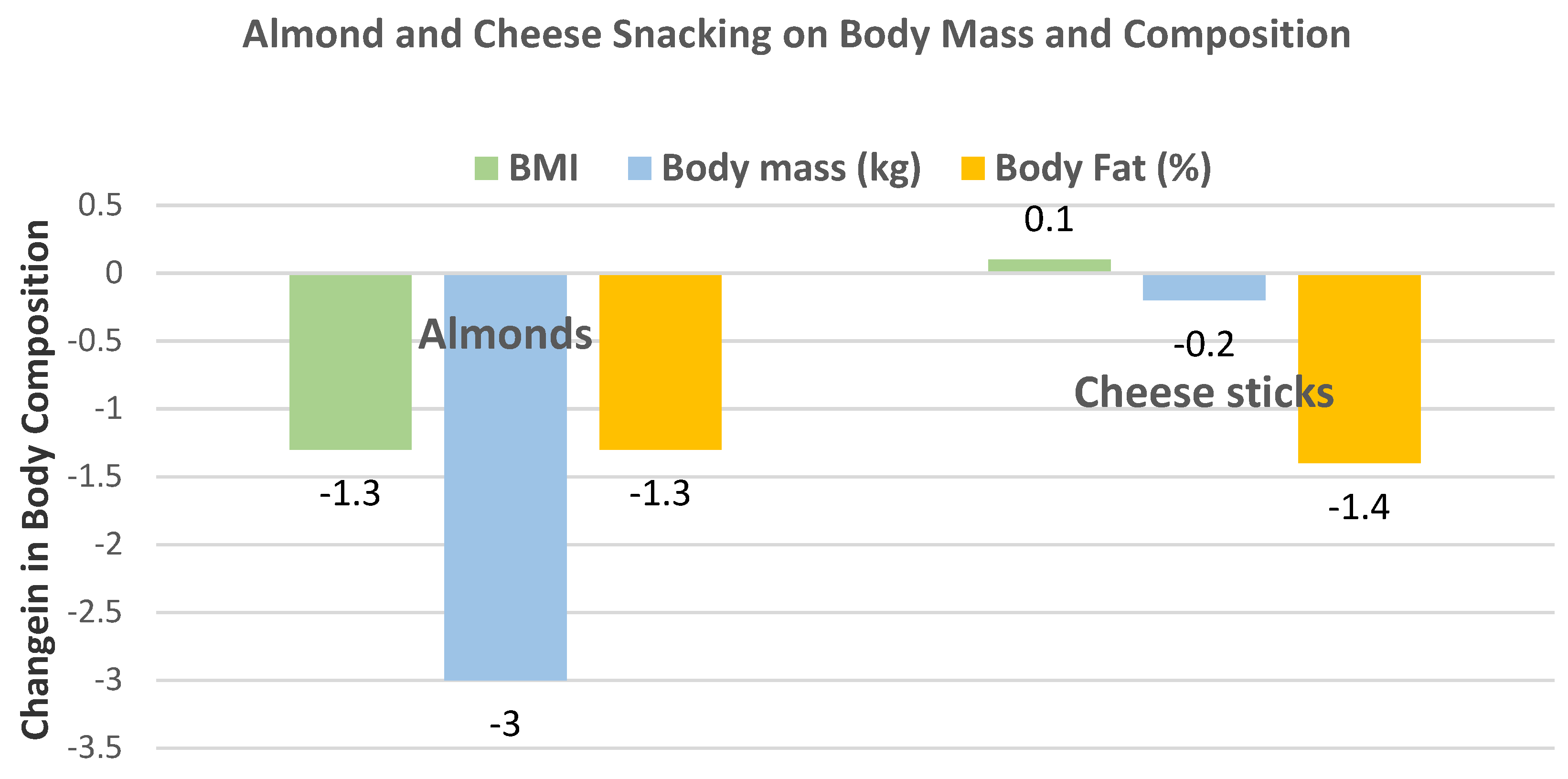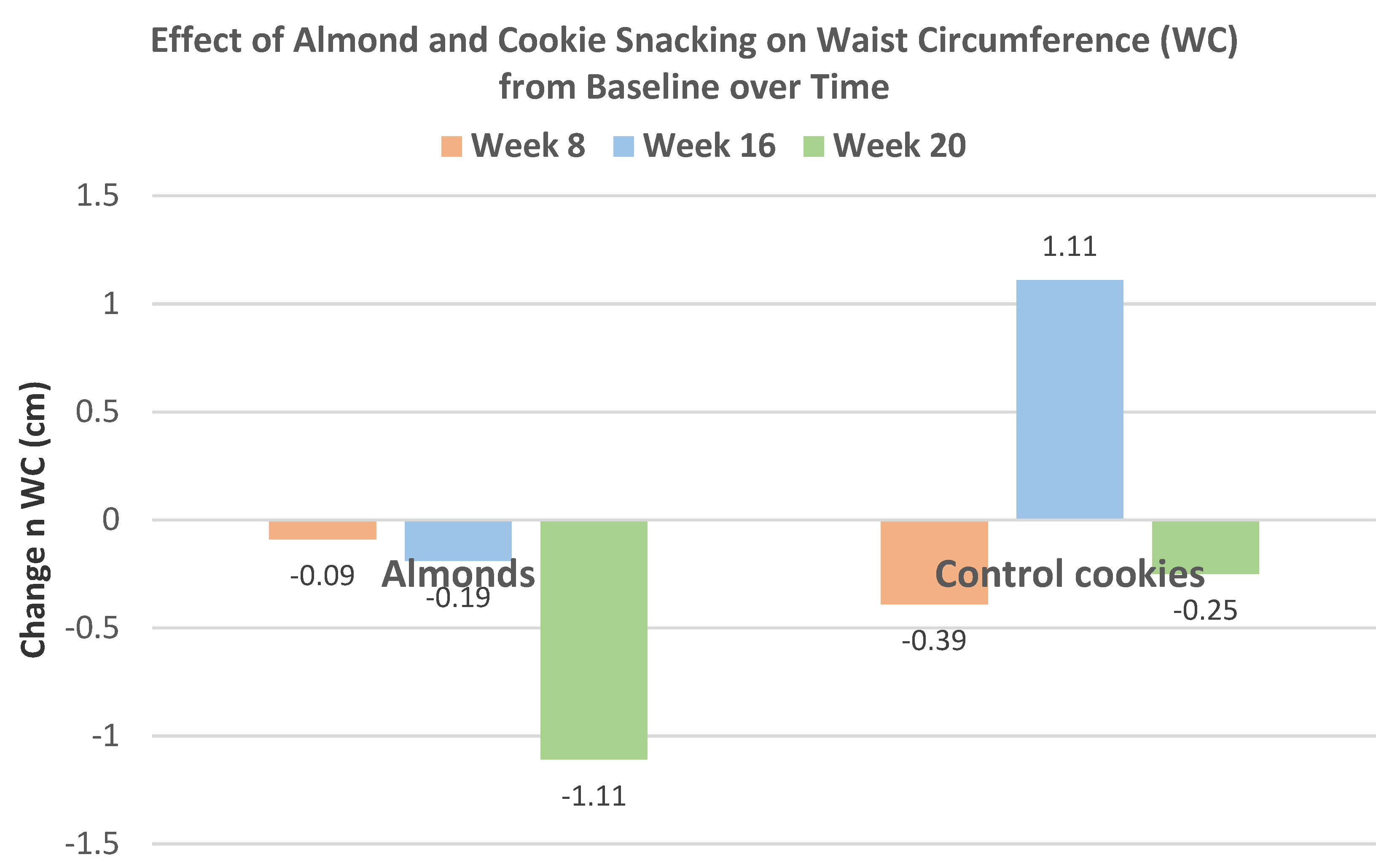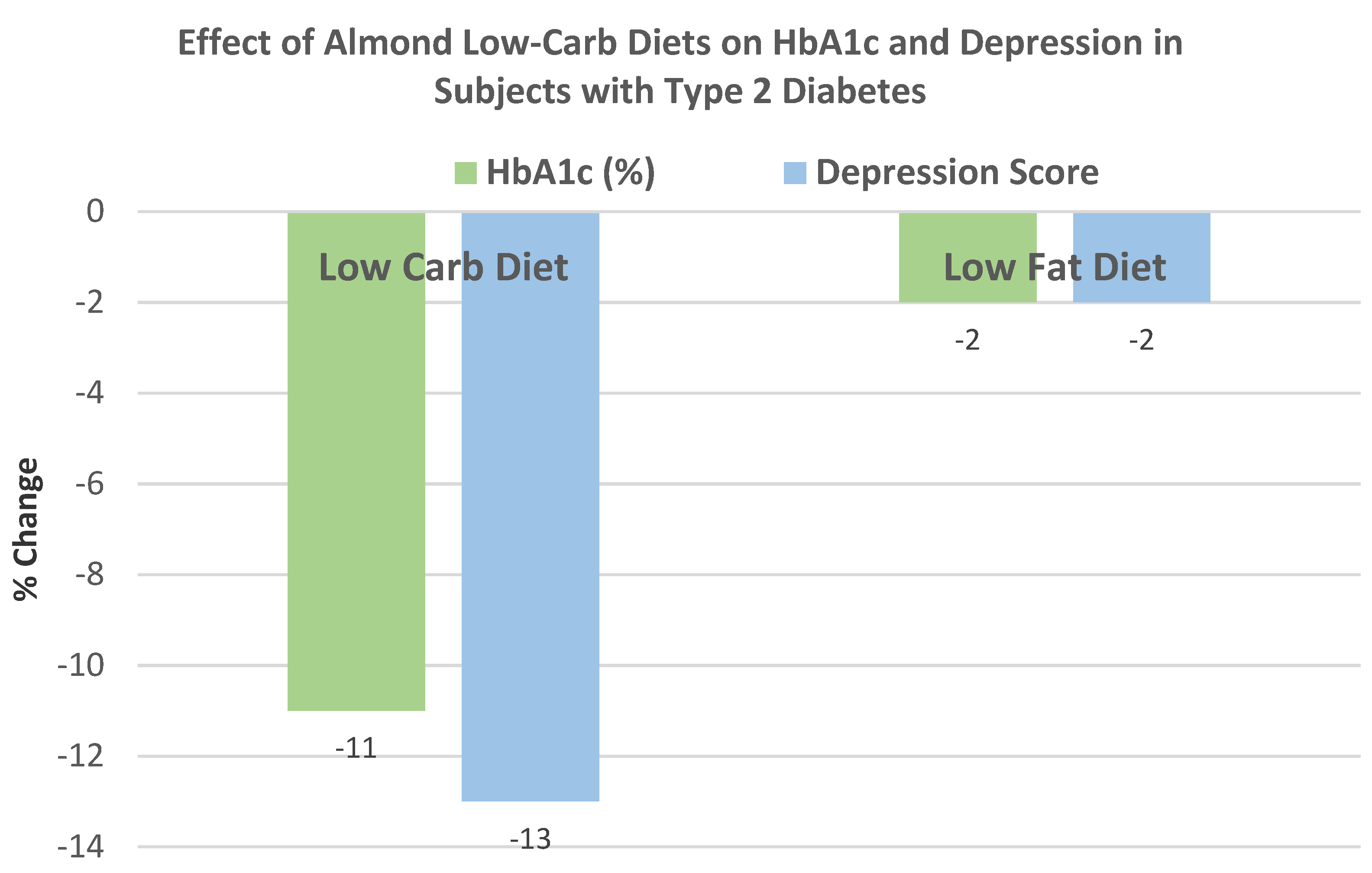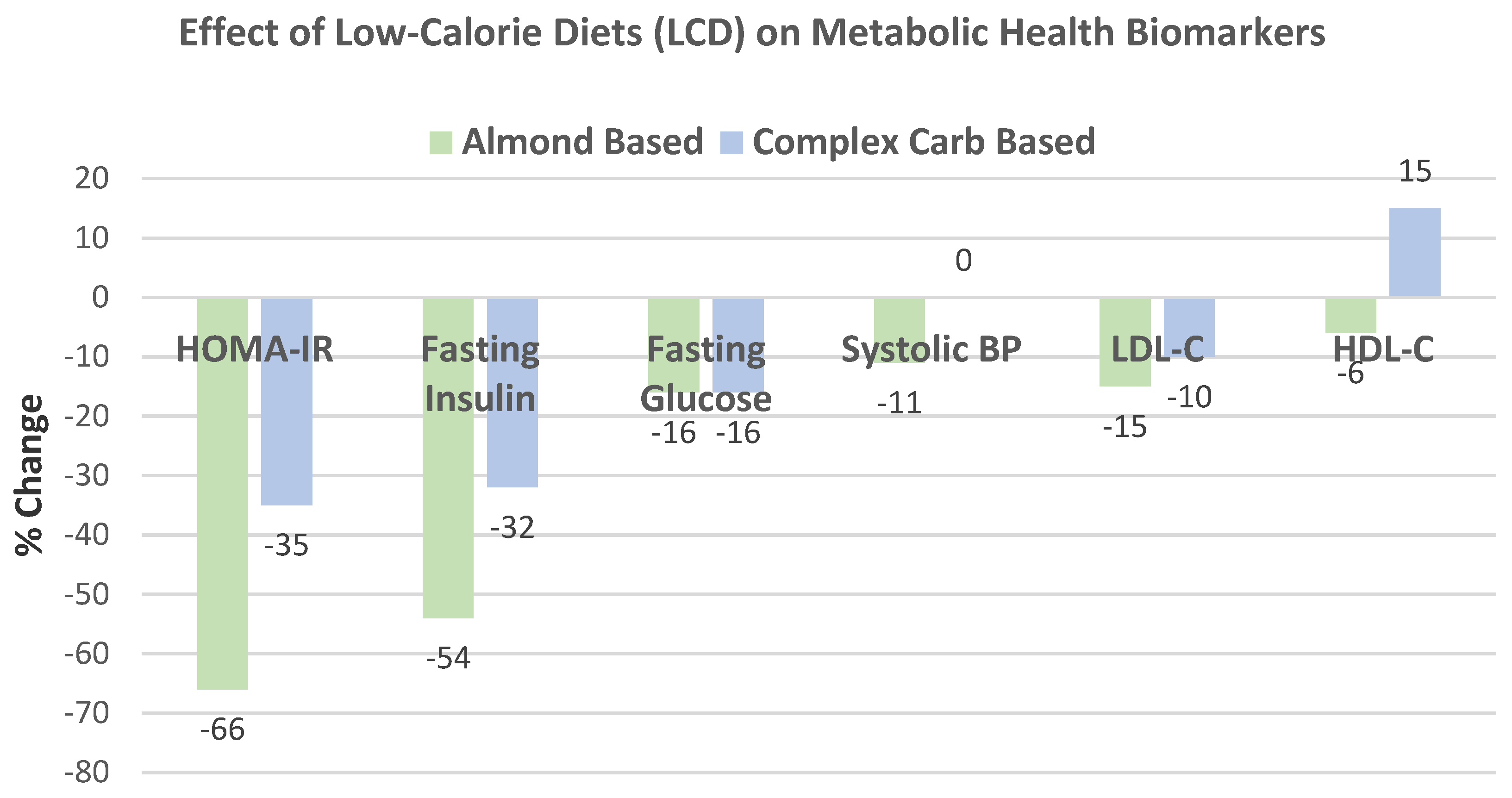3.1.2. Individual Almond RCTs
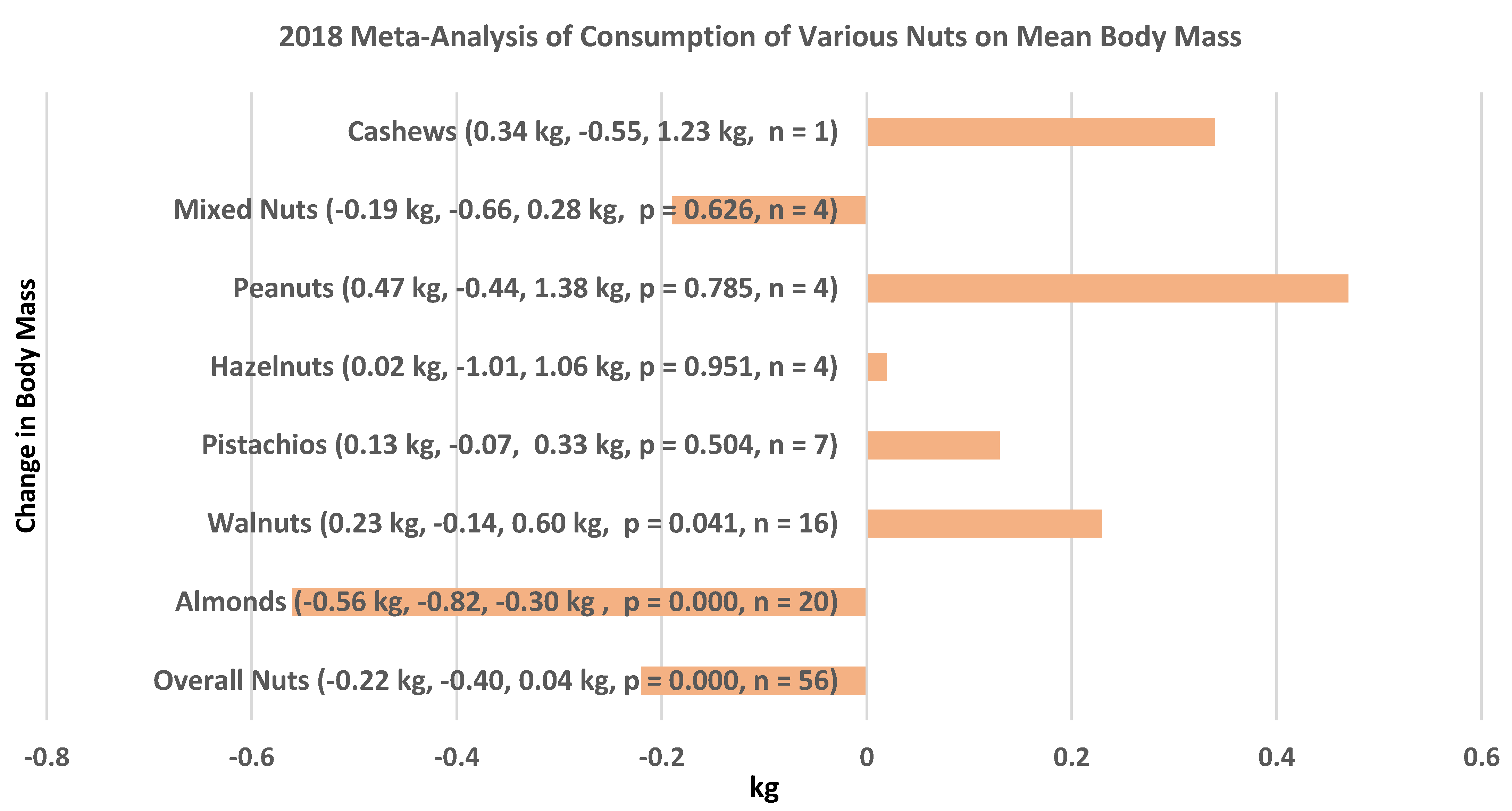
Thirteen RCTs evaluated the effects of habitual diet with added almonds compared to almond-free control diets on changes in body mass and composition, meal and total daily energy intake, energy density (ED) measurement methods, displacement of other snacks and discretionary foods, and hunger and appetite control [
5,
6,
7,
29,
30,
31,
32,
33,
34,
35,
36,
37,
38,
39]. Although nuts are higher in ED (e.g., rich in lipids) with the potential to increase the risk of unintended body mass gain, RCTs consistently demonstrate that increased adiposity is not a risk with daily almond consumption.
Fraser et al. [
29] conducted a crossover RCT which randomly assigned 81 healthy adults (43% women, mean age 50 years, mean BMI 26) to add an average of 54 g raw or roasted almonds/day (about 320 kcals/day, 40–50 nuts) to their habitual diet or to continue their habitual diet without added almonds as a control for six months. Compared to the control diet, the almond diet did not statistically or clinically alter body mass, with an overall gain of + 0.4 kg compared to the predicted body fat mass gain of 6.4 kg based on the added almond calories (almonds provided an average of 57,500 kcals for subjects during this study). A sub-group analysis showed that almond effects on body mass varied by sex and BMI: women were better able to maintain baseline body mass than men, and baseline lower or medium tertile BMI subjects gained body mass and higher BMI individuals (>28 BMI) actually lowered their body mass to varying levels. However, according to the investigators, all the body mass changes were relatively small and probably not biologically significant. Dietary analyses from food recalls showed that 54% of the extra almond energy was displaced by reduced intake of other foods, and food diaries showed 78% displacement. Jaceldo-Siegl et al. [
30] showed that the addition of almonds to the habitual diet made for a healthier and more weight-controlled diet by significantly increasing levels of unsaturated fat, protein, fiber, magnesium, and alpha-tocopherol, and decreasing level of trans-fat, animal protein, cholesterol, sodium, and sugar.
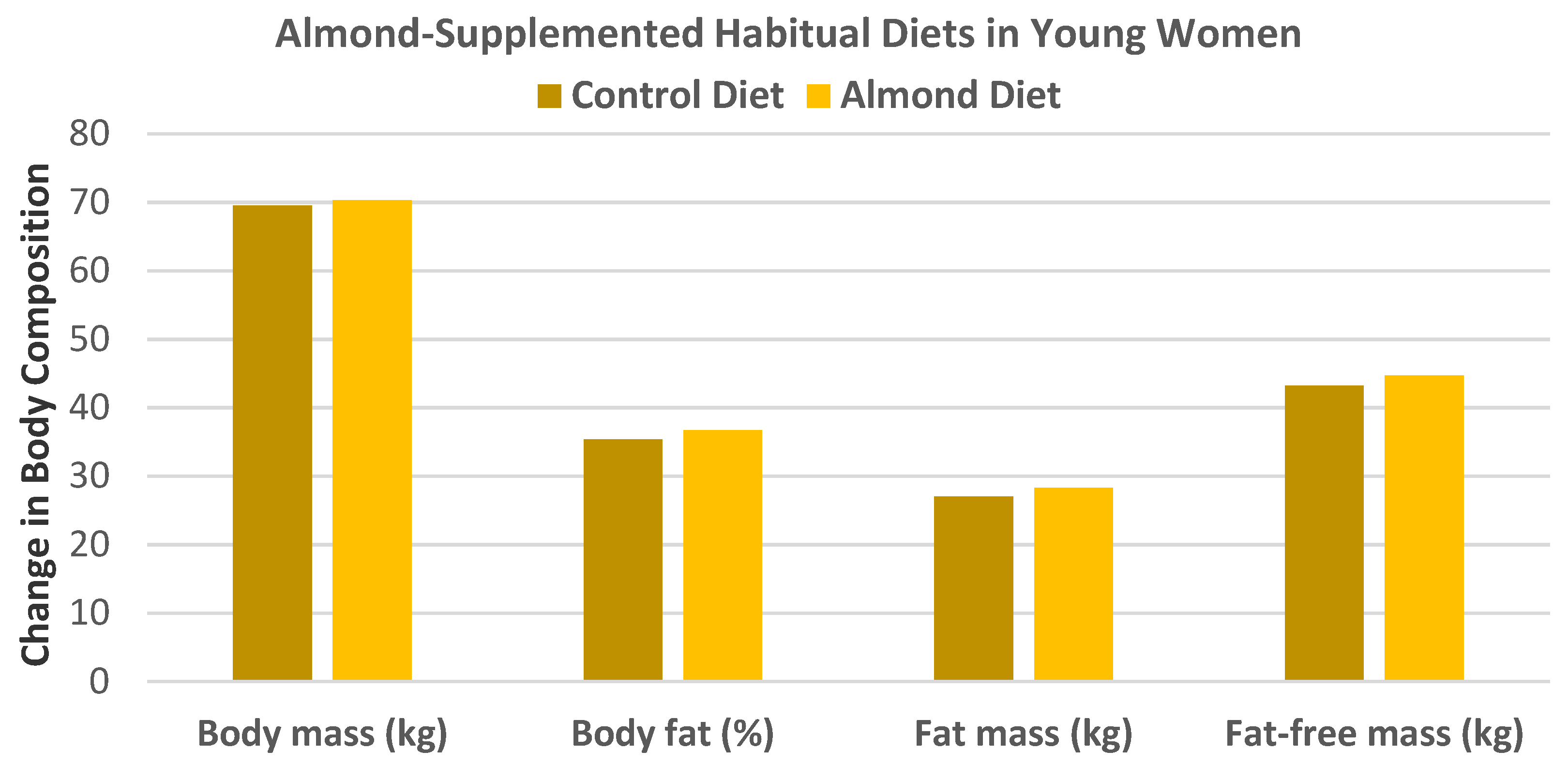
Hollis and Mattes [
31], in a crossover RCT, randomized 20 healthy women (mean age 24 years, mean BMI 26) into their habitual diet plus 60 g raw almonds/day (2.1 servings, 344 kcal) or an almond-free habitual diet for 10 weeks without any dietary advice for weight maintenance. Despite a predicted body fat gain of 3 kg, the almond-enriched diet did not significantly result in change in body mass (kg), body fat (%), fat mass (kg), fat-free mass (kg), or total food intake compared to the almond-free control diet (
p > 0.05 for all,
Figure 3). The biological processes by which added almond intake dissipated metabolizable energy (ME) included a 74% dietary energy compensation by reduced food intake from other sources, 7% increased fecal excretion of macronutrients, and 24% increased resting energy expenditure.
Lovejoy et al. [
32] conducted a clinical trial in which 20 healthy individuals (10 men and 10 women, mean age 25 years, mean BMI 23) consumed 100 g almonds/day (3.6 servings) (579 kcals) as whole almonds alone or in a variety of foods, such as trail mix, cookies, or muffins for 4 weeks. Compared to baseline, body mass increased slightly but significantly after 4 weeks of almond intake by 0.3 kg for women and by 0.9 kg for men (
p = 0.006), which was much lower than the predicted increase of about 2.1 kg of body fat based on the amount of added almond consumed.
Tan and Mattes [
33], in a parallel RCT, randomized 137 overweight or obese adults at risk for type 2 diabetes (65% women, mean BMI > 27, mean age 29 years) into 5 diets: nut-free usual diet (control), morning snack, afternoon snack (43 g almonds each), breakfast with almonds, and lunch with almonds (43 g almonds each) for 4 weeks without specific dietary guidance. There were no significant differences in body mass, body fat mass, BMI, and WC, between the almond-supplemented groups and the control group. Despite the additional 250 kcal/day from almonds, the daily energy intakes of the almond diet groups were not significantly different from the control diet group or baseline. Eating almonds either with meals or as snacks resulted in lower hunger levels at the next meal. Additionally, the consumption of almonds with breakfast was better at reducing post-meal hunger ratings than when almonds were consumed with lunch, but afternoon almond snacking was the most effective at suppressing hunger.
Sweazea et al. [
34], in a parallel RCT, randomly assigned 21 adults with type 2 diabetes (57% women, mean age 56 years, mean BMI 35) to a habitual diet with ≤2 servings of non-almond nuts (control) or a habitual diet plus 43 g (250 kcals) of almonds 5–7 times weekly for 12 weeks. Compared to the control diet, the almond diet insignificantly increased overall mean body mass by 1.2 kg (
p = 0.973), BMI by 0.4 kg/m
2 (
p = 0.973), WC by 1.3 cm (
p = 0.809), and body fat by 2.8% (
p = 0.468), which was less than the estimated increase in body mass of 6 kg predicted for the level of almond energy added to the diet over 12 weeks.
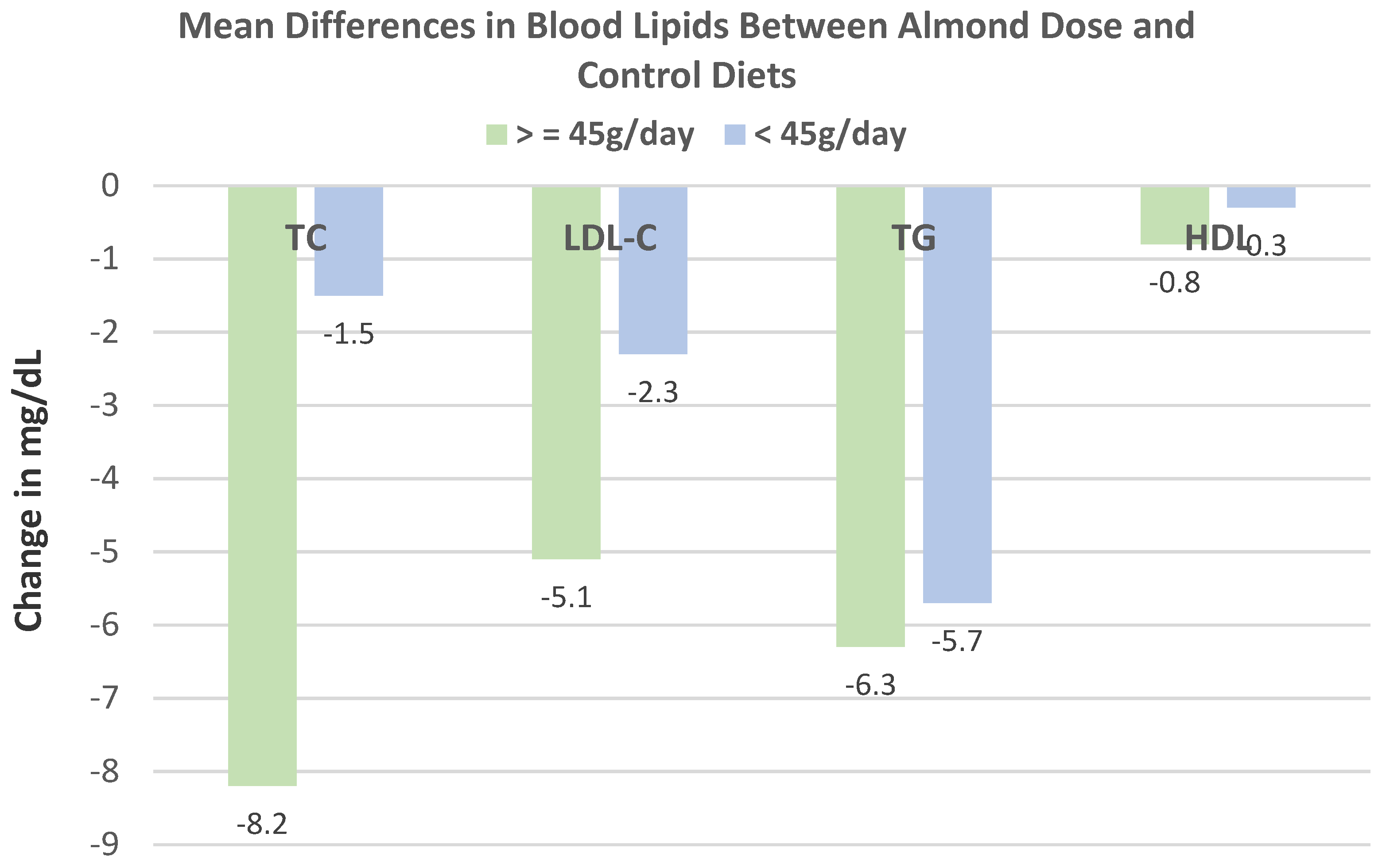
de Souza et al. [
35], in a parallel RCT, randomized 60 obese women (age 20–59 years, mean BMI 33) to habitual, isocaloric diets enriched with 20 g/day of roasted baru almonds (109 kcals) or a baru almond-free control diet for 8 weeks. Compared to control diets, baru almond-enriched diets significantly reduced WC by −2.45 cm (−3.90, −0.23 cm,
p = 0.03), but there were no significant differences between the diets for body mass, BMI, fat mass, and lean mass (
p ≤ 0.95), as shown in
Figure 4. The baru almond diet reduced abdominal adiposity in overweight/obese women compared to isocaloric control diets.
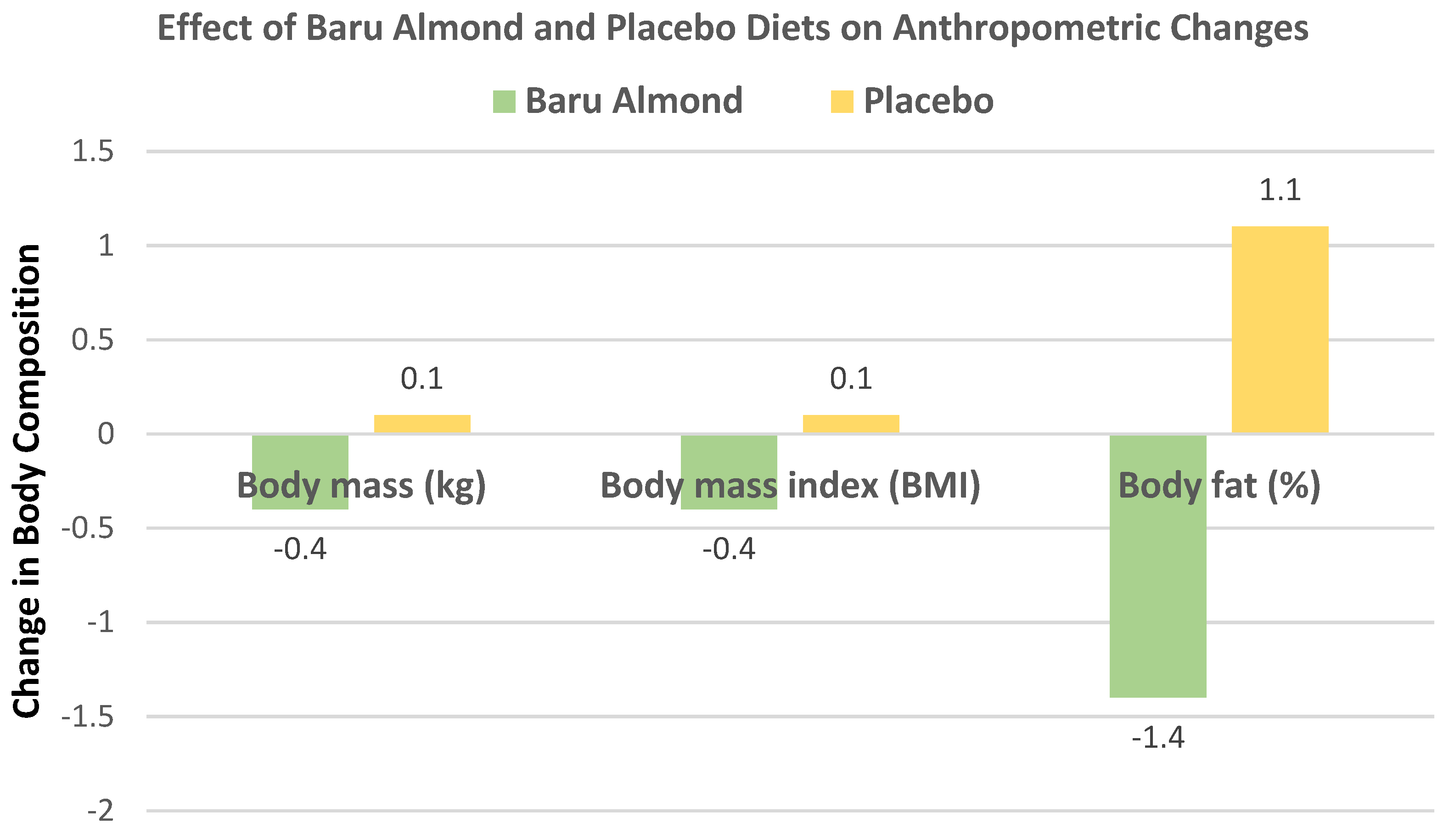
Bento et al. [
36], in a crossover RCT, randomized 20 mildly hypercholesterolemic subjects (8 men and 12 women, mean age 35 years, mean BMI 23) into their habitual diet plus 20 g (109 kcals) of baru almonds or a corn starch placebo capsule for 6 weeks. The baru almond-enriched diet tended to reduce body mass, BMI, and body fat (%) compared to the placebo (
Figure 5).
Jamshed et al. [
37], in a parallel RCT, randomized 150 adults with CHD (mean age 60 years, 25% women, mean body mass 76 kg) into a habitual diet plus 10 g of Pakistani or American almonds/day (soaked in water overnight, skin removed, and consumed before breakfast) or a control habitual diet (no added almonds or other nuts) for 12 weeks. Mean body mass was significantly reduced by −0.6 kg in almond groups compared to the control diet (
p < 0.05).
Spiller et al. [
38], in a parallel RCT, randomized 45 hyperlipidemic adults (73% women, mean age 53 years, mean body mass 66 kg) to the usual diet plus 630 kcals/day from: (1) raw whole and ground almonds (100 g/day), (2) olive oil (48 g), cottage cheese (113 g), and rye crackers (21 g), or (3) cheddar cheese (85 g), butter (28 g), and rye crackers (21 g) to match the total fat level for each diet. There were no significant differences in body mass for these diets after 4 weeks (
p > 0.05).
Cohen and Johnston [
39], in a parallel RCT, randomized 13 obese subjects with T2 diabetes (54% men, mean age 66 years, mean BMI 37) to their usual diets plus a serving of almonds (28 g/day, 164 kcals, 24 almonds) or a serving of cheese sticks (160 kcals) for 5 days per week for 12 weeks. Snacking on almonds significantly lowered BMI and tended to lower body mass compared to string cheese (
Figure 6).
Novotny et al. [
5] conducted a dose–response, crossover, controlled feeding RCT including 18 healthy adults (10 men and 8 women, mean age 56 years, mean BMI 27) who were randomized into a 3-arm typical American diet with 0.0, 42, and 84 g of whole raw almonds/day for 18 days to measure the energy density (ED) of whole almonds in the human diet. This study found that the mean ED for whole raw almonds was 4.6 ± 0.8 kcals/g or 129 kcals/serving compared to the USDA estimated Atwater ED 6.0 kcals/g or approximately 164 kcals/serving. The Atwater ED estimates when applied to whole raw almonds were overestimated by 23%. This lower ED value was primarily associated with more reduced almond fat bioavailability than that projected by the Atwater estimates. The fat digestibility was found to be decreased by 5% for 42.5 g raw almonds and by 10% for 84 g almonds (
p < 0.0001).
Gebauer et al. [
6] conducted a 5-period crossover, controlled feeding RCT in 18 healthy adults (56% men, mean age 57 years, mean body mass 89 kg) who were randomized into a typical American diet plus 42.5 g whole raw almonds, whole roasted almonds, chopped roasted almonds, or almond butter (data not shown), plus a control diet for 3 weeks to directly measure ED in kcals/g. The clinically measured EDs for whole raw almonds (4.42 kcals/g), whole roasted almonds (4.86 kcals/g), and chopped roasted almonds (5.04 kcals/g) were significantly lower than those predicted with Atwater factors, by 25%, 19%, and 17%, respectively (
p < 0.001;
Figure 7). The clinical ED of whole raw almonds was lower than that of roasted whole and roasted chopped almonds (
p < 0.05), which was due to lower hardness of whole roasted and chopped almonds compared to whole raw almonds (
p < 0.05). The harder whole raw almonds fractured into fewer, larger particles, inhibiting the release of lipids for gut bioavailability. Almond butter had an ED of 6.53 kcals/g which was basically the same as the Atwater estimated ED value of 6.62 kcals/g, because almond butter fat is readily bioavailable, and a better fit for the Atwater estimate.
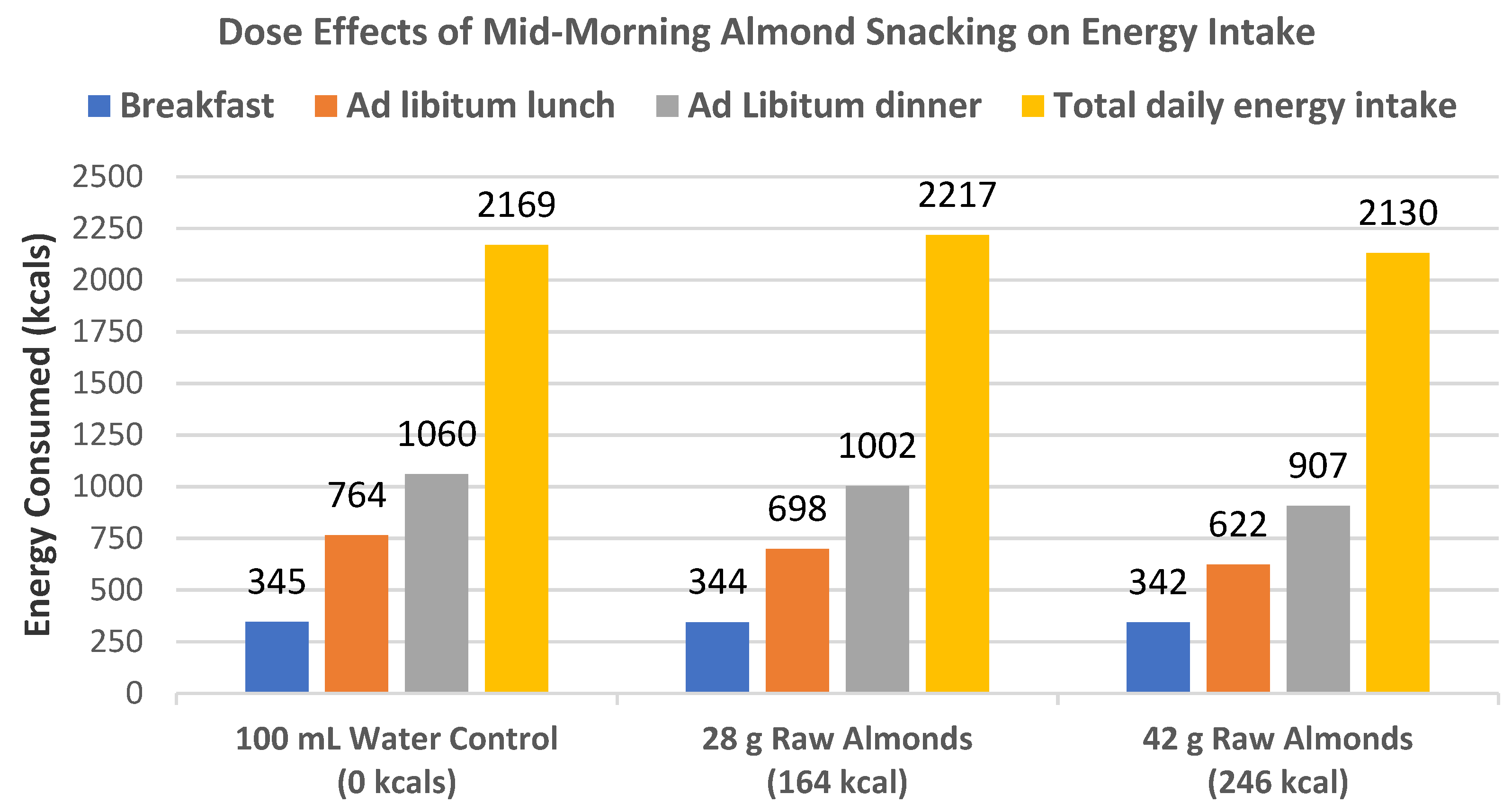
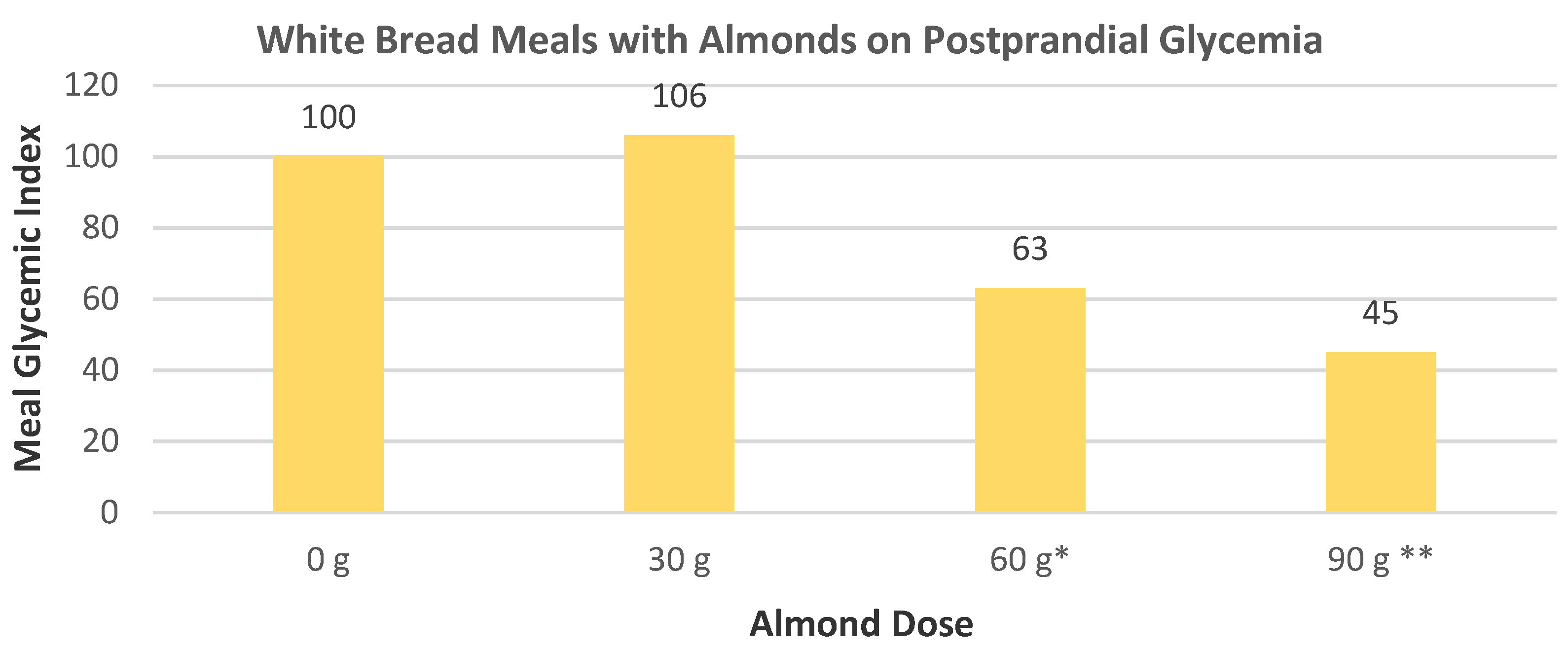
Hull et al. [
7], in a crossover RCT, randomly assigned 32 healthy women (mean age 48 years, mean BMI 23) to consume a standard breakfast followed by a mid-morning snack of a 100 mL water control and no almonds, 28, or 42 g of almonds with 100 mL of water and subsequent ad libitum meals at lunch and dinner to test for subjective appetite ratings of hunger and fullness, and both meal and total energy intake. The intake of 28 g raw almonds as a mid-morning snack significantly reduced lunch energy intake (
p = 0.016) but not dinner energy intake (
p = 0.28), and the 42 g mid-morning almond snack significantly lowered both lunch and dinner energy intake (
p ≤ 0.001) (
Figure 8). The almond snack decreased energy intake in the subsequent lunch and dinner meals in a dose–response manner for a net insignificant change of the daily total energy intake compared to the 0 g almond control. There was an almond dose-dependent increased subjective acute satiety rating for lower appetite in subsequent meals.
Ten RCTs evaluated the effects of “snacking” on 50–84 g almonds/day compared to isocaloric carbohydrate-based snacks consisting of cereal bars, cookies, crackers, mini-muffins, biscuits, potato chips, or a variety of snack choices, including low-fat potato chips, bagels, rice cakes, pretzels, vanilla cookies, dried mango slices, or pudding cups, with the usual diet in individuals ranging from 18 to >60 years for 4–20 weeks to examine changes in hunger, satiety, energy intake, and body weight and composition [
40,
41,
42,
43,
44,
45,
46,
47,
48,
49]. Snacking on almonds was more effective than snacking on savory crackers for acutely reducing the overall hunger drive, but there was no overall 24 h difference in energy intake [
40]. An almond pre-meal 18.5 g dose before each meal significantly reduced body fat mass and visceral fat compared to cookies between baseline and 8 weeks [
42]. In RCTs of 4–12 weeks duration, there were generally no or limited differences in body mass or composition measures between almond and carbohydrate-rich snacks [
41,
44,
45,
46,
47,
48,
49]. However, in RCTs of ≥16 weeks, almond snacking tended to improve both body fat and lean mass compared to high-carbohydrate control snacks [
42,
43].
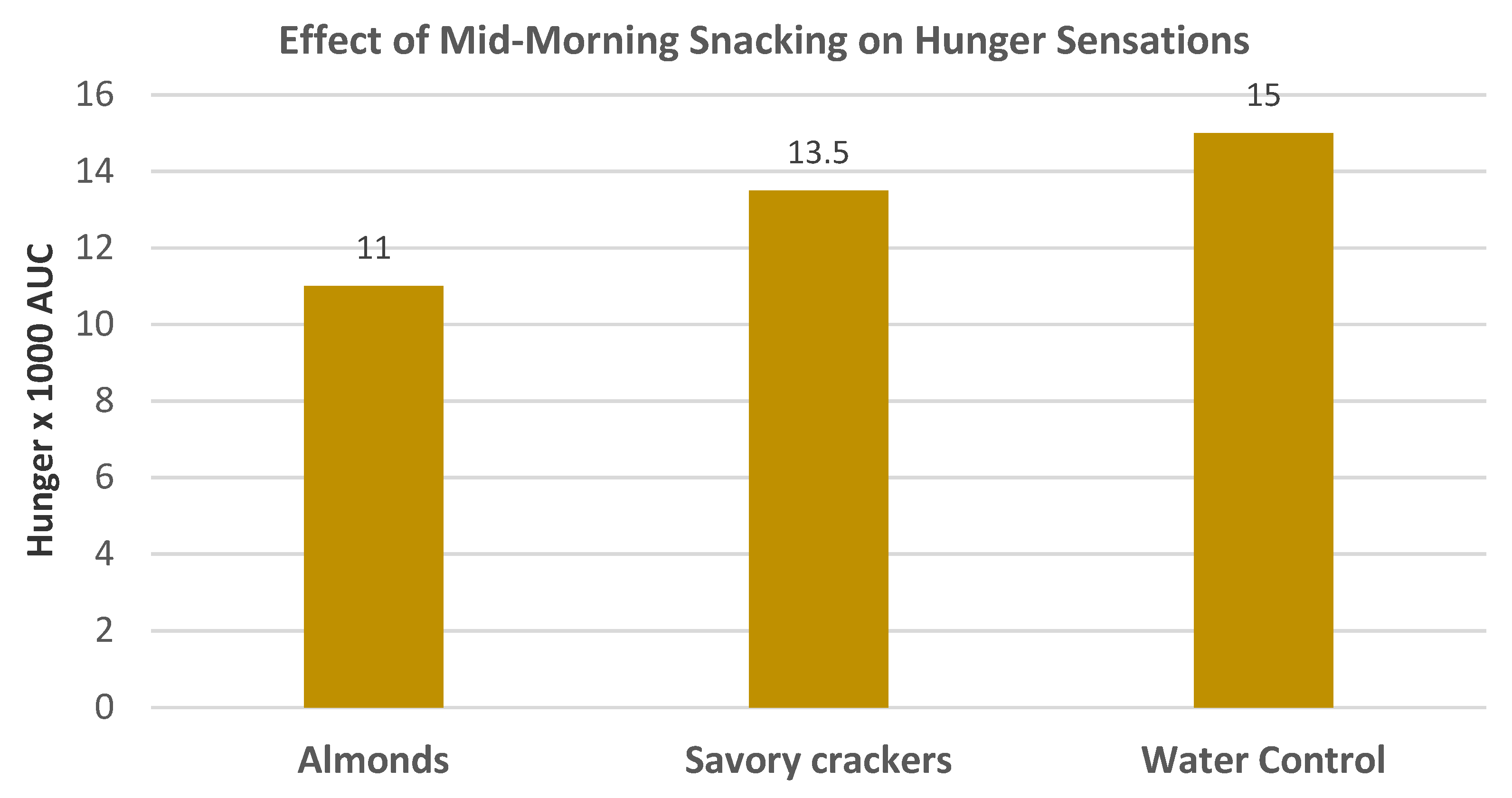
Hollingworth et al. [
40], in a crossover RCT, randomized 42 women (mean age 26 years, mean BMI 22) who consumed a standard breakfast into a water control or one of two mid-morning matched energy density snacks: raw almonds (53 g) or a savory cracker. They were evaluated for hunger ratings over 10 h. Almonds as a mid-morning snack significantly reduced hunger compared to the savory cracker and water control (
Figure 9). Additionally, almond snacking compared to the crackers reduced the desire to eat, reduced hedonic wanting for high-fat foods, and reduced prospective energy consumption. The satiating efficiency immediately after intake was greater for almonds compared to crackers (
p < 0.05). The satiety efficiency decreased over 120 min. This study showed that the addition of almonds to the diet as a mid-morning snack reduced overall hunger and reduced desire for high-fat foods compared to an energy-matched savory cracker snack.
Zaveri et al. [
41], in a parallel RCT, randomized 45 healthy men (mean age 40 years, mean BMI 30) to their habitual diet with the following added snacks: almonds (56 g, 3.9 g available carbs, 7.0 g fiber, 31 g fat, 12 g protein, 343 kcals) or cereal bar (60 g weight, 44 g available carbohydrate, 4.7 g fat, 3.0 g protein, 227 kcals) for 12 weeks. There were no significant differences in body mass, BMI, WC, or BF% between the almond and cereal bar groups but there were changes in waist–hip ratio and eating frequency. The cereal bars significantly increased the waist–hip ratio by 11% after 6 weeks and 22% after 12 weeks (
p ≤ 0.01), which could be an early indicator of metabolic syndrome risk. The almond snack significantly increased relative eating frequency from baseline by 23.5% compared to cereal bars at 12 weeks (
p ≤ 0.01) without resulting in higher energy intake or weight gain, which is an indicator of the almond effect on energy compensation via food displacement.
Liu et al. [
42], in a parallel RCT, randomized 169 healthy individuals (54% women, mean age 26 years, mean BMI 23) to the usual diet with: (1) 18.5 g almonds before each meal (pre-meal), (2) 56 g almonds as a snack consumed between meals, or (3) an isocaloric cookie (control) consumed between meals for 8–16 weeks. There were no significant differences in body mass or BMI from baseline among the three groups. The effects of almond and cookie intake on body fat measures for both 8 and 16 weeks are summarized in
Figure 10A,B. Pre-meal intake of almonds significantly reduced body fat mass (
p = 0.025) and visceral fat mass (
p = 0.000) at both 8 and 16 weeks and BF (%) after 16 weeks (
p = 0.032) compared to the cookie control. Snacking on almonds significantly reduced body fat (%) compared to the cookie control after 16 weeks (
p = 0.032) without significantly changing total body mass or BMI.
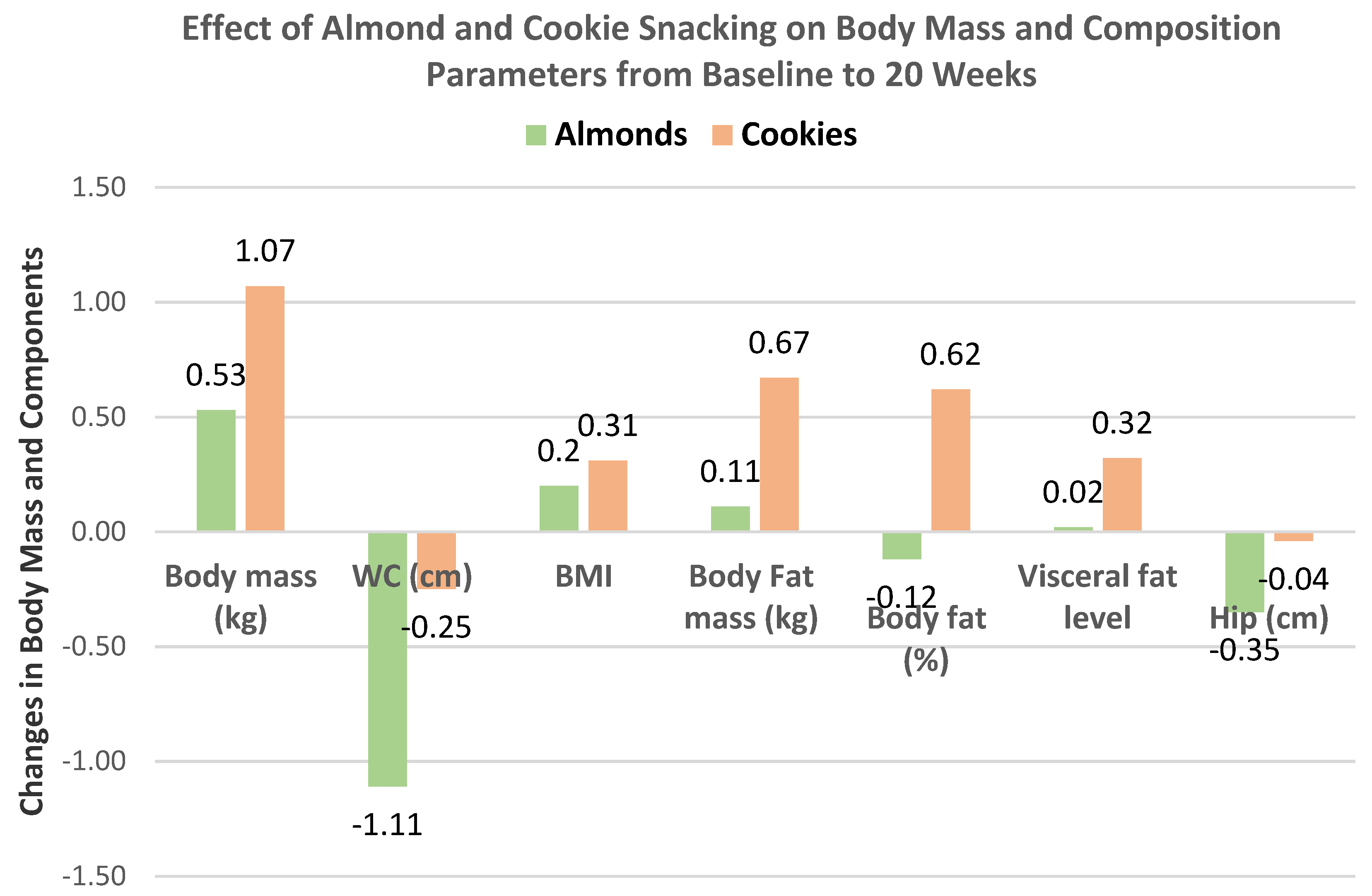
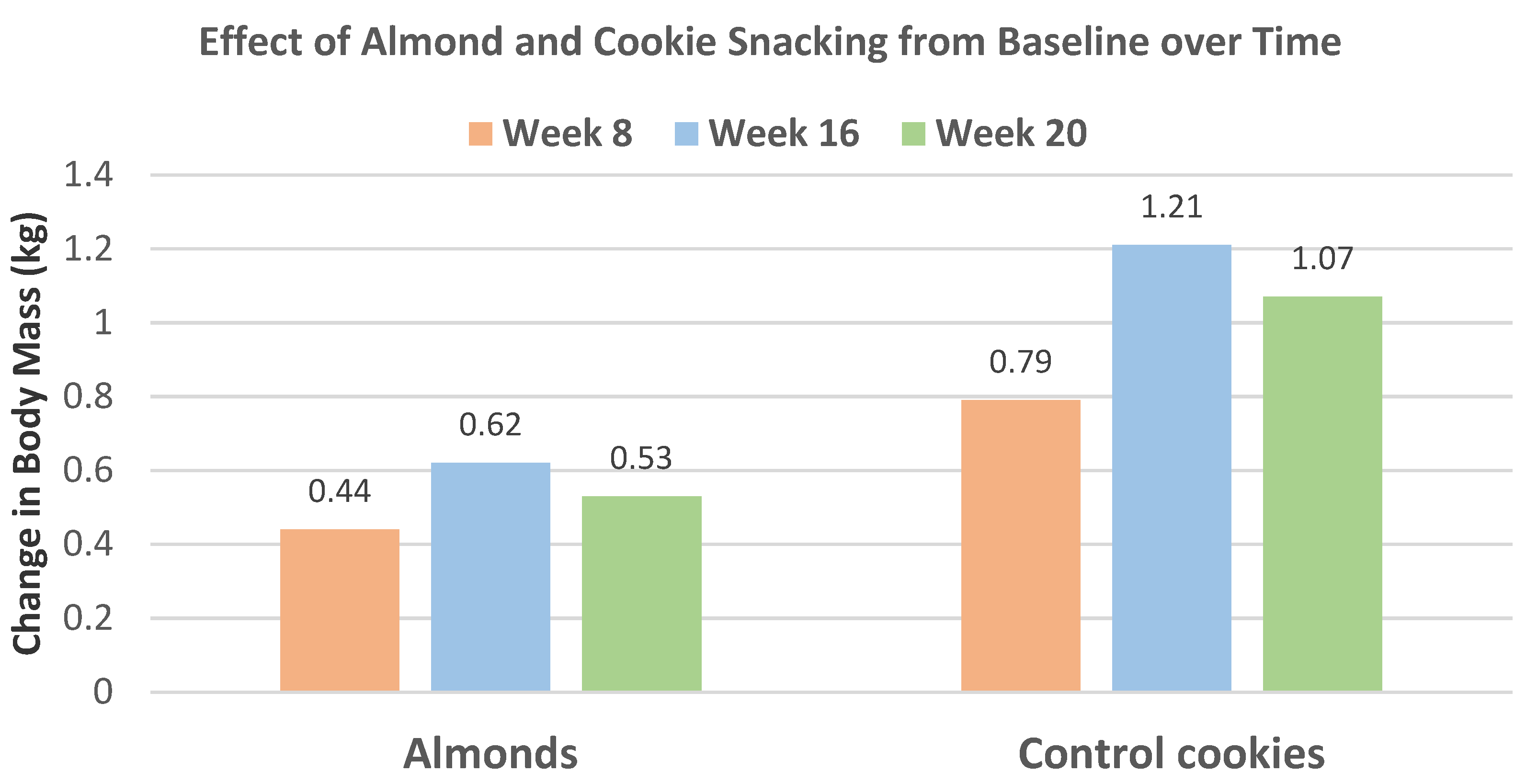
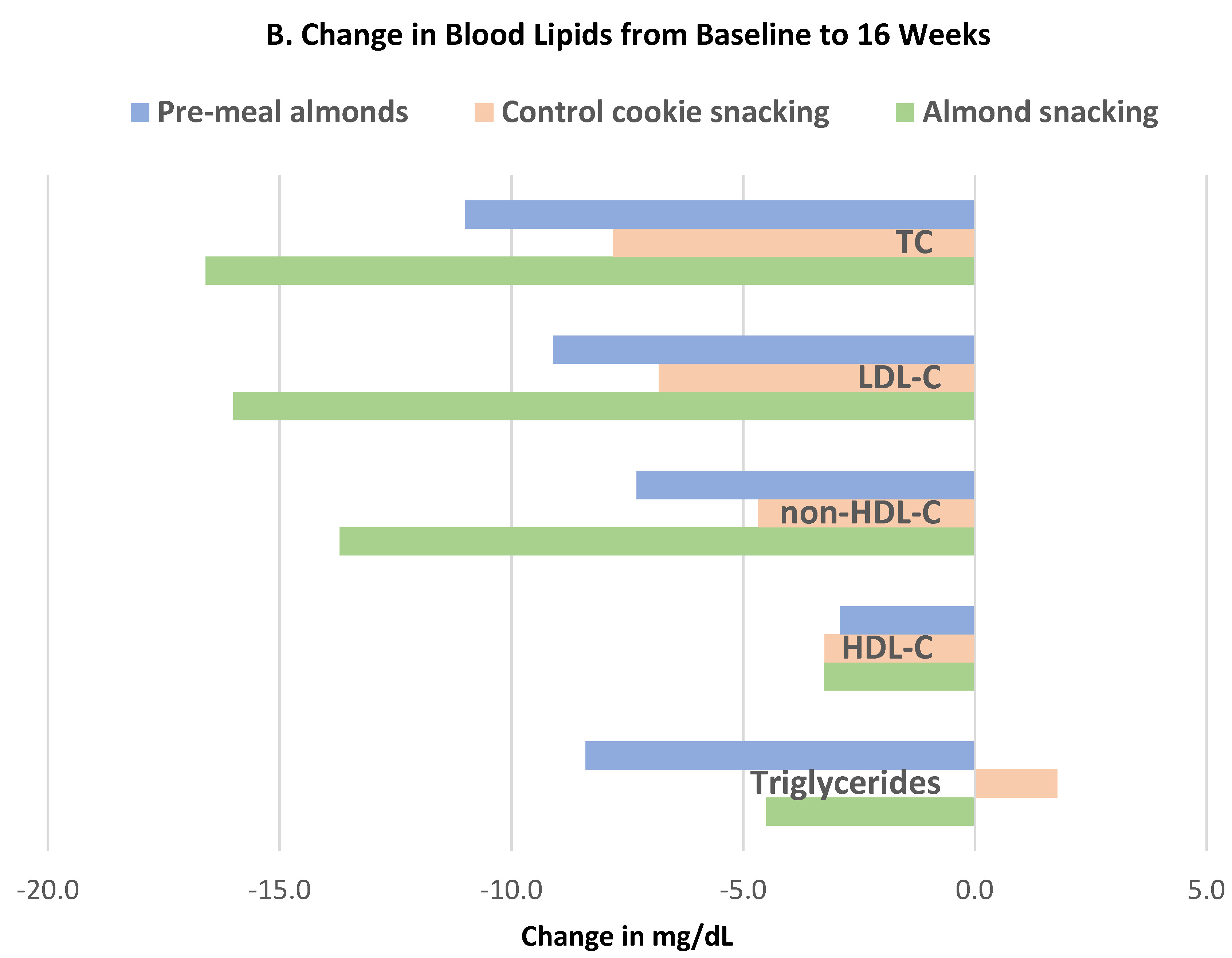
Liu et al. [
43] extended the previous trial [
42] to 20 weeks with 57 individuals from the almond (56 g) between meal snack group and 28 individuals from the isocaloric cookie control group (52% men, mean age 27 years, BMI 23). Changes in body mass and composition for almonds and cookies from baseline to week 20 are summarized in
Figure 11. Snacking on almonds for 20 weeks improved body composition in healthy young adults compared to the cookie control. The cookie control significantly increased body weight and BMI (
p < 0.001), body fat mass (
p = 0.027), and waist–hip ratio (
p = 0.035) from baseline to 20 weeks. Compared to the cookie control, almond snacking tended to decrease body mass (
p = 0.058), visceral fat level (
p = 0.056), and WC (
p = 0.064). Total body protein, fat-free mass, soft lean mass, and skeletal muscle mass were significantly increased after 20 weeks of almond intake (
p < 0.001 for all). Over 8 to 20 weeks, almond snacking tended to maintain body mass, but cookie snacking significantly increased body mass (
Figure 12) and almond snacking significantly lowered WC, while cookies significantly increased WC (
Figure 13).
Dhillon et al. [
44], in a parallel RCT, randomized 73 healthy college freshman students (mean age 18 years, 58% women, mean BMI 26) to their usual diet plus snacking on roasted almonds (57 g/day; 364 kcal) or Graham crackers (78 g/day; 338 kcal/day). Anthropometric and appetite changes were determined from baseline to 4 and 8 weeks for almond and Graham cracker snacks. Both snacks significantly increased body mass and fat-free mass from baseline to 8 weeks (
p < 0.05). Total body mass, trunk mass, trunk fat-free mass, and WC were not significantly different between baseline and week 8. For both snacks, mean 24 h hunger, desire to eat, and prospective consumption ratings significantly decreased from baseline to 4 weeks, with no difference after 8 weeks except for an increase in prospective consumption ratings. The 24 h fullness ratings did not significantly differ over the 8-week trial.
Dikariyanto et al. [
45], in a parallel RCT, randomly allocated 107 subjects with above-average risk of CVD (56 years, 70% women, mean BMI 27) to consume the typical United Kingdom diet with one of two snacks: (1) dry roasted almonds (63 g, 1/4 cup) or (2) sweet and savory mini-muffins as the control for 6 weeks, with dietary advice from a dietitian and instructions to only consume study snacks and to avoid the consumption of additional nuts or nut products during the study and maintain baseline fruit intake. There were no significant differences between the muffin control and almond snacks in BF (%), BMI, WC (cm), visceral fat volume (%), or liver fat (%). The almond group improved diet nutrition quality compared to the muffin group by reducing energy from starch by −7.0% and free sugar by −3.0%, lowering sodium by 671 mg/day, and increasing unsaturated fat: saturated fat ratio by 1.3, magnesium by 150 mg/day, vitamin E by 15 mg/day, and fiber by 7.4 g/day.
Jung et al. [
46], in a crossover RCT, randomized 84 “healthy” overweight/obese individuals (87% women, mean age 52 years, mean BMI 25) into a typical high-carbohydrate Korean diet (64% energy from carbohydrates, 22% energy from fat, and 15% energy from protein) plus daily snacking on roasted almonds (56 g, 340 kcals) or an isocaloric cookie (70 g, 340 kcals) for 4 weeks. Neither snacking on almonds nor cookies altered body or body fat mass, waist circumference (WC), or BMI, compared to baseline. However, the overall nutritional status was improved with almond snacking by decreasing % energy from carbohydrate by 14%, and increasing % energy from monounsaturated fat by 192%, polyunsaturated fat by 85%, vitamin E by 103%, and fiber by 12%. Bowen et al. [
47], in a parallel RCT, randomized 76 adults with elevated risk for or with type 2 (T2) diabetes (61 years, 59% male, mean BMI 34) to their usual diet: plus 2 servings of raw almonds (56 g/day) or 2 servings of Arnott’s biscuit snack (energy-matched) for 8 weeks, with individual advice on substituting their usual between meal snacks with their intervention snacks to daily minimize the caloric changes. There were no significant effects on body mass, fat mass, WC (cm), fat-free mass, muscle mass, visceral fat, or liver fat for either snack (
p > 0.7 for all) (
Table 3). The almond diet had 8.7% less energy from carbohydrates, 4.1% more energy from fat, 2.7% more energy from protein, and 8 g/day more fiber than the biscuit diet.
Palacios et al. [
48], in a crossover RCT, randomized 54 adults with prediabetes (48% women, mean age 48 years, mean BMI 31, 61% completers) into their usual diet plus snacks consisting of 84 g raw almonds/day (480 kcals) divided into two servings, or an isocaloric variety of carbohydrate snacks including baked chips, pretzels, dried mangos, pudding, cookies, mini-bagel, or rice cakes for 6 weeks. The subjects were instructed to use food substitution in their usual diet during each diet arm to maintain baseline energy intake. There were no significant changes from baseline in energy intake, body mass, or WC (cm) between the diets over 6 weeks (
p > 0.05). The almond diet significantly decreased % energy from sugar and total carbohydrates, and significantly increased fiber and % energy from unsaturated fat and protein compared to the control carbohydrate-rich snack diet.
Coates et al. [
49], in a parallel RCT, randomized 151 individuals (mean age 65 years, mean BMI 30) into 15% of energy snacks either from raw almonds (estimated 52 g based on a 2000 kcal diet) or isocaloric carbohydrate-rich biscuit or potato chip snacks incorporated into their habitual diets for 12 weeks. Subjects were advised to substitute almond or control snacks for discretionary foods and not to add foods to their usual diet. All subjects were evaluated every three weeks and weighed to check for weight stability. No differences in body mass, BMI, waist (cm), or body fat (%) between the two diets were observed. There was no significant difference in energy intake between the diets, but the almond diet had 20% more energy from fat, primarily from healthy unsaturated fat and a small reduction in saturated fat, lower energy from total carbohydrates and higher fiber intake, plus higher levels of vitamin E, potassium, magnesium, calcium, iron, and riboflavin.
Eleven clinical trials evaluating the effects of incorporating almonds (30–100 g/day) into healthy recommended diets for 3–12 weeks on body mass or composition found that almond-enriched diets generally supported improved body mass and composition compared to control diets [
50,
51,
52,
53,
54,
55,
56,
57,
58,
59,
60,
61].
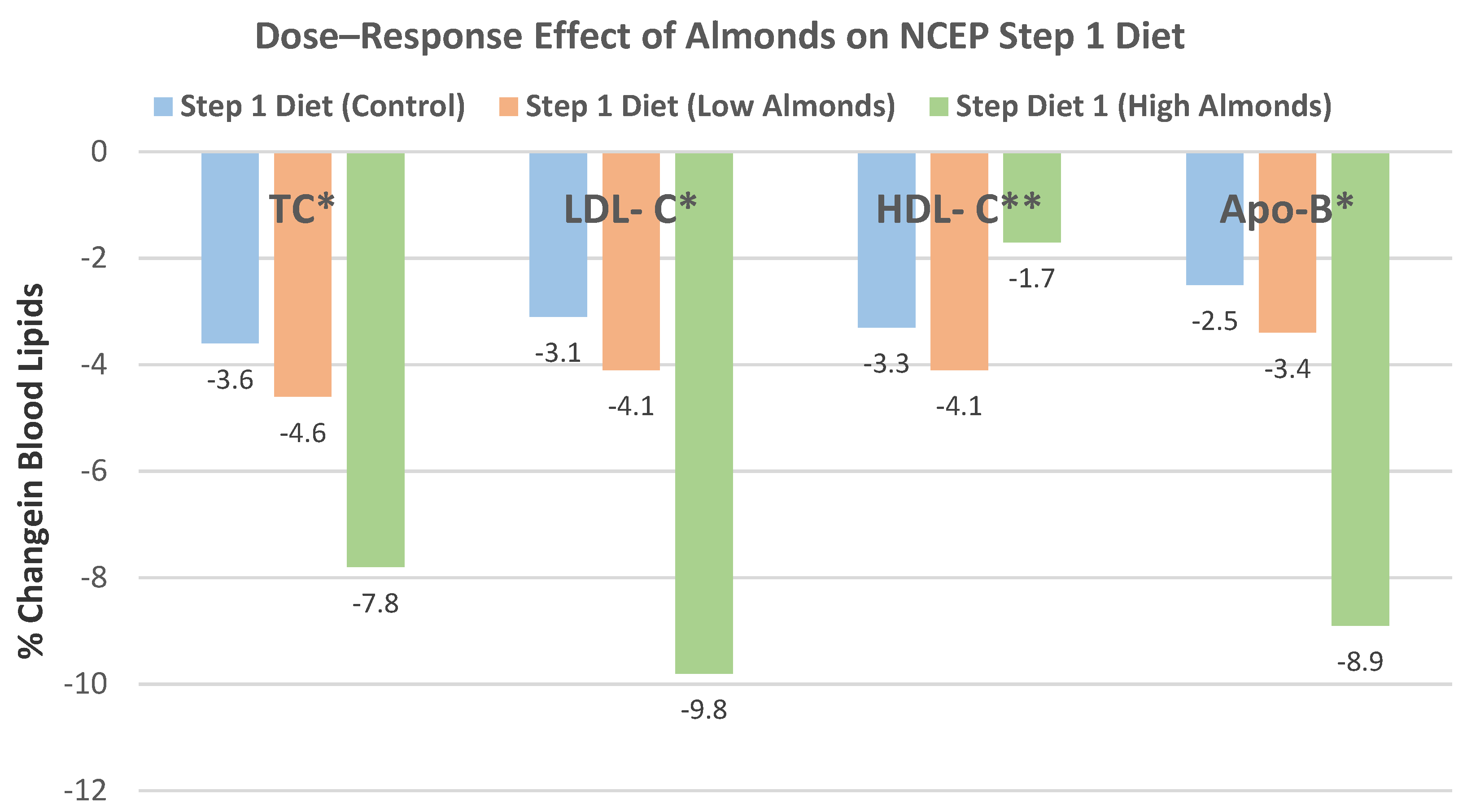
Sabate et al. [
50], in a dose–response, crossover RCT, randomly assigned 25 healthy subjects (56% men, mean age 41 years, overweight) to isocaloric National Cholesterol Education Program (NCEP) Step 1 diets containing 0%, 10%, and 20% of total energy from almonds for 4 weeks each, after a 2-week run-in Western diet. There were no significant body weight changes between the diets with the inclusion of 0%, 10%, or 20% energy from almonds (
p-trend = 0.11).
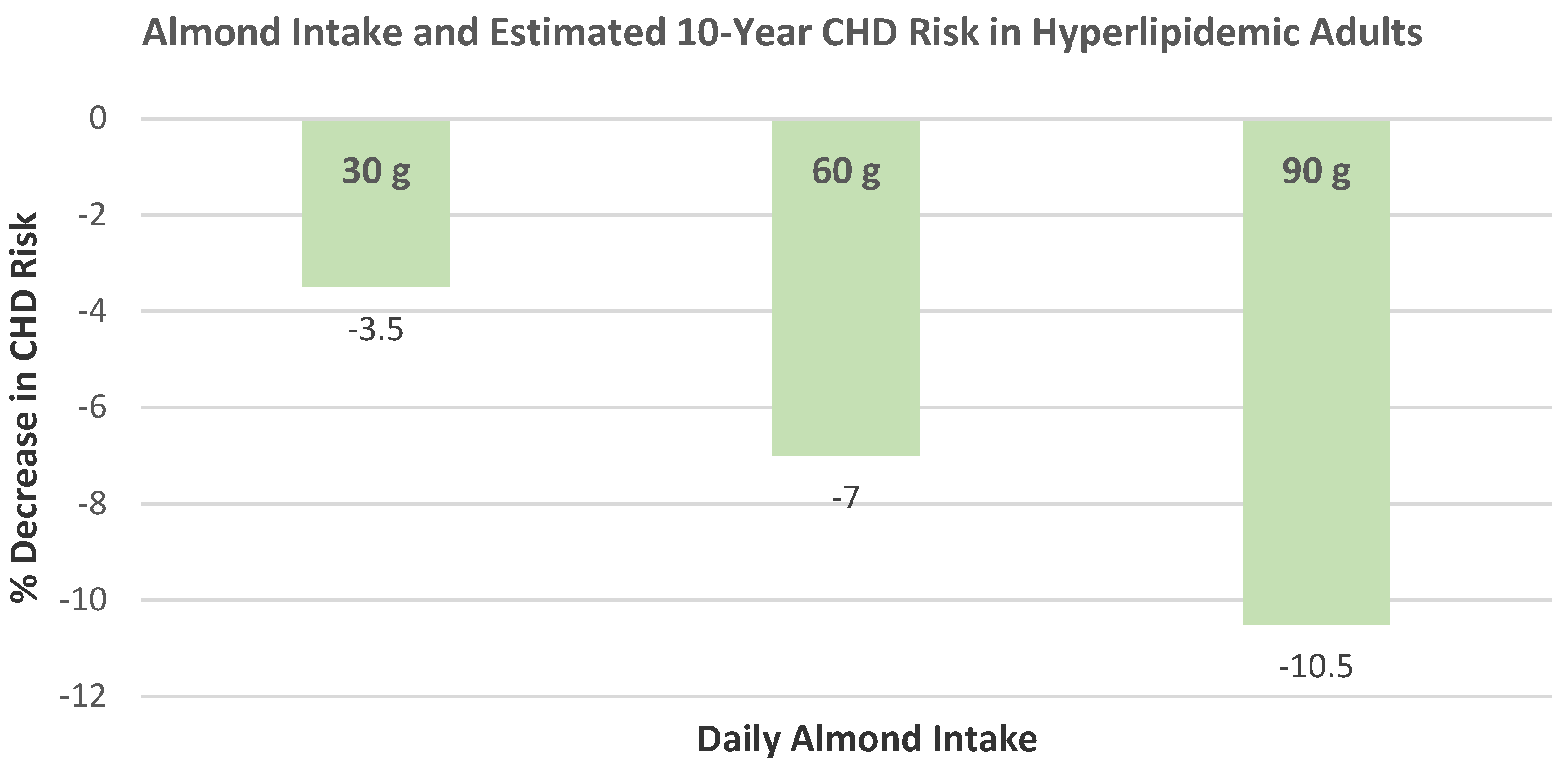
Jenkins et al. [
51,
52], in a dose–response, crossover RCT, randomized 27 overweight subjects (56% men, mean age 64 years, mean BMI 26) into NCEP Step 2 diets supplemented with 1 of 3 daily 423 kcal snacks: whole raw almonds (73 g), muffins control (147 g), or a half portion of each. Subjects were counseled on strategies to facilitate weight maintenance. The mean body weight differed by ≤300 g between all the diets from baseline to 4 weeks. The half-dose muffin and almond snack subjects lost significantly more weight than the full muffin snack (
p < 0.01).
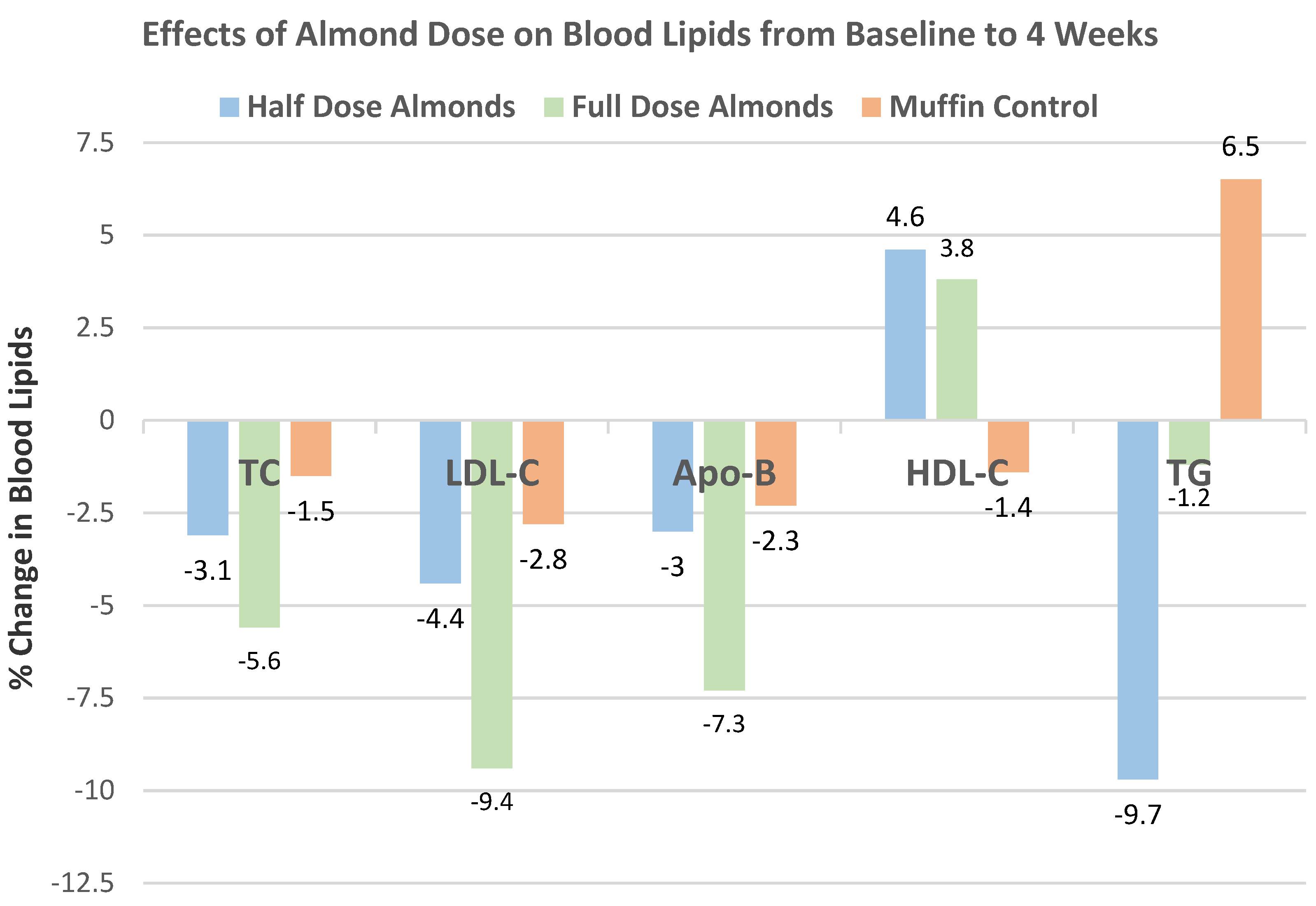
Berryman et al. [
53], in a 2-period, crossover, controlled-feeding RCT, had 48 healthy adults with mildly elevated cholesterol (54% women, mean age 50 years, mean BMI 26) consume 2100 kcal cholesterol-lowering nut-free diets with added daily snacks of raw almonds (42.5 g almonds, 250 kcals) or an isocaloric banana muffin (106 g plus 2.7 g butter, 270 kcals) for 6 weeks. Compared to the muffin control snack, the almond snack significantly reduced from baseline: WC (cm) (−0.8 cm ± 0.3), abdominal mass (−0.19 kg ± 0.08), abdominal fat mass (−0.07 kg ± 0.03), and leg fat mass (−0.12 kg ± 0.05) (
p = 0.02 for all), despite no significant body mass differences between the two diets. The % change in body weight measures from baseline to 6 weeks are summarized in
Figure 14.
Li et al. [
54], in a crossover RCT, randomized 20 adults with T2 diabetes (45% male, mean age 58 years, mean BMI 26) into two isocaloric NCEP Step 2 diets: (1) a nut-free control or (2) roasted almonds (56 g/day average), replacing 20% of the energy of the control diet for 4 weeks. Compared to the control diet group, the almond diet significantly reduced mean body fat mass by −0.8% (
p = 0.002) without altering total body mass or BMI. Compared to the control, the almond diet had 9.8% less energy from carbohydrate (
p ≤ 0.001), 9.9% more energy from fat (
p ≤ 0.001, mainly unsaturated fat), and increased fiber intake of 2.3 g/day (
p < 0.05).
Chen et al. [
55], in a crossover RCT with controlled feeding, randomized 33 patients with T2 diabetes (61% women, mean age 55 years, mean BMI 25) into two isocaloric NCEP Step 2 diets: (1) nut-free control diet or (2) roasted almond diet (60 g/day average), replacing 20% of the energy from the control diet for 12 weeks. Body mass was monitored weekly in order to adjust energy intake to maintain body mass during the study. Compared to the control diet, the almond diet insignificantly reduced mean body mass (−0.7 kg,
p = 0.241), BMI (−0.4,
p = 0.148), WC (−0.6 cm,
p = 0.477), and waist–hip ratio (−0.01,
p = 0.769). There was no significant difference in total energy intake between the diets. The almond group reduced the % energy from carbohydrates by 8.7% (
p < 0.001) and increased % energy from fat by 9.2%, primarily from monounsaturated fat (
p < 0.001), and increased fiber by 6.1 g/day (
p = 0.004).
Gulati et al. [
56], in a pre–post-clinical trial, assigned 63 adults with T2 diabetes (54% men, mean age 46 years, mean BMI 29) to a 3-week run-in diet based on the dietary guidelines for Asian Indians followed by an isocaloric modified run-in diet enriched with 20% energy from raw almonds (about 56 g/day average) for 24 weeks. There was a trend for lower body mass and BMI (
p < 0.192), and significant reductions in WC and waist–height ratio (
p ≤ 0.05) for the almond intervention in compliant subjects, which were 80% of all enrolled subjects.
Spiller et al. [
57], in a parallel RCT, randomized 38 free-living, hypercholesterolemic adults (70% women, mean age 61 years, mean weight 68 kg) into a heart-healthy diet including 100 g/day of raw almonds, roasted almonds, or almond butter for 4 weeks. The subjects were advised by nutritionists on how to substitute almonds for other foods and to keep total caloric intake similar to the baseline diet. The consumption of 100 g of almond products did not alter body weight (kg) from baseline with any of these almond sources.
Ruisinger et al. [
58], in a parallel RCT, randomized 48 hypercholesterolemic individuals receiving statins (50% men, mean age 61 years, mean BMI 29) to an NCEP Step 3 diet with 100 g raw almonds/day or a control Step 3 diet with dietary counseling for 4 weeks. The almond diet insignificantly increased body weight by 1.0 kg (
p = 0.145) and BMI by 0.3 kg/m
2 (
p = 0.262) compared to the control diet.
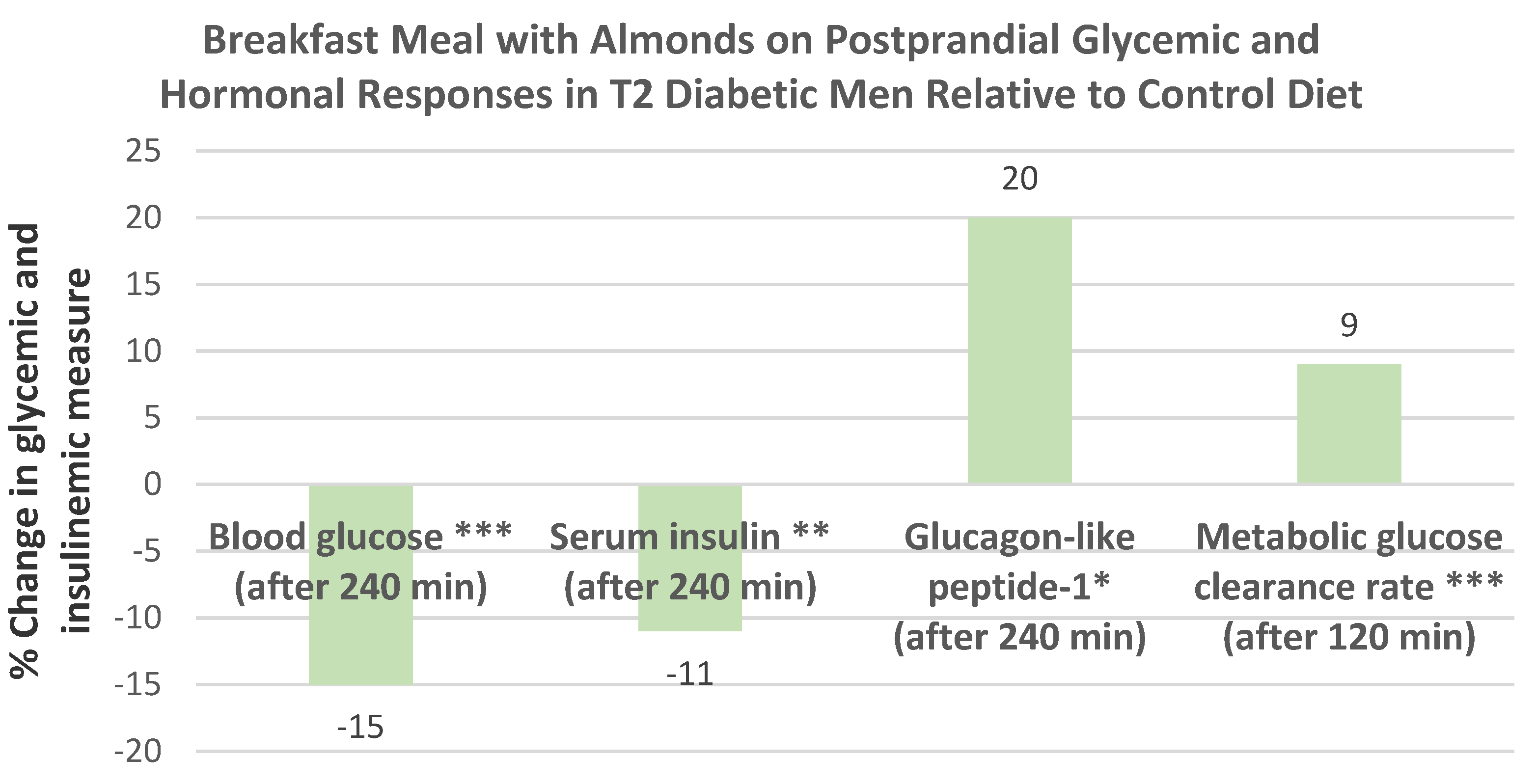
Ren et al. [
59], in a parallel RCT, randomly assigned 45 subjects with T2 diabetes (44% men, mean age 72 years, mean BMI 23) to one of two isocaloric diets: (1) an almond-based low-carbohydrate diet (56 g almonds replacing 150 g carbohydrate foods) or (2) a control low-fat (25% energy from fat) diet for 12 weeks with dietary counseling. The almond-based low-carbohydrate diet significantly reduced body weight by −7.3 kg and BMI by −0.5 (
p < 0.042) compared to the low-fat diet, which insignificantly lowered body weight by −0.5 kg and BMI by −0.16 (
p < 0.211).
Damasceno et al. [
60] conducted a crossover RCT with 18 hypercholesterolemic adults (mean age 56 years, mean BMI 26) after giving them general guidance for a Mediterranean diet. The study began with a run-in diet for 4 weeks, after which the subjects were randomized into 1 of 3 diets with healthy fat sources by replacing 40% of the run-in diet fat with: (1) virgin olive oil (35–50 g/day), (2) walnuts (40–65 g/day), or (3) raw almonds (50–75 g/day) for 4 weeks each for all subjects. There were no significant differences between the groups (
p < 0.601) in body weight as all three of these diets lowered mean body weight by −1.8 to −2.0 kg compared to the run-in diet.
Richman et al. [
61], in a crossover RCT, randomized 22 postmenopausal women with T2 diabetes (mean age 62 years, mean BMI 29) into a recommended dietary pattern for T2 diabetes with 30 g/day of almonds or sunflower seeds for 3 weeks. Compared to baseline, both almonds and sunflower seeds significantly reduced body mass and BMI (
p < 0.01), with no significant difference between them (
p > 0.540).
Six RCTs evaluated the effects of almond-based LCDs compared to nut-free control LCDs. Those trials with energy deficits of 250 to 500 kcals/day found very generally small significant or no significant differences in body mass and composition between the control and the almond LCDs [
62,
63,
64]. RCTs in the range of 1000 kcal deficits showed that almond LCDs significantly reduced body mass, BMI, body fat mass, waist, hip, and/or waist–hip ratio compared to control LCDs [
65,
66,
67].
Wien et al. [
62], in a parallel RCT, randomly assigned 65 adults with prediabetes (74% women, mean age 54 years, mean BMI 30, mean waist 96 cm) to the American Diabetes Association (ADA) diet containing 20% of energy from roasted almonds (60 g/day) or a control ADA diet without almonds for 16 weeks. This included individualized dietitian counseling to help subjects exchange added almonds for other foods or snacks with a prescribed target energy of 250–500 kcals/day deficit in subjects with BMI > 25 kg/m
2 (78% subjects). There were no significant differences in body mass, BMI, or WC between the groups during this ADA trial over 16 weeks (
p < 0.6). The authors’ post-trial analysis for the less than expected weight loss noted that it was likely due to an over-estimation of the subjects’ usual physical activity levels in the initial counseling session.
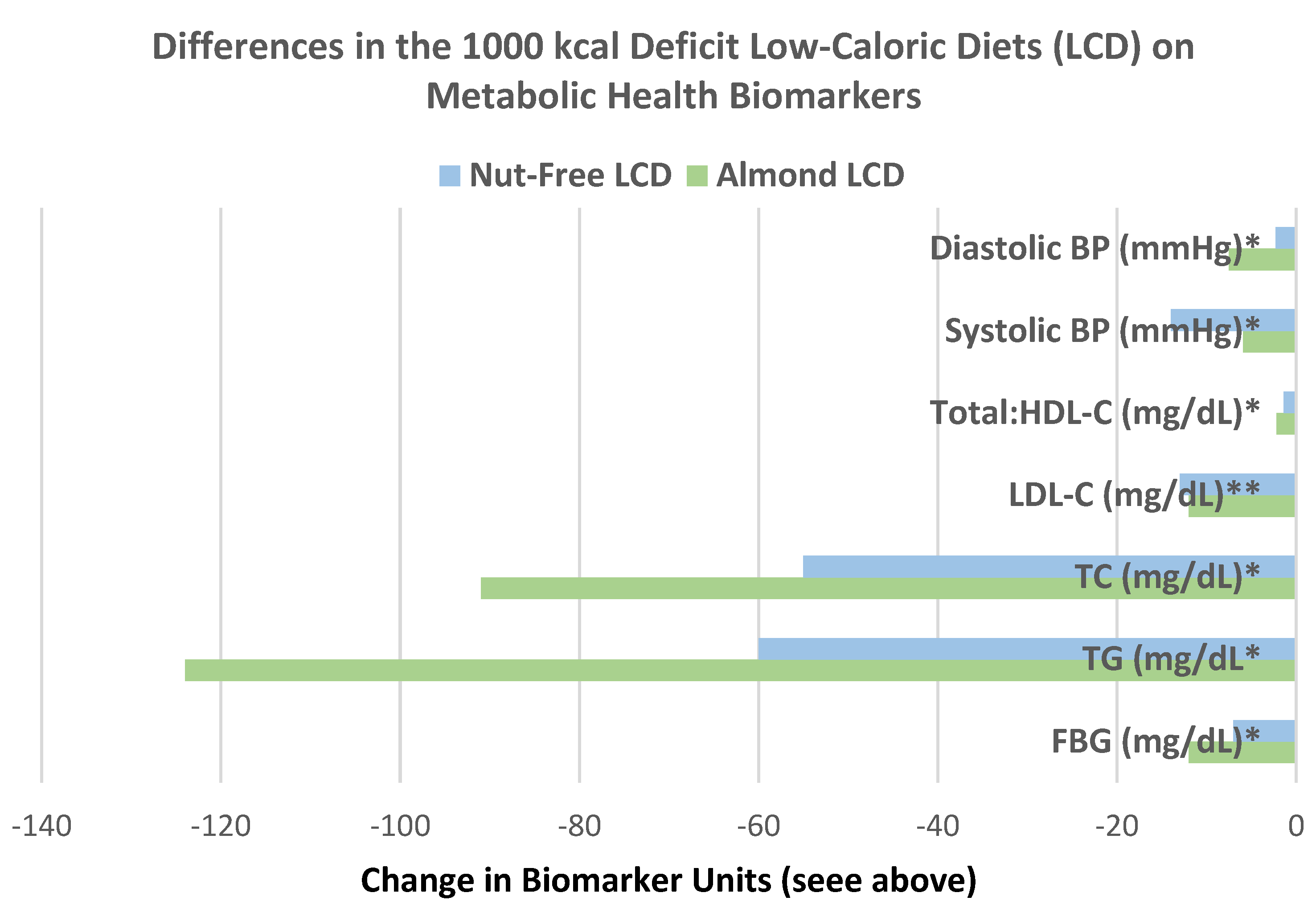
Foster et al. [
63], in a parallel RCT with 123 overweight and obese subjects (90% females, mean age 47 years, BMI 34), randomly assigned the subjects to 1 of 2 LCDs: (1) enriched with whole raw and roasted almonds (56 g/day consumed in two servings) or (2) a nut-free LCD. The targeted energy deficit was approximately 500 kcal/day (1200–1500 kcal/day for women and 1500–1800 kcal/day for men) with standard behavioral methods of hypocaloric weight loss for 18 months. The weight measures for the two LCDs are summarized in
Table 4. Those in the almond group lost slightly but significantly less body mass than did those in the nut-free group at 6 months, but there were no differences at 18 months. The loss in fat mass for the nut-free group was nearly statistically significant at 6 months, but no significant differences in changes in fat mass were found between the groups at 18 months. Although there were no significant differences in lean mass between the almond and control groups, the almond group tended to lose less lean mass by 1 kg at 18 months compared to the nut-free control group (
p = 0.09).
Dhillon, Tan, and Mattes [
64], in a parallel RCT, randomized 86 healthy adults (70% women, mean age 34 years, mean BMI 30) to 1 of 2 LCDs (targeted 500 kcal deficit/day) diets: (1) roasted almond-enriched diet (15% energy or estimated 38 g almonds/day) or a nut-free control diet for 12 weeks. Although the weight, visceral fat, WC (cm), and sagittal abdominal fat reductions did not significantly differ between the almond-enriched and control groups within either the intention-to-treat (ITT) or complier (58% of subjects) analyses, the almond group tended to lose more body mass and visceral adipose tissue (
p = 0.10) than the nut-free group in the complier analysis. Additionally, the complier analysis showed a significant decrease in percentage of total and trunk fat mass, and a significant increase in percentage of truncal and total fat-free mass in the almond-enriched diet vs. the control diet (
p < 0.05). The complier analysis more closely reflected better almond diet adherence and related efficacy than the ITT, which had a compliance rate of 65% for the almond and 67% for the control diet.
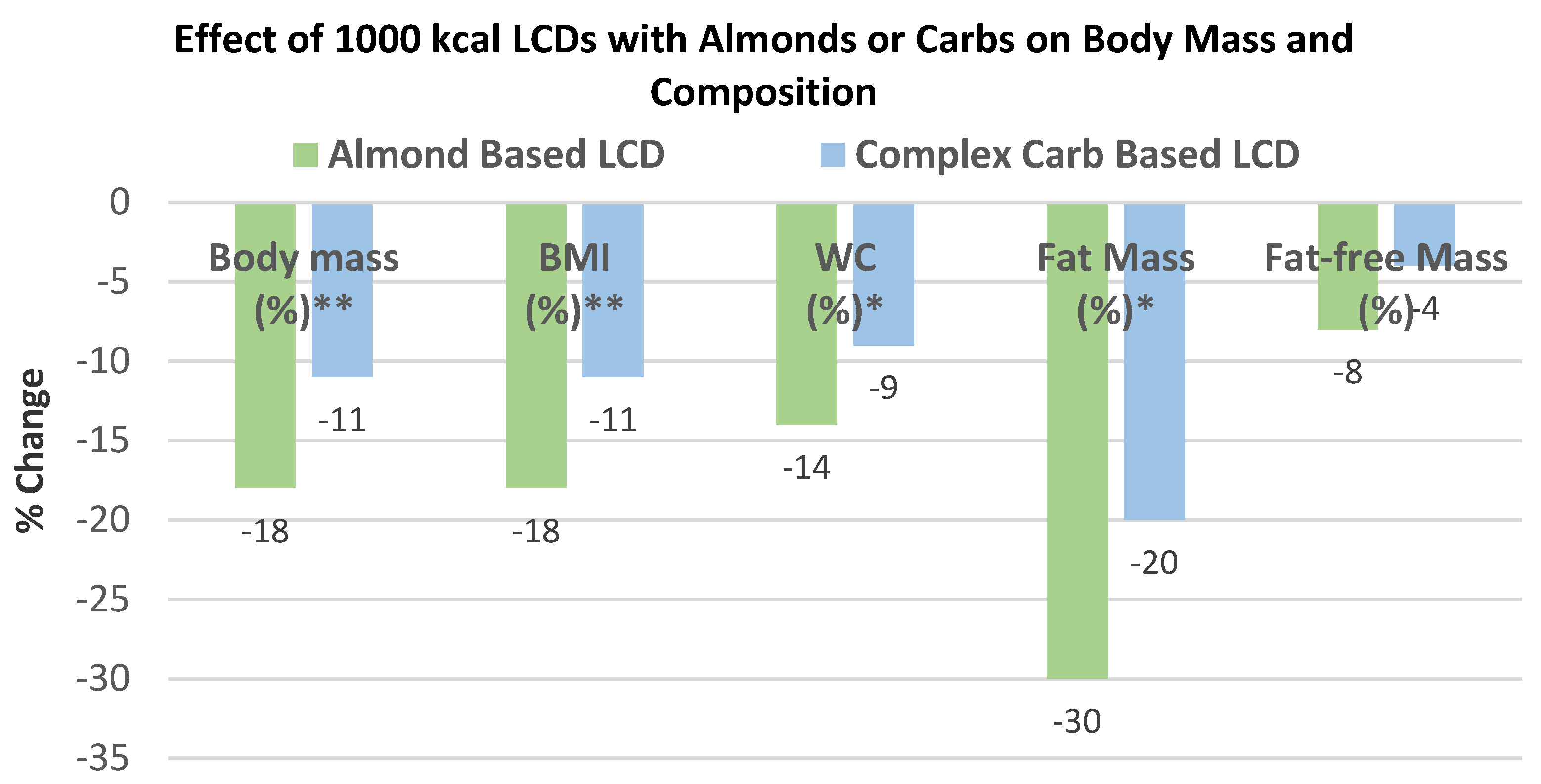
Wien et al. [
65], in a parallel weight loss RCT, randomly assigned 65 obese and metabolic syndrome middle-aged adults (57% women, mean age 55 years, mean BMI 38) into 1 of 2 diets: (1) a formula-based LCD of 1000 kcals/day, including 84 g almonds (485 kcals/day with 32% energy from carbohydrates, 29% energy from protein, 39% energy from fat, and 20 g fiber), or (2) self-selected complex carbohydrate diets, with 53% energy from carbs, 29% energy from protein, 18% energy from fat, and 32 g fiber, for 6 months under free-living conditions for 24 weeks. The almond LCD had greater % reduction in body mass and BMI (
p < 0.001), and WC and fat mass (
p < 0.05) than the carbohydrate LCD (
Figure 15). Ketone levels increased significantly in the almond LCD by 260% compared to no increase in the carbohydrate LCD (
p < 0.02). The carbohydrate group experienced a weight loss plateau at week 16, which did not occur with the almond LCD group.
Abazarfard et al. [
66], in a parallel weight loss (1000 kcals deficit) RCT, randomized 108 overweight and obese Iranian women (mean BMI 30, mean age 43 years, 93% completers) into 1 of 2 LCDs, with: (1) 50 g raw almonds/day divided into two servings or (2) a nut-free control for 12 weeks. Compared to the nut-free LCD, the almond-enriched LCD significantly lowered body mass by −2.4 kg, BMI by −1.0 kg/m
2, WC by −8.7 cm, and waist–hip ratio by −0.1 (
p < 0.001). Additionally, Abazarfard et al. [
67], in a parallel weight loss (1000 kcals deficit) RCT, showed similar results to their previous trial [
66]. They also found that the almond LCD reduced liver enzyme concentrations in obese women (
p < 0.001), which is likely associated with less accumulation of liver fat.