 2020, 12, 347. [Google Scholar] [CrossRef] [PubMed]
- Fine, J.P.; Gray, R.J. A Proportional Hazards Model for the Subdistribution of a Competing Risk. J. Am. Stat. Assoc. 1999, 94, 496–509. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, Q.; Shi, B.; Xu, P.; Hu, Z.; Bai, L.; Zhang, X. Development of a sandwich ELISA for evaluating soluble PD-L1 (CD274) in human sera of different ages as well as supernatants of PD-L1+ cell lines. Cytokine 2011, 56, 231–238. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Paz-Ares, L.; Caro, R.B.; Zurawski, B.; Kim, S.-W.; Costa, E.C.; Park, K.; Alexandru, A.; Lupinacci, L.; De la Mora, J.E.; et al. Nivolumab plus ipilimumab in advanced non–small-cell lung cancer. N. Engl. J. Med. 2019, 381, 2020–2031. [Google Scholar] [CrossRef]
- Schmid, P.; Adams, S.; Rugo, H.S.; Schneeweiss, A.; Barrios, C.H.; Iwata, H.; Diéras, V.; Hegg, R.; Im, S.-A.; Wright, G.S.; et al. Atezolizumab and Nab-Paclitaxel in Advanced Triple-Negative Breast Cancer. N. Engl. J. Med. 2018, 379, 2108–2121. [Google Scholar] [CrossRef]
- Eggermont, A.M.M.; Blank, C.U.; Mandalà, M.; Long, G.V.; Atkinson, V.; Dalle, S.; Haydon, A.; Lichinitser, M.; Khattak, A.; Carlino, M.S.; et al. Adjuvant Pembrolizumab versus Placebo in Resected Stage III Melanoma. N. Engl. J. Med. 2018, 378, 1789–1801. [Google Scholar] [CrossRef]
- Ng, K.; Nimeiri, H.S.; McCleary, N.J.; Abrams, T.A.; Yurgelun, M.B.; Cleary, J.M.; Rubinson, D.A.; Schrag, D.; Miksad, R.; Bullock, A.J.; et al. Effect of High-Dose vs. Standard-Dose Vitamin D3 Supplementation on Progression-Free Survival Among Patients With Advanced or Metastatic Colorectal Cancer. JAMA 2019, 321, 1370–1379. [Google Scholar] [CrossRef] [PubMed]
- Akutsu, T.; Kitamura, H.; Himeiwa, S.; Kitada, S.; Akasu, T.; Urashima, M. Vitamin D and Cancer Survival: Does Vitamin D Supplementation Improve the Survival of Patients with Cancer? Curr. Oncol. Rep. 2020, 22, 62. [Google Scholar] [CrossRef] [PubMed]
- Vaughan-Shaw, P.G.; Buijs, L.F.; Blackmur, J.P.; Theodoratou, E.; Zgaga, L.; Din, F.V.N.; Farrington, S.M.; Dunlop, M.G. The effect of vitamin D supplementation on survival in patients with colorectal cancer: Systematic review and meta-analysis of randomized controlled trials. Br. J. Cancer 2020, 123, 1705–1712. [Google Scholar] [CrossRef] [PubMed]
- Keum, N.; Lee, D.H.; Greenwood, D.C.; Manson, J.E.; Giovannucci, E. Vitamin D supplementation and total cancer incidence and mortality: A meta-analysis of randomized controlled trials. Ann. Oncol. 2019, 30, 733–743. [Google Scholar] [CrossRef]
- Zhang, Y.; Fang, F.; Tang, J.; Jia, L.; Feng, Y.; Xu, P.; Faramand, A. Association between vitamin D supplementation and mortality: Systematic review and meta-analysis. BMJ 2019, 366, l4673. [Google Scholar] [CrossRef]
- Harrington, D.; D’Agostino, R.B.; Gatsonis, C.; Hogan, J.W.; Hunter, D.J.; Normand, S.-L.T.; Drazen, J.M.; Hamel, M.B. New Guidelines for Statistical Reporting in the Journal. N. Engl. J. Med. 2019, 381, 285–286. [Google Scholar] [CrossRef]
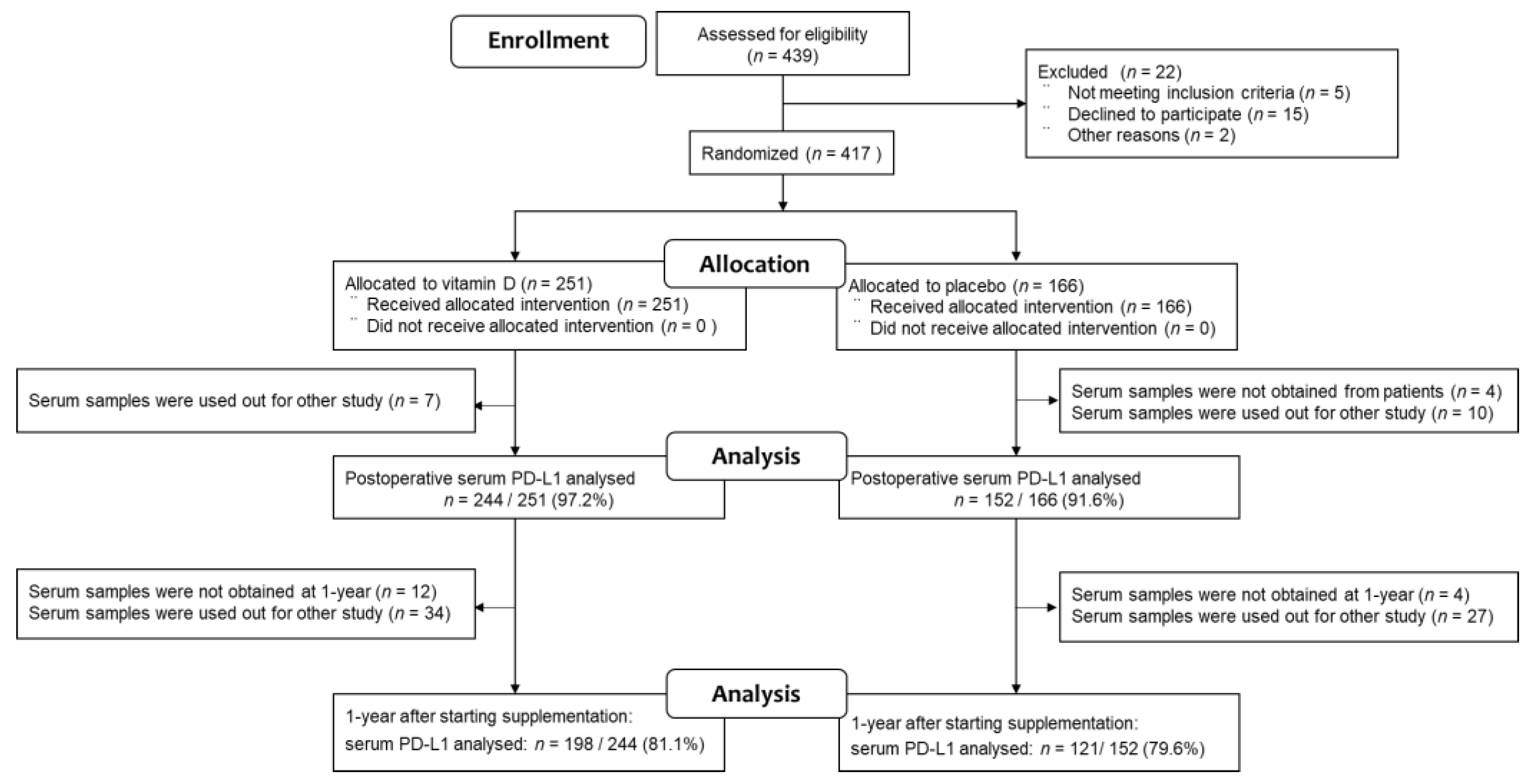
| n = 396 | Vitamin D n = 244 | Placebo n = 152 |
|---|---|---|
| 25(OH)D, ng/mL | n = 241 | n = 152 |
| median | 21 | 21 |
| IQR a (25–75%) | (17–27) | (14.5–26) |
| 25(OH)D, ng/mL 1 year after supplementation | n = 208 | n = 132 |
| median | 41 | 21 |
| IQR a (25–75%) | (33–55) | (15–27) |
| Bioavailable 25(OH)D, ng/mL | n = 214 | n = 136 |
| median | 1.8 | 1.6 |
| IQR a (25–75%) | (1.2–2.8) | (1.1–2.3) |
| Bioavailable 25(OH)D, ng/mL 1 year after supplementation | n = 177 | n = 117 |
| median | 5.0 | 2.2 |
| IQR a (25–75%) | (3.4–7.5) | (1.6–3.3) |
| Sex, n (%) | n = 244 | n = 152 |
| Male | 171 (70) | 94 (62) |
| Female | 73 (30) | 58 (38) |
| Age, y | n = 244 | n = 152 |
| median | 67 | 64 |
| IQR a (25–75%) | (61–75) | (58–70) |
| Body mass index (kg/m2) | n = 242 | n = 151 |
| median | 21.9 | 22.1 |
| IQR a (25–75%) | (19.8–24.0) | (20.0–23.7) |
| History of other cancers, n (%) | 8 (3) | 7 (5) |
| Comorbid condition, n (%) | n = 244 | n = 152 |
| Hypertension | 101 (41) | 54 (36) |
| Diabetes Mellitus | 44 (18) | 21 (14) |
| Endocrine Disease | 32 (13) | 16 (11) |
| Coronary Artery Disease | 16 (7) | 2 (1) |
| Stroke | 10 (4) | 6 (4) |
| Chronic Kidney Disease | 4 (2) | 1 (0.7) |
| Asthma | 3 (1) | 0 (0) |
| Orthopaedic disease | 1 (0.4) | 1 (0.7) |
| Site of cancer, n (%) | n = 244 | n = 152 |
| Oesophagus | 22 (9) | 15 (10) |
| Stomach | 104 (43) | 64 (42) |
| Small bowel | 1 (0.4) | 1 (0.7) |
| Colorectal | 117 (48) | 72 (47) |
| Stage, n (%) | n = 244 | n = 152 |
| I | 113 (46) | 61 (40) |
| II | 61 (25) | 43 (28) |
| III | 70 (29) | 48 (32) |
| Pathology b | ||
| Adenocarcinoma, n (%) | n = 244 | n = 152 |
| Well-differentiated | 137 (56) | 75 (49) |
| Moderately differentiated | 93 (38) | 66 (43) |
| Poorly differentiated | 43 (18) | 32 (21) |
| Signet ring cell | 18 (7) | 22 (14) |
| Mucinous | 18 (7) | 8 (5) |
| Papillary | 11 (5) | 4 (3) |
| Squamous cell carcinoma, n (%) | 20 (8) | 11 (7) |
| P53 expression, n (%) | n = 214 | n = 140 |
| None | 35 (16) | 28 (20) |
| Faintly expressed: >0% & <10% | 43 (20) | 31 (22) |
| Strongly expressed: ≥10% & <50% | 30 (14) | 26 (19) |
| Overexpressed: ≥50% | 106 (50) | 55 (39) |
| Vitamin D receptor expression, n (%) | n = 214 | n = 140 |
| Quartile 1, | 59 (28) | 33 (24) |
| Quartile 2, | 54 (25) | 35 (25) |
| Quartile 3, | 51 (24) | 35 (25) |
| Quartile 4, | 50 (23) | 37 (26) |
| Ki67 expression, n (%) | n = 214 | n = 140 |
| Quartile 1, | 37 (17) | 25 (18) |
| Quartile 2, | 76 (36) | 40 (29) |
| Quartile 3, | 30 (14) | 29 (21) |
| Quartile 4, | 71 (33) | 46 (33) |
| Adjuvant chemotherapy, n (%) | 84 (34) | 56 (37) |
| Total n = 396 | Q1 n = 79 | Q2 n = 80 | Q3 n = 79 | Q4 n = 79 | Q5 n = 79 | |
|---|---|---|---|---|---|---|
| Median (IQR b), pg/mL | 55.5 (44.2–70.2) | 34.4 (26.1–38.7) | 45.7 (44.2–49.1) | 55.5 (53.4–58.7) | 67.3 (64.7–70.2) | 86.8 (78.8–103.6) |
| Intervention | ||||||
| Vitamin D, n (%) | 244 (62) | 54 (68) | 41 (51) | 44 (56) | 48 (61) | 57 (72) |
| Placebo, n (%) | 152 (38) | 25 (32) | 39 (49) | 35 (44) | 31 (39) | 22 (28) |
| 25(OH)D c, ng/mL median (IQR b) | ||||||
| All | 21 (16–27) | 22 (17–28) | 20 (17–27) | 20 (14–25) | 22 (17–28) | 20 (14–26) |
| Vitamin D supplementation | 21 (17–27) | 23 (18–28) | 22 (18–28) | 19 (15–25) | 23 (19–30) | 20 (14–26) |
| Placebo supplementation | 21 (15–26) | 21 (16–25) | 20 (15–27) | 22 (13–26) | 21 (14–28) | 19 (15–26) |
| 25(OH)D c, ng/mL 1 year after supplementation median (IQR b) | ||||||
| All | 33 (21–47) | 32 (20–47) | 32 (19–42) | 32 (21–41) | 31 (22–47) | 37 (21–55) |
| Vitamin D supplementation. | 41 (33–55) | 40 (30–54) | 40 (35–54) | 40 (33–54) | 35 (45–58) | 44 (35–60) |
| Placebo supplementation | 21 (15–27) | 23 (17–30) | 19 (13–28) | 22 (16–29) | 22 (18–25) | 16 (11–23) |
| Bioavailable 25(OH)D c, ng/mL median (IQR b) | ||||||
| All | 1.71 (1.18–2.59) | 1.73 (1.33–2.73) | 1.79 (1.28–2.62) | 1.75 (1.04–2.31) | 1.94 (1.26–2.87) | 1.45 (1.00–2.16) |
| Vitamin D supplementation | 1.80 (1.23–2.79) | 1.97 (1.17–3.06) | 1.95 (1.54–2.74) | 1.70 (1.14–2.22) | 2.17 (1.57–3.12) | 1.52 (1.02–2.48) |
| Placebo supplementation | 1.63 (1.08–2.28) | 1.68 (1.53–2.02) | 1.71 (1.25–2.42) | 1.90 (0.97–2.62) | 1.47 (0.97–2.40) | 1.26 (0.97–1.65) |
| Bioavailable 25(OH)D c, ng/mL 1 year after supplementation median (IQR b) | ||||||
| All | 3.62 (2.16–6.20) | 3.59 (2.43–5.05) | 3.48 (2.00–6.25) | 3.35 (1.82–5.71) | 3.93 (2.07–6.94) | 4.20 (2.16–6.88) |
| Vitamin D supplementation. | 5.05 (3.37–7.51) | 4.31 (3.25–7.13) | 4.85 (3.60–7.58) | 5.21 (3.28–6.96) | 5.58 (3.43–8.36) | 6.19 (2.82–7.54) |
| Placebo supplementation | 2.25 (1.60–3.32) | 2.43 (1.76–3.64) | 2.21 (1.27–3.31) | 2.22 (1.66–3.74) | 2.84 (1.64–3.55) | 2.16 (1.41–2.53) |
| Sex, n (%) | ||||||
| Male | 265 (67) | 48 (61) | 47 (59) | 58 (73) | 50 (63) | 62 (78) |
| Female | 131 (33) | 31 (39) | 33 (41) | 21 (27) | 29 (37) | 17 (22) |
| Age, y | ||||||
| median (IQR b) | 66 (60–74) | 63 (57–70) | 64 (59–73) | 64 (57–70) | 70 (62–75) | 72 (64–78) |
| Body mass index (kg/m2) d | ||||||
| median (IQR b) | 21.9 (19.8–23.8) | 22.4 (20.0–24.0) | 21.4 (20.0–23.5) | 21.7 (20.4–24.2) | 21.9 (20.0–23.7) | 21.6 (19.2–24.1) |
| History of other cancers, n (%) | 15 (3.8) | 4 (5.1) | 3 (3.8) | 5 (6.3) | 2 (2.5) | 1 (1.3) |
| Comorbid condition, n (%) a | ||||||
| Hypertension | 155 (39) | 25 (32) | 25 (32) | 41 (52) | 28 (35) | 36 (46) |
| Diabetes Mellitus | 65 (16) | 11 (14) | 11 (14) | 13 (16) | 12 (15) | 18 (23) |
| Endocrine Disease | 48 (12) | 9 (11) | 13 (16) | 11 (14) | 7 (8.9) | 8 (10) |
| Coronary Artery Disease | 18 (4.5) | 2 (2.5) | 3 (3.8) | 3 (3.8) | 2 (2.5) | 8 (10.1) |
| Stroke | 16 (4.0) | 1 (1.3) | 2 (2.5) | 4 (5.1) | 3 (3.8) | 6 (7.6) |
| Chronic Kidney Disease | 5 (1.3) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 2. (2.5) | 3 (3.8) |
| Asthma | 3 (0.8) | 0 (0.0) | 0 (0.0) | 0 (0.0) | 1 (1.3) | 2 (2.5) |
| Orthopaedic disease | 2 (0.5) | 0 (0.0) | 1 (1.3) | 1 (1.3) | 0 (0.0) | 0 (0.0) |
| Site of cancer, n (%) a | ||||||
| Oesophagus | 37 (9.3) | 3 (3.8) | 6 (7.5) | 7 (8.9) | 11 (13.9) | 10 (12.7) |
| Stomach | 168 (42.4) | 39 (49.4) | 37 (46.3) | 29 (36.7) | 28 (35.4) | 35 (44.3) |
| Small bowel | 2 (0.5) | 0 (0.0) | 0 (0.0) | 1 (1.3) | 1 (1.3) | 0 (0.0) |
| Colorectal | 189 (47.7) | 37 (46.8) | 37 (46.3) | 42 (53.2) | 39 (49.4) | 34 (43.0) |
| Stage, n (%) a | ||||||
| I | 174 (43.9) | 40 (50.6) | 29 (36.3) | 38 (48.1) | 36 (45.6) | 31 (39.2) |
| II | 104 (26.3) | 19 (24.1) | 23 (28.8) | 18 (22.8) | 21 (26.6) | 23 (29.1) |
| III | 118 (29.8) | 20 (25.3) | 28 (35.0) | 23 (29.1) | 22 (27.8) | 25 (31.6) |
| Pathology, n (%) e | ||||||
| Adenocarcinoma | ||||||
| Well-differentiated | 212 (53.5) | 41 (51.9) | 36 (45.0) | 47 (59.5) | 41 (51.9) | 47 (59.5) |
| Moderately differentiated | 159 (40.2) | 33 (41 8) | 34 (42.5) | 25 (31.6) | 38 (48.1) | 29 (36.7) |
| Poorly differentiated | 75 (18.9) | 18 (22.8) | 22 (27.5) | 11 (13.9) | 11 (13.9) | 13 (16.5) |
| Signet ring cell | 40 (10.1) | 14 (17.7) | 9 (11.3) | 9 (11.4) | 5 (6.3) | 3 (3.8) |
| Mucinous | 26 (6.6) | 3 (3.8) | 7 (8.8) | 7 (8.9) | 1 (1.3) | 8 (10.1) |
| Papillary | 15 (3.8) | 3 (3.8) | 2 (2.5) | 2 (2.5) | 4 (5.1) | 4 (5.1) |
| Squamous cell carcinoma, n (%) | 31 (7.8) | 2 (2.5) | 5 (6.3) | 7 (8.9) | 9 (11.4) | 8 (10.1) |
| P53 expression, n (%) a | ||||||
| None | 63 (17.8) | 13 (19.7) | 16 (22.5) | 13 (18.3) | 13 (17.3) | 8 (11.3) |
| Faintly expressed: >0% & <10% | 74 (20.9) | 14 (21.2) | 19 (26.8) | 11 (15.5) | 13 (17.3) | 17 (23.9) |
| Strongly expressed: ≥10% & <50% | 56 (15.8) | 13 (19.7) | 14 (19.7) | 12 (16.9) | 10 (13.3) | 7 (9.9) |
| Overexpressed: ≥50% | 161 (45.5) | 26 (39.4) | 22 (31.0) | 35 (49.3) | 39 (52.0) | 39 (54.9) |
| Vitamin D receptor expression, n (%) a | ||||||
| Q1, | 92 (26.0) | 20 (30.3) | 22 (31.0) | 17 (23.9) | 17 (22.7) | 16 (22.5) |
| Q2, | 89 (25.1) | 15 (22.7) | 17 (23.9) | 15 (21.1) | 19 (25.3) | 23 (32.4) |
| Q3, | 86 (24.3) | 18 (27.3) | 15 (21.1) | 20 (28.2) | 17 (22.7) | 16 (22.5) |
| Q4, | 87 (24.6) | 13 (19.7) | 17 (23.9) | 19 (26.8) | 22 (29.3) | 16 (22.5) |
| Ki67 expression, n (%) a | ||||||
| Q1, | 62 (17.5) | 15 (22.7) | 16 (22.5) | 10 (14.1) | 10 (13.3) | 11 (15.5) |
| Q2, | 116 (32.8) | 21 (31.8) | 24 (33.8) | 28 (39.4) | 18 (24.0) | 25 (32.2) |
| Q3, | 59 (16.7) | 15 (22.7) | 8 (11.3) | 11 (15.5) | 15 (20.0) | 10 (14.1) |
| Q4, | 117 (33.1) | 15 (22.7) | 23 (32.4) | 22 (31.0) | 32 (42.7) | 25 (35.2) |
| Adjuvant chemotherapy, n (%) a | 140 (35.4) | 28 (35.4) | 29 (36.3) | 28 (35.4) | 25 (31.6) | 30 (38.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morita, M.; Okuyama, M.; Akutsu, T.; Ohdaira, H.; Suzuki, Y.; Urashima, M. Vitamin D Supplementation Regulates Postoperative Serum Levels of PD-L1 in Patients with Digestive Tract Cancer and Improves Survivals in the Highest Quintile of PD-L1: A Post Hoc Analysis of the AMATERASU Randomized Controlled Trial. Nutrients 2021, 13, 1987. https://doi.org/10.3390/nu13061987
Morita M, Okuyama M, Akutsu T, Ohdaira H, Suzuki Y, Urashima M. Vitamin D Supplementation Regulates Postoperative Serum Levels of PD-L1 in Patients with Digestive Tract Cancer and Improves Survivals in the Highest Quintile of PD-L1: A Post Hoc Analysis of the AMATERASU Randomized Controlled Trial. Nutrients. 2021; 13(6):1987. https://doi.org/10.3390/nu13061987
Chicago/Turabian StyleMorita, Makoto, Mai Okuyama, Taisuke Akutsu, Hironori Ohdaira, Yutaka Suzuki, and Mitsuyoshi Urashima. 2021. "Vitamin D Supplementation Regulates Postoperative Serum Levels of PD-L1 in Patients with Digestive Tract Cancer and Improves Survivals in the Highest Quintile of PD-L1: A Post Hoc Analysis of the AMATERASU Randomized Controlled Trial" Nutrients 13, no. 6: 1987. https://doi.org/10.3390/nu13061987
APA StyleMorita, M., Okuyama, M., Akutsu, T., Ohdaira, H., Suzuki, Y., & Urashima, M. (2021). Vitamin D Supplementation Regulates Postoperative Serum Levels of PD-L1 in Patients with Digestive Tract Cancer and Improves Survivals in the Highest Quintile of PD-L1: A Post Hoc Analysis of the AMATERASU Randomized Controlled Trial. Nutrients, 13(6), 1987. https://doi.org/10.3390/nu13061987






