Caffeine Consumption Habits of New Zealand Tertiary Students
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Experimental Procedures
2.4. Caffeine Consumption Habits (CaffCo) Questionnaire
2.5. Statistical Analysis
3. Results
3.1. Participants
3.2. Caffeine Intake
3.3. Exceeding the Adverse Effect Level (3 mg·kgbw−1·day−1)
3.4. Exceeding the Suggested ‘Safe Limit’ (400 mg·day−1)
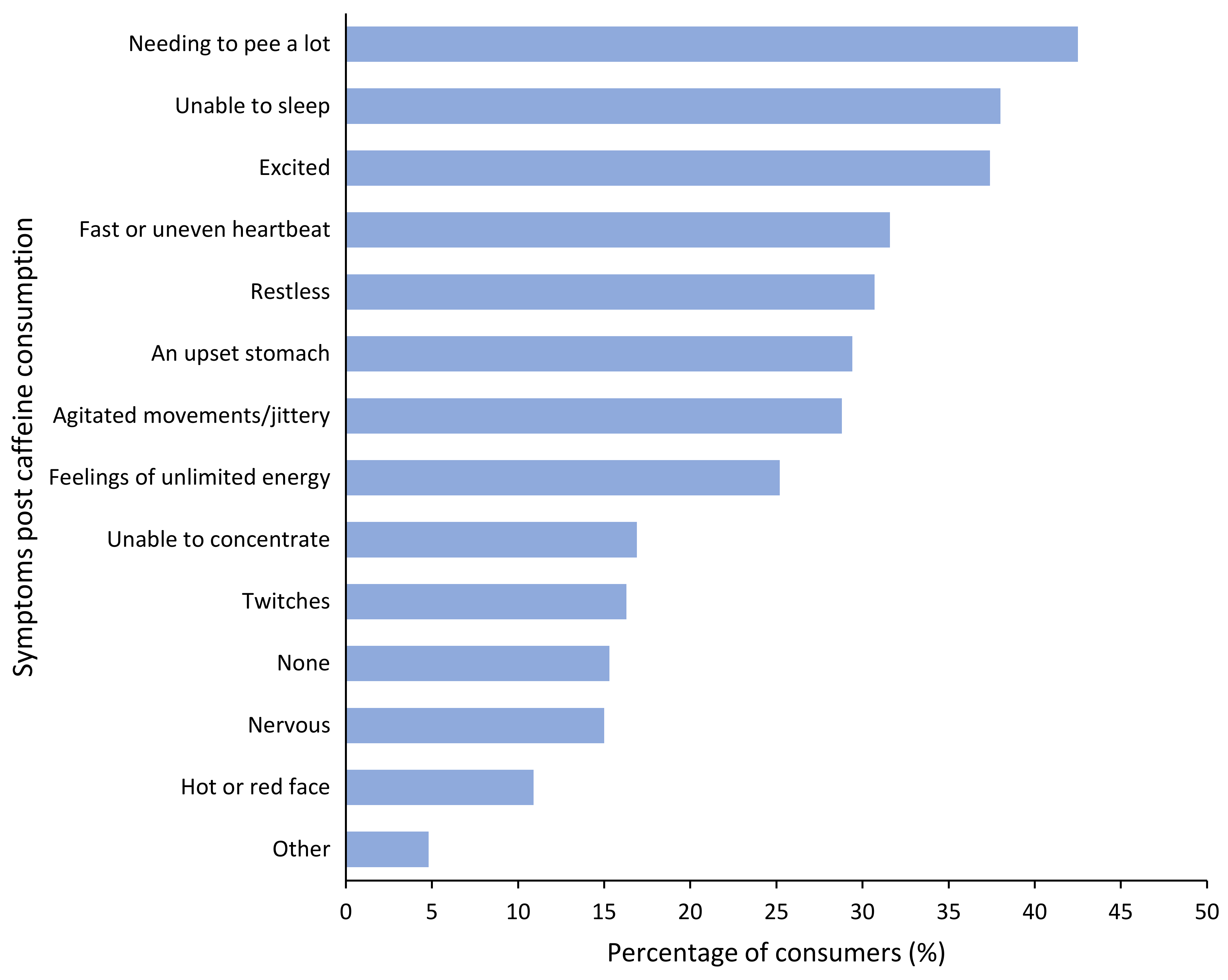
3.5. Perceived ‘Adverse Symptoms’ Post Caffeine Consumption
3.6. Caffeine Dependence
3.7. Withdrawal Symptoms
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weinberg, B.A.; Bealer, B.K. The World of Caffeine: The Science and Culture of the World’s Most Popular Drug; Routledge: London, UK, 2001. [Google Scholar]
- Heckman, M.A.; Weil, J.; de Mejia, E.G. Caffeine (1,3,7-trimethylxanthine) in foods: A comprehensive review on consumption, functionality, safety, and regulatory matters. J. Food Sci. 2010, 75, R77–R87. [Google Scholar] [CrossRef]
- Thomson, B.M.; Schiess, S. Risk Profile: Caffeine in Energy Drinks and Energy Shots. In New Zealand Food Safety Authority under Project CFS/09.04; 2010. Available online: https://www.mpi.govt.nz/dmsdocument/25706/direct (accessed on 30 January 2019).
- Barone, J.J.; Roberts, H.R. Caffeine consumption. Food Chem. Toxicol. 1996, 34, 119–129. [Google Scholar] [CrossRef]
- Frary, C.D.; Johnson, R.K.; Wang, M.Q. Food sources and intakes of caffeine in the diets of persons in the United States. J. Am. Diet. Assoc. 2005, 105, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Olmos, V.; Bardoni, N.; Ridolfi, A.S.; Villaamil Lepori, E.C. Caffeine levels in beverages from Argentina’s market: Application to caffeine dietary intake assessment. Food Addit. Contam. Part A Chem. Anal. Control Exp. Risk Assess. 2009, 26, 275–281. [Google Scholar] [CrossRef]
- Yamada, M.; Sasaki, S.; Murakami, K.; Takahashi, Y.; Okubo, H.; Hirota, N.; Notsu, A.; Todoriki, H.; Miura, A.; Fukui, M.; et al. Estimation of caffeine intake in Japanese adults using 16 d weighed diet records based on a food composition database newly developed for Japanese populations. Public Health Nutr. 2010, 13, 663–672. [Google Scholar] [CrossRef]
- Bunker, M.L.; McWilliams, M. Caffeine content of common beverages. J. Am. Diet. Assoc. 1979, 74, 28–32. [Google Scholar] [PubMed]
- Desbrow, B.; Henry, M.; Scheelings, P. An examination of consumer exposure to caffeine from commercial coffee and coffee-flavoured milk. J. Food Compost Anal. 2012, 28, 114–118. [Google Scholar] [CrossRef]
- Matissek, R. Evaluation of xanthine derivatives in chocolate–nutritional and chemical aspects. Z. Lebensm. Unters. Forch. 1997, 205, 175–184. [Google Scholar] [CrossRef]
- Food Standards Australia New Zealand. Food Standards Code; 2015. Available online: https://www2.health.vic.gov.au/public-health/food-safety/food-businesses/food-standards-code (accessed on 30 January 2019).
- World Health Organisation; The New Zealand Ministry of Health. Dietary Supplements Regulations 1985 (SR 1985/208); New Zealand Government: Wellington, New Zealand, 2010.
- University of Otago and Ministry of Health. A Focus on Nutrition: Key Findings of the 2008/2009 New Zealand Adult Nutrition Survey; Ministry of Health: Wellington, New Zealand, 2011.
- Wong, J.E.; Parnell, W.R.; Black, K.E.; Skidmore, P.M. Reliability and relative validity of a food frequency questionnaire to assess food group intakes in New Zealand adolescents. Nutr. J. 2012, 11. [Google Scholar] [CrossRef]
- Sundborn, G.; Gentles, D.; Metcalf, P. Carbonated beverage consumption in New Zealand adults. PAC Health Dialog. 2014, 20, 87–88. [Google Scholar]
- Australian Beverages Council. Caffeine: The Facts. 2013. Available online: http://australianbeverages.org/for-consumers/caffeine-facts/ (accessed on 30 January 2019).
- Thomson, B.M.; Campbell, D.M.; Cressey, P.; Egan, U.; Horn, B. Energy drink consumption and impact on caffeine risk. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess. 2014, 31, 1476–1488. [Google Scholar] [CrossRef]
- Cheng, M.; Hu, Z.; Lu, X.; Huang, J.; Gu, D. Caffeine intake and atrial fibrillation incidence: Dose response meta-analysis of prospective cohort studies. Can. J. Cardiol. 2014, 30, 448–454. [Google Scholar] [CrossRef] [PubMed]
- Crippa, A.; Discacciati, A.; Larsson, S.; Wolk, A.; Orsini, N. Coffee consumption and mortality from all causes, cardiovascular disease, and cancer: A dose-response meta-analysis. Am. J. Epidemiol. 2014, 180, 763–775. [Google Scholar] [CrossRef] [PubMed]
- Nawrot, P.; Jordan, S.; Eastwood, J.; Rotstein, J.; Hugenholtz, A.; Feeley, M. Effects of caffeine on human health. Food Addit. Contam. 2003, 20, 1–30. [Google Scholar] [CrossRef]
- Taylor, M.R. FDA to investigate added caffeine. FDA Consumer, 15 November 2013. [Google Scholar]
- Lorist, M.M.; Tops, M. Caffeine, fatigue, and cognition. Brain Cogn. 2003, 53, 82–94. [Google Scholar] [CrossRef]
- Puckeridge, M.; Fulcher, B.D.; Phillips, A.; Robinson, P.A. Incorporation of caffeine into a quantitative model of fatigue and sleep. J. Theor. Biol. 2011, 273, 44–54. [Google Scholar] [CrossRef] [PubMed]
- EFSA Panel on Dietetic Products and Nutrition and Allergies (NDA). Scientific opinion on the substantiation of health claims related to caffeine and increase in physical performance during short-term high-intensity exercise (ID 737, 1486, 1489), increase in endurance performance (ID 737, 1486), increase in endurance capacity (ID 1488) and reduction in the rated perceived exertion/effort during exercise (ID 1488, 1490) pursuant to Article 13(1) of Regulation (EC) No 1924/2006. EFSA J. 2011, 9, 2053. [Google Scholar]
- Cohen, J.; Cohen, P.; West, S.; Aiken, L.S. Applied Multiple Regression/Correlation Analysis for the Behavioral Sciences; Routledge: London, UK, 2013. [Google Scholar]
- Seifert, S.M.; Schaechter, J.L.; Hershorin, E.R.; Lipshultz, S.E. Health effects of energy drinks on children, adolescents, and young adults. Pediatrics 2011, 127, 511–528. [Google Scholar] [CrossRef]
- Yang, A.; Palmer, A.A.; de Wit, H. Genetics of caffeine consumption and responses to caffeine. Psychopharmacology 2010, 211, 245–257. [Google Scholar] [CrossRef]
- Abernethy, D.R.; Todd, E.L. Impairment of caffeine clearance by chronic use of low-dose oestrogen-containing oral contraceptives. Eur. J. Clin. Pharmacol. 1985, 28, 425–428. [Google Scholar] [CrossRef]
- Aranda, J.V.; Collinge, J.M.; Zinman, R.; Watters, G. Maturation of caffeine elimination in infancy. Arch. Dis. Child 1979, 54, 946–949. [Google Scholar] [CrossRef] [PubMed]
- Carrillo, J.A.; Benitez, J. CYP1A2 activity, gender and smoking, as variables influencing the toxicity of caffeine. Br. J. Clin. Pharmacol. 1996, 41, 605–608. [Google Scholar] [CrossRef] [PubMed]
- Fisher, C.D.; Lickteig, A.J.; Augustine, L.M.; Ranger-Moore, J.; Jackson, J.P.; Ferguson, S.S.; Cherrington, N.J. Hepatic cytochrome P450 enzyme alterations in humans with progressive stages of nonalcoholic fatty liver disease. Drug Metab. Dispos. 2009, 37, 2087–2094. [Google Scholar] [CrossRef]
- Kalow, W.; Tang, B.K. Caffeine as a metabolic probe: Exploration of the enzyme-inducing effect of cigarette smoking. Clin. Pharmacol. Ther. 1991, 49, 44–48. [Google Scholar] [CrossRef]
- Krul, C.; Hageman, G. Analysis of urinary caffeine metabolites to assess biotransformation enzyme activities by reversed-phase high-performance liquid chromatography. J. Chromatogr. B Biomed. Sci. Appl. 1998, 709, 27–34. [Google Scholar] [CrossRef]
- Sachse, C.; Brockmöller, J.; Bauer, S.; Roots, I. Functional significance of a C→A polymorphism in intron 1 of the cytochrome P450 CYP1A2 gene tested with caffeine. Br. J. Clin. Pharmacol. 1999, 47, 445–449. [Google Scholar] [CrossRef]
- Tsutsumi, K.; Kotegawa, T.; Matsuki, S.; Tanaka, Y.; Ishii, Y.; Kodama, Y.; Kuranari, M.; Miyakawa, I.; Nakano, S. The effect of pregnancy on cytochrome P4501A2, xanthine oxidase, and N-acetyltransferase activities in humans. Clin. Pharmacol. Ther. 2001, 70, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.R.; McHugh, P.; Holtzman, S. Alcohol and drug abuse: Caffeine and schizophrenia. Psychiatr. Serv. 1998, 49, 1415–1417. [Google Scholar] [CrossRef]
- Pelchovitz, D.J.; Goldberger, J.J. Caffeine and cardiac arrhythmias: A review of the evidence. Am. J. Med. 2011, 124, 284–289. [Google Scholar] [CrossRef]
- Ferré, S. An update on the mechanisms of the psychostimulant effects of caffeine. J. Neurochem. 2008, 105, 1067–1079. [Google Scholar] [CrossRef]
- Center for Behavioural Health Statistics and Quality. The DAWN Report; Update on Emergency Department Visits Involving Energy Drinks: A Continuing Public Health Concern; Substance Abuse and Mental Health Service Administration: Rockville, MD, USA, 2013.
- Gunja, N.; Brown, J.A. Energy drinks: Health risks and toxicity. Med. J. Aust. 2012, 196, 46–49. [Google Scholar] [CrossRef]
- Stachyshyn, S. Caffeine Consumption Habits, Motivations, and Experiences of New Zealand Tertiary Students. Master’s Thesis, Massey University, Albany, New Zealand, 2017. [Google Scholar]
- Rowe, K.; Wham, C.A.; Rutherfurd-Markwick, K.J.; Ali, A. CaffCo: A valid and reliable tool to assess caffeine consumption habits, caffeine expectancies, and caffeine withdrawal effects in adults. J. Caffeine Adenosine Res. 2020, 10, 154–160. [Google Scholar] [CrossRef]
- Qualtrics. Qualtrics Software (Version 3.673s). 2015. Available online: http://qualtrics.com (accessed on 30 January 2019).
- Rowe, K. Caffeine Intake, Influences and Experiences: The Development of CaffCo: A New Zealand Caffeine Consumption Habits Questionnaire. Master’s Thesis, Massey University, Albany, New Zealand, 2015. [Google Scholar]
- Cochran, W.G. Some methods for strengthening the common χ2 tests. Biometrics 1954, 10, 417–451. [Google Scholar] [CrossRef]
- Yates, D.; Moore, D.; McCabe, G. The Practice of Statistics; Freeman: New York, NY, USA, 1999. [Google Scholar]
- Fritz, C.O.; Morris, P.E.; Richler, J.J. Effect size estimates: Current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012, 141, 2–18. [Google Scholar] [CrossRef] [PubMed]
- Field, A. Discovering Statistics Using IBM SPSS Statistics; Sage: London, UK, 2013. [Google Scholar]
- Fulgoni Iii, V.L.; Keast, D.R.; Lieberman, H.R. Trends in intake and sources of caffeine in the diets of US adults: 2001–2010. Am. J. Clin. Nutr. 2015, 101, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-H.; Human, G.P.; Fourie, J.J.; Louw, W.A.; Larson, C.O.; Joubert, G. Medical students’ use of caffeine for ‘academic purposes’ and their knowledge of its benefits, side-effects and withdrawal symptoms. S. Afr. Fam. Pract. 2009, 51, 322–327. [Google Scholar] [CrossRef][Green Version]
- Lieberman, H.; Marriott, B.; Judelson, D.; Glickman, E.; Geiselman, P.; Giles, G.; Mahoney, C. Intake of caffeine from all sources including energy drinks and reasons for use in US college students. FASEB J. 2015, 29 (Suppl. 1). [Google Scholar] [CrossRef]
- Mackus, M.; van de Loo, A.J.A.E.; Benson, S.; Scholey, A.; Verster, J.C. Consumption of caffeinated beverages and the awareness of their caffeine content among Dutch students. Appetite 2016, 103, 353–357. [Google Scholar] [CrossRef]
- Norton, T.R.; Lazev, A.B.; Sullivan, M.J. The “buzz” on caffeine: Patterns of caffeine use in a convenience sample of college students. J. Caffeine Res. 2011, 1, 35–40. [Google Scholar] [CrossRef]
- Tannous, M.; Al Kalash, Y. Prevalence of caffeinated-beverage consumption by University Students in North Lebanon. Public Health Res. 2014, 4, 173–178. [Google Scholar]
- Mahoney, C.R.; Giles, G.E.; Marriott, B.P.; Judelson, D.A.; Glickman, E.L.; Geiselman, P.J.; Lieberman, H.R. Intake of caffeine from all sources and reasons for use by college students. Clin. Nutr. 2019, 38, 668–675. [Google Scholar] [CrossRef]
- Brice, C.F.; Smith, A.P. Factors associated with caffeine consumption. Int. J. Food Sci. Nutr. 2002, 53, 55–64. [Google Scholar]
- Global Data. Global Energy Drinks Report 2015: Comprehensive Analysis of Trends in the Global Energy Drinks Market Including Long-Term Forecasts; Market Research Reports, Inc.: New York, NY, USA, 2015. [Google Scholar]
- Pettit, M.L.; DeBarr, K.A. Perceived stress, energy drink consumption, and academic performance among college students. J. Am. Coll. Health 2011, 59, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Booth, N.; Saxton, J.; Rodda, S.N. Estimates of caffeine use disorder, caffeine withdrawal, harm and help-seeking in New Zealand: A cross-sectional survey. Addict. Behav. 2020, 109, 106470. [Google Scholar] [CrossRef] [PubMed]
- Ministry of Health. Percentage of Adult Current Smokers, 2006/07–2018/19. In New Zealand Health Survey n.d. [cited 2020 24/11/2020]. Available online: https://minhealthnz.shinyapps.io/nz-health-survey-2018-19-annual-data-explorer/_w_fca0c755/_w_8ba7f72d/_w_701caea6/#!/key-indicators (accessed on 30 January 2019).
- Jones, S.C.; Barrie, L.; Berry, N. Why (not) alcohol energy drinks? A qualitative study with Australian university students. Drug Alcohol Rev. 2012, 31, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, S. The Co-Ingestion of Caffeine and Alcohol: Influences and Experiences: A Qualitative Study. Master’s Thesis, Massey University, Albany, New Zealand, 2019. [Google Scholar]
- Fulgoni, V. Intake and exposure to caffeine. Various aspects of caffeine intake in America: Analysis of NHANES. In Caffeine in Food and Dietary Supplements: Examining Safety: Workshop Summary; The National Academies Press: Washington, DC, USA, 2014; pp. 30–37. [Google Scholar]
- Romero-Corral, A.; Somers, V.K.; Sierra-Johnson, J.; Thomas, R.J.; Collazo-Clavell, M.L.; Korinek, J.; Allison, T.G.; Batsis, J.A.; Sert-Kuniyoshi, F.H.; Lopez-Jimenez, F. Accuracy of body mass index in diagnosing obesity in the adult general population. Int. J. Obes. 2008, 3, 959–966. [Google Scholar] [CrossRef] [PubMed]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders (DSM-5®); American Psychiatric Association: Arlington, VA, USA, 2013. [Google Scholar]
- Coughlin, S.S. Recall bias in epidemiologic studies. J. Clin. Epidemiol. 1990, 43, 87–91. [Google Scholar] [CrossRef]
| Variable | Participants, n = 317 n(%)/Median [IQR] |
|---|---|
| Sex | |
| Male | 148 (46.7) |
| Female | 169 (53.3) |
| Age group | |
| 16–18 years | 51 (16) |
| 19–30 years | 236 (74.4) |
| 31–50 years | 25 (7.9) |
| 51+ years | 5 (1.6) |
| Ethnicity 1 | |
| NZ European | 150 (47.3) |
| Other European | 56 (17.7) |
| Māori | 17 (5.4) |
| Asian | 128 (40.4) |
| Pacific Peoples | 22 (6.9) |
| Middle Eastern/Latin American/African | 16 (5.0) |
| Living situation | |
| Living alone | 23 (7.4) |
| Living with family | 174 (54.9) |
| Flatting with others | 108 (34.1) |
| Halls of residence | 7 (2.2) |
| Living with partner | 5 (1.6) |
| Employment status | |
| No paid employment | 211 (66.6) |
| Part-time employment | 102 (32.2) |
| Full-time employment | 4 (1.3) |
| Body mass index, BMI (kg/m2) 2 | |
| Total | 22.9 [20.8, 25.1] |
| Male | 23.4 [21.0, 26.1] |
| Female | 22.3 [20.3, 24.2] |
| Caffeine Source | Male, mg·day−1 | Female, mg·day−1 | Mann-Whitney (U) | p |
|---|---|---|---|---|
| Tea | 26.43 | 26.53 | 6207.5 | 0.996 |
| Coffee | 100.00 | 108.76 | 6825.0 | 0.496 |
| Chocolate | 8.91 | 8.88 | 8112.0 | 0.864 |
| Kola drinks | 15.31 | 9.94 | 2046.0 | <0.001 |
| Energy drinks | 32.20 | 11.54 | 1191.0 | <0.001 |
| Caffeinated RTD | 6.10 | 3.00 | 275.0 | 0.022 |
| Supplements 1 | 90.52 | 19.34 | 31.0 | 0.176 |
| Caffeine tablets | 3.30 | 6.70 | 8.5 | 0.282 |
| Caffeine Source | Proportion of Participants Who Are Dependent (%) 1 |
|---|---|
| Tea (n = 227) | 24.8 |
| Coffee (n = 242) | 59.3 |
| Chocolate (n = 259) | 19.8 |
| Kola drinks (n = 156) | 8.3 |
| Energy drinks (n = 128) | 32.8 |
| Caffeinated RTD (n = 58) | 1.7 |
| Caffeine-containing sports supplements (n = 21) | 19.0 |
| Caffeine tablets (n = 11) | 18.2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stachyshyn, S.; Ali, A.; Wham, C.; Knightbridge-Eager, T.; Rutherfurd-Markwick, K. Caffeine Consumption Habits of New Zealand Tertiary Students. Nutrients 2021, 13, 1493. https://doi.org/10.3390/nu13051493
Stachyshyn S, Ali A, Wham C, Knightbridge-Eager T, Rutherfurd-Markwick K. Caffeine Consumption Habits of New Zealand Tertiary Students. Nutrients. 2021; 13(5):1493. https://doi.org/10.3390/nu13051493
Chicago/Turabian StyleStachyshyn, Saskia, Ajmol Ali, Carol Wham, Tayla Knightbridge-Eager, and Kay Rutherfurd-Markwick. 2021. "Caffeine Consumption Habits of New Zealand Tertiary Students" Nutrients 13, no. 5: 1493. https://doi.org/10.3390/nu13051493
APA StyleStachyshyn, S., Ali, A., Wham, C., Knightbridge-Eager, T., & Rutherfurd-Markwick, K. (2021). Caffeine Consumption Habits of New Zealand Tertiary Students. Nutrients, 13(5), 1493. https://doi.org/10.3390/nu13051493









