Seven Weeks of High-Dose Vitamin D Treatment Reduces the Need for Infliximab Dose-Escalation and Decreases Inflammatory Markers in Crohn’s Disease during One-Year Follow-Up
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Approval and Conduction
2.2. Former Intervention Study
- Infliximab and vitamin D3 (Ifx + VitD);
- Infliximab and placebo vitamin D3 (Ifx + placeboVitD);
- Placebo infliximab and vitamin D3 (placeboIfx + VitD);
- Placebo infliximab and placebo vitamin D3 (placeboIfx + placeboVitD).
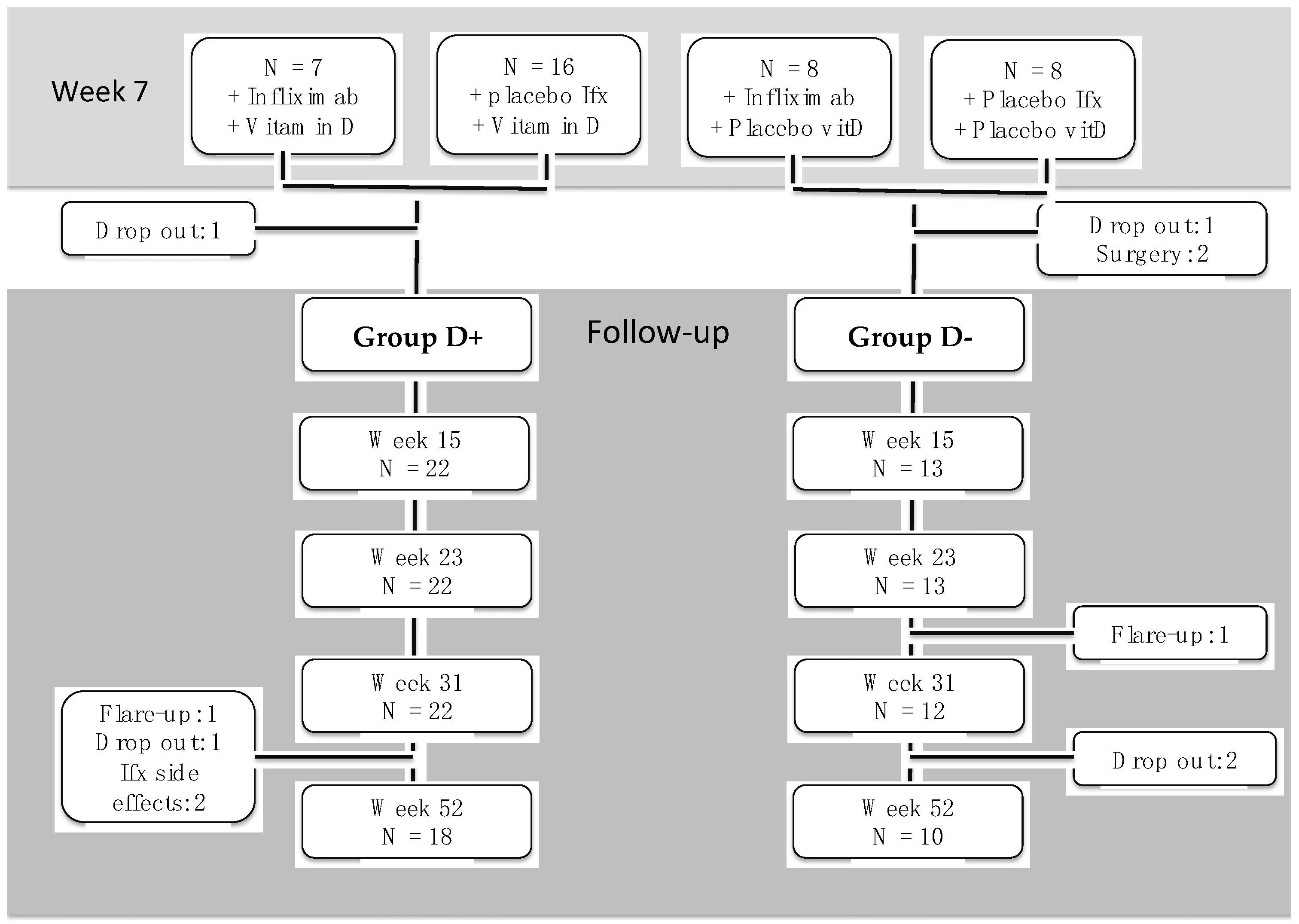
2.3. Follow-Up
- Group D+ (containing the Ifx+VitD and placIfx+vitD groups)
- Group D- (containing the Ifx+placVitD and placIfx+placVitD groups)
2.4. Vitamin D Safety Markers
2.5. Statistical Analyses
3. Results
3.1. Treatment Failures and Need for Dose-Escalation during Follow-Up
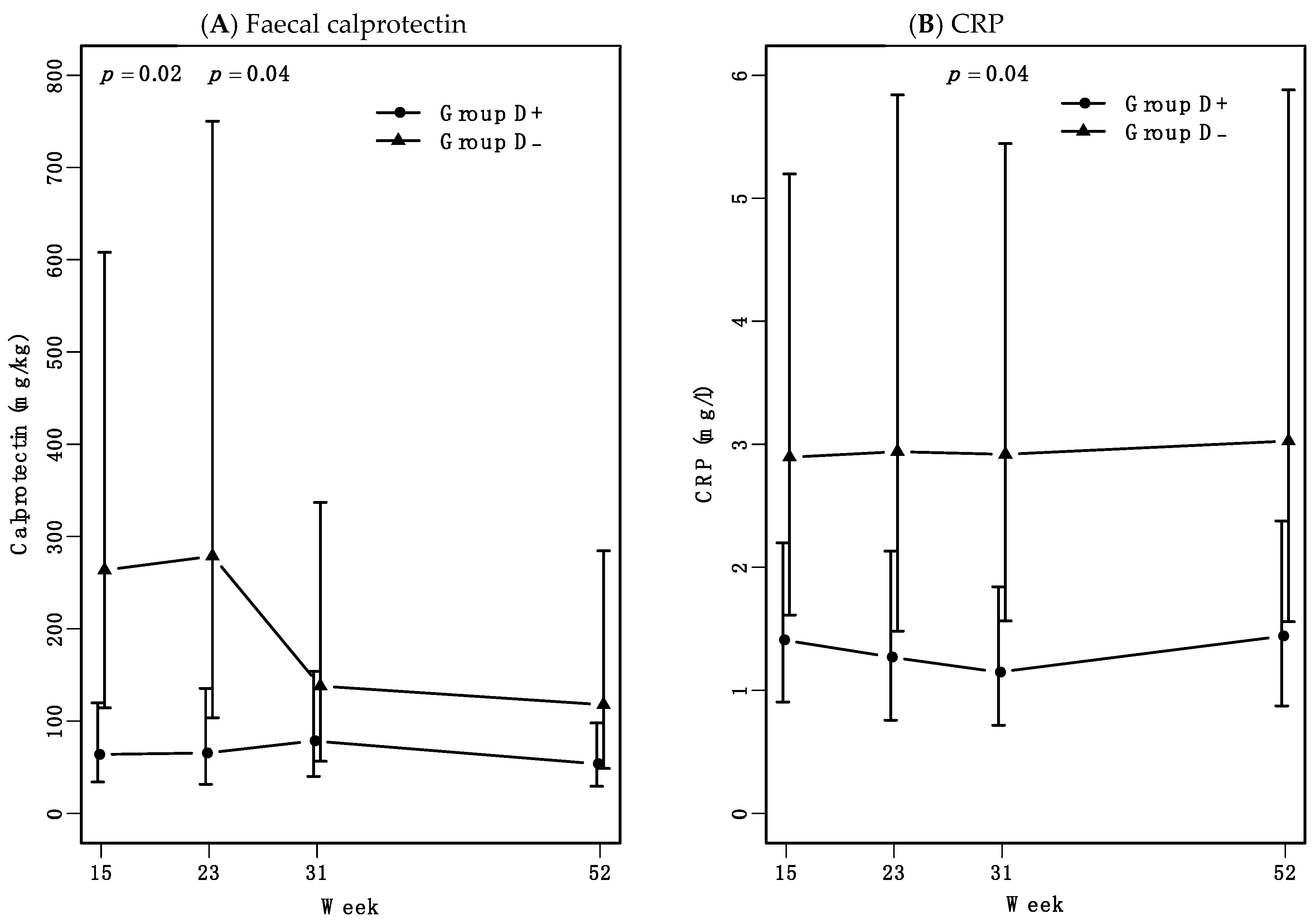
3.2. Initial High-Dose Vitamin D Treatment Reduces Faecal Calprotectin and CRP Levels
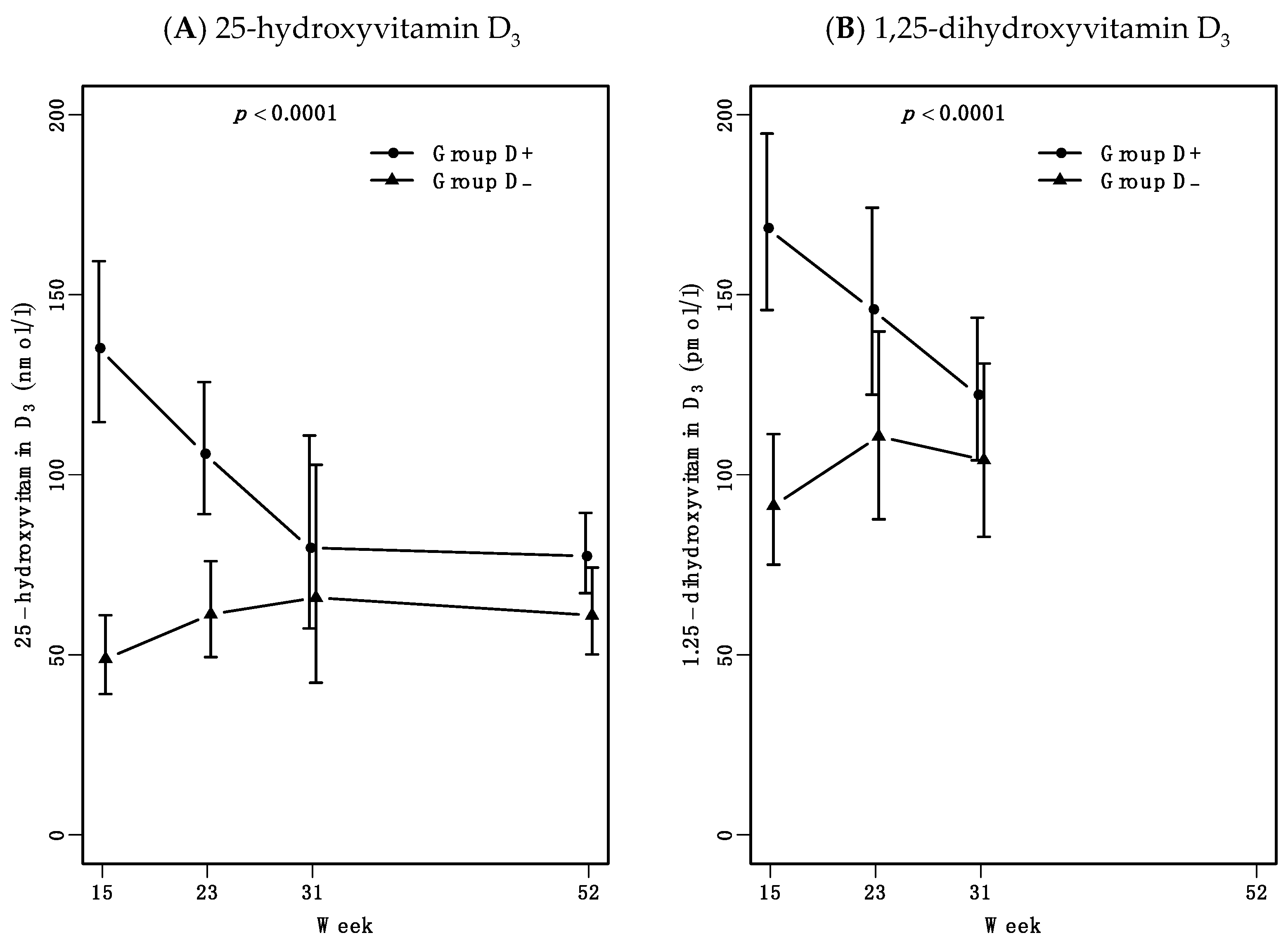
3.3. Increased Vitamin D Levels Are Maintained for 24 Weeks after 7 Weeks High-Dose Treatment
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Feagan, B.G.; Sandborn, W.J.; Gasink, C.; Jacobstein, D.; Lang, Y.; Friedman, J.R.; Blank, M.A.; Johanns, J.; Gao, L.L.; Miao, Y.; et al. Ustekinumab as Induction and Maintenance Therapy for Crohn’s Disease. N. Engl. J. Med. 2016, 375, 1946–1960. [Google Scholar] [CrossRef] [PubMed]
- Sandborn, W.J.; Feagan, B.G.; Rutgeerts, P.; Hanauer, S.; Colombel, J.F.; Sands, B.E.; Lukas, M.; Fedorak, R.N.; Lee, S.; Bressler, B.; et al. Vedolizumab as induction and maintenance therapy for Crohn’s disease. N. Engl. J. Med. 2013, 369, 711–721. [Google Scholar] [CrossRef] [Green Version]
- Sandborn, W.J.; Hanauer, S.B.; Rutgeerts, P.; Fedorak, R.N.; Lukas, M.; MacIntosh, D.G.; Panaccione, R.; Wolf, D.; Kent, J.D.; Bittle, B.; et al. Adalimumab for maintenance treatment of Crohn’s disease: Results of the CLASSIC II trial. Gut 2007, 56, 1232–1239. [Google Scholar] [CrossRef] [Green Version]
- Hanauer, S.B.; Feagan, B.G.; Lichtenstein, G.R.; Mayer, L.F.; Schreiber, S.; Colombel, J.F.; Rachmilewitz, D.; Wolf, D.C.; Olson, A.; Bao, W.; et al. Maintenance infliximab for Crohn’s disease: The ACCENT I randomised trial. Lancet 2002, 359, 1541–1549. [Google Scholar] [CrossRef]
- Kabbani, T.A.; Koutroubakis, I.E.; Schoen, R.E.; Ramos-Rivers, C.; Shah, N.; Swoger, J.; Regueiro, M.; Barrie, A.; Schwartz, M.; Hashash, J.G.; et al. Association of Vitamin D Level With Clinical Status in Inflammatory Bowel Disease: A 5-Year Longitudinal Study. Am. J. Gastroenterol. 2016, 111, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Bouillon, R.; Carmeliet, G.; Verlinden, L.; van Etten, E.E.; Verstuyf, A.; Luderer, H.F.; Lieben, L.; Mathieu, C.; Demay, M. Vitamin D and human health: Lessons from vitamin D receptor null mice. Endocr. Rev. 2008, 29, 726–776. [Google Scholar] [CrossRef] [PubMed]
- Bendix, M.; Dige, A.; Deleuran, B.; Dahlerup, J.F.; Peter Jorgensen, S.; Bartels, L.E.; Husted, L.B.; Harslof, T.; Langdahl, B.; Agnholt, J. Flow cytometry detection of vitamin D receptor changes during vitamin D treatment in Crohn’s disease. Clin. Exp. Immunol. 2015, 181, 19–28. [Google Scholar] [CrossRef] [Green Version]
- Dankers, W.; Colin, E.M.; van Hamburg, J.P.; Lubberts, E. Vitamin D in Autoimmunity: Molecular Mechanisms and Therapeutic Potential. Front. Immunol. 2016, 7, 697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakke, D.; Sun, J. Ancient Nuclear Receptor VDR With New Functions: Microbiome and Inflammation. Inflamm. Bowel Dis. 2018, 24, 1149–1154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jorgensen, S.P.; Agnholt, J.; Glerup, H.; Lyhne, S.; Villadsen, G.E.; Hvas, C.L.; Bartels, L.E.; Kelsen, J.; Christensen, L.A.; Dahlerup, J.F. Clinical trial: Vitamin D3 treatment in Crohn’s disease—A randomised double-blind placebo-controlled study. Aliment. Pharmacol. Ther. 2010, 32, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Frigstad, S.O.; Hoivik, M.; Jahnsen, J.; Dahl, S.R.; Cvancarova, M.; Grimstad, T.; Berset, I.P.; Huppertz-Hauss, G.; Hovde, O.; Torp, R.; et al. Vitamin D deficiency in inflammatory bowel disease: Prevalence and predictors in a Norwegian outpatient population. Scand. J. Gastroenterol. 2017, 52, 100–106. [Google Scholar] [CrossRef]
- Guzman-Prado, Y.; Samson, O.; Segal, J.P.; Limdi, J.K.; Hayee, B. Vitamin D Therapy in Adults With Inflammatory Bowel Disease: A Systematic Review and Meta-Analysis. Inflamm. Bowel Dis. 2020, 26, 1819–1830. [Google Scholar] [CrossRef]
- Yang, Y.; Cui, X.; Li, J.; Wang, H.; Li, Y.; Chen, Y.; Zhang, H. Clinical evaluation of vitamin D status and its relationship with disease activity and changes of intestinal immune function in patients with Crohn’s disease in the Chinese population. Scand. J. Gastroenterol. 2021, 56, 20–29. [Google Scholar] [CrossRef] [PubMed]
- Ham, M.; Longhi, M.S.; Lahiff, C.; Cheifetz, A.; Robson, S.; Moss, A.C. Vitamin D levels in adults with Crohn’s disease are responsive to disease activity and treatment. Inflamm. Bowel Dis. 2014, 20, 856–860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Winter, R.W.; Collins, E.; Cao, B.; Carrellas, M.; Crowell, A.M.; Korzenik, J.R. Higher 25-hydroxyvitamin D levels are associated with greater odds of remission with anti-tumour necrosis factor-alpha medications among patients with inflammatory bowel diseases. Aliment. Pharmacol. Ther. 2017, 45, 653–659. [Google Scholar] [CrossRef] [Green Version]
- Zator, Z.A.; Cantu, S.M.; Konijeti, G.G.; Nguyen, D.D.; Sauk, J.; Yajnik, V.; Ananthakrishnan, A.N. Pretreatment 25-Hydroxyvitamin D Levels and Durability of Anti-Tumor Necrosis Factor-alpha Therapy in Inflammatory Bowel Diseases. JPEN. J. Parenter. Enteral Nutr. 2014, 38, 385–391. [Google Scholar] [CrossRef]
- Mechie, N.C.; Mavropoulou, E.; Ellenrieder, V.; Kunsch, S.; Cameron, S.; Amanzada, A. Distinct Association of Serum Vitamin D Concentration with Disease Activity and Trough Levels of Infliximab and Adalimumab during Inflammatory Bowel Disease Treatment. Digestion 2020, 101, 761–770. [Google Scholar] [CrossRef]
- Bendix, M.; Dige, A.; Jørgensen, S.P.; Dahlerup, J.F.; Bibby, B.M.; Deleuran, B.; Agnholt, J. Decrease in Mucosal IL17A, IFNγ and IL10 Expressions in Active Crohn’s Disease Patients Treated with High-Dose Vitamin D Alone or Combined with Infliximab. Nutrients 2020, 12, 3699. [Google Scholar] [CrossRef] [PubMed]
- Baumgart, D.C.; Sandborn, W.J. Crohn’s disease. Lancet 2012, 380, 1590–1605. [Google Scholar] [CrossRef] [Green Version]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [Green Version]
- Harvey, R.F.; Bradshaw, J.M. A simple index of Crohn’s-disease activity. Lancet 1980, 1, 514. [Google Scholar] [CrossRef]
- Schoepfer, A.M.; Beglinger, C.; Straumann, A.; Trummler, M.; Vavricka, S.R.; Bruegger, L.E.; Seibold, F. Fecal calprotectin correlates more closely with the Simple Endoscopic Score for Crohn’s disease (SES-CD) than CRP, blood leukocytes, and the CDAI. Am. J. Gastroenterol. 2010, 105, 162–169. [Google Scholar] [CrossRef] [Green Version]
- Wright, E.K.; Kamm, M.A.; De Cruz, P.; Hamilton, A.L.; Ritchie, K.J.; Keenan, J.I.; Leach, S.; Burgess, L.; Aitchison, A.; Gorelik, A.; et al. Comparison of Fecal Inflammatory Markers in Crohn’s Disease. Inflamm. Bowel Dis. 2016, 22, 1086–1094. [Google Scholar] [CrossRef] [Green Version]
- Mumolo, M.G.; Bertani, L.; Ceccarelli, L.; Laino, G.; Di Fluri, G.; Albano, E.; Tapete, G.; Costa, F. From bench to bedside: Fecal calprotectin in inflammatory bowel diseases clinical setting. World J. Gastroenterol. 2018, 24, 3681–3694. [Google Scholar] [CrossRef]
- Bafutto, M.; Oliveira, E.; Filho, J. Use of Vitamin D With Anti-Tumor Necrosis Factor Therapy for Crohn’s Disease. Gastroenterol Res. 2020, 13, 101–106. [Google Scholar] [CrossRef]
- Mechie, N.; Mavropoulou, E.; Ellenrieder, V.; Petzold, G.; Kunsch, S.; Neesse, A.; Amanzada, A. Serum vitamin D but not zinc levels are associated with different disease activity status in patients with inflammatory bowel disease. Medicine 2019, 98, e15172. [Google Scholar] [CrossRef]
- De Cruz, P.; Kamm, M.A.; Prideaux, L.; Allen, P.B.; Moore, G. Mucosal healing in Crohn’s disease: A systematic review. Inflamm. Bowel Dis. 2013, 19, 429–444. [Google Scholar] [CrossRef] [PubMed]
- Valvano, M.; Magistroni, M.; Mancusi, A.; D’Ascenzo, D.; Longo, S.; Stefanelli, G.; Vernia, F.; Viscido, A.; Necozione, S.; Latella, G. The Usefulness of Serum Vitamin D Levels in the Assessment of IBD Activity and Response to Biologics. Nutrients 2021, 13, 323. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Chen, Y.; Golan, M.A.; Annunziata, M.L.; Du, J.; Dougherty, U.; Kong, J.; Musch, M.; Huang, Y.; Pekow, J.; et al. Intestinal epithelial vitamin D receptor signaling inhibits experimental colitis. J. Clin. Investig. 2013, 123, 3983–3996. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Du, J.; Zhang, Z.; Liu, T.; Shi, Y.; Ge, X.; Li, Y.C. MicroRNA-346 mediates tumor necrosis factor alpha-induced downregulation of gut epithelial vitamin D receptor in inflammatory bowel diseases. Inflamm. Bowel Dis. 2014, 20, 1910–1918. [Google Scholar] [CrossRef] [PubMed]
- Gibson, P.R. Increased gut permeability in Crohn’s disease: Is TNF the link? Gut 2004, 53, 1724–1725. [Google Scholar] [CrossRef] [Green Version]
- Edelblum, K.L.; Yan, F.; Yamaoka, T.; Polk, D.B. Regulation of apoptosis during homeostasis and disease in the intestinal epithelium. Inflamm. Bowel Dis. 2006, 12, 413–424. [Google Scholar] [CrossRef] [PubMed]
- Schaffler, H.; Herlemann, D.P.; Klinitzke, P.; Berlin, P.; Kreikemeyer, B.; Jaster, R.; Lamprecht, G. Vitamin D administration leads to a shift of the intestinal bacterial composition in Crohn’s disease patients, but not in healthy controls. J. Dig. Dis. 2018, 19, 225–234. [Google Scholar] [CrossRef] [PubMed]
- Cantorna, M.T.; Lin, Y.D.; Arora, J.; Bora, S.; Tian, Y.; Nichols, R.G.; Patterson, A.D. Vitamin D Regulates the Microbiota to Control the Numbers of RORgammat/FoxP3+ Regulatory T Cells in the Colon. Front. Immunol. 2019, 10, 1772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Group D+ | Group D- | p-Value | |
|---|---|---|---|
| Patients in treatment groups, n | 22 | 13 | |
| Relapse and infliximab stopped, n | 1 (5%) | 1 (8%) | 0.7 |
| Infliximab dose escalation, n * | 3 (14%) | 6 (46%) | 0.05 |
| Infliximab dose escalation week 15, n | 0 | 1 (8%) | 0.4 |
| Infliximab dose escalation week 23, n | 0 | 2 (15%) | 0.1 |
| Infliximab dose escalation week 31, n | 0 | 2 (15%) | 0.1 |
| Infliximab dose escalation week 32–52, n | 3 (14%) | 1 (8%) | 1.0 |
| Oral prednisolone 50 mg added week 15 | 0 | 1 (8%) | 0.4 |
| Stop infliximab patients in remission (patient wish), n | 1 (5%) | 2 (15%) | 0.3 |
| Stop infliximab due to side effects, n | 2 (9%) | 0 | 0.5 |
| Azathioprine users week 23, n | 18 (82%) | 13 (100%) | 0.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bendix, M.; Dige, A.; Jørgensen, S.P.; Dahlerup, J.F.; Bibby, B.M.; Deleuran, B.; Agnholt, J. Seven Weeks of High-Dose Vitamin D Treatment Reduces the Need for Infliximab Dose-Escalation and Decreases Inflammatory Markers in Crohn’s Disease during One-Year Follow-Up. Nutrients 2021, 13, 1083. https://doi.org/10.3390/nu13041083
Bendix M, Dige A, Jørgensen SP, Dahlerup JF, Bibby BM, Deleuran B, Agnholt J. Seven Weeks of High-Dose Vitamin D Treatment Reduces the Need for Infliximab Dose-Escalation and Decreases Inflammatory Markers in Crohn’s Disease during One-Year Follow-Up. Nutrients. 2021; 13(4):1083. https://doi.org/10.3390/nu13041083
Chicago/Turabian StyleBendix, Mia, Anders Dige, Søren Peter Jørgensen, Jens Frederik Dahlerup, Bo Martin Bibby, Bent Deleuran, and Jørgen Agnholt. 2021. "Seven Weeks of High-Dose Vitamin D Treatment Reduces the Need for Infliximab Dose-Escalation and Decreases Inflammatory Markers in Crohn’s Disease during One-Year Follow-Up" Nutrients 13, no. 4: 1083. https://doi.org/10.3390/nu13041083
APA StyleBendix, M., Dige, A., Jørgensen, S. P., Dahlerup, J. F., Bibby, B. M., Deleuran, B., & Agnholt, J. (2021). Seven Weeks of High-Dose Vitamin D Treatment Reduces the Need for Infliximab Dose-Escalation and Decreases Inflammatory Markers in Crohn’s Disease during One-Year Follow-Up. Nutrients, 13(4), 1083. https://doi.org/10.3390/nu13041083







