Bad Prognosis in Critical Ill Patients with COVID-19 during Short-Term ICU Stay regarding Vitamin D Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Subjects
2.2. Data Collection
2.3. Blood Sampling and Biochemical Parameters
2.4. Analytical Determination of Vitamin D
2.5. Statistical Analysis
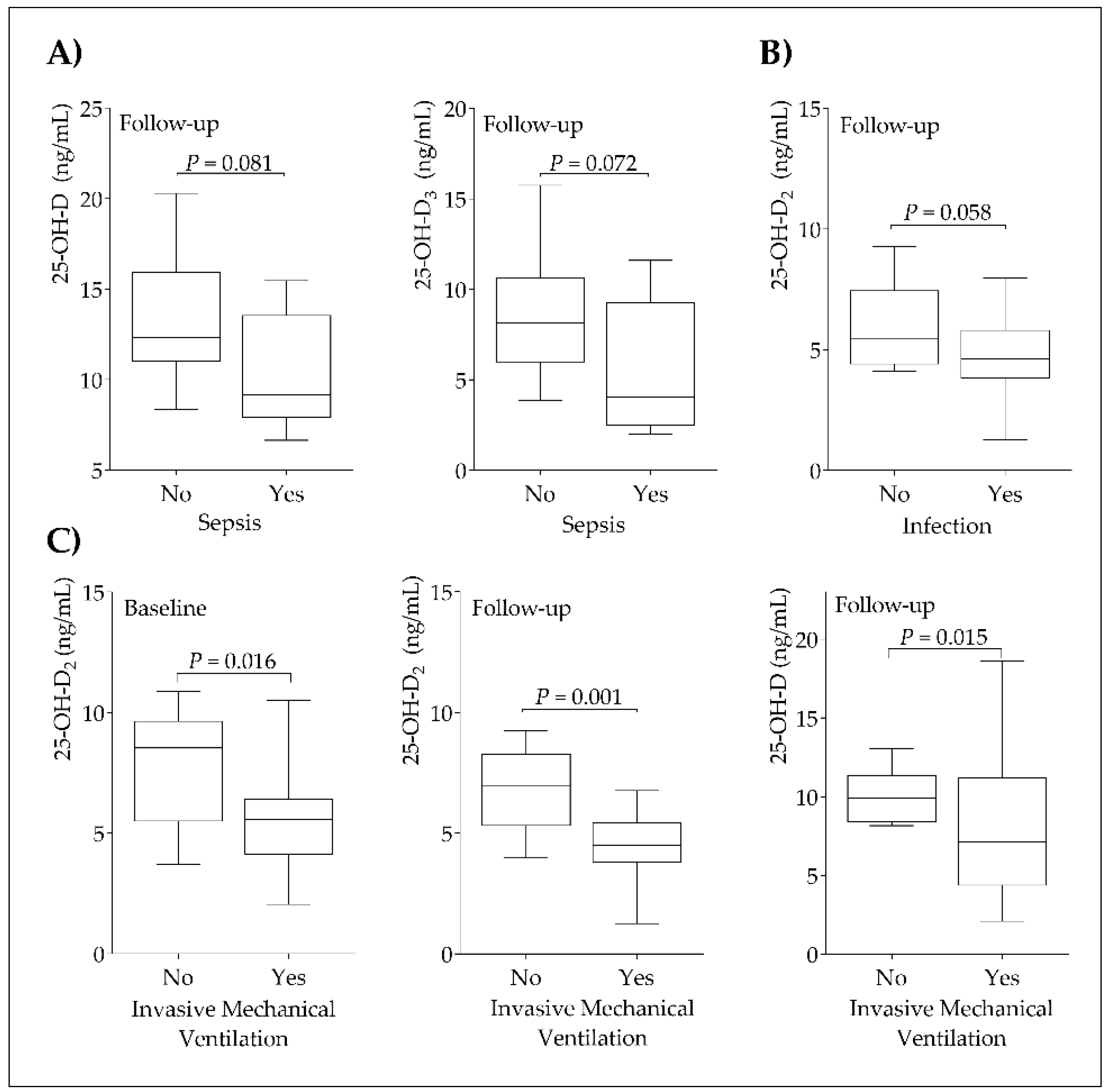
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ali, N. Role of Vitamin D in Preventing of COVID-19 Infection, Progression and Severity. J. Infect. Public Health 2020, 13, 1373–1380. [Google Scholar] [CrossRef] [PubMed]
- De las Heras, N.; Martín Giménez, V.M.; Ferder, L.; Manucha, W.; Lahera, V. Implications of Oxidative Stress and Potential Role of Mitochondrial Dysfunction in COVID-19: Therapeutic Effects of Vitamin D. Antioxidants 2020, 9, 897. [Google Scholar] [CrossRef] [PubMed]
- Han, H.; Ma, Q.; Li, C.; Liu, R.; Zhao, L.; Wang, W.; Zhang, P.; Liu, X.; Gao, G.; Liu, F.; et al. Profiling Serum Cytokines in COVID-19 Patients Reveals IL-6 and IL-10 Are Disease Severity Predictors. Emerg. Microbes Infect. 2020, 9, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Aygun, H. Vitamin D Can Prevent COVID-19 Infection-Induced Multiple Organ Damage. Naunyn. Schmiedebergs Arch. Pharmacol. 2020, 393, 1157–1160. [Google Scholar] [CrossRef] [PubMed]
- Abilés, J.; Aguayo, E.; Moreno-Torres, R.; Llopis, J.; Aranda, P.; Argüelles, S.; Ayala, A. Oxidative Stress Is Increased in Critically Ill Patients According to Antioxidant Vitamins Intake, Independent of Severity: A Cohort Study. Crit. Care 2006, 10, R146. [Google Scholar] [CrossRef] [PubMed]
- Gamarra, Y.; Santiago, F.C.; Molina-López, J.; Castaño, J.; Herrera-Quintana, L.; Domínguez, Á.; Planells, E. Pyroglutamic Acidosis by Glutathione Regeneration Blockage in Critical Patients with Septic Shock. Crit. Care 2019, 23, 162. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.; Li, Y.; Ge, Y.; Shi, Y.; Lv, P.; Zhang, J.; Fu, G.; Zhou, Y.; Jiang, K.; Lin, N.; et al. Evaluation of Nutrition Risk and Its Association with Mortality Risk in Severely and Critically Ill COVID-19 Patients. J. Parenter. Enter. Nutr. 2020, 45, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Laviano, A.; Koverech, A.; Zanetti, M. Nutrition Support in the Time of SARS-CoV-2 (COVID-19). Nutrition 2020, 74, 110834. [Google Scholar] [CrossRef] [PubMed]
- Barazzoni, R.; Bischoff, S.C.; Breda, J.; Wickramasinghe, K.; Krznaric, Z.; Nitzan, D.; Pirlich, M.; Singer, P. ESPEN Expert Statements and Practical Guidance for Nutritional Management of Individuals with SARS-CoV-2 Infection. Clin. Nutr. 2020, 39, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Zabetakis, I.; Lordan, R.; Norton, C.; Tsoupras, A. COVID-19: The Inflammation Link and the Role of Nutrition in Potential Mitigation. Nutrients 2020, 12, 1466. [Google Scholar] [CrossRef]
- Caccialanza, R.; Laviano, A.; Lobascio, F.; Montagna, E.; Bruno, R.; Ludovisi, S.; Corsico, A.G.; Di Sabatino, A.; Belliato, M.; Calvi, M.; et al. Early Nutritional Supplementation in Non-Critically Ill Patients Hospitalized for the 2019 Novel Coronavirus Disease (COVID-19): Rationale and Feasibility of a Shared Pragmatic Protocol. Nutrition 2020, 74, 110835. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.J.; Martindale, R.G.; McClave, S.A. Relevant Nutrition Therapy in COVID-19 and the Constraints on Its Delivery by a Unique Disease Process. Nutr. Clin. Pract. 2020, 35, 792–799. [Google Scholar] [CrossRef] [PubMed]
- Alexander, J.; Tinkov, A.; Strand, T.A.; Alehagen, U.; Skalny, A.; Aaseth, J. Early Nutritional Interventions with Zinc, Selenium and Vitamin D for Raising Anti-Viral Resistance Against Progressive COVID-19. Nutrients 2020, 12, 2358. [Google Scholar] [CrossRef] [PubMed]
- Van den Ouweland, J.M.W. Analysis of Vitamin D Metabolites by Liquid Chromatography-Tandem Mass Spectrometry. TrAC Trends Anal. Chem. 2016, 84, 117–130. [Google Scholar] [CrossRef]
- Higashi, T.; Shimada, K.; Toyo’oka, T. Advances in Determination of Vitamin D Related Compounds in Biological Samples Using Liquid Chromatography–Mass Spectrometry: A Review. J. Chromatogr. B 2010, 878, 1654–1661. [Google Scholar] [CrossRef]
- Gottschlich, M.M.; Mayes, T.; Khoury, J.; Kagan, R.J. Clinical Trial of Vitamin D 2 vs. D 3 Supplementation in Critically Ill Pediatric Burn Patients. J. Parenter. Enter. Nutr. 2017, 41, 412–421. [Google Scholar] [CrossRef] [PubMed]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence That Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed]
- Martín Giménez, V.M.; Inserra, F.; Tajer, C.D.; Mariani, J.; Ferder, L.; Reiter, R.J.; Manucha, W. Lungs as Target of COVID-19 Infection: Protective Common Molecular Mechanisms of Vitamin D and Melatonin as a New Potential Synergistic Treatment. Life Sci. 2020, 254, 117808. [Google Scholar] [CrossRef] [PubMed]
- Mansur, J.L.; Tajer, C.; Mariani, J.; Inserra, F.; Ferder, L.; Manucha, W. Vitamin D high doses supplementation could represent a promising alternative to prevent or treat COVID-19 infection. Clín. Investig. Arterioscler. 2020, 32, 267–277. [Google Scholar] [CrossRef] [PubMed]
- Weir, E.K.; Thenappan, T.; Bhargava, M.; Chen, Y. Does Vitamin D Deficiency Increase the Severity of COVID-19? Clin. Med. 2020, 20, e107–e108. [Google Scholar] [CrossRef] [PubMed]
- Waldron, J.L.; Ashby, H.L.; Cornes, M.P.; Bechervaise, J.; Razavi, C.; Thomas, O.L.; Chugh, S.; Deshpande, S.; Ford, C.; Gama, R. Vitamin D: A Negative Acute Phase Reactant. J. Clin. Pathol. 2013, 66, 620–622. [Google Scholar] [CrossRef] [PubMed]
- Heijboer, A.C.; Blankenstein, M.A.; Kema, I.P.; Buijs, M.M. Accuracy of 6 Routine 25-Hydroxyvitamin D Assays: Influence of Vitamin D Binding Protein Concentration. Clin. Chem. 2012, 58, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Teymoori-Rad, M.; Marashi, S.M. Vitamin D and Covid-19: From Potential Therapeutic Effects to Unanswered Questions. Rev. Med. Virol. 2020, 31, e2159. [Google Scholar] [CrossRef] [PubMed]
- Bilezikian, J.P.; Bikle, D.; Hewison, M.; Lazaretti-Castro, M.; Formenti, A.M.; Gupta, A.; Madhavan, M.V.; Nair, N.; Babalyan, V.; Hutchings, N.; et al. Mechanisms in endocrinology: Vitamin D and COVID-19. Eur. J. Endocrinol. 2020, 183, R133–R147. [Google Scholar] [CrossRef] [PubMed]
- Xu, Y.; Baylink, D.J.; Chen, C.-S.; Reeves, M.E.; Xiao, J.; Lacy, C.; Lau, E.; Cao, H. The Importance of Vitamin d Metabolism as a Potential Prophylactic, Immunoregulatory and Neuroprotective Treatment for COVID-19. J. Transl. Med. 2020, 18, 322. [Google Scholar] [CrossRef] [PubMed]
- Carpagnano, G.E.; Di Lecce, V.; Quaranta, V.N.; Zito, A.; Buonamico, E.; Capozza, E.; Palumbo, A.; Di Gioia, G.; Valerio, V.N.; Resta, O. Vitamin D Deficiency as a Predictor of Poor Prognosis in Patients with Acute Respiratory Failure Due to COVID-19. J. Endocrinol. Investig. 2020, 44, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Meltzer, D.O.; Best, T.J.; Zhang, H.; Vokes, T.; Arora, V.; Solway, J. Association of Vitamin D Status and Other Clinical Characteristics With COVID-19 Test Results. JAMA Netw. Open 2020, 3, e2019722. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.Y.; Yan, J.Y.; Qu, J.M. Interpretations of “Diagnosis and Treatment Protocol for Novel Coronavirus Pneumonia (Trial Version 7)”. Chin. Med. J. 2020, 133, 1347–1349. [Google Scholar] [CrossRef]
- Cohen, J. A Power Primer. Psychol. Bull. 1992, 112, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Rapp, J.; Lieberman-Cribbin, W.; Tuminello, S.; Taioli, E. Male Sex, Severe Obesity, Older Age, and Chronic Kidney Disease Are Associated With COVID-19 Severity and Mortality in New York City. Chest 2020, 159, 112–115. [Google Scholar] [CrossRef] [PubMed]
- Chiappetta, S.; Sharma, A.M.; Bottino, V.; Stier, C. COVID-19 and the Role of Chronic Inflammation in Patients with Obesity. Int. J. Obes. 2020, 44, 1790–1792. [Google Scholar] [CrossRef] [PubMed]
- Ponti, G.; Maccaferri, M.; Ruini, C.; Tomasi, A.; Ozben, T. Biomarkers associated with COVID-19 disease progression. Crit. Rev. Clin. Lab. Sci. 2020, 57, 389–399. [Google Scholar] [CrossRef] [PubMed]
- Christopher, K.B. Vitamin D and Critical Illness Outcomes. Curr. Opin. Crit. Care 2016, 22, 332–338. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Zajic, P.; Schnedl, C.; Waltensdorfer, A.; Fruhwald, S.; Holl, A.; Purkart, T.; Wünsch, G.; Valentin, T.; Grisold, A.; et al. Vitamin D Status and Its Association with Season, Hospital and Sepsis Mortality in Critical Illness. Crit. Care 2014, 18, R47. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. The Vitamin D Deficiency Pandemic: Approaches for Diagnosis, Treatment and Prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef]
- Kaufman, H.W.; Niles, J.K.; Kroll, M.H.; Bi, C.; Holick, M.F. SARS-CoV-2 Positivity Rates Associated with Circulating 25-Hydroxyvitamin D Levels. PLoS ONE 2020, 15, e0239252. [Google Scholar] [CrossRef] [PubMed]
- Benskin, L.L. A Basic Review of the Preliminary Evidence That COVID-19 Risk and Severity Is Increased in Vitamin D Deficiency. Front. Public Health 2020, 8, 513. [Google Scholar] [CrossRef] [PubMed]
- Ilie, P.C.; Stefanescu, S.; Smith, L. The Role of Vitamin D in the Prevention of Coronavirus Disease 2019 Infection and Mortality. Aging Clin. Exp. Res. 2020, 32, 1195–1198. [Google Scholar] [CrossRef] [PubMed]
- Daneshkhah, A.; Agrawal, V.; Eshein, A.; Subramanian, H.; Roy, H.K.; Backman, V. Evidence for possible association of vitamin D status with cytokine storm and unregulated inflammation in COVID-19 patients. Aging Clin. Exp. Res. 2020, 32, 2141–2158. [Google Scholar] [CrossRef] [PubMed]
- Lips, P.; Cashman, K.D.; Lamberg-Allardt, C.; Bischoff-Ferrari, H.A.; Obermayer-Pietsch, B.; Bianchi, M.L.; Stepan, J.; Fuleihan, G.E.; Bouillon, R. Current vitamin D status in European and Middle East coun-tries and strategies to prevent vitamin D deficiency: A position statement of the European Calcified Tissue Society. Eur. J. Endocrinol. 2019, 180, P23–P54. [Google Scholar] [CrossRef] [PubMed]
- Maghbooli, Z.; Sahraian, M.A.; Ebrahimi, M.; Pazoki, M.; Kafan, S.; Tabriz, H.M.; Hadadi, A.; Montazeri, M.; Nasiri, M.; Shirvani, A.; et al. Vitamin D Sufficiency, a Serum 25-Hydroxyvitamin D at Least 30 Ng/ML Reduced Risk for Adverse Clinical Outcomes in Patients with COVID-19 Infection. PLoS ONE 2020, 15, e0239799. [Google Scholar] [CrossRef] [PubMed]
- Cereda, E.; Bogliolo, L.; Klersy, C.; Lobascio, F.; Masi, S.; Crotti, S.; De Stefano, L.; Bruno, R.; Corsico, A.G.; Di Sabatino, A.; et al. Vitamin D 25OH Deficiency in COVID-19 Patients Admitted to a Tertiary Referral Hospital. Clin. Nutr. 2020, 40, 2469–2472. [Google Scholar] [CrossRef] [PubMed]
- Vassiliou, A.G.; Jahaj, E.; Pratikaki, M.; Orfanos, S.E.; Dimopoulou, I.; Kotanidou, A. Low 25-Hydroxyvitamin D Levels on Admission to the Intensive Care Unit May Predispose COVID-19 Pneumonia Patients to a Higher 28-Day Mortality Risk: A Pilot Study on a Greek ICU Cohort. Nutrients 2020, 12, 3773. [Google Scholar] [CrossRef]
- Vázquez-Lorente, H.; Herrera-Quintana, L.; Molina-López, J.; Gamarra-Morales, Y.; López-González, B.; Miralles-Adell, C.; Planells, E. Response of Vitamin D after Magnesium Intervention in a Postmenopausal Population from the Province of Granada, Spain. Nutrients 2020, 12, 2283. [Google Scholar] [CrossRef] [PubMed]
- Swanson, C.M.; Nielson, C.M.; Shrestha, S.; Lee, C.G.; Barrett-Connor, E.; Jans, I.; Cauley, J.A.; Boonen, S.; Bouillon, R.; Vanderschueren, D.; et al. Higher 25(OH)D2 Is Associated with Lower 25(OH)D3 and 1,25(OH)2D3. J. Clin. Endocrinol. Metab. 2014, 99, 2736–2744. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Liu, X.; Hou, J.; Wei, D.; Liu, P.; Fan, K.; Zhang, L.; Nie, L.; Li, X.; Huo, W.; et al. Serum Vitamin D Affected Type 2 Diabetes Though Altering Lipid Profile and Modified the Effects of Testosterone on Diabetes Status. Nutrients 2020, 13, 90. [Google Scholar] [CrossRef]
- Cannell, J.J.; Vieth, R.; Umhau, J.C.; Holick, M.F.; Grant, W.B.; Madronich, S.; Garland, C.F.; Giovannucci, E. Epidemic Influenza and Vitamin D. Epidemiol. Infect. 2006, 134, 1129–1140. [Google Scholar] [CrossRef] [PubMed]
- Buetti, N.; Ruckly, S.; de Montmollin, E.; Reignier, J.; Terzi, N.; Cohen, Y.; Shiami, S.; Dupuis, C.; Timsit, J.-F. COVID-19 Increased the Risk of ICU-Acquired Bloodstream Infections: A Case–Cohort Study from the Multicentric OUTCOMEREA Network. Intensive Care Med. 2021, 47, 180–187. [Google Scholar] [CrossRef]
- Infante, M.; Buoso, A.; Pieri, M.; Lupisella, S.; Nuccetelli, M.; Bernardini, S.; Fabbri, A.; Iannetta, M.; Andreoni, M.; Colizzi, V.; et al. Low Vitamin D Status at Admission as a Risk Factor for Poor Survival in Hospitalized Patients with COVID-19: An Italian Retrospective Study. J. Am. Coll. Nutr. 2021, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Houghton, L.A.; Vieth, R. The Case against Ergocalciferol (Vitamin D2) as a Vitamin Supplement. Am. J. Clin. Nutr. 2006, 84, 694–697. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. The D-Sparaging of Vitamin D2: How Physiologically and Pharmacologically Relevant Is It for the Clinician? J. Clin. Endocrinol. Metab. 2020, 105, e1913–e1915. [Google Scholar] [CrossRef] [PubMed]
- Bigman, G. Vitamin D Metabolites, D3 and D2, and Their Independent Associations with Depression Symptoms among Adults in the United States. Nutr. Neurosci. 2020, 1–9. [Google Scholar] [CrossRef]
- Feng, X.; Li, S.; Sun, Q.; Zhu, J.; Chen, B.; Xiong, M.; Cao, G. Immune-Inflammatory Parameters in COVID-19 Cases: A Systematic Review and Meta-Analysis. Front. Med. 2020, 7, 301. [Google Scholar] [CrossRef] [PubMed]
- Terpos, E.; Ntanasis-Stathopoulos, I.; Elalamy, I.; Kastritis, E.; Sergentanis, T.N.; Politou, M.; Psaltopoulou, T.; Gerotziafas, G.; Dimopoulos, M.A. Hematological Findings and Complications of COVID -19. Am. J. Hematol. 2020, 95, 834–847. [Google Scholar] [CrossRef] [PubMed]
- Parikh, S.; Guo, D.; Pollock, N.K.; Petty, K.; Bhagatwala, J.; Gutin, B.; Houk, C.; Zhu, H.; Dong, Y. Circulating 25-Hydroxyvitamin D Concentrations Are Correlated with Cardiometabolic Risk Among American Black and White Adolescents Living in a Year-Round Sunny Climate. Diabetes Care 2012, 35, 1133–1138. [Google Scholar] [CrossRef] [PubMed]
- Saghir Afifeh, A.M.; Verdoia, M.; Nardin, M.; Negro, F.; Viglione, F.; Rolla, R.; De Luca, G. Determinants of Vitamin D Activation in Patients with Acute Coronary Syndromes and Its Correlation with Inflammatory Markers. Nutr. Metab. Cardiovasc. Dis. 2020, 31, 36–43. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.M.; Reintam-Blaser, A.; Calder, P.C.; Casaer, M.; Hiesmayr, M.J.; Mayer, K.; Montejo, J.C.; Pichard, C.; Preiser, J.-C.; van Zanten, A.R.H.; et al. Monitoring Nutrition in the ICU. Clin. Nutr. 2019, 38, 584–593. [Google Scholar] [CrossRef] [PubMed]
- Vázquez-Lorente, H.; Herrera-Quintana, L.; Quintero-Osso, B.; Molina-López, J.; Planells, E. Current Trends in the Analytical Determination of Vitamin D. Nutr. Hosp. 2019, 36, 1418–1423. [Google Scholar] [PubMed]
- Tahsin-Swafiri, S.; Blanco-Navarro, I.; Pérez-Sacristán, B.; Millán, I.; Granado-Lorencio, F. The Prevalence of Vitamin Deficiency in Clinical Practice Is Assay-Dependent. Clin. Nutr. 2012, 31, 1011–1014. [Google Scholar] [CrossRef]
- Rousseau, A.-F.; Damas, P.; Janssens, M.; Kalin, S.; Ledoux, D.; Le Goff, C.; Gadisseur, R.; Delanaye, P.; Cavalier, E. Critical Care and Vitamin D Status Assessment: What about Immunoassays and Calculated Free 25OH-D? Clin. Chim. Acta 2014, 437, 43–47. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Hanotte, B.; Grandin de l’Eprevier, C.; Sabatier, J.-M.; Lafaie, L.; Célarier, T. Vitamin D and Survival in COVID-19 Patients: A Quasi-Experimental Study. J. Steroid Biochem. Mol. Biol. 2020, 204, 105771. [Google Scholar] [CrossRef] [PubMed]
- Entrenas Castillo, M.; Entrenas Costa, L.M.; Vaquero Barrios, J.M.; Alcalá Díaz, J.F.; Miranda, J.L.; Bouillon, R.; Quesada Gómez, J.M. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef] [PubMed]
- Ebadi, M.; Montano-Loza, A.J. Perspective: Improving Vitamin D Status in the Management of COVID-19. Eur. J. Clin. Nutr. 2020, 74, 856–859. [Google Scholar] [CrossRef] [PubMed]

| Baseline Characteristics (N = 37) | Mean ± SD | Min–Max | 95% CI |
|---|---|---|---|
| Age (years) | 60.0 ± 10.2 | 41.0–74.0 | 56.6–63.4 |
| Sex (M/F, %) | 26/11 (70.3/29.7) | - | - |
| BMI (kg/m2) | 30.77 ± 4.17 | 22.8–42.2 | 29.4–32.2 |
| BMI < 25 kg/m2 (n/N, %) | 3/37 (8.10) | - | - |
| BMI 25–30 kg/m2 (n/N, %) | 14/37 (37.8) | - | - |
| BMI > 30 kg/m2 (n/N, %) | 20/37 (54.1) | - | - |
| Smoking habit (n/N, %) | |||
| Smokers | 3/37 (8.10) | - | - |
| Ex-smokers | 12/37 (32.4) | - | - |
| Never smokers | 22/37 (59.5) | - | - |
| Patients with comorbidity (n/N, %) | 26/37 (70.3) | - | - |
| Diabetes | 13/37 (35.1) | - | - |
| Hypertension | 20/37 (54.1) | - | - |
| Dyslipidemia | 11/37 (29.7) | - | - |
| Chronic kidney disease | 2/37 (5.40) | - | - |
| COPD | 10/37 (27.0) | - | - |
| Cardiovascular disease | 6/37 (16.2) | - | - |
| APACHE-II score | 12.3 ± 3.77 | 6.00–21.0 | 11.1–13.6 |
| SOFA score | 6.54 ± 2.60 | 2.00–13.0 | 5.67–7.41 |
| Bacterial and fungal infection (n/N, %) | 26/37 (70.3) | - | - |
| Sepsis (n/N, %) | 7/36 (19.4) | - | - |
| PaO2/FiO2 | 212.9 ± 103.8 | 15.0–550.0 | 169.2–248.6 |
| ARDS (PaO2/FiO2 < 300) (n/N, %) | 26/37 (70.0) | - | - |
| Mild (300 < PaO2/FiO2 ≤ 200) (n/N, %) | 12/37 (32.4) | - | - |
| Moderate (200 < PaO2/FiO2 ≤ 100) (n/N, %) | 10/37 (27.0) | - | - |
| Severe (PaO2/FiO2 < 100) (n/N, %) | 4/37 (10.8) | - | - |
| IMV (n/N, %) | 30/37 (81.1) | - | - |
| Duration of IMV (days) | 21.7 ± 14.6 | 1.00–73.0 | 16.0–27.4 |
| ICU length of stay (days) | 25.4 ± 22.6 | 6.00–104.0 | 17.8–32.9 |
| Length of hospitalization (days) | 39.5 ± 27.0 | 9.00–131.0 | 30.5–48.5 |
| Patient 28-day mortality (n/N, %) | 26/37 (70.3) | - | - |
| Reference | Baseline Median (IQR) N = 37 | Follow-Up Median (IQR) N = 37 | Z | p-Value Initial–Final | ES | |
|---|---|---|---|---|---|---|
| Clinical | ||||||
| Heart rate (bpm) | 60–100 | 80.0 (28.7) | 64.0 (38.0) | −1.69 | 0.091 | 0.411 |
| Respiratory rate (brpm) | 15–20 | 30.0 (3.50) | 22.0 (4.50) | −1.63 | 0.102 | 0.582 |
| Mean blood pressure (mmHg) | 70–105 | 93.5 (18.0) | 91.5 (25.7) | −0.31 | 0.753 | 0.095 |
| PEEP (cm H2O) | 2–5 | 14.0 (3.50) | 12.0 (2.00) | −2.76 | 0.006 | 0.779 |
| FiO2 | >68% | 0.70 (0.25) | 0.60 (0.15) | −3.81 | 0.001 | 0.825 |
| PaO2/FiO2 | 200–300 | 200.0 (101.5) | 222.0 (119.0) | −0.05 | 0.964 | 0.010 |
| Biochemical | ||||||
| Total Proteins (g/dL) | 6.60–8.30 | 6.40 (0.90) | 6.10 (1.13) | −2.51 | 0.012 | 0.513 |
| Albumin (g/dL) | 3.50–5.20 | 3.20 (0.65) | 3.00 (0.60) | −2.11 | 0.035 | 0.444 |
| Prealbumin (mg/dL) | 16.0–42.0 | 9.00 (16.2) | 25.0 (23.0) | −2.39 | 0.017 | 0.782 |
| Ferritin (ng/mL) | 20.0–275.0 | 1139.3 (1772.9) | 1490.1 (1815.7) | −0.52 | 0.603 | 0.117 |
| Transferrin (mg/dL) | 200.0–360.0 | 132.0 (31.7) | 136.0 (68.0) | −0.82 | 0.410 | 0.269 |
| Glucose (mg/dL) | 75.0–115.0 | 154.0 (81.0) | 184.5 (113.5) | −1.62 | 0.106 | 0.328 |
| Total Cholesterol (mg/dL) | 140.0–200.0 | 138.5 (51.5) | 159.0 (103.0) | −2.02 | 0.044 | 0.574 |
| GOT or AST (U/L) | 5.00–40.0 | 37.0 (32.5) | 31.0 (32.0) | −1.76 | 0.078 | 0.351 |
| GPT or ALT (U/L) | 0.00–55.0 | 35.0 (40.0) | 36.5 (46.5) | −1.21 | 0.228 | 0.248 |
| LDH (U/L) | 0.00–248.0 | 490.5 (183.0) | 429.0 (138.0) | −3.05 | 0.002 | 0.590 |
| CRP (mg/L) | 0.00–5.00 | 153.7 (210.7) | 35.4 (56.4) | −4.66 | 0.001 | 0.991 |
| PCT (ng/dL) | 0.02–0.50 | 0.22 (0.44) | 0.11 (0.46) | −1.59 | 0.112 | 0.360 |
| Hemoglobin (g/dL) | 11.0–17.0 | 13.3 (2.80) | 12.6 (3.93) | −4.06 | 0.001 | 0.789 |
| Leukocytes (*103/μL) | 3.50–10.5 | 9.67 (6.94) | 9.45 (7.44) | −1.25 | 0.212 | 0.240 |
| Neutrophils (%) | 42.0–77.0 | 88.5 (8.15) | 88.0 (6.82) | −0.34 | 0.737 | 0.067 |
| Lymphocytes (%) | 20.0–44.0 | 6.40 (5.68) | 5.75 (4.13) | −0.28 | 0.777 | 0.056 |
| Platelets (*103/µL) | 120.0–450.0 | 212.0 (135.5) | 266.0 (131.5) | −2.52 | 0.012 | 0.482 |
| D-dimer (ng/dL) | 0.00–500.0 | 1080.0 (1647.5) | 1520.0 (3050.0) | −1.14 | 0.254 | 0.231 |
| Fibrinogen (mg/dL) | 200.0–350.0 | 750.5 (356.5) | 556.0 (336.7) | −3.52 | 0.001 | 0.683 |
| Ca (mg/dL) | 8.80–10.6 | 8.40 (0.48) | 8.10 (0.98) | −0.81 | 0.421 | 0.190 |
| P (mg/dL) | 2.30–4.50 | 3.55 (1.93) | 3.15 (1.43) | −0.02 | 0.984 | 0.005 |
| Mg (mg/dL) | 1.60–2.60 | 2.23 (0.37) | 2.20 (0.50) | −0.80 | 0.421 | 0.253 |
| 25–OH–D (ng/mL) | 20.0–100.0 | 13.6 (9.02) | 12.2 (6.01) | −2.53 | 0.011 | 0.600 |
| 25–OH–D3 (ng/mL) | - | 8.45 (6.38) | 7.92 (5.85) | −1.35 | 0.176 | 0.520 |
| 25–OH–D2 (ng/mL) | - | 5.85 (2.95) | 4.66 (2.02) | −2.74 | 0.006 | 0.278 |
| Baseline | Follow-Up | |||||
|---|---|---|---|---|---|---|
| 25-OH-D (ng/mL) | 25-OH-D3 (ng/mL) | 25-OH-D2 (ng/mL) | 25-OH-D (ng/mL) | 25-OH-D3 (ng/mL) | 25-OH-D2 (ng/mL) | |
| Age (years) | −0.129 | −0.132 | −0.035 | −0.089 | −0.132 | 0.065 |
| BMI (kg/m2) | 0.111 | 0.165 | −0.026 | 0.091 | 0.261 | −0.231 |
| APACHE-II | −0.103 | −0.066 | −0.040 | - | - | - |
| SOFA | −0.154 | −0.053 | −0.167 | −0.133 | 0.008 | −0.290 |
| Respiratory rate (brpm) | −0.176 | 0.053 | −0.456 a | −0.277 | 0.047 | −0.374 |
| Albumin (g/dL) | 0.403 a | 0.285 | 0.390 a | 0.015 | −0.097 | 0.234 |
| PCT (ng/dL) | −0.270 | −0.294 | −0.026 | −0.587 b | −0.458 a | −0.331 |
| Hemoglobin (g/dL) | 0.387 a | 0.307 | 0.261 | 0.301 | 0.223 | 0.224 |
| Lymphocytes (%) | −0.046 | −0.059 | 0.084 | 0.364 a | 0.371 a | 0.034 |
| D-dimer (ng/dL) | −0.521 b | −0.644 b | 0.148 | −0.264 | −0.405 a | 0.302 |
| Fibrinogen (mg/dL) | 0.350 a | 0.370 a | 0.128 | 0.116 | 0.335 | −0.521 b |
| Ca (mg/dL) | 0.285 | 0.180 | 0.306 | −0.333 | −0.527 b | 0.426 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Herrera-Quintana, L.; Gamarra-Morales, Y.; Vázquez-Lorente, H.; Molina-López, J.; Castaño-Pérez, J.; Machado-Casas, J.F.; Coca-Zúñiga, R.; Pérez-Villares, J.M.; Planells, E. Bad Prognosis in Critical Ill Patients with COVID-19 during Short-Term ICU Stay regarding Vitamin D Levels. Nutrients 2021, 13, 1988. https://doi.org/10.3390/nu13061988
Herrera-Quintana L, Gamarra-Morales Y, Vázquez-Lorente H, Molina-López J, Castaño-Pérez J, Machado-Casas JF, Coca-Zúñiga R, Pérez-Villares JM, Planells E. Bad Prognosis in Critical Ill Patients with COVID-19 during Short-Term ICU Stay regarding Vitamin D Levels. Nutrients. 2021; 13(6):1988. https://doi.org/10.3390/nu13061988
Chicago/Turabian StyleHerrera-Quintana, Lourdes, Yenifer Gamarra-Morales, Héctor Vázquez-Lorente, Jorge Molina-López, José Castaño-Pérez, Juan Francisco Machado-Casas, Ramón Coca-Zúñiga, José Miguel Pérez-Villares, and Elena Planells. 2021. "Bad Prognosis in Critical Ill Patients with COVID-19 during Short-Term ICU Stay regarding Vitamin D Levels" Nutrients 13, no. 6: 1988. https://doi.org/10.3390/nu13061988
APA StyleHerrera-Quintana, L., Gamarra-Morales, Y., Vázquez-Lorente, H., Molina-López, J., Castaño-Pérez, J., Machado-Casas, J. F., Coca-Zúñiga, R., Pérez-Villares, J. M., & Planells, E. (2021). Bad Prognosis in Critical Ill Patients with COVID-19 during Short-Term ICU Stay regarding Vitamin D Levels. Nutrients, 13(6), 1988. https://doi.org/10.3390/nu13061988









