Micronutrients Deficiency, Supplementation and Novel Coronavirus Infections—A Systematic Review and Meta-Analysis
Abstract
1. Introduction
2. Methods
2.1. Search Identification and Selection
- Type of intervention/exposure:
- Prevention: Supplementation and/or deficiency of any micronutrient singly or in combination with other micronutrients.
- Treatment: Treatment using any micronutrient singly, or in combination with other micronutrients or therapeutical drugs.
- Types of outcomes:
- Prevention: Incidence or episodes of novel coronavirus disease.
- Treatment: Severity of novel coronavirus disease episode, defined by clinical se-verity, mortality, intensive care unit (ICU) admission, hospitalization duration, or progression to respiratory-related complications.
- Type of study: Peer-reviewed publications on interventional (randomized controlled trials) and observational (cohort studies, cross-sectional studies and case-controlled studies) studies, with relevant comparator groups, e.g., infected versus non-infected, groups with varying degrees of severity.
- Type of participants: Adults >19 years, infection-free (prevention) or diagnosed with (treatment) any novel coronavirus disease at time of study recruitment
2.2. Data Extraction
2.3. Quality Assessment
2.4. Statistical Analysis
3. Results
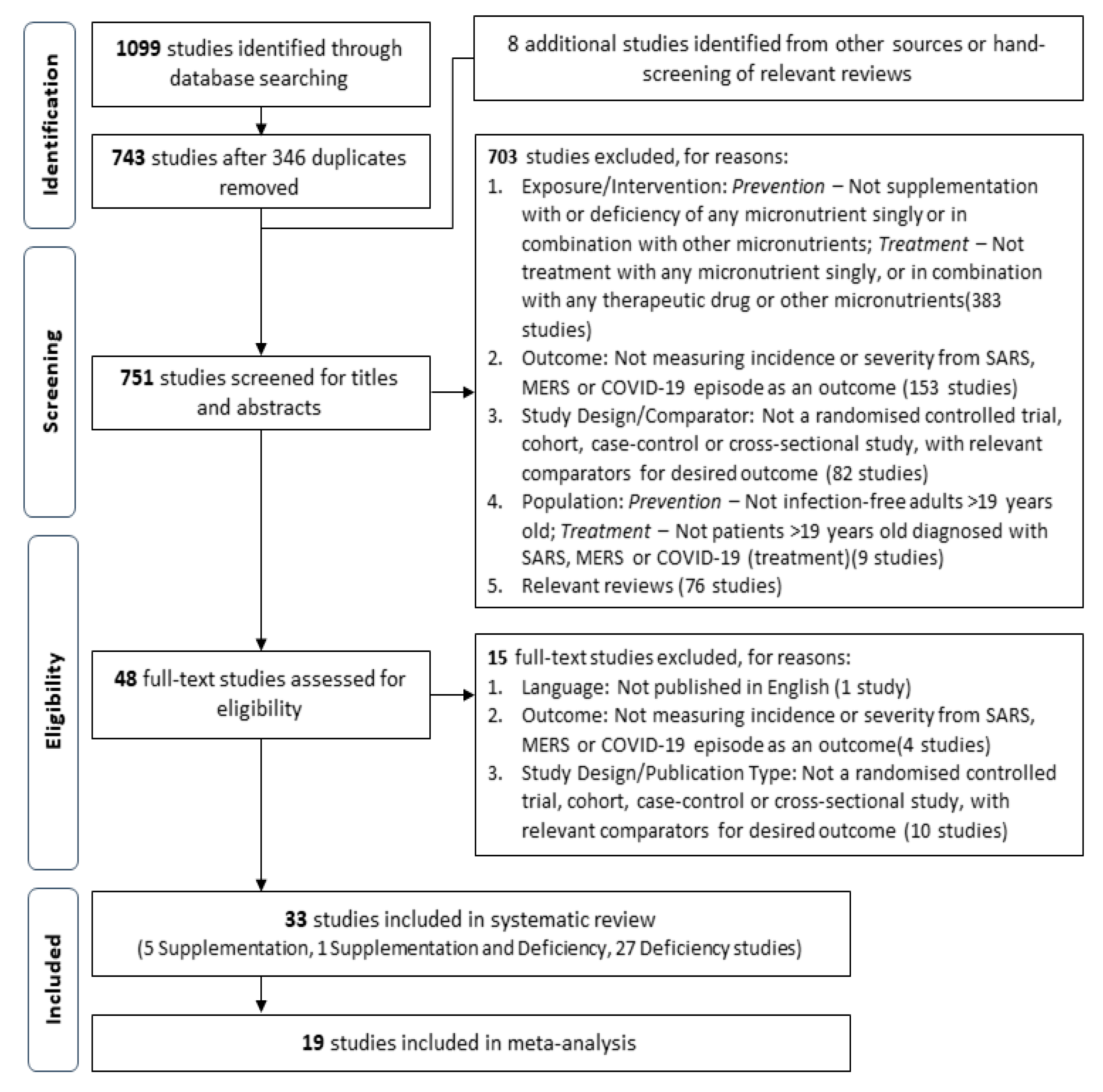
3.1. Screening Results and Characteristics of Included Studies
3.2. Methodological Quality of Included Studies
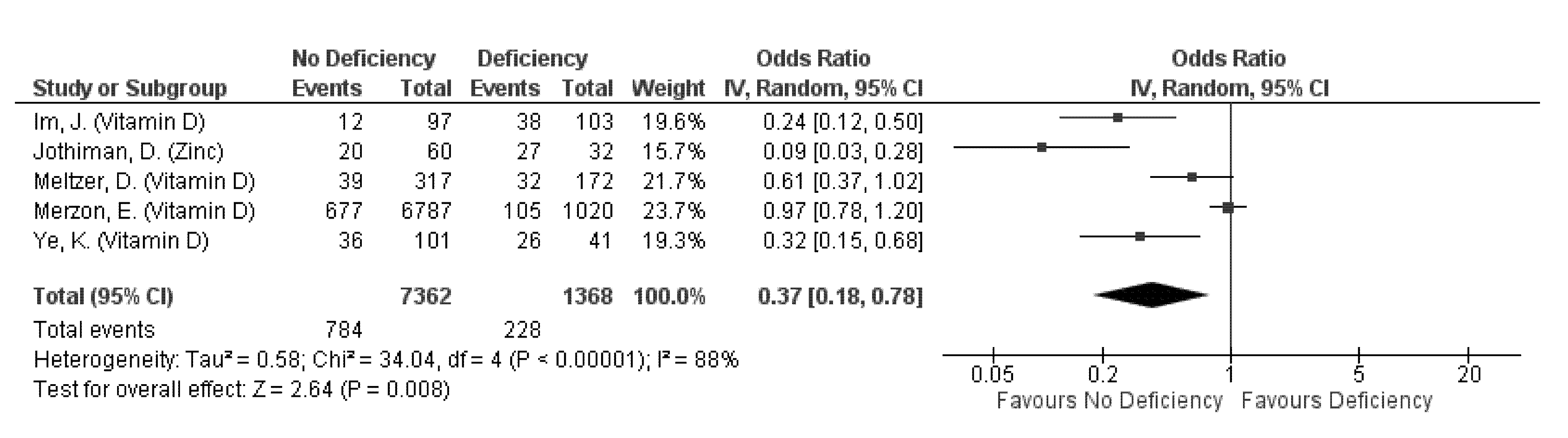
3.3. Outcome: Prevention of COVID-19 Episode
3.4. Outcome: Severity of COVID-19 Episode
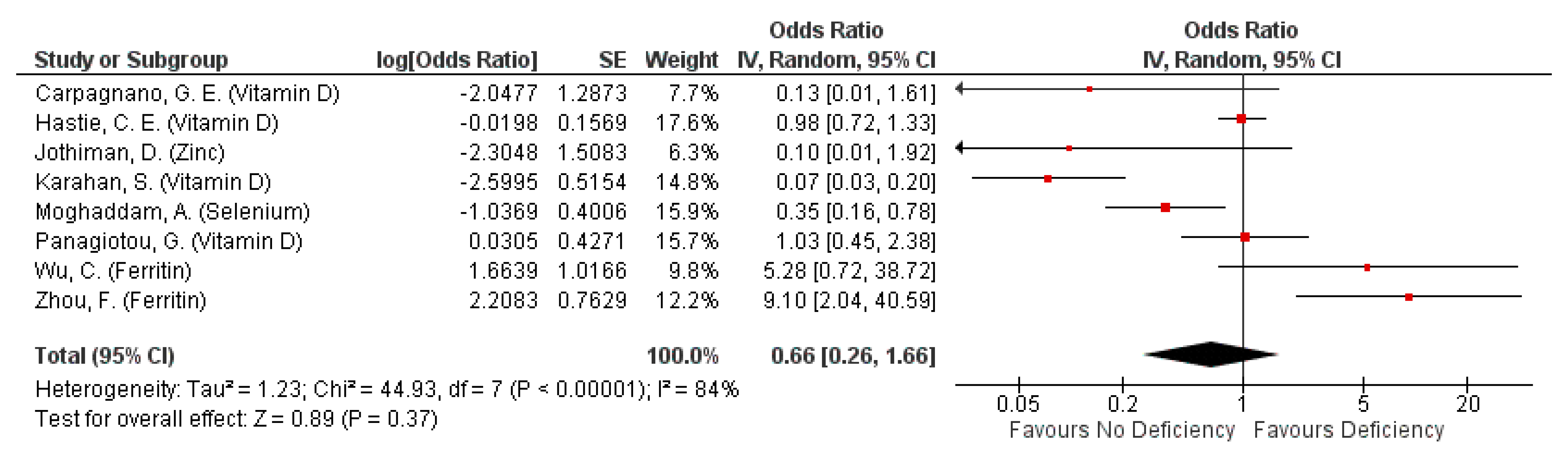
3.4.1. Severity: Mortality from COVID-19
3.4.2. Severity: Hospitalisation Duration from COVID-19
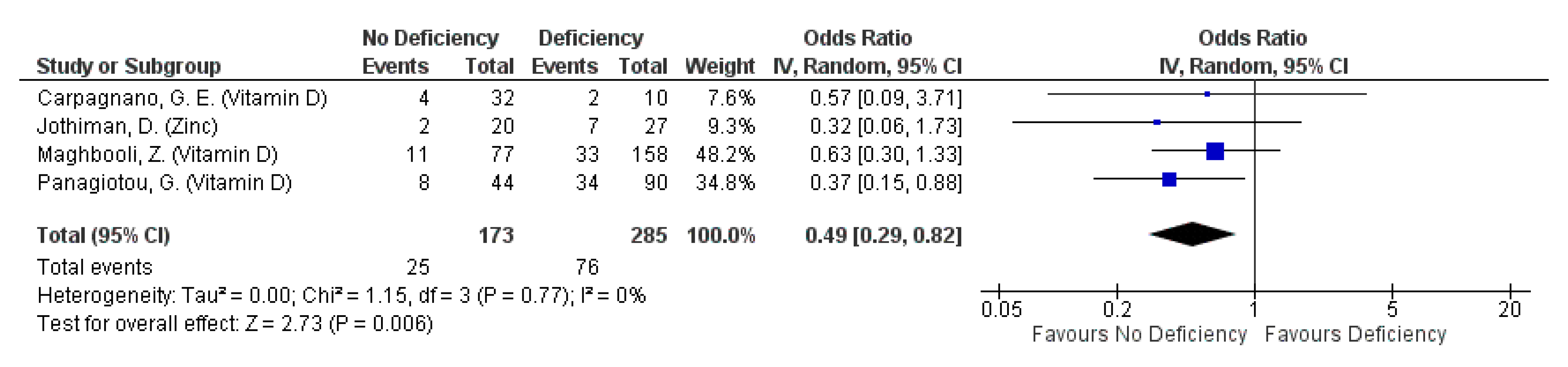
3.4.3. Severity: ICU Admission
3.4.4. Severity: Progression to Respiratory-Related Complications
3.4.5. Severity: Clinical Severity
3.4.6. Severity: Composite Outcomes
4. Discussion
Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- V’kovski, P.; Kratzel, A.; Steiner, S.; Stalder, H.; Thiel, V. Coronavirus biology and replication: Implications for SARS-CoV-2. Nat. Rev. Microbiol. 2021, 19, 155–170. [Google Scholar] [CrossRef]
- World Health Organisation. Update 95—SARS: Chronology of a Serial Killer. Disease Outbreak News 2002. Available online: https://www.who.int/csr/don/2003_07_04/en/ (accessed on 16 March 2021).
- World Health Organisation. Cumulative Number of Reported Probable Cases of SARS. Severe Acute Respiratory Syndrime (SARS) 2003. Available online: https://www.who.int/csr/sars/country/2003_07_11/en/ (accessed on 16 March 2021).
- World Health Organisation (Eastern Mediterranean). MERS Situation Update, December 2020, in Middle East Respiratory Syndrome Monthly Updates. 2020. Available online: http://www.emro.who.int/health-topics/mers-cov/mers-outbreaks.html (accessed on 16 March 2021).
- Coronavirus Update (Live): 74,746,810 Cases and 1,659,186 Deaths from COVID-19 Virus Pandemic—Worldometer. Available online: https://www.worldometers.info/coronavirus/ (accessed on 17 December 2020).
- National Institute of Health, Therapeutic Management of Adults with COVID-19. 2021. Available online: https://www.covid19treatmentguidelines.nih.gov/therapeutic-management/ (accessed on 16 March 2021).
- Gombart, A.F.; Pierre, A.; Maggini, S. A Review of Micronutrients and the Immune System–Working in Harmony to Reduce the Risk of Infection. Nutrients 2020, 12, 236. [Google Scholar] [CrossRef] [PubMed]
- Maggini, S.; Wintergerst, E.S.; Beveridge, S.; Hornig, D.H. Selected vitamins and trace elements support immune function by strengthening epithelial barriers and cellular and humoral immune responses. Br. J. Nutr. 2007, 98, S29–S35. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.X.; Koh, J.; Pang, J. Association between micronutrient deficiency and acute respiratory infections in healthy adults: A systematic review of observational studies. Nutr. J. 2019, 18, 1–12. [Google Scholar] [CrossRef]
- Wang, M.X.; Win, S.S.; Pang, J. Zinc Supplementation Reduces Common Cold Duration among Healthy Adults: A Systematic Review of Randomized Controlled Trials with Micronutrients Supplementation. Am. J. Trop. Med. Hyg. 2020, 103, 86–99. [Google Scholar] [CrossRef]
- Abioye, A.I.; Bromage, S.; Fawzi, W. Effect of micronutrient supplements on influenza and other respiratory tract infections among adults: A systematic review and meta-analysis. BMJ Glob. Health 2021, 6, e003176. [Google Scholar] [CrossRef] [PubMed]
- Hemilä, H.; Chalker, E. Vitamin C Can Shorten the Length of Stay in the ICU: A Meta-Analysis. Nutrients 2019, 11, 708. [Google Scholar] [CrossRef]
- Gorji, A.; Ghadiri, M.K. The potential roles of micronutrient deficiency and immune system dysfunction in COVID-19 pandemic. Nutrition 2020, 111047. [Google Scholar] [CrossRef]
- McAuliffe, S.; Ray, S.; Fallon, E.; Bradfield, J.; Eden, T.; Kohlmeier, M. Dietary micronutrients in the wake of COVID-19: An appraisal of evidence with a focus on high-risk groups and preventative healthcare. BMJ Nutr. Prev. Health 2020, 3, 93–99. [Google Scholar] [CrossRef]
- National Lung, H.B.I. NHLBI Study Quality Assessment Tools. Available online: https://www.nhlbi.nih.gov/health-topics/study-quality-assessment-tools (accessed on 16 March 2021).
- Cochrane. RoB 2: A Revised Cochrane Risk-of-Bias Tool for Randomized Trials. Cochrane Methods: Bias. Available online: https://methods.cochrane.org/bias/resources/rob-2-revised-cochrane-risk-bias-tool-randomized-trials (accessed on 16 March 2021).
- Hastie, C.E.; Pell, J.P.; Sattar, N. Vitamin D and COVID-19 infection and mortality in UK Biobank. Eur. J. Nutr. 2021, 60, 545–548. [Google Scholar] [CrossRef]
- Annweiler, C.; Hanotte, B.; de L’Eprevier, C.G.; Sabatier, J.-M.; Lafaie, L.; Célarier, T. Vitamin D and survival in COVID-19 patients: A quasi-experimental study. J. Steroid Biochem. Mol. Biol. 2020, 204, 105771. [Google Scholar] [CrossRef]
- Sun, Y.; Dong, Y.; Wang, L.; Xie, H.; Li, B.; Chang, C.; Wang, F.-S. Characteristics and prognostic factors of disease severity in patients with COVID-19: The Beijing experience. J. Autoimmun. 2020, 112, 102473. [Google Scholar] [CrossRef] [PubMed]
- Baktash, V.; Hosack, T.; Patel, N.; Shah, S.; Kandiah, P.; Abbeele, K.V.D.; Mandal, A.K.J.; Missouris, C.G. Vitamin D status and outcomes for hospitalised older patients with COVID-19. Postgrad. Med. J. 2020. [Google Scholar] [CrossRef] [PubMed]
- Bellmann-Weiler, R.; Lanser, L.; Barket, R.; Rangger, L.; Schapfl, A.; Schaber, M.; Fritsche, G.; Wöll, E.; Weiss, G. Prevalence and Predictive Value of Anemia and Dysregulated Iron Homeostasis in Patients with COVID-19 Infection. J. Clin. Med. 2020, 9, 2429. [Google Scholar] [CrossRef] [PubMed]
- Carpagnano, G.E.; Di Lecce, V.; Quaranta, V.N.; Zito, A.; Buonamico, E.; Capozza, E.; Palumbo, A.; Di Gioia, G.; Valerio, V.N.; Resta, O. Vitamin D deficiency as a predictor of poor prognosis in patients with acute respiratory failure due to COVID-19. J. Endocrinol. Investig. 2021, 44, 765–771. [Google Scholar] [CrossRef] [PubMed]
- D’Avolio, A. 25-Hydroxyvitamin D Concentrations Are Lower in Patients with Positive PCR for SARS-CoV-2. Nutrients 2020, 12, 1359. [Google Scholar] [CrossRef]
- Dahan, S.; Segal, G.; Katz, I.; Hellou, T.; Tietel, M.; Bryk, G.; Amital, H.; Shoenfeld, Y.; Dagan, A. Ferritin as a Marker of Severity in COVID-19 Patients: A Fatal Correlation. Isr. Med Assoc. J. 2020, 22, 494–500. [Google Scholar] [PubMed]
- Im, J.H.; Je, Y.S.; Baek, J.; Chung, M.-H.; Kwon, H.Y.; Lee, J.-S. Nutritional status of patients with COVID-19. Int. J. Infect. Dis. 2020, 100, 390–393. [Google Scholar] [CrossRef]
- Jothimani, D.; Kailasam, E.; Danielraj, S.; Nallathambi, B.; Ramachandran, H.; Sekar, P.; Manoharan, S.; Ramani, V.; Narasimhan, G.; Kaliamoorthy, I.; et al. COVID-19: Poor outcomes in patients with zinc deficiency. Int. J. Infect. Dis. 2020, 100, 343–349. [Google Scholar] [CrossRef]
- Karahan, S.; Katkat, F. Impact of Serum 25(OH) Vitamin D Level on Mortality in Patients with COVID-19 in Turkey. J. Nutr. Health Aging 2021, 25, 189–196. [Google Scholar] [CrossRef]
- Liu, J.; Han, P.; Wu, J.; Gong, J.; Tian, D. Prevalence and predictive value of hypocalcemia in severe COVID-19 patients. J. Infect. Public Health 2020, 13, 1224–1228. [Google Scholar] [CrossRef] [PubMed]
- Macaya, F.; Espejo Paeres, C.; Valls, A.; Fernandez-Ortiz, A.; Gonzalez Del Castillo, J.; Martin-Sanchez, F.J.; Runkle, I.; Rubio Herrera, M.A. Interaction between age and vitamin D deficiency in severe COVID-19 infection. Nutr. Hosp. 2020, 37, 1039–1042. [Google Scholar] [CrossRef]
- Maghbooli, Z.; Sahraian, M.A.; Ebrahimi, M.; Pazoki, M.; Kafan, S.; Tabriz, H.M.; Hadadi, A.; Montazeri, M.; Nasiri, M.; Shirvani, A.; et al. Vitamin D sufficiency, a serum 25-hydroxyvitamin D at least 30 ng/mL reduced risk for adverse clinical outcomes in patients with COVID-19 infection. PLoS ONE 2020, 15, e0239799. [Google Scholar] [CrossRef] [PubMed]
- Mardani, R.; Alamdary, A.; Nasab, S.M.; Gholami, R.; Ahmadi, N. Association of vitamin D with the modulation of the disease severity in COVID-19. Virus Res. 2020, 289, 198148. [Google Scholar] [CrossRef]
- Meltzer, D.O.; Best, T.J.; Zhang, H.; Vokes, T.; Arora, V.; Solway, J. Association of Vitamin D Status and Other Clinical Characteristics With COVID-19 Test Results. JAMA Netw. Open 2020, 3, e2019722. [Google Scholar] [CrossRef]
- Merzon, E.; Tworowski, D.; Gorohovski, A.; Vinker, S.; Cohen, A.G.; Green, I.; Frenkel-Morgenstern, M. Low plasma 25(OH) vitamin D level is associated with increased risk of COVID-19 infection: An Israeli population-based study. FEBS J. 2020, 287, 3693–3702. [Google Scholar] [CrossRef]
- Moghaddam, A.; Heller, R.A.; Sun, Q.; Seelig, J.; Cherkezov, A.; Seibert, L.; Hackler, J.; Seemann, P.; Diegmann, J.; Pilz, M.; et al. Selenium Deficiency Is Associated with Mortality Risk from COVID-19. Nutrients 2020, 12, 2098. [Google Scholar] [CrossRef]
- Panagiotou, G.; Tee, S.A.; Ihsan, Y.; Athar, W.; Marchitelli, G.; Kelly, D.; Boot, C.S.; Stock, N.; Macfarlane, J.; Martineau, A.R.; et al. Low serum 25-hydroxyvitamin D (25[OH]D) levels in patients hospitalized with COVID-19 are associated with greater disease severity. Clin. Endocrinol. 2020, 93, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Pizzini, A.; Aichner, M.; Sahanic, S.; Böhm, A.; Egger, A.; Hoermann, G.; Kurz, K.; Widmann, G.; Bellmann-Weiler, R.; Weiss, G.; et al. Impact of Vitamin D Deficiency on COVID-19—A Prospective Analysis from the CovILD Registry. Nutrients 2020, 12, 2775. [Google Scholar] [CrossRef]
- Radujkovic, A. Vitamin D Deficiency and Outcome of COVID-19 Patients. Nutrients 2020, 12, 2757. [Google Scholar] [CrossRef]
- Raisi-Estabragh, Z.; McCracken, C.; Bethell, M.S.; Cooper, J.; Cooper, C.; Caulfield, M.J.; Munroe, P.B.; Harvey, N.C.; Petersen, S.E. Greater risk of severe COVID-19 in Black, Asian and Minority Ethnic populations is not explained by cardiometabolic, socioeconomic or behavioural factors, or by 25(OH)-vitamin D status: Study of 1326 cases from the UK Biobank. J. Public Health 2020, 42, 451–460. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.; Lara, O.D.; O’Cearbhaill, R.; Knisely, A.; Mceachron, J.; Gabor, L.; Carr, C.; Blank, S.; Prasad-Hayes, M.; Frey, M.; et al. Inflammatory markers in gynecologic oncology patients hospitalized with COVID-19 infection. Gynecol. Oncol. 2020, 159, 618–622. [Google Scholar] [CrossRef]
- Sonnweber, T.; Boehm, A.; Sahanic, S.; Pizzini, A.; Aichner, M.; Sonnweber, B.; Kurz, K.; Koppelstätter, S.; Haschka, D.; Petzer, V.; et al. Persisting alterations of iron homeostasis in COVID-19 are associated with non-resolving lung pathologies and poor patients’ performance: A prospective observational cohort study. Respir. Res. 2020, 21, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Chen, X.; Cai, Y.; Xia, J.; Zhou, X.; Xu, S.; Huang, H.; Zhang, L.; Zhou, X.; Du, C.; et al. Risk Factors Associated With Acute Respiratory Distress Syndrome and Death in Patients With Coronavirus Disease 2019 Pneumonia in Wuhan, China. JAMA Intern. Med. 2020, 180, 934–943. [Google Scholar] [CrossRef] [PubMed]
- Yasui, Y.; Yasui, H.; Suzuki, K.; Saitou, T.; Yamamoto, Y.; Ishizaka, T.; Nishida, K.; Yoshihara, S.; Gohma, I.; Ogawa, Y. Analysis of the predictive factors for a critical illness of COVID-19 during treatment—Relationship between serum zinc level and critical illness of COVID-19. Int. J. Infect. Dis. 2020, 100, 230–236. [Google Scholar] [CrossRef]
- Ye, K.; Tang, F.; Liao, X.; Shaw, B.A.; Deng, M.; Huang, G.; Qin, Z.; Peng, X.; Xiao, H.; Chen, C.; et al. Does Serum Vitamin D Level Affect COVID-19 Infection and Its Severity?-A Case-Control Study. J. Am. Coll. Nutr. 2020. [Google Scholar] [CrossRef]
- Zhao, K.; Huang, J.; Dai, D.; Feng, Y.; Liu, L.; Nie, S. Serum Iron Level as a Potential Predictor of Coronavirus Disease 2019 Severity and Mortality: A Retrospective Study. Open Forum Infect. Dis. 2020, 7, ofaa250. [Google Scholar] [CrossRef]
- Zhou, F.; Yu, T.; Du, R.; Fan, G.; Liu, Y.; Liu, Z.; Xiang, J.; Wang, Y.; Song, B.; Gu, X.; et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: A retrospective cohort study. Lancet 2020, 395, 1054–1062. [Google Scholar] [CrossRef]
- Capone, S.; Abramyan, S.; Ross, B.; Rosenberg, J.; Zeibeq, J.; Vasudevan, V.; Samad, R.; Gerolemou, L.; Pinelis, E.; Gasperino, J.; et al. Characterization of Critically Ill COVID-19 Patients at a Brooklyn Safety-Net Hospital. Cureus 2020, 12, e9809. [Google Scholar] [CrossRef] [PubMed]
- Castillo, M.E.; Costa, L.M.E.; Barrios, J.M.V.; Díaz, J.F.A.; Miranda, J.L.; Bouillon, R.; Gomez, J.M.Q. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef]
- Fasano, A. COVID-19 in Parkinson’s Disease Patients Living in Lombardy, Italy. Mov. Disord. 2020, 35, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Tan, C.W.; Ho, L.P.; Kalimuddin, S.; Cherng, B.P.Z.; Teh, Y.E.; Thien, S.Y.; Wong, H.M.; Tern, P.J.W.; Chandran, M.; Chay, J.W.M.; et al. Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B12 in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrients 2020, 79–80, 111017. [Google Scholar] [CrossRef]
- Aranow, C. Vitamin D and the Immune System. J. Investig. Med. 2011, 59, 881–886. [Google Scholar] [CrossRef]
- Diabetes, T.L. Vitamin D and COVID-19: Why the Controversy? Elsevier: Amsterdam, The Netherlands, 2021. [Google Scholar]
- National Institute for Health and Care Excellence; Public Health England, and Scientific Advisory Commitee on Nutrition. COVID-19 Rapid Guideline: Vitamin D 2020. Available online: https://www.nice.org.uk/guidance/ng187 (accessed on 16 March 2021).
- Martineau, A.R.; Jolliffe, D.A.; Hooper, R.L.; Greenberg, L.; Aloia, J.F.; Bergman, P.; Dubnov-Raz, G.; Esposito, S.; Ganmaa, D.; Ginde, A.A.; et al. Vitamin D supplementation to prevent acute respiratory tract infections: Systematic review and meta-analysis of individual participant data. BMJ 2017, 356, i6583. [Google Scholar] [CrossRef] [PubMed]
- Ge, X.; Guo, Y.; Cheng, J.; Hu, R.; Feng, X. Epidemiology and Seasonality of Respiratory Viruses Detected from Children with Respiratory Tract Infections in Wuxi, East China. Med Sci. Monit. 2018, 24, 1856–1862. [Google Scholar] [CrossRef]
- Moan, J.E.; Dahlback, A.; Ma, L.; Juzeniene, A. Influenza, solar radiation and vitamin D. Derm. Endocrinol. 2009, 1, 308–310. [Google Scholar] [CrossRef] [PubMed]
- Forrest, K.Y.; Stuhldreher, W.L. Prevalence and correlates of vitamin D deficiency in US adults. Nutr. Res. 2011, 31, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Laird, E.; Walsh, J.B.; Lanham-New, S.; O’Sullivan, M.; Kenny, R.A.; Scully, H.; Crowley, V.; Healy, M. A High Prevalence of Vitamin D Deficiency Observed in an Irish South East Asian Population: A Cross-Sectional Observation Study. Nutrients 2020, 12, 3674. [Google Scholar] [CrossRef]
- Sze, S. Ethnicity and clinical outcomes in COVID-19: A systematic Review and Meta-analysis. EClinicalMedicine 2020, 29–30, 100630. [Google Scholar] [CrossRef]
- Thompson, D.-C.; Barbu, M.-G.; Beiu, C.; Popa, L.G.; Mihai, M.M.; Berteanu, M.; Popescu, M.N. The Impact of COVID-19 Pandemic on Long-Term Care Facilities Worldwide: An Overview on International Issues. BioMed Res. Int. 2020, 2020, 8870249. [Google Scholar] [CrossRef]
- Ali, N. Role of vitamin D in preventing of COVID-19 infection, progression and severity. J. Infect. Public Health 2020, 13, 1373–1380. [Google Scholar] [CrossRef] [PubMed]
- Holford, P.; Carr, A.C.; Jovic, T.H.; Ali, S.R.; Whitaker, I.S.; Marik, P.E.; Smith, A.D. Vitamin C—An Adjunctive Therapy for Respiratory Infection, Sepsis and COVID-19. Nutrients 2020, 12, 3760. [Google Scholar] [CrossRef]
- ClinicalTrials.Gov. Studies Found for Zinc (recruiting, not yet recruiting, completed) interventional studies for COVID-19 and SARS-CoV-2. 2021. Available online: https://clinicaltrials.gov/ct2/results?term=Zinc&cond=Covid-19&recrs=b&recrs=a&recrs=f&recrs=e&age_v=&gndr=&type=Intr&rslt=&Search=Apply (accessed on 15 March 2021).
- Wessells, K.R.; King, J.C.; Brown, K.H. Development of a Plasma Zinc Concentration Cutoff to Identify Individuals with Severe Zinc Deficiency Based on Results from Adults Undergoing Experimental Severe Dietary Zinc Restriction and Individuals with Acrodermatitis Enteropathica. J. Nutr. 2014, 144, 1204–1210. [Google Scholar] [CrossRef]
- Kell, D.B.; Pretorius, E. Serum ferritin is an important inflammatory disease marker, as it is mainly a leakage product from damaged cells. Metallomics 2014, 6, 748–773. [Google Scholar] [CrossRef] [PubMed]
- World Health Organisation. Who Guideline on Use of Ferritin Concentrations to Assess Iron Status in Individuals and Populations; 2020. Available online: https://www.who.int/publications/i/item/9789240000124 (accessed on 16 March 2021).
- Petersen, E. Comparing SARS-CoV-2 with SARS-CoV and influenza pandemics. Lancet Infect. Dis. 2020, 20, e238–e244. [Google Scholar] [CrossRef]
- Petrosillo, N.; Viceconte, G.; Ergonul, O.; Ippolito, G.; Petersen, E. COVID-19, SARS and MERS: Are they closely related? Clin. Microbiol. Infect. 2020, 26, 729–734. [Google Scholar] [CrossRef]
- Benskin, L.L. A basic review of the preliminary evidence that COVID-19 risk and severity is increased in vitamin D deficiency. Front. Public Health 2020, 8, 513. [Google Scholar] [CrossRef] [PubMed]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Köstenberger, M.; Berisha, A.T.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D deficiency 2.0: An update on the current status worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef] [PubMed]
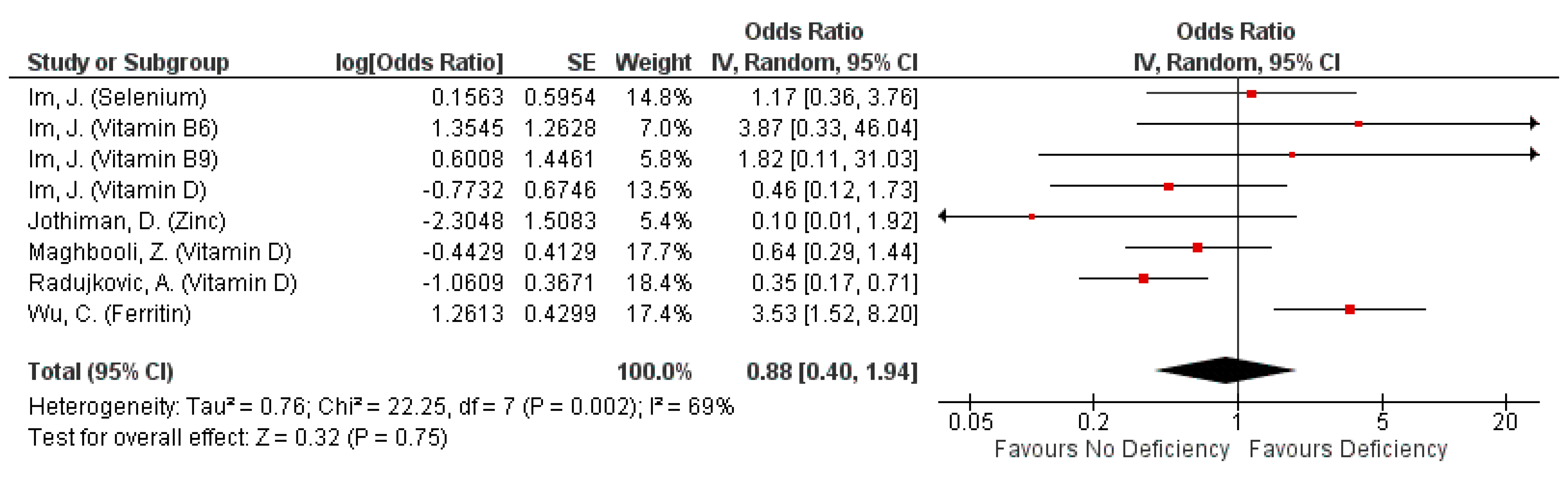
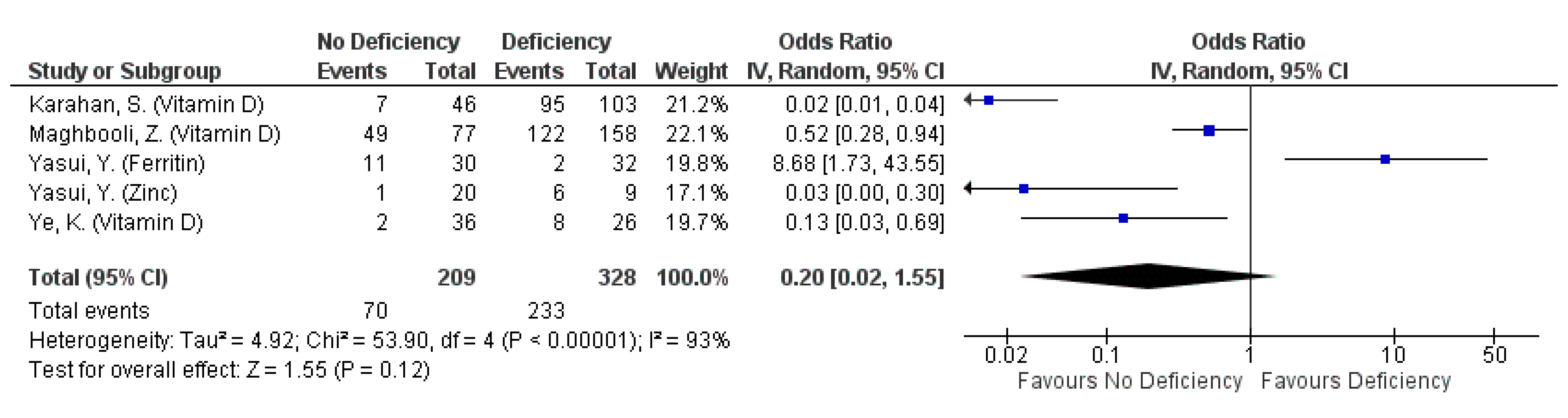
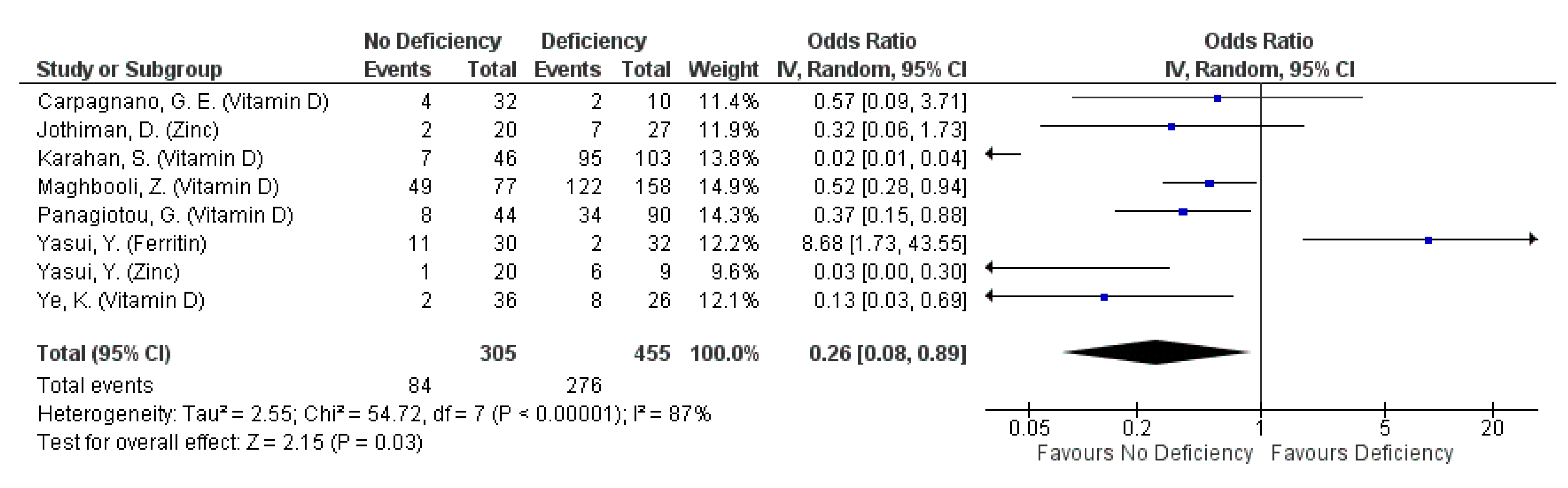
| Author, Year [Ref.] | Reported Study Design, Country of Study | Population Source: Period of Data Collection | Population Size (% Men); # Subjects with ≥1 Comorbidity | Mean Age in Years (SD) | Micronutrient in Question | Blood Sampling Timepoint | Micronutrient Categories, as Defined by Study: Cut-off for Each Category, as Defined by Study | Outcome Reported [I: Incidence of COVID-19 Episode; S, Severity of COVID-19 Episode] | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Quintile 1 | Quintile 2 | Quintile 3 | ||||||||
| Baktash, 2020 [20] | Prospective cohort; UK | Hospital: 1 Mar–30 Apr 2020 | 105 (54.3); 54 | Cases (deficient): 79.46 (89.52) Cases (non-deficient): 81.16 (7.23) Controls: 83.44 (8.08) | Vitamin D | At admission | NA: measured as a continuous outcome |
| ||
| Bellmann-Weiler, 2020 [21] | Retrospective cohort; Austria | Hospital: 25 Feb–20 May 2020 | 259 (60.6);152 | 68 (53–80) * | Iron | At admission (Day 1 ± 1) | No Iron deficiency (ID) | Absolute ID: TSAT <20% + Serum ferritin <100 µg/L | Functional ID: TSAT <20% + Serum ferritin >100 µg/L |
|
| Carpagnano, 2020 [22] | Retrospective cohort; Italy | Hospital: 11 Mar–30 Apr 2020 | 42 (71.4); 36 | ≥30 ng/mL: 64 (18) 30 >Vitamin D ≥20 ng/mL: 64 (13) 20 > Vitamin D ≥10 ng/mL: 60 (6.9) <10 ng/mL: 74 (11) | Vitamin D | ≤12 h following RICU admission | NR: ≥10 ng/mL | NR: <10 ng/mL | - |
|
| D’Avolio, 2020 [23] | Retrospective cohort; Switzerland | Hospital: 1 Mar–14 Apr 2019, 1 Mar–14 Apr 2020 | 107 (54.2); NR | 73 (63–81) * | Vitamin D | <7 weeks after PCR result, overall median days from result = 3.0 (IQR:0.0 to 7.0) | NA: measured as a continuous outcome |
| ||
| NR (controls): 1 March to 14 April 2019 | 1377 (45.3); NR | 63 (46–76) * | ||||||||
| Dahan, 2020 [24] | Cross-sectional; Israel | Hospital: 21 Feb–30 Mar 2020 | 39 (59); 22 | 52.46 (2.76) | Iron (Serum ferritin) | On admission | NA: measured as a continuous outcome |
| ||
| Hastie, 2020 [17] | Retrospective cohort; UK | Health registries: 5 Mar–25 Apr 2020 | 341,484 (NR); NR | 37–73 (between 2006–2010) # | Vitamin D | Baseline measurement taken between 2006–2010 | Sufficient: ≥50 nmol/L | Insufficient: <50 nmol/L | Deficient: <25 nmol/L |
|
| NA: measured as a continuous outcome (per 10 nmol/L) | ||||||||||
| Im, 2020 [25] | Case-control; South Korea | Hospital: Feb–Jun 2020 | Cases: 50 (42); NR Controls: 150 (sex-matched to cases); NR NR | Cases: 57.5 (34.5–68) Controls: NR. but age-matched to controls | Vitamin D3 | ≤7 days of admission (median number of days from admission = 2 days) | No deficiency | Deficiency: ≤20 ng/dL | Severe Deficiency: ≤10 ng/dL |
|
| Vitamin B6 | Deficiency: ≤5 µg/L | - |
| |||||||
| Vitamin B9 | Deficiency: ≤4 µg/L | - | ||||||||
| Selenium | Deficiency: ≤95 µg/L | - | ||||||||
| ≥1 deficiency | Any of the above | - | ||||||||
| Jothimani, 2020 [26] | Prospective case-control; India | Hospital: 17 May–27 May 2020 | Cases: 47 (61.5); NR Controls: 45 (67.7); 0 | Cases: 34 (18–77) * Controls: 32 (18–60) * | Zinc a | 6 h from admission | No deficiency | Deficiency: ≤80 µg/L | - |
|
| Karahan, 2020 [27] | Retrospective cohort; Turkey | Hospital: 1 Apr–20 May 2020 | 149 (54.4); 85 | 63.5 (15.3) | Vitamin D3 | NR | Normal: ≥ 30 ng/mL | Insufficiency: 21–29 ng/mL | Deficiency: ≤20 ng/mL |
|
| Liu, 2020 [28] | Retrospective cohort; China | Hospital: 9 Feb–15 Feb 2020 (follow-up till 25 Feb 2020) | 107 (49); 40 | 68 (61–76) * | Calcium (Serum Calcium) | ≤24 h from admission | Normal: 2.15–2.5 mmol/L | Hypocalcemia: <2.15 mmol/L | - |
|
| Macaya, 2020 [29] | Retrospective cohort; Spain | Hospital: 5 Mar–31 Mar 2020 | 80 (43.8); 50 | non-severe patients: 63 (50–72) * severe patients: 75 (66–84) * | Vitamin D3 | At admission or ≤3 months before admission | No deficiency: ≥20 ng/mL | Deficiency: <20 ng/mL | - |
|
| Maghbooli, 2020 [30] | Cross-sectional; Iran | Hospital: Till 1 May 2020 (start date unspecified) | 235 (61.3); NR | 58.72 (15.22) | Vitamin D3 | At admission | No deficiency: ≥30 ng/mL | Deficiency/ Insufficiency: <30 ng/mL | - |
|
| Mardani, 2020 [31] | Cross-sectional; Iran | Medical Center: Mar 2020 | 123 (52.8); NR | 42 (NR) | Vitamin D | At admission | NA: measured as a continuous outcome |
| ||
| Meltzer, 2020 [32] | Retrospective cohort; United States | Hospital: 3 Mar–10 Apr 2020 | 489 (25); 261 | 49.2 (18.4) | Vitamin D | Baseline measurement taken within 1 year to 14 days before patient’s COVID-19 test | No deficiency: ≥20 ng/mL OR ≥18 pg/mL | Deficiency: <20 ng/mL | - |
|
| Merzon, 2020 [33] | Retrospective cohort; Israel | Medical Center: 1 Feb–30 Apr 2020 | 7807 (41.4); 2136 | COVID-19 positive: 35.58 (0.56) COVID-19 negative: 47.35 (0.24) | Vitamin D | NR | Sufficiency: ≥30 ng/mL | Insufficiency: 29–20 ng/mL | Deficiency: <20 ng/mL |
|
| Normal: ≥30 ng/mL | Low: < 30 ng/mL | - | ||||||||
| NA: measured as a continuous outcome |
| |||||||||
| Moghaddam, 2020 [34] | Cross-sectional; Germany | Hospital: NR | 33 (42.4); 22 | 77 (38–94) * | Selenium (Serum Selenium) | Throughout hospitalisation; mean (SD) samples drawn per patient = 5.03 (4.27) | Normal: 45.7–131.6 μg/L | Deficiency: <45.7 μg/L | - |
|
| NA: measured as a continuous outcome | ||||||||||
| Panagiotou, 2020 [35] | Cross-sectional; UK | Hospital: NR | 134 (54.5); 114 | severe: 61.1 (11.8) mild: 76.4 (14.9) | Vitamin D | NR | Normal: ≥50 nmol/L | Deficient: <50 nmol/L | - |
|
| Pizzini, 2020 [36] | Prospective cohort; Austria | Medical Center: From 29 Apr 2020 (end date unspecified) | 109 (60); 88 | 58 (14) | Vitamin D | 8 weeks after COVID-19 diagnosis | NA: measured as a continuous outcome |
| ||
| Calcium (Total, ionised) | NA: measured as a continuous outcome | |||||||||
| Iron (Serum ferritin) | NA: measured as a continuous outcome | |||||||||
| Radujkovic, 2020 [37] | Prospective cohort; Germany | Hospital: 18 Mar–18 Jun 2020 | 185 (51); 77 | 60 (49–70) * | Vitamin D | At admission and SARS-CoV-2 testing | No deficiency: ≥12 ng/mL | Deficiency: <12 ng/mL (<30 nM) | - |
|
| No insufficiency: ≥20 ng/mL | Insufficiency: <20 ng/mL | - |
| |||||||
| NA: measured as a continuous outcome |
| |||||||||
| Raisi-Estabragh, 2020 [38] | Prospective cohort; UK | Health registries: 16 Mar–18 May 2020 | 4510 (48.8); 2081 | COVID-19 positive: 68.11 (9.23) COVID-19 negative: 68.91 (8.72) | Vitamin D | Baseline measurement taken between 2006–2010 | NA: measured as a continuous outcome |
| ||
| Smith, 2020 [39] | Retrospective multi-centre cohort; United States | Hospital: 1 May–30 Mar 2020 | 86 (0); 86 | 68.5 (59–74.8) | Iron (Ferritin) | At admission | NA: measured as a continuous outcome |
| ||
| Sonnweber, 2020 [40] | Prospective multi-centre cohort; Austria | Hospital: NR | 109 (60); 88 | 58 (14) | Iron (Ferritin) | 60 days (SD ± 12) after the onset of first COVID-19 symptoms | NA: measured as a continuous outcome |
| ||
| Sun, 2020 [19] | Retrospective cohort; China | NR: NR | 63 (58.7); 12 | Median: 47 (Range: 3–85) | Iron (Serum Ferritin) | On admission | NA: measured as a continuous outcome |
| ||
| Wu, 2020 [41] | Retrospective cohort; China | Hospital: 25 Dec 2019–26 Jan 2020 (follow-up till 13 Feb 2020) | 201 (63.7); 39 | 51 (43–60) * | Iron (Serum Ferritin) | ≤24 h from admission | NR: >300 ng/mL | NR: ≤300 ng/mL | - |
|
| NA: measured as a continuous outcome | ||||||||||
| Yasui, 2020 [42] | Retrospective cohort; Japan | Health Center: 24 Mar–24 May 2020 | 62 (54.8); | NR, but 17 (27.4%) are aged ≥65 years | Iron (Ferritin) | Multiple timepoints: on first day of hospitalisation and 2–3 days later on an empty stomach in the early morning after hospitalisation | NR: ≥300 ng/mL (male), ≥200 ng/mL (female) | NR: <300 ng/mL (male), <200 ng/mL (female) | - |
|
| NA: measured as a continuous outcome | ||||||||||
| Zinc | NR: ≥70 µg/dL | NR: <70 µg/dL | - | |||||||
| NA: measured as a continuous outcome | ||||||||||
| Ye, 2020 [43] | Case-control; China | Hospital: 16 Feb–16 Mar 2020 | Cases: 62 (37); 16 Controls: 80 (42); 0 | Cases: 43 (32–59) * Controls: 42 (31–52) * | Vitamin D | At admission | Sufficiency: ≥75 nmol/L | Insufficiency: 50 nmol/L ≤25(OH)D <75 nmol/L | Deficiency: <50 nmol/L |
|
| Non-deficiency: ≥50 nmol/L | Deficiency: <50 nmol/L | - |
| |||||||
| NA: measured as a continuous outcome | ||||||||||
| Zhao, 2020 [44] | Retrospective cohort; China | Hospital: 1 Feb–29 Feb 2020 | 50 (60); 17 | 55 (44–66) * | Iron (Pre- and post-treatment serum iron) | NR | NA: measured as a continuous outcome |
| ||
| Zhou, 2020 [45] | Retrospective cohort; China | Hospital: 29 Dec 2019–31 Jan 2020 | 191 (62.3); 91 | Survivor: 52 (45–58) * Non-survivor: 69 (63–76) * | Iron (Serum Ferritin) | NR: frequency of examinations determined by treating physician | NR: >300 ng/mL | NR: ≤300 ng/mL | - |
|
| NA: measured as a continuous outcome | ||||||||||
| Author, Year [Ref.] | Reported Study Design; Country of Study | Population Source: Period of Data Collection | Population Size (% Men); Number of Subjects with ≥1 Comorbidity | Mean Age (SD) | Micronutrient in Question | Outcome Reported [I: Incidence of COVID-19 Episode; S, Severity of COVID-19 Episode] |
|---|---|---|---|---|---|---|
| Annweiler, 2020 [18] | Retrospective cohort; France | Nursing Home: Mar–Apr 2020 | 66 (22.7); 66 | 87.7 (9.0) | Vitamin D3 a |
|
| Capone, 2020 [46] | Retrospective cohort; United States | Hospital: till 20 Apr 2020 (start date unspecified) | 102 (53.9); 61 | 63.22 (53.3–74.3) * | Vitamin C & zinc b |
|
| Castillo, 2020 [47] | Open-label, double blind randomised controlled trial; Spain | Hospital: Unspecified | 76 (58.2); 26 | 53 (10) | Vitamin D3 c |
|
| Fasano, 2020 [48] | Retrospective, single-center case-control; Italy | Health registry: NR | 1486 (56.9); 1486 | Case: 70.5 (10.1) Controls: 73.0 (9.5) | Vitamin D b |
|
| Macaya, 2020 [29] | Retrospective cohort; Spain | Hospital: 5 Mar–31 Mar 2020 | 80 (43.8); 50 | non-severe patients: 63 (50–72) * severe patients: 75 (66–84) * | Vitamin D b |
|
| Tan, 2020 [49] | Retrospective cohort; Singapore | Hospital: 15 Jan–15 Apr 2020 | 43 (60.4); 24 | Non-supplemented: 64.1 (7.9) Supplemented: 58.4 (7) | Vitamin D, Magnesium & Vitamin B12 d |
|
| Author [Ref] | Micronutrient in question | Odds Ratio (OR) (95% Confidence Interval); % Population Infected (Infected/Population Size) | Key Findings | ||||
|---|---|---|---|---|---|---|---|
| Supplementation | Deficiency | ||||||
| Supplemented | Non-supplemented | Quintile 1 | Quintile 2 | Quintile 3 | |||
| Baktash [20] | Vitamin D |
| |||||
| D’Avolio [23] | Vitamin D |
| |||||
| Fasano [48] | Vitamin D | 0.56 (0.32- 0.99) *,a; 4.0% (13/329) | Reference; 8.0% (92/1157) * |
| |||
| Im [25] | Vitamin D3 | NR; 12.4% (12/97) # | NR; 32.5% (26/80) # | NR; 52.2% (12/23) # |
| ||
| Mardani [31] | Vitamin D |
| |||||
| Meltzer [32] | Vitamin D | NR; 12.3% (39/317) | NR; 18.6% (32/172) |
| |||
| Merzon [33] | Vitamin D ᶧ | Reference; 69.4% (79/1139) # | 1.59 (1.29–2.02); 10.6% (598/5648) # | 1.58 (1.13–2.09); 10.3% (105/1020)# |
| ||
| Vitamin D ᶲ | Reference; 69.4% (79/1139) | 1.50 (1.13–1.98) b; 10.5% (703/6668) * | |||||
| Raisi-Estabragh [38] | Vitamin D |
| |||||
| Ye [43] | Vitamin D | Reference; 35.6% (36/101) # | 3.13 (1.47–6.66); 63.4% (26/41) # |
| |||
| Jothimani [26] | Zinc | NR; 33.3% (20/60) # | NR; 84.4% (27/32) # |
| |||
| Author [Ref] | Micronutrient in Question | Reported Summary Risk Estimate: Odds Ratio (OR) (95% Confidence Interval)/Mean (SD); % Population Infected (Infected/Population Size) | Key Findings | ||||
|---|---|---|---|---|---|---|---|
| Supplementation | Deficiency | ||||||
| Supplemented | Non-Supplemented | Quintile 1 | Quintile 2 | Quintile 3 | |||
| Outcome: Death due to COVID-19 episode/during hospitalisation due to COVID-19 episode | |||||||
| Annweiler [18] | Vitamin D3 | 0.11 (0.03–0.48) *,a; 17.5% (10/57) | Reference; 55.6% (5/9) * |
| |||
| Carpagnano [22] | Vitamin D | NR; 3.1% (1/32) # | NR; 20% (2/10) # |
| |||
| Castillo [47] | Vitamin D | NR; 0% (0/50) # | NR; 7.7% (2/26) # |
| |||
| Hastie [17] | Vitamin D | Reference | 1.21 (0.83–1.76) b | 1.02 (0.75–1.38) b |
| ||
| Karahan [27] | Vitamin D3 | NR; 0% (0/12) * | NR; 14.7% (5/34) * | NR; 62.1% (64/103) * |
| ||
| Mardani [31] | Vitamin D |
| |||||
| Panagiotou [35] | Vitamin D | Reference; NR | 0.97 (0.42, 2.23); NR |
| |||
| Radujkovic [37] | Vitamin D ᶧ | Reference, NR | For all subjects: 14.73 (4.16–52.19) c; NR For inpatients only: 11.51 (3.24–40.92) c; NR |
| |||
| Vitamin D ᶲ | Reference, NR | For all subjects: 11.27 (1.48–85.55) c; NR For inpatients only: 7.97 (1.05–60.60) c; NR |
| ||||
| Tan [49] | Vitamin D, Magnesium & Vitamin B12 | NR; 0% (0/17) # | NR; 0% (0/26) # |
| |||
| Capone [46] | Vitamin C & Zinc |
| |||||
| Jothimani [26] | Zinc | Reference; 0% (0/20) | 5.48 (0.61–49.35); 18.5% (5/27) |
| |||
| Bellmann-Weiler [21] | Iron | Reference; NR | 0.458 (0.082–2.572); NR | 0.418 (0.15–1.165); NR |
| ||
| Wu [41] | Iron (Serum Ferritin) | 5.28 (0.72–38.48); NR | Reference; NR |
| |||
| Zhao [44] | Iron (Serum iron, Pre-treatment) |
| |||||
| Iron (Serum iron, Post-treatment) |
| ||||||
| Zhou [45] | Iron (Serum Ferritin) | 9.1 (2.04–40.58); 43.1% (44/102) * | Reference; 7.7% (2/26) * |
| |||
| Moghaddam [34] | Selenium (Serum) | NR; 13.0% (12/92) # | NR; 29.7% (22/74) # |
| |||
| Outcome: Hospitalisation Duration (Unit: days) | |||||||
| Carpagnano [22] | Vitamin D | 12.5 (8–20.5) #,f | 8 (6–11.25) #,f |
| |||
| Maghbooli [30] | Vitamin D3 | 5 (1–19) g | 5 (1–23) g |
| |||
| Jothimani [26] | Zinc | 5.7 (NR) * | 7.9 (NR) * |
| |||
| Outcome: Intensive care unit (ICU) Admission | |||||||
| Bellmann-Weiler [21] | Iron | Reference; NR | 0.147 (0.017–1.297); NR | 0.556 (0.225–1.373); NR |
| ||
| Carpagnano [22] | Vitamin D | NR; 12.5% (4/32) # | NR; 20% (2/10) # |
| |||
| Castillo [47] | Vitamin D | 0.03 (0.003–0.25) e; 2% (1/50) * | Reference; 50% (13/26) * |
| |||
| Maghbooli [30] | Vitamin D3 | NR; 14.3% (11/77) | NR; 20.9% (33/158) |
| |||
| Panagiotou [35] | Vitamin D | NR; 18.2% (8/44) * | NR; 37.8% (34/90) * |
| |||
| Jothimani [26] | Zinc | Reference; 10% (2/20) | 3.15 (0.58–17.67); 25.9% (7/27) |
| |||
| Author [Ref] | Micronutrient in Question | Outcome Definition (Study-Specific) | Reported Summary Risk Estimate: Odds Ratio (OR) (95% Confidence Interval); % Population Infected (Infected/Population Size) | Key Findings | ||||
|---|---|---|---|---|---|---|---|---|
| Supplementation | Deficiency | |||||||
| Supplemented | Non-supplemented | Quintile 1 | Quintile 2 | Quintile 3 | ||||
| Outcome: Clinical severity | ||||||||
| Hastie [17] | Vitamin D | Hospitalisation rate | Reference; NR | 1.06 (0.89–1.26) a; NR | 1.1 (0.88–1.37) a; NR |
| ||
| Merzon [33] | Vitamin D | Hospitalisation rate |
| |||||
| Radujkovic [37] | Vitamin D | Hospitalisation rate | NR; 44.4% (64/144) * | NR; 70.7% (29/41) * |
| |||
| Karahan [27] | Vitamin D3 | Chinese Clinical Guideline (Moderate disease) | NR; 100% (12/12) * | NR; 79.4% (27/34) * | NR; 7.8% (8/103) * |
| ||
| Chinese Clinical Guideline (Severe/critical disease) | NR; 0% (0/12) * | NR; 18.9% (7/37)* | NR; 92.2% (95/103) | |||||
| Maghbooli [30] | Vitamin D3 | CDC criteria (Severe/critical disease) | Reference; 63.6% (49/77) * | 1.59 (1.05–2.41) *,b; 77.2% (122/158) * |
| |||
| Ye [43] | Vitamin D | Chinese National Health Commission Guidelines (6th edition) (Mild/moderate disease) | Reference; 88.9% (32/36) # | NR; 69.2% (18/26) # |
| |||
| Chinese National Health Commission Guidelines (6th edition) (Severe/critical disease) | Reference; 5.6% (2/36) # | 15.18 (1.23–187.45) c; 30.8% (8/26) # | ||||||
| Number of symptomatic patients | NR; 80% (8/10) | NR; 100% (26/26) | NR; 100% (26/26) |
| ||||
| Pizzini [36] | Vitamin D | Requiring hospitalisation, respiratory support or intensive care treatment |
| |||||
| Calcium (total, ionised) |
| |||||||
| Iron (Serum Ferritin) |
| |||||||
| Dahan [24] | Iron (Serum Ferritin) | Report of the WHO-China Joint Mission |
| |||||
| Sun [19] | Iron (Serum Ferritin) | New Coronavirus Pneumonia Prevention and Control Program, 7th edition |
| |||||
| Zhao [44] | Iron (Serum) | Chinese National Health Commission Guidelines (7th edition) |
| |||||
| Smith [39] | Iron (Ferritin) | Hospitalisation and/or ICU admission, requiring mechanical ventilation and/or death |
| |||||
| Sonnweber [40] | Iron/Ferritin | ICU admission, requiring oxygen therapy or respiratory support |
| |||||
| Yasui [42] | Iron (Ferritin) | ICU admission, requiring oxygen therapy or respiratory support | All patients: NR; 36.7% (11/30) * Subset of inpatient: NR; 28.6% (6/21) | All patients: NR; 6.3% (2/32) * Subset of inpatient: NR; 12.5% (1/8) |
| |||
| Zinc | Subset of inpatient: NR; 5% (1/20) * | Subset of inpatient: NR; 66.7% (6/9) * |
| |||||
| Jothimani [26] | Zinc | Number of symptomatic patients | Reference; 90% (18/20) | 3.15 (0.58–17.67); 96.3% (26/27) |
| |||
| Outcome: Progression to respiratory-related complication | ||||||||
| Jothimani [26] | Zinc | ARDS development | NR; 0% (0/20) | NR; 18.5% (5/27) |
| |||
| Wu [41] | Iron (Serum Ferritin) | ARDS development | 3.53 (1.52-8-16); NR | Reference; NR |
| |||
| Maghbooli [30] | Vitamin D3 | ARDS development | NR; 11.7% (9/77) | NR; 17.1% (27/158) |
| |||
| Im [25] | Vitamin D3 | Pneumonia incidence, or requiring high-flow nasal cannula, mechanical ventilator, and extracorporeal membrane oxygenation or death | NR; 50% (6/12) # | NR; 68.4% (26/38) # |
| |||
| Vitamin B6 | NR; 66% (31/47) # | NR; 33.3% (1/3) # | ||||||
| Vitamin B9 | NR; 64.6% (31/48) # | NR; 50% (1/2) # | ||||||
| Selenium | NR; 65.5% (19/29) # | NR 61.9% (13/21) # | ||||||
| ≥1 deficiency | NR; 44.4% (4/9) # | NR; 68.3% (28/41) # | ||||||
| Capone [46] | Vitamin C & Zinc | Requiring invasive mechanical ventilation |
| |||||
| Radujkovic [37] | Vitamin D | Requiring any form of oxygen therapy | NR; 35.4% (54/144) * | NR;63.4% (26/41) * |
| |||
| Tan [49] | Vitamin D, Magnesium & Vitamin B12 | Requiring oxygen therapy | 0.195 (0.041–0.926) d, 0.182 (0.038–0.859) e; 11.7% (2/17) * | Reference; 30.8% (8/26) * |
| |||
| Outcome: Composite outcome (with multiple outcomes) | ||||||||
| Liu [28] | Calcium (Serum calcium) | Need for mechanical ventilation, ICU admission due to COVID-19 episode, or all-cause mortality during admission | Reference; 25% (10/40) * | 2.962 (1.085–8.090) f; 47.8% (32/67) * |
| |||
| Macaya [29] | Vitamin D3 α | Death, ICU admission or requiring high flow oxygen (greater than nasal cannula) | Reference; 31.4% (11/35) | 3.2 (0.9–11.4) g; 44.4% (20/45) |
| |||
| Vitamin D3 β | NR; 45.5% (20/44) | NR; 20.6% (11/36) |
| |||||
| Radujkovic [37] | Vitamin D3 ᶧ | Mechanical invasive ventilation and/or death from COVID-19 episode | For all subjects: Reference; NRFor inpatients only: Reference; 87.5% (56/64) # | For all subjects: 6.12(2.79–13.42) h; NR For inpatients only: 4.65 (2.11–10.25) h; 89.7% (26/29) # |
| |||
| Vitamin D3 ᶲ | Reference; NR | For all subjects: 5.75 (1.73–19.09) h; NR For inpatients only: 3.99 (1.2–13.28) h; NR |
| |||||
| Tan [49] | Vitamin D, Magnesium & Vitamin B12 | Requiring oxygen therapy or ICU admission due to COVID-19 episode | NR; 5.9% (1/17) * | NR; 30.8% (8/26) * |
| |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, M.X.; Gwee, S.X.W.; Pang, J. Micronutrients Deficiency, Supplementation and Novel Coronavirus Infections—A Systematic Review and Meta-Analysis. Nutrients 2021, 13, 1589. https://doi.org/10.3390/nu13051589
Wang MX, Gwee SXW, Pang J. Micronutrients Deficiency, Supplementation and Novel Coronavirus Infections—A Systematic Review and Meta-Analysis. Nutrients. 2021; 13(5):1589. https://doi.org/10.3390/nu13051589
Chicago/Turabian StyleWang, Min Xian, Sylvia Xiao Wei Gwee, and Junxiong Pang. 2021. "Micronutrients Deficiency, Supplementation and Novel Coronavirus Infections—A Systematic Review and Meta-Analysis" Nutrients 13, no. 5: 1589. https://doi.org/10.3390/nu13051589
APA StyleWang, M. X., Gwee, S. X. W., & Pang, J. (2021). Micronutrients Deficiency, Supplementation and Novel Coronavirus Infections—A Systematic Review and Meta-Analysis. Nutrients, 13(5), 1589. https://doi.org/10.3390/nu13051589






