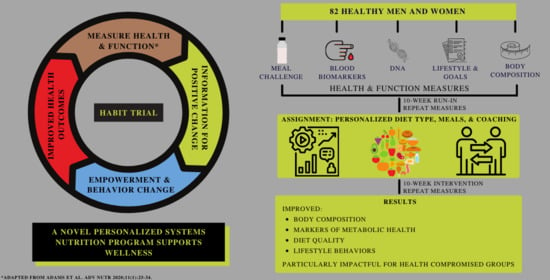
A Novel Personalized Systems Nutrition Program Improves Dietary Patterns, Lifestyle Behaviors and Health-Related Outcomes: Results from the Habit Study
Abstract
1. Introduction
2. Materials and Methods
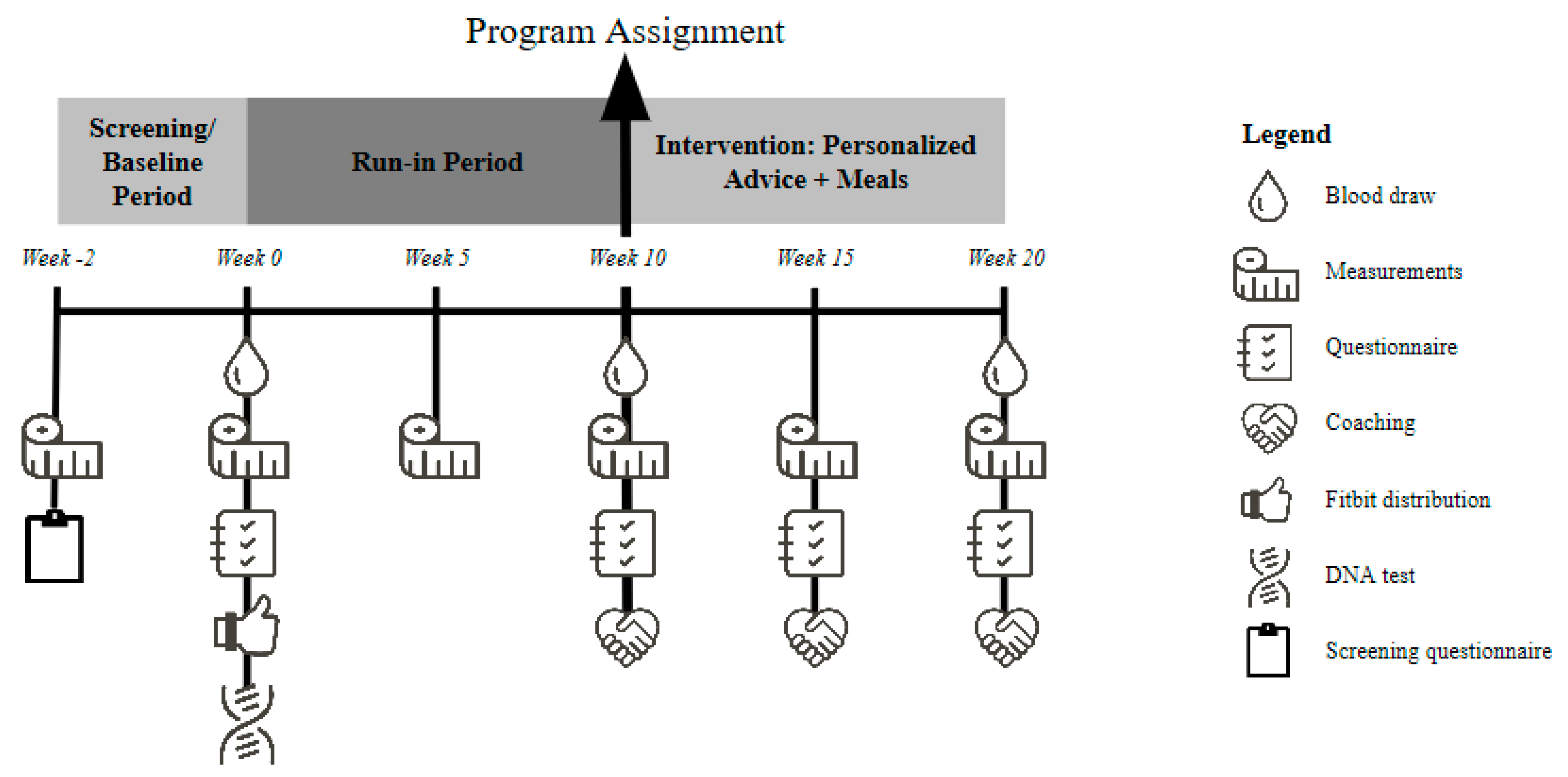
2.1. Study Design
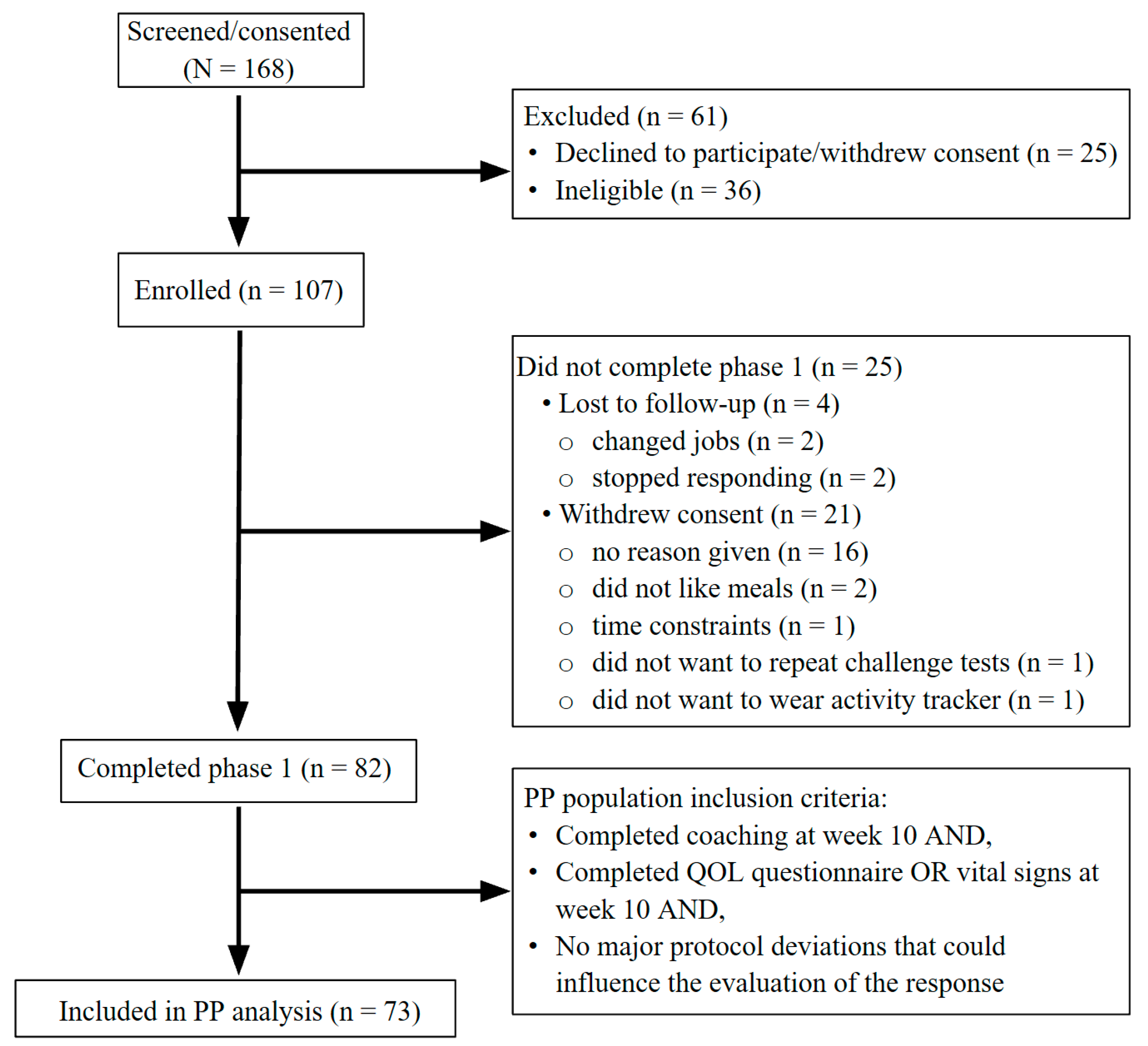
2.2. Participants
2.3. Study Procedures Overview
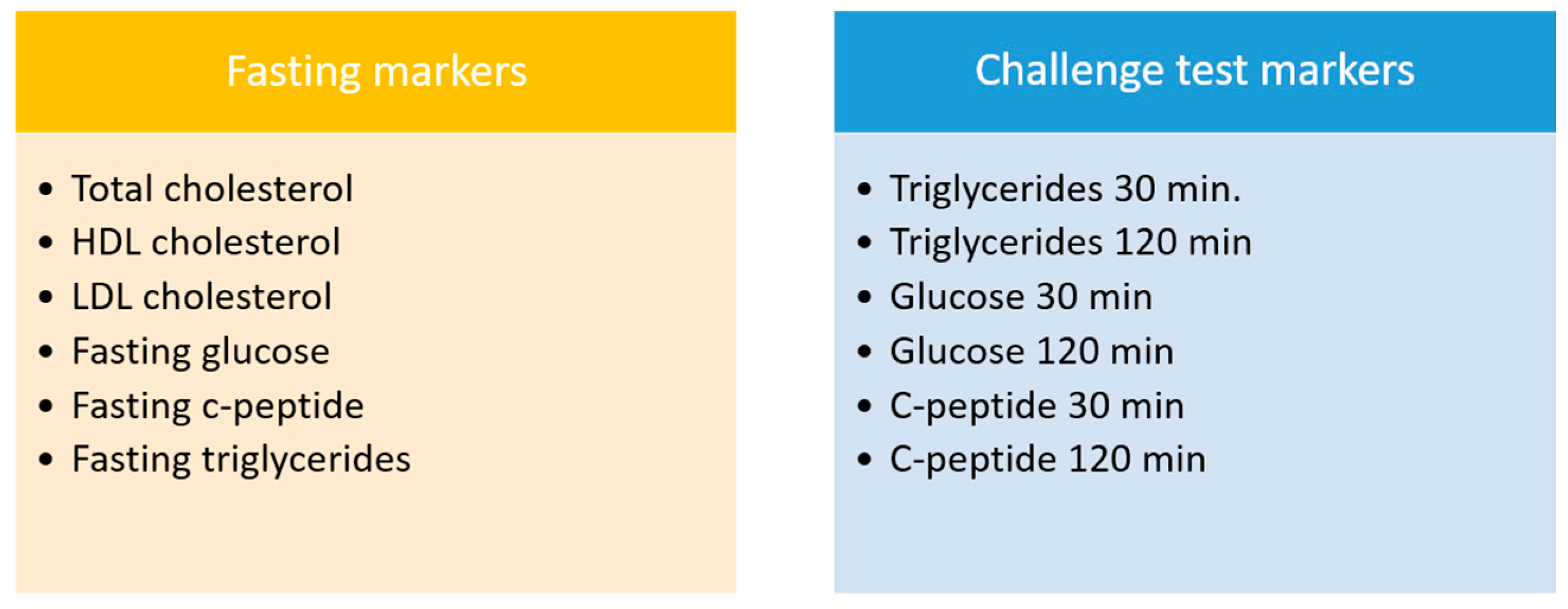
2.4. At-Home Sample Collection and Challenge Test
2.5. Personalized Systems Nutrition Program
2.6. Study Meals and Compliance
2.7. Dietary Intake
2.8. Anthropometrics and Vitals
2.9. Wellbeing and Lifestyle
2.10. Laboratory Analyses
2.11. Calculation of Insulin Sensitivity Indices
2.12. Statistical Analysis and Data Management
2.12.1. Health Space Model
2.12.2. Wellbeing Questionnaires and Activity/Sleep Data Analysis
2.12.3. Linear Mixed Model Univariate Analysis
3. Results
3.1. Study Logistics and Baseline Characteristics
3.2. Run-in Period Effects (Qualitative Control)
3.3. Intervention Effects
3.3.1. Dietary Intake
3.3.2. Wellbeing and Lifestyle
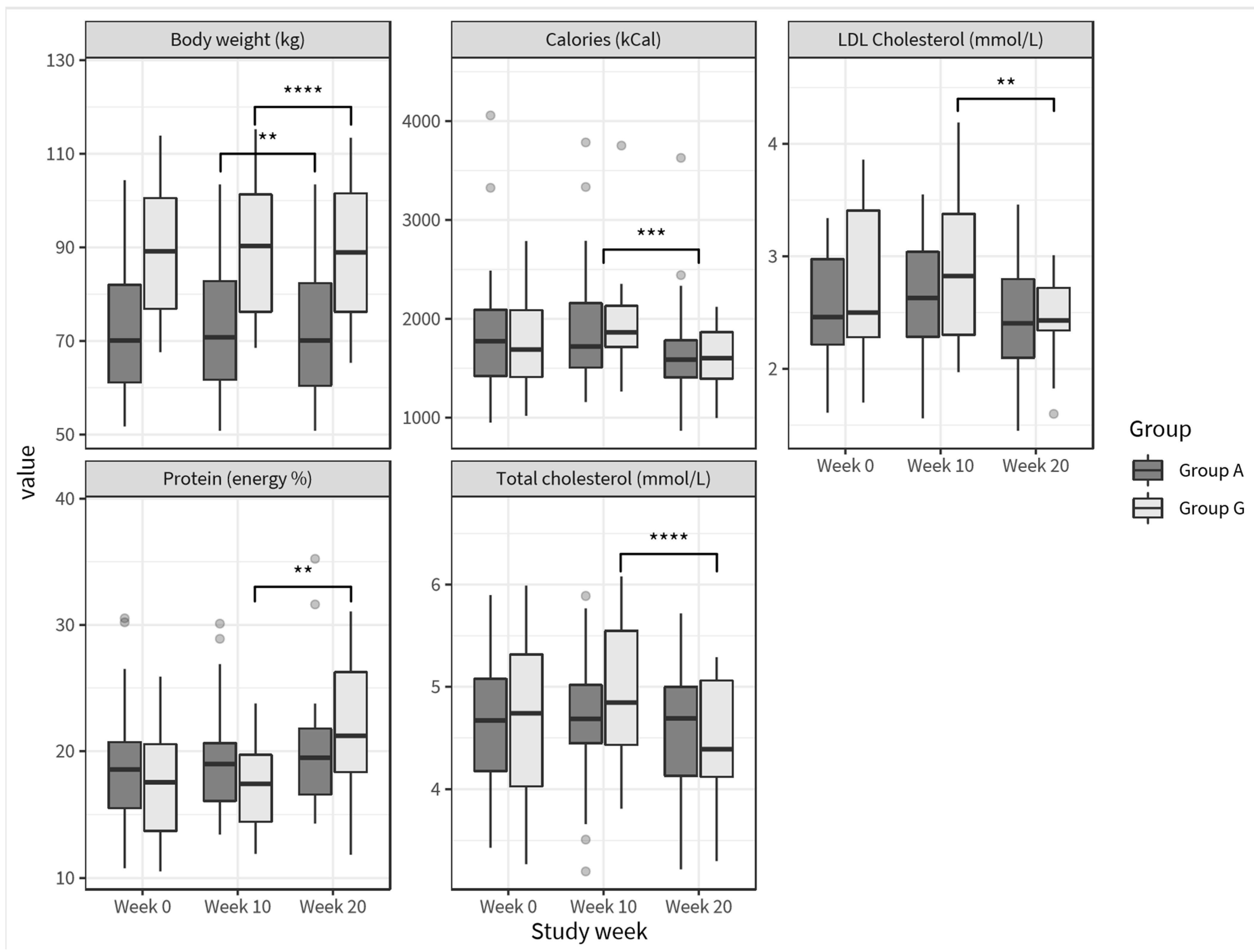
3.3.3. Anthropometrics and Vitals
3.3.4. Clinical Chemistry
3.3.5. Health Space
4. Discussion
4.1. Limitations
4.2. Strengths
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- U.S. Department of Health; Human Services; U.S. Department of Agriculture. 2015–2020 Dietary Guidelines for Americans. Available online: https://health.gov/our-work/food-nutrition/2015-2020-dietary-guidelines/guidelines/ (accessed on 29 July 2020).
- Yubero-Serrano, E.M.; Delgado-Lista, J.; Tierney, A.C.; Perez-Martinez, P.; Garcia-Rios, A.; Alcala-Diaz, J.F.; Castaño, J.P.; Tinahones, F.J.; Drevon, C.A.; Defoort, C.; et al. Insulin Resistance Determines a Differential Response to Changes in Dietary Fat Modification on Metabolic Syndrome Risk Factors: The LIPGENE Study. Am. J. Clin. Nutr. 2015, 102, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Kirwan, L.; Walsh, M.C.; Celis-Morales, C.; Marsaux, C.F.M.; Livingstone, K.M.; Navas-Carretero, S.; Fallaize, R.; O’Donovan, C.B.; Woolhead, C.; Forster, H.; et al. Phenotypic Factors Influencing the Variation in Response of Circulating Cholesterol Level to Personalised Dietary Advice in the Food4Me Study. Br. J. Nutr. 2016, 116, 2011–2019. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Rojo, R.; Alcala-Diaz, J.F.; Wopereis, S.; Perez-Martinez, P.; Quintana-Navarro, G.M.; Marin, C.; Ordovas, J.M.; van Ommen, B.; Perez-Jimenez, F.; Delgado-Lista, J.; et al. The Insulin Resistance Phenotype (Muscle or Liver) Interacts with the Type of Diet to Determine Changes in Disposition Index after 2 Years of Intervention: The CORDIOPREV-DIAB Randomised Clinical Trial. Diabetologia 2016, 59, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Kreuter, M.W.; Wray, R.J. Tailored and Targeted Health Communication: Strategies for Enhancing Information Relevance. Am. J. Health Behav. 2003, 27, S227–S232. [Google Scholar] [CrossRef]
- Stewart-Knox, B.J.; Bunting, B.P.; Gilpin, S.; Parr, H.J.; Pinhão, S.; Strain, J.J.; de Almeida, M.D.V.; Gibney, M. Attitudes toward Genetic Testing and Personalised Nutrition in a Representative Sample of European Consumers. Br. J. Nutr. 2009, 101, 982–989. [Google Scholar] [CrossRef]
- Fallaize, R.; Macready, A.L.; Butler, L.T.; Ellis, J.A.; Lovegrove, J.A. An Insight into the Public Acceptance of Nutrigenomic-Based Personalised Nutrition. Nutr. Res. Rev. 2013, 26, 39–48. [Google Scholar] [CrossRef]
- Sandercock, V.; Andrade, J. Evaluation of Worksite Wellness Nutrition and Physical Activity Programs and Their Subsequent Impact on Participants’ Body Composition. J. Obes. 2018, 2018. [Google Scholar] [CrossRef]
- Doets, E.L.; de Hoogh, I.M.; Holthuysen, N.; Wopereis, S.; Verain, M.C.D.; van den Puttelaar, J.; Hogenelst, K.; Boorsma, A.; Bouwman, E.P.; Timmer, M.; et al. Beneficial Effect of Personalized Lifestyle Advice Compared to Generic Advice on Wellbeing among Dutch Seniors–An Explorative Study. Physiol. Behav. 2019, 210, 112642. [Google Scholar] [CrossRef]
- Parekh, S.; Vandelanotte, C.; King, D.; Boyle, F.M. Improving Diet, Physical Activity and Other Lifestyle Behaviours Using Computer-Tailored Advice in General Practice: A Randomised Controlled Trial. Int. J. Behav. Nutr. Phys. Act. 2012, 9. [Google Scholar] [CrossRef] [PubMed]
- Krebs, P.; Prochaska, J.O.; Rossi, J.S. A Meta-Analysis of Computer-Tailored Interventions for Health Behavior Change. Prev. Med. 2010, 214–221. [Google Scholar] [CrossRef] [PubMed]
- Celis-Morales, C.; Lara, J.; Mathers, J.C. Personalising Nutritional Guidance for More Effective Behaviour Change. Proc. Nutr. Soc. 2015, 74, 130–138. [Google Scholar] [CrossRef]
- Adams, S.H.; Anthony, J.C.; Carvajal, R.; Chae, L.; Khoo, C.S.H.; Latulippe, M.E.; Matusheski, N.V.; McClung, H.L.; Rozga, M.; Schmid, C.H.; et al. Perspective: Guiding Principles for the Implementation of Personalized Nutrition Approaches That Benefit Health and Function. Adv. Nutr. 2020, 11, 25–34. [Google Scholar] [CrossRef]
- Van Ommen, B.; Van Den Broek, T.; De Hoogh, I.; Van Erk, M.; Van Someren, E.; Rouhani-Rankouhi, T.; Anthony, J.C.; Hogenelst, K.; Pasman, W.; Boorsma, A.; et al. Systems Biology of Personalized Nutrition. Nutr. Rev. 2017. [Google Scholar] [CrossRef]
- Bush, C.L.; Blumberg, J.B.; El-Sohemy, A.; Minich, D.M.; Ordovás, J.M.; Reed, D.G.; Behm, V.A.Y. Toward the Definition of Personalized Nutrition: A Proposal by The American Nutrition Association. J. Am. Coll. Nutr. 2020, 39, 5–15. [Google Scholar] [CrossRef] [PubMed]
- Burgess, E.; Hassmén, P.; Welvaert, M.; Pumpa, K.L. Behavioural Treatment Strategies Improve Adherence to Lifestyle Intervention Programmes in Adults with Obesity: A Systematic Review and Meta-Analysis. Clin. Obes. 2017, 7, 105–114. [Google Scholar] [CrossRef]
- Glanz, K.; Bishop, D.B. The Role of Behavioral Science Theory in Development and Implementation of Public Health Interventions. Annu. Rev. Public Health 2010, 399–418. [Google Scholar] [CrossRef]
- Brierley, M.L.; Chater, A.M.; Smith, L.R.; Bailey, D.P. The Effectiveness of Sedentary Behaviour Reduction Workplace Interventions on Cardiometabolic Risk Markers: A Systematic Review. Sports Med. 2019, 1739–1767. [Google Scholar] [CrossRef]
- Van Rhoon, L.; Byrne, M.; Morrissey, E.; Murphy, J.; McSharry, J. A Systematic Review of the Behaviour Change Techniques and Digital Features in Technology-Driven Type 2 Diabetes Prevention Interventions. Digit. Health 2020. [Google Scholar] [CrossRef]
- Szinay, D.; Jones, A.; Chadborn, T.; Brown, J.; Naughton, F. Influences on the Uptake of and Engagement with Health and Well-Being Smartphone Apps: Systematic Review. J. Med. Internet Res. 2020, 22, e17572. [Google Scholar] [CrossRef]
- Michie, S.; Abraham, C.; Whittington, C.; McAteer, J.; Gupta, S. Effective Techniques in Healthy Eating and Physical Activity Interventions: A Meta-Regression. Health Psychol. 2009, 28, 690–701. [Google Scholar] [CrossRef]
- Beishuizen, C.R.L.; Stephan, B.C.M.; Van Gool, W.A.; Brayne, C.; Peters, R.J.G.; Andrieu, S.; Kivipelto, M.; Soininen, H.; Busschers, W.B.; Van Charante, E.P.M.; et al. Web-Based Interventions Targeting Cardiovascular Risk Factors in Middle-Aged and Older People: A Systematic Review and Meta-Analysis. J. Med. Internet Res. 2016. [Google Scholar] [CrossRef]
- Fjeldsoe, B.; Neuhaus, M.; Winkler, E.; Eakin, E. Systematic Review of Maintenance of Behavior Change Following Physical Activity and Dietary Interventions. Health Psychol. 2011, 30, 99–109. [Google Scholar] [CrossRef]
- Webb, T.L.; Joseph, J.; Yardley, L.; Michie, S. Using the Internet to Promote Health Behavior Change: A Systematic Review and Meta-Analysis of the Impact of Theoretical Basis, Use of Behavior Change Techniques, and Mode of Delivery on Efficacy. J. Med. Internet Res. 2010, 12. [Google Scholar] [CrossRef] [PubMed]
- Dennison, L.; Morrison, L.; Lloyd, S.; Phillips, D.; Stuart, B.; Williams, S.; Bradbury, K.; Roderick, P.; Murray, E.; Michie, S.; et al. Does Brief Telephone Support Improve Engagement with a Web-Based Weight Management Intervention? Randomized Controlled Trial. J. Med. Internet Res. 2014, 16. [Google Scholar] [CrossRef]
- Wallace, A.M.; Bogard, M.T.; Zbikowski, S.M. Intrapersonal Variation in Goal Setting and Achievement in Health Coaching: Cross-Sectional Retrospective Analysis. J. Med. Internet Res. 2018, 20. [Google Scholar] [CrossRef] [PubMed]
- Adriaanse, M.A.; De Ridder, D.T.D.; De Wit, J.B.F. Finding the Critical Cue: Implementation Intentions to Change One’s Diet Work Best When Tailored to Personally Relevant Reasons for Unhealthy Eating. Personal. Soc. Psychol. Bull. 2009, 35, 60–71. [Google Scholar] [CrossRef]
- Prestwich, A.; Ayres, K.; Lawton, R. Crossing Two Types of Implementation Intentions with a Protection Motivation Intervention for the Reduction of Saturated Fat Intake: A Randomized Trial. Soc. Sci. Med. 2008, 67, 1550–1558. [Google Scholar] [CrossRef]
- De Vet, E.; Oenema, A.; Brug, J. More or Better: Do the Number and Specificity of Implementation Intentions Matter in Increasing Physical Activity? Psychol. Sport Exerc. 2011, 12, 471–477. [Google Scholar] [CrossRef]
- Huber, M.; André Knottnerus, J.; Green, L.; Van Der Horst, H.; Jadad, A.R.; Kromhout, D.; Leonard, B.; Lorig, K.; Loureiro, M.I.; Van Der Meer, J.W.M.; et al. How Should We Define Health? BMJ 2011, 343. [Google Scholar] [CrossRef]
- The Lancet. What Is Health? The Ability to Adapt. Lancet 2009, 781. [Google Scholar] [CrossRef]
- Van Ommen, B.; van der Greef, J.; Ordovas, J.M.; Daniel, H. Phenotypic Flexibility as Key Factor in the Human Nutrition and Health Relationship. Genes Nutr. 2014. [Google Scholar] [CrossRef] [PubMed]
- Blaak, E.E.; Hul, G.; Verdich, C.; Stich, V.; Martinez, A.; Petersen, M.; Feskens, E.F.M.; Patel, K.; Oppert, J.M.; Barbe, P.; et al. Fat Oxidation before and after a High Fat Load in the Obese Insulin-Resistant State. J. Clin. Endocrinol. Metab. 2006, 91, 1462–1469. [Google Scholar] [CrossRef] [PubMed]
- Van Ommen, B.; Keijer, J.; Heil, S.G.; Kaput, J. Challenging Homeostasis to Define Biomarkers for Nutrition Related Health. Mol. Nutr. Food Res. 2009, 795–804. [Google Scholar] [CrossRef]
- Stroeve, J.H.M.; van Wietmarschen, H.; Kremer, B.H.A.; van Ommen, B.; Wopereis, S. Phenotypic Flexibility as a Measure of Health: The Optimal Nutritional Stress Response Test. Genes Nutr. 2015, 10. [Google Scholar] [CrossRef]
- Kardinaal, A.F.M.; Van Erk, M.J.; Dutman, A.E.; Stroeve, J.H.M.; Van De Steeg, E.; Bijlsma, S.; Kooistra, T.; Van Ommen, B.; Wopereis, S. Quantifying Phenotypic Flexibility as the Response to a High-Fat Challenge Test in Different States of Metabolic Health. FASEB J. 2015, 29, 4600–4613. [Google Scholar] [CrossRef] [PubMed]
- Hoevenaars, F.P.; Esser, D.; Schutte, S.; Priebe, M.G.; Vonk, R.J.; van den Brink, W.J.; van der Kamp, J.-W.; Stroeve, J.H.; Afman, L.A.; Wopereis, S. Whole Grain Wheat Consumption Affects Postprandial Inflammatory Response in a Randomized Controlled Trial in Overweight and Obese Adults with Mild Hypercholesterolemia in the Graandioos S. J. Nutr. 2019, 149, 2133–2144. [Google Scholar] [CrossRef]
- Kalache, A.; de Hoogh, I.M.; Howlett, S.E.; Kennedy, B.; Eggersdorfer, M.; Marsman, D.S.; Shao, A.; Griffiths, J.C. Nutrition Interventions for Healthy Ageing across the Lifespan: A Conference Report. Eur. J. Nutr. 2019, 58 (Suppl. S1). [Google Scholar] [CrossRef]
- Bouwman, J.; Vogels, J.T.; Wopereis, S.; Rubingh, C.M.; Bijlsma, S.; Van Ommen, B. Visualization and Identification of Health Space, Based on Personalized Molecular Phenotype and Treatment Response to Relevant Underlying Biological Processes. BMC Med. Genomics 2012, 5. [Google Scholar] [CrossRef]
- Van den Broek, T.J.; Bakker, G.C.M.; Rubingh, C.M.; Bijlsma, S.; Stroeve, J.H.M.; van Ommen, B.; van Erk, M.J.; Wopereis, S. Ranges of Phenotypic Flexibility in Healthy Subjects. Genes Nutr. 2017, 12. [Google Scholar] [CrossRef] [PubMed]
- World Medical Association Declaration of Helsinki: Ethical Principles for Medical Research Involving Human Subjects. JAMA 2013, 2191–2194. [CrossRef]
- World Health Organization. Waist Circumference and Waist-Hip Ratio: Report of a WHO Expert Consultation; WHO: Geneva, Switzerland, 2008. [Google Scholar]
- Winters, B.L.; Anthony, J.C.; Remensperger, A.; Boorsma, A.; Vandenbroek, T.; Maki, K. Validation of a Macronutrient Mixed Challenge Beverage for Personalized Nutrition Applications Using a Reduced Sampling Period. FASEB J. 2017, 31, lb289. [Google Scholar] [CrossRef]
- Grimaldi, K.A.; van Ommen, B.; Ordovas, J.M.; Parnell, L.D.; Mathers, J.C.; Bendik, I.; Brennan, L.; Celis-Morales, C.; Cirillo, E.; Daniel, H.; et al. Proposed Guidelines to Evaluate Scientific Validity and Evidence for Genotype-Based Dietary Advice. Genes Nutr. 2017, 35. [Google Scholar] [CrossRef] [PubMed]
- Institute of Medicine. Dietary Reference Intakes for Energy, Carbohydrate, Fiber, Fat, Fatty Acids, Cholesterol, Protein, and Amino Acids (Macronutrients); National Academies Press: Washington, DC, USA, 2005. [Google Scholar] [CrossRef]
- De Luis, D.A.; Aller, R.; Izaola, O.; Primo, D.; Urdiales, S.; Romero, E. Effects of a High-Protein/Low-Carbohydrate Diet versus a Standard Hypocaloric Diet on Weight and Cardiovascular Risk Factors: Role of a Genetic Variation in the Rs9939609 FTO Gene Variant. J. Nutrigenet. Nutr. 2015, 8, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Qi, Q.; Li, Y.; Hu, F.B.; Bray, G.A.; Sacks, F.M.; Williamson, D.A.; Qi, L. FTO Genotype, Dietary Protein, and Change in Appetite: The Preventing Overweight Using Novel Dietary Strategies Trial. Am. J. Clin. Nutr. 2014, 99, 1126–1130. [Google Scholar] [CrossRef] [PubMed]
- Sonestedt, E.; Roos, C.; Gullberg, B.; Ericson, U.; Wirfält, E.; Orho-Melander, M. Fat and Carbohydrate Intake Modify the Association between Genetic Variation in the FTO Genotype and Obesity. Am. J. Clin. Nutr. 2009, 90, 1418–1425. [Google Scholar] [CrossRef]
- Mifflin, M.D.; St Jeor, S.T.; Hill, L.A.; Scott, B.J.; Daugherty, S.A.; Koh, Y.O. A New Predictive Equation for Resting Energy Expenditure in Healthy Individuals. Am. J. Clin. Nutr. 1990, 51, 241–247. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Sacks, G.; Chandramohan, D.; Chow, C.C.; Wang, Y.C.; Gortmaker, S.L.; Swinburn, B.A. Quantification of the Effect of Energy Imbalance on Bodyweight. Lancet 2011, 826–837. [Google Scholar] [CrossRef]
- Ainsworth, B.E.; Haskell, W.L.; Herrmann, S.D.; Meckes, N.; Bassett, D.R.; Tudor-Locke, C.; Greer, J.L.; Vezina, J.; Whitt-Glover, M.C.; Leon, A.S. Compendium of Physical Activities: A Second Update of Codes and MET Values. Med. Sci. Sports Exerc. 2011, 43, 1575–1581. [Google Scholar] [CrossRef]
- Horigan, G.; McNulty, H.; Ward, M.; Strain, J.J.J.; Purvis, J.; Scott, J.M. Riboflavin Lowers Blood Pressure in Cardiovascular Disease Patients Homozygous for the 677C→T Polymorphism in MTHFR. J. Hypertens. 2010, 28, 478–486. [Google Scholar] [CrossRef]
- Wilson, C.P.; McNulty, H.; Ward, M.; Strain, J.J.; Trouton, T.G.; Hoeft, B.A.; Weber, P.; Roos, F.F.; Horigan, G.; McAnena, L.; et al. Blood Pressure in Treated Hypertensive Individuals with the Mthfr 677tt Genotype Is Responsive to Intervention with Riboflavin: Findings of a Targeted Randomized Trial. Hypertension 2013, 61, 1302–1308. [Google Scholar] [CrossRef]
- McNulty, H.; Strain, J.J.; Hughes, C.F.; Ward, M. Riboflavin, MTHFR Genotype and Blood Pressure: A Personalized Approach to Prevention and Treatment of Hypertension. Mol. Aspects Med. 2017, 53, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Wilson, C.P.; Ward, M.; McNulty, H.; Strain, J.J.; Trouton, T.G.; Horigan, G.; Purvis, J.; Scott, J.M. Riboflavin Offers a Targeted Strategy for Managing Hypertension in Patients with the MTHFR 677TT Genotype: A 4-y Follow-Up. Am. J. Clin. Nutr. 2012, 95, 766–772. [Google Scholar] [CrossRef] [PubMed]
- European Food Safety Authority. Scientific Opinion on the Substantiation of Health Claims Related to EPA, DHA, DPA and Maintenance of Normal Blood Pressure (ID 502), Maintenance of Normal HDL-Cholesterol Concentrations (ID 515), Maintenance of Normal (Fasting) Blood Concentratio. EFSA J. 2009, 7, 1–26. [Google Scholar] [CrossRef]
- Barnes, R.D.; Ivezaj, V. A Systematic Review of Motivational Interviewing for Weight Loss among Adults in Primary Care. Obes. Rev. 2015, 304–318. [Google Scholar] [CrossRef] [PubMed]
- Resnicow, K.; McMaster, F. Motivational Interviewing: Moving from Why to How with Autonomy Support. Int. J. Behav. Nutr. Phys. Act. 2012. [Google Scholar] [CrossRef]
- Gardner, J.; Kjolhaug, J.; Linde, J.A.; Sevcik, S.; Lytle, L.A. Teaching Goal-Setting for Weight-Gain Prevention in a College Population: Insights from the CHOICES Study. J. Health Educ. Teach. 2013, 4, 39–49. [Google Scholar] [PubMed]
- NIH—National Cancer Institute. Dietary Assessment Primer–Food Record. Available online: https://dietassessmentprimer.cancer.gov/profiles/record/index.html (accessed on 1 January 2017).
- Hunot, C.; Fildes, A.; Croker, H.; Llewellyn, C.H.; Wardle, J.; Beeken, R.J. Appetitive Traits and Relationships with BMI in Adults: Development of the Adult Eating Behaviour Questionnaire. Appetite 2016, 105, 356–363. [Google Scholar] [CrossRef]
- Harper, A.; Power, M.; Orley, J.; Herrman, H.; Schofield, H.; Murphy, B.; Metelko, Z.; Szabo, S.; Pibernik-Okanovic, M.; Quemada, N.; et al. Development of the World Health Organization WHOQOL-BREF Quality of Life Assessment. The WHOQOL Group. Psychol. Med. 1998, 28, 551–558. [Google Scholar] [CrossRef]
- O’Mara, M.; Hudson-Curtis, B.; Olson, K.; Yueh, Y.; Dunn, J.; Spooner, N. The Effect of Hematocrit and Punch Location on Assay Bias during Quantitative Bioanalysis of Dried Blood Spot Samples. Bioanalysis 2011, 3, 2335–2347. [Google Scholar] [CrossRef]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the Concentration of Low-Density Lipoprotein Cholesterol in Plasma, without Use of the Preparative Ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis Model Assessment: Insulin Resistance and β-Cell Function from Fasting Plasma Glucose and Insulin Concentrations in Man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Wallace, T.M.; Levy, J.C.; Matthews, D.R. Use and Abuse of HOMA Modeling. Diabetes Care 2004, 1487–1495. [Google Scholar] [CrossRef]
- Schutte, S.; Esser, D.; Hoevenaars, F.P.M.; Hooiveld, G.J.E.J.; Priebe, M.G.; Vonk, R.J.; Wopereis, S.; Afman, L.A. A 12-Wk Whole-Grain Wheat Intervention Protects against Hepatic Fat: The Graandioos Study, a Randomized Trial in Overweight Subjects. Am. J. Clin. Nutr. 2018, 108, 1264–1274. [Google Scholar] [CrossRef] [PubMed]
- Friedman, J.; Hastie, T.; Tibshirani, R. Regularization Paths for Generalized Linear Models via Coordinate Descent. J. Stat. Softw. 2010, 33, 1–22. [Google Scholar] [CrossRef]
- Duthie, S.J.; Duthie, G.G.; Russell, W.R.; Kyle, J.A.M.; Macdiarmid, J.I.; Rungapamestry, V.; Stephen, S.; Megias-Baeza, C.; Kaniewska, J.J.; Shaw, L.; et al. Effect of Increasing Fruit and Vegetable Intake by Dietary Intervention on Nutritional Biomarkers and Attitudes to Dietary Change: A Randomised Trial. Eur. J. Nutr. 2018, 57, 1855–1872. [Google Scholar] [CrossRef]
- Mielgo-Ayuso, J.; Valtueña, J.; Huybrechts, I.; Breidenassel, C.; Cuenca-García, M.; De Henauw, S.; Stehle, P.; Kafatos, A.; Kersting, M.; Widhalm, K.; et al. Fruit and Vegetables Consumption Is Associated with Higher Vitamin Intake and Blood Vitamin Status among European Adolescents. Eur. J. Clin. Nutr. 2017, 71, 458–467. [Google Scholar] [CrossRef] [PubMed]
- Souverein, O.W.; De Vries, J.H.M.; Freese, R.; Watzl, B.; Bub, A.; Miller, E.R.; Castenmiller, J.J.M.; Pasman, W.J.; Van Het Hof, K.; Chopra, M.; et al. Prediction of Fruit and Vegetable Intake from Biomarkers Using Individual Participant Data of Diet-Controlled Intervention Studies. Br. J. Nutr. 2015, 113, 1396–1409. [Google Scholar] [CrossRef]
- Silva, D.A.S.; De Lima, T.R.; Tremblay, M.S. Association between Resting Heart Rate and Health-Related Physical Fitness in Brazilian Adolescents. BioMed Res. Int. 2018, 2018. [Google Scholar] [CrossRef]
- Cornelissen, V.A.; Verheyden, B.; Aubert, A.E.; Fagard, R.H. Effects of Aerobic Training Intensity on Resting, Exercise and Post-Exercise Blood Pressure, Heart Rate and Heart-Rate Variability. J. Hum. Hypertens. 2010, 24, 175–182. [Google Scholar] [CrossRef]
- Jensen, M.T.; Suadicani, P.; Hein, H.O.; Gyntelberg, F. Elevated Resting Heart Rate, Physical Fitness and All-Cause Mortality: A 16-Year Follow-up in the Copenhagen Male Study. Heart 2013, 99, 882–887. [Google Scholar] [CrossRef]
- Ryan, D.; Barquera, S.; Barata Cavalcanti, O.; Ralston, J. The Global Pandemic of Overweight and Obesity. In Handbook of Global Health; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–35. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Dias, S.; Hoffmann, G. Impact of Long-Term Lifestyle Programmes on Weight Loss and Cardiovascular Risk Factors in Overweight/Obese Participants: A Systematic Review and Network Meta-Analysis. Syst. Rev. 2015, 3. [Google Scholar] [CrossRef]
- Cioffi, I.; Evangelista, A.; Ponzo, V.; Ciccone, G.; Soldati, L.; Santarpia, L.; Contaldo, F.; Pasanisi, F.; Ghigo, E.; Bo, S. Intermittent versus Continuous Energy Restriction on Weight Loss and Cardiometabolic Outcomes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. J. Transl. Med. 2018. [Google Scholar] [CrossRef] [PubMed]
- Verheggen, R.J.H.M.; Maessen, M.F.H.; Green, D.J.; Hermus, A.R.M.M.; Hopman, M.T.E.; Thijssen, D.H.T. A Systematic Review and Meta-Analysis on the Effects of Exercise Training versus Hypocaloric Diet: Distinct Effects on Body Weight and Visceral Adipose Tissue. Obes. Rev. 2016, 664–690. [Google Scholar] [CrossRef]
- Bowen, J.; Brindal, E.; James-Martin, G.; Noakes, M. Randomized Trial of a High Protein, Partial Meal Replacement Program with or without Alternate Day Fasting: Similar Effects on Weight Loss, Retention Status, Nutritional, Metabolic, and Behavioral Outcomes. Nutrients 2018, 10, 1145. [Google Scholar] [CrossRef]
- Aller, E.E.J.G.; Larsen, T.M.; Claus, H.; Lindroos, A.K.; Kafatos, A.; Pfeiffer, A.; Martinez, J.A.; Handjieva-Darlenska, T.; Kunesova, M.; Stender, S.; et al. Weight Loss Maintenance in Overweight Subjects on Ad Libitum Diets with High or Low Protein Content and Glycemic Index: The DIOGENES Trial 12-Month Results. Int. J. Obes. 2014, 38, 1511–1517. [Google Scholar] [CrossRef]
- Wycherley, T.P.; Moran, L.J.; Clifton, P.M.; Noakes, M.; Brinkworth, G.D. Effects of Energy-Restricted High-Protein, Low-Fat Compared with Standard-Protein, Low-Fat Diets: A Meta-Analysis of Randomized Controlled Trials. Am. J. Clin. Nutr. 2012, 96, 1281–1298. [Google Scholar] [CrossRef] [PubMed]
- Van Baak, M.A.; Mariman, E.C.M. Dietary Strategies for Weight Loss Maintenance. Nutrients 2019, 11, 1916. [Google Scholar] [CrossRef] [PubMed]
- Leidy, H.J.; Clifton, P.M.; Astrup, A.; Wycherley, T.P.; Westerterp-Plantenga, M.S.; Luscombe-Marsh, N.D.; Woods, S.C.; Mattes, R.D. The Role of Protein in Weight Loss and Maintenance. Am. J. Clin. Nutr. 2015, 1320S–1329S. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S. The Significance of Protein in Food Intake and Body Weight Regulation. Curr. Opin. Clin. Nutr. Metab. Care 2003, 635–638. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Hoffmann, G. Comparison of Effects of Long-Term Low-Fat vs High-Fat Diets on Blood Lipid Levels in Overweight or Obese Patients: A Systematic Review and Meta-Analysis. J. Acad. Nutr. Diet. 2013, 113, 1640–1661. [Google Scholar] [CrossRef]
- Vafeiadou, K.; Weech, M.; Altowaijri, H.; Todd, S.; Yaqoob, P.; Jackson, K.G.; Lovegrove, J.A. Replacement of Saturated with Unsaturated Fats Had No Impact on Vascular Function but Beneficial Effects on Lipid Biomarkers, E-Selectin, and Blood Pressure: Results from the Randomized, Controlled Dietary Intervention and VAScular Function (DIVAS) Study. Am. J. Clin. Nutr. 2015, 102, 40–48. [Google Scholar] [CrossRef]
- Lu, M.; Wan, Y.; Yang, B.; Huggins, C.E.; Li, D. Effects of Low-Fat Compared with High-Fat Diet on Cardiometabolic Indicators in People with Overweight and Obesity without Overt Metabolic Disturbance: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Br. J. Nutr. 2018, 119, 96–108. [Google Scholar] [CrossRef]
- Ferguson, L.R.; De Caterina, R.; Görman, U.; Allayee, H.; Kohlmeier, M.; Prasad, C.; Choi, M.S.; Curi, R.; De Luis, D.A.; Gil, Á.; et al. Guide and Position of the International Society of Nutrigenetics/Nutrigenomics on Personalised Nutrition: Part 1—Fields of Precision Nutrition. J. Nutr. Nutr. 2016, 9, 12–27. [Google Scholar] [CrossRef]
- Horne, J.; Gilliland, J.; O’Connor, C.; Seabrook, J.; Madill, J. Enhanced Long-Term Dietary Change and Adherence in a Nutrigenomics-Guided Lifestyle Intervention Compared to a Population-Based (GLB/DPP) Lifestyle Intervention for Weight Management: Results from the NOW Randomised Controlled Trial. BMJ Nutr. Prev. Health 2020, 3, 49–59. [Google Scholar] [CrossRef]
- Nielsen, D.E.; El-Sohemy, A. Disclosure of Genetic Information and Change in Dietary Intake: A Randomized Controlled Trial. PLoS ONE 2014, 9, e112665. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, J.C.; De Vries, J.; McBurney, M.I.; Wopereis, S.; Serttas, S.; Marsman, D.S. Measuring Health Promotion: Translating Science into Policy. Eur. J. Nutr. 2020, 59, 11–23. [Google Scholar] [CrossRef]
- Ndanuko, R.N.; Tapsell, L.C.; Charlton, K.E.; Neale, E.P.; Batterham, M.J. Effect of Individualised Dietary Advice for Weight Loss Supplemented with Walnuts on Blood Pressure: The HealthTrack Study. Eur. J. Clin. Nutr. 2018, 72, 894–903. [Google Scholar] [CrossRef]
- Celis-Morales, C.; Livingstone, K.M.; Marsaux, C.F.; Macready, A.L.; Fallaize, R.; O’donovan, C.B.; Woolhead, C.; Forster, H.; Walsh, M.C.; Navas-Carretero, S.; et al. Effect of Personalized Nutrition on Health-Related Behaviour Change: Evidence from the Food4Me European Randomized Controlled Trial. Int. J. Epidemiol. 2017, 578–588. [Google Scholar] [CrossRef] [PubMed]
- Hollands, G.J.; French, D.P.; Griffin, S.J.; Prevost, A.T.; Sutton, S.; King, S.; Marteau, T.M. The Impact of Communicating Genetic Risks of Disease on Riskreducing Health Behaviour: Systematic Review with Meta-Analysis. BMJ 2016, 352. [Google Scholar] [CrossRef]
- Robinson, K.; Rozga, M.; Braakhuis, A.; Ellis, A.; Monnard, C.R.; Sinley, R.; Wanner, A.; Vargas, A.J. Effect of Incorporating Genetic Testing Results into Nutrition Counseling and Care on Dietary Intake: An Evidence Analysis Center Systematic Review—Part I. J. Acad. Nutr. Diet. 2020, 121, 553–581.e3. [Google Scholar] [CrossRef]
- Braakhuis, A.; Monnard, C.R.; Ellis, A.; Rozga, M. Consensus Report of the Academy of Nutrition and Dietetics: Incorporating Genetic Testing into Nutrition Care. J. Acad. Nutr. Diet. 2020, 121. [Google Scholar] [CrossRef] [PubMed]
- Ellis, A.; Rozga, M.; Braakhuis, A.; Monnard, C.R.; Robinson, K.; Sinley, R.; Wanner, A.; Vargas, A.J. Effect of Incorporating Genetic Testing Results into Nutrition Counseling and Care on Health Outcomes: An Evidence Analysis Center Systematic Review—Part II. J. Acad. Nutr. Diet. 2020, 121, 582–605.e17. [Google Scholar] [CrossRef] [PubMed]
- Ordovas, J.M.; Ferguson, L.R.; Tai, E.S.; Mathers, J.C. Personalised Nutrition and Health. BMJ 2018, 361, bmj.k2173. [Google Scholar] [CrossRef]
- Kebbe, M.; Perez, A.; Buchholz, A.; McHugh, T.L.F.; Scott, S.S.; Richard, C.; Mohipp, C.; Dyson, M.P.; Ball, G.D.C. Barriers and Enablers for Adopting Lifestyle Behavior Changes in Adolescents with Obesity: A Multi-Centre, Qualitative Study. PLoS ONE 2018, 13, e0209219. [Google Scholar] [CrossRef] [PubMed]
- Den Braver, N.R.; de Vet, E.; Duijzer, G.; ter Beek, J.; Jansen, S.C.; Hiddink, G.J.; Feskens, E.J.M.; Haveman-Nies, A. Determinants of Lifestyle Behavior Change to Prevent Type 2 Diabetes in High-Risk Individuals. Int. J. Behav. Nutr. Phys. Act. 2017, 14. [Google Scholar] [CrossRef] [PubMed]
- Jeanette, M.O.; Bonnie, J.N. Health Coaching to Improve Healthy Lifestyle Behaviors: An Integrative Review. Am. J. Health Promot. 2010. [Google Scholar] [CrossRef]
- Coppini, L.Z.; Waitzberg, D.L.; Campos, A.C.L. Limitations and Validation of Bioelectrical Impedance Analysis in Morbidly Obese Patients. Curr. Opin. Clin. Nutr. Metab. Care 2005, 329–332. [Google Scholar] [CrossRef] [PubMed]
- Maes, L.; Van Cauwenberghe, E.; Van Lippevelde, W.; Spittaels, H.; De Pauw, E.; Oppert, J.M.; Van Lenthe, F.J.; Brug, J.; De Bourdeaudhuij, I. Effectiveness of Workplace Interventions in Europe Promoting Healthy Eating: A Systematic Review. Eur. J. Public Health 2012, 22, 677–683. [Google Scholar] [CrossRef] [PubMed]
- Rongen, A.; Robroek, S.J.W.; Van Lenthe, F.J.; Burdorf, A. Workplace Health Promotion: A Meta-Analysis of Effectiveness. Am. J. Prev. Med. 2013, 406–415. [Google Scholar] [CrossRef] [PubMed]
- Weiner, B.J.; Lewis, M.A.; Linnan, L.A. Using Organization Theory to Understand the Determinants of Effective Implementation of Worksite Health Promotion Programs. Health Educ. Res. 2009, 292–305. [Google Scholar] [CrossRef] [PubMed]
- Linnan, L.A.; Sorensen, G.; Colditz, G.; Klar, N.; Emmons, K.M. Using Theory to Understand the Multiple Determinants of Low Participation in Worksite Health Promotion Programs. Health Educ. Behav. 2001, 28, 591–607. [Google Scholar] [CrossRef] [PubMed]
- Mache, S.; Jensen, S.; Jahn, R.; Steudtner, M.; Ochsmann, E.; Preuß, G. Worksite Health Program Promoting Changes in Eating Behavior and Health Attitudes. Health Promot. Pract. 2015, 16, 826–836. [Google Scholar] [CrossRef] [PubMed]
- Robroek, S.J.W.; Lindeboom, D.E.M.; Burdorf, A. Initial and Sustained Participation in an Internet-Delivered Long-Term Worksite Health Promotion Program on Physical Activity and Nutrition. J. Med. Internet Res. 2012, 14, 42–55. [Google Scholar] [CrossRef] [PubMed]
- Seaverson, E.L.D.; Gingerich, S.B.; Mangen, D.J.; Anderson, D.R. Measuring Participation in Employer-Sponsored Health and Well-Being Programs: A Participation Index and Its Association with Health Risk Change. Am. J. Health. Promot. 2019, 33, 1002–1008. [Google Scholar] [CrossRef] [PubMed]
- Wopereis, S.; Stroeve, J.H.M.; Stafleu, A.; Bakker, G.C.M.; Burggraaf, J.; van Erk, M.J.; Pellis, L.; Boessen, R.; Kardinaal, A.A.F.; van Ommen, B. Multi-Parameter Comparison of a Standardized Mixed Meal Tolerance Test in Healthy and Type 2 Diabetic Subjects: The PhenFlex Challenge. Genes Nutr. 2017, 12, 21. [Google Scholar] [CrossRef] [PubMed]
- Berry, S.E.; Valdes, A.M.; Drew, D.A.; Asnicar, F.; Mazidi, M.; Wolf, J.; Capdevila, J.; Hadjigeorgiou, G.; Davies, R.; Al Khatib, H.; et al. Human Postprandial Responses to Food and Potential for Precision Nutrition. Nat. Med. 2020, 26, 964–973. [Google Scholar] [CrossRef]
- Division for Heart Disease and Stroke Prevention, Centers for Disease Control and Prevention. DHDSP Data Trends & Maps. Available online: https://www.cdc.gov/dhdsp/maps/dtm/index.html (accessed on 3 May 2021).

| Advice Category | Personalized Advice | Personalization Factor 2 | Classification | Personalization Based on SNP |
|---|---|---|---|---|
| Energy intake advice | Caloric intake | body weight, height, age, gender, physical activity | Mifflin St. Jeor equation: BMR (kcal/day) = 10 × weight (kg) + 6.25 × height (cm) − 5 × age (y) + a1 (kcal/day), a1 = +5 for males and −161 for females. Total energy expenditure = BMR × daily PAL Total PAL = PAL daily pattern + PAL sport/leisure1 + … + PAL sport/leisuren | - |
| Personalized Diet Types & SNP-based macronutrient advice | Protein intake | glucose tolerance, disposition index 3, blood-pressure | normal = normal, glucose intolerance = IFG, IGT, IFG/IGT or T2D low disposition index (<1.5), normal disposition index (>1.5) optimal (SBP <120 and DBP <80 mmHg), elevated (SBP ≥120 or DBP ≥80 mmHg) | FTO rs99396095 |
| Carbohydrate intake | waist circumference, 2-h glucose | normal (M ≤ 40-inch, F ≤ 35 in), elevated (M > 40-in, F > 35 in) normal (<7.77 mmol/L; <140 mg/dL); elevated (≥7.77 mmol/L; ≥140 mg/dL) | FTO rs99396095 ADAMTS9 rs4607103 GCKR rs780094 | |
| Fat intake | waist circumference, blood pressure, LDL cholesterol | normal (M ≤ 40 in, F ≤ 35 in), elevated (M > 40 in, F > 35 in) optimal (SBP < 120 and DBP < 80 mmHg), elevated (SBP ≥ 120 or DBP ≥ 80 mmHg) normal (≤3.36 mmol/L; ≤130 mg/dL); elevated (>3.36 mmol/L; >130 mg/dL) | FTO rs99396095 FTO rs1121980 TCF7L2 rs7903146 FADS1 rs174546 FADS1 rs174548 IGF2BP2 rs4402960 PPARG rs1801282 | |
| Micronutrient advice | Fiber intake | fasting glucose, 2-h glucose, LDL cholesterol, blood pressure, waist circumference | normal (<5.55 mmol/L; <100 mg/dL); elevated (≥5.55 mmol/L; ≥100 mg/dL) normal (<7.77 mmol/L; <140 mg/dL); elevated (≥7.77 mmol/L; ≥140 mg/dL) optimal (<2.59 mmol/L; <100 mg/dL); increased (≥2.59 mmol/L; ≥100 mg/dL) optimal (SBP < 120 and DBP < 80 mmHg), elevated (SBP ≥ 120 or DBP ≥ 80 mmHg) normal (M ≤ 40 in, F ≤ 35 in), elevated (M > 40 in, F > 35 in) | ADAMTS9 rs4607103 TCF7L2 rs7903146 |
| MUFA intake | disposition index 3 LDL cholesterol, blood pressure, fasting TG, postprandial TG 4, | low disposition index (<1.5), normal disposition index (>1.5) normal (≤3.36 mmol/L; ≤130 mg/dL); elevated (>3.36 mmol/L; >130 mg/dL) optimal (SBP <120 and DBP <80 mmHg), elevated (SBP ≥120 or DBP ≥80 mmHg) normal (≤1.7 mmol/L; ≤150 mg/dL); elevated (>1.7 mmol/L; >150 mg/dL) normal (≤2.5 mmol/L); elevated (>2.5 mmol/L) | - | |
| Omega-3 intake | blood pressure, fasting TG, postprandial TG 4, omega-3 index | optimal (SBP <120 and DBP <80 mmHg), elevated (SBP ≥120 or DBP ≥80 mmHg) normal (≤1.7 mmol/L; ≤150 mg/dL); elevated (>1.7 mmol/L; >150 mg/dL) normal (≤2.5 mmol/L); elevated (>2.5 mmol/L) optimal (>8 %); intermediate or low (≤8 %) | FADS1 rs1745465 FADS1 rs1745485 | |
| Phytosterols | LDL cholesterol | optimal (<2.59 mmol/L); increased (≥2.59-≤3.36 mmol/L); elevated (>3.36 mmol/L) | - | |
| Vitamin C intake | blood pressure, age, gender | optimal (SBP <120 and DBP <80 mmHg), elevated (SBP ≥120 or DBP ≥80 mmHg) | - | |
| Vitamin B2 intake | blood pressure, age, gender | optimal (SBP <120 and DBP <80 mmHg), elevated (SBP ≥120 or DBP ≥80 mmHg) | MTHFR rs18011335 | |
| SNP-based narratives | Physical activity | - | ACTN3 rs1815739 FTO rs1121980 | |
| Vitamin D | - | GC rs7041 | ||
| - | GC rs4588 | |||
| - | GC rs2282679 | |||
| Lactose intolerance | - | MCM6 rs182549 | ||
| - | MCM6 rs4988235 | |||
| Caffeine sensitivity | - | CYP1A2 rs762551 | ||
| Salt sensitivity | - | AGT rs5051 | ||
| - | AGT rs699 |
| Carbohydrates | Fat | Protein | |
|---|---|---|---|
| PDT | % of Total Energy (Target %) | ||
| A | 45–65 (50) | 20–40 (30) | 10–22 (20) |
| B | 45–65 (60) | 20–30 (20) | 10–22 (20) |
| C | 35–50 (45) | 20–40 (40) | 10–22 (15) |
| D | 45–65 (45) | 20–40 (25) | 18–35 (30) |
| E | 45–65 (45) | 20–30 (20) | 18–35 (35) |
| F | 35–50 (35) | 20–40 (30) | 18–35 (35) |
| G | 35–50 (40) | 20–30 (25) | 18–35 (35) |
| Variable | PP (n = 73) | Group A (n = 48) | Group G (n = 22) | |||
|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | Mean | SD | |
| Gender (n, men/women) | 25/48 | 15/33 | 9/13 | |||
| Age (years) ** | 43.1 | 8.7 | 40.9 | 8.1 | 47.8 | 8.3 |
| Anthropometrics and Vitals | ||||||
| BMI (kg/m2) *** | 27.4 | 4.0 | 26.0 | 3.3 | 30.5 | 3.6 |
| Body weight (kg) ** | 77.8 | 15.5 | 72.6 | 13.2 | 89.4 | 14.6 |
| Body fat (%) **** | 32.0 | 7.6 | 30.3 | 6.8 | 36.8 | 7.1 |
| Muscle mass (kg) | 50.1 | 10.7 | 48.3 | 9.9 | 53.7 | 11.8 |
| Waist circumference (cm) **** | 94.6 | 13.0 | 89.7 | 10.7 | 105.7 | 10.7 |
| Hip circumference (cm) ** | 104.8 | 10.1 | 102.4 | 8.4 | 111.9 | 7.0 |
| Systolic blood pressure (mmHg) | 119.2 | 16.4 | 116.6 | 16.1 | 123.1 | 16.4 |
| Diastolic blood pressure (mmHg) | 73.7 | 8.7 | 72.2 | 8.0 | 75.6 | 8.6 |
| Clinical Chemistry (fasting) | ||||||
| C-peptide (nmol/L) | 0.48 | 0.20 | 0.43 | 0.15 | 0.54 | 0.21 |
| Glucose (mmol/L) | 4.41 | 0.47 | 4.30 | 0.44 | 4.64 | 0.47 |
| HDL (mmol/L) | 1.52 | 0.40 | 1.60 | 0.43 | 1.39 | 0.27 |
| LDL (mmol/L) | 2.62 | 0.54 | 2.54 | 0.47 | 2.78 | 0.65 |
| Total cholesterol (mmol/L) | 4.68 | 0.66 | 4.66 | 0.62 | 4.70 | 0.75 |
| Triglycerides (mmol/L) | 1.15 | 0.57 | 1.11 | 0.61 | 1.25 | 0.51 |
| Indices | ||||||
| HOMA-IR | 0.094 | 0.043 | 0.082 | 0.029 | 0.119 | 0.057 |
| Matsuda index | 212.0 | 82.4 | 230.8 | 77.5 | 176.2 | 79.0 |
| Week 10: Mean (SD) | Week 20: Mean (SD) | Difference: | ||||
|---|---|---|---|---|---|---|
| Week 20–Week 10 (%) 1 | ||||||
| A (n = 48) | G (n = 22) | A (n = 48) | G (n = 22) | A (n = 48) | G (n = 22) | |
| Macronutrient intake 1 | ||||||
| Calories (kcal) *** | 1877.1 (554.4) | 1974.0 (520.0) | 1681.5 (490.6) | 1613.5 (324.5) | −10.40% | −18.30% |
| Carbohydrates (g) ** | 189.9 (64.9) | 217.4 (83.1) | 182.0 (58.3) | 163.6 (43.5) | −4.20% | −24.70% |
| Carbohydrates (en%) | 40.6 (7.2) | 43.4 (5.4) | 43 (6.6) | 40.9 (8.5) | 6% | −5.80% |
| Protein (g) | 87.9 (24.8) | 82.7 (16.2) | 79.9 (24.6) | 88.8 (27.6) | −9.10% | 7.40% |
| Protein (en%) | 19.1 (3.8) | 17.3 (3.5) | 19.9 (4.3) | 22.0 (5.5) | 4.10% | 27.7% ** |
| Fat (g) **** | 80.2 (27.7) | 82.6 (21.2) | 65.0 (22.2) | 62.4 (16.8) | −18.90% | −24.70% |
| Fat (en%) ** | 38.2 (5.4) | 37.9 (5.8) | 34.6 (6.2) | 34.7 (5.0) | −9.50% | −8.50% |
| SFA (g) *** | 25.8 (10.3) | 25.2 (7.1) | 20.6 (7.2) | 18.8 (6.6) | −20.30% | −25.50% |
| SFA (en%) | 12.3 (3.0) | 11.7 (3.0) | 11.0 (2.6) | 10.4 (2.3) | −10.60% | −11.70% |
| PUFA (g) ** | 12.4 (5.9) | 11.7 (5.5) | 9.6 (4.7) | 9.3 (4.3) | −22.60% | −20.50% |
| PUFA (en%) | 6.0 (2.3) | 5.4 (2.5) | 5.2 (2.0) | 5.1 (2.0) | −12.60% | −5.60% |
| MUFA (g) | 22.0 (10.6) | 20.4 (10.8) | 17.8 (8.6) | 17.0 (8.0) | −19.10% | −16.80% |
| MUFA (en%) | 10.5 (3.5) | 9.2 (3.6) | 9.7 (3.4) | 9.4 (3.3) | −8.50% | 1.60% |
| Total sugar (g) **** | 63.9 (33.5) | 82.7 (40.5) | 55.8 (30.2) | 55.6 (23.9) | −12.70% | −32.70% |
| Total sugar (en%) | 13.7 (5.7) | 16.3 (5.1) | 13.3 (5.4) | 13.8 (5.4) | −3.90% | −15.70% |
| Total fiber (g) 2 | 17.3 (5.6) | 17.6 (8.0) | 19.0 (6.1) | 17.8 (5.9) | 9.70% | 1.60% |
| Total fiber (en%) 2 **** | 1.9 (0.6) | 1.8 (0.6) | 2.4 (0.8) | 2.2 (0.7) | 25.50% | 24.70% |
| Micronutrient intake 1 | ||||||
| Sodium (mg) *** | 2799.6 (895.5) | 2795.3 (885.2) | 2212.8 (892.3) | 2371.7 (829.7) | −21% | −15.20% |
| Potassium (mg) * | 1983.7 (781.0) | 1777.7 (714.6) | 2241.1 (716.4) | 2233.8 (831.9) | 13% | 59.40% |
| Magnesium (mg) ** | 187.3 (66.7) | 222.3 (150.5) | 238.5 (82.5) | 257.9 (78.2) | 27.30% | 15.90% |
| Vitamin C (mg) *** | 74.4 (55.2) | 72.3 (39.1) | 106.1 (67.3) | 111.8 (58.4) | 42.60% | 54.50% |
| Beta-carotene (mcg) | 3074.0 (4330.5) | 1534.2 (2740.1) | 3415.7 (2518.0) | 5970.8 (4316.1) | 11.10% | 289.2% **** |
| Physical activity 3 | ||||||
| Resting heart rate (bpm) **** | 63.4 (6.9) | 66.0 (7.1) | 62.4 (6.7) | 63.6 (7.0) | −1.70% | −2.90% |
| Steps (n/day) **** | 9319 (3073) | 8558 (1856) | 10234 (3206) | 8957 (1865) | 8.50% | 6.50% |
| Sleep 3 | ||||||
| Sleep (h/day) | 7.3 (0.7) | 6.9 (1.1) | 7.1 (1.2) | 7.0 (1.1) | −2.80% | 1.40% |
| Variable 1 | Week 10: Mean (SD) | Week 20: Mean (SD) | Difference: Week 20–Week 10 (%) 1 | |||
|---|---|---|---|---|---|---|
| A (n = 48) | G (n = 22) | A (n = 48) | G (n = 22) | A (n = 48) | G (n = 22) | |
| Anthropometrics and Vitals | ||||||
| BMI (kg/m2) **** | 26.0 (3.4) | 30.7 (3.8) | 25.7 (3.3) | 29.9 (3.8) | −1.20% | −2.60% |
| Body weight (kg) | 73.0 (13.4) | 90.0 (15.0) | 72.1 (13.3) | 89.0 (15.4) | −1.2% ** | −1.1% **** |
| Body fat (%) * | 30.5 (7.1) | 36.2 (6.0) | 29.8 (6.9) | 35.9 (6.7) | −2.30% | −0.90% |
| Muscle mass (kg) | 48.3 (9.7) | 54.2 (11.6) | 48.1 (10.0) | 54.4 (11.7) | −0.50% | 0.40% |
| Waist circumference (cm) | 89.5 (11.0) | 104.5 (11.8) | 89.0 (9.5) | 104.3 (11.8) | −0.60% | −0.20% |
| Hip circumference (cm) **** | 101.2 (8.4) | 111.5 (7.3) | 99.6 (8.4) | 108.0 (7.8) | −1.60% | −3.10% |
| Systolic BP (mmHg) | 118.0 (13.8) | 122.8 (15.8) | 114.7 (15.0) | 119.2 (14.3) | −2.80% | −2.90% |
| Diastolic BP (mmHg) | 73.8 (7.7) | 76.5 (8.2) | 70.9 (7.1) | 74.8 (9.1) | −3.90% | −2.20% |
| Clinical Chemistry (fasting) | ||||||
| C-peptide fasting (nmol/L) | 0.54 (0.24) | 0.68 (0.23) | 0.47 (0.19) | 0.66 (0.25) | −12.10% | −2.70% |
| C-peptide 2 h (nmol/L) | 1.39 (0.69) | 1.91 (0.73) | 1.42 (0.79) | 1.73 (0.69) | 1.90% | −9.70% |
| Glucose fasting (mmol/L) | 4.48 (0.47) | 4.75 (0.41) | 4.68 (0.51) | 5.18 (0.49) | 4.50% | 9.00% |
| Glucose 2 h (mmol/L) | 5.37 (0.83) | 5.94 (0.87) | 5.56 (0.72) | 6.10 (0.63) | 3.60% | 2.70% |
| HDL cholesterol (mmol/L) | 1.54 (0.39) | 1.38 (0.40) | 1.62 (0.40) | 1.28 (0.36) | 5.10% | −6.80% |
| LDL cholesterol (mmol/L) | 2.67 (0.53) | 2.88 (0.64) | 2.44 (0.50) | 2.44 (0.40) | −8.40% | −15.4% ** |
| Total cholesterol (mmol/L) | 4.70 (0.60) | 4.96 (0.74) | 4.61 (0.61) | 4.46 (0.61) | −2% | −9.9% **** |
| Triglycerides (mmol/L) | 1.16 (0.61) | 1.58 (0.56) | 1.32 (1.0) | 1.64 (0.47) | 13.20% | 3.70% |
| Indices | ||||||
| HOMA-IR | 0.108 (0.050) | 0.142 (0.046) | 0.100 (0.045) | 0.154 (0.062) | −7.40% | 8.50% |
| Matsuda index | 202.8 (89.2) | 129.9 (40.6) | 191.1 (66.9) | 125.8 (45.7) | −5.80% | −3.20% |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Hoogh, I.M.; Winters, B.L.; Nieman, K.M.; Bijlsma, S.; Krone, T.; van den Broek, T.J.; Anderson, B.D.; Caspers, M.P.M.; Anthony, J.C.; Wopereis, S. A Novel Personalized Systems Nutrition Program Improves Dietary Patterns, Lifestyle Behaviors and Health-Related Outcomes: Results from the Habit Study. Nutrients 2021, 13, 1763. https://doi.org/10.3390/nu13061763
de Hoogh IM, Winters BL, Nieman KM, Bijlsma S, Krone T, van den Broek TJ, Anderson BD, Caspers MPM, Anthony JC, Wopereis S. A Novel Personalized Systems Nutrition Program Improves Dietary Patterns, Lifestyle Behaviors and Health-Related Outcomes: Results from the Habit Study. Nutrients. 2021; 13(6):1763. https://doi.org/10.3390/nu13061763
Chicago/Turabian Stylede Hoogh, Iris M., Barbara L. Winters, Kristin M. Nieman, Sabina Bijlsma, Tanja Krone, Tim J. van den Broek, Barbara D. Anderson, Martien P. M. Caspers, Joshua C. Anthony, and Suzan Wopereis. 2021. "A Novel Personalized Systems Nutrition Program Improves Dietary Patterns, Lifestyle Behaviors and Health-Related Outcomes: Results from the Habit Study" Nutrients 13, no. 6: 1763. https://doi.org/10.3390/nu13061763
APA Stylede Hoogh, I. M., Winters, B. L., Nieman, K. M., Bijlsma, S., Krone, T., van den Broek, T. J., Anderson, B. D., Caspers, M. P. M., Anthony, J. C., & Wopereis, S. (2021). A Novel Personalized Systems Nutrition Program Improves Dietary Patterns, Lifestyle Behaviors and Health-Related Outcomes: Results from the Habit Study. Nutrients, 13(6), 1763. https://doi.org/10.3390/nu13061763