Associations between Age-Related Hearing Loss and DietaryAssessment Using Data from Korean National Health andNutrition Examination Survey
Abstract
1. Introduction
2. Materials and Methods
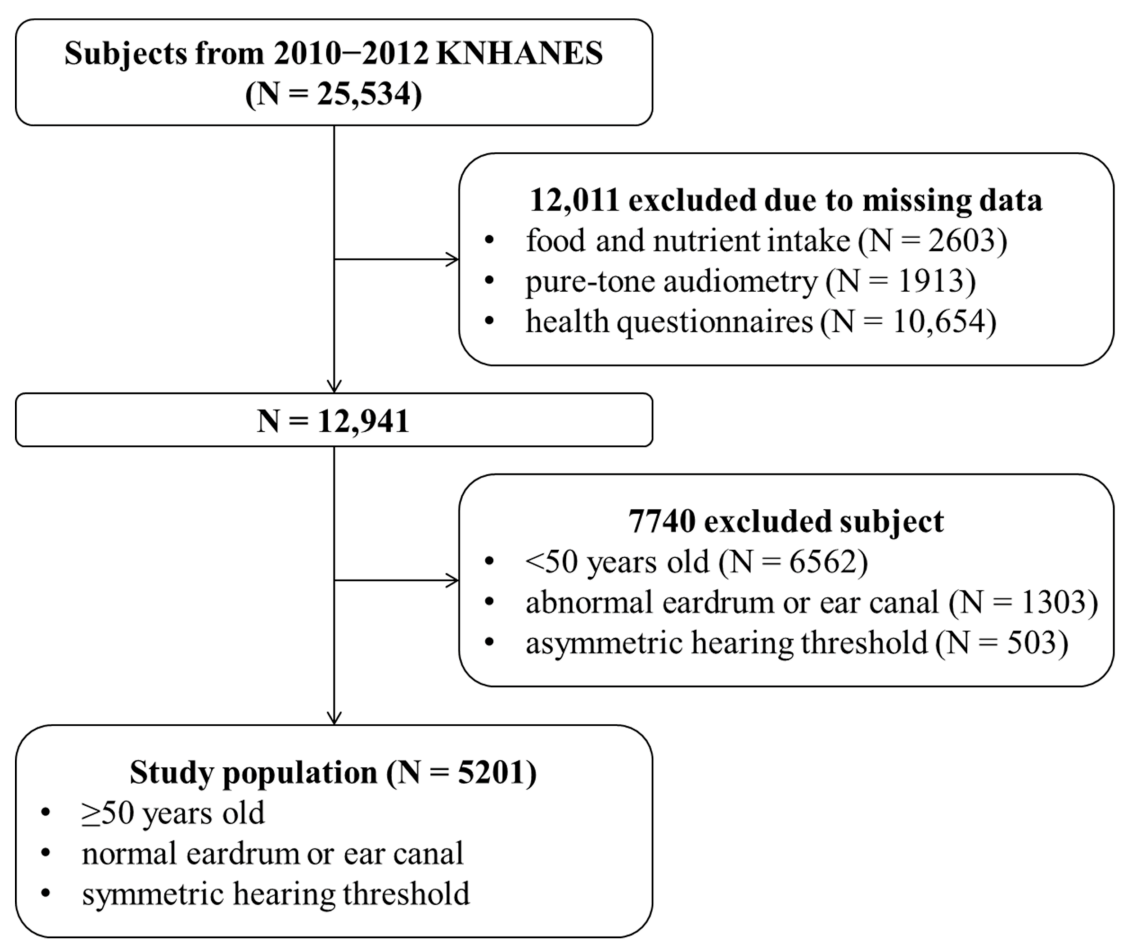
2.1. Data Source and Study Population
2.2. Assessment of Hearing and Risk Factors
2.3. Assessment of Dietary Characteristics
2.4. Statistical Analyses
3. Results
3.1. General Characteristics
3.2. Food Consumption
3.3. Nutrient Intake
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nelson, E.G.; Hinojosa, R. Presbycusis: A Human Temporal Bone Study of Individuals with Downward Sloping Audiometric Patterns of Hearing Loss and Review of the Literature. Laryngoscope 2006, 116, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Wattamwar, K.; Qian, Z.J.; Otter, J.; Leskowitz, M.J.; Caruana, F.F.; Siedlecki, B.; Spitzer, J.B.; Lalwani, A.K. Increases in the Rate of Age-Related Hearing Loss in the Older Old. JAMA Otolaryngol. Head Neck Surg. 2017, 143, 41–45. [Google Scholar] [CrossRef] [PubMed]
- Gopinath, B.; Rochtchina, E.; Wang, J.J.; Schneider, J.; Leeder, S.R.; Mitchell, P. Prevalence of Age-Related Hearing Loss in Older Adults: Blue Mountains Study. Arch. Intern. Med. 2009, 169, 415–418. [Google Scholar] [CrossRef] [PubMed]
- Sprinzl, G.M.; Riechelmann, H. Current Trends in Treating Hearing Loss in Elderly People: A Review of the Technology and Treatment Options—A Mini-Review. Gerontology 2010, 56, 351–358. [Google Scholar] [CrossRef]
- Lin, F.R.; Thorpe, R.; Gordon-Salant, S.; Ferrucci, L. Hearing Loss Prevalence and Risk Factors Among Older Adults in the United States. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2011, 66, 582–590. [Google Scholar] [CrossRef]
- Choi, J.E.; Ahn, J.; Park, H.W.; Baek, S.-Y.; Kim, S.; Moon, I.J. Prevalence of minimal hearing loss in South Korea. PLoS ONE 2017, 12, e0171635. [Google Scholar] [CrossRef]
- Mathers, C.D.; Loncar, D. Projections of Global Mortality and Burden of Disease from 2002 to 2030. PLoS Med. 2006, 3, e442. [Google Scholar] [CrossRef]
- Olusanya, B.O.; Davis, A.C.; Hoffman, H.J. Hearing loss: Rising prevalence and impact. Bull. World Health Organ. 2019, 97, 646. [Google Scholar] [CrossRef]
- Mick, P.; Kawachi, I.; Lin, F.R. The Association between Hearing Loss and Social Isolation in Older Adults. Otolaryngol. Head Neck Surg. 2014, 150, 378–384. [Google Scholar] [CrossRef] [PubMed]
- Ford, A.H.; Hankey, G.J.; Yeap, B.B.; Golledge, J.; Flicker, L.; Almeida, O.P. Hearing loss and the risk of dementia in later life. Maturitas 2018, 112, 1–11. [Google Scholar] [CrossRef]
- Harithasan, D.; Mukari, S.Z.S.; Ishak, W.S.; Shahar, S.; Yeong, W.L. The impact of sensory impairment on cognitive performance, quality of life, depression, and loneliness in older adults. Int. J. Geriatr. Psychiatr. 2020, 35, 358–364. [Google Scholar] [CrossRef] [PubMed]
- Schuknecht, H.F.; Gacek, M.R. Cochlear Pathology in Presbycusis. Ann. Otol. Rhinol. Laryngol. 1993, 102, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Seidman, M.D. Effects of Dietary Restriction and Antioxidants on Presbyacusis. Laryngoscope 2000, 110, 727–738. [Google Scholar] [CrossRef] [PubMed]
- Seidman, M.D.; Ahmad, N.; Joshi, D.; Seidman, J.; Thawani, S.; Quirk, W.S. Age-related hearing loss and its association with reactive oxygen species and mitochondrial DNA damage. Acta Otolaryngol. 2004, 124, 16–24. [Google Scholar] [CrossRef]
- Yamasoba, T.; Lin, F.R.; Someya, S.; Kashio, A.; Sakamoto, T.; Kondo, K. Current concepts in age-related hearing loss: Epidemiology and mechanistic pathways. Hear. Res. 2013, 303, 30–38. [Google Scholar] [CrossRef]
- Watson, N.; Ding, B.; Zhu, X.; Frisina, R.D. Chronic inflammation—inflammaging—in the ageing cochlea: A novel target for future presbycusis therapy. Ageing Res. Rev. 2017, 40, 142–148. [Google Scholar] [CrossRef]
- Van Eyken, E.; Van Camp, G.; Van Laer, L. The Complexity of Age-Related Hearing Impairment: Contributing Environmental and Genetic Factors. Audiol. Neurotol. 2007, 12, 345–358. [Google Scholar] [CrossRef]
- Hultcrantz, M.; Simonoska, R.; Stenberg, A.E. Estrogen and hearing: A summary of recent investigations. Acta Otolaryngol. 2006, 126, 10–14. [Google Scholar] [CrossRef]
- Ohlemiller, K.K. Mechanisms and genes in human strial presbycusis from animal models. Brain Res. 2009, 1277, 70–83. [Google Scholar] [CrossRef]
- Uchida, Y.; Sugiura, S.; Ando, F.; Nakashima, T.; Shimokata, H. Diabetes reduces auditory sensitivity in middle-aged listeners more than in elderly listeners: A population- based study of age-related hearing loss. Med. Sci. Monit. 2010, 16, 63–68. [Google Scholar]
- Saito, T.; Sato, K.; Saito, H. An experimental study of auditory dysfunction associated with hyperlipoproteinemia. Eur. Arch. Otorinolaryngol. 1986, 243, 242–245. [Google Scholar] [CrossRef]
- Sikora, M.A.; Morizono, T.; Ward, W.D.; Paparella, M.M.; Leslie, K. Diet-induced Hyperlipidemia and Auditory Dysfunction. Acta Otolaryngol. 1986, 102, 372–381. [Google Scholar] [CrossRef]
- Du, Z.; Yang, Y.; Hu, Y.; Sun, Y.; Zhang, S.; Peng, W.; Zhong, Y.; Huang, X.; Kong, W. A long-term high-fat diet increases oxidative stress, mitochondrial damage and apoptosis in the inner ear of d-galactose-induced aging rats. Hear. Res. 2012, 287, 15–24. [Google Scholar] [CrossRef]
- Choi, Y.-H.; Miller, J.M.; Tucker, K.L.; Hu, H.; Park, S.K. Antioxidant vitamins and magnesium and the risk of hearing loss in the US general population. Am. J. Clin. Nutr. 2013, 99, 148–155. [Google Scholar] [CrossRef]
- Michikawa, T.; Nishiwaki, Y.; Kikuchi, Y.; Hosoda, K.; Mizutari, K.; Saito, H.; Asakura, K.; Milojevic, A.; Iwasawa, S.; Nakano, M.; et al. Serum Levels of Retinol and Other Antioxidants for Hearing Impairment Among Japanese Older Adults. J. Gerontol. Ser. A Biol. Sci. Med Sci. 2009, 64, 910–915. [Google Scholar] [CrossRef]
- Ackah, S.E.H.; Juhn, S.K.; Huang, T.C.; Wiedmann, T.S. A combination antioxidant therapy prevents age-related hearing loss in C57BL/6 mice. Otolaryngol. Head Neck Surg. 2010, 143, 429–434. [Google Scholar] [CrossRef]
- Fujimoto, C.; Yamasoba, T. Oxidative Stresses and Mitochondrial Dysfunction in Age-Related Hearing Loss. Oxidative Med. Cell. Longev. 2014, 2014, 1–6. [Google Scholar] [CrossRef]
- Alvarado, J.C.; Fuentes-Santamaria, V.; Melgar-Rojas, P.; Valero, M.L.; Gabaldon-Ull, M.C.; Miller, J.M.; Juiz, J.M. Synergistic effects of free radical scavengers and cochlear vasodilators: A new otoprotective strategy for age-related hearing loss. Front. Aging Neurosci. 2015, 7, 86. [Google Scholar] [CrossRef]
- Ünal, M.; Tamer, L.; Doğruer, Z.N.; Yildirim, H.; Vayisoğlu, Y.; Çamdeviren, H. N-Acetyltransferase 2 Gene Polymorphism and Presbycusis. Laryngoscope 2005, 115, 2238–2241. [Google Scholar] [CrossRef]
- Ferrucci, L.; Corsi, A.; Lauretani, F.; Bandinelli, S.; Bartali, B.; Taub, D.D.; Guralnik, J.M.; Longo, D.L. The origins of age-related proinflammatory state. Blood 2005, 105, 2294–2299. [Google Scholar] [CrossRef]
- Sanada, F.; Taniyama, Y.; Muratsu, J.; Otsu, R.; Shimizu, H.; Rakugi, H.; Morishita, R. Source of Chronic Inflammation in Aging. Front. Cardiovasc. Med. 2018, 5, 12. [Google Scholar] [CrossRef] [PubMed]
- Verschuur, C.; Agyemang-Prempeh, A.; Newman, T.A. Inflammation is associated with a worsening of presbycusis: Evidence from the MRC national study of hearing. Int. J. Audiol. 2014, 53, 469–475. [Google Scholar] [CrossRef]
- Frye, M.D.; Yang, W.; Zhang, C.; Xiong, B.; Hu, B.H. Dynamic activation of basilar membrane macrophages in response to chronic sensory cell degeneration in aging mouse cochleae. Hear. Res. 2017, 344, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Bogani, P.; Galli, C.; Villa, M.; Visioli, F. Postprandial anti-inflammatory and antioxidant effects of extra virgin olive oil. Atherosclerosis 2007, 190, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Riso, P.; Visioli, F.; Grande, S.; Guarnieri, S.; Gardana, C.; Simonetti, P.; Porrini, M. Effect of a Tomato-Based Drink on Markers of Inflammation, Immunomodulation, and Oxidative Stress. J. Agric. Food Chem. 2006, 54, 2563–2566. [Google Scholar] [CrossRef] [PubMed]
- Steptoe, A.; Gibson, E.L.; Vuononvirta, R.; Hamer, M.; Wardle, J.; Rycroft, J.A.; Martin, J.F.; Erusalimsky, J.D. The effects of chronic tea intake on platelet activation and inflammation: A double-blind placebo controlled trial. Atherosclerosis 2007, 193, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Shargorodsky, J.; Curhan, S.G.; Eavey, R.; Curhan, G.C. A prospective study of vitamin intake and the risk of hearing loss in men. Otolaryngol. Head Neck Surg. 2010, 142, 231–236. [Google Scholar] [CrossRef] [PubMed]
- Sardone, R.; Lampignano, L.; Guerra, V.; Zupo, R.; Donghia, R.; Castellana, F.; Battista, P.; Bortone, I.; Procino, F.; Castellana, M.; et al. Relationship between Inflammatory Food Consumption and Age-Related Hearing Loss in a Prospective Observational Cohort: Results from the Salus in Apulia Study. Nutrients 2020, 12, 426. [Google Scholar] [CrossRef]
- Kim, T.S.; Chung, J.W. Associations of Dietary Riboflavin, Niacin, and Retinol with Age-related Hearing Loss: An Analysis of Korean National Health and Nutrition Examination Survey Data. Nutrients 2019, 11, 896. [Google Scholar] [CrossRef]
- Kang, J.W.; Choi, H.S.; Kim, K.; Choi, J.Y. Dietary vitamin intake correlates with hearing thresholds in the older population: The Korean National Health and Nutrition Examination Survey. Am. J. Clin. Nutr. 2014, 99, 1407–1413. [Google Scholar] [CrossRef]
- Saylor, J.; Friedmann, E.; Lee, H.J. Navigating Complex Sample Analysis Using National Survey Data. Nurs. Res. 2012, 61, 231–237. [Google Scholar] [CrossRef]
- Kim, S.Y.; Sim, S.; Kim, H.-J.; Choi, H.G. Low-fat and low-protein diets are associated with hearing discomfort among the elderly of Korea. Br. J. Nutr. 2015, 114, 1711–1717. [Google Scholar] [CrossRef]
- Kweon, S.; Kim, Y.; Jang, M.-J.; Kim, Y.; Kim, K.; Choi, S.; Chun, C.; Khang, Y.-H.; Oh, K. Data resource profile: The Korea National Health and Nutrition Examination Survey (KNHANES). Int. J. Epidemiol. 2014, 43, 69–77. [Google Scholar] [CrossRef]
- Ros, E. Health Benefits of Nut Consumption. Nutrition 2010, 2, 652–682. [Google Scholar] [CrossRef]
- Corsetto, P.A.; Montorfano, G.; Zava, S.; Colombo, I.; Ingadottir, B.; Jonsdottir, R.; Sveinsdottir, K.; Rizzo, A.M. Characterization of Antioxidant Potential of Seaweed Extracts for Enrichment of Convenience Food. Antioxidants 2020, 9, 249. [Google Scholar] [CrossRef]
- Jiang, R.; Jacobs, D.R., Jr.; Mayer-Davis, E.; Szklo, M.; Herrington, D.; Jenny, N.S.; Kronmal, R.; Barr, R.G. Nut and Seed Consumption and Inflammatory Markers in the Multi-Ethnic Study of Atherosclerosis. Am. J. Epidemiol. 2006, 163, 222–231. [Google Scholar] [CrossRef]
- Noble, K.V.; Liu, T.; Matthews, L.J.; Schulte, B.A.; Lang, H. Age-Related Changes in Immune Cells of the Human Cochlea. Front. Neurol. 2019, 10, 895. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Dai, M.; Fridberger, A.; Hassan, A.; DeGagne, J.; Neng, L.; Zhang, F.; He, W.; Ren, T.; Trune, D.; et al. Perivascular-resident macrophage-like melanocytes in the inner ear are essential for the integrity of the intrastrial fluid-blood barrier. Proc. Natl. Acad. Sci. USA 2012, 109, 10388–10393. [Google Scholar] [CrossRef]
- Fujioka, M.; Okano, H.; Ogawa, K. Inflammatory and immune responses in the cochlea: Potential therapeutic targets for sensorineural hearing loss. Front. Pharmacol. 2014, 5, 287. [Google Scholar] [CrossRef]
- Iwai, H.; Baba, S.; Omae, M.; Lee, S.; Yamashita, T.; Ikehara, S. Maintenance of systemic immune functions prevents accelerated presbycusis. Brain Res. 2008, 1208, 8–16. [Google Scholar] [CrossRef]
- Iwai, H.; Lee, S.; Inaba, M.; Sugiura, K.; Baba, S.; Tomoda, K.; Yamashita, T.; Ikehara, S. Correlation between accelerated presbycusis and decreased immune functions. Exp. Gerontol. 2003, 38, 319–325. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; McMahon, C.M.; Burlutsky, G.; Spankovich, C.; Hood, L.J.; Mitchell, P. Dietary antioxidant intake is associated with the prevalence but not incidence of age-related hearing loss. J. Nutr. Health Aging 2011, 15, 896–900. [Google Scholar] [CrossRef]
- Michikawa, T.; Nishiwaki, Y.; Kikuchi, Y.; Nakano, M.; Iwasawa, S.; Asakura, K.; Milojevic, A.; Mizutari, K.; Saito, H.; Ishida, S.; et al. Gender-specific associations of vision and hearing impairments with adverse health outcomes in older Japanese: A population-based cohort study. BMC Geriatr. 2009, 9, 50. [Google Scholar] [CrossRef]
- Péneau, S.; Jeandel, C.; Déjardin, P.; Andreeva, V.A.; Hercberg, S.; Galan, P.; Kesse-Guyot, E.; SU.VI.MAX 2 Research Group. Intake of specific nutrients and foods and hearing level measured 13 years later. Br. J. Nutr. 2012, 109, 2079–2088. [Google Scholar] [CrossRef]
- Frenz, D.A.; Liu, W.; Cvekl, A.; Xie, Q.; Wassef, L.; Quadro, L.; Niederreither, K.; Maconochie, M.; Shanske, A. Retinoid signaling in inner ear development: A “goldilocks” phenomenon. Am. J. Med. Genet. A 2010, 152A, 2947–2961. [Google Scholar] [CrossRef]
- Romand, R.; Dollé, P.; Hashino, E. Retinoid signaling in inner ear development. J. Neurobiol. 2006, 66, 687–704. [Google Scholar] [CrossRef]
- Denke, M.A. Dietary retinol—A double-edged sword. JAMA 2002, 287, 102–104. [Google Scholar] [CrossRef]
- Le Prell, C.G.; Hughes, L.F.; Miller, J.M. Free radical scavengers vitamins A, C, and E plus magnesium reduce noise trauma. Free. Radic. Biol. Med. 2007, 42, 1454–1463. [Google Scholar] [CrossRef]
- Tesoriere, L.; Ciaccio, M.; Bongiorno, A.; Riccio, A.; Pintaudi, A.; Livrea, M. Antioxidant Activity of All-trans-retinol in Homogeneous Solution and in Phosphatidylcholine Liposomes. Arch. Biochem. Biophys. 1993, 307, 217–223. [Google Scholar] [CrossRef]
- Krinsky, N.I. Antioxidant functions of carotenoids. Free. Radic. Biol. Med. 1989, 7, 617–635. [Google Scholar] [CrossRef]
- Das, N.P. Effects of Vitamin A and Its Analogs on Nonenzymatic Lipid Peroxidation in Rat Brain Mitochondria. J. Neurochem. 1989, 52, 585–588. [Google Scholar] [CrossRef]
- Parker, R.S. Carotenoids in Human Blood and Tissues. J. Nutr. 1989, 119, 101–104. [Google Scholar] [CrossRef]
- Sena, L.A.; Chandel, N.S. Physiological Roles of Mitochondrial Reactive Oxygen Species. Mol. Cell 2012, 48, 158–167. [Google Scholar] [CrossRef]
- Chen, G.-D.; Li, M.; Tanaka, C.; Bielefeld, E.C.; Hu, B.-H.; Kermany, M.H.; Salvi, R.; Henderson, D. Aging outer hair cells (OHCs) in the Fischer 344 rat cochlea: Function and morphology. Hear. Res. 2009, 248, 39–47. [Google Scholar] [CrossRef]
- Seidman, M.D.; Ahmad, N.; Bai, U. Molecular mechanisms of age-related hearing loss. Ageing Res. Rev. 2002, 1, 331–343. [Google Scholar] [CrossRef]
- Kalinec, G.M.; Lomberk, G.; Urrutia, R.A.; Kalinec, F. Resolution of Cochlear Inflammation: Novel Target for Preventing or Ameliorating Drug-, Noise- and Age-related Hearing Loss. Front. Cell. Neurosci. 2017, 11, 192. [Google Scholar] [CrossRef]
- Jung, S.Y.; Kim, S.H.; Yeo, S.G. Association of Nutritional Factors with Hearing Loss. Nutrients 2019, 11, 307. [Google Scholar] [CrossRef]
| PTA (dB HL) | 4FA | HF | |
|---|---|---|---|
| Variables | Values | 44.0 ± 3.0 (38.2, 50.0) | 61.5 ± 3.0 (55.7, 67.3) |
| P-Value | P-Value | ||
| Age, years | 60.9 ± 0.2 (61.6, 61.2) | 0.007 | <0.001 |
| 50–59 | 2164 (53%) | ||
| 60–69 | 1742 (28%) | ||
| ≥70 | 1295 (19%) | ||
| Sex | |||
| Men | 2576 (52%) | 0.542 | 0.029 |
| Women | 2625 (48%) | ||
| Household income | |||
| Low | 1441 (25%) | 0.344 | 0.285 |
| Middle-low | 1356 (26%) | 0.386 | 0.322 |
| Middle-high | 1169 (24%) | 0.969 | 0.997 |
| High | 1235 (25%) | ||
| Education level | |||
| ≤Elementary school | 2174 (41%) | 0.004 | 0.002 |
| Middle school | 942 (19%) | 0.022 | 0.011 |
| High school | 1370 (27%) | 0.031 | 0.021 |
| ≥College | 715 (13%) | ||
| Occupation | |||
| Employed | 2865 (61%) | 0.720 | 0.727 |
| Unemployed | 2,336 (39%) | ||
| Body mass index (kg/m2) | 24.1 ± 0.1 (24.0, 24.2) | 0.116 | 0.128 |
| History of hypertension | 2057 (37%) | 0.500 | 0.482 |
| History of diabetes | 719 (14%) | 0.017 | 0.019 |
| History of noise exposure | 698 (15%) | 0.433 | 0.526 |
| Current smoking | 906 (21%) | 0.491 | 0.560 |
| Alcohol drinking within past year | 4019 (79%) | 0.530 | 0.595 |
| Food Groups | Values | 4FA | HF | ||
|---|---|---|---|---|---|
| Estimate | p * | Estimate | p * | ||
| (95% CI) | (95% CI) | ||||
| Cereals and grain products | 314.2 ± 2.9 (308.4, 320.0) | −0.02 (−0.06, 0.01) | 0.105 | −0.02 (−0.05, 0.01) | 0.122 |
| Potatoes and starches | 36.5 ± 2.2 (32.1, 40.8) | −0.01 (0.04, 0.02) | 0.516 | −0.01 (0.04, 0.02) | 0.533 |
| Sugars and sweets | 8.3 ± 0.3 (7.8, 8.9) | −0.20 (−0.66, 0.27) | 0.389 | −0.21 (−0.67, 0.25) | 0.355 |
| Legumes and their products | 40.6 ± 1.4 (37.9, 43.4) | 0.02 (−0.08, 0.11) | 0.719 | 0.02 (−0.08, 0.11) | 0.732 |
| Seeds and nuts | 6.1 ± 0.5 (5.1, 7.0) | −0.08 (−0.15, −0.01) | 0.013 | −0.07 (−0.14, 0.00) | 0.025 |
| Vegetables | 355.2 ± 5.4 (344.7, 365.7) | 0.01 (−0.01, 0.02) | 0.895 | 0.01 (0.00, 0.02) | 0.742 |
| Mushrooms | 4.1 ± 0.4 (3.3, 4.9) | 0.18 (−0.18, 0.54) | 0.317 | 0.18 (−0.18, 0.53) | 0.335 |
| Fruits | 198.2 ± 7.5 (183.6, 212.9) | −0.01 (−0.02, 0.00) | <0.001 | −0.01 (−0.02, 0.00) | <0.001 |
| Meats, poultry, and their products | 73.3 ± 2.5 (183.6, 212.9) | 0.03 (−0.02, 0.09) | 0.236 | 0.03 (−0.02, 0.09) | 0.262 |
| Eggs | 73.3 ± 2.5 (68.4, 78.2) | −0.05 (−0.18, 0.08) | 0.611 | −0.05 (−0.18, 0.07) | 0.594 |
| Fish and shellfish | 15.2 ± 0.6 (14.1, 16.3) | 0.04 (−0.05, 0.14) | 0.452 | 0.04 (−0.06, 0.13) | 0.481 |
| Seaweed | 54.8 ± 1.9 (51.1, 58.5) | −0.17 (−0.30, −0.04) | 0.001 | −0.16 (−0.29, −0.03) | 0.003 |
| Milk and dairy products | 61.3 ± 2.5 (56.3, 66.3) | −0.01 (−0.05, 0.02) | 0.508 | −0.01 (−0.05, 0.02) | 0.505 |
| Fats and oils | 6.4 ± 0.2 (6.1, 6.8) | 0.16 (−0.75, 1.08) | 0.865 | 0.15 (−0.76, 1.05) | 0.896 |
| Beverages | 218.0 ± 9.3 (199.7, 236.3) | 0.0 (−0.01, 0.02) | 0.710 | 0.0 (−0.01, 0.02) | 0.708 |
| Seasonings | 33.7 ± 0.7 (32.3, 35.1) | −0.07 (−0.20, 0.05) | 0.164 | −0.08 (−0.20, 0.04) | 0.143 |
| Nutrients | Values | 4FA | HF | ||
|---|---|---|---|---|---|
| Estimate | p * | Estimate | p * | ||
| (95% CI) | (95% CI) | ||||
| Macro-nutrients | |||||
| Total energy (kcal/day) | 1952.5 ± 15.3 (1922.4, 1982.6) | 0.01 (−0.02, 0.04) | 0.397 | 0.01 (−0.02, 0.04) | 0.433 |
| Carbohydrate (g/day) | 328.2 ± 2.6 (323.0, 333.4) | −0.11 (−0.22, 0.00) | 0.060 | −0.10 (−0.21, 0.04) | 0.072 |
| Protein (g/day) | 67.5 ± 0.7 (66.2, 68.8) | 0.09 (−0.26, 0.45) | 0.605 | 0.10 (−0.25, 0.45) | 0.582 |
| Fat (g/day) | 32.9 ± 0.5 (31.9, 33.9) | −0.25 (−0.64, 0.14) | 0.205 | −0.25 (−0.63, 0.14) | 0.208 |
| Micro-nutrients | |||||
| Crude fiber (g/day) | 8.1 ± 0.1 (7.8, 8.4) | −0.29 (−0.82, 0.23) | 0.271 | −0.26 (0.78, 0.26) | 0.323 |
| Ash (g/day) | 19.8 ± 0.2 (19.4, 20.3) | 0.22 (−0.74, 1.17) | 0.658 | 0.25 (−0.70, 1.20) | 0.599 |
| Calcium (mg/day) | 505.6 ± 6.7 (492.4, 518.8) | −0.01 (−0.03, 0.02) | 0.638 | −0.01 (−0.03, 0.02) | 0.630 |
| Iron (mg/day) | 15.9 ± 0.3 (15.2, 16.5) | −0.10 (−0.25, 0.06) | 0.214 | −0.10 (−0.25, 0.05) | 0.196 |
| Potassium (mg/day) | 3090.4 ± 33.1 (3205.4, 3155.4) | 0.00 (−0.01, 0.00) | 0.458 | 0.00 (−0.01, 0.00) | 0.497 |
| Sodium (mg/day) | 4829.1 ± 63.7 (4704.0, 4954.2) | 0.0 (0.0, 0.0) | 0.750 | 0.0 (0.0, 0.0) | 0.803 |
| Phosphorus (mg/day) | 1162.7 ± 9.4 (114.3, 1181.2) | 0.00 (−0.02, 0.03) | 0.727 | 0.01 (0.00, 0.02) | 0.748 |
| Retinol (μg/day) | 75.7 ± 3.9 (68.1, 83.4) | 0.01 (0.00, 0.02) | 0.279 | 0.01 (0.00, 0.02) | 0.177 |
| β-carotene (μg/day) | 4455.5 ± 138.1 (4184.1, 4726.8) | 0.0 (0.0, 0.0) | 0.102 | 0.0 (0.0, 0.0) | 0.050 |
| Vitamin A (μgRE/day) | 836.8 ± 24.6 (788.4, 885.2) | −0.01 (−0.01, 0.00) | 0.047 | −0.01 (−0.01, 0.00) | 0.024 |
| Thiamin (mg/day) | 1.3 ± 0.0 (1.2, 1.3) | −5.61 (−19.39, 8.17) | 0.424 | −4.77 (−18.27, 8.72) | 0.487 |
| Riboflavin (mg/day) | 1.1 ± 0.0 (1.1, 1.2) | 2.70 (−4.50, 9.90) | 0.462 | 2.32 (−4.75, 9.39) | 0.520 |
| Niacin (mg/day) | 16.4 ± 0.2 (16.0, 16.7) | 0.36 (−0.50, 1.22) | 0.414 | 0.26 (−0.59, 1.10) | 0.552 |
| Vitamin C (mg/day) | 109.8 ± 1.9 (106.0, 113.5) | 0.00 (−0.06, 0.06) | 0.974 | 0.00 (−0.07, 0.06) | 0.880 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, J.E.; Ahn, J.; Moon, I.J. Associations between Age-Related Hearing Loss and DietaryAssessment Using Data from Korean National Health andNutrition Examination Survey. Nutrients 2021, 13, 1230. https://doi.org/10.3390/nu13041230
Choi JE, Ahn J, Moon IJ. Associations between Age-Related Hearing Loss and DietaryAssessment Using Data from Korean National Health andNutrition Examination Survey. Nutrients. 2021; 13(4):1230. https://doi.org/10.3390/nu13041230
Chicago/Turabian StyleChoi, Ji Eun, Jungmin Ahn, and Il Joon Moon. 2021. "Associations between Age-Related Hearing Loss and DietaryAssessment Using Data from Korean National Health andNutrition Examination Survey" Nutrients 13, no. 4: 1230. https://doi.org/10.3390/nu13041230
APA StyleChoi, J. E., Ahn, J., & Moon, I. J. (2021). Associations between Age-Related Hearing Loss and DietaryAssessment Using Data from Korean National Health andNutrition Examination Survey. Nutrients, 13(4), 1230. https://doi.org/10.3390/nu13041230






