The Role of Exercise, Diet, and Cytokines in Preventing Obesity and Improving Adipose Tissue
Abstract
1. Introduction
2. Adipose Tissue Biology: Why Our Body Is a Fat-Storing Machine?
2.1. White Adipose Tissue
2.2. Brown and Beige Adipose Tissue
3. Exercise Strategies to Prevent Obesity and Improve Adipose Tissue Health
3.1. Continuous Exercise and Adipose Tissue
Molecular Mechanisms Underlying the Reduction in Total BF, SAT, and VAT with Exercise
3.2. High-Intensity Interval Training and Adipose Tissue
3.3. Resistance Exercise Training and Adipose Tissue
3.4. Concurrent Training and Adipose Tissue
| Continuous Exercise and Adipose Tissue | ||||||||
| Author | Year | Participants ( O2max) (mL/kg/min) | n (M/F) | Duration, Frequency, Mode | Protocols | Main Findings | Ref | |
| 1 | Ross et al. | 2000 | Obese males (NR) | (52/0) | 12 weeks, daily, brisk walking or light jogging | Group 1: Diet (reducing total daily energy intake to 700 kcal/day) Group 2: Exercise (80% of HRmax until 700 kcal is expended) Group 3: Exercise without weight loss (enough calories given to compensate for the energy expended during the daily exercise sessions) Group 4: Control group | Reduction in total fat was greater in group 2 compared with group 1. Group 2: Substantial decreased in both SAT and VAT Group 3: Attenuation in abdominal fat and prevented further weight gain. | [59] |
| 2 | Miyatake et al. | 2002 | Obese males (NR) | (31/0) | 1 year follow up study, daily, walking | An increase of daily steps from 7013 to 8840 | Significantly decreased in SAT, VAT, and body composition. | [61] |
| 3 | Racette et al. | 2006 | Healthy, non-obese adults (NR) | (18/30) | 1 year, 6 days/wk, running/cycling/rowing ergometers/elliptical machines/stairclimbers | Group 1: 20% calorically-restricted diet Group 2: Training at 58% of VO2max Group 3: Healthy lifestyle control group | Significant reduction in fat mass, SAT, and VAT for both group 1 and 2. | [60] |
| 4 | Durheim et al. | 2008 | Sedentary, dyslipidemic, overweight males (~32.8 VO2peak) females (~23.9 VO2peak) | (40/33) | 8–9 months, 3 days/wk, aerobic training | Group 1: ∼20 miles/wk of jogging (65–80% VO2max) Group 2: 12 miles/wk of jogging (65%-80% VO2max) Group 3: 12 miles/wk of brisk walking (40–55% VO2max) | Significantly reduced in thigh SAT for all three groups, but VAT decreased substantially in men only. | [62] |
| 5 | Ross et al. | 2015 | Abdominally obese adults (NR) | (104/196) | 24 weeks, 5 days/wk, walking/jogging/treadmill training | Group 1: Training at a low-amount, moderate-intensity exercise at 50% VO2max (31 min/session) Group 2: Training at a high-amount, moderate-intensity exercise at 50% VO2max (58 min/session) Group 3: Training at a high-amount, high-intensity exercise at 75% VO2max (40-min/session) Group 4: Control group | Similar reductions were resulted in total BF, SAT, and VAT in all training groups. | [63] |
| 6 | Wilmore et al. | 1999 | Overweight adults (NR) | (258/299) | 20 weeks, 3 days/wk, cycling | Training at 55% VO2max to at 75% VO2max for 30 min to 50 min. | Males had a greater loss in abdominal SAT and VAT than females. A higher rate of decline was also seen in abdominal SAT compared to VAT. | [65] |
| 7 | Irving et al. | 2008 | Middle-aged obese women (~21 VO2peak) | (0/27) | 16 weeks, 5 days/wk, aerobic training | Group 1: Moderate-intensity training (5 days per week at an intensity ≤ LT Group 2: High-intensity training (3 days per week at an intensity > LT and 2 days per week ≤ LT) Group 3: No-exercise training | No significant changes of SAT and VAT were observed in group 1, whereas group 2 resulted in reduced total abdominal fat, SAT, and VAT. | [66] |
| 8 | Coker et al. | 2009 | Overweight elderly adults (NR) | (9/9) | 12 weeks, 4–5 days/wk, aerobic training | Group 1: Moderate-intensity (50% VO2peak) Group 2: High-intensity (75% VO2peak) | A remarkable reduction in VAT in the high-intensity group exhibited, while no change was observed in the moderate group. | [67] |
| 9 | Tanaka et al. | 2020 | Healthy adults (NR) | (87/145) | 4 months, NR, walking/aerobic training | Group 1: WM Group 2: WM + vigorous-intensity physical (VPA) activities (VWM) | VPA activities resulted in high BAT density, particularly in men. BAT-density is related to visceral fat area and VWM in men, and related to body fat percentage in women. | [68] |
| 10 | Owens et al. | 1999 | Obese children (NR) | (25/49) | 4 months, 5 days/wk, exercising on machines and sports activities | Group 1: 40 min/day at 70–75% HRmax Group 2: Control group | Significant decrease in BF%, total BF, and SAT for group 1. | [69] |
| 11 | Lee et al. | 2005 | Lean and obese male with and without T2D (~61.2% VO2peak) | (24/0) | 13 weeks: 5 days/wk, aerobic training | All participants trained for 60 min/day at a moderate intensity (∼60% VO2peak) | Significant reductions in total abdominal SAT and VAT in all groups (lean and obese males with and without T2D). Reduction in VAT was greater in the obese and T2D groups when compared with the lean group. | [70] |
| 12 | Keating et al. | 2015 | Inactive and overweight/obese adults (~22.4 VO2peak) | (17/31) | 8 weeks, 3–4 days/wk, brisk walking/cycling | Group 1: Cycling and brisk walk at 50% VO2peak for 3 days and 1 day/wk, respectively. (From 45 min in week one to 60 min by the 3rd week, totaling 180–240 min/wk) Group 2: Cycling and brisk walk at 50% VO2peak for 2 days and 1 day/wk, respectively. (From 30 min in week one to 45 min by the 3rd week, totaling 90–135 min/wk) Group 3: Cycling and brisk walk at 60–70% VO2peak for 2 days and 1 day/wk, respectively. (From 30 min in week one to 45 min at 70% VO2peak by the third week, totaling 90–135 min/wk) Group 4: Control group | Reduction in liver fat and VAT for all three groups. | [71] |
| 13 | Christiansen et al. | 2009 | Obese adults (NR) | 79 | 12 weeks, 3 days/wk, aerobic training | Group 1: exercise (60–75 min at 70% VO2max per training session) Group 2: hypocaloric diet (600 kcal/day) Group 3: hypocaloric diet and exercise | Reduction in BW 3.5 kg and VAT 18% in group 1. Higher reduction in BW (12.3 kg) and VAT (30–37%) in group 2 and 3 than group 1. | [72] |
| 14 | Walhin et al. | 2016 | Sedentary overweight men and postmenopausal women (31.5 VO2max) | (24/14) | 3 weeks, 5 days/wk, treadmill | Group 1: Moderate intensity training (50% VO2max) with caloric restriction (5000 kcal/wk) Group 2: Vigorous-intensity training (70% VO2max) with caloric restriction (5000 kcal/wk) | Both groups resulted similar reductions in total fat and abdominal fat mass, as well as similarly affected the expression of the lipogenic enzymes. | [73] |
| 15 | Islam et al. | 2018 | Active young males (NR) | (8/0) | 1 day, acute session, running | Group 1: 30 min continuous running at 65% VO2max Group 2: 30 min of running at 85% VO2max Group 3: 4 × 30 s “all-out” sprints with 4 min of rest (SIT) Group 4: No exercise | Increased whole-body fat oxidation during the post-exercise recovery period in all exercise groups and it was greatest in group 3. | [74] |
| 16 | Henderson et al. | 2007 | Healthy males (56.6% VO2peak) and females (48.9% VO2peak) | (10/8) | 1 day, acute session, aerobic exercise | Group 1: 90 min of exercise at 45% VO2peak Group 2: 60 min of exercise at 65% VO2peak | Substantial increased for the oxidation of total lipid and plasma fatty acid in both groups. Women was more dependent on lipid during exercise, whereas during recovery, lipid metabolism is accentuated to a greater extent in men. | [75] |
| 17 | Perreault et al. | 2004 | Healthy lean males (59.4 VO2max) and females (60 VO2max) | (10/10) | 1 day, acute session, aerobic exercise | Exercised at 85% LT for 90 min | Significantly increased the amount of the adipose tissue lipoprotein lipase (56%) in men but not women. | [76] |
| 18 | Ludzki et al. | 2020 | Obese adults (NR) | (3/7) | 1 day, acute session, aerobic exercise | Group 1: 60 min acute session at 80% HRpeak Group 2: No acute exercise session | Increased the number of the adipose tissue progenitor cell phenotype in exercise group, as well as decreased of preadipocyte content was shown in the stromal vascular cells fraction of SAT twelve hours after exercise. | [77] |
| 19 | Hojbjerre et al. | 2007 | Overweight (54.6 VO2max) and lean males (57.1 VO2max) | (16/0) | 1 day, acute session, aerobic exercise | Exercised for 1 h at 55% of VO2max | Modification of adipose tissue mRNA and interstitial cytokine concentration in overweight males. An increased concentration of interstitial adiponectin and IL-6, while the response at the mRNA level was different, with IL-6 mRNA increasing but adiponectin mRNA decreasing. | [78] |
| 20 | Van et al. | 2017 | Overweight and obese adults that active (51 VO2peak) and sedentary (42 VO2peak) | (8/12) | 1 day, acute session, aerobic exercise | 60 min of acute moderate-intensity exercise at 65% VO2max | Increased SAT mRNA expression of VEGFA. | [79] |
| 21 | Fabre et al. | 2018 | Healthy young males (46.88 VO2max) | (15/0) | 1 day, acute session, aerobic exercise | A single session of 15 min exercise at 80% VO2max | Induction of more than 3800 genes in adipose tissue from lean and overweight individuals. Among them were the genes responsible for monocyte infiltration. | [80] |
| 22 | Campbell et al. | 2013 | Overweight/obese postmenopausal women (24.4 VO2max) | (0/45) | 12 months, 5 days/wk, aerobic exercise | Group 1: Exercise (≥45 min of moderate-to-vigorous intensity exercise) Group 2: Diet (reducing total daily energy intake to 1200–2000 kcal/day) Group 3: Diet plus exercise Group 4: Control | Compared to the control, the mean percent BF loss was: diet, −12.6%, exercise, −3.1%, diet + exercise, −13.2% | [81] |
| 23 | Richterova et al. | 2004 | Obese women (NR) | (0/11) | 12 weeks, 3 days/wk, home-based training | Trained at 50% VO2peak at 40 min | No changed in genes involved in the control of SAT lipolysis. | [83] |
| 24 | Polak et al. | 2006 | Obese sedentary premenopausal women (24.6 VO2max) | (0/25) | 12 weeks, 5 days/wk, aerobic training/cycling | 2 sessions/wk of supervised aerobic exercise (50% VO2max) and 3 sessions/wk of home-based exercise (cycling) | No changes of gene expression of adipocytokines in obese women, while a decrease of plasma leptin was detected. | [84] |
| 25 | Leick et al. | 2007 | Obese and non-obese Adults (NR) | (18/24) | 8 weeks, 3 days/wk, home-based training | 30 min/session at 70% VO2max | Reduction of adipose tissue IL-18 mRNA content by 20% in obese individuals. | [85] |
| 26 | Christiansen et al. | 2010 | Obese adults (NR) | (9/10) | 12 weeks, 3 days/wk, home-based training | 60–75 min/session at 70% 35–40% VO2max | Significant elevation of the mRNA expression of adipose tissue adiponectin and adiponectin receptors. | [86] |
| 27 | Stanford et al. | 2015 | Trained or sedentary donor mice (NR) | 6 | 11 days, daily, running | Running daily inside the wheel cage. | Increased expression of many beige adipocyte marker genes in rodent SAT. | [49] |
| 28 | Trevellin et al. | 2014 | 8 weeks old male mice (NR) | (36/0) | 30 days, daily, swimming | 90 min of swimming | Increased expression of UCP1 and Prdm16 in mice SAT. | [88] |
| 29 | Otero-Diaz et al. | 2018 | Non-diabetic adults (NR) | (14/19) | 12 weeks, 3 days/wk, cycling | 60 min/session at 43–70% VO2max | Induction of the mRNA expression of beige/BAT makers of UCP1, TBX1, CPT1B in SAT of sedentary subjects. | [91] |
| High-Intensity Exercise and Adipose Tissue | ||||||||
| Author | Year | Participants ( O2max) (mL/kg/min) | n (M/F) | Duration, Frequency, Mode | Protocols | Main Findings | Ref | |
| 1 | Higgins et al. | 2016 | Inactive overweight/obese young women (NR) | (0/52) | 6 weeks, 3 days/wk, SIT/cycling | Group 1: SIT (30 s “all-out” sprints followed by 4 min of active recovery) Group 2: moderate-intensity continuous training (MICT) at 45–55% HRmax, for 20–30-min | SIT resulted greater loss in total BF and android fat than MICT cycling. | [94] |
| 2 | Zhang et al. | 2017 | Obese young women (NR) | (0/43) | 12 weeks, 3–4 days/wk, cycling | Group 1: prolonged MICT 60% VO2max Group 2: HIIT (90% VO2max, 4 min bout with 3 min recovery) | Similar reduction in abdominal SAT and VAT in both groups. | [97] |
| 3 | Riis et al. | 2019 | Healthy young males (43.9 VO2max) | (10/0) | 10 weeks, 3 days/wk, cycling | The first session was 40 min at 70% VO2max, the second session 2 × 20 min at 80%−90% VO2max (5 min easy biking in between), and the third session was 8 × 5 min at 90–100% (1 min easy biking in between). | Improvement in adipose tissue insulin sensitivity. | [98] |
| 4 | Dohlmann et al. | 2018 | Healthy sedentary adults (27 VO2max) | (5/7) | 6 weeks, 3 days/wk, HIIT | 7 × 1 min at 95–100% VO2max, with 1 min recovery | No change for BF% in overweight subjects, whereas the mitochondrial respiratory capacity in SAT was reduced after training. | [99] |
| 5 | Leggate et al. | 2012 | Overweight/obese sedentary males (NR) | (12/0) | 2 weeks, 3 days/wk, HIIT | 10 × 4 min at 85% VO2max, 2 min rest | Decreased in waist circumference, as well as reductions in IL6 and fatty acid synthase content in SAT biopsies. | [100] |
| 6 | Islam et al. | 2020 | Overweight women (30.3 VO2peak) | (0/10) | 1 day, Acute session, HIIE | HIIE: 10 × 4 min 90% HRmax, separated by 2 min recovery | β-adrenergic and insulin signaling in subcutaneous abdominal adipose tissue remained unchanged following acute HIIE, while there was a significant decrease in the respiratory exchange ratio. | [101] |
| 7 | Astorino et al. | 2013 | Sedentary women (24.2 VO2max) | (0/23) | 12 weeks, 3 days/wk, HIIT | Group 1: 6–10 × 60 s intervals at 80–90% peak power output, with 75 s recovery Group 2: 6–10 × 60 s intervals at 60–80% peak power output, with 75 or 60 s recovery | Increased fat oxidation rate in sedentary (including both normal weight to obese) women. | [102] |
| 8 | Taylor et al. | 2020 | Coronary artery disease patients (NR) | 42 | 12 months, 3 days/wk for 4 weeks, followed by three home-based sessions/wk for 11 months, HIIT/MICT | HIIT: 4 × 4 min high-intensity interval training MICT: 40 min of usual care | Both exercise interventions reduced VAT over 3 and 12 months, while HIIT resulted in a slightly greater reduction in liver fat compared with MICT. | [103] |
| 9 | Larsen et al. | 2015 | Overweight adults (NR) | NR | 6 weeks, 3 days/wk, HIIT | 5 × 60 s at ~128% of peak power, 90 s recovery | No alteration in BF% or adipose tissue mitochondrial function. | [105] |
| 10 | Whyte et al. | 2010 | Overweight/obese sedentary men (NR) | (10/0) | 2 weeks, 3 days/wk, Wingate-based SIT | 4 to 6 repeats of 30 s Wingate anaerobic sprints on an electromagnetically braked cycle ergometer, with 4.5 min recovery. | Significant reduction in waist and hip circumference in overweight/obese sedentary men, as well as an elevated resting fat oxidation rate. | [106] |
| 11 | Honkala et al. | 2020 | Inactive, healthy adults with IR (<40 VO2peak) | (28/26) | 2 weeks, 3 days/wk, Wingate-based SIT | SIT: 4–6 × 30 s at maximum effort MICT: 40–60 min at 60% VO2max | Both groups resulted in a similar reduction in BF%, abdominal SAT and VAT, as well as decreased CD26 and ANGPTL4 gene expression in SAT. | [107] |
| 12 | Cooper et al. | 2016 | Overweight men (NR) | (30/0) | 12 weeks, 3 days/wk, SIT | SIT: 4–10 × 30 s sprint efforts with passive or active recovery | No significant changes in FM or abdominal VAT. | [108] |
| Resistance Exercise and Adipose Tissue | ||||||||
| Author | Year | Participants ( O2max) (mL/kg/min) | n (M/F) | Duration, Frequency, Mode | Protocols | Main Findings | Ref | |
| 1 | Schmitz et al. | 2003 | Midlife women (NR) | (0/60) | 15 weeks, 2 days/wk, RT | The treatment group performed twice-weekly supervised strength training followed by 6 months of unsupervised training. | Reduction in FM and VAT. | [109] |
| 2 | Ku et al. | 2010 | Women with T2D (NR) | (0/44) | 12 weeks, 5 days/wk, RT/aerobic training | RT: elastic band training Aerobic training: Walking for 60 min at moderate-intensity (3.6–5.2 metabolic equivalents) | RT resulted in a greater reduction in SAT than aerobic training, as well as only RT, which decreased subfascial adipose tissue at the mid-thigh level. | [110] |
| 3 | Treuth et al. | 1994 | Healthy men (NR) | (13/0) | 16 weeks, RT | 16-week strength-training program | Reduction in FM and VAT. | [120] |
| 4 | Prabhakaran et al. | 1999 | Healthy, sedentary, premenopausal women (NR) | (0/24) | 14 weeks, 3 days/wk, RT | Group 1: 45–50 min RT sessions (85% of 1 RM) Group 2: no exercise | Reduction in FM and VAT for group 1. | [121] |
| 5 | Ross et al. | 1996 | Obese men (NR) | (33/0) | 16 weeks, 5 days/wk, RT/RT combined with diet interventions/only diet intervention | Group 1: RT Group 2: RT combined with diet interventions Group 3: Diet intervention | RT group has shown a decrease in FM and VAT, whereas 40% reduction in visceral fat only observed in the RT combined diet group. | [122] |
| 6 | Treuth et al. | 1995 | Older women (NR) | (0/14) | 16 weeks, 3 days/wk, RT | Strength was assessed by one-repetition maximum tests, with training intensity gradually increased to approximately 67% of one repetition maximum | Significant reduction in visceral fat. | [123] |
| 7 | Hunter et al. | 2002 | Older adults (NR) | (14/12) | 25 weeks, 3 days/wk, RT | Training consisted of two sets of 10 repetitions at 65–80% of 1 RM | Improvement in fat-free mass and a reduction in fat mass in older males and females. Substantial loss of intra-abdominal adipose tissue (IAAT) and abdominal SAT in overweight females, but not in overweight men. | [124] |
| 8 | Rosety et al. | 2015 | Obese women (NR) | (0/48) | 12 weeks, 3 days/wk, resistance circuit training program | This training was circularly performed in six stations: arm curl, leg extension, seated row, leg curl, triceps extension and leg press. | Reduced thickness of epicardial adipose tissue. | [125] |
| 9 | Ross and Rissanen | 1994 | Obese women (NR) | (0/24) | 16 weeks, 3 days/wk, RT/aerobic training combined with caloric restriction | Group 1: RT Group 2: aerobic training combined with caloric restriction (reduced by 1000 kcal) | Substantial similar decrease in the volume ratio of VAT to SAT after RT and aerobic training combined with caloric restriction. | [126] |
| 10 | Slentz et al. | 2011 | Overweight adults (NR) | (44/56) | 8–10 weeks, 3 days/wk, RT or high-intensity aerobic training | Group 1: RT (3 times/wk, 3 sets of 8–12 repetitions/set) Group 2: high-intensity aerobic training (12 miles/wk at 75% VO2max) | High-intensity training provided a greater reduction in VAT and total abdominal fat than RT. | [127] |
| 11 | Ormsbee et al. | 2007 | Trained men (NR) | (8/0) | 1 day, acute session, RE | Three sets of 10 repetitions with a load at 85–100% of the individual’s one 1RM, 90 s rest periods between all sets and exercises, for a total of 40–45 min | Increased SAT lipolysis during RE, while SAT lipolysis and whole-body fat oxidation were higher immediately post RE. | [128] |
| 12 | Allman et al. | 2019 | Trained women (NR) | (0/13) | 1 day, acute session, RE | One set of 10 repetitions at 40% 1RM and three sets of 10 repetitions at 65% 1RM | İncreased in post-exercise whole-body fat oxidation and SAT lipolysis. | [129] |
| 13 | Chatzinikolaou et al. | 2008 | Lean men and obese males (NR) | (17/0) | 1 day, 30 min session, RE | Three cycles on 10 machines selected to stress the major muscle groups, 10–12 repetitions/set at 70–75% of one-repetition maximum with 30 s rests between sets and 2 min rests between cycles | Adipose tissue triacylglycerol lipase activity was elevated by 18-fold after 5 min of exercise in lean subjects, whereas a 16-fold increase was observed 10 min after exercise in obese males. | [130] |
| Concurrent Training and Adipose Tissue | ||||||||
| Author | Year | Participants ( O2max) (mL/kg/min) | n (M/F) | Duration, Frequency, Mode | Protocols | Main Findings | Ref | |
| 1 | Slentz et al. | 2011 | Overweight adults (NR) | (41/51) | 8–10 weeks, 3 days/wk, aerobic plus RT or aerobic training | Aerobic plus RT: 12 miles/wk at 75% VO2max plus 3 sets of 8–12 repetitions/set High-intensity aerobic training: 12 miles/wk at 75% VO2max | Similar significant reductions in VAT, SAT, and total abdominal fat for both groups. | [127] |
| 2 | Monteiro et al. | 2015 | Obese adolescents (NR) | 32 | 20 weeks, 3 days/wk, CT or aerobic training | CT: 60 min of 50 % of RM, followed by 30 min of 65 and 85% VO2max aerobic training. Aerobic training: 50 min continuous exercise at 65–85% VO2max | Significant reduction in waist circumferences and BF% after CT and aerobic training. | [136] |
| 3 | Damaso et al. | 2014 | Obese adolescents (NR) | 139 | 1 year, 3 days/wk, aerobic plus RT or aerobic training | Group 1: aerobic plus RT Group 2: AT | Aerobic plus RT provided a greater reduction in visceral fat and pro-inflammatory adipokines than AT alone intervention. | [138] |
| 4 | de Mello et al. | 2011 | Obese adolescents (NR) | (20/10) | 1 year, 3 days/wk, aerobic plus RT | Aerobic (30 min of aerobic exercise at 50–70% VO2max) plus RT (3 sets, 6–20 repetitions, 90–45 s/exercise/session) | Induced higher changes in body composition, waist circumference, and BF in obese adolescents than aerobic exercise only. | [139] |
| 5 | Davis et al. | 2011 | Obese adolescents (NR) | (0/38) | 16 weeks, 2 days/wk, CT | 30–45 min/session 70–85% HRmax plus 30–45 min, 12–14 repetitions, low-heavy weights | Significant reduction of waist circumference (~3%), VAT (~10%), and SAT (~10%). | [140] |
| 6 | Norheim et al. | 2014 | Overweight males (NR) | (26/0) | 12 weeks, 4 days/wk, aerobic plus RT | 12 weeks of CT | Chronic training increased the mRNA level of PGC-1α of SAT by 1.2-fold and 1.6-fold in the control group and the pre-diabetes group, respectively, whereas no significant changes neither in the brown-fat-selective gene Prdm16 or other known browning genes TBX1, TMEM26, and CD137 for both groups. | [141] |
| 7 | Stinkens et al. | 2018 | Obese males (NR) | (21/0) | 12 weeks, 3 days/wk, CT | Aerobic exercise (30 min at 70% of maximal power output) + resistance exercise (3 × 10 repetitions at 60% of 1 repeated maximum) | No significant changes in abdominal subcutaneous adipocyte size, β2-adrenergic sensitivity of lipolysis, and adipose tissue gene expression of markers involved in browning and lipolysis in obese subjects. | [142] |
4. Diet Strategies to Prevent Obesity and Improve Adipose Tissue Health
4.1. Manipulation of Diet Composition
4.2. Elimination/Restriction of a Specific Food Group: Plant-Based Diets
4.3. Manipulation of Timing: Intermittent Fasting
| Effects of Diet Composition Manipulation on Body Weight and Health | ||||||||
| Author | Year | Diet | Participants | n (M/F) | Duration | Main Findings | Ref | |
| 1 | Hall et al. | 2016 | Low-CHO ketogenic isocaloric diet or high-CHO diet | Overweight and obese men Mean age: 33 ± 1.8 y BMI: 28.8 ± 0.8 kg/m2 | (17/0) | 4 weeks high-CHO diet and 4 weeks ketogenic diet | Weight loss KD: 2.2 ± 0.3 kg (0.5 ± 0.2 kg from loss of body fat) BD: 0.8 ± 0.2 kg (0.5 ± 0.1 kg from loss of body fat) Increase in EEchamber, sleeping EE and EEDLW, decrease in RQ compared with baseline diet | [152] |
| 2 | Foster et al. | 2010 | LCD or LFD (limiting energy intake to 1200 to 1500 kcal/d for women and 1500 to 1800 kcal/d for men, 55% CHO, 30% fat, 15% protein) | Obese adults 45.5 ± 9.7 y BMI: 36.1± 3.5 kg/m2 | (99/208) | 2 years | Weight loss LCD: −6.34 kg LFD: −7.37 kg Fat mass loss LCD: −3.99 kg LFD: −3.84 kg Higher increase in HDL in LCD group Similar reductions in TG, LDL, VLDL, systolic blood pressure | [157] |
| 3 | Ebbeling et al. | 2012 | Isocaloric LFD (60% CHO 20% fat, 20% protein) or low-glycemic index diet (40% CHO, 40% fat, 20% protein), or VLCD (10% CHO, 60% fat, 30% protein) | Overweight and obese young adults Mean age: 30.3 ± 5.7 y BMI: 34.4 ± 4.9 kg/m2 | (13/8) | Crossover design; 12 weeks | Highest decreases in REE and TEE with LFD. Leptin level was highest in the LFD and lowest in the VLCD. HDL was highest in VLCD and lowest in LFD. | [159] |
| 4 | Hall et al. | 2015 | Isocaloric reduced fat diet or reduced CHO diet | Obese adults Mean age: 35.4 ± 1.74 y BMI: 35.9 ± 1.1 kg/m2 | (10/9) | 5 to 7 weeks | Greater weight loss and increased fat oxidation in RC diet than RF diet at the 6th day and greater fat loss (463 ± 37 g) in the RF diet compared to the RC diet (245 ± 21 g). | [160] |
| 5 | Dyson et al. | 2007 | LCD (≤40 g CHO/day) or healthy-eating diet | Overweight or obese with T2DM or non-diabetic Mean age: 52 ± 9 y BMI: 35.1 ± 7.0 kg/m2 | (8/18) | 3 months | Weight loss LCD: −6.9 kg Healthy eating diet: −2.1 kg No difference in changes in HbA1c, ketone, or lipid levels. | [169] |
| 6 | Goday et al. | 2016 | Very low-calorie-ketogenic (VLCK) or low-calorie diet | Obese adults with T2DM Mean age: 54.5 ± 8.4 y BMI: 33.07 ± 1.56 kg/m2 | (31/58) | 4 months | Weight loss VLCK: −14.7 kg LC: −5 kg The reduction in HbA1c and glycemic control was greater in the VLCK group. | [170] |
| 7 | Harvey et al. | 2019 | VLCKD (5% CHO) or LCD (15% CHO) or moderate-low CHO diet (MCD) (25% CHO) | Healthy adults Mean age: 38.9 ± 7.1 y BMI: 27.0 ± 3.96 kg/m2 | (14/25) | 12 weeks | Weight loss VLCKD: −4.12 kg LCD: −3.93 kg MCD: −2.97 kg Similar reductions in total cholesterol, LDL, TG, and increase in HDL | [171] |
| 8 | Dalle Grave et al. | 2013 | HPD (34% protein, 46% CHO) or HCD (17% proteins, 64% CHO) | Obese adults Mean age: 46.7 ± 11.1 y BMI: 45.6 ± 6.7 kg/m2 | (37/51) | 1 year | Weight loss HPD: −18.1 kg (15.0%) HCD: −15.9 (13.3%) Similar reductions in TG, LDL, total cholesterol, glucose, and insulin levels and increase in HDL. | [179] |
| Effects of Plant-Based Diets on Body Weight and Health | ||||||||
| Author | Year | Diet | Participants | n (M/F) | Duration | Main Findings | Ref | |
| 1 | Wright et al. | 2017 | Low-fat plant-based diet (7–15% total energy from fat) or control | Obese, overweight, and diagnosed with at least one of T2DM, ischaemic heart disease, hypertension or hypercholesterolaemia Mean age: 56 ± 9.7 y BMI: 34.3 ± 1.9 kg/m2 | (26/39) | 6 to 12 months | Significant reduction in BMI (4.2 kg/m2) in diet group | [191] |
| 2 | Thompson et al. | 2005 | Standard diet or high-dairy diet or high-fiber and high-dairy diet | Obese adults Mean age: 41.4 ± 8.9 y BMI: 34.8 ± 3.1 kg/m2 | (72/0) | 48 weeks | Similar weight loss in all diet groups Standard diet: 10.1 kg High-dairy diet: 11.7 kg High fiber and high dairy diet: 10.4 Similar fat mass loss in all diet groups Standard diet: −7.5 kg High-dairy diet: −9.0 kg High fiber and high dairy diet: −8.5 kg Similar increase in HDL and reductions in total cholesterol, LDL, fasting glucose and insulin, leptin, hs-CRP | [197] |
| 3 | Turner-McGrievy et al. | 2015 | Vegan Vegetarian Pesco-vegetarian Semi-vegetarian Omnivorous | Overweight or obese adults Mean age: 48.74 ± 7.5 y BMI: 34.96 ± 5.2 kg/m2 | (17/46) | 6 months | Weight loss Vegan: −7.5% Vegetarian: −6.3% Pesco-vegetarian: −3.2% Semi-vegetarian: −3.2% Omnivorous: −3.1% | [205] |
| Effects of Intermittent Fasting on Body Weight and Health | ||||||||
| Author | Year | Diet | Participants | n (M/F) | Duration | Main Findings | Ref | |
| 1 | Varady et al. | 2009 | Alternate day fasting (Fast day: 25% of energy needs, alternated day: ad libitum food intake) | Obese adults Mean age: 46.0 ± 2.4 y BMI: 33.8 ± 1.0 kg/m2 | (4/12) | 10 weeks | Weight loss −5.6 ± 1.0 kg Body fat percent decreased from 45 ± 2% to 42 ± 2% Significant decreases in total cholesterol, LDL, TG, and blood pressure | [209] |
| 2 | Harvie et al. | 2011 | IER (∼2710 kJ/day for 2 days/week) or CER (∼6276 kJ/day for 7 days/week) | Overweight or obese premenopausal women Mean age: 40.05 y BMI: 30.6 ± 5.1 kg/m2 | (0/107) | 6 months | Weight loss IER: −6.4 kg CER: −5.6 kg Similar reductions in hs-CRP, leptin, total cholesterol, LDL, TG, and blood pressure. Reductions in fasting insulin and insulin resistance in both groups greater in the IER group. | [212] |
| 3 | Schübel et al. | 2018 | ICR (5:2, weekly energy deficit ∼20%) or CCR (daily energy deficit ∼20%) or control group | Overweight and obese adults Mean age: 50.2 ± 8 y BMI: 31.4 ± 3.8 kg/m2 | ICR: n: 49 CCR: n: 49 Control: n:52 | 50 weeks | Weight loss ICR: −5.2% ± 1.2%, CCR: −4.9% ± 1.1% Control: −1.7% ± 0.8% Similar reductions LDL, HDL, cholesterol, TG, insulin, HOMA-IR levels, and adipokines (adiponectin, leptin) | [213] |
5. Cytokines That Burn Our Fire: Are They the Cornerstones of Obesity Treatment for the Foreseeable Future?
5.1. Brain-Derived Neurotrophic Factor
5.2. β-aminoisobutyric Acid
5.3. Interleukin-6
5.4. Interleukin-15
5.5. Irisin
5.6. Meteorin-Like
6. Summary and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chooi, Y.C.; Ding, C.; Magkos, F. The epidemiology of obesity. Metabolism 2019, 92, 6–10. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Obesity and Overweight. Fact Sheet. Available online: https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed on 25 February 2021).
- Stamatakis, E.; Chau, J.Y.; Pedisic, Z.; Bauman, A.; Macniven, R.; Coombs, N.; Hamer, M. Are Sitting Occupations Associated with Increased All-Cause, Cancer, and Cardiovascular Disease Mortality Risk? A Pooled Analysis of Seven British Population Cohorts. PLoS ONE 2013, 8, e73753. [Google Scholar] [CrossRef] [PubMed]
- Bray, G.A.; Kim, K.K.; Wilding, J.P.H. Obesity: A chronic relapsing progressive disease process. A position statement of the World Obesity Federation. Obes. Rev. 2017, 18, 715–723. [Google Scholar] [CrossRef] [PubMed]
- Sarma, S.; Sockalingam, S.; Dash, S. Obesity as a multisystem disease: Trends in obesity rates and obesity-related complications. Diabetes Obes. Metab. 2021, 23 (Suppl. 1), 3–16. [Google Scholar] [CrossRef]
- Di Angelantonio, E.; Bhupathiraju Sh, N.; Wormser, D.; Gao, P.; Kaptoge, S.; Berrington de Gonzalez, A.; Cairns, B.J.; Huxley, R.; Jackson Ch, L.; Joshy, G.; et al. Body-mass index and all-cause mortality: Individual-participant-data meta-analysis of 239 prospective studies in four continents. Lancet 2016, 388, 776–786. [Google Scholar] [CrossRef]
- Bhaskaran, K.; Dos-Santos-Silva, I.; Leon, D.A.; Douglas, I.J.; Smeeth, L. Association of BMI with overall and cause-specific mortality: A population-based cohort study of 3·6 million adults in the UK. Lancet Diabetes Endocrinol. 2018, 6, 944–953. [Google Scholar] [CrossRef]
- Piché, M.E.; Tchernof, A.; Després, J.P. Obesity Phenotypes, Diabetes, and Cardiovascular Diseases. Circ. Res. 2020, 126, 1477–1500. [Google Scholar] [CrossRef]
- Boles, A.; Kandimalla, R.; Reddy, P.H. Dynamics of diabetes and obesity: Epidemiological perspective. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1026–1036. [Google Scholar] [CrossRef]
- Vekic, J.; Zeljkovic, A.; Stefanovic, A.; Jelic-Ivanovic, Z.; Spasojevic-Kalimanovska, V. Obesity and dyslipidemia. Metab. Clin. Exp. 2019, 92, 71–81. [Google Scholar] [CrossRef] [PubMed]
- Gerges, S.H.; Wahdan, S.A.; Elsherbiny, D.A.; El-Demerdash, E. Non-alcoholic fatty liver disease: An overview of risk factors, pathophysiological mechanisms, diagnostic procedures, and therapeutic interventions. Life Sci. 2021, 271, 119220. [Google Scholar] [CrossRef] [PubMed]
- Das, E.; Moon, J.H.; Lee, J.H.; Thakkar, N.; Pausova, Z.; Sung, H.K. Adipose Tissue and Modulation of Hypertension. Curr. Hypertens. Rep. 2018, 20, 96. [Google Scholar] [CrossRef] [PubMed]
- Câmara, N.O.; Iseki, K.; Kramer, H.; Liu, Z.H.; Sharma, K. Kidney disease and obesity: Epidemiology, mechanisms and treatment. Nat. Rev. Nephrol. 2017, 13, 181–190. [Google Scholar] [CrossRef] [PubMed]
- Basen-Engquist, K.; Chang, M. Obesity and cancer risk: Recent review and evidence. Curr. Oncol. Rep. 2011, 13, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Avgerinos, K.I.; Spyrou, N.; Mantzoros, C.S.; Dalamaga, M. Obesity and cancer risk: Emerging biological mechanisms and perspectives. Metab. Clin. Exp. 2019, 92, 121–135. [Google Scholar] [CrossRef]
- Tremmel, M.; Gerdtham, U.G.; Nilsson, P.M.; Saha, S. Economic Burden of Obesity: A Systematic Literature Review. Int. J. Environ. Res. Public Health 2017, 14, 435. [Google Scholar] [CrossRef]
- Pedisic, Z.; Grunseit, A.; Ding, D.; Chau, J.Y.; Banks, E.; Stamatakis, E.; Jalaludin, B.B.; Bauman, A.E. High sitting time or obesity: Which came first? Bidirectional association in a longitudinal study of 31,787 Australian adults. Obesity 2014, 22, 2126–2130. [Google Scholar] [CrossRef]
- Shrestha, N.; Pedisic, Z.; Neil-Sztramko, S.; Kukkonen-Harjula, K.T.; Hermans, V. The Impact of Obesity in the Workplace: A Review of Contributing Factors, Consequences and Potential Solutions. Curr. Obes. Rep. 2016, 5, 344–360. [Google Scholar] [CrossRef]
- Dai, H.; Alsalhe, T.A.; Chalghaf, N.; Riccò, M.; Bragazzi, N.L.; Wu, J. The global burden of disease attributable to high body mass index in 195 countries and territories, 1990-2017: An analysis of the Global Burden of Disease Study. PLoS Med. 2020, 17, e1003198. [Google Scholar] [CrossRef]
- Chouchani, E.T.; Kajimura, S. Metabolic adaptation and maladaptation in adipose tissue. Nat. Metab. 2019, 1, 189–200. [Google Scholar] [CrossRef]
- Feng, B.; Zhang, T.; Xu, H. Human adipose dynamics and metabolic health. Ann. N. Y. Acad. Sci. 2013, 1281, 160–177. [Google Scholar] [CrossRef]
- Tsatsoulis, A.; Paschou, S.A. Metabolically Healthy Obesity: Criteria, Epidemiology, Controversies, and Consequences. Curr. Obes. Rep. 2020, 9, 109–120. [Google Scholar] [CrossRef]
- Ghaben, A.L.; Scherer, P.E. Adipogenesis and metabolic health. Nat. Rev. Mol. Cell Biol. 2019, 20, 242–258. [Google Scholar] [CrossRef]
- Goossens, G.H. The Metabolic Phenotype in Obesity: Fat Mass, Body Fat Distribution, and Adipose Tissue Function. Obes. Facts 2017, 10, 207–215. [Google Scholar] [CrossRef]
- Iacobini, C.; Pugliese, G.; Blasetti Fantauzzi, C.; Federici, M.; Menini, S. Metabolically healthy versus metabolically unhealthy obesity. Metabolism 2019, 92, 51–60. [Google Scholar] [CrossRef]
- Goossens, G.H.; Blaak, E.E. Adipose tissue dysfunction and impaired metabolic health in human obesity: A matter of oxygen? Front. Endocrinol. 2015, 6, 55. [Google Scholar] [CrossRef] [PubMed]
- Kumanyika, S.K.; Obarzanek, E.; Stettler, N.; Bell, R.; Field, A.E.; Fortmann, S.P.; Franklin, B.A.; Gillman, M.W.; Lewis, C.E.; Poston, W.C., 2nd; et al. Population-based prevention of obesity: The need for comprehensive promotion of healthful eating, physical activity, and energy balance: A scientific statement from American Heart Association Council on Epidemiology and Prevention, Interdisciplinary Committee for Prevention (formerly the expert panel on population and prevention science). Circulation 2008, 118, 428–464. [Google Scholar] [CrossRef]
- Lagerros, Y.T.; Rossner, S. Obesity management: What brings success? Therap. Adv. Gastroenterol. 2013, 6, 77–88. [Google Scholar] [CrossRef] [PubMed]
- Kheniser, K.; Saxon, D.R.; Kashyap, S.R. Long-term weight loss strategies for obesity. J. Clin. Endocrinol. Metab. 2021. [Google Scholar] [CrossRef] [PubMed]
- Kolahdouzi, S.; Talebi-Garakani, E.; Hamidian, G.; Safarzade, A. Exercise training prevents high-fat diet-induced adipose tissue remodeling by promoting capillary density and macrophage polarization. Life Sci. 2019, 220, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.; Soni, S.; Houle, S.A. Negative Energy Balance Induced by Exercise or Diet: Effects on Visceral Adipose Tissue and Liver Fat. Nutrients 2020, 12, 891. [Google Scholar] [CrossRef] [PubMed]
- Verheggen, R.J.; Maessen, M.F.; Green, D.J.; Hermus, A.R.; Hopman, M.T.; Thijssen, D.H. A systematic review and meta-analysis on the effects of exercise training versus hypocaloric diet: Distinct effects on body weight and visceral adipose tissue. Obes. Rev. 2016, 17, 664–690. [Google Scholar] [CrossRef] [PubMed]
- Rabiee, A. Beige Fat Maintenance; Toward a Sustained Metabolic Health. Front. Endocrinol. 2020, 11, 634. [Google Scholar] [CrossRef] [PubMed]
- Pedersen, B.K.; Febbraio, M.A. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Garg, A. Adipose tissue dysfunction in obesity and lipodystrophy. Clin. Cornerstone 2006, 8 (Suppl. 4), S7–S13. [Google Scholar] [CrossRef]
- Kucuk Baloglu, F.; Garip, S.; Heise, S.; Brockmann, G.; Severcan, F. FTIR imaging of structural changes in visceral and subcutaneous adiposity and brown to white adipocyte transdifferentiation. Analyst 2015, 140, 2205–2214. [Google Scholar] [CrossRef] [PubMed]
- Thompson, D.; Karpe, F.; Lafontan, M.; Frayn, K. Physical activity and exercise in the regulation of human adipose tissue physiology. Physiol. Rev. 2012, 92, 157–191. [Google Scholar] [CrossRef] [PubMed]
- Kershaw, E.E.; Flier, J.S. Adipose tissue as an endocrine organ. J. Clin. Endocrinol. Metab. 2004, 89, 2548–2556. [Google Scholar] [CrossRef]
- Yan, X.; Zhu, M.J.; Dodson, M.V.; Du, M. Developmental programming of fetal skeletal muscle and adipose tissue development. J. Genom. 2013, 1, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Grundy, S.M. Adipose tissue and metabolic syndrome: Too much, too little or neither. Eur. J. Clin. Investig. 2015, 45, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Saely, C.H.; Geiger, K.; Drexel, H. Brown versus white adipose tissue: A mini-review. Gerontology 2012, 58, 15–23. [Google Scholar] [CrossRef]
- Tam, C.S.; Lecoultre, V.; Ravussin, E. Brown adipose tissue: Mechanisms and potential therapeutic targets. Circulation 2012, 125, 2782–2791. [Google Scholar] [CrossRef]
- Korta, P.; Pochec, E.; Mazur-Bialy, A. Irisin as a Multifunctional Protein: Implications for Health and Certain Diseases. Medicina 2019, 55, 485. [Google Scholar] [CrossRef]
- Wronska, A.; Kmiec, Z. Structural and biochemical characteristics of various white adipose tissue depots. Acta Physiol. 2012, 205, 194–208. [Google Scholar] [CrossRef]
- Stanford, K.I.; Goodyear, L.J. Exercise regulation of adipose tissue. Adipocyte 2016, 5, 153–162. [Google Scholar] [CrossRef]
- Wu, J.; Bostrom, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366–376. [Google Scholar] [CrossRef] [PubMed]
- Lehnig, A.C.; Stanford, K.I. Exercise-induced adaptations to white and brown adipose tissue. J. Exp. Biol. 2018, 221. [Google Scholar] [CrossRef] [PubMed]
- Petrovic, N.; Walden, T.B.; Shabalina, I.G.; Timmons, J.A.; Cannon, B.; Nedergaard, J. Chronic peroxisome proliferator-activated receptor gamma (PPARgamma) activation of epididymally derived white adipocyte cultures reveals a population of thermogenically competent, UCP1-containing adipocytes molecularly distinct from classic brown adipocytes. J. Biol. Chem. 2010, 285, 7153–7164. [Google Scholar] [CrossRef]
- Stanford, K.I.; Middelbeek, R.J.; Townsend, K.L.; Lee, M.Y.; Takahashi, H.; So, K.; Hitchcox, K.M.; Markan, K.R.; Hellbach, K.; Hirshman, M.F.; et al. A novel role for subcutaneous adipose tissue in exercise-induced improvements in glucose homeostasis. Diabetes 2015, 64, 2002–2014. [Google Scholar] [CrossRef] [PubMed]
- Stanford, K.I.; Middelbeek, R.J.; Goodyear, L.J. Exercise Effects on White Adipose Tissue: Beiging and Metabolic Adaptations. Diabetes 2015, 64, 2361–2368. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Choi, E.Y.; Liu, X.; Martin, A.; Wang, C.; Xu, X.; During, M.J. White to brown fat phenotypic switch induced by genetic and environmental activation of a hypothalamic-adipocyte axis. Cell Metab. 2011, 14, 324–338. [Google Scholar] [CrossRef]
- Elabd, C.; Chiellini, C.; Carmona, M.; Galitzky, J.; Cochet, O.; Petersen, R.; Penicaud, L.; Kristiansen, K.; Bouloumie, A.; Casteilla, L.; et al. Human multipotent adipose-derived stem cells differentiate into functional brown adipocytes. Stem Cells 2009, 27, 2753–2760. [Google Scholar] [CrossRef] [PubMed]
- Bishop, D.J.; Botella, J.; Genders, A.J.; Lee, M.J.; Saner, N.J.; Kuang, J.; Yan, X.; Granata, C. High-Intensity Exercise and Mitochondrial Biogenesis: Current Controversies and Future Research Directions. Physiology 2019, 34, 56–70. [Google Scholar] [CrossRef] [PubMed]
- Pedisic, Z.; Shrestha, N.; Kovalchik, S.; Stamatakis, E.; Liangruenrom, N.; Grgic, J.; Titze, S.; Biddle, S.J.; Bauman, A.E.; Oja, P. Is running associated with a lower risk of all-cause, cardiovascular and cancer mortality, and is the more the better? A systematic review and meta-analysis. Br. J. Sports Med. 2020, 54, 898–905. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Li, Y.; Atakan, M.M.; Kuang, J.; Hu, Y.; Bishop, D.J.; Yan, X. The Molecular Adaptive Responses of Skeletal Muscle to High-Intensity Exercise/Training and Hypoxia. Antioxidants 2020, 9, 656. [Google Scholar] [CrossRef] [PubMed]
- Vidal, P.; Stanford, K.I. Exercise-Induced Adaptations to Adipose Tissue Thermogenesis. Front. Endocrinol. 2020, 11, 270. [Google Scholar] [CrossRef] [PubMed]
- Wareham, N. Physical activity and obesity prevention. Obes. Rev. 2007, 8, 109–114. [Google Scholar] [CrossRef]
- Wareham, N.J.; van Sluijs, E.M.; Ekelund, U. Physical activity and obesity prevention: A review of the current evidence. Proc. Nutr. Soc. 2005, 64, 229–247. [Google Scholar] [CrossRef]
- Ross, R.; Dagnone, D.; Jones, P.J.; Smith, H.; Paddags, A.; Hudson, R.; Janssen, I. Reduction in obesity and related comorbid conditions after diet-induced weight loss or exercise-induced weight loss in men. A randomized, controlled trial. Ann. Intern. Med. 2000, 133, 92–103. [Google Scholar] [CrossRef]
- Racette, S.B.; Weiss, E.P.; Villareal, D.T.; Arif, H.; Steger-May, K.; Schechtman, K.B.; Fontana, L.; Klein, S.; Holloszy, J.O.; Washington University School of Medicine, C.G. One year of caloric restriction in humans: Feasibility and effects on body composition and abdominal adipose tissue. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 943–950. [Google Scholar] [CrossRef]
- Miyatake, N.; Nishikawa, H.; Morishita, A.; Kunitomi, M.; Wada, J.; Suzuki, H.; Takahashi, K.; Makino, H.; Kira, S.; Fujii, M. Daily walking reduces visceral adipose tissue areas and improves insulin resistance in Japanese obese subjects. Diabetes Res. Clin. Pract. 2002, 58, 101–107. [Google Scholar] [CrossRef]
- Durheim, M.T.; Slentz, C.A.; Bateman, L.A.; Mabe, S.K.; Kraus, W.E. Relationships between exercise-induced reductions in thigh intermuscular adipose tissue, changes in lipoprotein particle size, and visceral adiposity. Am. J. Physiol. Endocrinol. Metab. 2008, 295, E407–E412. [Google Scholar] [CrossRef] [PubMed]
- Ross, R.; Hudson, R.; Stotz, P.J.; Lam, M. Effects of exercise amount and intensity on abdominal obesity and glucose tolerance in obese adults: A randomized trial. Ann. Intern. Med. 2015, 162, 325–334. [Google Scholar] [CrossRef] [PubMed]
- Brennan, A.M.; Day, A.G.; Cowan, T.E.; Clarke, G.J.; Lamarche, B.; Ross, R. Individual Response to Standardized Exercise: Total and Abdominal Adipose Tissue. Med. Sci. Sports Exerc. 2019. [Google Scholar] [CrossRef]
- Wilmore, J.H.; Despres, J.P.; Stanforth, P.R.; Mandel, S.; Rice, T.; Gagnon, J.; Leon, A.S.; Rao, D.; Skinner, J.S.; Bouchard, C. Alterations in body weight and composition consequent to 20 wk of endurance training: The HERITAGE Family Study. Am. J. Clin. Nutr. 1999, 70, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Irving, B.A.; Davis, C.K.; Brock, D.W.; Weltman, J.Y.; Swift, D.; Barrett, E.J.; Gaesser, G.A.; Weltman, A. Effect of exercise training intensity on abdominal visceral fat and body composition. Med. Sci. Sports Exerc. 2008, 40, 1863–1872. [Google Scholar] [CrossRef] [PubMed]
- Coker, R.H.; Williams, R.H.; Kortebein, P.M.; Sullivan, D.H.; Evans, W.J. Influence of exercise intensity on abdominal fat and adiponectin in elderly adults. Metab. Syndr. Relat. Disord. 2009, 7, 363–368. [Google Scholar] [CrossRef]
- Tanaka, R.; Fuse, S.; Kuroiwa, M.; Amagasa, S.; Endo, T.; Ando, A.; Kime, R.; Kurosawa, Y.; Hamaoka, T. Vigorous-Intensity Physical Activities Are Associated with High Brown Adipose Tissue Density in Humans. Int. J. Environ. Res. Public Health 2020, 17, 2796. [Google Scholar] [CrossRef] [PubMed]
- Owens, S.; Gutin, B.; Allison, J.; Riggs, S.; Ferguson, M.; Litaker, M.; Thompson, W. Effect of physical training on total and visceral fat in obese children. Med. Sci. Sports Exerc. 1999, 31, 143–148. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.; Kuk, J.L.; Davidson, L.E.; Hudson, R.; Kilpatrick, K.; Graham, T.E.; Ross, R. Exercise without weight loss is an effective strategy for obesity reduction in obese individuals with and without Type 2 diabetes. J. Appl. Physiol. 2005, 99, 1220–1225. [Google Scholar] [CrossRef]
- Keating, S.E.; Hackett, D.A.; Parker, H.M.; O’Connor, H.T.; Gerofi, J.A.; Sainsbury, A.; Baker, M.K.; Chuter, V.H.; Caterson, I.D.; George, J.; et al. Effect of aerobic exercise training dose on liver fat and visceral adiposity. J. Hepatol. 2015, 63, 174–182. [Google Scholar] [CrossRef]
- Christiansen, T.; Paulsen, S.K.; Bruun, J.M.; Overgaard, K.; Ringgaard, S.; Pedersen, S.B.; Positano, V.; Richelsen, B. Comparable reduction of the visceral adipose tissue depot after a diet-induced weight loss with or without aerobic exercise in obese subjects: A 12-week randomized intervention study. Eur. J. Endocrinol. 2009, 160, 759–767. [Google Scholar] [CrossRef]
- Walhin, J.P.; Dixon, N.C.; Betts, J.A.; Thompson, D. The impact of exercise intensity on whole body and adipose tissue metabolism during energy restriction in sedentary overweight men and postmenopausal women. Physiol. Rep. 2016, 4. [Google Scholar] [CrossRef] [PubMed]
- Islam, H.; Townsend, L.K.; Hazell, T.J. Excess Postexercise Oxygen Consumption and Fat Utilization Following Submaximal Continuous and Supramaximal Interval Running. Res. Q. Exerc. Sport 2018, 89, 450–456. [Google Scholar] [CrossRef] [PubMed]
- Henderson, G.C.; Fattor, J.A.; Horning, M.A.; Faghihnia, N.; Johnson, M.L.; Mau, T.L.; Luke-Zeitoun, M.; Brooks, G.A. Lipolysis and fatty acid metabolism in men and women during the postexercise recovery period. J. Physiol. 2007, 584, 963–981. [Google Scholar] [CrossRef]
- Perreault, L.; Lavely, J.M.; Kittelson, J.M.; Horton, T.J. Gender differences in lipoprotein lipase activity after acute exercise. Obes. Res. 2004, 12, 241–249. [Google Scholar] [CrossRef]
- Ludzki, A.C.; Krueger, E.M.; Baldwin, T.C.; Schleh, M.W.; Porsche, C.E.; Ryan, B.J.; Muir, L.A.; Singer, K.; Lumeng, C.N.; Horowitz, J.F. Acute Aerobic Exercise Remodels the Adipose Tissue Progenitor Cell Phenotype in Obese Adults. Front. Physiol. 2020, 11, 903. [Google Scholar] [CrossRef] [PubMed]
- Hojbjerre, L.; Rosenzweig, M.; Dela, F.; Bruun, J.M.; Stallknecht, B. Acute exercise increases adipose tissue interstitial adiponectin concentration in healthy overweight and lean subjects. Eur. J. Endocrinol. 2007, 157, 613–623. [Google Scholar] [CrossRef]
- Van Pelt, D.W.; Guth, L.M.; Horowitz, J.F. Aerobic exercise elevates markers of angiogenesis and macrophage IL-6 gene expression in the subcutaneous adipose tissue of overweight-to-obese adults. J. Appl. Physiol. 2017, 123, 1150–1159. [Google Scholar] [CrossRef]
- Fabre, O.; Ingerslev, L.R.; Garde, C.; Donkin, I.; Simar, D.; Barres, R. Exercise training alters the genomic response to acute exercise in human adipose tissue. Epigenomics 2018, 10, 1033–1050. [Google Scholar] [CrossRef]
- Campbell, K.L.; Foster-Schubert, K.E.; Makar, K.W.; Kratz, M.; Hagman, D.; Schur, E.A.; Habermann, N.; Horton, M.; Abbenhardt, C.; Kuan, L.Y.; et al. Gene expression changes in adipose tissue with diet- and/or exercise-induced weight loss. Cancer Prev. Res. 2013, 6, 217–231. [Google Scholar] [CrossRef]
- Tam, C.S.; Covington, J.D.; Ravussin, E.; Redman, L.M. Little evidence of systemic and adipose tissue inflammation in overweight individuals. Front. Genet. 2012, 3, 58. [Google Scholar] [CrossRef]
- Richterova, B.; Stich, V.; Moro, C.; Polak, J.; Klimcakova, E.; Majercik, M.; Harant, I.; Viguerie, N.; Crampes, F.; Langin, D.; et al. Effect of endurance training on adrenergic control of lipolysis in adipose tissue of obese women. J. Clin. Endocrinol. Metab. 2004, 89, 1325–1331. [Google Scholar] [CrossRef][Green Version]
- Polak, J.; Klimcakova, E.; Moro, C.; Viguerie, N.; Berlan, M.; Hejnova, J.; Richterova, B.; Kraus, I.; Langin, D.; Stich, V. Effect of aerobic training on plasma levels and subcutaneous abdominal adipose tissue gene expression of adiponectin, leptin, interleukin 6, and tumor necrosis factor alpha in obese women. Metab. Clin. Exp. 2006, 55, 1375–1381. [Google Scholar] [CrossRef] [PubMed]
- Leick, L.; Lindegaard, B.; Stensvold, D.; Plomgaard, P.; Saltin, B.; Pilegaard, H. Adipose tissue interleukin-18 mRNA and plasma interleukin-18: Effect of obesity and exercise. Obesity 2007, 15, 356–363. [Google Scholar] [CrossRef] [PubMed]
- Christiansen, T.; Paulsen, S.K.; Bruun, J.M.; Ploug, T.; Pedersen, S.B.; Richelsen, B. Diet-induced weight loss and exercise alone and in combination enhance the expression of adiponectin receptors in adipose tissue and skeletal muscle, but only diet-induced weight loss enhanced circulating adiponectin. J. Clin. Endocrinol. Metab. 2010, 95, 911–919. [Google Scholar] [CrossRef]
- Stanford, K.I.; Goodyear, L.J. Muscle-Adipose Tissue Cross Talk. Cold Spring Harb. Perspect. Med. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Trevellin, E.; Scorzeto, M.; Olivieri, M.; Granzotto, M.; Valerio, A.; Tedesco, L.; Fabris, R.; Serra, R.; Quarta, M.; Reggiani, C.; et al. Exercise training induces mitochondrial biogenesis and glucose uptake in subcutaneous adipose tissue through eNOS-dependent mechanisms. Diabetes 2014, 63, 2800–2811. [Google Scholar] [CrossRef]
- Cypess, A.M.; Lehman, S.; Williams, G.; Tal, I.; Rodman, D.; Goldfine, A.B.; Kuo, F.C.; Palmer, E.L.; Tseng, Y.H.; Doria, A.; et al. Identification and importance of brown adipose tissue in adult humans. N. Engl. J. Med. 2009, 360, 1509–1517. [Google Scholar] [CrossRef] [PubMed]
- Vosselman, M.J.; Hoeks, J.; Brans, B.; Pallubinsky, H.; Nascimento, E.B.; van der Lans, A.A.; Broeders, E.P.; Mottaghy, F.M.; Schrauwen, P.; van Marken Lichtenbelt, W.D. Low brown adipose tissue activity in endurance-trained compared with lean sedentary men. Int. J. Obes. 2015, 39, 1696–1702. [Google Scholar] [CrossRef]
- Otero-Diaz, B.; Rodriguez-Flores, M.; Sanchez-Munoz, V.; Monraz-Preciado, F.; Ordonez-Ortega, S.; Becerril-Elias, V.; Baay-Guzman, G.; Obando-Monge, R.; Garcia-Garcia, E.; Palacios-Gonzalez, B.; et al. Exercise Induces White Adipose Tissue Browning Across the Weight Spectrum in Humans. Front. Physiol. 2018, 9, 1781. [Google Scholar] [CrossRef]
- Donnelly, J.E.; Blair, S.N.; Jakicic, J.M.; Manore, M.M.; Rankin, J.W.; Smith, B.K. American College of Sports Medicine Position Stand. Appropriate physical activity intervention strategies for weight loss and prevention of weight regain for adults. Med. Sci. Sports Exerc. 2009, 41, 459–471. [Google Scholar] [CrossRef] [PubMed]
- Hallal, P.C.; Andersen, L.B.; Bull, F.C.; Guthold, R.; Haskell, W.; Ekelund, U. Global physical activity levels: Surveillance progress, pitfalls, and prospects. Lancet 2012, 380, 247–257. [Google Scholar] [CrossRef]
- Higgins, S.; Fedewa, M.V.; Hathaway, E.D.; Schmidt, M.D.; Evans, E.M. Sprint interval and moderate-intensity cycling training differentially affect adiposity and aerobic capacity in overweight young-adult women. Appl. Physiol. Nutr. Metab. 2016, 41, 1177–1183. [Google Scholar] [CrossRef] [PubMed]
- Atakan, M.M.; Güzel, Y.; Bulut, S.; Koşar, N.; McConell, G.K.; Turnagöl, H.H. Six high-intensity interval training sessions over 5 days increases maximal oxygen uptake, endurance capacity, and sub-maximal exercise fat oxidation as much as 6 high-intensity interval training sessions over 2 weeks. J. Sport Health Sci. 2020. [Google Scholar] [CrossRef]
- Little, J.P.; Safdar, A.; Wilkin, G.P.; Tarnopolsky, M.A.; Gibala, M.J. A practical model of low-volume high-intensity interval training induces mitochondrial biogenesis in human skeletal muscle: Potential mechanisms. J. Physiol. 2010, 588, 1011–1022. [Google Scholar] [CrossRef]
- Zhang, H.; Tong, T.K.; Qiu, W.; Zhang, X.; Zhou, S.; Liu, Y.; He, Y. Comparable Effects of High-Intensity Interval Training and Prolonged Continuous Exercise Training on Abdominal Visceral Fat Reduction in Obese Young Women. J. Diabetes Res. 2017, 2017, 5071740. [Google Scholar] [CrossRef] [PubMed]
- Riis, S.; Christensen, B.; Nellemann, B.; Moller, A.B.; Husted, A.S.; Pedersen, S.B.; Schwartz, T.W.; Jorgensen, J.O.L.; Jessen, N. Molecular adaptations in human subcutaneous adipose tissue after ten weeks of endurance exercise training in healthy males. J. Appl. Physiol. 2019, 126, 569–577. [Google Scholar] [CrossRef]
- Dohlmann, T.L.; Hindso, M.; Dela, F.; Helge, J.W.; Larsen, S. High-intensity interval training changes mitochondrial respiratory capacity differently in adipose tissue and skeletal muscle. Physiol. Rep. 2018, 6, e13857. [Google Scholar] [CrossRef]
- Leggate, M.; Carter, W.G.; Evans, M.J.; Vennard, R.A.; Sribala-Sundaram, S.; Nimmo, M.A. Determination of inflammatory and prominent proteomic changes in plasma and adipose tissue after high-intensity intermittent training in overweight and obese males. J. Appl. Physiol. 2012, 112, 1353–1360. [Google Scholar] [CrossRef]
- Islam, H.; Smith, M.M.W.; Scribbans, T.D.; McCrady, E.; Castellani, L.N.; Allen, M.D.; Wright, D.C.; Simpson, C.A.; Gurd, B.J. Effect of Acute High-intensity Interval Exercise on Whole-body Fat Oxidation and Subcutaneous Adipose Tissue Cell Signaling in Overweight Women. Int. J. Exerc. Sci. 2020, 13, 554–566. [Google Scholar]
- Astorino, T.A.; Schubert, M.M.; Palumbo, E.; Stirling, D.; McMillan, D.W. Effect of two doses of interval training on maximal fat oxidation in sedentary women. Med. Sci. Sports Exerc. 2013, 45, 1878–1886. [Google Scholar] [CrossRef]
- Taylor, J.L.; Holland, D.J.; Mielke, G.I.; Bailey, T.G.; Johnson, N.A.; Leveritt, M.D.; Gomersall, S.R.; Rowlands, A.V.; Coombes, J.S.; Keating, S.E. Effect of High-Intensity Interval Training on Visceral and Liver Fat in Cardiac Rehabilitation: A Randomized Controlled Trial. Obesity 2020, 28, 1245–1253. [Google Scholar] [CrossRef] [PubMed]
- Keating, S.E.; Johnson, N.A.; Mielke, G.I.; Coombes, J.S. A systematic review and meta-analysis of interval training versus moderate-intensity continuous training on body adiposity. Obes. Rev. 2017, 18, 943–964. [Google Scholar] [CrossRef] [PubMed]
- Larsen, S.; Danielsen, J.H.; Sondergard, S.D.; Sogaard, D.; Vigelsoe, A.; Dybboe, R.; Skaaby, S.; Dela, F.; Helge, J.W. The effect of high-intensity training on mitochondrial fat oxidation in skeletal muscle and subcutaneous adipose tissue. Scand. J. Med. Sci. Sports 2015, 25, e59–e69. [Google Scholar] [CrossRef]
- Whyte, L.J.; Gill, J.M.; Cathcart, A.J. Effect of 2 weeks of sprint interval training on health-related outcomes in sedentary overweight/obese men. Metabolism 2010, 59, 1421–1428. [Google Scholar] [CrossRef]
- Honkala, S.M.; Motiani, P.; Kivela, R.; Hemanthakumar, K.A.; Tolvanen, E.; Motiani, K.K.; Eskelinen, J.J.; Virtanen, K.A.; Kemppainen, J.; Heiskanen, M.A.; et al. Exercise training improves adipose tissue metabolism and vasculature regardless of baseline glucose tolerance and sex. BMJ Open Diabetes Res. Care 2020, 8. [Google Scholar] [CrossRef]
- Cooper, J.H.; Collins, B.E.; Adams, D.R.; Robergs, R.A.; Donges, C.E. Limited Effects of Endurance or Interval Training on Visceral Adipose Tissue and Systemic Inflammation in Sedentary Middle-Aged Men. J. Obes. 2016, 2016, 2479597. [Google Scholar] [CrossRef]
- Schmitz, K.H.; Jensen, M.D.; Kugler, K.C.; Jeffery, R.W.; Leon, A.S. Strength training for obesity prevention in midlife women. Int. J. Obes. Relat. Metab. Disord. 2003, 27, 326–333. [Google Scholar] [CrossRef]
- Ku, Y.H.; Han, K.A.; Ahn, H.; Kwon, H.; Koo, B.K.; Kim, H.C.; Min, K.W. Resistance exercise did not alter intramuscular adipose tissue but reduced retinol-binding protein-4 concentration in individuals with type 2 diabetes mellitus. J. Int. Med. Res. 2010, 38, 782–791. [Google Scholar] [CrossRef]
- Grgic, J.; Schoenfeld, B.J.; Davies, T.B.; Lazinica, B.; Krieger, J.W.; Pedisic, Z. Effect of Resistance Training Frequency on Gains in Muscular Strength: A Systematic Review and Meta-Analysis. Sports Med. 2018, 48, 1207–1220. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Grgic, J.; Ogborn, D.; Krieger, J.W. Strength and Hypertrophy Adaptations Between Low- vs. High-Load Resistance Training: A Systematic Review and Meta-analysis. J. Strength Cond. Res. 2017, 31, 3508–3523. [Google Scholar] [CrossRef] [PubMed]
- Grgic, J.; Garofolini, A.; Orazem, J.; Sabol, F.; Schoenfeld, B.J.; Pedisic, Z. Effects of Resistance Training on Muscle Size and Strength in Very Elderly Adults: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Sports Med. 2020, 50, 1983–1999. [Google Scholar] [CrossRef] [PubMed]
- Braith, R.W.; Stewart, K.J. Resistance exercise training: Its role in the prevention of cardiovascular disease. Circulation 2006, 113, 2642–2650. [Google Scholar] [CrossRef] [PubMed]
- Baumgartner, R.N.; Koehler, K.M.; Gallagher, D.; Romero, L.; Heymsfield, S.B.; Ross, R.R.; Garry, P.J.; Lindeman, R.D. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef]
- Pollock, M.L.; Franklin, B.A.; Balady, G.J.; Chaitman, B.L.; Fleg, J.L.; Fletcher, B.; Limacher, M.; Piña, I.L.; Stein, R.A.; Williams, M.; et al. AHA Science Advisory. Resistance exercise in individuals with and without cardiovascular disease: Benefits, rationale, safety, and prescription: An advisory from the Committee on Exercise, Rehabilitation, and Prevention, Council on Clinical Cardiology, American Heart Association; Position paper endorsed by the American College of Sports Medicine. Circulation 2000, 101, 828–833. [Google Scholar] [CrossRef]
- Clinical guidelines on the identification, evaluation, and treatment of overweight and obesity in adults—the evidence report. National Institutes of Health. Obes. Res. 1998, 6, 51S–179S. [CrossRef]
- Vaughan, L.; Zurlo, F.; Ravussin, E. Aging and energy expenditure. Am. J. Clin. Nutr. 1991, 53, 821–825. [Google Scholar] [CrossRef] [PubMed]
- American College of Sports Medicine Position Stand. The recommended quantity and quality of exercise for developing and maintaining cardiorespiratory and muscular fitness, and flexibility in healthy adults. Med. Sci. Sports Exerc. 1998, 30, 975–991. [CrossRef]
- Treuth, M.S.; Ryan, A.S.; Pratley, R.E.; Rubin, M.A.; Miller, J.P.; Nicklas, B.J.; Sorkin, J.; Harman, S.M.; Goldberg, A.P.; Hurley, B.F. Effects of strength training on total and regional body composition in older men. J. Appl. Physiol. 1994, 77, 614–620. [Google Scholar] [CrossRef]
- Prabhakaran, B.; Dowling, E.A.; Branch, J.D.; Swain, D.P.; Leutholtz, B.C. Effect of 14 weeks of resistance training on lipid profile and body fat percentage in premenopausal women. Br. J. Sports Med. 1999, 33, 190–195. [Google Scholar] [CrossRef]
- Ross, R.; Rissanen, J.; Pedwell, H.; Clifford, J.; Shragge, P. Influence of diet and exercise on skeletal muscle and visceral adipose tissue in men. J. Appl. Physiol. 1996, 81, 2445–2455. [Google Scholar] [CrossRef]
- Treuth, M.S.; Hunter, G.R.; Kekes-Szabo, T.; Weinsier, R.L.; Goran, M.I.; Berland, L. Reduction in intra-abdominal adipose tissue after strength training in older women. J. Appl. Physiol. 1995, 78, 1425–1431. [Google Scholar] [CrossRef]
- Hunter, G.R.; Bryan, D.R.; Wetzstein, C.J.; Zuckerman, P.A.; Bamman, M.M. Resistance training and intra-abdominal adipose tissue in older men and women. Med. Sci. Sports Exerc. 2002, 34, 1023–1028. [Google Scholar] [CrossRef]
- Rosety, M.A.; Pery, M.T.; Rodriguez-Pareja, M.A.; Diaz, A.; Rosety, J.; Garcia, N.; Brenes-Martin, F.; Rosety-Rodriguez, M.; Toro, R.; Ordonez, F.J.; et al. A Short-Term Circuit Resistance Programme Reduced Epicardial Fat in Obese Aged Women. Nutr. Hosp. 2015, 32, 2193–2197. [Google Scholar] [CrossRef]
- Ross, R.; Rissanen, J. Mobilization of visceral and subcutaneous adipose tissue in response to energy restriction and exercise. Am. J. Clin. Nutr. 1994, 60, 695–703. [Google Scholar] [CrossRef] [PubMed]
- Slentz, C.A.; Bateman, L.A.; Willis, L.H.; Shields, A.T.; Tanner, C.J.; Piner, L.W.; Hawk, V.H.; Muehlbauer, M.J.; Samsa, G.P.; Nelson, R.C.; et al. Effects of aerobic vs. resistance training on visceral and liver fat stores, liver enzymes, and insulin resistance by HOMA in overweight adults from STRRIDE AT/RT. Am. J. Physiol. Endocrinol. Metab. 2011, 301, E1033–E1039. [Google Scholar] [CrossRef] [PubMed]
- Ormsbee, M.J.; Thyfault, J.P.; Johnson, E.A.; Kraus, R.M.; Choi, M.D.; Hickner, R.C. Fat metabolism and acute resistance exercise in trained men. J. Appl. Physiol. 2007, 102, 1767–1772. [Google Scholar] [CrossRef]
- Allman, B.R.; Morrissey, M.C.; Kim, J.S.; Panton, L.B.; Contreras, R.J.; Hickner, R.C.; Ormsbee, M.J. Fat metabolism and acute resistance exercise in trained women. J. Appl. Physiol. 2019, 126, 739–745. [Google Scholar] [CrossRef]
- Chatzinikolaou, A.; Fatouros, I.; Petridou, A.; Jamurtas, A.; Avloniti, A.; Douroudos, I.; Mastorakos, G.; Lazaropoulou, C.; Papassotiriou, I.; Tournis, S.; et al. Adipose tissue lipolysis is upregulated in lean and obese men during acute resistance exercise. Diabetes Care 2008, 31, 1397–1399. [Google Scholar] [CrossRef]
- Bishop, D.J.; Bartlett, J.; Fyfe, J.; Lee, M. Methodological Considerations for Concurrent Training. In Concurrent Aerobic and Strength Training: Scientific Basics and Practical Applications; Schumann, M., Rønnestad, B.R., Eds.; Springer International Publishing: Cham, Swithzerland, 2019; pp. 183–196. [Google Scholar] [CrossRef]
- Wilhelm, E.N.; Pinto, R.S. Concurrent Aerobic and Strength Training for Body Composition and Health. In Concurrent Aerobic and Strength Training: Scientific Basics and Practical Applications; Schumann, M., Rønnestad, B.R., Eds.; Springer International Publishing: Cham, Swithzerland, 2019; pp. 293–307. [Google Scholar] [CrossRef]
- Mikkola, J.; Rusko, H.; Nummela, A.; Pollari, T.; Häkkinen, K. Concurrent endurance and explosive type strength training improves neuromuscular and anaerobic characteristics in young distance runners. Int. J. Sports Med. 2007, 28, 602–611. [Google Scholar] [CrossRef]
- Schumann, M.; Rønnestad, B.R. A Brief Historical Overview on the Science of Concurrent Aerobic and Strength Training. In Concurrent Aerobic and Strength Training: Scientific Basics and Practical Applications; Schumann, M., Rønnestad, B.R., Eds.; Springer International Publishing: Cham, Swithzerland, 2019; pp. 1–6. [Google Scholar] [CrossRef]
- Lee, M.J.; Ballantyne, J.K.; Chagolla, J.; Hopkins, W.G.; Fyfe, J.J.; Phillips, S.M.; Bishop, D.J.; Bartlett, J.D. Order of same-day concurrent training influences some indices of power development, but not strength, lean mass, or aerobic fitness in healthy, moderately-active men after 9 weeks of training. PLoS ONE 2020, 15, e0233134. [Google Scholar] [CrossRef]
- Monteiro, P.A.; Chen, K.Y.; Lira, F.S.; Saraiva, B.T.; Antunes, B.M.; Campos, E.Z.; Freitas, I.F., Jr. Concurrent and aerobic exercise training promote similar benefits in body composition and metabolic profiles in obese adolescents. Lipids Health Dis. 2015, 14, 153. [Google Scholar] [CrossRef]
- Wilson, J.M.; Marin, P.J.; Rhea, M.R.; Wilson, S.M.; Loenneke, J.P.; Anderson, J.C. Concurrent training: A meta-analysis examining interference of aerobic and resistance exercises. J. Strength Cond. Res. 2012, 26, 2293–2307. [Google Scholar] [CrossRef]
- Damaso, A.R.; da Silveira Campos, R.M.; Caranti, D.A.; de Piano, A.; Fisberg, M.; Foschini, D.; de Lima Sanches, P.; Tock, L.; Lederman, H.M.; Tufik, S.; et al. Aerobic plus resistance training was more effective in improving the visceral adiposity, metabolic profile and inflammatory markers than aerobic training in obese adolescents. J. Sports Sci. 2014, 32, 1435–1445. [Google Scholar] [CrossRef]
- de Mello, M.T.; de Piano, A.; Carnier, J.; Sanches Pde, L.; Correa, F.A.; Tock, L.; Ernandes, R.M.; Tufik, S.; Damaso, A.R. Long-term effects of aerobic plus resistance training on the metabolic syndrome and adiponectinemia in obese adolescents. J. Clin. Hypertens. 2011, 13, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.N.; Gyllenhammer, L.E.; Vanni, A.A.; Meija, M.; Tung, A.; Schroeder, E.T.; Spruijt-Metz, D.; Goran, M.I. Startup circuit training program reduces metabolic risk in Latino adolescents. Med. Sci. Sports Exerc. 2011, 43, 2195–2203. [Google Scholar] [CrossRef]
- Norheim, F.; Langleite, T.M.; Hjorth, M.; Holen, T.; Kielland, A.; Stadheim, H.K.; Gulseth, H.L.; Birkeland, K.I.; Jensen, J.; Drevon, C.A. The effects of acute and chronic exercise on PGC-1alpha, irisin and browning of subcutaneous adipose tissue in humans. FEBS J. 2014, 281, 739–749. [Google Scholar] [CrossRef]
- Stinkens, R.; Brouwers, B.; Jocken, J.W.; Blaak, E.E.; Teunissen-Beekman, K.F.; Hesselink, M.K.; van Baak, M.A.; Schrauwen, P.; Goossens, G.H. Exercise training-induced effects on the abdominal subcutaneous adipose tissue phenotype in humans with obesity. J. Appl. Physiol. 2018, 125, 1585–1593. [Google Scholar] [CrossRef]
- Coelho, M.; Oliveira, T.; Fernandes, R. Biochemistry of adipose tissue: An endocrine organ. Arch. Med. Sci. 2013, 9, 191–200. [Google Scholar] [CrossRef]
- Ezquerro, S.; Rodríguez, A.; Portincasa, P.; Frühbeck, G. Effects of Diets on Adipose Tissue. Curr. Med. Chem. 2019, 26, 3593–3612. [Google Scholar] [CrossRef] [PubMed]
- Raynor, H.A.; Champagne, C.M. Position of the Academy of Nutrition and Dietetics: Interventions for the Treatment of Overweight and Obesity in Adults. J. Acad. Nutr. Diet. 2016, 116, 129–147. [Google Scholar] [CrossRef]
- Abete, I.; Astrup, A.; Martínez, J.A.; Thorsdottir, I.; Zulet, M.A. Obesity and the metabolic syndrome: Role of different dietary macronutrient distribution patterns and specific nutritional components on weight loss and maintenance. Nutr. Rev. 2010, 68, 214–231. [Google Scholar] [CrossRef]
- Capel, F.; Viguerie, N.; Vega, N.; Dejean, S.; Arner, P.; Klimcakova, E.; Martinez, J.A.; Saris, W.H.; Holst, C.; Taylor, M.; et al. Contribution of energy restriction and macronutrient composition to changes in adipose tissue gene expression during dietary weight-loss programs in obese women. J. Clin. Endocrinol. Metab. 2008, 93, 4315–4322. [Google Scholar] [CrossRef] [PubMed]
- Clément, K.; Viguerie, N.; Poitou, C.; Carette, C.; Pelloux, V.; Curat, C.A.; Sicard, A.; Rome, S.; Benis, A.; Zucker, J.D.; et al. Weight loss regulates inflammation-related genes in white adipose tissue of obese subjects. FASEB J. 2004, 18, 1657–1669. [Google Scholar] [CrossRef]
- Ludwig, D.S.; Friedman, M.I. Increasing adiposity: Consequence or cause of overeating? JAMA 2014, 311, 2167–2168. [Google Scholar] [CrossRef]
- Taubes, G. The science of obesity: What do we really know about what makes us fat? An essay by Gary Taubes. BMJ 2013, 346. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.S.; Ebbeling, C.B. The carbohydrate-insulin model of obesity: Beyond “calories in, calories out”. JAMA Intern. Med. 2018, 178, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Chen, K.Y.; Guo, J.; Lam, Y.Y.; Leibel, R.L.; Mayer, L.E.; Reitman, M.L.; Rosenbaum, M.; Smith, S.R.; Walsh, B.T.; et al. Energy expenditure and body composition changes after an isocaloric ketogenic diet in overweight and obese men. Am. J. Clin. Nutr. 2016, 104, 324–333. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.C.; Siervo, M. Obesity and energy balance: Is the tail wagging the dog? Eur. J. Clin. Nutr. 2011, 65, 1173–1189. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Guo, J. Obesity Energetics: Body Weight Regulation and the Effects of Diet Composition. Gastroenterology 2017, 152, 1718–1727.e3. [Google Scholar] [CrossRef]
- Banting, W. Letter on Corpulence, Addressed to the Public... with Addenda; Harrison: London, UK, 1869. [Google Scholar]
- Fleming, J.A.; Kris-Etherton, P.M. Macronutrient Content of the Diet: What Do We Know About Energy Balance and Weight Maintenance? Curr. Obes. Rep. 2016, 5, 208–213. [Google Scholar] [CrossRef] [PubMed]
- Foster, G.D.; Wyatt, H.R.; Hill, J.O.; Makris, A.P.; Rosenbaum, D.L.; Brill, C.; Stein, R.I.; Mohammed, B.S.; Miller, B.; Rader, D.J. Weight and metabolic outcomes after 2 years on a low-carbohydrate versus low-fat diet: A randomized trial. Ann. Intern. Med. 2010, 153, 147–157. [Google Scholar] [CrossRef] [PubMed]
- Mansoor, N.; Vinknes, K.J.; Veierød, M.B.; Retterstøl, K. Effects of low-carbohydrate diets v. low-fat diets on body weight and cardiovascular risk factors: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2016, 115, 466–479. [Google Scholar] [CrossRef]
- Ebbeling, C.B.; Swain, J.F.; Feldman, H.A.; Wong, W.W.; Hachey, D.L.; Garcia-Lago, E.; Ludwig, D.S. Effects of dietary composition on energy expenditure during weight-loss maintenance. JAMA 2012, 307, 2627–2634. [Google Scholar] [CrossRef] [PubMed]
- Hall, K.D.; Bemis, T.; Brychta, R.; Chen, K.Y.; Courville, A.; Crayner, E.J.; Goodwin, S.; Guo, J.; Howard, L.; Knuth, N.D. Calorie for calorie, dietary fat restriction results in more body fat loss than carbohydrate restriction in people with obesity. Cell Metab. 2015, 22, 427–436. [Google Scholar] [CrossRef]
- Bueno, N.B.; de Melo, I.S.; de Oliveira, S.L.; da Rocha Ataide, T. Very-low-carbohydrate ketogenic diet v. low-fat diet for long-term weight loss: A meta-analysis of randomised controlled trials. Br. J. Nutr. 2013, 110, 1178–1187. [Google Scholar] [CrossRef]
- Sackner-Bernstein, J.; Kanter, D.; Kaul, S. Dietary Intervention for Overweight and Obese Adults: Comparison of Low-Carbohydrate and Low-Fat Diets. A Meta-Analysis. PLoS ONE 2015, 10, e0139817. [Google Scholar] [CrossRef]
- Tobias, D.K.; Chen, M.; Manson, J.E.; Ludwig, D.S.; Willett, W.; Hu, F.B. Effect of low-fat diet interventions versus other diet interventions on long-term weight change in adults: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2015, 3, 968–979. [Google Scholar] [CrossRef]
- Osuna-Prieto, F.J.; Martinez-Tellez, B.; Sanchez-Delgado, G.; Aguilera, C.M.; Lozano-Sanchez, J.; Arraez-Roman, D.; Segura-Carretero, A.; Ruiz, J.R. Activation of Human Brown Adipose Tissue by Capsinoids, Catechins, Ephedrine, and Other Dietary Components: A Systematic Review. Adv. Nutr. 2019, 10, 291–302. [Google Scholar] [CrossRef] [PubMed]
- Heenan, K.A.; Carrillo, A.E.; Fulton, J.L.; Ryan, E.J.; Edsall, J.R.; Rigopoulos, D.; Markofski, M.M.; Flouris, A.D.; Dinas, P.C. Effects of Nutrition/Diet on Brown Adipose Tissue in Humans: A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 2752. [Google Scholar] [CrossRef]
- Noakes, T.D.; Windt, J. Evidence that supports the prescription of low-carbohydrate high-fat diets: A narrative review. Br. J. Sports Med. 2017, 51, 133. [Google Scholar] [CrossRef] [PubMed]
- Wheless, J.W. History of the ketogenic diet. Epilepsia 2008, 49 (Suppl. 8), 3–5. [Google Scholar] [CrossRef]
- Stubbs, B.J.; Newman, J.C. Ketogenic diet and adipose tissue inflammation-a simple story? Fat chance! Nat. Metab. 2020, 2, 3–4. [Google Scholar] [CrossRef] [PubMed]
- Dyson, P.A.; Beatty, S.; Matthews, D.R. A low-carbohydrate diet is more effective in reducing body weight than healthy eating in both diabetic and non-diabetic subjects. Diabet Med. 2007, 24, 1430–1435. [Google Scholar] [CrossRef] [PubMed]
- Goday, A.; Bellido, D.; Sajoux, I.; Crujeiras, A.B.; Burguera, B.; García-Luna, P.P.; Oleaga, A.; Moreno, B.; Casanueva, F.F. Short-term safety, tolerability and efficacy of a very low-calorie-ketogenic diet interventional weight loss program versus hypocaloric diet in patients with type 2 diabetes mellitus. Nutr. Diabetes 2016, 6, e230. [Google Scholar] [CrossRef] [PubMed]
- Harvey, C.; Schofield, G.M.; Zinn, C.; Thornley, S.J.; Crofts, C.; Merien, F.L.R. Low-carbohydrate diets differing in carbohydrate restriction improve cardiometabolic and anthropometric markers in healthy adults: A randomised clinical trial. PeerJ 2019, 7, e6273. [Google Scholar] [CrossRef]
- Freire, R. Scientific evidence of diets for weight loss: Different macronutrient composition, intermittent fasting, and popular diets. Nutrition 2020, 69, 110549. [Google Scholar] [CrossRef]
- Seidelmann, S.B.; Claggett, B.; Cheng, S.; Henglin, M.; Shah, A.; Steffen, L.M.; Folsom, A.R.; Rimm, E.B.; Willett, W.C.; Solomon, S.D. Dietary carbohydrate intake and mortality: A prospective cohort study and meta-analysis. Lancet Public Health 2018, 3, e419–e428. [Google Scholar] [CrossRef]
- Westerterp-Plantenga, M.S.; Lemmens, S.G.; Westerterp, K.R. Dietary protein—Its role in satiety, energetics, weight loss and health. Br. J. Nutr. 2012, 108 (Suppl. 2), S105–S112. [Google Scholar] [CrossRef]
- Dansinger, M.L.; Gleason, J.A.; Griffith, J.L.; Selker, H.P.; Schaefer, E.J. Comparison of the Atkins, Ornish, Weight Watchers, and Zone diets for weight loss and heart disease risk reduction: A randomized trial. JAMA 2005, 293, 43–53. [Google Scholar] [CrossRef] [PubMed]
- McAuley, K.A.; Hopkins, C.M.; Smith, K.J.; McLay, R.T.; Williams, S.M.; Taylor, R.W.; Mann, J.I. Comparison of high-fat and high-protein diets with a high-carbohydrate diet in insulin-resistant obese women. Diabetologia 2005, 48, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Due, A.; Toubro, S.; Skov, A.R.; Astrup, A. Effect of normal-fat diets, either medium or high in protein, on body weight in overweight subjects: A randomised 1-year trial. Int. J. Obes. Relat. Metab. Disord. 2004, 28, 1283–1290. [Google Scholar] [CrossRef] [PubMed]
- Claessens, M.; van Baak, M.A.; Monsheimer, S.; Saris, W.H. The effect of a low-fat, high-protein or high-carbohydrate ad libitum diet on weight loss maintenance and metabolic risk factors. Int. J. Obes. 2009, 33, 296–304. [Google Scholar] [CrossRef] [PubMed]
- Dalle Grave, R.; Calugi, S.; Gavasso, I.; El Ghoch, M.; Marchesini, G. A randomized trial of energy-restricted high-protein versus high-carbohydrate, low-fat diet in morbid obesity. Obesity 2013, 21, 1774–1781. [Google Scholar] [CrossRef]
- Santesso, N.; Akl, E.A.; Bianchi, M.; Mente, A.; Mustafa, R.; Heels-Ansdell, D.; Schünemann, H.J. Effects of higher- versus lower-protein diets on health outcomes: A systematic review and meta-analysis. Eur. J. Clin. Nutr. 2012, 66, 780–788. [Google Scholar] [CrossRef]
- Gustafson, B. Adipose tissue, inflammation and atherosclerosis. J. Atheroscler. Thromb. 2010, 17, 332–341. [Google Scholar] [CrossRef]
- Fischer, K.; Pick, J.A.; Moewes, D.; Noethlings, U. Qualitative aspects of diet affecting visceral and subcutaneous abdominal adipose tissue: A systematic review of observational and controlled intervention studies. Nutr. Rev. 2015, 73, 191–215. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.; Mandes, T.; Crimarco, A. A plant-based diet for overweight and obesity prevention and treatment. J. Geriatr. Cardiol. 2017, 14, 369–374. [Google Scholar] [CrossRef]
- Barnard, N.D.; Levin, S.M.; Yokoyama, Y. A systematic review and meta-analysis of changes in body weight in clinical trials of vegetarian diets. J. Acad. Nutr. Diet. 2015, 115, 954–969. [Google Scholar] [CrossRef]
- Kanerva, N.; Rissanen, H.; Knekt, P.; Havulinna, A.S.; Eriksson, J.G.; Männistö, S. The healthy Nordic diet and incidence of Type 2 Diabetes--10-year follow-up. Diabetes Res. Clin. Pract. 2014, 106, e34–e37. [Google Scholar] [CrossRef]
- Eichelmann, F.; Schwingshackl, L.; Fedirko, V.; Aleksandrova, K. Effect of plant-based diets on obesity-related inflammatory profiles: A systematic review and meta-analysis of intervention trials. Obes. Rev. 2016, 17, 1067–1079. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.Y.; Huang, C.C.; Hu, F.B.; Chavarro, J.E. Vegetarian Diets and Weight Reduction: A Meta-Analysis of Randomized Controlled Trials. J. Gen. Intern. Med. 2016, 31, 109–116. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Yang, B.; Zheng, J.; Li, G.; Wahlqvist, M.L.; Li, D. Cardiovascular disease mortality and cancer incidence in vegetarians: A meta-analysis and systematic review. Ann. Nutr. Metab. 2012, 60, 233–240. [Google Scholar] [CrossRef]
- Shahavandi, M.; Djafari, F.; Shahinfar, H.; Davarzani, S.; Babaei, N.; Ebaditabar, M.; Djafarian, K.; Clark, C.C.T.; Shab-Bidar, S. The association of plant-based dietary patterns with visceral adiposity, lipid accumulation product, and triglyceride-glucose index in Iranian adults. Complement. Ther. Med. 2020, 53, 102531. [Google Scholar] [CrossRef] [PubMed]
- Tonstad, S.; Butler, T.; Yan, R.; Fraser, G.E. Type of vegetarian diet, body weight, and prevalence of type 2 diabetes. Diabetes Care 2009, 32, 791–796. [Google Scholar] [CrossRef]
- Wright, N.; Wilson, L.; Smith, M.; Duncan, B.; McHugh, P. The BROAD study: A randomised controlled trial using a whole food plant-based diet in the community for obesity, ischaemic heart disease or diabetes. Nutr. Diabetes 2017, 7, e256. [Google Scholar] [CrossRef] [PubMed]
- Westerterp-Plantenga, M.S.; Nieuwenhuizen, A.; Tomé, D.; Soenen, S.; Westerterp, K.R. Dietary protein, weight loss, and weight maintenance. Annu. Rev. Nutr. 2009, 29, 21–41. [Google Scholar] [CrossRef]
- Jayarathne, S.; Koboziev, I.; Park, O.H.; Oldewage-Theron, W.; Shen, C.L.; Moustaid-Moussa, N. Anti-Inflammatory and Anti-Obesity Properties of Food Bioactive Components: Effects on Adipose Tissue. Prev. Nutr. Food Sci. 2017, 22, 251–262. [Google Scholar] [CrossRef]
- Siriwardhana, N.; Kalupahana, N.S.; Cekanova, M.; LeMieux, M.; Greer, B.; Moustaid-Moussa, N. Modulation of adipose tissue inflammation by bioactive food compounds. J. Nutr. Biochem. 2013, 24, 613–623. [Google Scholar] [CrossRef] [PubMed]
- Torres-Fuentes, C.; Schellekens, H.; Dinan, T.G.; Cryan, J.F. A natural solution for obesity: Bioactives for the prevention and treatment of weight gain. A review. Nutr. Neurosci. 2015, 18, 49–65. [Google Scholar] [CrossRef]
- Sobiecki, J.G.; Appleby, P.N.; Bradbury, K.E.; Key, T.J. High compliance with dietary recommendations in a cohort of meat eaters, fish eaters, vegetarians, and vegans: Results from the European Prospective Investigation into Cancer and Nutrition-Oxford study. Nutr. Res. 2016, 36, 464–477. [Google Scholar] [CrossRef]
- Thompson, W.G.; Holdman, N.R.; Janzow, D.J.; Slezak, J.M.; Morris, K.L.; Zemel, M.B. Effect of energy-reduced diets high in dairy products and fiber on weight loss in obese adults. Obes. Res. 2005, 13, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.; Rosner, B.; Willett, W.W.; Sacks, F.M. Cholesterol-lowering effects of dietary fiber: A meta-analysis. Am. J. Clin. Nutr. 1999, 69, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Melina, V.; Craig, W.; Levin, S. Position of the Academy of Nutrition and Dietetics: Vegetarian diets. J. Acad. Nutr. Diet. 2016, 116, 1970–1980. [Google Scholar] [CrossRef]
- Baden, M.Y.; Satija, A.; Hu, F.B.; Huang, T. Change in plant-based diet quality is associated with changes in plasma adiposity-associated biomarker concentrations in women. J. Nutr. 2019, 149, 676–686. [Google Scholar] [CrossRef] [PubMed]
- McCarty, M.F. Promotion of hepatic lipid oxidation and gluconeogenesis as a strategy for appetite control. Med. Hypotheses 1994, 42, 215–225. [Google Scholar] [CrossRef]
- McCarty, M.F. Vegan proteins may reduce risk of cancer, obesity, and cardiovascular disease by promoting increased glucagon activity. Med. Hypotheses 1999, 53, 459–485. [Google Scholar] [CrossRef]
- Chen, Z.; Schoufour, J.D.; Rivadeneira, F.; Lamballais, S.; Ikram, M.A.; Franco, O.H.; Voortman, T. Plant-based Diet and Adiposity Over Time in a Middle-aged and Elderly Population: The Rotterdam Study. Epidemiology 2019, 30, 303–310. [Google Scholar] [CrossRef] [PubMed]
- Ratjen, I.; Morze, J.; Enderle, J.; Both, M.; Borggrefe, J.; Müller, H.-P.; Kassubek, J.; Koch, M.; Lieb, W. Adherence to a plant-based diet in relation to adipose tissue volumes and liver fat content. Am. J. Clin. Nutr. 2020, 112, 354–363. [Google Scholar] [CrossRef]
- Turner-McGrievy, G.M.; Davidson, C.R.; Wingard, E.E.; Wilcox, S.; Frongillo, E.A. Comparative effectiveness of plant-based diets for weight loss: A randomized controlled trial of five different diets. Nutrition 2015, 31, 350–358. [Google Scholar] [CrossRef]
- McEvoy, C.T.; Temple, N.; Woodside, J.V. Vegetarian diets, low-meat diets and health: A review. Public Health Nutr. 2012, 15, 2287–2294. [Google Scholar] [CrossRef]
- Patterson, R.E.; Laughlin, G.A.; LaCroix, A.Z.; Hartman, S.J.; Natarajan, L.; Senger, C.M.; Martínez, M.E.; Villaseñor, A.; Sears, D.D.; Marinac, C.R.; et al. Intermittent Fasting and Human Metabolic Health. J. Acad. Nutr. Diet. 2015, 115, 1203–1212. [Google Scholar] [CrossRef]
- Mattson, M.P.; Moehl, K.; Ghena, N.; Schmaedick, M.; Cheng, A. Intermittent metabolic switching, neuroplasticity and brain health. Nat. Rev. Neurosci. 2018, 19, 63–80. [Google Scholar] [CrossRef] [PubMed]
- Varady, K.A.; Bhutani, S.; Church, E.C.; Klempel, M.C. Short-term modified alternate-day fasting: A novel dietary strategy for weight loss and cardioprotection in obese adults. Am. J. Clin. Nutr. 2009, 90, 1138–1143. [Google Scholar] [CrossRef]
- Barnosky, A.R.; Hoddy, K.K.; Unterman, T.G.; Varady, K.A. Intermittent fasting vs daily calorie restriction for type 2 diabetes prevention: A review of human findings. Transl. Res. 2014, 164, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Varady, K.A. Intermittent versus daily calorie restriction: Which diet regimen is more effective for weight loss? Obes. Rev. 2011, 12, e593–e601. [Google Scholar] [CrossRef]
- Harvie, M.N.; Pegington, M.; Mattson, M.P.; Frystyk, J.; Dillon, B.; Evans, G.; Cuzick, J.; Jebb, S.A.; Martin, B.; Cutler, R.G.; et al. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: A randomized trial in young overweight women. Int. J. Obes. 2011, 35, 714–727. [Google Scholar] [CrossRef] [PubMed]
- Schübel, R.; Nattenmüller, J.; Sookthai, D.; Nonnenmacher, T.; Graf, M.E.; Riedl, L.; Schlett, C.L.; von Stackelberg, O.; Johnson, T.; Nabers, D.; et al. Effects of intermittent and continuous calorie restriction on body weight and metabolism over 50 wk: A randomized controlled trial. Am. J. Clin. Nutr. 2018, 108, 933–945. [Google Scholar] [CrossRef]
- Trepanowski, J.F.; Kroeger, C.M.; Barnosky, A.; Klempel, M.C.; Bhutani, S.; Hoddy, K.K.; Gabel, K.; Freels, S.; Rigdon, J.; Rood, J.; et al. Effect of Alternate-Day Fasting on Weight Loss, Weight Maintenance, and Cardioprotection Among Metabolically Healthy Obese Adults: A Randomized Clinical Trial. JAMA Intern. Med. 2017, 177, 930–938. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M.; Howell, A. Potential Benefits and Harms of Intermittent Energy Restriction and Intermittent Fasting Amongst Obese, Overweight and Normal Weight Subjects-A Narrative Review of Human and Animal Evidence. Behav. Sci. 2017, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Jones, W. (Trans.) Hippocrates; Harvard University Press: Cambridge, MA, USA, 1952. [Google Scholar]
- Holloszy, J.O. Biochemical Adaptations in Muscle: Effects of Exercise on Mitochondrial Oxygen Uptake and Respiratory Enzyme Activity in Skeletal Muscle. J. Biol. Chem. 1967, 242, 2278–2282. [Google Scholar] [CrossRef]
- Bergström, J.; Hermansen, L.; Hultman, E.; Saltin, B. Diet, muscle glycogen and physical performance. Acta Physiol. Scand. 1967, 71, 140–150. [Google Scholar] [CrossRef] [PubMed]
- Hermansen, L.; Hultman, E.; Saltin, B. Muscle glycogen during prolonged severe exercise. Acta Physiol. Scand. 1967, 71, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Booth, F.W.; Roberts, C.K.; Laye, M.J. Lack of exercise is a major cause of chronic diseases. Compr. Physiol. 2012, 2, 1143–1211. [Google Scholar] [CrossRef]
- Barde, Y.A.; Edgar, D.; Thoenen, H. Purification of a new neurotrophic factor from mammalian brain. EMBO J. 1982, 1, 549–553. [Google Scholar] [CrossRef] [PubMed]
- Matthews, V.B.; Astrom, M.B.; Chan, M.H.; Bruce, C.R.; Krabbe, K.S.; Prelovsek, O.; Akerstrom, T.; Yfanti, C.; Broholm, C.; Mortensen, O.H.; et al. Brain-derived neurotrophic factor is produced by skeletal muscle cells in response to contraction and enhances fat oxidation via activation of AMP-activated protein kinase. Diabetologia 2009, 52, 1409–1418. [Google Scholar] [CrossRef]
- Schnyder, S.; Handschin, C. Skeletal muscle as an endocrine organ: PGC-1alpha, myokines and exercise. Bone 2015, 80, 115–125. [Google Scholar] [CrossRef]
- Xu, B.; Goulding, E.H.; Zang, K.; Cepoi, D.; Cone, R.D.; Jones, K.R.; Tecott, L.H.; Reichardt, L.F. Brain-derived neurotrophic factor regulates energy balance downstream of melanocortin-4 receptor. Nat. Neurosci. 2003, 6, 736–742. [Google Scholar] [CrossRef]
- Rios, M.; Fan, G.; Fekete, C.; Kelly, J.; Bates, B.; Kuehn, R.; Lechan, R.M.; Jaenisch, R. Conditional deletion of brain-derived neurotrophic factor in the postnatal brain leads to obesity and hyperactivity. Mol. Endocrinol. 2001, 15, 1748–1757. [Google Scholar] [CrossRef]
- Kernie, S.G.; Liebl, D.J.; Parada, L.F. BDNF regulates eating behavior and locomotor activity in mice. EMBO J. 2000, 19, 1290–1300. [Google Scholar] [CrossRef]
- Lyons, W.E.; Mamounas, L.A.; Ricaurte, G.A.; Coppola, V.; Reid, S.W.; Bora, S.H.; Wihler, C.; Koliatsos, V.E.; Tessarollo, L. Brain-derived neurotrophic factor-deficient mice develop aggressiveness and hyperphagia in conjunction with brain serotonergic abnormalities. Proc. Natl. Acad. Sci. USA 1999, 96, 15239–15244. [Google Scholar] [CrossRef]
- Yang, H.; An, J.J.; Sun, C.; Xu, B. Regulation of Energy Balance via BDNF Expressed in Nonparaventricular Hypothalamic Neurons. Mol. Endocrinol. 2016, 30, 494–503. [Google Scholar] [CrossRef]
- Wang, C.; Bomberg, E.; Billington, C.; Levine, A.; Kotz, C.M. Brain-derived neurotrophic factor in the hypothalamic paraventricular nucleus increases energy expenditure by elevating metabolic rate. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007, 293, R992–R1002. [Google Scholar] [CrossRef] [PubMed]
- Westerterp-Plantenga, M.S.; Verwegen, C.R.; Ijedema, M.J.; Wijckmans, N.E.; Saris, W.H. Acute effects of exercise or sauna on appetite in obese and nonobese men. Physiol. Behav. 1997, 62, 1345–1354. [Google Scholar] [CrossRef]
- Egan, B.; Zierath, J.R. Exercise metabolism and the molecular regulation of skeletal muscle adaptation. Cell Metab. 2013, 17, 162–184. [Google Scholar] [CrossRef] [PubMed]
- Hazell, T.J.; Islam, H.; Townsend, L.K.; Schmale, M.S.; Copeland, J.L. Effects of exercise intensity on plasma concentrations of appetite-regulating hormones: Potential mechanisms. Appetite 2016, 98, 80–88. [Google Scholar] [CrossRef] [PubMed]
- Hawley, J.A.; Hargreaves, M.; Joyner, M.J.; Zierath, J.R. Integrative Biology of Exercise. Cell 2014, 159, 738–749. [Google Scholar] [CrossRef]
- Cheng, A.; Wan, R.; Yang, J.L.; Kamimura, N.; Son, T.G.; Ouyang, X.; Luo, Y.; Okun, E.; Mattson, M.P. Involvement of PGC-1alpha in the formation and maintenance of neuronal dendritic spines. Nat. Commun. 2012, 3, 1250. [Google Scholar] [CrossRef]
- Krabbe, K.S.; Nielsen, A.R.; Krogh-Madsen, R.; Plomgaard, P.; Rasmussen, P.; Erikstrup, C.; Fischer, C.P.; Lindegaard, B.; Petersen, A.M.; Taudorf, S.; et al. Brain-derived neurotrophic factor (BDNF) and type 2 diabetes. Diabetologia 2007, 50, 431–438. [Google Scholar] [CrossRef]
- Han, J.C.; Liu, Q.R.; Jones, M.; Levinn, R.L.; Menzie, C.M.; Jefferson-George, K.S.; Adler-Wailes, D.C.; Sanford, E.L.; Lacbawan, F.L.; Uhl, G.R.; et al. Brain-derived neurotrophic factor and obesity in the WAGR syndrome. N. Engl. J. Med. 2008, 359, 918–927. [Google Scholar] [CrossRef]
- Neeper, S.A.; Gómez-Pinilla, F.; Choi, J.; Cotman, C. Exercise and brain neurotrophins. Nature 1995, 373, 109. [Google Scholar] [CrossRef] [PubMed]
- Vaynman, S.; Ying, Z.; Gomez-Pinilla, F. Hippocampal BDNF mediates the efficacy of exercise on synaptic plasticity and cognition. Eur. J. Neurosci. 2004, 20, 2580–2590. [Google Scholar] [CrossRef]
- Dinoff, A.; Herrmann, N.; Swardfager, W.; Liu, C.S.; Sherman, C.; Chan, S.; Lanctot, K.L. The Effect of Exercise Training on Resting Concentrations of Peripheral Brain-Derived Neurotrophic Factor (BDNF): A Meta-Analysis. PLoS ONE 2016, 11, e0163037. [Google Scholar] [CrossRef]
- Szuhany, K.L.; Bugatti, M.; Otto, M.W. A meta-analytic review of the effects of exercise on brain-derived neurotrophic factor. J. Psychiatr. Res. 2015, 60, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Carling, D.; Mayer, F.V.; Sanders, M.J.; Gamblin, S.J. AMP-activated protein kinase: Nature’s energy sensor. Nat. Chem. Biol. 2011, 7, 512–518. [Google Scholar] [CrossRef]
- Wrann, C.D.; White, J.P.; Salogiannnis, J.; Laznik-Bogoslavski, D.; Wu, J.; Ma, D.; Lin, J.D.; Greenberg, M.E.; Spiegelman, B.M. Exercise induces hippocampal BDNF through a PGC-1alpha/FNDC5 pathway. Cell Metab. 2013, 18, 649–659. [Google Scholar] [CrossRef]
- Fujimura, H.; Altar, C.A.; Chen, R.; Nakamura, T.; Nakahashi, T.; Kambayashi, J.; Sun, B.; Tandon, N.N. Brain-derived neurotrophic factor is stored in human platelets and released by agonist stimulation. Thromb. Haemost. 2002, 87, 728–734. [Google Scholar] [CrossRef]
- Crumpler, H.R.; Dent, C.E.; Harris, H.; Westall, R.G. beta-Aminoisobutyric acid (alpha-methyl-beta-alanine); a new amino-acid obtained from human urine. Nature 1951, 167, 307–308. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.D.; Bostrom, P.; O’Sullivan, J.F.; Schinzel, R.T.; Lewis, G.D.; Dejam, A.; Lee, Y.K.; Palma, M.J.; Calhoun, S.; Georgiadi, A.; et al. beta-Aminoisobutyric acid induces browning of white fat and hepatic beta-oxidation and is inversely correlated with cardiometabolic risk factors. Cell Metab. 2014, 19, 96–108. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Åkerström, T.C.A.; Nielsen, A.R.; Fischer, C.P. Role of myokines in exercise and metabolism. J. Appl. Physiol. 2007, 103, 1093–1098. [Google Scholar] [CrossRef]
- Tanianskii, D.A.; Jarzebska, N.; Birkenfeld, A.L.; O’Sullivan, J.F.; Rodionov, R.N. Beta-Aminoisobutyric Acid as a Novel Regulator of Carbohydrate and Lipid Metabolism. Nutrients 2019, 11, 524. [Google Scholar] [CrossRef]
- Stautemas, J.; Van Kuilenburg, A.B.P.; Stroomer, L.; Vaz, F.; Blancquaert, L.; Lefevere, F.B.D.; Everaert, I.; Derave, W. Acute Aerobic Exercise Leads to Increased Plasma Levels of R- and S-β-Aminoisobutyric Acid in Humans. Front. Physiol. 2019, 10, 1240. [Google Scholar] [CrossRef]
- Fazelzadeh, P.; Hangelbroek, R.W.; Tieland, M.; de Groot, L.C.; Verdijk, L.B.; van Loon, L.J.; Smilde, A.K.; Alves, R.D.; Vervoort, J.; Müller, M.; et al. The Muscle Metabolome Differs between Healthy and Frail Older Adults. J. Proteome Res. 2016, 15, 499–509. [Google Scholar] [CrossRef]
- Begriche, K.; Massart, J.; Abbey-Toby, A.; Igoudjil, A.; Letteron, P.; Fromenty, B. Beta-aminoisobutyric acid prevents diet-induced obesity in mice with partial leptin deficiency. Obesity 2008, 16, 2053–2067. [Google Scholar] [CrossRef]
- Maisonneuve, C.; Igoudjil, A.; Begriche, K.; Lettéron, P.; Guimont, M.C.; Bastin, J.; Laigneau, J.P.; Pessayre, D.; Fromenty, B. Effects of zidovudine, stavudine and beta-aminoisobutyric acid on lipid homeostasis in mice: Possible role in human fat wasting. Antivir. Ther. 2004, 9, 801–810. [Google Scholar] [PubMed]
- Rector, R.S.; Uptergrove, G.M.; Morris, E.M.; Borengasser, S.J.; Laughlin, M.H.; Booth, F.W.; Thyfault, J.P.; Ibdah, J.A. Daily exercise vs. caloric restriction for prevention of nonalcoholic fatty liver disease in the OLETF rat model. Am. J. Physiol. Gastrointest. Liver Physiol. 2011, 300, G874–G883. [Google Scholar] [CrossRef]
- Jung, T.W.; Hwang, H.J.; Hong, H.C.; Yoo, H.J.; Baik, S.H.; Choi, K.M. BAIBA attenuates insulin resistance and inflammation induced by palmitate or a high fat diet via an AMPK-PPARδ-dependent pathway in mice. Diabetologia 2015, 58, 2096–2105. [Google Scholar] [CrossRef] [PubMed]
- Shi, C.X.; Zhao, M.X.; Shu, X.D.; Xiong, X.Q.; Wang, J.J.; Gao, X.Y.; Chen, Q.; Li, Y.H.; Kang, Y.M.; Zhu, G.Q. β-aminoisobutyric acid attenuates hepatic endoplasmic reticulum stress and glucose/lipid metabolic disturbance in mice with type 2 diabetes. Sci Rep. 2016, 6, 21924. [Google Scholar] [CrossRef] [PubMed]
- Kitase, Y.; Vallejo, J.A.; Gutheil, W.; Vemula, H.; Jähn, K.; Yi, J.; Zhou, J.; Brotto, M.; Bonewald, L.F. β-aminoisobutyric Acid, l-BAIBA, Is a Muscle-Derived Osteocyte Survival Factor. Cell Rep. 2018, 22, 1531–1544. [Google Scholar] [CrossRef]
- Note, R.; Maisonneuve, C.; Lettéron, P.; Peytavin, G.; Djouadi, F.; Igoudjil, A.; Guimont, M.C.; Biour, M.; Pessayre, D.; Fromenty, B. Mitochondrial and metabolic effects of nucleoside reverse transcriptase inhibitors (NRTIs) in mice receiving one of five single- and three dual-NRTI treatments. Antimicrob. Agents Chemother. 2003, 47, 3384–3392. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.E.; Miller, R.H.; Block, K.P. Branched-chain amino acid metabolism. Annu. Rev. Nutr. 1984, 4, 409–454. [Google Scholar] [CrossRef] [PubMed]
- Short, K.R.; Chadwick, J.Q.; Teague, A.M.; Tullier, M.A.; Wolbert, L.; Coleman, C.; Copeland, K.C. Effect of Obesity and Exercise Training on Plasma Amino Acids and Amino Metabolites in American Indian Adolescents. J. Clin. Endocrinol. Metab. 2019, 104, 3249–3261. [Google Scholar] [CrossRef] [PubMed]
- van Hall, G.; Steensberg, A.; Sacchetti, M.; Fischer, C.; Keller, C.; Schjerling, P.; Hiscock, N.; Moller, K.; Saltin, B.; Febbraio, M.A.; et al. Interleukin-6 stimulates lipolysis and fat oxidation in humans. J. Clin. Endocrinol. Metab. 2003, 88, 3005–3010. [Google Scholar] [CrossRef]
- Steensberg, A.; van Hall, G.; Osada, T.; Sacchetti, M.; Saltin, B.; Klarlund Pedersen, B. Production of interleukin-6 in contracting human skeletal muscles can account for the exercise-induced increase in plasma interleukin-6. J. Physiol. 2000, 529, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Kern, L.; Mittenbuhler, M.J.; Vesting, A.J.; Ostermann, A.L.; Wunderlich, C.M.; Wunderlich, F.T. Obesity-Induced TNFalpha and IL-6 Signaling: The Missing Link between Obesity and Inflammation-Driven Liver and Colorectal Cancers. Cancers 2018, 11, 24. [Google Scholar] [CrossRef]
- Bastard, J.P.; Jardel, C.; Bruckert, E.; Blondy, P.; Capeau, J.; Laville, M.; Vidal, H.; Hainque, B. Elevated levels of interleukin 6 are reduced in serum and subcutaneous adipose tissue of obese women after weight loss. J. Clin. Endocrinol. Metab. 2000, 85, 3338–3342. [Google Scholar] [CrossRef]
- Fischer, C.P.; Berntsen, A.; Perstrup, L.B.; Eskildsen, P.; Pedersen, B.K. Plasma levels of interleukin-6 and C-reactive protein are associated with physical inactivity independent of obesity. Scand. J. Med. Sci. Sports 2007, 17, 580–587. [Google Scholar] [CrossRef]
- Bruun, J.M.; Verdich, C.; Toubro, S.; Astrup, A.; Richelsen, B. Association between measures of insulin sensitivity and circulating levels of interleukin-8, interleukin-6 and tumor necrosis factor-alpha. Effect of weight loss in obese men. Eur. J. Endocrinol. 2003, 148, 535–542. [Google Scholar] [CrossRef]
- Kado, S.; Nagase, T.; Nagata, N. Circulating levels of interleukin-6, its soluble receptor and interleukin-6/interleukin-6 receptor complexes in patients with type 2 diabetes mellitus. Acta Diabetol. 1999, 36, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Fisman, E.Z.; Benderly, M.; Esper, R.J.; Behar, S.; Boyko, V.; Adler, Y.; Tanne, D.; Matas, Z.; Tenenbaum, A. Interleukin-6 and the risk of future cardiovascular events in patients with angina pectoris and/or healed myocardial infarction. Am. J. Cardiol. 2006, 98, 14–18. [Google Scholar] [CrossRef]
- Cottam, D.R.; Mattar, S.G.; Barinas-Mitchell, E.; Eid, G.; Kuller, L.; Kelley, D.E.; Schauer, P.R. The chronic inflammatory hypothesis for the morbidity associated with morbid obesity: Implications and effects of weight loss. Obes. Surg. 2004, 14, 589–600. [Google Scholar] [CrossRef]
- Wedell-Neergaard, A.S.; Lang Lehrskov, L.; Christensen, R.H.; Legaard, G.E.; Dorph, E.; Larsen, M.K.; Launbo, N.; Fagerlind, S.R.; Seide, S.K.; Nymand, S.; et al. Exercise-Induced Changes in Visceral Adipose Tissue Mass Are Regulated by IL-6 Signaling: A Randomized Controlled Trial. Cell Metab. 2019, 29, 844–855.e3. [Google Scholar] [CrossRef]
- Pedersen, B.K.; Febbraio, M.A. Muscle as an endocrine organ: Focus on muscle-derived interleukin-6. Physiol. Rev. 2008, 88, 1379–1406. [Google Scholar] [CrossRef] [PubMed]
- Ostrowski, K.; Rohde, T.; Asp, S.; Schjerling, P.; Pedersen, B.K. Pro- and anti-inflammatory cytokine balance in strenuous exercise in humans. J. Physiol. 1999, 515, 287–291. [Google Scholar] [CrossRef] [PubMed]
- Bruunsgaard, H.; Galbo, H.; Halkjaer-Kristensen, J.; Johansen, T.L.; MacLean, D.A.; Pedersen, B.K. Exercise-induced increase in serum interleukin-6 in humans is related to muscle damage. J. Physiol. 1997, 499, 833–841. [Google Scholar] [CrossRef]
- Nybo, L.; Nielsen, B.; Pedersen, B.K.; Møller, K.; Secher, N.H. Interleukin-6 release from the human brain during prolonged exercise. J. Physiol. 2002, 542, 991–995. [Google Scholar] [CrossRef]
- Fischer, C.P. Interleukin-6 in acute exercise and training: What is the biological relevance? Exerc. Immunol. Rev. 2006, 12, 6–33. [Google Scholar] [PubMed]
- Starkie, R.L.; Angus, D.J.; Rolland, J.; Hargreaves, M.; Febbraio, M.A. Effect of prolonged, submaximal exercise and carbohydrate ingestion on monocyte intracellular cytokine production in humans. J. Physiol. 2000, 528, 647–655. [Google Scholar] [CrossRef]
- Wallenius, V.; Wallenius, K.; Ahrén, B.; Rudling, M.; Carlsten, H.; Dickson, S.L.; Ohlsson, C.; Jansson, J.O. Interleukin-6-deficient mice develop mature-onset obesity. Nat. Med. 2002, 8, 75–79. [Google Scholar] [CrossRef]
- Carey, A.L.; Steinberg, G.R.; Macaulay, S.L.; Thomas, W.G.; Holmes, A.G.; Ramm, G.; Prelovsek, O.; Hohnen-Behrens, C.; Watt, M.J.; James, D.E.; et al. Interleukin-6 increases insulin-stimulated glucose disposal in humans and glucose uptake and fatty acid oxidation in vitro via AMP-activated protein kinase. Diabetes 2006, 55, 2688–2697. [Google Scholar] [CrossRef]
- Petersen, E.W.; Carey, A.L.; Sacchetti, M.; Steinberg, G.R.; Macaulay, S.L.; Febbraio, M.A.; Pedersen, B.K. Acute IL-6 treatment increases fatty acid turnover in elderly humans in vivo and in tissue culture in vitro. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E155–E162. [Google Scholar] [CrossRef] [PubMed]
- Kahn, B.B.; Alquier, T.; Carling, D.; Hardie, D.G. AMP-activated protein kinase: Ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab. 2005, 1, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Wolsk, E.; Mygind, H.; Grøndahl, T.S.; Pedersen, B.K.; van Hall, G. IL-6 selectively stimulates fat metabolism in human skeletal muscle. Am. J. Physiol. Endocrinol. Metab. 2010, 299, E832–E840. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, L.; Aguer, C. Interleukin-15 as a myokine: Mechanistic insight into its effect on skeletal muscle metabolism. Appl. Physiol. Nutr. Metab. 2019, 44, 229–238. [Google Scholar] [CrossRef]
- Tagaya, Y.; Kurys, G.; Thies, T.A.; Losi, J.M.; Azimi, N.; Hanover, J.A.; Bamford, R.N.; Waldmann, T.A. Generation of secretable and nonsecretable interleukin 15 isoforms through alternate usage of signal peptides. Proc. Natl. Acad. Sci. USA 1997, 94, 14444–14449. [Google Scholar] [CrossRef]
- Azimi, N.; Brown, K.; Bamford, R.N.; Tagaya, Y.; Siebenlist, U.; Waldmann, T.A. Human T cell lymphotropic virus type I Tax protein trans-activates interleukin 15 gene transcription through an NF-kappaB site. Proc. Natl. Acad. Sci. USA 1998, 95, 2452–2457. [Google Scholar] [CrossRef] [PubMed]
- Tamura, Y.; Watanabe, K.; Kantani, T.; Hayashi, J.; Ishida, N.; Kaneki, M. Upregulation of circulating IL-15 by treadmill running in healthy individuals: Is IL-15 an endocrine mediator of the beneficial effects of endurance exercise? Endocr. J. 2011, 58, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, C.; Rosati, M.; Jalah, R.; Valentin, A.; Kulkarni, V.; Alicea, C.; Zhang, G.M.; Patel, V.; Felber, B.K.; Pavlakis, G.N. Intracellular interaction of interleukin-15 with its receptor alpha during production leads to mutual stabilization and increased bioactivity. J. Biol. Chem. 2008, 283, 4189–4199. [Google Scholar] [CrossRef] [PubMed]
- Quinn, L.S.; Anderson, B.G. Interleukin-15, IL-15 Receptor-Alpha, and Obesity: Concordance of Laboratory Animal and Human Genetic Studies. J. Obes. 2011, 2011, 456347. [Google Scholar] [CrossRef] [PubMed]
- Quinn, L.S.; Anderson, B.G.; Strait-Bodey, L.; Stroud, A.M.; Argilés, J.M. Oversecretion of interleukin-15 from skeletal muscle reduces adiposity. Am. J. Physiol. Endocrinol. Metab. 2009, 296, E191–E202. [Google Scholar] [CrossRef]
- Almendro, V.; Busquets, S.; Ametller, E.; Carbo, N.; Figueras, M.; Fuster, G.; Argiles, J.M.; Lopez-Soriano, F.J. Effects of interleukin-15 on lipid oxidation: Disposal of an oral [(14)C]-triolein load. Biochim. Biophys. Acta 2006, 1761, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Nadeau, L.; Patten, D.A.; Caron, A.; Garneau, L.; Pinault-Masson, E.; Foretz, M.; Haddad, P.; Anderson, B.G.; Quinn, L.S.; Jardine, K.; et al. IL-15 improves skeletal muscle oxidative metabolism and glucose uptake in association with increased respiratory chain supercomplex formation and AMPK pathway activation. Biochim. Biophys. Acta Gen. Subj. 2019, 1863, 395–407. [Google Scholar] [CrossRef]
- Ye, J. Beneficial metabolic activities of inflammatory cytokine interleukin 15 in obesity and type 2 diabetes. Front. Med. 2015, 9, 139–145. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, A.R.; Hojman, P.; Erikstrup, C.; Fischer, C.P.; Plomgaard, P.; Mounier, R.; Mortensen, O.H.; Broholm, C.; Taudorf, S.; Krogh-Madsen, R.; et al. Association between Interleukin-15 and Obesity: Interleukin-15 as a Potential Regulator of Fat Mass. J. Clin. Endocrinol. Metab. 2008, 93, 4486–4493. [Google Scholar] [CrossRef]
- Alvarez, B.; Carbó, N.; López-Soriano, J.; Drivdahl, R.H.; Busquets, S.; López-Soriano, F.J.; Argilés, J.M.; Quinn, L.S. Effects of interleukin-15 (IL-15) on adipose tissue mass in rodent obesity models: Evidence for direct IL-15 action on adipose tissue. Biochim. Biophys. Acta 2002, 1570, 33–37. [Google Scholar] [CrossRef]
- Barra, N.G.; Palanivel, R.; Denou, E.; Chew, M.V.; Gillgrass, A.; Walker, T.D.; Kong, J.; Richards, C.D.; Jordana, M.; Collins, S.M.; et al. Interleukin-15 modulates adipose tissue by altering mitochondrial mass and activity. PLoS ONE 2014, 9, e114799. [Google Scholar] [CrossRef] [PubMed]
- Al-Shukaili, A.; Al-Ghafri, S.; Al-Marhoobi, S.; Al-Abri, S.; Al-Lawati, J.; Al-Maskari, M. Analysis of inflammatory mediators in type 2 diabetes patients. Int. J. Endocrinol. 2013, 2013, 976810. [Google Scholar] [CrossRef]
- Pérez-López, A.; Valadés, D.; Vázquez Martínez, C.; de Cos Blanco, A.I.; Bujan, J.; García-Honduvilla, N. Serum IL-15 and IL-15Rα levels are decreased in lean and obese physically active humans. Scand. J. Med. Sci. Sports 2018, 28, 1113–1120. [Google Scholar] [CrossRef]
- Riechman, S.E.; Balasekaran, G.; Roth, S.M.; Ferrell, R.E. Association of interleukin-15 protein and interleukin-15 receptor genetic variation with resistance exercise training responses. J. Appl. Physiol. 2004, 97, 2214–2219. [Google Scholar] [CrossRef]
- Nielsen, A.R.; Mounier, R.; Plomgaard, P.; Mortensen, O.H.; Penkowa, M.; Speerschneider, T.; Pilegaard, H.; Pedersen, B.K. Expression of interleukin-15 in human skeletal muscle effect of exercise and muscle fibre type composition. J. Physiol. 2007, 584, 305–312. [Google Scholar] [CrossRef] [PubMed]
- Crane, J.D.; MacNeil, L.G.; Lally, J.S.; Ford, R.J.; Bujak, A.L.; Brar, I.K.; Kemp, B.E.; Raha, S.; Steinberg, G.R.; Tarnopolsky, M.A. Exercise-stimulated interleukin-15 is controlled by AMPK and regulates skin metabolism and aging. Aging Cell 2015, 14, 625–634. [Google Scholar] [CrossRef] [PubMed]
- Rinnov, A.; Yfanti, C.; Nielsen, S.; Akerstrom, T.C.; Peijs, L.; Zankari, A.; Fischer, C.P.; Pedersen, B.K. Endurance training enhances skeletal muscle interleukin-15 in human male subjects. Endocrine 2014, 45, 271–278. [Google Scholar] [CrossRef]
- Ostrowski, K.; Hermann, C.; Bangash, A.; Schjerling, P.; Nielsen, J.N.; Pedersen, B.K. A trauma-like elevation of plasma cytokines in humans in response to treadmill running. J. Physiol. 1998, 513, 889–894. [Google Scholar] [CrossRef] [PubMed]
- Stoklasek, T.A.; Schluns, K.S.; Lefrançois, L. Combined IL-15/IL-15Ralpha immunotherapy maximizes IL-15 activity in vivo. J. Immunol. 2006, 177, 6072–6080. [Google Scholar] [CrossRef]
- Tsiloulis, T.; Watt, M.J. Exercise and the Regulation of Adipose Tissue Metabolism. Prog. Mol. Biol. Transl. Sci. 2015, 135, 175–201. [Google Scholar] [CrossRef]
- Gorgens, S.W.; Eckardt, K.; Jensen, J.; Drevon, C.A.; Eckel, J. Exercise and Regulation of Adipokine and Myokine Production. Prog. Mol. Biol. Transl. Sci. 2015, 135, 313–336. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.; Elbelt, U.; Stengel, A. Irisin as a muscle-derived hormone stimulating thermogenesis—A critical update. Peptides 2014, 54, 89–100. [Google Scholar] [CrossRef]
- Huh, J.Y.; Panagiotou, G.; Mougios, V.; Brinkoetter, M.; Vamvini, M.T.; Schneider, B.E.; Mantzoros, C.S. FNDC5 and irisin in humans: I. Predictors of circulating concentrations in serum and plasma and II. mRNA expression and circulating concentrations in response to weight loss and exercise. Metab. Clin. Exp. 2012, 61, 1725–1738. [Google Scholar] [CrossRef]
- Fox, J.; Rioux, B.V.; Goulet, E.D.B.; Johanssen, N.M.; Swift, D.L.; Bouchard, D.R.; Loewen, H.; Sénéchal, M. Effect of an acute exercise bout on immediate post-exercise irisin concentration in adults: A meta-analysis. Scand. J. Med. Sci. Sports 2018, 28, 16–28. [Google Scholar] [CrossRef]
- Qiu, S.; Cai, X.; Sun, Z.; Schumann, U.; Zügel, M.; Steinacker, J.M. Chronic Exercise Training and Circulating Irisin in Adults: A Meta-Analysis. Sports Med. 2015, 45, 1577–1588. [Google Scholar] [CrossRef] [PubMed]
- Boström, P.; Wu, J.; Jedrychowski, M.P.; Korde, A.; Ye, L.; Lo, J.C.; Rasbach, K.A.; Boström, E.A.; Choi, J.H.; Long, J.Z.; et al. A PGC1-α-dependent myokine that drives brown-fat-like development of white fat and thermogenesis. Nature 2012, 481, 463–468. [Google Scholar] [CrossRef] [PubMed]
- Lee, P.; Linderman, J.D.; Smith, S.; Brychta, R.J.; Wang, J.; Idelson, C.; Perron, R.M.; Werner, C.D.; Phan, G.Q.; Kammula, U.S.; et al. Irisin and FGF21 are cold-induced endocrine activators of brown fat function in humans. Cell Metab. 2014, 19, 302–309. [Google Scholar] [CrossRef] [PubMed]
- Vaughan, R.A.; Gannon, N.P.; Barberena, M.A.; Garcia-Smith, R.; Bisoffi, M.; Mermier, C.M.; Conn, C.A.; Trujillo, K.A. Characterization of the metabolic effects of irisin on skeletal muscle in vitro. Diabetes Obes. Metab. 2014, 16, 711–718. [Google Scholar] [CrossRef]
- Bi, J.; Zhang, J.; Ren, Y.; Du, Z.; Li, Q.; Wang, Y.; Wei, S.; Yang, L.; Zhang, J.; Liu, C.; et al. Irisin alleviates liver ischemia-reperfusion injury by inhibiting excessive mitochondrial fission, promoting mitochondrial biogenesis and decreasing oxidative stress. Redox Biol. 2019, 20, 296–306. [Google Scholar] [CrossRef]
- Moreno-Navarrete, J.M.; Ortega, F.; Serrano, M.; Guerra, E.; Pardo, G.; Tinahones, F.; Ricart, W.; Fernandez-Real, J.M. Irisin is expressed and produced by human muscle and adipose tissue in association with obesity and insulin resistance. J. Clin. Endocrinol. Metab. 2013, 98, E769–E778. [Google Scholar] [CrossRef] [PubMed]
- Puigserver, P.; Wu, Z.; Park, C.W.; Graves, R.; Wright, M.; Spiegelman, B.M. A cold-inducible coactivator of nuclear receptors linked to adaptive thermogenesis. Cell 1998, 92, 829–839. [Google Scholar] [CrossRef]
- Roca-Rivada, A.; Castelao, C.; Senin, L.L.; Landrove, M.O.; Baltar, J.; Belén Crujeiras, A.; Seoane, L.M.; Casanueva, F.F.; Pardo, M. FNDC5/irisin is not only a myokine but also an adipokine. PLoS ONE 2013, 8, e60563. [Google Scholar] [CrossRef]
- Perakakis, N.; Triantafyllou, G.A.; Fernandez-Real, J.M.; Huh, J.Y.; Park, K.H.; Seufert, J.; Mantzoros, C.S. Physiology and role of irisin in glucose homeostasis. Nat. Rev. Endocrinol. 2017, 13, 324–337. [Google Scholar] [CrossRef]
- Polyzos, S.A.; Mathew, H.; Mantzoros, C.S. Irisin: A true, circulating hormone. Metab. Clin. Exp. 2015, 64, 1611–1618. [Google Scholar] [CrossRef]
- Polyzos, S.A.; Anastasilakis, A.D.; Efstathiadou, Z.A.; Makras, P.; Perakakis, N.; Kountouras, J.; Mantzoros, C.S. Irisin in metabolic diseases. Endocrine 2018, 59, 260–274. [Google Scholar] [CrossRef]
- Huh, J.Y.; Dincer, F.; Mesfum, E.; Mantzoros, C.S. Irisin stimulates muscle growth-related genes and regulates adipocyte differentiation and metabolism in humans. Int. J. Obes. 2014, 38, 1538–1544. [Google Scholar] [CrossRef] [PubMed]
- Aydin, S.; Aydin, S.; Kuloglu, T.; Yilmaz, M.; Kalayci, M.; Sahin, I.; Cicek, D. Alterations of irisin concentrations in saliva and serum of obese and normal-weight subjects, before and after 45 min of a Turkish bath or running. Peptides 2013, 50, 13–18. [Google Scholar] [CrossRef]
- Choi, Y.-K.; Kim, M.-K.; Bae, K.H.; Seo, H.-A.; Jeong, J.-Y.; Lee, W.-K.; Kim, J.-G.; Lee, I.-K.; Park, K.-G. Serum irisin levels in new-onset type 2 diabetes. Diabetes Res. Clin. Pract. 2013, 100, 96–101. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Li, R.; Meng, Y.; Li, S.; Donelan, W.; Zhao, Y.; Qi, L.; Zhang, M.; Wang, X.; Cui, T.; et al. Irisin stimulates browning of white adipocytes through mitogen-activated protein kinase p38 MAP kinase and ERK MAP kinase signaling. Diabetes 2014, 63, 514–525. [Google Scholar] [CrossRef]
- Xiong, X.Q.; Chen, D.; Sun, H.J.; Ding, L.; Wang, J.J.; Chen, Q.; Li, Y.H.; Zhou, Y.B.; Han, Y.; Zhang, F.; et al. FNDC5 overexpression and irisin ameliorate glucose/lipid metabolic derangements and enhance lipolysis in obesity. Biochim. Biophys. Acta 2015, 1852, 1867–1875. [Google Scholar] [CrossRef]
- Park, K.H.; Zaichenko, L.; Peter, P.; Davis, C.R.; Crowell, J.A.; Mantzoros, C.S. Diet quality is associated with circulating C-reactive protein but not irisin levels in humans. Metab. Clin. Exp. 2014, 63, 233–241. [Google Scholar] [CrossRef]
- Crujeiras, A.B.; Zulet, M.A.; Lopez-Legarrea, P.; de la Iglesia, R.; Pardo, M.; Carreira, M.C.; Martínez, J.A.; Casanueva, F.F. Association between circulating irisin levels and the promotion of insulin resistance during the weight maintenance period after a dietary weight-lowering program in obese patients. Metab. Clin. Exp. 2014, 63, 520–531. [Google Scholar] [CrossRef] [PubMed]
- Castillo-Quan, J.I. From white to brown fat through the PGC-1α-dependent myokine irisin: Implications for diabetes and obesity. Dis. Models Mech. 2012, 5, 293–295. [Google Scholar] [CrossRef]
- Murawska-Cialowicz, E.; Wojna, J.; Zuwala-Jagiello, J. Crossfit training changes brain-derived neurotrophic factor and irisin levels at rest, after wingate and progressive tests, and improves aerobic capacity and body composition of young physically active men and women. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2015, 66, 811–821. [Google Scholar]
- Safarimosavi, S.; Mohebbi, H.; Rohani, H. High-Intensity Interval vs. Continuous Endurance Training: Preventive Effects on Hormonal Changes and Physiological Adaptations in Prediabetes Patients. J. Strength Cond. Res. 2018. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, Y.; Ijichi, T.; Goto, K. Effect of sprint training on resting serum irisin concentration—Sprint training once daily vs. twice every other day. Metab. Clin. Exp. 2016, 65, 492–495. [Google Scholar] [CrossRef]
- Dundar, A.; Kocahan, S.; Sahin, L. Associations of apelin, leptin, irisin, ghrelin, insulin, glucose levels, and lipid parameters with physical activity during eight weeks of regular exercise training. Arch. Physiol. Biochem. 2019, 1–5. [Google Scholar] [CrossRef]
- Loffler, D.; Muller, U.; Scheuermann, K.; Friebe, D.; Gesing, J.; Bielitz, J.; Erbs, S.; Landgraf, K.; Wagner, I.V.; Kiess, W.; et al. Serum Irisin Levels Are Regulated by Acute Strenuous Exercise. J. Clin. Endocrinol. Metab. 2015, 100, 1289–1299. [Google Scholar] [CrossRef] [PubMed]
- Hunag, J.H.; Wang, S.; Xu, F.P.; Wang, D.; Yin, H.G.; Lai, Q.H.; Liao, J.W.; Hou, X.H.; Hu, M. Exercise training with dietary restriction enhances circulating irisin level associated with increasing endothelial progenitor cell number in obese adults: An intervention study. PeerJ 2017, 5, 18. [Google Scholar] [CrossRef]
- Bluher, S.; Panagiotou, G.; Petroff, D.; Markert, J.; Wagner, A.; Klemm, T.; Filippaios, A.; Keller, A.; Mantzoros, C.S. Effects of a 1-year exercise and lifestyle intervention on irisin, adipokines, and inflammatory markers in obese children. Obesity 2014, 22, 1701–1708. [Google Scholar] [CrossRef] [PubMed]
- Briken, S.; Rosenkranz, S.C.; Keminer, O.; Patra, S.; Ketels, G.; Heesen, C.; Hellweg, R.; Pless, O.; Schulz, K.H.; Gold, S.M. Effects of exercise on Irisin, BDNF and IL-6 serum levels in patients with progressive multiple sclerosis. J. Neuroimmunol. 2016, 299, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Lee, H.J.; So, B.; Son, J.S.; Yoon, D.; Song, W. Effect of aerobic training and resistance training on circulating irisin level and their association with change of body composition in overweight/obese adults: A pilot study. Physiol. Res. 2016, 65, 271–279. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Ando, D.; Takamatsu, K.; Goto, K. Resistance exercise induces a greater irisin response than endurance exercise. Metab. Clin. Exp. 2015, 64, 1042–1050. [Google Scholar] [CrossRef]
- Tsuchiya, Y.; Ando, D.; Goto, K.; Kiuchi, M.; Yamakita, M.; Koyama, K. High-intensity exercise causes greater irisin response compared with low-intensity exercise under similar energy consumption. Tohoku J. Exp. Med. 2014, 233, 135–140. [Google Scholar] [CrossRef]
- Rao, R.R.; Long, J.Z.; White, J.P.; Svensson, K.J.; Lou, J.; Lokurkar, I.; Jedrychowski, M.P.; Ruas, J.L.; Wrann, C.D.; Lo, J.C.; et al. Meteorin-like is a hormone that regulates immune-adipose interactions to increase beige fat thermogenesis. Cell 2014, 157, 1279–1291. [Google Scholar] [CrossRef]
- Zheng, S.L.; Li, Z.Y.; Song, J.; Liu, J.M.; Miao, C.Y. Metrnl: A secreted protein with new emerging functions. Acta Pharmacol. Sin. 2016, 37, 571–579. [Google Scholar] [CrossRef]
- Li, Z.-Y.; Song, J.; Zheng, S.-L.; Fan, M.-B.; Guan, Y.-F.; Qu, Y.; Xu, J.; Wang, P.; Miao, C.-Y. Adipocyte Metrnl Antagonizes Insulin Resistance Through PPARγ Signaling. Diabetes 2015, 64, 4011–4022. [Google Scholar] [CrossRef]
- Jørgensen, J.R.; Fransson, A.; Fjord-Larsen, L.; Thompson, L.H.; Houchins, J.P.; Andrade, N.; Torp, M.; Kalkkinen, N.; Andersson, E.; Lindvall, O.; et al. Cometin is a novel neurotrophic factor that promotes neurite outgrowth and neuroblast migration in vitro and supports survival of spiral ganglion neurons in vivo. Exp. Neurol. 2012, 233, 172–181. [Google Scholar] [CrossRef] [PubMed]
- Onalan, E.; Cavli, C.; Dogan, Y.; Onalan, E.; Gozel, N.; Buran, I.; Yakar, B.; Donder, E. Low serum levels of meteorin-like/subfatin: An indicator of diabetes mellitus and insulin resistance? Endokrynol. Pol. 2020, 71, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Sobieh, B.H.; Kassem, D.H.; Zakaria, Z.M.; El-Mesallamy, H.O. Potential emerging roles of the novel adipokines adipolin/CTRP12 and meteorin-like/METRNL in obesity-osteoarthritis interplay. Cytokine 2021, 138, 155368. [Google Scholar] [CrossRef]
- AlKhairi, I.; Cherian, P.; Abu-Farha, M.; Madhoun, A.A.; Nizam, R.; Melhem, M.; Jamal, M.; Al-Sabah, S.; Ali, H.; Tuomilehto, J.; et al. Increased Expression of Meteorin-Like Hormone in Type 2 Diabetes and Obesity and Its Association with Irisin. Cells 2019, 8, 1283. [Google Scholar] [CrossRef]
- Wang, K.; Li, F.; Wang, C.; Deng, Y.; Cao, Z.; Cui, Y.; Xu, K.; Ln, P.; Sun, Y. Serum Levels of Meteorin-Like (Metrnl) Are Increased in Patients with Newly Diagnosed Type 2 Diabetes Mellitus and Are Associated with Insulin Resistance. Med. Sci. Monit. 2019, 25, 2337–2343. [Google Scholar] [CrossRef] [PubMed]
- Bae, J.Y. Aerobic Exercise Increases Meteorin-Like Protein in Muscle and Adipose Tissue of Chronic High-Fat Diet-Induced Obese Mice. Biomed. Res. Int. 2018, 2018, 6283932. [Google Scholar] [CrossRef] [PubMed]
- Amano, Y.; Nonaka, Y.; Takeda, R.; Kano, Y.; Hoshino, D. Effects of electrical stimulation-induced resistance exercise training on white and brown adipose tissues and plasma meteorin-like concentration in rats. Physiol. Rep. 2020, 8, e14540. [Google Scholar] [CrossRef]
- Eaton, M.; Granata, C.; Barry, J.; Safdar, A.; Bishop, D.; Little, J.P. Impact of a single bout of high-intensity interval exercise and short-term interval training on interleukin-6, FNDC5, and METRNL mRNA expression in human skeletal muscle. J. Sport Health Sci. 2018, 7, 191–196. [Google Scholar] [CrossRef] [PubMed]
 ↑ small change or no change. ↑↑ modest change. ↑↑↑ large change. FM fat mass. SKM skeletal muscle.
↑ small change or no change. ↑↑ modest change. ↑↑↑ large change. FM fat mass. SKM skeletal muscle.
 ↑ small change or no change. ↑↑ modest change. ↑↑↑ large change. FM fat mass. SKM skeletal muscle.
↑ small change or no change. ↑↑ modest change. ↑↑↑ large change. FM fat mass. SKM skeletal muscle.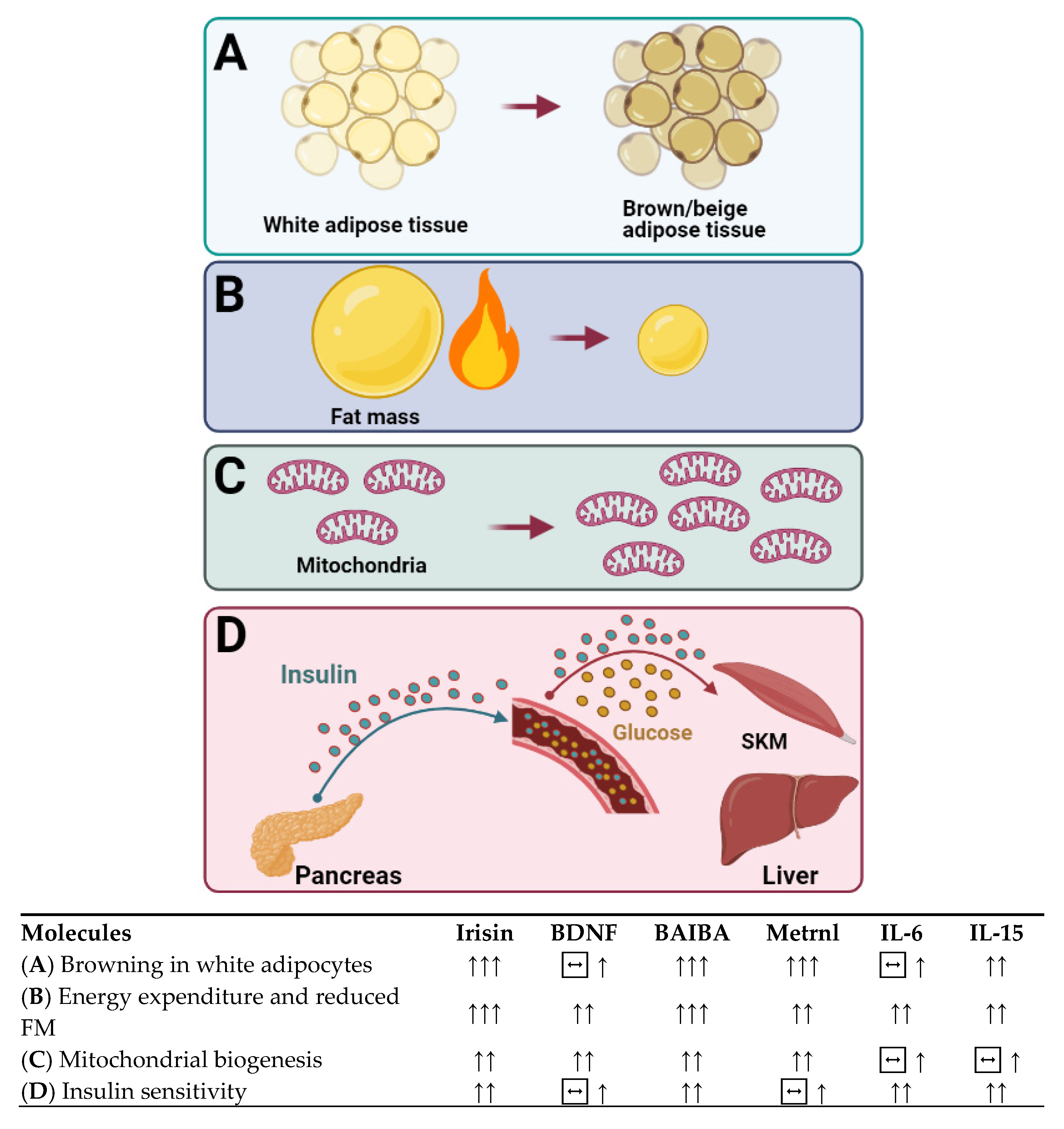
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atakan, M.M.; Koşar, Ş.N.; Güzel, Y.; Tin, H.T.; Yan, X. The Role of Exercise, Diet, and Cytokines in Preventing Obesity and Improving Adipose Tissue. Nutrients 2021, 13, 1459. https://doi.org/10.3390/nu13051459
Atakan MM, Koşar ŞN, Güzel Y, Tin HT, Yan X. The Role of Exercise, Diet, and Cytokines in Preventing Obesity and Improving Adipose Tissue. Nutrients. 2021; 13(5):1459. https://doi.org/10.3390/nu13051459
Chicago/Turabian StyleAtakan, Muhammed Mustafa, Şükran Nazan Koşar, Yasemin Güzel, Hiu Tung Tin, and Xu Yan. 2021. "The Role of Exercise, Diet, and Cytokines in Preventing Obesity and Improving Adipose Tissue" Nutrients 13, no. 5: 1459. https://doi.org/10.3390/nu13051459
APA StyleAtakan, M. M., Koşar, Ş. N., Güzel, Y., Tin, H. T., & Yan, X. (2021). The Role of Exercise, Diet, and Cytokines in Preventing Obesity and Improving Adipose Tissue. Nutrients, 13(5), 1459. https://doi.org/10.3390/nu13051459







