Can Mixed Strains of Lactobacillus and Bifidobacterium Reduce Eczema in Infants under Three Years of Age? A Meta-Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources and Literature Search
2.2. Inclusion and Exclusion Criteria
2.3. Quality Assessment
2.4. Data Extraction
2.5. Data Synthesis and Statistical Analyses
3. Results
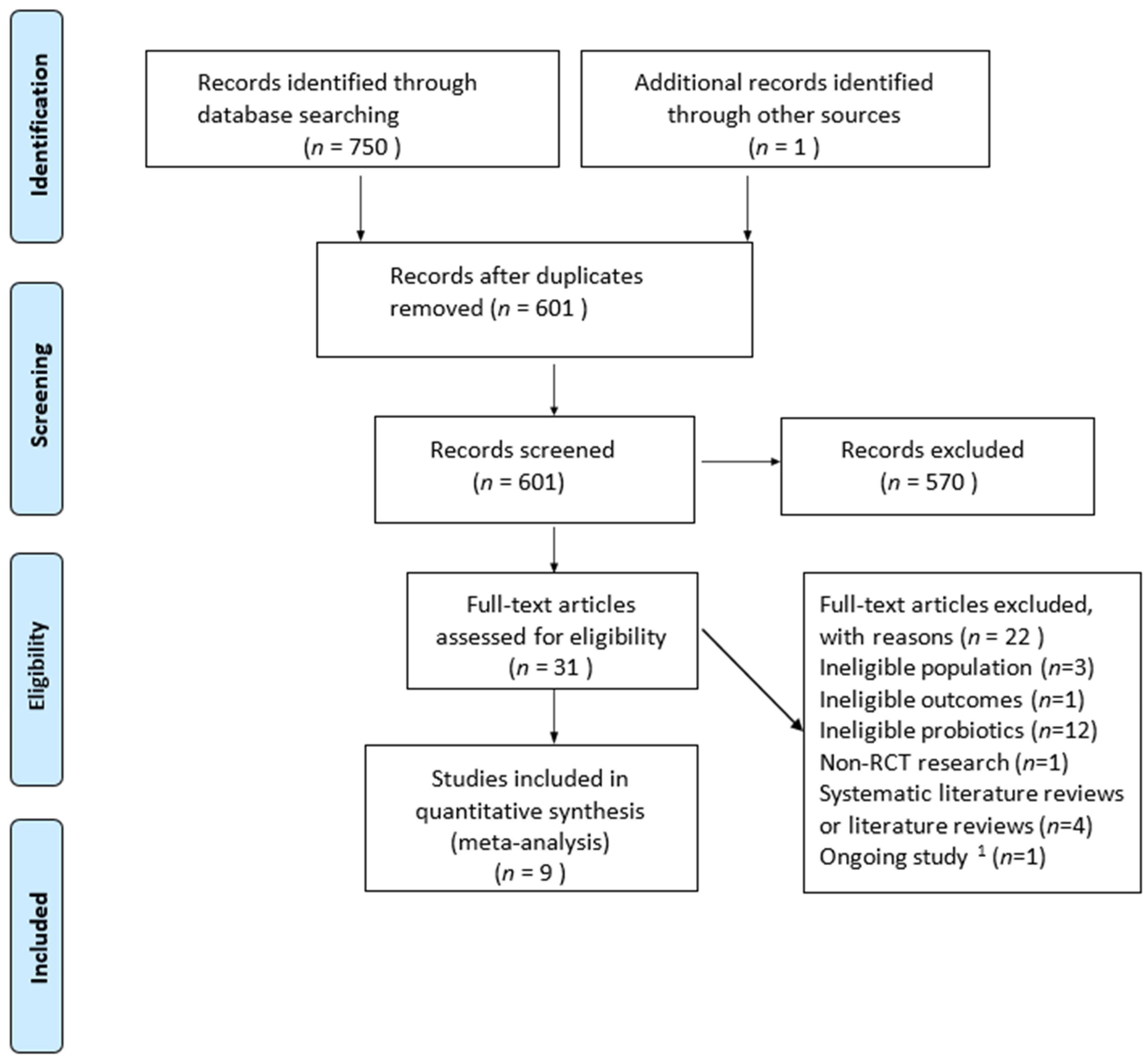
3.1. Search Results
3.2. Study Characteristics
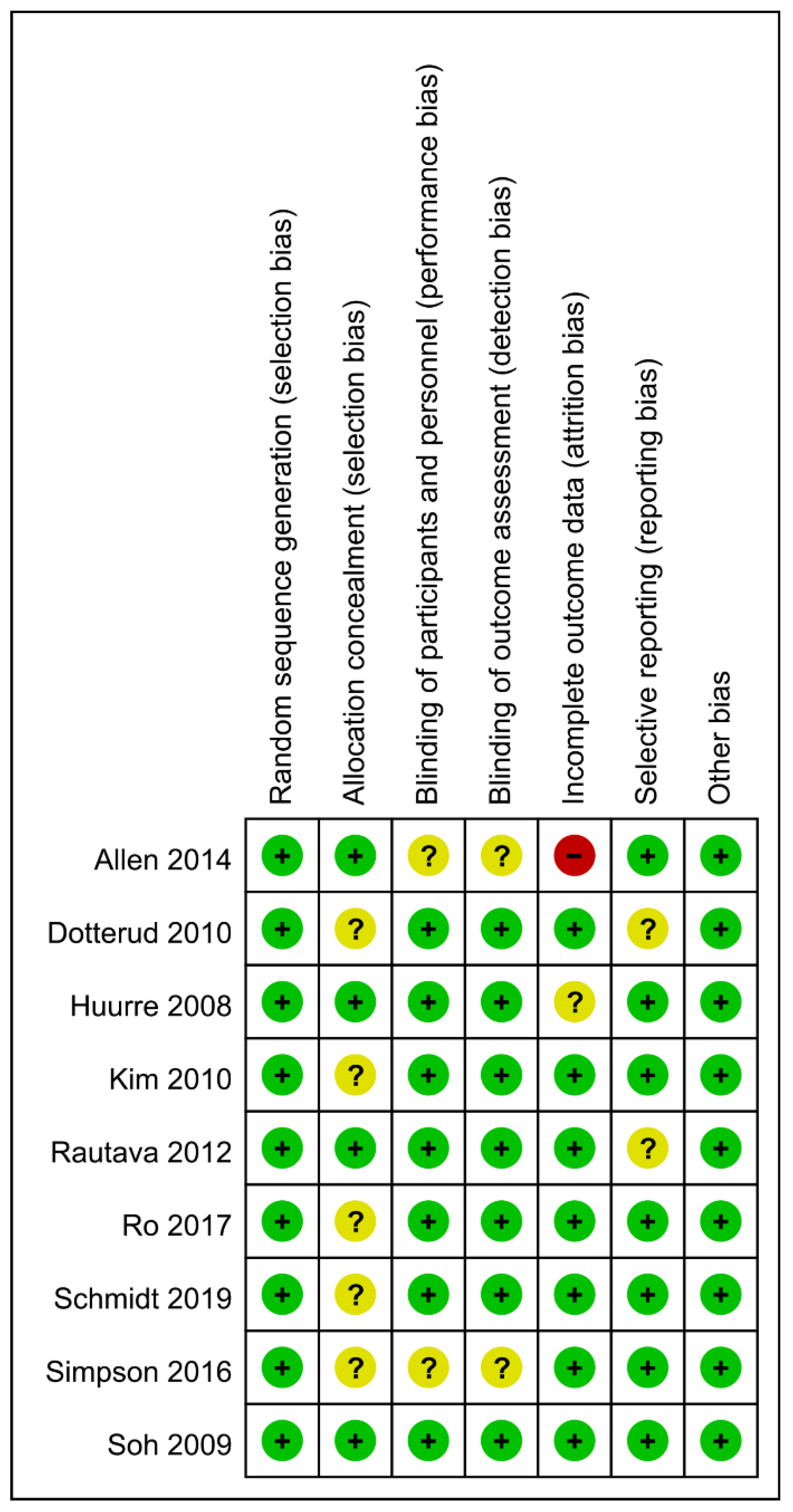
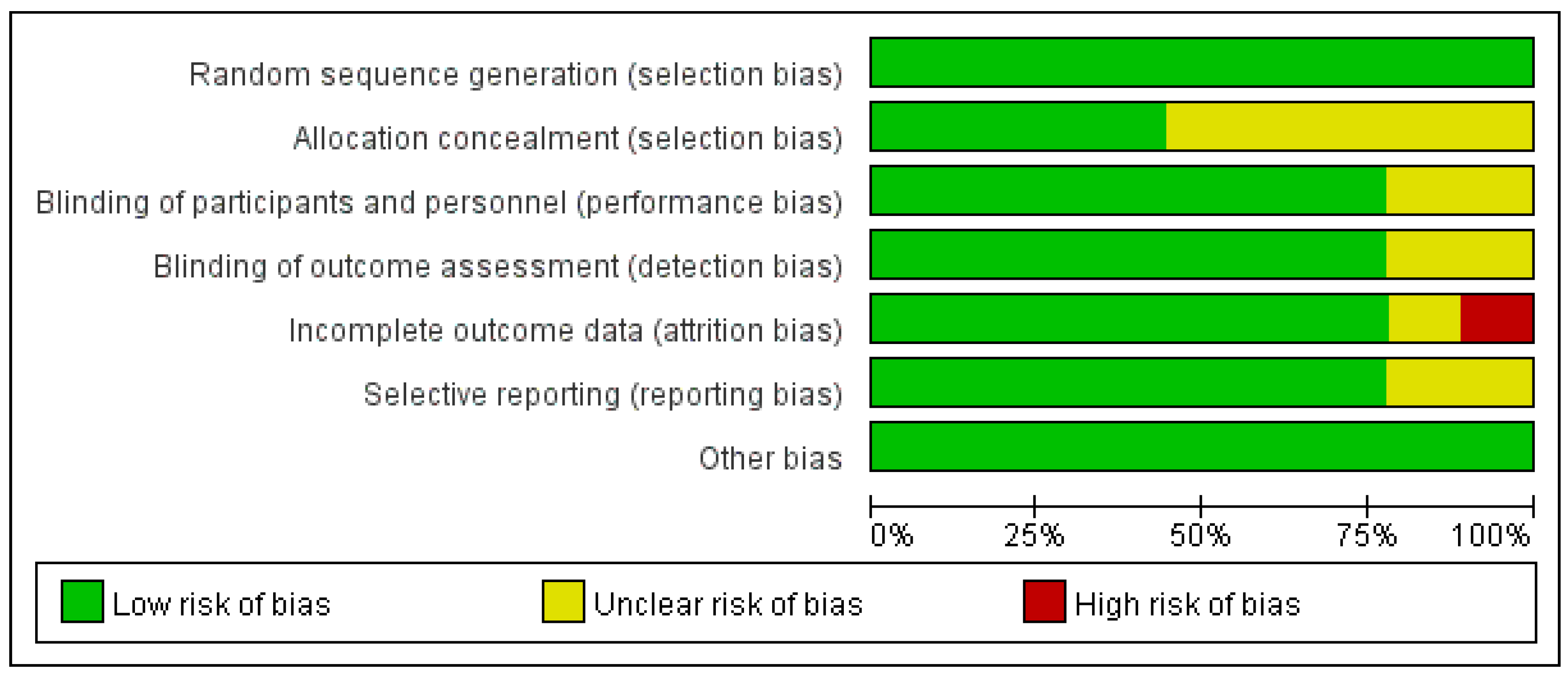
3.3. Bias Risk Assessment
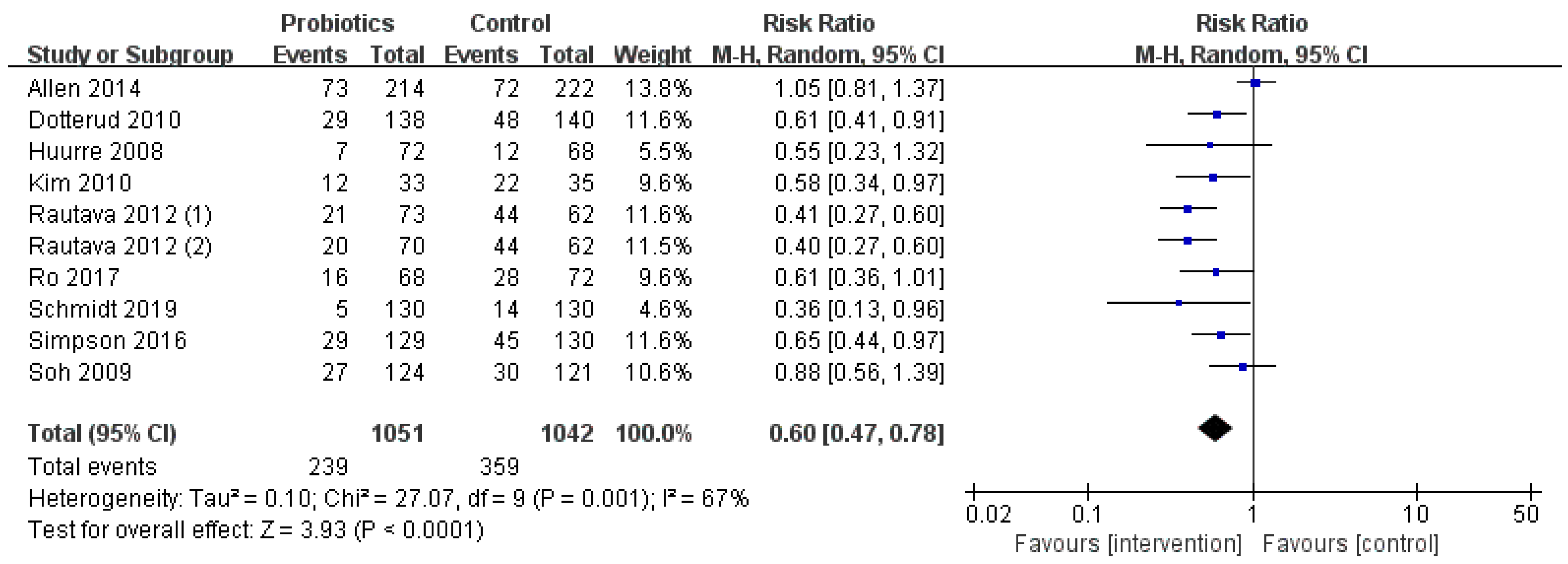
3.4. Effect of Lactobacillus and Bifidobacterium on Prevention of Infant Eczema
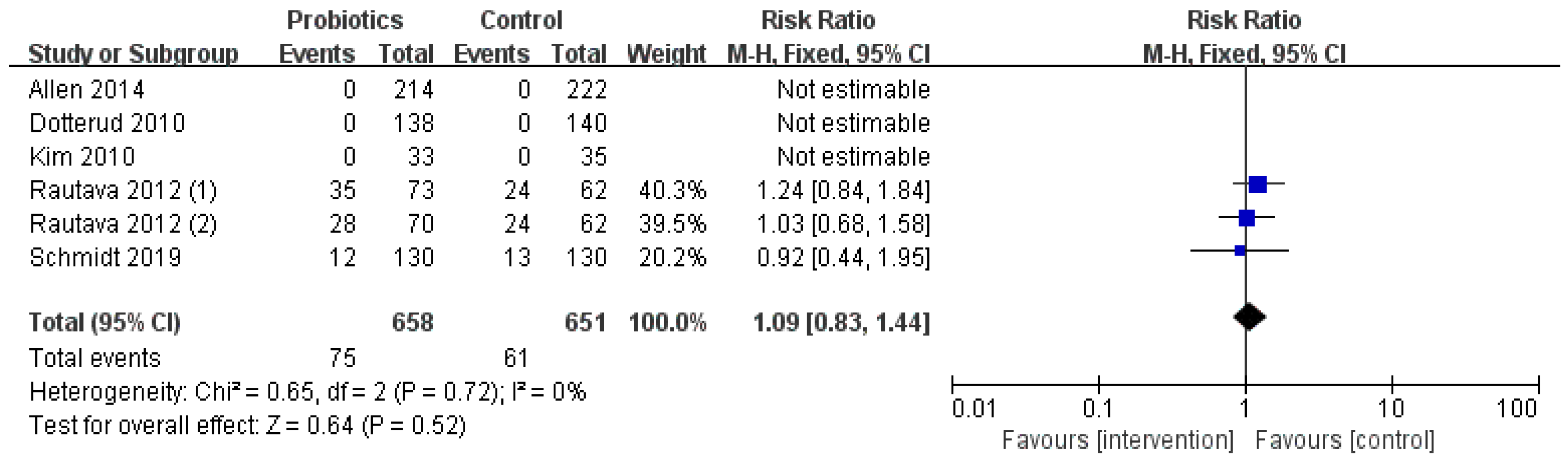
3.5. Occurrence of Adverse Events
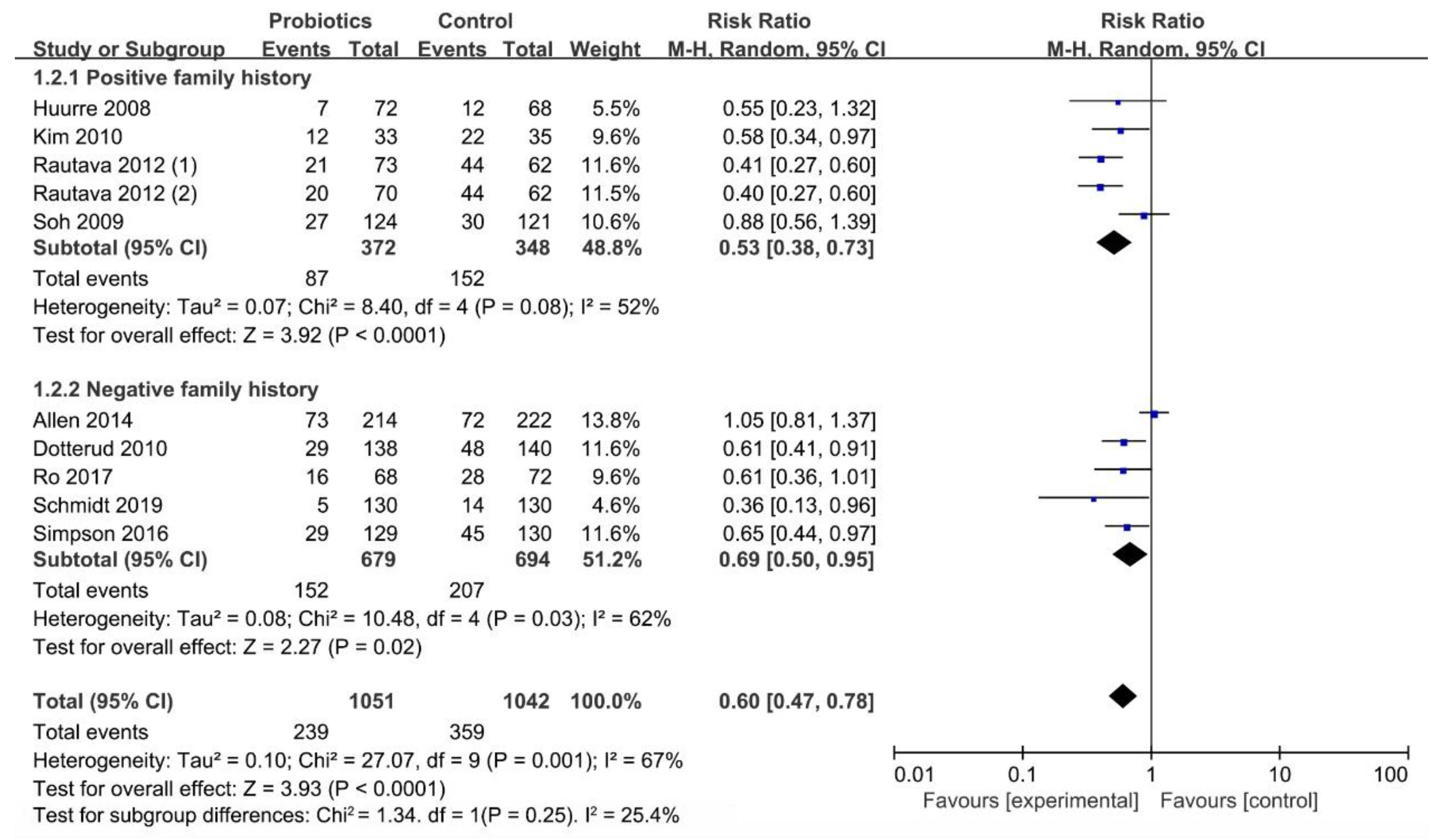
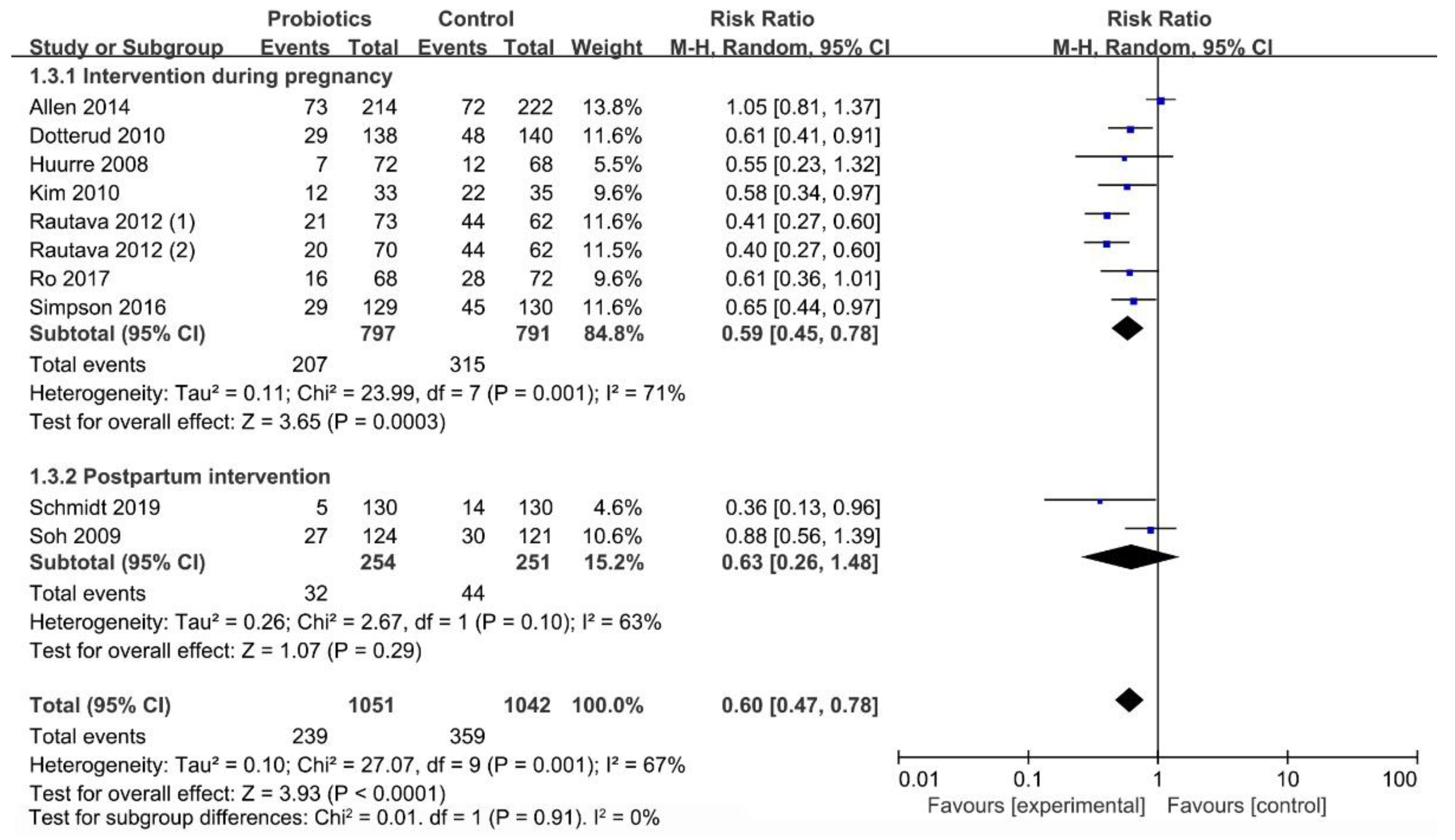
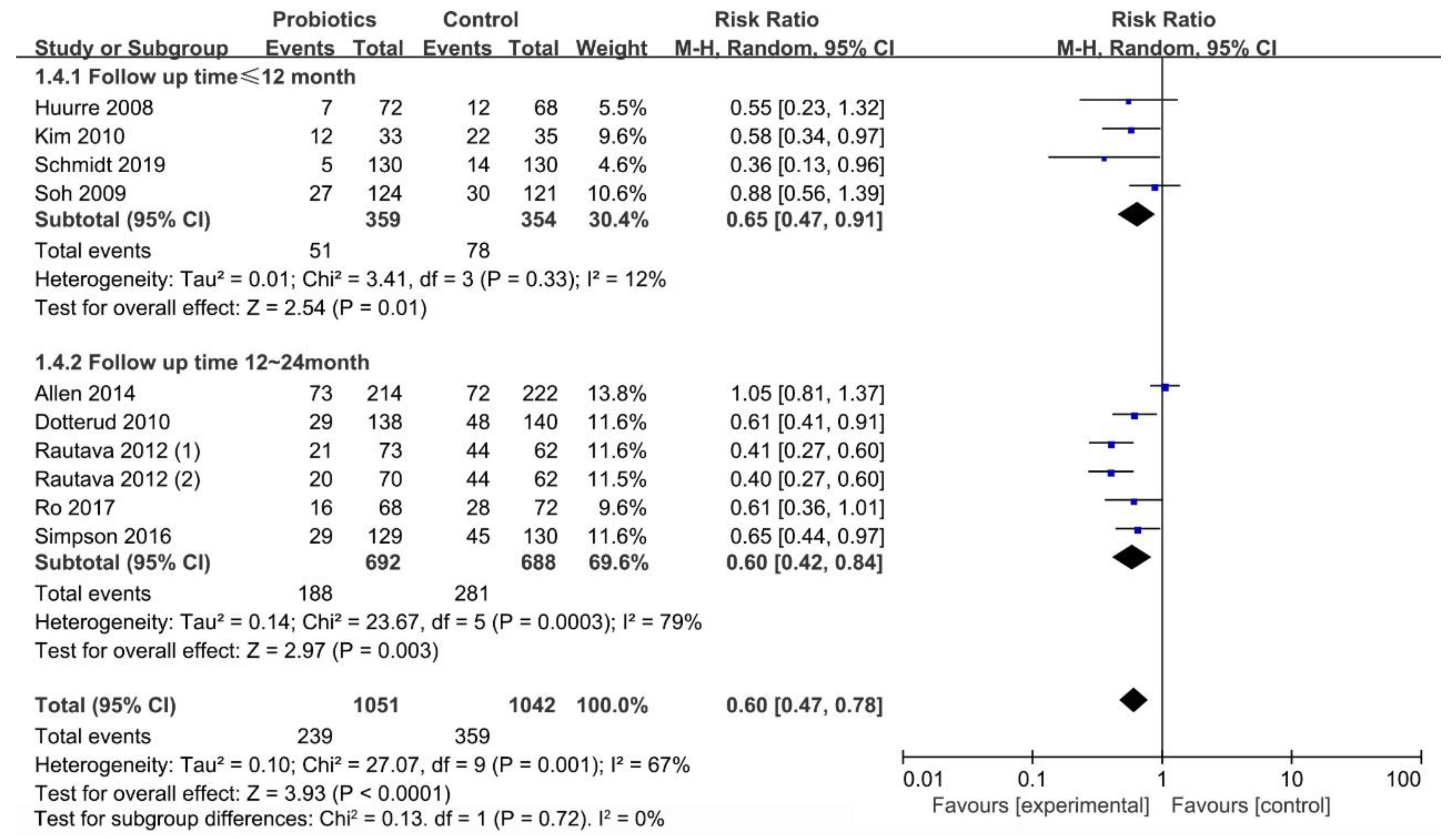
3.6. Subgroup Analysis Results
3.6.1. Influence of Family History on Occurrence of Infant Eczema
3.6.2. Influence of Initial Time of Intervention on Occurrence of Infant Eczema
3.6.3. Effect of Follow-up Time (≤12 Months/12–24 Months) on Occurrence of Infant Eczema
3.6.4. Effect of Probiotics Dosage and Strains on Occurrence of Infant Eczema
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Xiaohui, M.; Yan, H. Research status of related factors of infantile eczema. Dig. World Latest Med. Inf. 2018, 18, 31–32. [Google Scholar]
- Pawankar, R.; Canonica, G.; Holgate, S.; Lockey, R.; Blaiss, M. World Allergy Organization (WAO) White Book on Allergy, Updat; World Allergy Organization: Milwaukee, WI, USA, 2013; pp. 9–11. [Google Scholar]
- Chu, L.M.; Rennie, D.C.; Cockcroft, D.W.; Pahwa, P.; Dosman, J.; Hagel, L.; Karunanayake, C.; Pickett, W.; Lawson, J.A. Prevalence and determinants of atopy and allergic diseases among school-age children in rural Saskatchewan, Canada. Ann. Allergy Asthma Immunol. 2014, 113, 430–439. [Google Scholar] [CrossRef] [PubMed]
- Dell, S.D.; Foty, R.G.; Gilbert, N.L.; Jerret, M.; To, T.; Walter, S.D.; Stieb, D.M. Asthma and allergic disease prevalence in a diverse sample of Toronto school children: Results from the Toronto Child Health Evaluation Questionnaire (T-CHEQ) Study. Can. Respir. J. 2010, 17, e1–e6. [Google Scholar] [CrossRef] [PubMed]
- Elbert, N.J.; Duijts, L.; den Dekker, H.T.; Jaddoe, V.W.; Sonnenschein-van der Voort, A.M.; de Jongste, J.C.; Pasmans, S.G. Role of environmental exposures and filaggrin mutations on associations of ethnic origin with risk of childhood eczema. The Generation R Study. Pediatric Allergy Immunol. 2016, 27, 627–635. [Google Scholar] [CrossRef] [PubMed]
- Silvers, K.M.; Frampton, C.M.; Wickens, K.; Epton, M.J.; Pattemore, P.K.; Ingham, T.; Fishwick, D.; Crane, J.; Town, G.I. Breastfeeding protects against adverse respiratory outcomes at 15 months of age. Matern. Child. Nutr. 2009, 5, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Mengzhi, H. Clinical analysis of 222 cases of infantile eczema. J. Chin. Med. Cosmetol. 2014, 4, 118. [Google Scholar]
- Spergel, J.M. From atopic dermatitis to asthma: The atopic march. Ann. Allergy Asthma Immunol. 2010, 105, 99–106. [Google Scholar] [CrossRef]
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514. [Google Scholar] [CrossRef]
- Lee, J.; Seto, D.; Bielory, L. Meta-analysis of clinical trials of probiotics for prevention and treatment of pediatric atopic dermatitis. J. Allergy Clin. Immunol. 2008, 121, 116–121.e111. [Google Scholar] [CrossRef]
- Zhang, Y.; Jin, S.; Wang, J.; Zhang, L.; Mu, Y.; Huang, K.; Zhao, B.; Zhang, K.; Cui, Y.; Li, S. Variations in early gut microbiome are associated with childhood eczema. FEMS Microbiol. Lett. 2019, 366. [Google Scholar] [CrossRef]
- Lodge, C.J.; Tan, D.J.; Lau, M.X.; Dai, X.; Tham, R.; Lowe, A.J.; Bowatte, G.; Allen, K.J.; Dharmage, S.C. Breastfeeding and asthma and allergies: A systematic review and meta-analysis. Acta Paediatr. 2015, 104, 38–53. [Google Scholar] [CrossRef]
- Egeberg, A.; Andersen, Y.M.; Gislason, G.; Skov, L.; Thyssen, J.P. Neonatal risk factors of atopic dermatitis in Denmark - Results from a nationwide register-based study. Pediatric Allergy Immunol. 2016, 27, 368–374. [Google Scholar] [CrossRef] [PubMed]
- Ting, W.; Fuyan, L.; Ming, M. Efficacy and hidden danger of probiotics. J. Sci. 2019, 71, 29–33. [Google Scholar]
- Murray, C.S.; Tannock, G.W.; Simon, M.A.; Harmsen, H.J.; Welling, G.W.; Custovic, A.; Woodcock, A. Fecal microbiota in sensitized wheezy and non-sensitized non-wheezy children: A nested case-control study. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2005, 35, 741–745. [Google Scholar] [CrossRef]
- Song, H.; Yoo, Y.; Hwang, J.; Na, Y.C.; Kim, H.S. Faecalibacterium prausnitzii subspecies-level dysbiosis in the human gut microbiome underlying atopic dermatitis. J. Allergy Clin. Immunol. 2016, 137, 852–860. [Google Scholar] [CrossRef] [PubMed]
- Malik, K.; Heitmiller, K.D.; Czarnowicki, T. An Update on the Pathophysiology of Atopic Dermatitis. Dermatol. Clin. 2017, 35, 317–326. [Google Scholar] [CrossRef]
- Vitaliti, G.; Pavone, P.; Guglielmo, F.; Spataro, G.; Falsaperla, R. The immunomodulatory effect of probiotics beyond atopy: An update. J. Asthma 2014, 51, 320–332. [Google Scholar] [CrossRef] [PubMed]
- Cabana, M.D.; McKean, M.; Caughey, A.B.; Fong, L.; Lynch, S.; Wong, A.; Leong, R.; Boushey, H.A.; Hilton, J.F. Early Probiotic Supplementation for Eczema and Asthma Prevention: A Randomized Controlled Trial. Pediatrics 2017, 140. [Google Scholar] [CrossRef]
- Chenxi, H. Preventive Effect of Probiotics on Neonatal Eczema. Master’s Thesis, North China University of Science and Technology, Tangshan, China, 2019. [Google Scholar]
- Fiocchi, A.; Pawankar, R.; Cuello-Garcia, C.; Ahn, K.; Al-Hammadi, S.; Agarwal, A.; Beyer, K.; Burks, W.; Canonica, G.W.; Ebisawa, M.; et al. World Allergy Organization-McMaster University Guidelines for Allergic Disease Prevention (GLAD-P): Probiotics. World Allergy Organ. J. 2015, 8, 4. [Google Scholar] [CrossRef]
- Muraro, A.; Halken, S.; Arshad, S.H.; Beyer, K.; Dubois, A.E.; Du Toit, G.; Eigenmann, P.A.; Grimshaw, K.E.; Hoest, A.; Lack, G.; et al. EAACI food allergy and anaphylaxis guidelines. Primary prevention of food allergy. Allergy 2014, 69, 590–601. [Google Scholar] [CrossRef]
- de Silva, D.; Halken, S.; Singh, C.; Antonella, M.; Angier, E.; Arasi, S.; Arshad, H.; Beyer, K.; Boyle, R.; du Toit, G.; et al. Preventing food allergy in infancy and childhood: Systematic review of randomised controlled trials. Pediatric Allergy Immunol. 2020. [Google Scholar] [CrossRef]
- Forsberg, A.; West, C.E.; Prescott, S.L.; Jenmalm, M.C. Pre- and probiotics for allergy prevention: Time to revisit recommendations? Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2016, 46, 1506–1521. [Google Scholar] [CrossRef] [PubMed]
- Zhuoyun, M.; Ze, Y.; Jialong, Y. Application status and efficacy of probiotics in food. J. Mod. Food 2020, 129–131. [Google Scholar] [CrossRef]
- Dang, D.; Zhou, W.; Lun, Z.J.; Mu, X.; Wang, D.X.; Wu, H. Meta-analysis of probiotics and/or prebiotics for the prevention of eczema. J. Int. Med. Res. 2013, 41, 1426–1436. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.; Morgan, X.C.; Wang, X.Y.; Wickens, K.; Purdie, G.; Fitzharris, P.; Otal, A.; Lawley, B.; Stanley, T.; Barthow, C.; et al. Eczema-protective probiotic alters infant gut microbiome functional capacity but not composition: Sub-sample analysis from a RCT. Benef. Microbes 2019, 10, 5–17. [Google Scholar] [CrossRef] [PubMed]
- David, M.; Alessandro, L.; Jennifer, T.; Douglas, G.A.; Group, T.P. Preferred Reporting Items for Systematic Reviews and Meta-Analyses: The PRISMA Statement. PLoS Med. 2009, 6, e1000097. [Google Scholar]
- Gu, S.; Yang, A.W.; Xue, C.C.; Li, C.G.; Pang, C.; Zhang, W.; Williams, H.C. Chinese herbal medicine for atopic eczema. Cochrane Database Syst. Rev. 2013. [Google Scholar] [CrossRef]
- Hardy, R.J.; Thompson, S.G. Detecting and describing heterogeneity in meta-analysis. Stat. Med. 1998, 17, 841–856. [Google Scholar] [CrossRef]
- Allen, S.J.; Jordan, S.; Storey, M.; Thornton, C.A.; Gravenor, M.B.; Garaiova, I.; Plummer, S.F.; Wang, D.; Morgan, G. Probiotics in the prevention of eczema: A randomised controlled trial. Arch. Dis. Child. 2014, 99, 1014–1019. [Google Scholar] [CrossRef]
- Dotterud, C.K.; Storro, O.; Johnsen, R.; Oien, T. Probiotics in pregnant women to prevent allergic disease: A randomized, double-blind trial. Br. J. Dermatol. 2010, 163, 616–623. [Google Scholar] [CrossRef]
- Huurre, A.; Laitinen, K.; Rautava, S.; Korkeamaki, M.; Isolauri, E. Impact of maternal atopy and probiotic supplementation during pregnancy on infant sensitization: A double-blind placebo-controlled study. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2008, 38, 1342–1348. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kwon, J.H.; Ahn, S.H.; Lee, S.I.; Han, Y.S.; Choi, Y.O.; Lee, S.Y.; Ahn, K.M.; Ji, G.E. Effect of probiotic mix (Bifidobacterium bifidum, Bifidobacterium lactis, Lactobacillus acidophilus) in the primary prevention of eczema: A double-blind, randomized, placebo-controlled trial. Pediatric Allergy Immunol. 2010, 21, e386–e393. [Google Scholar] [CrossRef] [PubMed]
- Rautava, S.; Kainonen, E.; Salminen, S.; Isolauri, E. Maternal probiotic supplementation during pregnancy and breast-feeding reduces the risk of eczema in the infant. J. Allergy Clin. Immunol. 2012, 130, 1355–1360. [Google Scholar] [CrossRef]
- Ro, A.D.B.; Simpson, M.R.; Ro, T.B.; Storro, O.; Johnsen, R.; Videm, V.; Oien, T. Reduced Th22 cell proportion and prevention of atopic dermatitis in infants following maternal probiotic supplementation. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2017, 47, 1014–1021. [Google Scholar] [CrossRef]
- Schmidt, R.M.; Pilmann Laursen, R.; Bruun, S.; Larnkjaer, A.; Molgaard, C.; Michaelsen, K.F.; Host, A. Probiotics in late infancy reduce the incidence of eczema: A randomized controlled trial. Pediatric Allergy Immunol. 2019, 30, 335–340. [Google Scholar] [CrossRef] [PubMed]
- Simpson, M.R.; Ro, A.D.; Grimstad, O.; Johnsen, R.; Storro, O.; Oien, T. Atopic dermatitis prevention in children following maternal probiotic supplementation does not appear to be mediated by breast milk TSLP or TGF-beta. Clin. Transl. Allergy 2016, 6, 27. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Soh, S.E.; Aw, M.; Gerez, I.; Chong, Y.S.; Rauff, M.; Ng, Y.P.; Wong, H.B.; Pai, N.; Lee, B.W.; Shek, L.P. Probiotic supplementation in the first 6 months of life in at risk Asian infants—Effects on eczema and atopic sensitization at the age of 1 year. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2009, 39, 571–578. [Google Scholar] [CrossRef] [PubMed]
- Lafang, H.; Zhen, H.; Daogen, Y.; Qingchuan, W.; Xiaobing, L.; Xueyan, D. Effects of probiotics supplementation during pregnancy and infancy to prevent allergic eczema in children: A meta analysis. Chin. J. Child. Health Care 2018, 26, 873–877. [Google Scholar]
- Zuccotti, G.; Meneghin, F.; Aceti, A.; Barone, G.; Callegari, M.L.; Di Mauro, A.; Fantini, M.P.; Gori, D.; Indrio, F.; Maggio, L.; et al. Probiotics for prevention of atopic diseases in infants: Systematic review and meta-analysis. Allergy 2015, 70, 1356–1371. [Google Scholar] [CrossRef]
- West, C.E.; Jenmalm, M.C.; Prescott, S.L. The gut microbiota and its role in the development of allergic disease: A wider perspective. Clin. Exp. Allergy J. Br. Soc. Allergy Clin. Immunol. 2015, 45, 43–53. [Google Scholar] [CrossRef]
- Yuejie, Z. Application of probiotics in children with allergic diseases. Chin. J. Pract. Pediatrics 2017, 32, 114–117. [Google Scholar]
- Lujing, T.; Jie, C.; Yi, S. Probiotics for prevention of allergic diseases in children: A meta analysis. Chin. J. Pediatrics 2012, 50, 504–509. [Google Scholar]
- Yu, W.; Guo, Z.; Cairong, Z.; Lujiao, H.; Ke, L.; Li, Y. Probiotics in the prevention of infantile eczema and atopic eczema: A systematic review. Chin. J. Evid. Based Med. 2012, 12, 1372–1378. [Google Scholar]
- Loo, E.X.; Llanora, G.V.; Lu, Q.; Aw, M.M.; Lee, B.W.; Shek, L.P. Supplementation with probiotics in the first 6 months of life did not protect against eczema and allergy in at-risk Asian infants: A 5-year follow-up. Int. Arch. Allergy Immunol. 2014, 163, 25–28. [Google Scholar] [CrossRef]
- Allen, S.J.; Jordan, S.; Storey, M.; Thornton, C.A.; Gravenor, M.; Garaiova, I.; Plummer, S.F.; Wang, D.; Morgan, G. Dietary supplementation with lactobacilli and bifidobacteria is well tolerated and not associated with adverse events during late pregnancy and early infancy. J. Nutr. 2010, 140, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Pan, S.J.; Kuo, C.H.; Lam, K.P.; Chu, Y.T.; Wang, W.L.; Hung, C.H. Probiotics and allergy in children—An update review. Pediatric Allergy Immunol. 2010, 21, e659–e666. [Google Scholar] [CrossRef]
| Trial | Sample Size | Eczema Cases | Family History (Negative/Positive) | Adverse Events Cases | Intervention vs. Placebo | Specific Strains and Dosage (cfu/Day) | Intervention Time (Start–End) | Follow-Up Time |
|---|---|---|---|---|---|---|---|---|
| Allen 2014 | T: 214 C: 222 | T: 73 C: 72 | Negative | T: 0 C: 0 | Lactobacillus and Bifidobacterium vs. Maltodextrin | Lactobacillus salivarius (L. salivarius) (6.25 × 109), L. paracasei (1.25 × 109), Bifidobacterium animalis subspecies lactis (B. animals subsp. lactis) (1.25 × 109), Bifidobacterium bifidum (B. bifidum) (1.25 × 109) | Mother: from gestational week 36 until delivery Infants: 0–6 months of age | 2 years |
| Dotterud 2010 | T: 138 C: 140 | T: 29 C: 48 | Negative | T: 0 C: 0 | Lactobacillus and Bifidobacterium vs. Skim fermented milk | LGG (5 × 1010), B. animals subsp. lactis (5 × 1010), Lactobacillus acidophilus (L.acidophilus) (5 × 109) | Mother: 36 weeks of pregnancy–3 months postpartum | 2 years |
| Huurre 2008 | T: 72 C: 68 | T: 7 C: 12 | Positive | Not report. | Lactobacillus and Bifidobacterium vs. Microcrystalline cellulose and anhydrous glucose | LGG (1 × 1010), B. animals subsp. lactis (1 × 1010) | Mother: the first trimester of pregnancy–the end of exclusive breastfeeding | 1 year |
| Kim 2010 | T: 33 C: 35 | T: 12 C: 22 | Positive | T: 0 C: 0 | Lactobacillus and Bifidobacterium vs. Maltodextrin | B. bifidum (1.6 × 109), B. animals subsp. lactis (1.6 × 109), L. acidophilus (1.6 × 109) | Mother: 8 weeks before delivery–6 months after delivery | 1 year |
| Rautava 2012 (1) | T: 73 C: 62 | T: 21 C: 44 | Positive | T: 35 C: 24 | Lactobacillus and Bifidobacterium vs. Dietary supplement without probiotics | LGG (1 × 109), B. longum (1 × 109) | Mother: 2 months before delivery–2 months after delivery | 2 years |
| Rautava 2012 (2) | T: 70 C: 62 | T: 20 C: 44 | Positive | T: 28 C: 24 | Lactobacillus and Bifidobacterium vs. Dietary supplement without probiotics | L. paracasei (1 × 109), B. longum (1 × 109) | Mother: 2 months before delivery–2 months after delivery | 2 years |
| Ro 2017 | T: 68 C: 72 | T: 16 C: 28 | Negative | Not report. | Lactobacillus and Bifidobacterium vs. placebo | LGG (5 × 1010), B. animals subsp. lactis (5 × 1010), L. acidophilus (5 × 109) | Mother: 36 weeks of pregnancy–3 months postpartum | 2 years |
| Schmidt 2019 | T: 130 C: 130 | T: 5 C: 14 | Negative | T: 12 C: 13 | Lactobacillus and Bifidobacterium vs. Maltodextrin | LGG (1 × 109), B. animals subsp. lactis (1 × 109) | Infant: 6 months | half a year |
| Simpson 2016 | T: 129 C: 130 | T: 29 C: 45 | Negative | Not report. | Lactobacillus and Bifidobacterium vs. Fermented skimmed milk | LGG (5 × 1010), B. animals subsp. lactis (5 × 1010), L. acidophilus (5 × 109) | Mother: 36 weeks of pregnancy–3 months postpartum | 2 years |
| Soh 2009 | T: 124 C: 121 | T: 27 C: 30 | Positive | Not report. | Lactobacillus and Bifidobacterium vs. infant formula | B. longum (1 × 107), LGG (2 × 107) | Infants: 0–6 months of age | 1 year |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, M.; Luo, J.; Liu, H.; Xi, Y.; Lin, Q. Can Mixed Strains of Lactobacillus and Bifidobacterium Reduce Eczema in Infants under Three Years of Age? A Meta-Analysis. Nutrients 2021, 13, 1461. https://doi.org/10.3390/nu13051461
Sun M, Luo J, Liu H, Xi Y, Lin Q. Can Mixed Strains of Lactobacillus and Bifidobacterium Reduce Eczema in Infants under Three Years of Age? A Meta-Analysis. Nutrients. 2021; 13(5):1461. https://doi.org/10.3390/nu13051461
Chicago/Turabian StyleSun, Minghui, Jing Luo, Hanmei Liu, Yue Xi, and Qian Lin. 2021. "Can Mixed Strains of Lactobacillus and Bifidobacterium Reduce Eczema in Infants under Three Years of Age? A Meta-Analysis" Nutrients 13, no. 5: 1461. https://doi.org/10.3390/nu13051461
APA StyleSun, M., Luo, J., Liu, H., Xi, Y., & Lin, Q. (2021). Can Mixed Strains of Lactobacillus and Bifidobacterium Reduce Eczema in Infants under Three Years of Age? A Meta-Analysis. Nutrients, 13(5), 1461. https://doi.org/10.3390/nu13051461







