Effects of Oro-Sensory Exposure on Satiation and Underlying Neurophysiological Mechanisms—What Do We Know So Far?
Abstract
1. Introduction
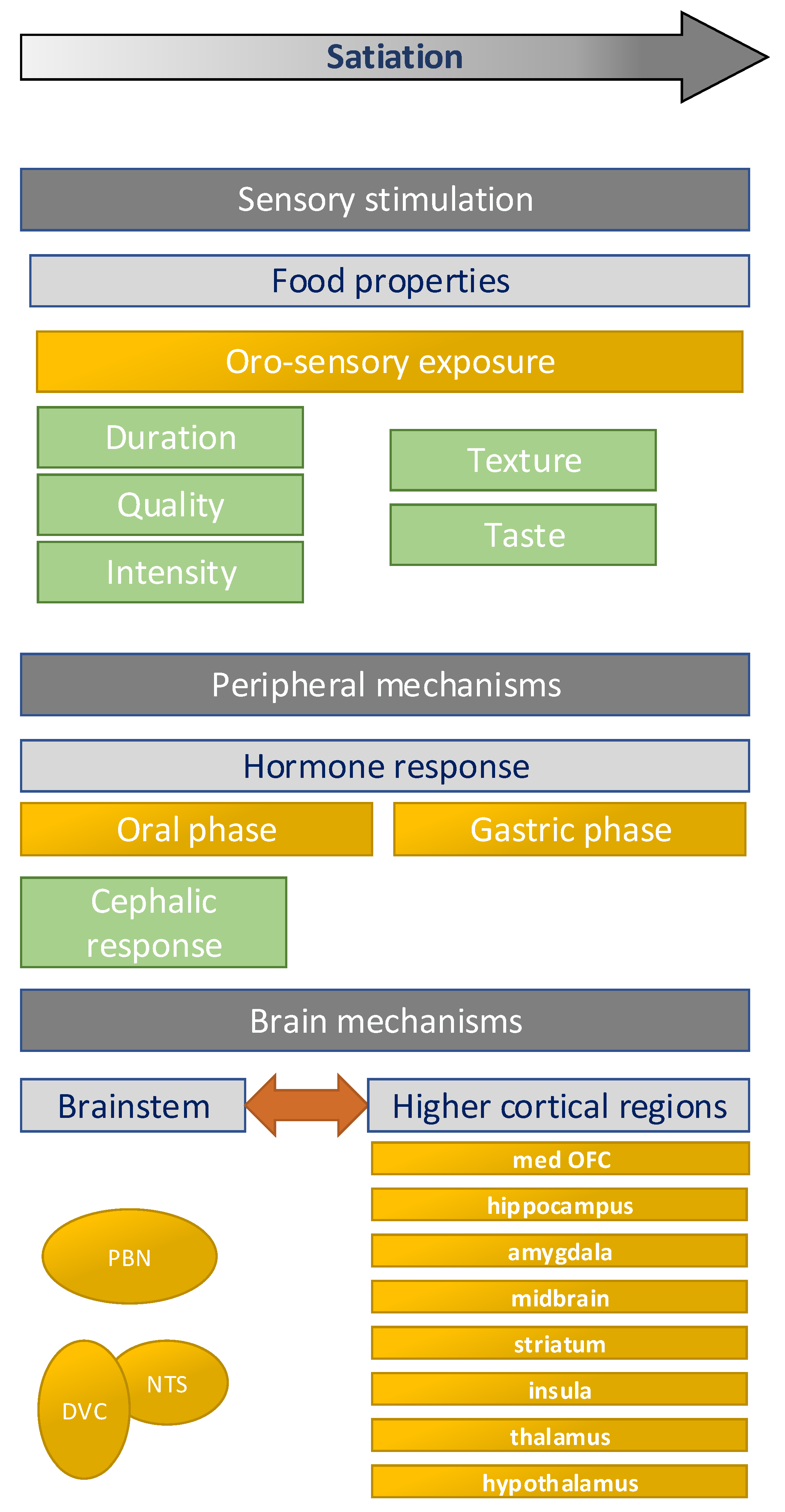
2. Effects on Satiation
2.1. Factors Affecting Oro-Sensory Exposure
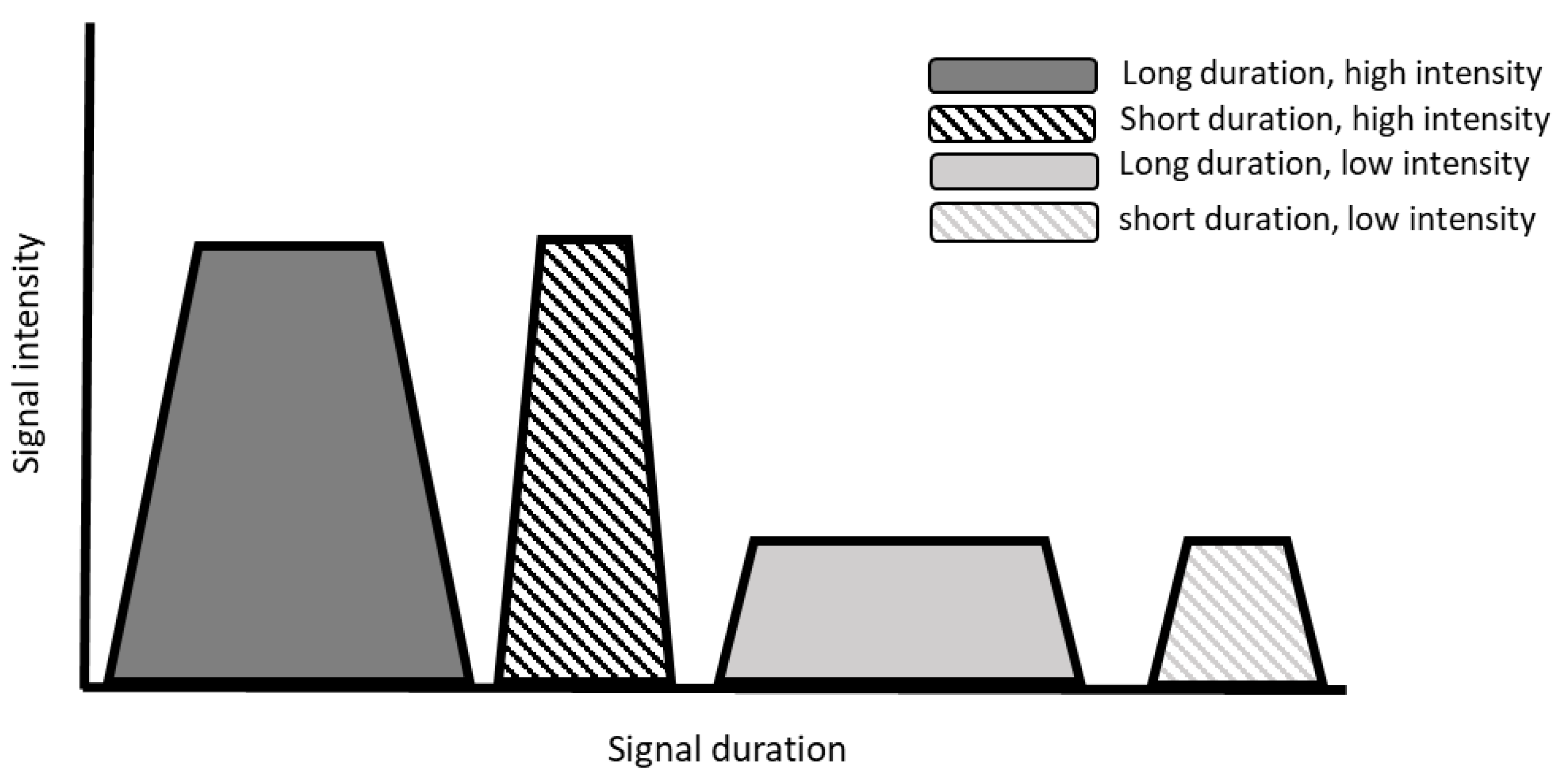
2.2. Oral Exposure Duration
2.3. Taste Intensity and Quality
2.4. Discussion Effects of Oro-Sensory Exposure on Satiation
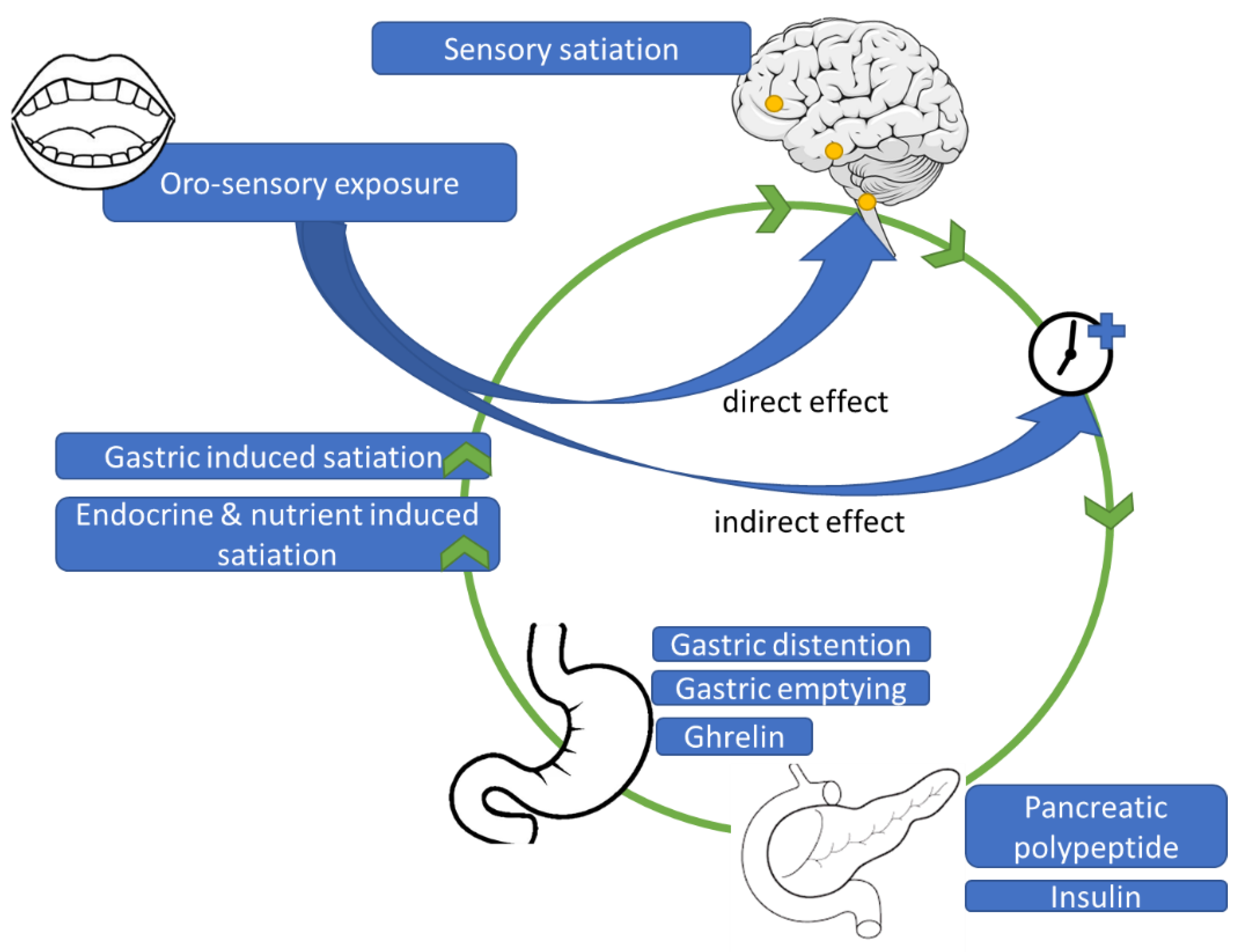
3. Neurophysiological Mechanisms
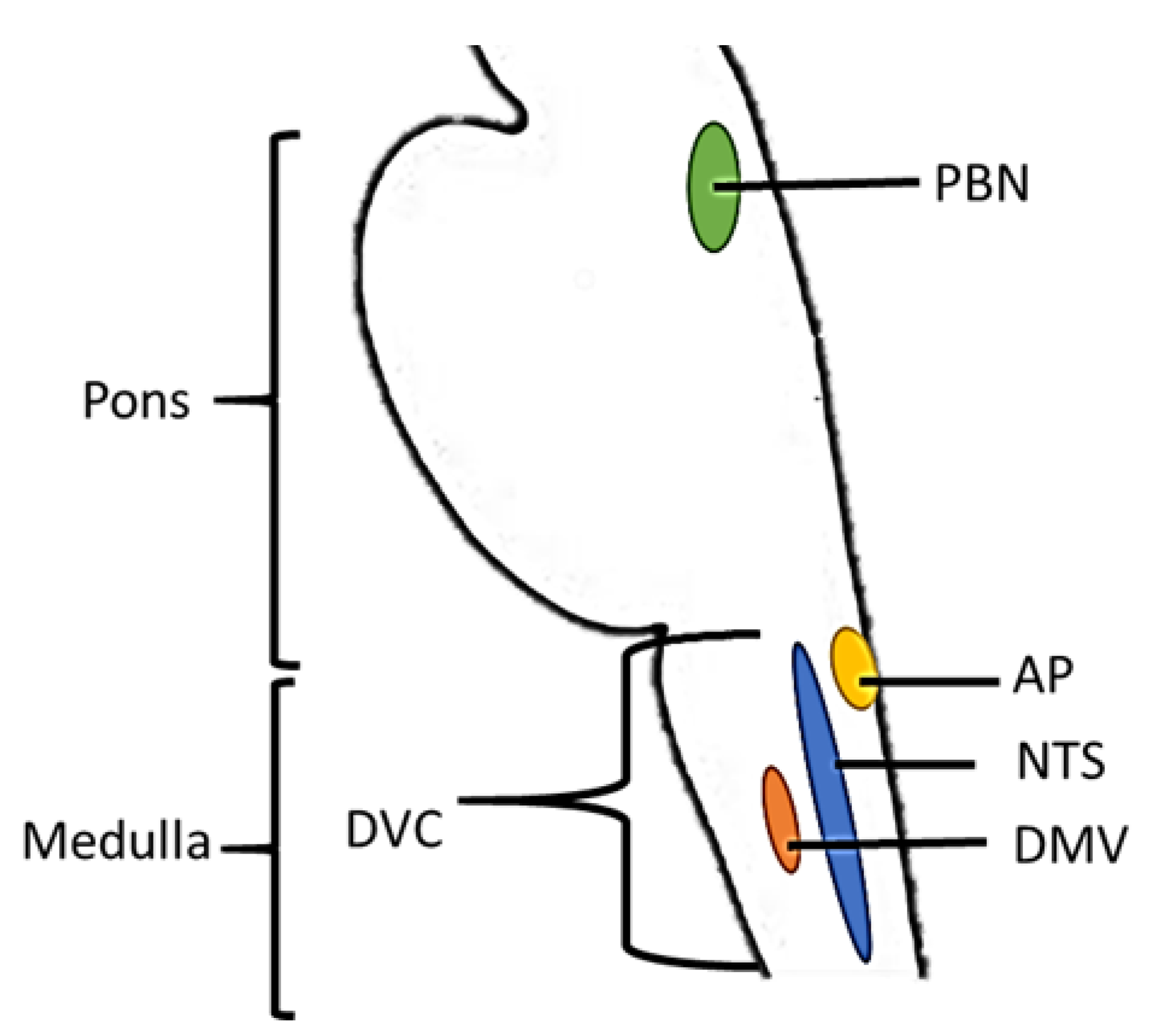
3.1. Brain Stem
3.2. Higher Cortical Regions
3.3. Cephalic Phase Responses
3.4. Discussion Neurophysiological Mechanisms in Satiation
4. Recommendations for Future Research
5. Conclusions
6. Implications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Blundell, J.; de Graaf, C.; Hulshof, T.; Jebb, S.; Livingstone, B.; Lluch, A.; Mela, D.; Salah, S.; Schuring, E.; van der Knaap, H.; et al. Appetite control: Methodological aspects of the evaluation of foods. Obes. Rev. 2010, 11, 251–270. [Google Scholar] [CrossRef] [PubMed]
- Delzenne, N.; Blundell, J.; Brouns, F.; Cunningham, K.; De Graaf, K.; Erkner, A.; Lluch, A.; Mars, M.; Peters, H.P.; Westerterp-Plantenga, M. Gastrointestinal targets of appetite regulation in humans. Obes. Rev. 2010, 11, 234–250. [Google Scholar] [CrossRef]
- Stieger, M.; van de Velde, F. Microstructure, texture and oral processing: New ways to reduce sugar and salt in foods. Curr. Opin. Colloid Interface Sci. 2013, 18, 334–348. [Google Scholar] [CrossRef]
- Lasschuijt, M.P.; Mars, M.; de Graaf, C.; Smeets, P.A.M. Endocrine cephalic phase responses to food cues: A systematic review. Adv. Nutr. 2020, 11, 1364–1383. [Google Scholar] [CrossRef] [PubMed]
- Smeets, P.A.; Erkner, A.; de Graaf, C. Cephalic phase responses and appetite. Nutr. Rev. 2010, 68, 643–655. [Google Scholar] [CrossRef] [PubMed]
- Power, M.L.; Schulkin, J. Anticipatory physiological regulation in feeding biology: Cephalic phase responses. Appetite 2008, 50, 194–206. [Google Scholar] [CrossRef] [PubMed]
- Woods, S.C. The eating paradox: How we tolerate food. Psychol. Rev. 1991, 98, 488–505. [Google Scholar] [CrossRef] [PubMed]
- Rolls, B.J.; Rolls, E.T.; Rowe, E.A.; Sweeney, K. Sensory specific satiety in man. Physiol. Behav. 1981, 27, 137–142. [Google Scholar] [CrossRef]
- Hopkins, M.; Blundell, J.; Halford, J.; King, N.; Finlayson, G. The Regulation of Food Intake in Humans. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dhatariya, K., Dungan, K., Grossman, A., Hershman, J.M., Hofland, J., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- de Graaf, C. Texture and satiation: The role of oro-sensory exposure time. Physiol. Behav. 2012, 107, 496–501. [Google Scholar] [CrossRef] [PubMed]
- Bolhuis, D.P.; Lakemond, C.M.; de Wijk, R.A.; Luning, P.A.; Graaf, C. Both longer oral sensory exposure to and higher intensity of saltiness decrease ad libitum food intake in healthy normal-weight men. J. Nutr. 2011, 141, 2242–2248. [Google Scholar] [CrossRef] [PubMed]
- Forde, C.G.; Mars, M.; de Graaf, K. Ultra-processing or oral processing? A role for energy density and eating rate in moderating energy intake from processed foods. Curr. Dev. Nutr. 2020, 4, nzaa019. [Google Scholar] [CrossRef]
- van den Boer, J.; Werts, M.; Siebelink, E.; de Graaf, C.; Mars, M. The availability of slow and fast calories in the dutch diet: The current situation and opportunities for interventions. Foods 2017, 6, 87. [Google Scholar] [CrossRef] [PubMed]
- Devezeaux de Lavergne, M.; Derks, J.A.M.; Ketel, E.C.; de Wijk, R.A.; Stieger, M. Eating behaviour explains differences between individuals in dynamic texture perception of sausages. Food Qual. Pref. 2015, 41, 189–200. [Google Scholar] [CrossRef]
- Zijlstra, N.; Mars, M.; de Wijk, R.A.; Westerterp-Plantenga, M.S.; de Graaf, C. The effect of viscosity on ad libitum food intake. Int. J. Obes. 2008, 32, 676–683. [Google Scholar] [CrossRef] [PubMed]
- Szczesniak, A.S. Texture is a sensory property. Food Qual. Pref. 2002, 13, 215–225. [Google Scholar] [CrossRef]
- van Vliet, T.; van Aken, G.A.; de Jongh, H.H.; Hamer, R.J. Colloidal aspects of texture perception. Adv. Colloid. Interface. Sci. 2009, 150, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Mosca, A.C.; Torres, A.P.; Slob, E.; de Graaf, K.; McEwan, J.A.; Stieger, M. Small food texture modifications can be used to change oral processing behaviour and to control ad libitum food intake. Appetite 2019, 142, 104375. [Google Scholar] [CrossRef] [PubMed]
- Lasschuijt, M.; Mars, M.; de Graaf, C.; Smeets, P.A.M. How oro-sensory exposure and eating rate affect satiation and associated endocrine responses-a randomized trial. Am. J. Clin. Nutr. 2020, 111, 1137–1149. [Google Scholar] [CrossRef]
- Robinson, E.; Almiron-Roig, E.; Rutters, F.; de Graaf, C.; Forde, C.G.; Tudur Smith, C.; Nolan, S.J.; Jebb, S.A. A systematic review and meta-analysis examining the effect of eating rate on energy intake and hunger. Am. J. Clin. Nutr. 2014, 100, 123–151. [Google Scholar] [CrossRef]
- Zijlstra, N.; de Wijk, R.A.; Mars, M.; Stafleu, A.; de Graaf, C. Effect of bite size and oral processing time of a semisolid food on satiation. Am. J. Clin. Nutr. 2009, 90, 269–275. [Google Scholar] [CrossRef]
- den Boer, A.; Boesveldt, S.; Lawlor, J.B. How sweetness intensity and thickness of an oral nutritional supplement affects intake and satiety. Food Qual. Pref. 2019, 71, 406–414. [Google Scholar] [CrossRef]
- Hogenkamp, P.S.; Mars, M.; Stafleu, A.; de Graaf, C. Intake during repeated exposure to low- and high-energy-dense yogurts by different means of consumption. Am. J. Clin. Nutr. 2010, 91, 841–847. [Google Scholar] [CrossRef] [PubMed]
- Forde, C.G.; van Kuijk, N.; Thaler, T.; de Graaf, C.; Martin, N. Texture and savoury taste influences on food intake in a realistic hot lunch time meal. Appetite 2013, 60, 180–186. [Google Scholar] [CrossRef] [PubMed]
- Bolhuis, D.P.; Forde, C.G.; Cheng, Y.; Xu, H.; Martin, N.; de Graaf, C. Slow food: Sustained impact of harder foods on the reduction in energy intake over the course of the day. PLoS ONE 2014, 9, e93370. [Google Scholar] [CrossRef] [PubMed]
- Zijlstra, N.; Mars, M.; Stafleu, A.; de Graaf, C. The effect of texture differences on satiation in 3 pairs of solid foods. Appetite 2010, 55, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Cahayadi, J.; Leong, S.Y.; Oey, I.; Peng, M. Textural effects on perceived satiation and ad libitum intake of potato chips in males and females. Foods 2020, 9, 85. [Google Scholar] [CrossRef] [PubMed]
- Bolhuis, D.P.; Forde, C.G. Application of food texture to moderate oral processing behaviors and energy intake. Trends Food Sci. Technol. 2020, 106, 445–456. [Google Scholar] [CrossRef]
- Weijzen, P.L.; Smeets, P.A.; de Graaf, C. Sip size of orangeade: Effects on intake and sensory-specific satiation. Br. J. Nutr. 2009, 102, 1091–1097. [Google Scholar] [CrossRef]
- Weijzen, P.L.; Liem, D.G.; Zandstra, E.H.; de Graaf, C. Sensory specific satiety and intake: The difference between nibble- and bar-size snacks. Appetite 2008, 50, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Cook, D.J.; Hollowood, T.A.; Taylor, A.J. Effects of viscosity on flavor perception: Multi-modal approach. Abstr. Pap. Am. Chem. Soc. 2002, 224, U75. [Google Scholar]
- Hollowood, T.A.; Linforth, R.S.; Taylor, A.J. The effect of viscosity on the perception of flavour. Chem. Senses 2002, 27, 583–591. [Google Scholar] [CrossRef] [PubMed]
- Nishinari, K. Rheology, food texture and mastication. J. Text. Stud. 2004, 35, 113–124. [Google Scholar] [CrossRef]
- Bourne, M. Food Texture and Viscosity: Concept and Measurement; Academic Press: Cambridge, MA, USA, 2002. [Google Scholar]
- Lucas, F.; Bellisle, F. The measurement of food preferences in humans: Do taste-and-spit tests predict consumption? Phys. Behav. 1987, 39, 739–743. [Google Scholar] [CrossRef]
- Vickers, Z.; Holton, E.; Wang, J. Effect of ideal-relative sweetness on yogurt consumption. Food Qual. Pref. 2001, 12, 521–526. [Google Scholar] [CrossRef]
- Vickers, Z.; Holton, E.; Wang, J. Effect of yogurt sweetness on sensory specific satiety. J. Sens. Stud. 1998, 13, 377–388. [Google Scholar] [CrossRef]
- Bolhuis, D.P.; Lakemond, C.M.; de Wijk, R.A.; Luning, P.A.; de Graaf, C. Effect of salt intensity in soup on ad libitum intake and on subsequent food choice. Appetite 2012, 58, 48–55. [Google Scholar] [CrossRef]
- Griffioen-Roose, S.; Mars, M.; Finlayson, G.; Blundell, J.E.; de Graaf, C. Satiation due to equally palatable sweet and savory meals does not differ in normal weight young adults. J. Nutr. 2009, 139, 2093–2098. [Google Scholar] [CrossRef] [PubMed]
- Lasschuijt, M.P.; Mars, M.; Stieger, M.; Miquel-Kergoat, S.; de Graaf, C.; Smeets, P. Comparison of oro-sensory exposure duration and intensity manipulations on satiation. Physiol. Behav. 2017, 176, 76–83. [Google Scholar] [CrossRef] [PubMed]
- McCrickerd, K.; Forde, C.G. Sensory influences on food intake control: Moving beyond palatability. Obes. Rev. 2016, 17, 18–29. [Google Scholar] [CrossRef] [PubMed]
- Stribitcaia, E.; Evans, C.E.L.; Gibbons, C.; Blundell, J.; Sarkar, A. Food texture influences on satiety: Systematic review and meta-analysis. Sci. Rep. 2020, 10, 12929. [Google Scholar] [CrossRef]
- Griffioen-Roose, S.; Finlayson, G.; Mars, M.; Blundell, J.E.; de Graaf, C. Measuring food reward and the transfer effect of sensory specific satiety. Appetite 2010, 55, 648–655. [Google Scholar] [CrossRef] [PubMed]
- Mars, M.; Hogenkamp, P.S.; Gosses, A.M.; Stafleu, A.; De Graaf, C. Effect of viscosity on learned satiation. Physiol. Behav. 2009, 98, 60–66. [Google Scholar] [CrossRef]
- Hogenkamp, P.S.; Stafleu, A.; Mars, M.; Brunstrom, J.M.; de Graaf, C. Texture, not flavor, determines expected satiation of dairy products. Appetite 2011, 57, 635–641. [Google Scholar] [CrossRef] [PubMed]
- Ruijschop, R.M.; Zijlstra, N.; Boelrijk, A.E.; Dijkstra, A.; Burgering, M.J.; Graaf, C.; Westerterp-Plantenga, M.S. Effects of bite size and duration of oral processing on retro-nasal aroma release—Features contributing to meal termination. Br. J. Nutr. 2011, 105, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Ruijschop, R.M.; Boelrijk, A.E.; de Ru, J.A.; de Graaf, C.; Westerterp-Plantenga, M.S. Effects of retro-nasal aroma release on satiation. Br. J. Nutr. 2008, 99, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- Ketel, E.C.; Aguayo-Mendoza, M.G.; de Wijk, R.A.; de Graaf, C.; Piqueras-Fiszman, B.; Stieger, M. Age, gender, ethnicity and eating capability influence oral processing behaviour of liquid, semi-solid and solid foods differently. Food. Res. Int. 2019, 119, 143–151. [Google Scholar] [CrossRef] [PubMed]
- de Graaf, C.; Kok, F.J. Slow food, fast food and the control of food intake. Nat. Rev. Endocrinol. 2010, 6, 290–293. [Google Scholar] [CrossRef] [PubMed]
- van Dongen, M.V.; van den Berg, M.C.; Vink, N.; Kok, F.J.; de Graaf, C. Taste-nutrient relationships in commonly consumed foods. Br. J. Nutr. 2012, 108, 140–147. [Google Scholar] [CrossRef]
- Sessle, B.J. Mechanisms of oral somatosensory and motor functions and their clinical correlates. J. Oral. Rehabil. 2006, 33, 243–261. [Google Scholar] [CrossRef]
- Harvey, A.K.; Pattinson, K.T.; Brooks, J.C.; Mayhew, S.D.; Jenkinson, M.; Wise, R.G. Brainstem functional magnetic resonance imaging: Disentangling signal from physiological noise. J. Magn. Reson. Imaging. 2008, 28, 1337–1344. [Google Scholar] [CrossRef]
- Brooks, J.C.; Faull, O.K.; Pattinson, K.T.; Jenkinson, M. Physiological noise in brainstem fmri. Front. Hum. Neurosci. 2013, 7, 623. [Google Scholar] [CrossRef] [PubMed]
- Beissner, F. Functional mri of the brainstem: Common problems and their solutions. Clin. Neuroradiol. 2015, 25 (Suppl. 2), 251–257. [Google Scholar] [CrossRef]
- Scott, T.R.; Yaxley, S.; Sienkiewicz, Z.J.; Rolls, E.T. Gustatory responses in the nucleus tractus solitarius of the alert cynomolgus monkey. J. Neurophysiol. 1986, 55, 182–200. [Google Scholar] [CrossRef] [PubMed]
- Alhadeff, A.L.; Baird, J.P.; Swick, J.C.; Hayes, M.R.; Grill, H.J. Glucagon-like peptide-1 receptor signaling in the lateral parabrachial nucleus contributes to the control of food intake and motivation to feed. Neuropsychopharmacology 2014, 39, 2233–2243. [Google Scholar] [CrossRef] [PubMed]
- Baird, J.P.; Travers, S.P.; Travers, J.B. Integration of gastric distension and gustatory responses in the parabrachial nucleus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 281, R1581–R1593. [Google Scholar] [CrossRef] [PubMed]
- Baird, J.P.; Travers, J.B.; Travers, S.P. Parametric analysis of gastric distension responses in the parabrachial nucleus. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 281, R1568–R1580. [Google Scholar] [CrossRef] [PubMed]
- Blouet, C.; Schwartz, G.J. Brainstem nutrient sensing in the nucleus of the solitary tract inhibits feeding. Cell Metab. 2012, 16, 579–587. [Google Scholar] [CrossRef] [PubMed]
- Schneeberger, M.; Gomis, R.; Claret, M. Hypothalamic and brainstem neuronal circuits controlling homeostatic energy balance. J. Endocrinol. 2014, 220, T25–T46. [Google Scholar] [CrossRef]
- Small, D.M. Taste representation in the human insula. Brain. Struct. Funct. 2010, 214, 551–561. [Google Scholar] [CrossRef] [PubMed]
- Naidich, T.h.P.; Duvernoy, H.M.; Delman, B.N.; Sorensen, A.G.; Kollias, S.S.; Haacke, E.M. Duvernoy’s Atlas of The Human Brain Stem and Cerebellum: High-Field Mri, Surface Anatomy, Internal Structure, Vascularization And 3 D Sectional Anatomy; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2009. [Google Scholar]
- Small, D.M.; Gregory, M.D.; Mak, Y.E.; Gitelman, D.; Mesulam, M.M.; Parrish, T. Dissociation of neural representation of intensity and affective valuation in human gustation. Neuron 2003, 39, 701–711. [Google Scholar] [CrossRef]
- Komisaruk, B.R.; Mosier, K.M.; Liu, W.C.; Criminale, C.; Zaborszky, L.; Whipple, B.; Kalnin, A. Functional localization of brainstem and cervical spinal cord nuclei in humans with fmri. Am. J. Neuroradiol. 2002, 23, 609–617. [Google Scholar]
- Topolovec, J.C.; Gati, J.S.; Menon, R.S.; Shoemaker, J.K.; Cechetto, D.F. Human cardiovascular and gustatory brainstem sites observed by functional magnetic resonance imaging. J. Comp. Neurol. 2004, 471, 446–461. [Google Scholar] [CrossRef]
- Grill, H.J.; Norgren, R. Chronically decerebrate rats demonstrate satiation but not bait shyness. Science 1978, 201, 267–269. [Google Scholar] [CrossRef]
- Small, D.M.; Zatorre, R.J.; Dagher, A.; Evans, A.C.; Jones-Gotman, M. Changes in brain activity related to eating chocolate: From pleasure to aversion. Brain 2001, 124, 1720–1733. [Google Scholar] [CrossRef]
- Devoto, F.; Zapparoli, L.; Bonandrini, R.; Berlingeri, M.; Ferrulli, A.; Luzi, L.; Banfi, G.; Paulesu, E. Hungry brains: A meta-analytical review of brain activation imaging studies on food perception and appetite in obese individuals. Neurosci. Biobehav. Rev. 2018, 94, 271–285. [Google Scholar] [CrossRef]
- Smeets, P.A.; de Graaf, C.; Stafleu, A.; van Osch, M.J.; Nievelstein, R.A.; van der Grond, J. Effect of satiety on brain activation during chocolate tasting in men and women. Am. J. Clin. Nutr. 2006, 83, 1297–1305. [Google Scholar] [CrossRef] [PubMed]
- Winston, J.S.; Gottfried, J.A.; Kilner, J.M.; Dolan, R.J. Integrated neural representations of odor intensity and affective valence in human amygdala. J. Neurosci. 2005, 25, 8903–8907. [Google Scholar] [CrossRef] [PubMed]
- Paton, J.J.; Belova, M.A.; Morrison, S.E.; Salzman, C.D. The primate amygdala represents the positive and negative value of visual stimuli during learning. Nature 2006, 439, 865–870. [Google Scholar] [CrossRef] [PubMed]
- Garavan, H.; Pendergrass, J.C.; Ross, T.J.; Stein, E.A.; Risinger, R.C. Amygdala response to both positively and negatively valenced stimuli. Neuroreport 2001, 12, 2779–2783. [Google Scholar] [CrossRef] [PubMed]
- Hamann, S.; Mao, H. Positive and negative emotional verbal stimuli elicit activity in the left amygdala. Neuroreport 2002, 13, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Jin, J.; Zelano, C.; Gottfried, J.A.; Mohanty, A. Human amygdala represents the complete spectrum of subjective valence. J. Neurosci. 2015, 35, 15145–15156. [Google Scholar] [CrossRef] [PubMed]
- O’Doherty, J.; Rolls, E.T.; Francis, S.; Bowtell, R.; McGlone, F. Representation of pleasant and aversive taste in the human brain. J. Neurophysiol. 2001, 85, 1315–1321. [Google Scholar] [CrossRef] [PubMed]
- Morris, J.S.; Dolan, R.J. Involvement of human amygdala and orbitofrontal cortex in hunger-enhanced memory for food stimuli. J. Neurosci. 2001, 21, 5304–5310. [Google Scholar] [CrossRef]
- LaBar, K.S.; Gitelman, D.R.; Parrish, T.B.; Kim, Y.H.; Nobre, A.C.; Mesulam, M.M. Hunger selectively modulates corticolimbic activation to food stimuli in humans. Behav. Neurosci. 2001, 115, 493–500. [Google Scholar] [CrossRef]
- Anderson, A.K.; Christoff, K.; Stappen, I.; Panitz, D.; Ghahremani, D.G.; Glover, G.; Gabrieli, J.D.; Sobel, N. Dissociated neural representations of intensity and valence in human olfaction. Nat. Neurosci. 2003, 6, 196–202. [Google Scholar] [CrossRef] [PubMed]
- Berthoud, H.R. Vagal and hormonal gut-brain communication: From satiation to satisfaction. Neurogastroenterol. Motil. 2008, 20 (Suppl. 1), 64–72. [Google Scholar] [CrossRef] [PubMed]
- Richardson, C.T.; Feldman, M. Salivary response to food in humans and its effect on gastric acid secretion. Am. J. Physiol. 1986, 250, G85–G91. [Google Scholar] [CrossRef] [PubMed]
- Powley, T.L. The ventromedial hypothalamic syndrome, satiety, and a cephalic phase hypothesis. Psychol. Rev. 1977, 84, 89–126. [Google Scholar] [CrossRef] [PubMed]
- Mattes, R.D. Fat taste and lipid metabolism in humans. Physiol. Behav. 2005, 86, 691–697. [Google Scholar] [CrossRef] [PubMed]
- Teff, K.L.; Mattes, R.D.; Engelman, K. Cephalic phase insulin release in normal weight males: Verification and reliability. Am. J. Physiol. 1991, 261, E430–E436. [Google Scholar] [CrossRef] [PubMed]
- Katschinski, M. Nutritional implications of cephalic phase gastrointestinal responses. Appetite 2000, 34, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Zafra, M.A.; Molina, F.; Puerto, A. The neural/cephalic phase reflexes in the physiology of nutrition. Neurosci. Biobehav. Rev. 2006, 30, 1032–1044. [Google Scholar] [CrossRef]
- Yamashita, H.; Iwai, M.; Nishimura, K.; Kobayashi, N.; Shimazu, T. Altered lipid metabolism during enteral or parenteral nutrition in rats: Comparison with oral feeding. J. Nutr. Sci. Vitaminol. 1993, 39, 151–161. [Google Scholar] [CrossRef]
- Sakata, T.; Yoshimatsu, H.; Masaki, T.; Tsuda, K. Anti-obesity actions of mastication driven by histamine neurons in rats. Exp. Biol. Med. 2003, 228, 1106–1110. [Google Scholar] [CrossRef]
- Tucker, R.M.; Mattes, R.D. 10—Satiation, satiety: The puzzle of solids and liquids. In Satiation, Satiety and the Control of Food Intak; Blundell, J.E., Bellisle, F., Eds.; Woodhead Publishing: Sawston, UK, 2013; pp. 182–201. [Google Scholar]
- Dhillon, J.; Lee, J.Y.; Mattes, R.D. The cephalic phase insulin response to nutritive and low-calorie sweeteners in solid and beverage form. Physiol. Behav. 2017, 181, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Kashima, H.; Eguchi, K.; Miyamoto, K.; Fujimoto, M.; Endo, M.Y.; Aso-Someya, N.; Kobayashi, T.; Hayashi, N.; Fukuba, Y. Suppression of oral sweet taste sensation with gymnema sylvestre affects postprandial gastrointestinal blood flow and gastric emptying in humans. Chem. Senses 2017, 42, 295–302. [Google Scholar] [CrossRef]
- Cecil, J.E.; Francis, J.; Read, N.W. Comparison of the effects of a high-fat and high-carbohydrate soup delivered orally and intragastrically on gastric emptying, appetite, and eating behaviour. Physiol. Behav. 1999, 67, 299–306. [Google Scholar] [CrossRef]
- Mackie, A.R.; Rafiee, H.; Malcolm, P.; Salt, L.; van Aken, G. Specific food structures supress appetite through reduced gastric emptying rate. Am. J. Physiol. Gastrointest. Liver Physiol. 2013, 304, G1038–G1043. [Google Scholar] [CrossRef] [PubMed]
- Deng, R.; Mars, M.; Van Der Sman, R.G.M.; Smeets, P.A.M.; Janssen, A.E.M. The importance of swelling for in vitro gastric digestion of whey protein gels. Food. Chem. 2020, 330, 127182. [Google Scholar] [CrossRef] [PubMed]
- Marciani, L.; Hall, N.; Pritchard, S.E.; Cox, E.F.; Totman, J.J.; Lad, M.; Hoad, C.L.; Foster, T.J.; Gowland, P.A.; Spiller, R.C. Preventing gastric sieving by blending a solid/water meal enhances satiation in healthy humans. J. Nutr. 2012, 142, 1253–1258. [Google Scholar] [CrossRef] [PubMed]
- Ritter, R.C. Gastrointestinal mechanisms of satiation for food. Physiol. Behav. 2004, 81, 249–273. [Google Scholar] [CrossRef] [PubMed]
- Chambers, A.P.; Sandoval, D.A.; Seeley, R.J. Integration of satiety signals by the central nervous system. Curr. Biol. 2013, 23, R379–R388. [Google Scholar] [CrossRef] [PubMed]
- Rasoamanana, R.; Darcel, N.; Fromentin, G.; Tome, D. Nutrient sensing and signalling by the gut. Proc. Nutr. Soc. 2012, 71, 446–455. [Google Scholar] [CrossRef]
- Lemmens, S.G.; Martens, E.A.; Kester, A.D.; Westerterp-Plantenga, M.S. Changes in gut hormone and glucose concentrations in relation to hunger and fullness. Am. J. Clin. Nutr. 2011, 94, 717–725. [Google Scholar] [CrossRef] [PubMed]
- Bellisle, F.; Drewnowski, A.; Anderson, G.H.; Westerterp-Plantenga, M.; Martin, C.K. Sweetness, satiation, and satiety. J. Nutr. 2012, 142, 1149S–1154S. [Google Scholar] [CrossRef] [PubMed]
- Krop, E.M.; Hetherington, M.M.; Nekitsing, C.; Miquel, S.; Postelnicu, L.; Sarkar, A. Influence of oral processing on appetite and food intake—A systematic review and meta-analysis. Appetite 2018, 125, 253–269. [Google Scholar] [CrossRef] [PubMed]
- Llewellyn, C.H.; van Jaarsveld, C.H.; Boniface, D.; Carnell, S.; Wardle, J. Eating rate is a heritable phenotype related to weight in children. Am. J. Clin. Nutr. 2008, 88, 1560–1566. [Google Scholar] [CrossRef]
- Drucker, R.R.; Hammer, L.D.; Agras, W.S.; Bryson, S. Can mothers influence their child’s eating behavior? J. Dev. Behav. Pediatr. 1999, 20, 88–92. [Google Scholar] [CrossRef] [PubMed]
- Murakami, K.; Miyake, Y.; Sasaki, S.; Tanaka, K.; Arakawa, M. Self-reported rate of eating and risk of overweight in japanese children: Ryukyus child health study. J. Nutr. Sci. Vitaminol. 2012, 58, 247–252. [Google Scholar] [CrossRef]
- Fogel, A.; Fries, L.R.; McCrickerd, K.; Goh, A.T.; Quah, P.L.; Chan, M.J.; Toh, J.Y.; Chong, Y.S.; Tan, K.H.; Yap, F.; et al. Oral processing behaviours that promote children’s energy intake are associated with parent-reported appetitive traits: Results from the gusto cohort. Appetite 2018, 126, 8–15. [Google Scholar] [CrossRef]
- Fogel, A.; Goh, A.T.; Fries, L.R.; Sadananthan, S.A.; Velan, S.S.; Michael, N.; Tint, M.T.; Fortier, M.V.; Chan, M.J.; Toh, J.Y.; et al. Faster eating rates are associated with higher energy intakes during an ad libitum meal, higher bmi and greater adiposity among 4.5-year-old children: Results from the growing up in singapore towards healthy outcomes (gusto) cohort. Br. J. Nutr. 2017, 117, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Saper, C.B.; Chou, T.C.; Elmquist, J.K. The need to feed: Homeostatic and hedonic control of eating. Neuron 2002, 36, 199–211. [Google Scholar] [CrossRef]
- Anderson, J.W.; Konz, E.C. Obesity and disease management: Effects of weight loss on comorbid conditions. Obes. Res. 2001, 9 (Suppl. 4), 326S–334S. [Google Scholar] [CrossRef] [PubMed]
- Tuomilehto, J.; Lindstrom, J.; Eriksson, J.G.; Valle, T.T.; Hamalainen, H.; Ilanne-Parikka, P.; Keinanen-Kiukaanniemi, S.; Laakso, M.; Louheranta, A.; Rastas, M.; et al. Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance. N. Engl. J. Med. 2001, 344, 1343–1350. [Google Scholar] [CrossRef] [PubMed]
- Burke, M.V.; Small, D.M. Physiological mechanisms by which non-nutritive sweeteners may impact body weight and metabolism. Physiol. Behav. 2015, 152, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Katz, D.L.; O’Connell, M.; Yeh, M.C.; Nawaz, H.; Njike, V.; Anderson, L.M.; Cory, S.; Dietz, W.; The Community Preventive Services Task Force. Public health strategies for preventing and controlling overweight and obesity in school and worksite settings: A report on recommendations of the task force on community preventive services. Morb. Mortal. Wkly. Rep. Recomm. Rep. 2005, 54, 1–12. [Google Scholar]
- Jeltema, M.; Beckley, J.; Vahalik, J. Model for understanding consumer textural food choice. Food. Sci. Nutr. 2015, 3, 202–212. [Google Scholar] [CrossRef]
- Hermsen, S.; Mars, M.; Higgs, S.; Frost, J.H.; Hermans, R.C.J. Effects of eating with an augmented fork with vibrotactile feedback on eating rate and body weight: A randomized controlled trial. Int. J. Behav. Nutr. Phys. Act. 2019, 16, 90. [Google Scholar] [CrossRef]
- Ford, A.L.; Bergh, C.; Sodersten, P.; Sabin, M.A.; Hollinghurst, S.; Hunt, L.P.; Shield, J.P. Treatment of childhood obesity by retraining eating behaviour: Randomised controlled trial. BMJ 2009, 340, b5388. [Google Scholar] [CrossRef] [PubMed]
- Gezondheidsraad Onafhankelijk Wetenschappelijk Adviesorgraan voor Regering en Parlement. Available online: https://www.gezondheidsraad.nl/documenten/adviezen/2015/11/04/richtlijnen-goede-voeding-2015 (accessed on 11 April 2021).
- U.S. Department of Agriculture and hhs, Dietary Guidelines for Americans 2015–2020. Available online: http://health.Gov/dietaryguidelines/2015/ (accessed on 11 April 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lasschuijt, M.P.; de Graaf, K.; Mars, M. Effects of Oro-Sensory Exposure on Satiation and Underlying Neurophysiological Mechanisms—What Do We Know So Far? Nutrients 2021, 13, 1391. https://doi.org/10.3390/nu13051391
Lasschuijt MP, de Graaf K, Mars M. Effects of Oro-Sensory Exposure on Satiation and Underlying Neurophysiological Mechanisms—What Do We Know So Far? Nutrients. 2021; 13(5):1391. https://doi.org/10.3390/nu13051391
Chicago/Turabian StyleLasschuijt, Marlou P., Kees de Graaf, and Monica Mars. 2021. "Effects of Oro-Sensory Exposure on Satiation and Underlying Neurophysiological Mechanisms—What Do We Know So Far?" Nutrients 13, no. 5: 1391. https://doi.org/10.3390/nu13051391
APA StyleLasschuijt, M. P., de Graaf, K., & Mars, M. (2021). Effects of Oro-Sensory Exposure on Satiation and Underlying Neurophysiological Mechanisms—What Do We Know So Far? Nutrients, 13(5), 1391. https://doi.org/10.3390/nu13051391





