The Effects of Pre-Game Carbohydrate Intake on Running Performance and Substrate Utilisation during Simulated Gaelic Football Match Play
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Experimental Design
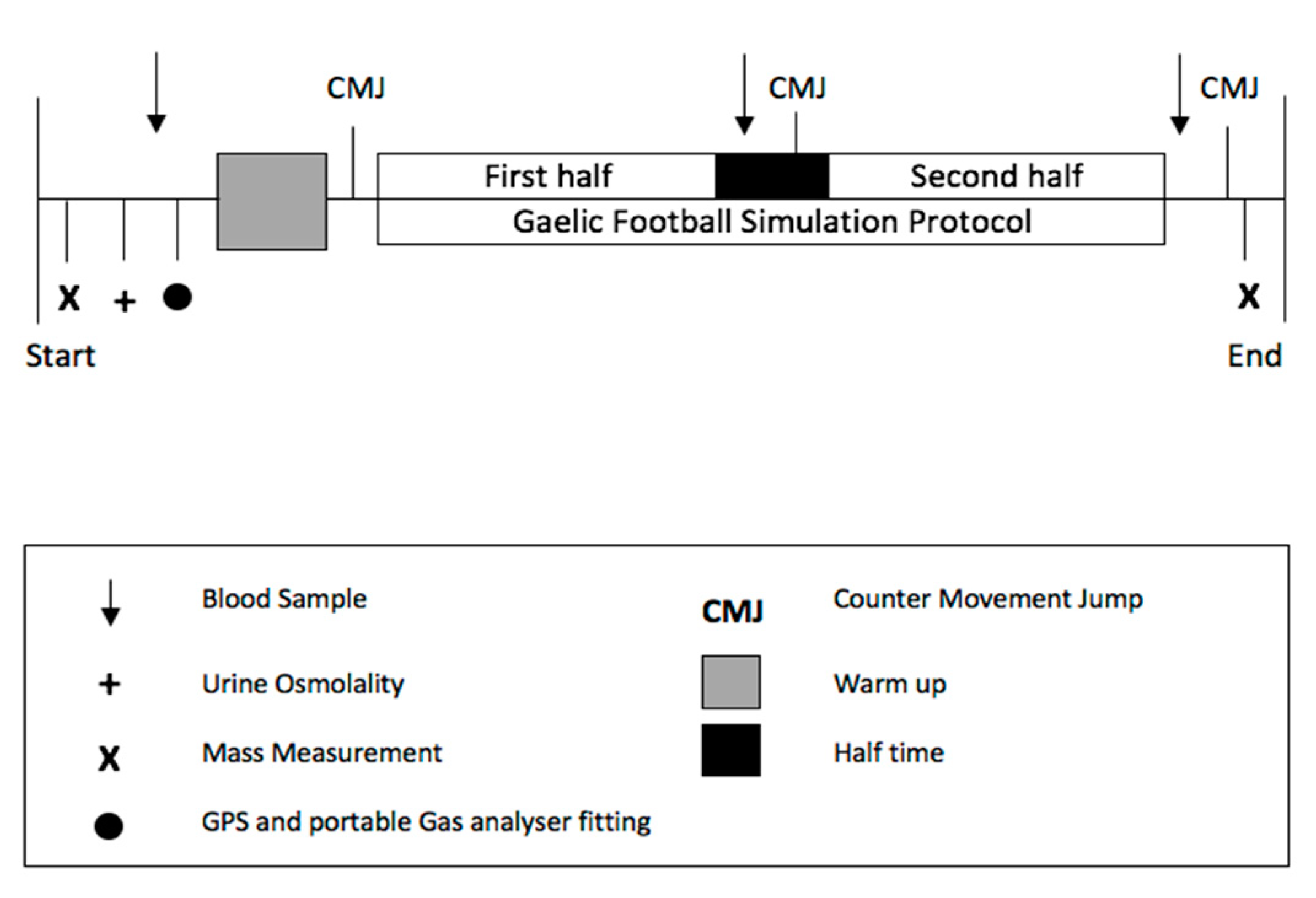
2.3. Gaelic Football Simulation Protocol
2.4. Preliminary Measurements and Familiarisation
2.5. Glycogen-Depleting Protocol
2.6. Main Trial
2.7. Movement and Physiological Measurements
2.7.1. GPS/Heart Rate
2.7.2. Substrate Utilisation
2.7.3. Repeated-Sprint Ability
2.7.4. Countermovement Jump
2.7.5. Blood Collection
2.7.6. Blood Analysis
2.8. Statistical Analysis
3. Results
3.1. Running Performances
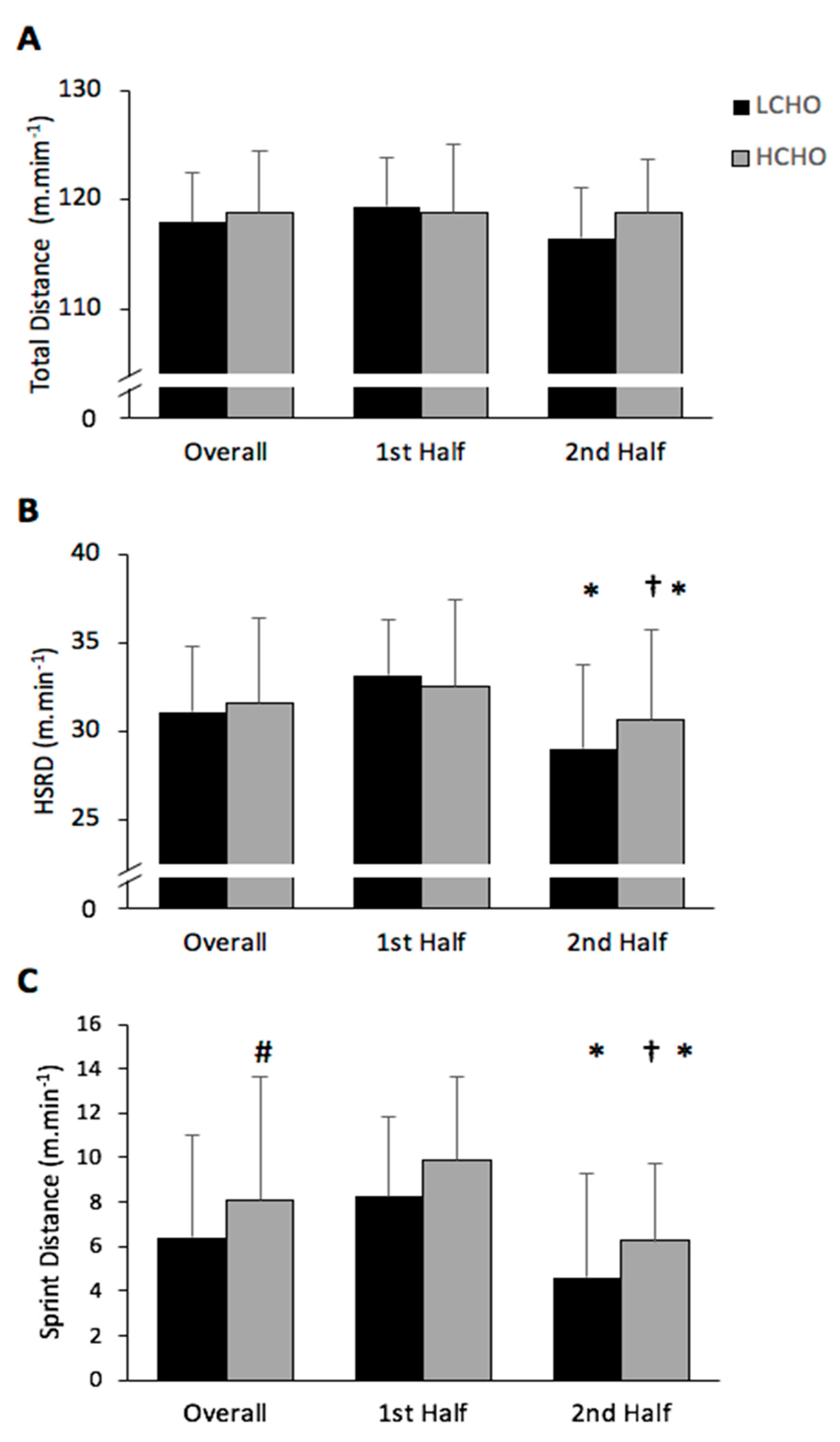
3.1.1. Total Distance Covered
3.1.2. High-Speed Distance Covered
3.1.3. Sprint Distance Covered
3.1.4. Peak Velocity
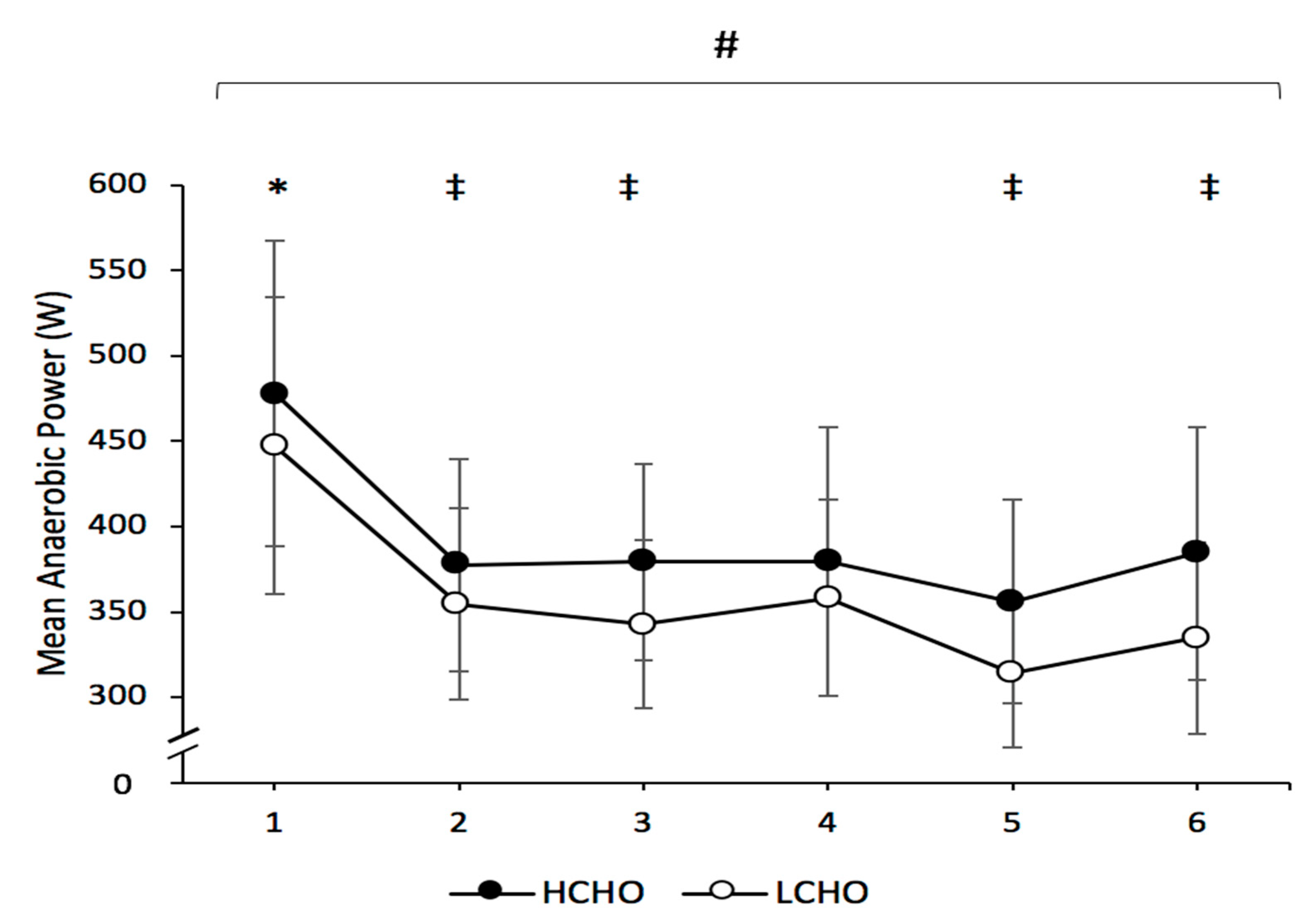
3.2. Repeated-Sprint Ability
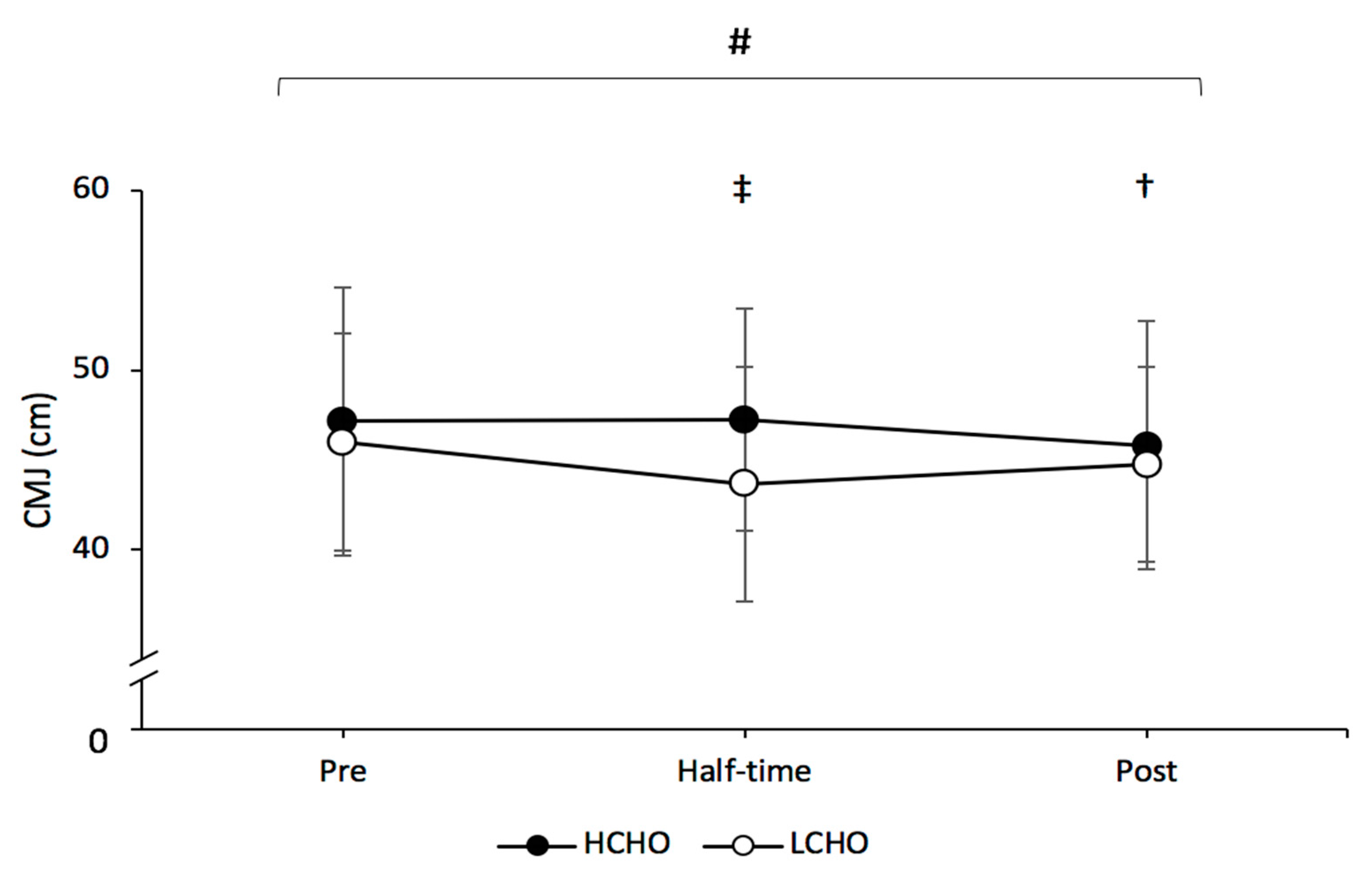
3.3. Countermovement Jump
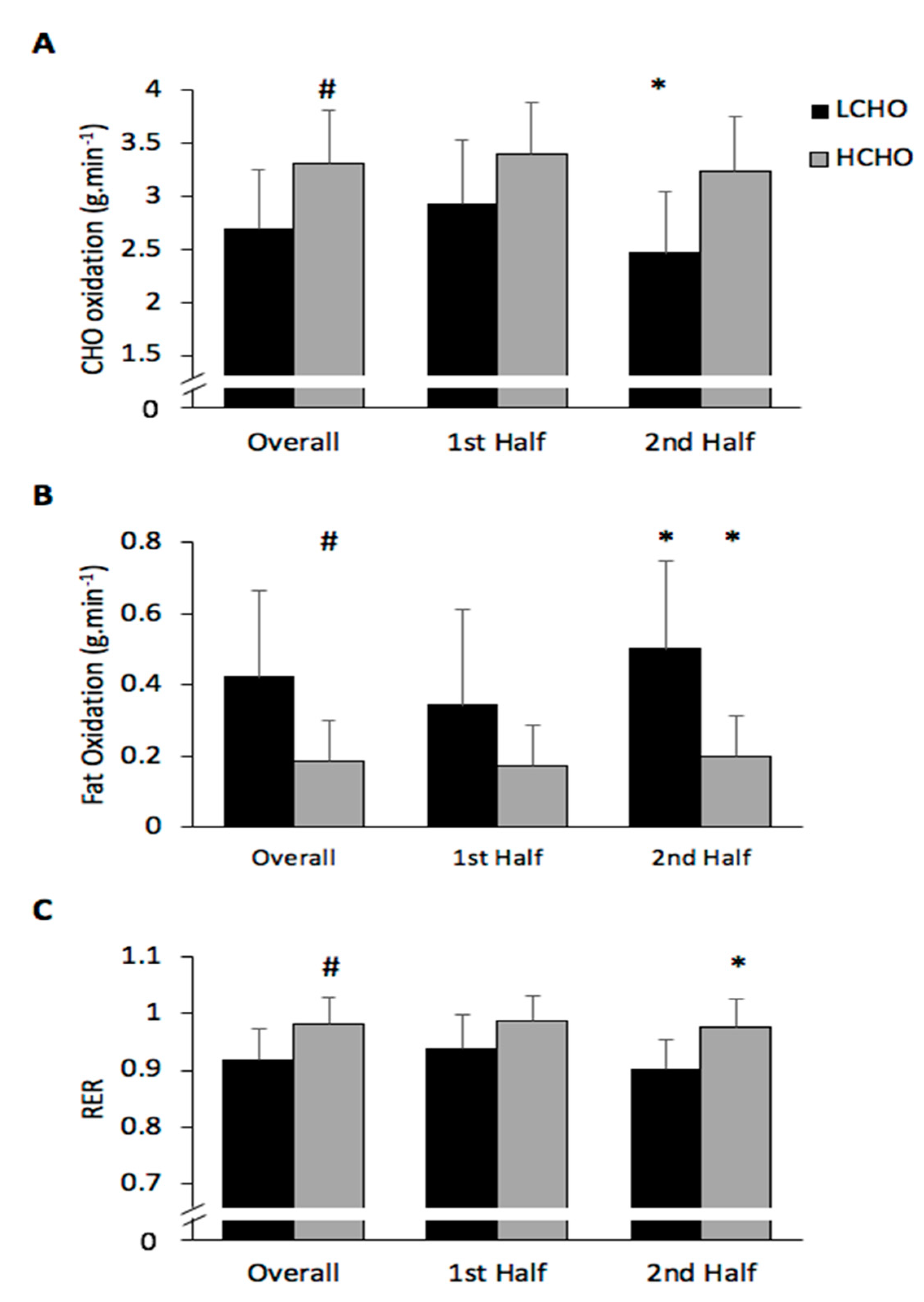
3.4. Substrate Utilisation
3.4.1. CHO Oxidation
3.4.2. Fat Oxidation
3.4.3. RER
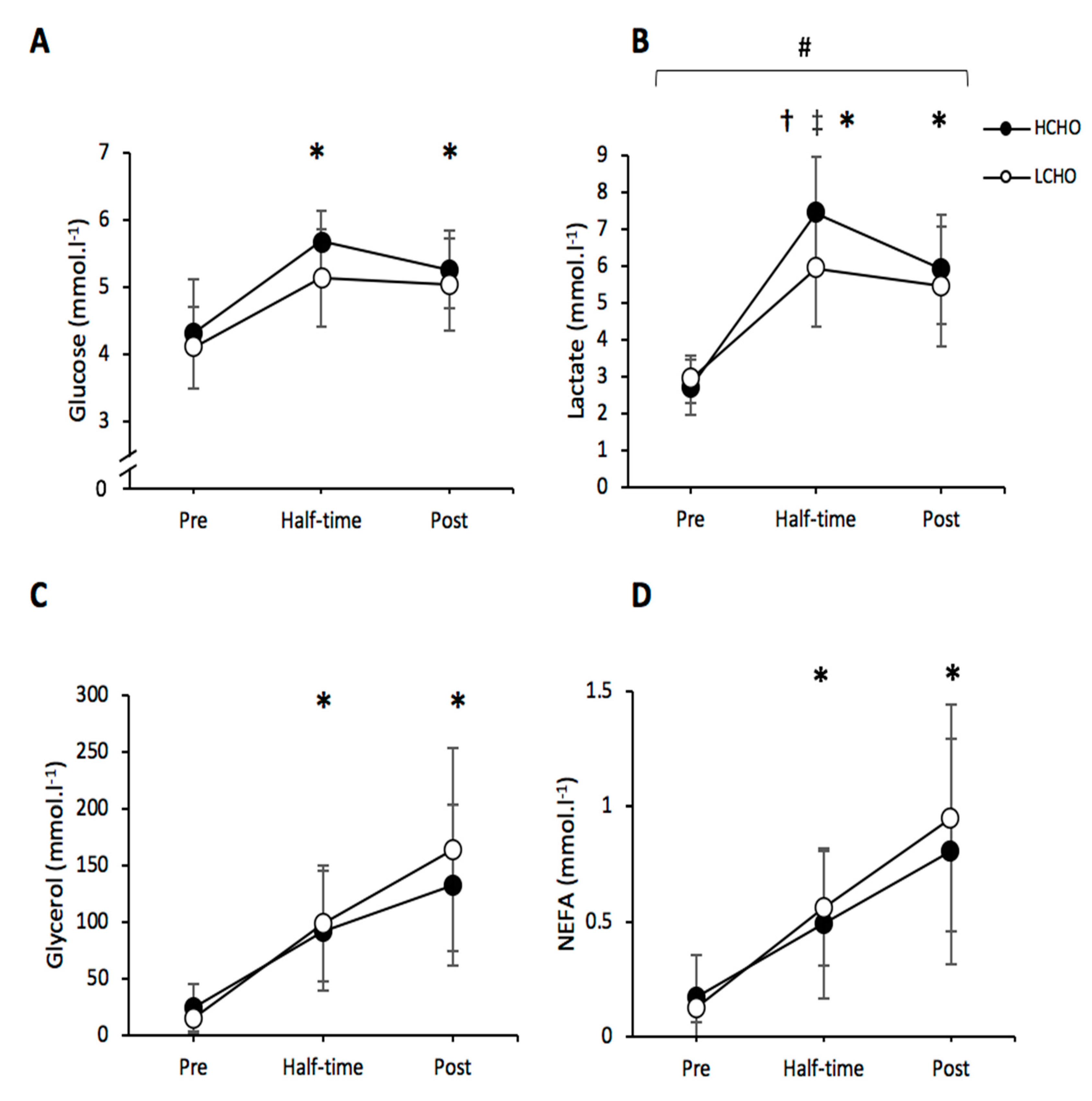
3.5. Blood Metabolites
3.5.1. Plasma Glucose
3.5.2. Lactate
3.5.3. Glycerol
3.5.4. NEFAs
3.6. Heart Rate
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malone, S.; Solan, B.; Collins, K. The running performance profile of elite Gaelic football match-play. J. Strength Cond. Res. 2017, 31, 30–36. [Google Scholar] [CrossRef]
- Malone, S.; Solan, B.; Collins, K.; Doran, D. The metabolic power and energetic demands of elite Gaelic football match play. J. Sports Med. Phys. Fit. 2016, 57, 543–549. [Google Scholar]
- Malone, S.; Solan, B.; Collins, K.D.; Doran, D.A. Positional match running performance in elite Gaelic football. J. Strength Cond. Res. 2016, 30, 2292–2298. [Google Scholar] [CrossRef]
- Malone, S.; Solan, B.; Hughes, B.; Collins, K. Duration specific running performance in elite Gaelic football. J. Strength Cond. Res. 2017. [Google Scholar] [CrossRef]
- Gamble, D.; Spencer, M.; McCarren, A.; Moyna, N. Activity profile, PlayerLoad™ and heart rate response of Gaelic football players: A pilot study. J. Hum. Sport Exerc. 2019, 14, 14. [Google Scholar] [CrossRef]
- Mohr, M.; Krustrup, P.; Bangsbo, J. Match performance of high-standard soccer players with special reference to development of fatigue. J. Sports Sci. 2003, 21, 519–528. [Google Scholar] [CrossRef]
- Sykes, D.; Twist, C.; Nicholas, C.; Lamb, K. Changes in locomotive rates during senior elite rugby league matches. J. Sports Sci. 2011, 29, 1263–1271. [Google Scholar] [CrossRef]
- Coutts, A.J.; Quinn, J.; Hocking, J.; Castagna, C.; Rampinini, E. Match running performance in elite Australian rules football. J. Sci. Med. Sport 2010, 13, 543–548. [Google Scholar] [CrossRef]
- Williams, C.; Rollo, I. Carbohydrate nutrition and team sport performance. Sports Med. 2015, 45, 13–22. [Google Scholar] [CrossRef]
- Waldron, M.; Highton, J. Fatigue and pacing in high-intensity intermittent team sport: An update. Sports Med. 2014, 44, 1645–1658. [Google Scholar] [CrossRef]
- Balsom, P.D.; Wood, K.; Olsson, P.; Ekblom, B. Carbohydrate intake and multiple sprint sports: With special reference to football (soccer). Int. J. Sports Med. 1999, 20, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Holway, F.E.; Spriet, L.L. Sport-specific nutrition: Practical strategies for team sports. J. Sports Sci. 2011, 29, S115–S125. [Google Scholar] [CrossRef] [PubMed]
- Cassidy, C.; Collins, K.; Shortall, M. The precompetition macronutrient intake of elite Gaelic football players. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 574–579. [Google Scholar] [CrossRef] [PubMed]
- Catháin, C.Ó.; Fleming, J.; Renard, M.; Kelly, D.T. Dietary intake of Gaelic football players during game preparation and recovery. Sports 2020, 8, 62. [Google Scholar] [CrossRef] [PubMed]
- Hulton, A.T.; Edwards, J.P.; Gregson, W.; MacLaren, D.; Doran, D.A. Effect of fat and CHO meals on intermittent exercise in soccer players. Int. J. Sports Med. 2012, 34, 165–169. [Google Scholar] [CrossRef] [PubMed]
- Raman, A.; MacDermid, P.W.; Mündel, T.; Mann, M.; Stannard, S.R. The effects of carbohydrate loading 48 hours before a simulated squash match. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 157–165. [Google Scholar] [CrossRef]
- Bangsbo, J.; Mohr, M.; Krustrup, P. Physical and metabolic demands of training and match-play in the elite football player. J. Sports Sci. 2006, 24, 665–674. [Google Scholar] [CrossRef] [PubMed]
- King, S.; O’Donoghue, P. The activity profile of men’s Gaelic football. Int. J. Perform. Anal. Sport 2003, 3, 130–144. [Google Scholar] [CrossRef]
- Collins, K.; Doran, D.; Morton, J.; McRobert, A. The reliability of a multidirectional hurling simulation protocol. In Proceedings of the 12th Annual Conference of the Faculty of Sport and Exercise Medicine, Royal College of Surgeons Ireland, Dublin, Ireland, 17–19 September 2015. [Google Scholar]
- Burgess, K.; Holt, T.; Munro, S.; Swinton, P. Reliability and validity of the running anaerobic sprint test (RAST) in soccer players. J. Trainol. 2016, 5, 24–29. [Google Scholar] [CrossRef]
- O’Brien, L. The physical demands and nutritional requirements of Gaelic football training and match-play. In School of Health Sciences; Liverpool Hope University: Liverpool, UK, 2020. [Google Scholar]
- Hawley, J.A.; Noakes, T.D. Peak power output predicts maximal oxygen uptake and performance time in trained cyclists. Graefe’s Arch. Clin. Exp. Ophthalmol. 1992, 65, 79–83. [Google Scholar] [CrossRef]
- Taylor, C.; Bartlett, J.D.; Van De Graaf, C.S.; Louhelainen, J.; Coyne, V.; Iqbal, Z.; MacLaren, D.P.M.; Gregson, W.; Close, G.L.; Morton, J.P. Protein ingestion does not impair exercise-induced AMPK signalling when in a glycogen-depleted state: Implications for train-low compete-high. Graefe’s Arch. Clin. Exp. Ophthalmol. 2012, 113, 1457–1468. [Google Scholar] [CrossRef]
- Thomas, D.T.; Erdman, K.A.; Burke, L.M. Nutrition and athletic performance. Med. Sci. Sports Exerc. 2016, 48, 543–568. [Google Scholar] [CrossRef]
- Nuzzo, J.L.; Anning, J.H.; Scharfenberg, J.M. The reliability of three devices used for measuring vertical jump height. J. Strength Cond. Res. 2011, 25, 2580–2590. [Google Scholar] [CrossRef] [PubMed]
- Fritz, C.O.; Morris, P.E.; Richler, J.J. Effect size estimates: Current use, calculations, and interpretation. J. Exp. Psychol. Gen. 2012, 141, 2–18. [Google Scholar] [CrossRef]
- Skein, M.; Duffield, R.; Kelly, B.T.; Marino, F.E. The effects of carbohydrate intake and muscle glycogen content on self-paced intermittent-sprint exercise despite no knowledge of carbohydrate manipulation. Graefe’s Arch. Clin. Exp. Ophthalmol. 2011, 112, 2859–2870. [Google Scholar] [CrossRef]
- Bradley, W.J.; Morehen, J.C.; Haigh, J.; Clarke, J.; Donovan, T.F.; Twist, C.; Cotton, C.; Shepherd, S.; Cocks, M.; Sharma, A.; et al. Muscle glycogen utilisation during Rugby match play: Effects of pre-game carbohydrate. J. Sci. Med. Sport 2016, 19, 1033–1038. [Google Scholar] [CrossRef] [PubMed]
- Spencer, M.; Bishop, D.; Dawson, B.; Goodman, C. Physiological and Metabolic Responses of Repeated-Sprint Activities. Sports Med. 2005, 35, 1025–1044. [Google Scholar] [CrossRef]
- Rampinini, E.; Bosio, A.; Ferraresi, I.; Petruolo, A.; Morelli, A.; Sassi, A. Match-Related Fatigue in Soccer Players. Med. Sci. Sports Exerc. 2011, 43, 2161–2170. [Google Scholar] [CrossRef]
- Bergström, J.; Hermansen, L.; Hultman, E.; Saltin, B. Diet, Muscle Glycogen and Physical Performance. Acta Physiol. Scand. 1967, 71, 140–150. [Google Scholar] [CrossRef]
- Jeukendrup, A.E.; Wallis, G.A. Measurement of substrate oxidation during exercise by means of gas exchange measurements. Int. J. Sports Med. 2004, 26, S28–S37. [Google Scholar] [CrossRef]
- Krustrup, P.; Mohr, M.; Steensberg, A.; Bencke, J.; Kjær, M.; Bangsbo, J. Muscle and Blood Metabolites during a Soccer Game. Med. Sci. Sports Exerc. 2006, 38, 1165–1174. [Google Scholar] [CrossRef]
- Chryssanthopoulos, C.; Williams, C.; Nowitz, A.; Bogdanis, G. Skeletal muscle glycogen concentration and metabolic responses following a high glycaemic carbohydrate breakfast. J. Sports Sci. 2004, 22, 1065–1071. [Google Scholar] [CrossRef]
- Foskett, A.; Williams, C.; Boobis, L.; Tsintzas, K. Carbohydrate availability and muscle energy metabolism during intermittent running. Med. Sci. Sports Exerc. 2008, 40, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Bangsbo, J. The physiology of soccer-with special reference to intense intermittent exercise. Acta Physiol. Scand. Suppl. 1994, 619, 1–155. [Google Scholar]
- Ludwig, D.S.; Hu, F.B.; Tappy, L.; Brand-Miller, J. Dietary carbohydrates: Role of quality and quantity in chronic disease. BMJ 2018, 361, k2340. [Google Scholar] [CrossRef]
| Time | High-Carbohydrate Diet | Low-Carbohydrate Diet |
|---|---|---|
| 8.00 a.m. | Cornflakes (100 g) | Cornflakes (50 g) |
| Semi skimmed milk (250 mL) | Semi skimmed milk (250 mL) | |
| Orange juice (250 mL) | 2 boiled eggs | |
| White bread sliced (60 g) | Water (500 mL) | |
| Strawberry jam (30 g) | ||
| 10.30 a.m. | Muller rice (180 g) | Cooked chicken pieces (60 g) |
| Banana | 1 medium banana | |
| Lucozade sport (500 mL) | Mixed nuts (60 g) | |
| Water (500 mL) | ||
| 1.00 p.m. | Uncle Ben’s basmati rice (250 g) | Uncle Ben’s basmati rice (250 g) |
| Chicken breast (90 g) | Chicken breast (90 g) | |
| Frozen mixed veg (163 g) | Frozen mixed veg (163 g) | |
| Water (500 mL) | Water (500 mL) | |
| 3.30 p.m. | Apple | Greek yogurt (150 g) |
| Nutri-grain bar (37 g) | 1 medium apple | |
| Lucozade sport (500 mL) | Water (500 mL) | |
| 6.00 p.m. | Pasta (250 g) | Pasta (250 g) |
| Domino tomato pasta sauce (125 g) | Cheese (120 g) | |
| Chicken breast (90 g) | Chicken breast (90 g) | |
| Frozen mixed veg (163 g) | Frozen mixed veg (163 g) | |
| Water (500 mL) | Water (500 mL) | |
| 9.00 p.m. | Muller rice (180 g) | Beef biltong (65 g) |
| Banana | 1 medium banana | |
| Water (500 mL) | Water (500 mL) |
| High-Carbohydrate Diet (72% of Total Energy Intake) | Low-Carbohydrate Diet (36% of Total Energy Intake) | |
|---|---|---|
| Energy (kcal/day) | 3126 | 3077 |
| Carbohydrate (g·kg−1·day−1) | 7 | 3.5 |
| Protein (g·kg−1·day−1) | 1.7 | 2.8 |
| Fat (g·kg−1·day−1) | 0.5 | 1.4 |
| Fluid (mL/day) | 3000 | 3000 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
O’Brien, L.; Collins, K.; Webb, R.; Davies, I.; Doran, D.; Amirabdollahian, F. The Effects of Pre-Game Carbohydrate Intake on Running Performance and Substrate Utilisation during Simulated Gaelic Football Match Play. Nutrients 2021, 13, 1392. https://doi.org/10.3390/nu13051392
O’Brien L, Collins K, Webb R, Davies I, Doran D, Amirabdollahian F. The Effects of Pre-Game Carbohydrate Intake on Running Performance and Substrate Utilisation during Simulated Gaelic Football Match Play. Nutrients. 2021; 13(5):1392. https://doi.org/10.3390/nu13051392
Chicago/Turabian StyleO’Brien, Luke, Kieran Collins, Richard Webb, Ian Davies, Dominic Doran, and Farzad Amirabdollahian. 2021. "The Effects of Pre-Game Carbohydrate Intake on Running Performance and Substrate Utilisation during Simulated Gaelic Football Match Play" Nutrients 13, no. 5: 1392. https://doi.org/10.3390/nu13051392
APA StyleO’Brien, L., Collins, K., Webb, R., Davies, I., Doran, D., & Amirabdollahian, F. (2021). The Effects of Pre-Game Carbohydrate Intake on Running Performance and Substrate Utilisation during Simulated Gaelic Football Match Play. Nutrients, 13(5), 1392. https://doi.org/10.3390/nu13051392








