Potential Protective Protein Components of Cow’s Milk against Certain Tumor Entities
Abstract
1. Introduction
2. Composition of Cow’s Milk
3. Milk Proteins and Processed Peptides with Chemopreventive Properties
3.1. Casein Proteins and Processed Peptides
3.1.1. Caseins and Casomorphines
3.1.2. Casein Phosphopeptides
3.2. Whey Proteins and Processed Peptides
3.2.1. Lactoferrin
3.2.2. Lactoferricin
3.2.3. α-Lactalbumin and Bovine α-Lactalbumin Made Lethal to Tumor Cells (BAMLET)
3.2.4. β-Lactoglobulin
3.3. Milk Fat Globule Membrane
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| BAMLET | bovine α-lactalbumin made lethal to tumor cells |
| BCM | βb-casomorphin |
| bLf | bovine lactoferrin |
| BSA | bovine serum albumin |
| BTN | butyrophilin |
| CLA | conjugated linoleic acid |
| CLD | cytoplasmatic lipid droplet |
| CN | casein |
| CPP | caseinphosphopeptide |
| DMBA | 7,12 dimethylbenz[a]anthracene |
| DMH-DSS | 1,2-dimethylhydrazine/dextran sulphate sodium |
| FAO | Food and Agriculture Organization of the United Nations |
| FAS | Foreign Agricultural Service |
| HAMLET | human α-lactalbumin made lethal to tumor cells |
| HBP | hamster buccal pouch |
| hLf | human lactoferrin |
| LAK | lymphokine-activated killer |
| Lf | lactoferrin |
| Lfcin | lactoferricin |
| LfcinB | bovine lactoferricin |
| LfR | Lf receptor |
| MFGM | milk fat globule membrane |
| MLD | microlipid droplet |
| MLN | mesenteric lymph nodes |
| MUC1 | mucin-1 |
| NK | natural killer |
| PAS6/7 | periodic acid Schiff 6/7 |
| NPC | nasopharyngeal carcinoma |
| PDK1 | 3-phosphoinositide-dependent protein kinase 1 |
| PEG | polyethylene glycol |
| PMNL | polymorphonuclear leukocyte |
| PP | Peyer´s patches |
| USDA | United States Department of Agriculture |
| WHO | World Health Organization |
| XO | xanthine oxidoreductase |
| α-LA | α-lactalbumin |
| β-LG | β-lactoglobulin |
References
- Itan, Y.; Powell, A.; Beaumont, M.A.; Burger, J.; Thomas, M.G. The Origins of Lactase Persistence in Europe. PLoS Comput. Biol. 2009, 5, e1000491. [Google Scholar] [CrossRef]
- United States Department of Agriculture (USDA); Foreign Agricultural Service (FAS). Dairy: World Markets and Trade. Release Date: December 2014. p. 14. Available online: https://downloads.usda.library.cornell.edu/usda-esmis/files/5t34sj56t/f4752h17r/05741s112/dairy-market-12-16-2014.pdf (accessed on 14 December 2020).
- United States Department of Agriculture (USDA); Foreign Agricultural Service (FAS). Dairy: World Markets and Trade. Release Date: December 2020. p. 9. Available online: https://apps.fas.usda.gov/psdonline/circulars/dairy.pdf (accessed on 14 December 2020).
- Bundesanstalt für Landwirtschaft und Ernährung (BLE); Bundesinformationszentrum Landwirtschaft. Bericht zur Markt- und Versorgungslage mit Milch und Milcherzeugnissen. Release Date: 8 May 2020. pp. 14, 64. Available online: https://www.ble.de/SharedDocs/Downloads/DE/BZL/Daten-Berichte/MilchUndMilcherzeugnisse/JaehrlicheErgebnisse/Deutschland/2020BerichtMilch.pdf?__blob=publicationFile&v=2 (accessed on 14 December 2020).
- Food and Agriculture Organization of the United Nation. Agricultural Data. Available online: http://www.fao.org/faostat/en/#data/QC (accessed on 24 February 2021).
- Food and Agriculture Organization of the United Nations. Food Outlook. Biannual Report on Global Food Markets; Food and Agriculture Organization of the United Nations: Rome, Italy, 2020; p. 8. ISBN 925132848X. [Google Scholar]
- Oberritter, H.; Schäbethal, K.; von Ruesten, A.; Boeing, H. The DGE Nutrition Circle—Presentation and Basis of the Food-Related Recommendations from the German Nutrition Society (DGE). Ernaehrungs Umsch. Int. 2013, 24–29. [Google Scholar] [CrossRef]
- Committee, D.G.A. Dietary Guidelines for Americans 2015–2020; U.S. Deptartment of Health and Human Services: Washington, DC, USA, 2016; ISBN 9780160934650.
- World Health Organization. WHO Report on Cancer: Setting Priorities, Investing Wisely and Providing Care for All; World Health Organization: Geneva, Switzerland, 2020; ISBN 9789240001305. [Google Scholar]
- Burkard, M.; Leischner, C.; Lauer, U.M.; Busch, C.; Venturelli, S.; Frank, J. Dietary flavonoids and modulation of natural killer cells: Implications in malignant and viral diseases. J. Nutr. Biochem. 2017, 46, 1–12. [Google Scholar] [CrossRef]
- Venturelli, S.; Burkard, M.; Biendl, M.; Lauer, U.M.; Frank, J.; Busch, C. Prenylated chalcones and flavonoids for the prevention and treatment of cancer. Nutrition 2016, 32, 1171–1178. [Google Scholar] [CrossRef]
- Leischner, C.; Burkard, M.; Pfeiffer, M.M.; Lauer, U.M.; Busch, C.; Venturelli, S. Nutritional immunology: Function of natural killer cells and their modulation by resveratrol for cancer prevention and treatment. Nutr. J. 2015, 15, 1–12. [Google Scholar] [CrossRef]
- Busch, C.; Burkard, M.; Leischner, C.; Lauer, U.M.; Frank, J.; Venturelli, S. Epigenetic activities of flavonoids in the prevention and treatment of cancer. Clin. Epigenetics 2015, 7, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Barrubés, L.; Babio, N.; Becerra-Tomás, N.; Rosique-Esteban, N.; Salas-Salvadó, J. Association Between Dairy Product Consumption and Colorectal Cancer Risk in Adults: A Systematic Review and Meta-Analysis of Epidemiologic Studies. Adv. Nutr. 2019, 10, S190–S211. [Google Scholar] [CrossRef] [PubMed]
- Savaiano, D.A.; Hutkins, R.W. Yogurt, cultured fermented milk, and health: A systematic review. Nutr. Rev. 2021, 79, 599–614. [Google Scholar] [CrossRef]
- Bermejo, L.M.; López-Plaza, B.; Santurino, C.; Cavero-Redondo, I.; Gómez-Candela, C. Milk and Dairy Product Consumption and Bladder Cancer Risk: A Systematic Review and Meta-Analysis of Observational Studies. Adv. Nutr. 2019, 10, S224–S238. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Preterre, A.L.; Iqbal, K.; Bechthold, A.; De Henauw, S.; Michels, N.; Devleesschauwer, B.; et al. Food groups and risk of colorectal cancer. Int. J. Cancer 2018, 142, 1748–1758. [Google Scholar] [CrossRef] [PubMed]
- Eskin, N.A.M.; Goff, H.D. Chapter 4—Milk. In Biochemistry of Foods, 3rd ed.; Eskin, N.M., Shahidi, F., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 187–214. ISBN 978-0-12-242352-9. [Google Scholar]
- Farrell, H.; Jimenez-Flores, R.; Bleck, G.; Brown, E.; Butler, J.; Creamer, L.; Hicks, C.; Hollar, C.; Ng-Kwai-Hang, K.; Swaisgood, H. Nomenclature of the Proteins of Cows’ Milk—Sixth Revision. J. Dairy Sci. 2004, 87, 1641–1674. [Google Scholar] [CrossRef]
- Park, Y.W.; Nam, M.S. Bioactive Peptides in Milk and Dairy Products: A Review. Food Sci. Anim. Resour. 2015, 35, 831–840. [Google Scholar] [CrossRef] [PubMed]
- Eigel, W.; Butler, J.; Ernstrom, C.; Farrell, H.; Harwalkar, V.; Jenness, R.; Whitney, R.M. Nomenclature of Proteins of Cow’s Milk: Fifth Revision. J. Dairy Sci. 1984, 67, 1599–1631. [Google Scholar] [CrossRef]
- Fuquay, J.W.; Roginski, H.; Fox, P.F. Encyclopedia of Dairy Sciences; Academic Press: Amsterdam, The Netherlands, 2003; ISBN1 0122272358. ISBN2 9780122272356. [Google Scholar]
- Fiocchi, A.; Brozek, J.; Schünemann, H.; Bahna, S.L.; von Berg, A.; Beyer, K.; Bozzola, M.; Bradsher, J.; Compalati, E.; Ebisawa, M.; et al. World Allergy Organization (WAO) Diagnosis and Rationale for Action against Cow’s Milk Allergy (DRACMA) Guidelines. World Allergy Organ. J. 2010, 3, 57–161. [Google Scholar] [CrossRef] [PubMed]
- Bonuccelli, G.; Castello-Cros, R.; Capozza, F.; Martinez-Outschoorn, U.E.; Lin, Z.; Tsirigos, A.; Xuanmao, J.; Whitaker-Menezes, D.; Howell, A.; Lisanti, M.P.; et al. The milk protein α-casein functions as a tumor suppressor via activation of STAT1 signaling, effectively preventing breast cancer tumor growth and metastasis. Cell Cycle 2012, 11, 3972–3982. [Google Scholar] [CrossRef]
- Nekipelaya, V.V.; Semenov, D.V.; Potapenko, M.O.; Kuligina, E.V.; Kit, Y.U.Y.; Romanova, I.V.; Richter, V.A. Lactaptin is a human milk protein inducing apoptosis of MCF-7 adenocarcinoma cells. Dokl. Biochem. Biophys. 2008, 419, 58–61. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Gu, F.; Wei, C.; Tang, Y.; Zheng, X.; Ren, M.; Qin, Y. PGPIPN, a Therapeutic Hexapeptide, Suppressed Human Ovarian Cancer Growth by Targeting BCL2. PLoS ONE 2013, 8, e60701. [Google Scholar] [CrossRef]
- Azevedo, R.A.; Ferreira, A.K.; Auada, A.V.V.; Pasqualoto, K.F.M.; Marques-Porto, R.; Maria, D.A.; Lebrun, I. Antitumor Effect of Cationic INKKI Peptide from Bovine β-Casein on Melanoma B16F10. J. Cancer Ther. 2012, 3, 237–244. [Google Scholar] [CrossRef]
- Kampa, M.; Bakogeorgou, E.; Hatzoglou, A.; Damianaki, A.; Martin, P.-M.; Castanas, E. Opioid alkaloids and casomorphin peptides decrease the proliferation of prostatic cancer cell lines (LNCaP, PC3 and DU145) through a partial interaction with opioid receptors. Eur. J. Pharmacol. 1997, 335, 255–265. [Google Scholar] [CrossRef]
- Hatzoglou, A.; Bakogeorgou, E.; Hatzoglou, C.; Martin, P.-M.; Castanas, E. Antiproliferative and receptor binding properties of α- and β-casomorphins in the T47D human breast cancer cell line. Eur. J. Pharmacol. 1996, 310, 217–223. [Google Scholar] [CrossRef]
- Hata, I.; Higashiyama, S.; Otani, H. Identification of a phosphopeptide in bovine αs1-casein digest as a factor influencing proliferation and immunoglobulin production in lymphocyte cultures. J. Dairy Res. 1998, 65, 569–578. [Google Scholar] [CrossRef]
- Perego, S.; Cosentino, S.; Fiorilli, A.; Tettamanti, G.; Ferraretto, A. Casein phosphopeptides modulate proliferation and apoptosis in HT-29 cell line through their interaction with voltage-operated L-type calcium channels. J. Nutr. Biochem. 2012, 23, 808–816. [Google Scholar] [CrossRef]
- Iigo, M.; Kuhara, T.; Ushida, Y.; Sekine, K.; Moore, M.A.; Tsuda, H. Inhibitory effects of bovine lactoferrin on colon carcinoma 26 lung metastasis in mice. Clin. Exp. Metastasis 1999, 17, 43–49. [Google Scholar] [CrossRef]
- Yoo, Y.-C.; Watanabe, S.; Watanabe, R.; Hata, K.; Shimazaki, K.-I.; Azuma, I. Bovine Lactoferrin and Lactoferricin, a Peptide Derived from Bovine Lactoferrin, Inhibit Tumor Metastasis in Mice. Jpn. J. Cancer Res. 1997, 88, 184–190. [Google Scholar] [CrossRef]
- Sekine, K.; Ushida, Y.; Kuhara, T.; Iigo, M.; Baba-Toriyama, H.; Moore, M.; Murakoshi, M.; Satomi, Y.; Nishino, H.; Kakizoe, T.; et al. Inhibition of initiation and early stage development of aberrant crypt foci and enhanced natural killer activity in male rats administered bovine lactoferrin concomitantly with azoxymethane. Cancer Lett. 1997, 121, 211–216. [Google Scholar] [CrossRef]
- Roy, M.; Kuwabara, Y.; Hara, K.; Watanabe, Y.; Tamai, Y. Peptides From the N-terminal End of Bovine Lactoferrin Induce Apoptosis in Human Leukemic (HL-60) Cells. J. Dairy Sci. 2002, 85, 2065–2074. [Google Scholar] [CrossRef]
- Sakai, T.; Banno, Y.; Kato, Y.; Nozawa, Y.; Kawaguchi, M. Pepsin-Digested Bovine Lactoferrin Induces Apoptotic Cell Death With JNK/SAPK Activation in Oral Cancer Cells. J. Pharmacol. Sci. 2005, 98, 41–48. [Google Scholar] [CrossRef]
- Mohan, K.V.P.C.; Kumaraguruparan, R.; Prathiba, D.; Nagini, S. Modulation of xenobiotic-metabolizing enzymes and redox status during chemoprevention of hamster buccal carcinogenesis by bovine lactoferrin. Nutrition 2006, 22, 940–946. [Google Scholar] [CrossRef] [PubMed]
- Cutone, A.; Colella, B.; Pagliaro, A.; Rosa, L.; Lepanto, M.S.; di Patti, M.C.B.; Valenti, P.; Di Bartolomeo, S.; Musci, G. Native and iron-saturated bovine lactoferrin differently hinder migration in a model of human glioblastoma by reverting epithelial-to-mesenchymal transition-like process and inhibiting interleukin-6/STAT3 axis. Cell. Signal. 2020, 65, 109461. [Google Scholar] [CrossRef]
- Sugihara, Y.; Zuo, X.; Takata, T.; Jin, S.; Miyauti, M.; Isikado, A.; Imanaka, H.; Tatsuka, M.; Qi, G.; Shimamoto, F. Inhibition of DMH-DSS-induced colorectal cancer by liposomal bovine lactoferrin in rats. Oncol. Lett. 2017, 14, 5688–5694. [Google Scholar] [CrossRef] [PubMed]
- Eliassen, L.T.; Berge, G.; Sveinbjørnsson, B.; Svendsen, J.S.; Vorland, L.H.; Rekdal, Ø. Evidence for a direct antitumor mecha-nism of action of bovine lactoferricin. Anticancer. Res. 2002, 22, 2703–2710. [Google Scholar]
- Rammer, P.; Groth-Pedersen, L.; Kirkegaard, T.; Daugaard, M.; Rytter, A.; Szyniarowski, P.; Høyer-Hansen, M.; Povlsen, L.K.; Nylandsted, J.; Larsen, J.E.; et al. BAMLET Activates a Lysosomal Cell Death Program in Cancer Cells. Mol. Cancer Ther. 2010, 9, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Li, P.; Yang, H.; Wang, Y.; Huang, G.; Wang, J.; Zheng, N. Investigation and comparison of the anti-tumor activities of lactoferrin, α-lactalbumin, and β-lactoglobulin in A549, HT29, HepG2, and MDA231-LM2 tumor models. J. Dairy Sci. 2019, 102, 9586–9597. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.T.; Law, A.J.R. The content and composition of protein in creamery milks in south-west Scotland. J. Dairy Res. 1980, 47, 83–90. [Google Scholar] [CrossRef]
- Miller, M.J.S.; Witherly, S.A.; Clark, D.A. Casein: A Milk Protein with Diverse Biologic Consequences. Exp. Biol. Med. 1990, 195, 143–159. [Google Scholar] [CrossRef] [PubMed]
- Korhonen, H.; Pihlanto-Leppäla, A.; Rantamäki, P.; Tupasela, T. Impact of processing on bioactive proteins and peptides. Trends Food Sci. Technol. 1998, 9, 307–319. [Google Scholar] [CrossRef]
- Gobbetti, M.; Stepaniak, L.; De Angelis, M.; Corsetti, A.; Di Cagno, R. Latent Bioactive Peptides in Milk Proteins: Proteolytic Activation and Significance in Dairy Processing. Crit. Rev. Food Sci. Nutr. 2002, 42, 223–239. [Google Scholar] [CrossRef]
- Kamiński, S.; Cieślińska, A.; Kostyra, E. Polymorphism of bovine beta-casein and its potential effect on human health. J. Appl. Genet. 2007, 48, 189–198. [Google Scholar] [CrossRef]
- Henschen, A.; Lottspeich, F.; Brantl, V.; Teschemacher, H. Novel opioid peptides derived from casein (beta-casomorphins). II. Structure of active components from bovine casein peptone. Hoppe-Seyler´s Zeitschrift für physiologische Chemie 1979, 360, 1217–1224. [Google Scholar]
- Liu, Z.; Udenigwe, C.C. Role of food-derived opioid peptides in the central nervous and gastrointestinal systems. J. Food Biochem. 2018, 43, e12629. [Google Scholar] [CrossRef]
- Noni, I.; FitzGerald, R.; Korhonen, H.; Livesey, C.; Thorsdottir, I.; Tomé, D.; Witkamp, R. Review of the potential health impact of β-casomorphins and related peptides. EFSA Sci. Rep. 2009, 7, 231. [Google Scholar] [CrossRef]
- Park, S.-W.; Kim, J.-Y.; Kim, Y.-S.; Lee, S.J.; Chung, M.K.; Lee, S.D. A Milk Protein, Casein, as a Proliferation Promoting Factor in Prostate Cancer Cells. World J. Men Health 2014, 32, 76–82. [Google Scholar] [CrossRef]
- Meisel, H.; Frister, H. Chemical Characterization of a Caseinophosphopeptide Isolated from in vivo Digests of a Casein Diet. Biol. Chem. Hoppe-Seyler 1988, 369, 1275–1280. [Google Scholar] [CrossRef]
- Berrocal, R.; Chanton, S.; Juillerat, M.A.; Favillare, B.; Scherz, J.-C.; Jost, R. Tryptic phosphopeptides from whole casein. II. Physicochemical properties related to the solubilization of calcium. J. Dairy Res. 1989, 56, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Kitts, D.D.; Nakamura, S. Calcium-enriched casein phosphopeptide stimulates release of IL-6 cytokine in human epithelial intestinal cell line. J. Dairy Res. 2005, 73, 44–48. [Google Scholar] [CrossRef] [PubMed]
- Otani, H.; Watanabe, T.; Tashiro, Y. Effects of Bovine β-Casein (1-28) and Its Chemically Synthesized Partial Fragments on Proliferative Responses and Immunoglobulin Production in Mouse Spleen Cell Cultures. Biosci. Biotechnol. Biochem. 2001, 65, 2489–2495. [Google Scholar] [CrossRef][Green Version]
- Cosentino, S.; Gravaghi, C.; Donetti, E.; Donida, B.M.; Lombardi, G.; Bedoni, M.; Fiorilli, A.; Tettamanti, G.; Ferraretto, A. Caseinphosphopeptide-induced calcium uptake in human intestinal cell lines HT-29 and Caco2 is correlated to cellular differentiation. J. Nutr. Biochem. 2010, 21, 247–254. [Google Scholar] [CrossRef]
- Metz-Boutigue, M.-H.; Jolles, J.; Mazurier, J.; Schoentgen, F.; Legrand, D.; Spik, G.; Montreuil, J.; Jolles, P. Human lactotransferrin: Amino acid sequence and structural comparisons with other transferrins. JBIC J. Biol. Inorg. Chem. 1984, 145, 659–676. [Google Scholar] [CrossRef]
- Pierce, A.; Colavizza, D.; Benaissa, M.; Maes, P.; Tartar, A.; Montreuil, J.; Spik, G. Molecular cloning and sequence analysis of bovine lactotransferrin. JBIC J. Biol. Inorg. Chem. 1991, 196, 177–184. [Google Scholar] [CrossRef] [PubMed]
- Anderson, B.F.; Baker, H.M.; Norris, G.E.; Rice, D.W.; Baker, E.N. Structure of human lactoferrin: Crystallographic structure analysis and refinement at 2·8 Å resolution. J. Mol. Biol. 1989, 209, 711–734. [Google Scholar] [CrossRef]
- Masson, P.L.; Heremans, J.F.; Schonne, E. Lactoferrin, an iron-binbing protein in neutrophilic leukocytes. J. Exp. Med. 1969, 130, 643–658. [Google Scholar] [CrossRef]
- Sørensen, M.; Sørensen, S.P.L. The Proteins in Whey; Hagerup in Komm: Copenhague, Denmark, 1939. [Google Scholar]
- Groves, M.L. The Isolation of a Red Protein from Milk2. J. Am. Chem. Soc. 1960, 82, 3345–3350. [Google Scholar] [CrossRef]
- Johanson, B. Isolation of an Iron-Containing Red Protein from Human Milk. Acta Chem. Scand. 1960, 14, 510–512. [Google Scholar] [CrossRef]
- Montreuil, J.; Tonnelat, J.; Mullet, S. Preparation and properties of lactosiderophilin (lactotransferrin) of human milk. Biochim. Biophys. Acta 1960, 45, 413–421. [Google Scholar] [CrossRef]
- Sanchez, L.; Calvo, M.; Brock, J.H. Biological role of lactoferrin. Arch. Dis. Child. 1992, 67, 657–661. [Google Scholar] [CrossRef]
- Van der Strate, B.; Beljaars, L.; Molema, G.; Harmsen, M.; Meijer, D. Antiviral activities of lactoferrin. Antivir. Res. 2001, 52, 225–239. [Google Scholar] [CrossRef]
- Levay, P.F.; Viljoen, M. Lactoferrin: A general review. Haematologica 1995, 80, 252–267. [Google Scholar]
- Cutone, A.; Rosa, L.; Ianiro, G.; Lepanto, M.S.; Di Patti, M.C.B.; Valenti, P.; Musci, G. Lactoferrin’s Anti-Cancer Properties: Safety, Selectivity, and Wide Range of Action. Biomolecules 2020, 10, 456. [Google Scholar] [CrossRef] [PubMed]
- Di Patti, M.C.B.; Cutone, A.; Polticelli, F.; Rosa, L.; Lepanto, M.S.; Valenti, P.; Musci, G. The ferroportin-ceruloplasmin system and the mammalian iron homeostasis machine: Regulatory pathways and the role of lactoferrin. BioMetals 2018, 31, 399–414. [Google Scholar] [CrossRef]
- Rosa, L.; Cutone, A.; Lepanto, M.S.; Paesano, R.; Valenti, P. Lactoferrin: A Natural Glycoprotein Involved in Iron and Inflammatory Homeostasis. Int. J. Mol. Sci. 2017, 18, 1985. [Google Scholar] [CrossRef]
- Czosnykowska-Łukacka, M.; Orczyk-Pawiłowicz, M.; Broers, B.; Krolak-Olejnik, B. Lactoferrin in Human Milk of Prolonged Lactation. Nutrients 2019, 11, 2350. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Franco, D.A.; Vázquez-Moreno, L.; Ramos-Clamont Montfort, G. Actividad antimicrobiana de la lactoferrina: Mecanismos y aplicaciones clínicas potenciales. Rev. Latinoam. Microbiol. 2005, 47, 102–111. [Google Scholar] [PubMed]
- Cheng, J.B.; Wang, J.Q.; Bu, D.P.; Liu, G.L.; Zhang, C.G.; Wei, H.Y.; Zhou, L.Y.; Wang, J.Z. Factors Affecting the Lactoferrin Concentration in Bovine Milk. J. Dairy Sci. 2008, 91, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Sánchez, L.; Aranda, P.; Pérez, M.; Calvo, M. Concentration of Lactoferrin and Transferrin throughout Lactation in Cow’s Colostrum and Milk. Biol. Chem. Hoppe-Seyler 1988, 369, 1005–1008. [Google Scholar] [CrossRef] [PubMed]
- Yamauchi, K.; Wakabayashi, H.; Shin, K.; Takase, M. Bovine lactoferrin: Benefits and mechanism of action against infectionsThis paper is one of a selection of papers published in this Special Issue, entitled 7th International Conference on Lactoferrin: Structure, Functions, and Applications, and has undergone the Journal’s usual peer review process. Biochem. Cell Biol. 2006, 84, 291–296. [Google Scholar] [CrossRef]
- Kell, D.B.; Heyden, E.L.; Pretorius, E. The Biology of Lactoferrin, an Iron-Binding Protein That Can Help Defend Against Viruses and Bacteria. Front. Immunol. 2020, 11, 1221. [Google Scholar] [CrossRef] [PubMed]
- Arias, M.; Hilchie, A.L.; Haney, E.F.; Bolscher, J.G.M.; Hyndman, M.E.; Hancock, R.E.W.; Vogel, H.J. Anticancer activities of bovine and human lactoferricin-derived peptides. Biochem. Cell Biol. 2017, 95, 91–98. [Google Scholar] [CrossRef] [PubMed]
- Drago-Serrano, M.E.; Campos-Rodriguez, R.; Carrero, J.C.; De La Garza, M. Lactoferrin and Peptide-derivatives: Antimicrobial Agents with Potential Use in Nonspecific Immunity Modulation. Curr. Pharm. Des. 2018, 24, 1067–1078. [Google Scholar] [CrossRef]
- Britigan, B.E.; Lewis, T.S.; Waldschmidt, M.; McCormick, M.L.; Krieg, A.M. Lactoferrin Binds CpG-Containing Oligonucleotides and Inhibits Their Immunostimulatory Effects on Human B Cells. J. Immunol. 2001, 167, 2921–2928. [Google Scholar] [CrossRef]
- Gibbons, J.A.; Kanwar, R.K.; Kanwar, J.R. Lactoferrin and cancer in different cancer models. Front. Biosci. 2011, 3, 1080–1088. [Google Scholar] [CrossRef]
- Legrand, D. Lactoferrin, a key molecule in immune and inflammatory processes1This article is part of Special Issue entitled Lactoferrin and has undergone the Journal’s usual peer review process. Biochem. Cell Biol. 2012, 90, 252–268. [Google Scholar] [CrossRef] [PubMed]
- Cohen, M.S.; Mao, J.; Rasmussen, G.T.; Serody, J.; Britigan, B.E. Interaction of Lactoferrin and Lipopolysaccharide (LPS): Effects on the Antioxidant Property of Lactoferrin and the Ability of LPS to Prime Human Neutrophils for Enhanced Superoxide Formation. J. Infect. Dis. 1992, 166, 1375. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Zhang, G.; Chen, H.; Cao, Y.; Dong, X.; Li, H.; Liu, C. Dose Effect of Bovine Lactoferrin Fortification on Iron Metabolism of Anemic Infants. J. Nutr. Sci. Vitaminol. 2020, 66, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Ellison, R.T.; Giehl, T.J. Killing of gram-negative bacteria by lactoferrin and lysozyme. J. Clin. Investig. 1991, 88, 1080–1091. [Google Scholar] [CrossRef] [PubMed]
- Kawakami, H.; Lonnerdal, B. Isolation and function of a receptor for human lactoferrin in human fetal intestinal brush-border membranes. Am. J. Physiol. Gastrointest. Liver Physiol. 1991, 261, G841–G846. [Google Scholar] [CrossRef]
- Lönnerdal, B.; Jiang, R.; Du, X. Bovine Lactoferrin Can Be Taken Up by the Human Intestinal Lactoferrin Receptor and Exert Bioactivities. J. Pediatr. Gastroenterol. Nutr. 2011, 53, 606–614. [Google Scholar] [CrossRef]
- Spik, G.; Brunet, B.; Mazurier-Dehaine, C.; Fontaine, G.; Montreuil, J. Characterization and properties of the human and bovine lactotransferrins extracted from the faeces of newborn infants. Acta Paediatr. 1982, 71, 979–985. [Google Scholar] [CrossRef]
- Liao, Y.; Jiang, R.; Lönnerdal, B. Biochemical and molecular impacts of lactoferrin on small intestinal growth and development during early life11This article is part of a Special Issue entitled Lactoferrin and has undergone the Journal’s usual peer review process. Biochem. Cell Biol. 2012, 90, 476–484. [Google Scholar] [CrossRef]
- Lepanto, M.S.; Rosa, L.; Cutone, A.; Conte, M.P.; Paesano, R.; Valenti, P. Efficacy of Lactoferrin Oral Administration in the Treatment of Anemia and Anemia of Inflammation in Pregnant and Non-pregnant Women: An Interventional Study. Front. Immunol. 2018, 9, 2123. [Google Scholar] [CrossRef] [PubMed]
- Cutone, A.; Lepanto, M.S.; Rosa, L.; Scotti, M.J.; Rossi, A.; Ranucci, S.; De Fino, I.; Bragonzi, A.; Valenti, P.; Musci, G.; et al. Aerosolized Bovine Lactoferrin Counteracts Infection, Inflammation and Iron Dysbalance in A Cystic Fibrosis Mouse Model of Pseudomonas aeruginosa Chronic Lung Infection. Int. J. Mol. Sci. 2019, 20, 2128. [Google Scholar] [CrossRef]
- Valenti, P.; Frioni, A.; Rossi, A.; Ranucci, S.; De Fino, I.; Cutone, A.; Rosa, L.; Bragonzi, A.; Berlutti, F. Aerosolized bovine lactoferrin reduces neutrophils and pro-inflammatory cytokines in mouse models of Pseudomonas aeruginosa lung infections. Biochem. Cell Biol. 2017, 95, 41–47. [Google Scholar] [CrossRef]
- Bezault, J.; Bhimani, R.; Wiprovnick, J.; Furmanski, P. Human lactoferrin inhibits growth of solid tumors and development of experimental metastases in mice. Cancer Res. 1994, 54, 2310–2312. [Google Scholar]
- Shau, H.; Kim, A.; Golub, S.H. Modulation of natural killer and lymphokine-activated killer cell cytotoxicity by lactoferrin. J. Leukoc. Biol. 1992, 51, 343–349. [Google Scholar] [CrossRef] [PubMed]
- Iigo, M.; Shimamura, M.; Matsuda, E.; Fujita, K.-I.; Nomoto, H.; Satoh, J.; Kojima, S.; Alexander, D.B.; Moore, M.A.; Tsuda, H. Orally administered bovine lactoferrin induces caspase-1 and interleukin-18 in the mouse intestinal mucosa: A possible explanation for inhibition of carcinogenesis and metastasis. Cytokine 2004, 25, 36–44. [Google Scholar] [CrossRef]
- Chaix, J.; Tessmer, M.S.; Hoebe, K.; Fuséri, N.; Ryffel, B.; Dalod, M.; Alexopoulou, L.; Beutler, B.; Brossay, L.; Vivier, E.; et al. Cutting Edge: Priming of NK Cells by IL-18. J. Immunol. 2008, 181, 1627–1631. [Google Scholar] [CrossRef] [PubMed]
- Nakanishi, K. Unique Action of Interleukin-18 on T Cells and Other Immune Cells. Front. Immunol. 2018, 9, 763. [Google Scholar] [CrossRef]
- Akita, K.; Ohtsuki, T.; Nukada, Y.; Tanimoto, T.; Namba, M.; Okura, T.; Takakura-Yamamoto, R.; Torigoe, K.; Gu, Y.; Su, M.S.-S.; et al. Involvement of Caspase-1 and Caspase-3 in the Production and Processing of Mature Human Interleukin 18 in Monocytic THP.1 Cells. J. Biol. Chem. 1997, 272, 26595–26603. [Google Scholar] [CrossRef] [PubMed]
- Kuhara, T.; Yamauchi, K.; Tamura, Y.; Okamura, H. Oral Administration of Lactoferrin Increases NK Cell Activity in Mice via Increased Production of IL-18 and Type I IFN in the Small Intestine. J. Interf. Cytokine Res. 2006, 26, 489–499. [Google Scholar] [CrossRef]
- Lorget, F.; Clough, J.; Oliveira, M.; Daury, M.-C.; Sabokbar, A.; Offord, E. Lactoferrin reduces in vitro osteoclast differentiation and resorbing activity. Biochem. Biophys. Res. Commun. 2002, 296, 261–266. [Google Scholar] [CrossRef]
- Damiens, E.; El Yazidi, I.; Mazurier, J.; Duthille, I.; Spik, G.; Boilly-Marer, Y. Lactoferrin inhibits G1 cyclin-dependent kinases during growth arrest of human breast carcinoma cells. J. Cell. Biochem. 1999, 74, 486–498. [Google Scholar] [CrossRef]
- Deng, M.; Zhang, W.; Tang, H.; Ye, Q.; Liao, Q.; Zhou, Y.; Wu, M.; Xiong, W.; Zheng, Y.; Guo, X.; et al. Lactotransferrin acts as a tumor suppressor in nasopharyngeal carcinoma by repressing AKT through multiple mechanisms. Oncogene 2012, 32, 4273–4283. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, R.R.; Mansour, D.F.; Salama, A.A.; Abdel-Rahman, R.F.; Hassan, A.M. Regulation of PKB/Akt-pathway in the chemopreventive effect of lactoferrin against diethylnitrosamine-induced hepatocarcinogenesis in rats. Pharmacol. Rep. 2019, 71, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Hayes, T.G.; Falchook, G.F.; Varadhachary, G.R.; Smith, D.P.; Davis, L.D.; Dhingra, H.M.; Hayes, B.P.; Varadhachary, A. Phase I trial of oral talactoferrin alfa in refractory solid tumors. Investig. New Drugs 2005, 24, 233–240. [Google Scholar] [CrossRef]
- Kuwata, H.; Yip, T.-T.; Tomita, M.; Hutchens, T. Direct evidence of the generation in human stomach of an antimicrobial peptide domain (lactoferricin) from ingested lactoferrin. Biochim. Biophys. Acta (BBA) Protein Struct. Mol. Enzym. 1998, 1429, 129–141. [Google Scholar] [CrossRef]
- Gifford, J.L.; Hunter, H.N.; Vogel, H.J. Lactoferricin. Cell. Mol. Life Sci. 2005, 62, 2588–2598. [Google Scholar] [CrossRef]
- Connor, J.; Bucana, C.; Fidler, I.J.; Schroit, A.J. Differentiation-dependent expression of phosphatidylserine in mammalian plasma membranes: Quantitative assessment of outer-leaflet lipid by prothrombinase complex formation. Proc. Natl. Acad. Sci. USA 1989, 86, 3184–3188. [Google Scholar] [CrossRef]
- Qasba, P.K.; Kumar, S.; Brew, K. Molecular Divergence of Lysozymes and α-Lactalbumin. Crit. Rev. Biochem. Mol. Biol. 1997, 32, 255–306. [Google Scholar] [CrossRef]
- Pettersson, J.; Mossberg, A.-K.; Svanborg, C. α-Lactalbumin species variation, HAMLET formation, and tumor cell death. Biochem. Biophys. Res. Commun. 2006, 345, 260–270. [Google Scholar] [CrossRef]
- Hakansson, A.; Zhivotovsky, B.; Orrenius, S.; Sabharwal, H.; Svanborg, C. Apoptosis induced by a human milk protein. Proc. Natl. Acad. Sci. USA 1995, 92, 8064–8068. [Google Scholar] [CrossRef]
- Svensson, M.; Hakansson, A.; Mossberg, A.-K.; Linse, S.; Svanborg, C. Conversion of alpha -lactalbumin to a protein inducing apoptosis. Proc. Natl. Acad. Sci. USA 2000, 97, 4221–4226. [Google Scholar] [CrossRef]
- Brinkmann, C.R.; Heegaard, C.W.; Petersen, T.E.; Jensenius, J.C.; Thiel, S. The toxicity of bovine α-lactalbumin made lethal to tumor cells is highly dependent on oleic acid and induces killing in cancer cell lines and noncancer-derived primary cells. FEBS J. 2011, 278, 1955–1967. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, K.; Oobatake, M.; Goto, Y. Salt-dependent monomer-dimer equilibrium of bovine β-lactoglobulin at pH 3. Protein Sci. 2008, 10, 2325–2335. [Google Scholar] [CrossRef]
- Jameson, G.B.; Adams, J.J.; Creamer, L.K. Flexibility, functionality and hydrophobicity of bovine β-lactoglobulin. Int. Dairy J. 2002, 12, 319–329. [Google Scholar] [CrossRef]
- Ragona, L.; Fogolari, F.; Catalano, M.; Ugolini, R.; Zetta, L.; Molinari, H. EF Loop Conformational Change Triggers Ligand Binding in β-Lactoglobulins. J. Biol. Chem. 2003, 278, 38840–38846. [Google Scholar] [CrossRef] [PubMed]
- Reddy, I.M.; Kella, N.K.D.; Kinsella, J.E. Structural and conformational basis of the resistance of.beta.-lactoglobulin to peptic and chymotryptic digestion. J. Agric. Food Chem. 1988, 36, 737–741. [Google Scholar] [CrossRef]
- Bijari, N.; Ghobadi, S.; Derakhshandeh, K. β-lactoglobulin-irinotecan inclusion complex as a new targeted nanocarrier for colorectal cancer cells. Res. Pharm. Sci. 2019, 14, 216–227. [Google Scholar] [CrossRef] [PubMed]
- Lišková, K.; Auty, M.A.E.; Chaurin, V.; Min, S.; Mok, K.H.; O’Brien, N.; Kelly, A.L.; Brodkorb, A. Cytotoxic complexes of sodium oleate with β-lactoglobulin. Eur. J. Lipid Sci. Technol. 2011, 113, 1207–1218. [Google Scholar] [CrossRef]
- Mather, I.H.; Keenan, T.W. Origin and Secretion of Milk Lipids. J. Mammary Gland. Biol. Neoplasia 1998, 3, 259–273. [Google Scholar] [CrossRef] [PubMed]
- Fong, B.Y.; Norris, C.S.; MacGibbon, A.K. Protein and lipid composition of bovine milk-fat-globule membrane. Int. Dairy J. 2007, 17, 275–288. [Google Scholar] [CrossRef]
- Kvistgaard, A.; Pallesen, L.; Arias, C.; Lopez, S.; Petersen, T.; Heegaard, C.; Rasmussen, J.T. Inhibitory Effects of Human and Bovine Milk Constituents on Rotavirus Infections. J. Dairy Sci. 2004, 87, 4088–4096. [Google Scholar] [CrossRef]
- Reinhardt, T.; Lippolis, J. Bovine Milk Fat Globule Membrane Proteome. J. Dairy Res. 2006, 73, 406–416. [Google Scholar] [CrossRef]
- Zanabria, R.; Griffiths, M.W.; Corredig, M. Does structure affect biological function? Modifications to the protein and phospholipids fraction of the milk fat globule membrane after extraction affect the antiproliferative activity of colon cancer cells. J. Food Biochem. 2020, 44, e13104. [Google Scholar] [CrossRef]
- Clare, D.A.; Zheng, Z.; Hassan, H.M.; Swaisgood, H.E.; Catignani, G.L. Antimicrobial Properties of Milkfat Globule Membrane Fractions. J. Food Prot. 2008, 71, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Rosenblatt, D.A.N.; Chan, D.S.M.; Vieira, A.R.; Vieira, R.; Greenwood, D.C.; Vatten, L.J.; Norat, T. Dairy products, calcium, and prostate cancer risk: A systematic review and meta-analysis of cohort studies. Am. J. Clin. Nutr. 2014, 101, 87–117. [Google Scholar] [CrossRef] [PubMed]
- Harrison, S.; Lennon, R.; Holly, J.; Higgins, J.P.T.; Gardner, M.; Perks, C.; Gaunt, T.; Tan, V.; Borwick, C.; Emmet, P.; et al. Does milk intake promote prostate cancer initiation or progression via effects on insulin-like growth factors (IGFs)? A systematic review and meta-analysis. Cancer Causes Control. 2017, 28, 497–528. [Google Scholar] [CrossRef]
- Song, Y.; Chavarro, J.E.; Cao, Y.; Qiu, W.; Mucci, L.; Sesso, H.D.; Stampfer, M.J.; Giovannucci, E.; Pollak, M.; Liu, S.; et al. Whole milk intake is associated with prostate cancer-specific mortality among U.S. male physicians. J. Nutr. 2012, 143, 189–196. [Google Scholar] [CrossRef]
- Park, S.-Y.; Murphy, S.P.; Wilkens, L.R.; Stram, D.O.; Henderson, B.E.; Kolonel, L.N. Calcium, Vitamin D, and Dairy Product Intake and Prostate Cancer Risk: The Multiethnic Cohort Study. Am. J. Epidemiol. 2007, 166, 1259–1269. [Google Scholar] [CrossRef] [PubMed]
- Tate, P.L.; Bibb, R.; Larcom, L.L. Milk Stimulates Growth of Prostate Cancer Cells in Culture. Nutr. Cancer 2011, 63, 1361–1366. [Google Scholar] [CrossRef]
- Qin, L.-Q.; Wang, P.-Y.; Xu, J.-Y.; Li, J.; Wang, J.; Sato, A. The Effects of Commercial whole Milk on the Prostate Carcinogenesis in Rats with or without Induction by 2-Amino-1-methyl-6-phenylimidazo[4,5-b]pyridine. J. Heal. Sci. 2006, 52, 419–424. [Google Scholar] [CrossRef][Green Version]
- Fraser, G.; Jaceldo-Siegl, K.; Orlich, M.; Mashchak, A.; Sirirat, R.; Knutsen, S. Dairy, soy, and risk of breast cancer: Those confounded milks. Int. J. Epidemiol. 2020, 49, 1526–1537. [Google Scholar] [CrossRef]
- Galván-Salazar, H.R.; Arreola-Cruz, A.; Madrigal-Pérez, D.; Soriano-Hernández, A.D.; Guzman-Esquivel, J.; Montes-Galindo, D.A.; López-Flores, R.A.; Gomez, F.E.-; Rodríguez-Sanchez, I.P.; Newton-Sanchez, O.A.; et al. Association of Milk and Meat Consumption with the Development of Breast Cancer in a Western Mexican Population. Breast Care 2015, 10, 393–396. [Google Scholar] [CrossRef]
- Pala, V.; Krogh, V.; Berrino, F.; Sieri, S.; Grioni, S.; Tjonneland, A.; Olsen, A.; Jakobsen, M.U.; Overvad, K.; Clavel-Chapelon, F.; et al. Meat, eggs, dairy products, and risk of breast cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort. Am. J. Clin. Nutr. 2009, 90, 602–612. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Sundquist, K. Lactose intolerance and risk of lung, breast and ovarian cancers: Aetiological clues from a population-based study in Sweden. Br. J. Cancer 2015, 112, 149–152. [Google Scholar] [CrossRef]
- Kong, F.; Singh, R.P. A Human Gastric Simulator (HGS) to Study Food Digestion in Human Stomach. J. Food Sci. 2010, 75, E627–E635. [Google Scholar] [CrossRef] [PubMed]
- Schwendel, B.H.; Wester, T.J.; Morel, P.C.; Fong, B.; Tavendale, M.H.; Deadman, C.; Shadbolt, N.M.; Otter, D.E. Pasture feeding conventional cows removes differences between organic and conventionally produced milk. Food Chem. 2017, 229, 805–813. [Google Scholar] [CrossRef] [PubMed]
- Tunick, M.H.; Van Hekken, D.L.; Paul, M.; Ingham, E.R.; Karreman, H.J. Case study: Comparison of milk composition from adjacent organic and conventional farms in the USA. Int. J. Dairy Technol. 2016, 69, 137–142. [Google Scholar] [CrossRef]
- Jahreis, G.; Fritsche, J.; Steinhart, H. Conjugated linoleic acid in milk fat: High variation depending on production system. Nutr. Res. 1997, 17, 1479–1484. [Google Scholar] [CrossRef]
- Kuczyńska, B.; Puppel, K.; Gołȩbiewski, M.; Metera, E.; Sakowski, T.; Słoniewski, K.; Gołębiewski, M. Differences in whey protein content between cow’s milk collected in late pasture and early indoor feeding season from conventional and organic farms in Poland. J. Sci. Food Agric. 2012, 92, 2899–2904. [Google Scholar] [CrossRef]
- Tsakali, E.; Chatzilazarou, A.; Houhoula, D.; Koulouris, S.; Tsaknis, J.; Van Impe, J. A rapid HPLC method for the determination of lactoferrin in milk of various species. J. Dairy Res. 2019, 86, 238–241. [Google Scholar] [CrossRef] [PubMed]
- Fardet, A.; Rock, E. In vitro and in vivo antioxidant potential of milks, yoghurts, fermented milks and cheeses: A narrative review of evidence. Nutr. Res. Rev. 2018, 31, 52–70. [Google Scholar] [CrossRef]
- Paul, M.; Somkuti, G.A. Hydrolytic breakdown of lactoferricin by lactic acid bacteria. J. Ind. Microbiol. Biotechnol. 2010, 37, 173–178. [Google Scholar] [CrossRef]
- Zhang, J.; Lai, S.; Cai, Z.; Chen, Q.; Huang, B.; Ren, Y. Determination of bovine lactoferrin in dairy products by ultra-high performance liquid chromatography–tandem mass spectrometry based on tryptic signature peptides employing an isotope-labeled winged peptide as internal standard. Anal. Chim. Acta 2014, 829, 33–39. [Google Scholar] [CrossRef]
- Campanella, L.; Martini, E.; Tomassetti, M. New immunosensor for Lactoferrin determination in human milk and several pharmaceutical dairy milk products recommended for the unweaned diet. J. Pharm. Biomed. Anal. 2008, 48, 278–287. [Google Scholar] [CrossRef]
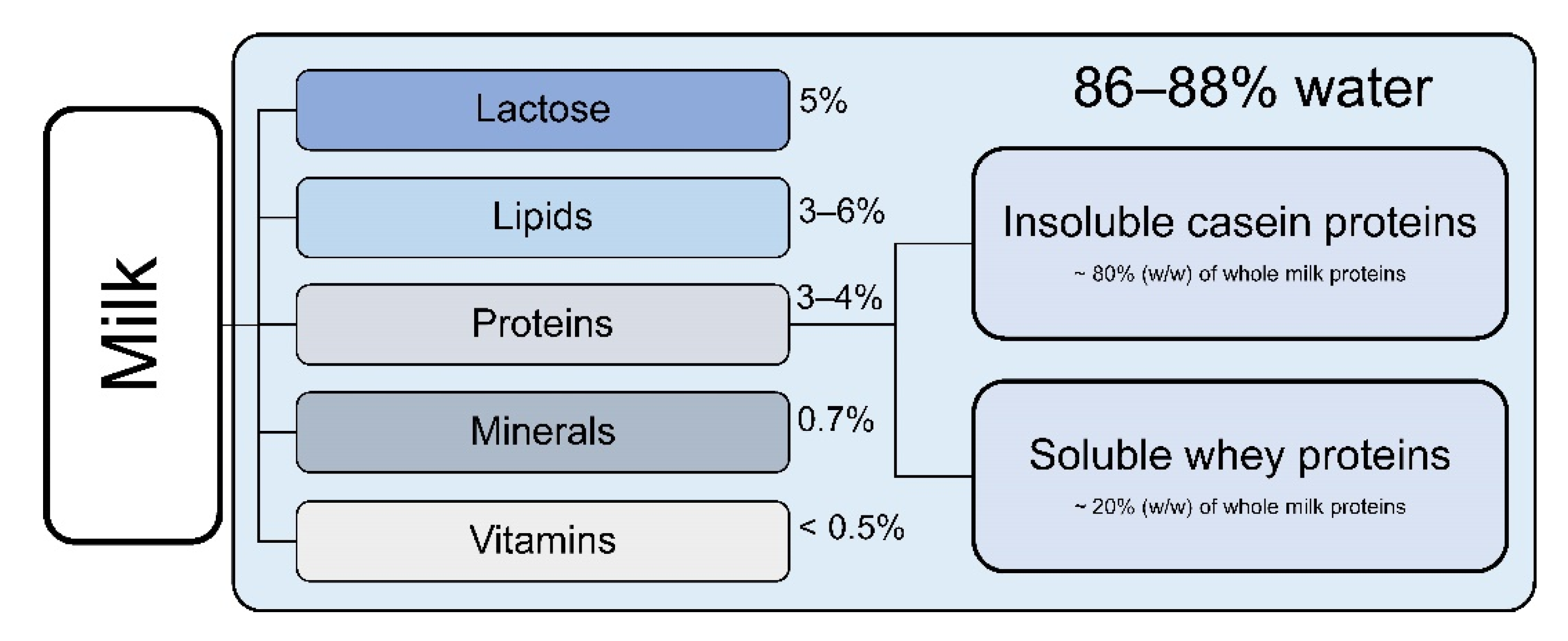
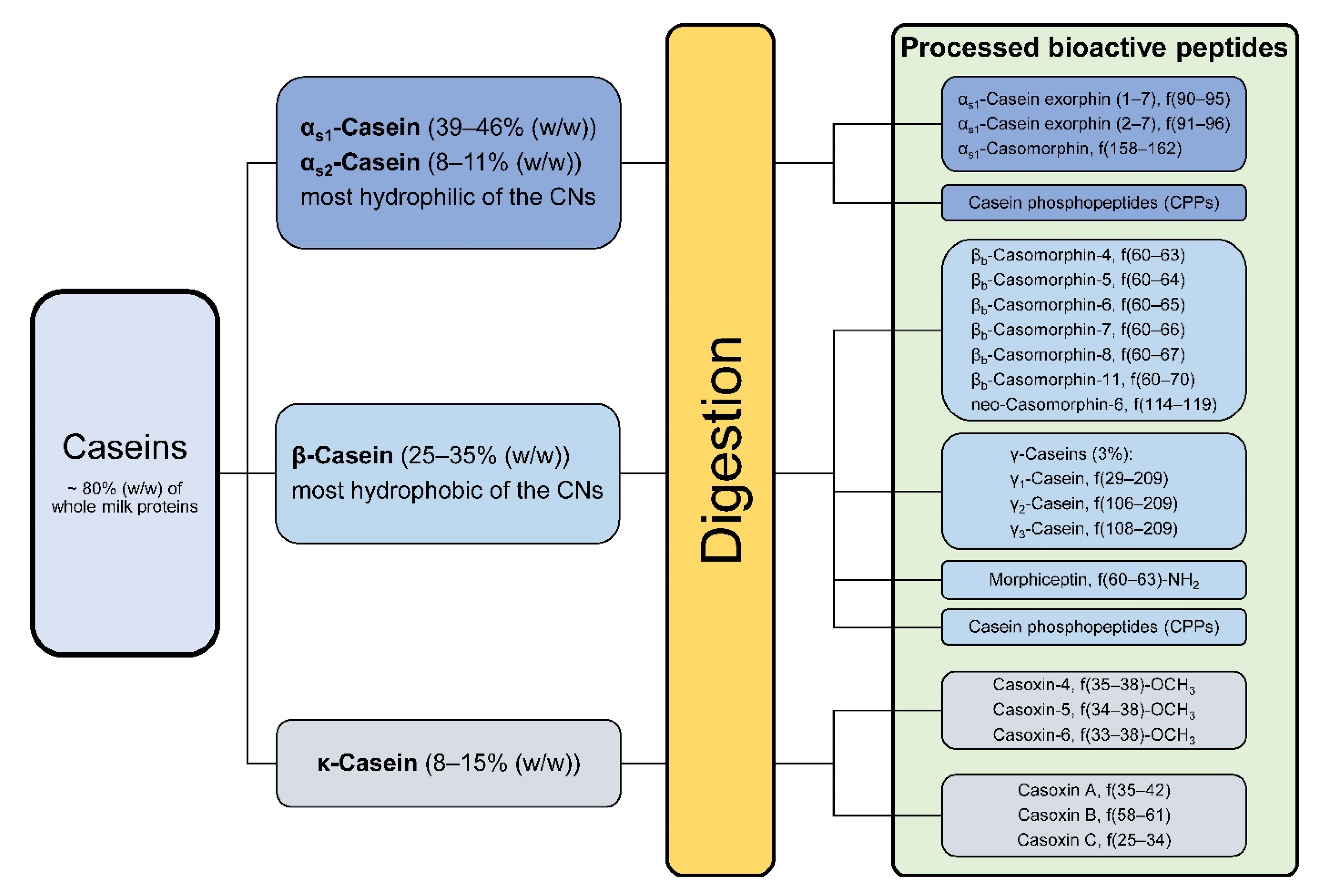
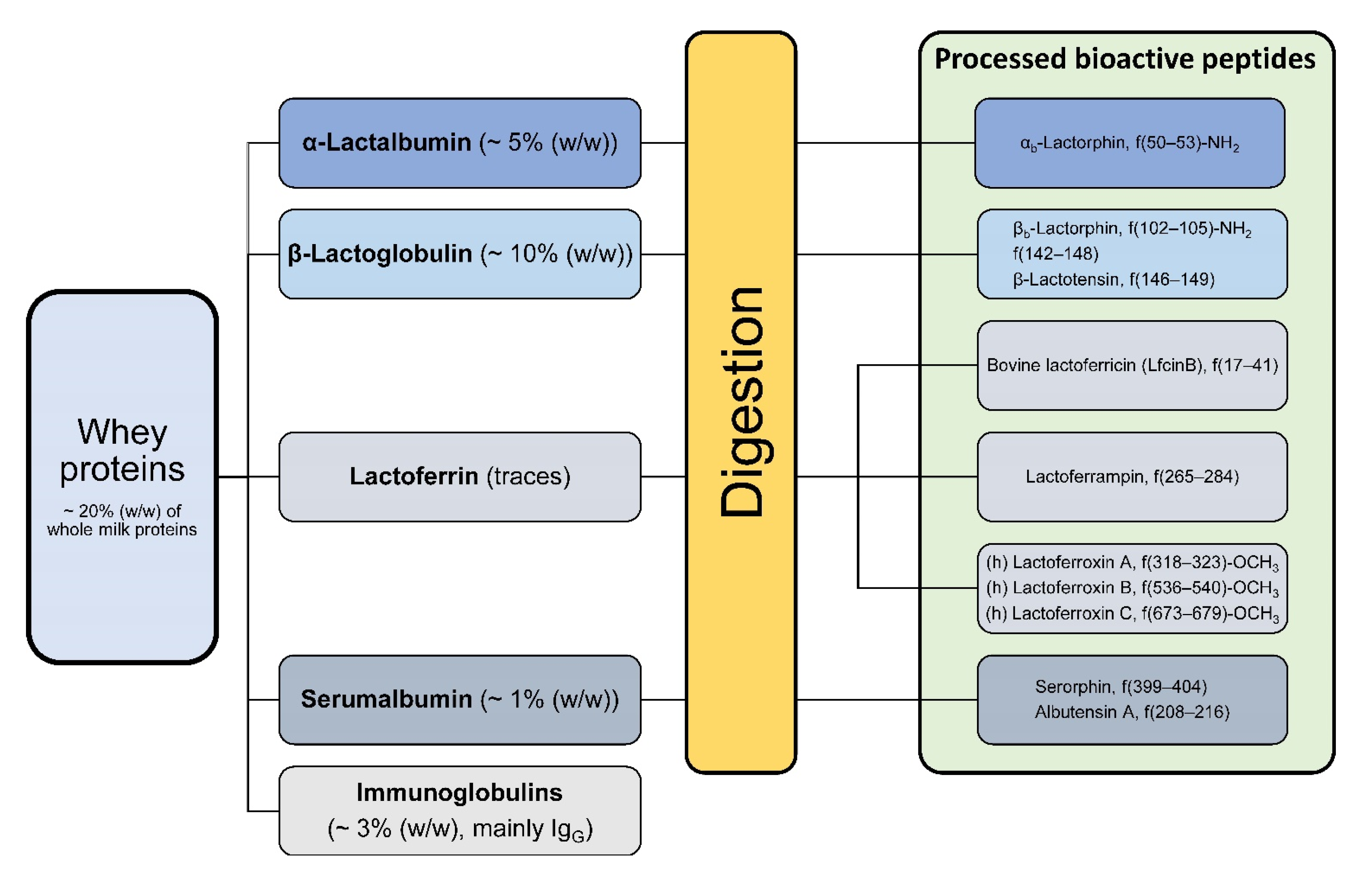
| Proteins | Tumor Species | Effects and Potential Mechanism | Ref. | ||
|---|---|---|---|---|---|
| Casein proteins | CNs | α-, β-, κ-caseins | Breast cancer (human): MCF10A H Ras (G12V), MDA-MB-231 | Decreased cell migration | [24] |
| Mammary tumor (murine): Met-1 | Decreased cell migration, tumor growth, Metastasis, activation of STAT1 signaling, apoptosis induction (shown for α-CN) | ||||
| Lactaptin (κ-casein fragment) | Breast carcinoma (human): MCF-7 | Apoptosis induction | [25] | ||
| PGPIPN (β-casein fragment) | Ovarian cancer (human): SKOV3 | BCL2 targeting | [26] | ||
| INKKI (β-casein fragment) | Melanoma (murine): B16F10 | Apoptosis induction | [27] | ||
| CMs | Prostatic cancer (human): LNCaP, PC-3, DU145 | Interaction with opioid receptors | [28] | ||
| Breast cancer (human): T47D | G0/G1 blocking | [29] | |||
| Promyeloic leukemia (human): HL-60 | Apoptosis induction | [30] | |||
| CPPs | Intestinal tumor (human): HT-29 | Interaction with voltage-operated L-type calcium channels, apoptosis | [31] | ||
| Whey proteins | Lf | Colon carcinoma (murine): Co26Lu | Inhibitory effects on lung metastatic colony formation in Balb/c mice due to Tand NK cell activation | [32] | |
| Melanoma (murine): B16-BL6 | Inhibition of lung metastasis in C57BL/6 mice (only apo-Lf) | [33] | |||
| Lymphoma (murine): L5178Y-ML25 | Inhibition of liver and spleen metastasis in C57BL/6 mice (only apo-Lf) | ||||
| Colon carcinoma (murine) | Reduced induction of aberrant crypt foci (ACF) by azoxymethane administration in male F344 rats | [34] | |||
| Pepsin hydrolysate f(17–38) | Promyeloic leukemia (human): HL-60 | Apoptosis induction | [35] | ||
| Pepsin hydrolysate (mixture) | Oral squamous cell carcinoma (human): SAS | Apoptosis induction by JNK/SAPK MAP kinase activation | [36] | ||
| Basal diet with 0.2% bLf | Hamster buccal pouch (HBP) carcinoma | Decreased incidence of DMBA-induced carcinogenesis, decreased levels of phase I enzymes, modulated lipid peroxidation, increased antioxidant and phase II enzyme activities | [37] | ||
| Native and iron saturated bLf | Glioblastoma (human): GL-15 | Down-regulation of Snail and vimentin expression, increase in cadherin levels Inhibition of EMT-like processes and IL-6/STAT3 axis mainly by the holo-form | [38] | ||
| Liposomal bLf | Colorectal cancer (rat): DMF-DSS induced colorectal | Suppression of inflammation and tumor cell proliferation | [39] | ||
| Lfcin | Fibrosarcoma (murine): Meth A | Reduction of tumor growth in CB6 mice Cytotoxic activity, lysis by pore formation (SEM) | [40] | ||
| Melanoma (murine): B16F10 | Cytotoxic activity | ||||
| Colon carcinoma (murine): C26 | Cytotoxic activity | ||||
| α-LA in complex with oleic acid (BAMLET) | Cervical epithelial carcinoma (human): HeLa | Accumulation in endolysosomal compartment, lysosomal membrane permeabilization inducing nonapoptotic lysosomal cell death | [41] | ||
| Bladder carcinoma (human): J82, RT4 | |||||
| Prostate carcinoma (human): PC-3 | |||||
| Astrocytoma (human): U118 | |||||
| Breast carcinoma (human): MCF-7 | |||||
| Osteosarcoma (human): U2-OS | |||||
| Lymphocytic leukemia (murine): L1210 | |||||
| β-LG | Lung adenocarcinoma (human): A549 | Apoptosis induction, upregulation of Bax and caspase-3, decreased level of Bcl-2, reduced chemotactic motility, tumor inhibition in BALB7c mice after oral administration | [42] | ||
| Intestinal tumor (human): HT-29 | |||||
| Hepatoblastoma (human): HepG2 | |||||
| Breast carcinoma (human): MDA231-LM2 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leischner, C.; Egert, S.; Burkard, M.; Venturelli, S. Potential Protective Protein Components of Cow’s Milk against Certain Tumor Entities. Nutrients 2021, 13, 1974. https://doi.org/10.3390/nu13061974
Leischner C, Egert S, Burkard M, Venturelli S. Potential Protective Protein Components of Cow’s Milk against Certain Tumor Entities. Nutrients. 2021; 13(6):1974. https://doi.org/10.3390/nu13061974
Chicago/Turabian StyleLeischner, Christian, Sarah Egert, Markus Burkard, and Sascha Venturelli. 2021. "Potential Protective Protein Components of Cow’s Milk against Certain Tumor Entities" Nutrients 13, no. 6: 1974. https://doi.org/10.3390/nu13061974
APA StyleLeischner, C., Egert, S., Burkard, M., & Venturelli, S. (2021). Potential Protective Protein Components of Cow’s Milk against Certain Tumor Entities. Nutrients, 13(6), 1974. https://doi.org/10.3390/nu13061974







