Abstract
Lifestyle interventions to reduce second stroke risk are complex. For effective translation into practice, interventions must be specific to end-user needs and described in detail for replication. This study used an Integrated Knowledge Translation (IKT) approach and the Template for Intervention Description and Replication (TIDieR) checklist to co-design and describe a telehealth-delivered diet program for stroke survivors. Stroke survivors and carers (n = 6), specialist dietitians (n = 6) and an IKT research team (n = 8) participated in a 4-phase co-design process. Phase 1: the IKT team developed the research questions, and identified essential program elements and workshop strategies for effective co-design. Phase 2: Participant co-design workshops used persona and journey mapping to create user profiles to identify barriers and essential program elements. Phase 3: The IKT team mapped Phase 2 data to the TIDieR checklist and developed the intervention prototype. Phase 4: Co-design workshops were conducted to refine the prototype for trial. Rigorous IKT co-design fundamentally influenced intervention development. Modifications to the protocol based on participant input included ensuring that all resources were accessible to people with aphasia, an additional support framework and resources specific to outcome of stroke. The feasibility and safety of this intervention is currently being pilot tested (randomised controlled trial; 2019/ETH11533, ACTRN12620000189921).
1. Introduction
Knowledge translation is the process of moving research into clinical practice. A commonly cited research flaw is the 17 years that this process takes [1,2,3], that is if the research is implemented at all. It has been estimated that $85 billion of research funding each year is lost due to lack of translation into clinical practice [4,5]. The Stroke Recovery and Rehabilitation Roundtable (SRRR) was assembled to provide direction to move the field of rehabilitation research forward. The SRRR identified a lack of rigorous intervention development and detailed reporting as barriers to successful translation of research findings into practice [6]. To improve research impact, they endorsed the use of the Template for Intervention Description and Replication (TIDieR) checklist as a development and reporting guide to accurately report complex interventions, enhancing reproducibility and validity [7]. The SRRR also recommends using a co-design framework to ensure feasibility in a real-world context.
Both stroke and recurrent stroke are largely preventable because their risk factors can be modified by lifestyle changes. The INTERSTROKE study found that 10 potentially modifiable lifestyle factors were responsible for over 90% population attributable risk (PAR) [8]. Poor diet quality is one of those factors. However, the majority of the remaining nine factors (cardiac causes, waist-to-hip ratio, hypertension, psychosocial factors, diabetes, apolipoprotein ratio-ApoB/ApoA1) also have the potential to be addressed by improving diet quality.
Several dietary patterns have been shown to lower risk factors associated with stroke in clinical trials-Mediterranean diet [9,10], Nordic diet [11,12] and Dietary Approaches to Stop Hypertension (DASH) [13,14] diets. The only dietary pattern that has been shown to be effective in reducing actual risk (not just reduction in risk factors) is the Mediterranean diet. The PREDIMED study showed a Mediterranean diet supplemented with olive oil showed a 35% decrease in risk for primary stroke and the same diet supplemented with nuts showed a 45% decrease [15]. Due to its variety and adaptability, high adherence to a Mediterranean diet pattern has been consistently shown to be possible in countries outside of the Mediterranean [16,17,18,19].
We developed the AusMed diet program specifically for older Australians. The core food components of this are a high intake of fruit, vegetables, legumes, nuts and wholegrains; moderate intake of fish, poultry and dairy foods; limited intake of commercial sweets, red and processed meats; olive oil as the main source of culinary fats [20]. It was tested in a small population and the 2 weeks program was found to be acceptable to participants and resulted in high adherence (mean Mediterranean diet score [21] 9.6 ± 2.0 out of 14, where <5 is considered low, 6–8 moderate and ≥9 high adherence) [20]. It takes time for any change in diet to lower risk factors and improve health. Adherence needs to be long lasting to be meaningful and for that, interventions need to be created with the specific needs of their end users at the forefront.
Integrated Knowledge Translation (IKT) has at its core, a collaborative approach, where all who might benefit from an intervention (participants, researchers, health care providers and clinicians) are called knowledge users and are equally involved in its design from inception [22]. IKT has been shown to increase the applicability and impact of the research developed using its framework [23]. It has also been identified as a method to effect change in stroke care by bridging the gap between research and clinical practice [24]. A recent rapid review of co-design research found that while co-design is frequently used, there is a lack of standard terminology, the method is seldom described in detail and outcomes are not well defined [25].
Interventions developed to prevent recurrent stroke by changes in lifestyle are complex and have multiple components. Complex interventions are often not being described in enough detail to be of use in clinical practice [7]. Numerous public health frameworks have been proposed to address this need for better intervention development and reporting—for example, the Medical Research Council (MRC) [26] guidelines for complex interventions, the Consolidated Standards of Reporting Trials (CONSORT) Statement [27] and the TIDieR checklist [7]. Research tells us that few interventions are meeting the guidelines set out by these frameworks [28,29,30] and that has not improved over time [31]. In most cases, the research setting, recipient, provider and schedule of interventions are well described, but detailed descriptions of the actual content of the interventions and supporting materials or equipment used are often lacking [28,29,30,32]. Frequently missing were adequate reporting of intervention development, content and materials and treatment fidelity [31].
The IKT co-design framework has been used to develop a supervised physical activity intervention delivered via telehealth for stroke survivors (i-Rebound after Stroke—Let’s get Moving) [33]. The dietary intervention developed by this study (i-Rebound after Stroke—Eat for Health) is being piloted alongside this as part of the ENAbLE pilot trial to reduce secondary stroke (2019/ETH11533 and ACTRN12620000189921). The aim of this study was to develop a Mediterranean dietary intervention (using the AusMed diet as a framework) for the prevention of second stroke using the TIDieR checklist for reporting (including the National Institute of Health Treatment Fidelity Framework [34]) and IKT co-design to improve its replicability and feasibility.
2. Materials and Methods
This study used the IKT methodology (conducted over 4 phases). The study process is described by Figure 1. Workshops were conducted to codesign a Mediterranean dietary intervention to be delivered via telehealth for secondary stroke risk reduction. This approach complies with both the SRRR roundtable recommendations and the TIDieR framework for complex intervention reporting, incorporating the NIH Treatment Fidelity Framework at all stages of development.
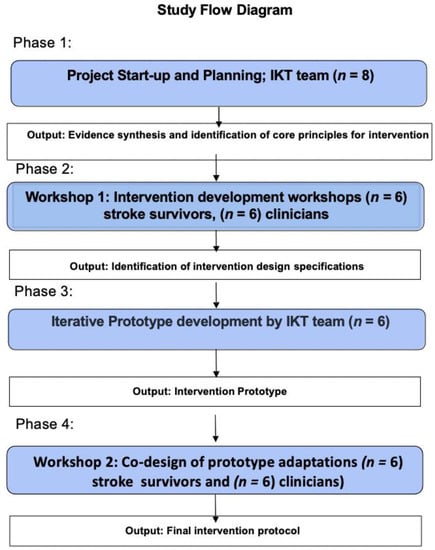
Figure 1.
Flow chart of Integrated Knowledge Translation (IKT) study processes.
2.1. Recruitment
The IKT research team (n = 8) was already in place. Purposive recruitment was utilised to ensure a breadth of experience across knowledge users. Participants were identified as possible end users of the program (stroke survivors, carers and dietitians responsible for program delivery) and were recruited from a variety of sources; from the initial ENAbLE trial codesign process (n = 4), utilizing professional networks (n = 6) and through word of mouth (n = 2). Written and informed consent was obtained from all participants.
2.2. Phase 1: Project Start up and Planning
The IKT research team consisted of researchers (n = 3), clinicians with experience in stroke rehabilitation and/or telehealth delivery (n = 3) and stroke survivors (n = 2). As the physical activity intervention to be trialled alongside this dietary intervention was already in place, the timeframe from Phase 1 to finalised intervention prototype was identified as 8 weeks. An iterative collaborative process was used to identify the essential elements of the program and define the research questions. Strategies to engage participants in the co-design process were identified to capture a breadth of information applicable to a variety of stroke outcomes.
2.3. Phase 2: Workshop 1. Intervention Development
A series of stakeholder workshops were held with stroke survivors (n = 4), carers (n = 2) and specialist disability dietitians (n = 6). Stroke survivor and clinician workshops were held separately. Researchers used persona [35] and journey mapping [36] processes to identify essential items to the intervention design. Persona and journey mapping are tools used frequently in marketing to explore a user’s experience of a program or product in order to improve it, similar to a case study [37]. Two personas of stroke survivors were developed by the research team using data derived from the Australian Institute of Health and Welfare (AIHW) [38] to represent common stroke-related impairments that may affect the ability to adhere to a diet program (Table S1). Persona characteristics were intentionally varied (different genders, employment status, support structures and stroke outcomes). The facilitator used a semi-structured question guide to describe the AusMed diet program in its original format, which included a resource booklet with Mediterranean diet education, recipes and a checklist, a 2 h group information session and a 2 weeks example meal plan and shopping list. Workshop facilitators mapped the journey of each persona through the program. Participants were asked to identify how the persona might complete the program, to describe enablers and barriers for each persona and identify essential elements needed for effective and feasible diet program adherence. Each workshop was audio recorded, transcribed and thematically analysed. Analysis was emailed to participants for member checking which gives them the opportunity to review and clarify any information or give additional data.
2.4. Phase 3: Prototype Development Using TIDieR
Essential elements and detailed data collected from the workshops were mapped to the 12 items of the TIDieR checklist (Table 1) and used to develop the intervention manual, resources and components necessary for the intervention prototype. The research team then used the NIH Treatment Fidelity Framework to develop a fidelity checklist to ensure the intervention will be delivered as intended.

Table 1.
Research Questions Resulting from Phase 1.
2.5. Phase 4: Co-Design of Prototype Adaptations
Another series of workshops was then held with knowledge users (n = 6 stroke survivors and carers and n = 6 dietitians) to refine and adapt the intervention prototype. Prior to the workshop, the prototype and resources were emailed to participants to allow time for formulation of feedback. The workshop facilitator summarised the thematic analysis from workshop 1 and presented the outcome developed as a result. A physical copy of the emailed prototype resources was also presented to participants. Participants feedback was sought after each section of the presentation. Each workshop was audio recorded, transcribed and thematically analysed by a member of the research team as per the six-phase guide outlined by Braun and Clarke [39]. Analysis was then emailed to participants for member checking. The data collected were then used to finalise the TIDieR checklist and intervention materials ready for trial.
3. Results
3.1. Phase 1: Project Start up and Planning
Iterative collaboration meetings, a literature search and prior research by the IKT team identified essential elements to be included in the intervention protocol, the research questions to be answered by the Phase 2 co-design workshops and strategies to facilitate co-design.
The key elements identified were the use of the Mediterranean diet pattern (modelled on the AusMed framework), individual dietary counselling sessions, delivery by telehealth using individual video calls and embedding behaviour change techniques within each session using the Behaviour Change Wheel [40].
To capture a broad scope of data, two personas were developed to represent differing stroke outcomes, gender, support needs and demographics. The research questions to be answered by the codesign process are displayed in the table below (Table 1).
3.2. Phase 2: Workshop 1 Intervention Development
The IKT research team identified the co-design of the program elements and structure as the goal of the workshops with the data collected to be mapped to the TIDieR checklist to ensure complete intervention development. The use of journey mapping gave the workshops focus and direction and enhanced participant engagement with the codesign process. The variety of characteristics of the developed persona meant that barriers and enablers described were more broadly applicable than would otherwise have been captured by the experience of workshop participants.
Thematic analysis of workshop transcripts (Table 2) identified five elements that stroke survivors and dietitians found were key to the successful delivery of the AusMed diet program: inclusion of a detailed initial assessment, accessible resource design, development of resources specific to stroke outcome (such as one-handed food preparation, fatigue strategies, convenience meals), creation of a support framework (including optional group and peer support, text messages and reminders) and resources to support effective telehealth delivery.

Table 2.
Thematic Analysis of Workshop 1 (n = 12).
3.3. Phase 3: Intervention Prototype Development
The IKT team combined initial essential elements identified in Phase 1 with data from the Phase 2 co-design workshops and mapped this to the TIDieR checklist, thereby developing the intervention protocol and supporting resources. A complete description of the intervention developed by the IKT co-design process is outlined in the TIDieR checklist (Table 3).

Table 3.
TIDieR Checklist.
As a result of the co-design process, there are six essential elements to applying the i-Rebound after Stroke-Eat for Health program;
- The end goal of the program is for participants to achieve high adherence to a Mediterranean dietary pattern.
- Delivery is via individual/tailored diet counselling sessions.
- Sessions are to be delivered by video call to the participant’s own home.
- Each session must include embedded behaviour change strategies to support participants’ long-term adherence.
- All resources must be accessible for all (including in a language and a format that are aphasia friendly) and be able to be tailored to different stroke-related impairments.
- The intervention must include optional, embedded support systems such as peer support groups and/or text messages to facilitate behaviour change.
The intervention protocol, which will be delivered by an Accredited Practicing Dietitian (APD), consists of 10 individual diet counselling sessions to be delivered over 6 months. The first two sessions (weekly) focus on detailed initial assessment, program education, current diet history and assessment of barriers to change. The next five sessions are fortnightly and aim to achieve high adherence to a Mediterranean diet pattern. All sessions have the same format; revision of previous goals, Mediterranean diet score calculation, identification of new goals, assessment of barriers, identification of behaviour change strategies to enable and resources required to support. The final three sessions are delivered monthly and aim for self-efficacy in maintaining dietary change.
Supporting resources were developed as a result of the co-design process utilising aphasia-friendly design guidelines with appropriate font size and type, spacing and visual representation; pictures and icons [53]. A participant program manual and resources were developed to support a variety of stroke outcomes and address barriers to change; fatigue, convenience meals, one-handed food preparation, templates for meal plans, shopping lists, goals setting. Also developed was a recipe book and a practitioner training manual including resources for effective communication via video call, technology instructions, behaviour change taxonomy to be used in session and a telehealth session manual to be used by the therapist for each participant—this includes session outlines, a validated 14-point Mediterranean diet score [54] to assess adherence and fidelity checklist (Table 4). The therapist and telehealth manuals will be used to train clinicians in delivery of the program to ensure fidelity.

Table 4.
NIH Treatment Fidelity Plan.
Co-design identified the need for resources to be specific to the needs of the individual. Additional resources will be developed according to need as identified within individual sessions. This will result in a bank of resources being compiled over the duration of the pilot trial for use in a larger roll out. All support sessions and text messaging are optional and will be tailored to individual need or request.
As part of the TIDieR framework, a fidelity assessment plan was produced using the National Institute of Health’s Treatment Fidelity Framework [34]. Fidelity is measured by the five domains of this framework; 1. design, 2. training, 3. delivery, 4. receipt and 5. enactment. (See Table 2 for a detailed description). Data will be collected and managed using Research Electronic Data Capture (REDCap) [56,57] hosted at the Hunter Medical Research Institute (HMRI).
3.4. Phase 4: Co-Design of Intervention Prototype Adaptations
The intervention prototype was presented for participant feedback at Phase 4 co-design workshops. All workshop participants agreed that the intervention prototype developed as a result of the co-design workshops was acceptable and feasible.
“Yeah. It’s easy to read, easy to understand. Excellent. Yeah. You’ve really got everything in there.” E.
“You’ve got some lovely, very simple recipes. Excellent resources applicable for a variety of clients. That actually looks deliverable…The best I’ve seen for a while. You’ve got me. It’s a really, really good book.” J.
Thematic analysis of transcribed interviews revealed three overarching themes to refine the intervention. The first was development of carer-specific resources; several clinicians suggested resources created to allow carers to facilitate opportunities for independence would be beneficial.
“You can often be more prescriptive with carers. Outline specific duties that you want them to undertake that will facilitate the behaviour change for the client. Things like kitchen preparation set up or meal preparation where the client will finish the meal off. Or teaching them to facilitate the meal planning and shopping with the client.” L.
The second was demonstration; stroke survivors re-iterated the importance of seeing themselves or someone like them demonstrate preparation skills. Video was seen as an important tool for self-efficacy.
“I think that at the very least, having videos by us that teach people how to do those things is great. If you made little demonstration videos, like of various things you need to do like prep set up or, or even for one recipe and now you’ve got to chop, you’ve got to blend, you got to stir it. This is how you can make it easier.” M.
The final theme identified was for support sessions to be themed and guided to allow time for participants to prepare their input for peer support. All participants emphasised that peer support and sharing their experience was an important method of skill building for them.
“Make them as useful as you can. You know, you’re your own best resource and your each other’s own best resource as well. Maybe like, you choose a theme for each session and then bring your best ideas for the group for that session. So, the theme of this week’s group session is going to be, how you open packets and cans.” R.
The final prototype was then revised by the IKT team to reflect participant adaptations with some ideas parked for future use due to time constraints. The intervention is currently in the pilot trial phase, with ongoing refinement.
4. Discussion
Lifestyle interventions to lower second stroke risk are inherently complex. Understanding their development process is essential for replication and translation into clinical practice yet rarely is this reported with enough detail to be effective [28,29,30,31]. The aim of this study was to use the IKT framework and TIDieR checklist with the NIH Treatment fidelity Framework to co-design and describe a Mediterranean dietary intervention specifically for stroke survivors. While the Stroke Recovery and Rehabilitation Roundtable (SRRR) recommends the use of the TIDieR checklist in reporting interventions [6], this study also used it as a framework to guide development. The checklist gave clear justification for and reliable description of all intervention components and ensured complete intervention development from the outset. Using both IKT and the TIDieR checklist allowed for a complete description of the diet program methodology using standardised terminology addressing a cited limitation of co-design research [25].
Both the SRRR and a recent Cochrane review identify co-design as necessary for intervention acceptability and feasibility both in trial and clinical practice [6,58]. Enablers to successful co-design using IKT include having multiple opportunities for interaction, a phased approach for iterative development, formalised processes and the establishment of partnership early in the process [59]. This intervention development timeframe was brief (8 weeks) in comparison to the same IKT process undertaken by the research team to develop the physical activity intervention (i-Rebound after Stroke-Let’s Get Moving) which took 7 months. However, a number of stakeholders participated across both studies [33]. As a result, relationships were already formed, and phased study processes formalised. Strategies were put in place to streamline development; the AusMed diet program was used as a framework for redevelopment rather than to design a completely new intervention, separate workshops were held for clinicians and stroke survivors, and persona and journey mapping used to guide those workshops. These strategies improved workshop focus and augmented the development of an intervention acceptable to both clinicians and stroke survivors within the required timeframe.
The IKT process was used to finalise an intervention prototype with six key elements. While not all of these elements are unique to the stroke survivor population, collectively, they address barriers to dietary change and intervention delivery that are unique to stroke survivors. Stroke causes a greater range of disabilities than any other condition. Debilitating fatigue affects up to 75% of stroke survivors [60,61], cognitive difficulty such as aphasia affects 40–50% [62,63] and physical disability up to 65% [64,65]. These outcomes make access and participation in standard diet programs to lower the risk of second stroke difficult [66]. Co-design highlighted key program elements for inclusion that address these barriers; delivery by telehealth to improve access, tailoring of the intervention by individual diet counselling and resource creation, embedding behaviour change techniques using the Behaviour Change Wheel [52] and support within the intervention processes and resources. A recent Cochrane review on self-management programs for quality of life in people with stroke found that in order to create effective programs that are meaningful and acceptable to people with stroke, stroke survivors need to be involved in their design, the interventions needs to be tailored to their ability and delivered by professionals and peers that are experts in stroke and its consequences [58].
Unique to this study is the development of accessible intervention components and resources designed specifically to address the barriers created by stroke outcome. Aphasia can significantly alter the understanding of language, speech and writing ability [62,63,67]. Lifestyle intervention programs often rely on written resources to enable participant adherence. In order for these to be useful to someone with aphasia, specific design guidelines must be used to ensure readability [53]. Materials must be designed using easily readable sans serif fonts (minimum 12–14 pts), 1.5–2 spacing, use of images and lots of white space on the page. Program resource materials were created to support participants with a variety of stroke outcomes (such as strategies for fatigue, adaptations for cooking with muscle weakness or paralysis, flavour without salt). All were developed using these design principles to improve accessibility and acceptability.
The SRRR recommends adoption of a fidelity framework in the intervention design phase to help identify which aspects are most effective and which need refining [6]. Using the National Institute of Health Fidelity Framework to design a fidelity plan has given this intervention a detailed description of delivery and audit processes to ensure it can be done well in any setting by any qualified therapist. The fidelity plan includes not only session data and behaviour change coding but also recruitment rate and retention. Treatment fidelity ensures that any outcome is a direct result of the intervention which adds to scientific confidence.
Interventions with high treatment fidelity have also been shown to have improved treatment outcomes [68]. Embedding measurement of fidelity from the beginning will improve study processes and translates into a rigorously designed trial.
5. Limitations
Several limitations must be noted when interpreting the study process described here. As stated, the timeframe between intervention development and finalisation for pilot trial was 8 weeks. This study shows that it is possible to co-design an intervention using the IKT process within that short time frame. However, time constraints limited the number of stakeholders involved and resulted in more structured workshop sessions, which may have resulted in fewer ideas from outside of that structure. Further workshops involving more participants, across a wider range of allied health disciplines (such as occupational and speech therapy) and representative of a variety of stroke outcomes, would have been of benefit to confirm the key themes and essential elements of the intervention. The facilitator of the workshops was also responsible for data analysis, which was valuable in terms of intervention development, as they had participated in discussions, but may provide a source of bias. Including independent data analysis would help prevent this.
6. Conclusions
Promoting lifestyle interventions aimed at lowering risk factors for stroke seems intuitively appropriate yet few stroke survivors are adhering to them [69]. A rigorously developed intervention, specific to the needs of both the people who will benefit from AND the people who will deliver the intervention will make long-term adherence more likely in a real-world setting. IKT uses the real-world experience and contexts of knowledge users to ensure that intervention design is both effective and achievable in practice. Creating interventions that are acceptable, feasible and replicable could improve translation and decrease money lost from underuse of interventions [4,5].
A recent Cochrane review looking at primary and secondary prevention of cardiovascular disease by the Mediterranean dietary pattern found that while observational studies confirmed the benefits, there was a distinct lack of trial evidence [70]. The intervention developed by this study is currently in pilot trial, testing feasibility and safety, with a process evaluation being conducted concurrently, which will add to trial evidence. The key elements developed by the co-design process and the detailed intervention description will allow for improved feasibility, sustainability and translation.
The focus of this article was to describe the development process of a novel, complex, Mediterranean dietary intervention for stroke survivors. The four phases of the IKT framework identified essential elements for inclusion and enabled a comprehensive description of the intervention using the TIDieR checklist. This process will provide a template to help researchers replicate the intervention with a view to closing the gap moving stroke research evidence into clinical practice.
Supplementary Materials
The following are available online at https://www.mdpi.com/article/10.3390/nu13041058/s1, Table S1: Description of Persona used in Workshop 1.
Author Contributions
Conceptualization, A.J.P., C.E. and L.M.-W.; methodology, A.J.P., C.E., E.R. and L.M.-W.; formal analysis, K.Z.; investigation, K.Z.; resources, K.Z., E.R., R.G. and M.B.; data curation, K.Z.; writing—original draft preparation, K.Z.; writing—review and editing, K.Z., A.J.P., C.E., E.R., M.G., R.G., M.B. and L.M.-W.; supervision, A.J.P., C.E. and L.M.-W.; project administration, K.Z. and M.G. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki and the ICH E6 Good Clinical Practice. It was approved by the Hunter New England Human Research Ethics Committee of the Hunter New England Local Health District (2019/ETH13034) on 3 December 2019.
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
Data Availability Statement
The data presented in this study are available upon reasonable request to the corresponding author. The data are not publicly available due to confidentiality reasons.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Westfall, J.M.; Mold, J.; Fagnan, L. Practice-Based Research—“Blue Highways” on the NIH Roadmap. JAMA 2007, 297, 403–406. [Google Scholar] [CrossRef] [PubMed]
- Green, L.W.; Ottoson, J.M.; García, C.; Hiatt, R.A. Diffusion Theory and Knowledge Dissemination, Utilization, and Integration in Public Health. Annu. Rev. Public Health 2009, 30, 151–174. [Google Scholar] [CrossRef] [PubMed]
- Grant, J.; Green, L.; Mason, B. Basic research and health: A reassessment of the scientific basis for the support of biomedical science. Res. Eval. 2003, 12, 217–224. [Google Scholar] [CrossRef]
- Glasziou, P.; Straus, S.; Brownlee, S.; Trevena, L.; Dans, L.; Guyatt, G.; Elshaug, A.G.; Janett, R.; Saini, V. Evidence for underuse of effective medical services around the world. Lancet 2017, 390, 169–177. [Google Scholar] [CrossRef]
- Glasziou, P.; Chalmers, I. Research waste is still a scandal—An essay by Paul Glasziou and Iain Chalmers. BMJ 2018, 363, k4645. [Google Scholar] [CrossRef]
- Walker, M.F.; Hoffmann, T.C.; Brady, M.C.; Dean, C.M.; Eng, J.J.; Farrin, A.J.; Felix, C.; Forster, A.; Langhorne, P.; Lynch, E.; et al. Improving the development, monitoring and reporting of stroke rehabilitation research: Consensus-based core recommendations from the Stroke Recovery and Rehabilitation Roundtable. Int. J. Stroke 2017, 12, 472–479. [Google Scholar] [CrossRef]
- Hoffmann, T.C.; Glasziou, P.P.; Boutron, I.; Milne, R.; Perera, R.; Moher, D.; Altman, D.G.; Barbour, V.; Macdonald, H.; Johnston, M.; et al. Better reporting of interventions: Template for intervention description and replication (TIDieR) checklist and guide. BMJ Br. Med. J. 2014, 348, g1687. [Google Scholar] [CrossRef] [PubMed]
- O’Donnell, M.J.; Chin, S.L.; Rangarajan, S.; Xavier, D.; Liu, L.; Zhang, H.; Rao-Melacini, P.; Zhang, X.; Pais, P.; Agapay, S.; et al. Global and regional effects of potentially modifiable risk factors associated with acute stroke in 32 countries (INTERSTROKE): A case-control study. Lancet 2016, 388, 761–775. [Google Scholar] [CrossRef]
- Davis, C.R.; Hodgson, J.M.; Woodman, R.; Bryan, J.; Wilson, C.; Murphy, K.J. A Mediterranean diet lowers blood pressure and improves endothelial function: Results from the MedLey randomized intervention trial. Am. J. Clin. Nutr. 2017, 105, 1305–1313. [Google Scholar] [CrossRef]
- Cowell, O.R.; Mistry, N.; Deighton, K.; Matu, J.; Griffiths, A.; Minihane, A.M.; Mathers, J.C.; Shannon, O.M.; Siervo, M. Effects of a Mediterranean diet on blood pressure: A systematic review and meta-analysis of randomized controlled trials and observational studies. J. Hypertens. 2021, 39, 729–739. [Google Scholar] [CrossRef]
- Adamsson, V.; Reumark, A.; Fredriksson, I.-B.; Hammarström, E.; Vessby, B.; Johansson, G.; Risérus, U. Effects of a healthy Nordic diet on cardiovascular risk factors in hypercholesterolaemic subjects: A randomized controlled trial (NORDIET). J. Intern. Med. 2010, 269, 150–159. [Google Scholar] [CrossRef]
- Ramezani-Jolfaie, N.; Mohammadi, M.; Salehi-Abargouei, A. The effect of healthy Nordic diet on cardio-metabolic markers: A systematic review and meta-analysis of randomized controlled clinical trials. Eur. J. Nutr. 2018, 58, 2159–2174. [Google Scholar] [CrossRef]
- Sacks, F.M.; Svetkey, L.P.; Vollmer, W.M.; Appel, L.J.; Bray, G.A.; Harsha, D.; Obarzanek, E.; Conlin, P.R.; Miller, E.R.; Simons-Morton, D.G.; et al. Effects on Blood Pressure of Reduced Dietary Sodium and the Dietary Approaches to Stop Hypertension (DASH) Diet. N. Engl. J. Med. 2001, 344, 3–10. [Google Scholar] [CrossRef] [PubMed]
- Saneei, P.; Salehi-Abargouei, A.; Esmaillzadeh, A.; Azadbakht, L. Influence of Dietary Approaches to Stop Hypertension (DASH) diet on blood pressure: A systematic review and meta-analysis on randomized controlled trials. Nutr. Metab. Cardiovasc. Dis. 2014, 24, 1253–1261. [Google Scholar] [CrossRef] [PubMed]
- Estruch, R.; Ros, E.; Salas-Salvadó, J.; Covas, M.-I.; Corella, D.; Arós, F.; Gómez-Gracia, E.; Ruiz-Gutiérrez, V.; Fiol, M.; Lapetra, J.; et al. Primary Prevention of Cardiovascular Disease with a Mediterranean Diet Supplemented with Extra-Virgin Olive Oil or Nuts. N. Engl. J. Med. 2018, 378, e34. [Google Scholar] [CrossRef]
- Davis, C.; Hodgson, J.; Bryan, J.; Garg, M.; Woodman, R.; Murphy, K. Older Australians Can Achieve High Adherence to the Mediterranean Diet during a 6 Month Randomised Intervention; Results from the Medley Study. Nutrients 2017, 9, 534. [Google Scholar] [CrossRef]
- Kouris-Blazos, A.; Gnardellis, C.; Wahlqvist, M.L.; Trichopoulos, D.; Lukito, W.; Trichopoulou, A. Are the advantages of the Mediterranean diet transferable to other populations? A cohort study in Melbourne, Australia. Br. J. Nutr. 1999, 82, 57–61. [Google Scholar] [CrossRef] [PubMed]
- Lara, J.; Turbett, E.; Mckevic, A.; Rudgard, K.; Hearth, H.; Mathers, J.C. The Mediterranean diet among British older adults: Its understanding, acceptability and the feasibility of a randomised brief intervention with two levels of dietary advice. Maturitas 2015, 82, 387–393. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Garcia, E.; Rodriguez-Artalejo, F.; Li, T.Y.; Fung, T.T.; Li, S.; Willett, W.C.; Rimm, E.B.; Hu, F.B. The Mediterranean-style dietary pattern and mortality among men and women with cardiovascular disease. Am. J. Clin. Nutr. 2013, 99, 172–180. [Google Scholar] [CrossRef]
- Zacharia, K.; Patterson, A.J.; English, C.; MacDonald-Wicks, L. Feasibility of the AusMed Diet Program: Translating the Mediterranean Diet for Older Australians. Nutrients 2020, 12, 1044. [Google Scholar] [CrossRef]
- Martínez-González, M.A.; García-Arellano, A.; Toledo, E.; Salas-Salvadó, J.; Buil-Cosiales, P.; Corella, D.; Covas, M.I.; Schröder, H.; Arós, F.; Gómez-Gracia, E.; et al. A 14-Item Mediterranean Diet Assessment Tool and Obesity Indexes among High-Risk Subjects: The PREDIMED Trial. PLoS ONE 2012, 7, e43134. [Google Scholar] [CrossRef] [PubMed]
- Graham, I.D.; Logan, J.; Harrison, M.B.; Straus, S.E.; Tetroe, J.; Caswell, W.; Robinson, N. Lost in knowledge translation: Time for a map? J. Contin. Educ. Health Prof. 2006, 26, 13–24. [Google Scholar] [CrossRef]
- Canadian Institutes of Health Research. Guide to Knowledge Translation Planning at CIHR: Integrated and End-of Grant Approaches; Canadian Institutes of Health Research: Ottawa, ON, Canada, 2012. [Google Scholar]
- Bayley, M.T.; Teasell, R.W.; Wolfe, D.L.; Gruen, R.L.; Eng, J.J.; Ghajar, J.; Tavender, E.; Kua, A.; Bragge, P. Where to Build the Bridge Between Evidence and Practice? J. Head Trauma Rehabil. 2014, 29, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Slattery, P.; Saeri, A.K.; Bragge, P. Research co-design in health: A rapid overview of reviews. Health Res. Policy Syst. 2020, 18, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Craig, P.; Dieppe, P.; Macintyre, S.; Michie, S.; Nazareth, I.; Petticrew, M. Developing and evaluating complex interventions: The new Medical Research Council guidance. BMJ 2008, 337, a1655. [Google Scholar] [CrossRef]
- Moher, D.; Schulz, K.F.; Altman, D.G. The CONSORT statement: Revised recommendations for improving the quality of reports of parallel-group randomised trials. Lancet 2001, 357, 1191–1194. [Google Scholar] [CrossRef]
- Ball, L.E.; Sladdin, I.K.; Mitchell, L.J.; Barnes, K.A.; Ross, L.J.; Williams, L.T. Quality of development and reporting of dietetic intervention studies in primary care: A systematic review of randomised controlled trials. J. Hum. Nutr. Diet. 2017, 31, 47–57. [Google Scholar] [CrossRef]
- Warner, M.M.; Kelly, J.T.; Reidlinger, D.P.; Hoffmann, T.C.; Campbell, K.L.; Burner, E.; Neubeck, L. Reporting of Telehealth-Delivered Dietary Intervention Trials in Chronic Disease: Systematic Review. J. Med. Internet Res. 2017, 19, e410. [Google Scholar] [CrossRef]
- Hoffmann, T.C.; Erueti, C.; Glasziou, P.P. Poor description of non-pharmacological interventions: Analysis of consecutive sample of randomised trials. BMJ 2013, 347, f3755. [Google Scholar] [CrossRef]
- Candy, B.; Vickerstaff, V.; Jones, L.; King, M. Description of complex interventions: Analysis of changes in reporting in randomised trials since 2002. Trials 2018, 19, 1–9. [Google Scholar] [CrossRef]
- Begg, C.; Cho, M.; Eastwood, S.; Horton, R.; Moher, D.; Olkin, I.; Pitkin, R.; Rennie, D.; Schulz, K.F.; Simel, D.; et al. Improving the Quality of Reporting of Randomized Controlled Trials. JAMA 1996, 276, 637–639. [Google Scholar] [CrossRef]
- Ramage, E.R.; Burke, M.; Galloway, M.; Janssen, H.; Lynch, E.A.; Marsden, D.L.; Patterson, A.J.; Said, V.M.; English, C. An integrated knowledge translation approach to co-design a complex exercise intervention for stroke survivors: A case report. In How We Work Together: The Integrated Knowledge Translation Research Network Casebook; Boland, L., Reszel, J., McCutcheon, C., Kothari, A., Graham, I.D., Eds.; Integrated Knowledge Translation Research Network: Ottawa, ON, Canada, 2020; Volume 3, pp. 13–18. [Google Scholar]
- Borrelli, B.; Sepinwall, D.; Ernst, D.; Bellg, A.J.; Czajkowski, S.; Breger, R.; DeFrancesco, C.; Levesque, C.; Sharp, D.L.; Ogedegbe, G.; et al. A new tool to assess treatment fidelity and evaluation of treatment fidelity across 10 years of health behavior research. J. Consult. Clin. Psychol. 2005, 73, 852–860. [Google Scholar] [CrossRef]
- Pruitt, J.; Adlin, T. Chapter 2 The Persona Lifecycle: A Framework for the Persona Approach. In The Persona Lifecycle: Keeping People in Mind throughout Product Design; Morgan Kaufmann: San Francisco, CA, USA, 2006. [Google Scholar]
- Endmann, A.; Keßner, D. User Journey Mapping—A Method in User Experience Design. i-com 2016, 15. [Google Scholar] [CrossRef]
- Howard, T. Journey mapping. Commun. Des. Q. Rev. 2014, 2, 10–13. [Google Scholar] [CrossRef]
- Australian Institute of Health and Welfare. Stroke Management in Australia: An Update; Cardiovascular Diseases Series No. 37. CVD 61; AIHW: Canberra, ACT, Australia, 2013. [Google Scholar]
- Braun, V.; Clarke, V. Using thematic analysis in psychology. Qual. Res. Psychol. 2006, 3, 77–101. [Google Scholar] [CrossRef]
- Michie, S.; Van Stralen, M.M.; West, R. The behaviour change wheel: A new method for characterising and designing behaviour change interventions. Implement. Sci. 2011, 6, 42. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.; Dyer, K.; Hyde, B.; Davis, C.; Hodgson, J.; Woodman, R. Australians Can Adopt Mediterranean Diet (MedDiet) Principles up to 1-Year Following Completion of the Medley Trail (OR22-01-19). Curr. Dev. Nutr. 2019, 3 (Suppl. 1). [Google Scholar] [CrossRef]
- Teasdale, N.; Elhussein, A.; Butcher, F.; Piernas, C.; Cowburn, G.; Hartmann-Boyce, J.; Saksena, R.; Scarborough, P. Systematic review and meta-analysis of remotely delivered interventions using self-monitoring or tailored feedback to change dietary behavior. Am. J. Clin. Nutr. 2018, 107, 247–256. [Google Scholar] [CrossRef] [PubMed]
- Whatnall, M.C.; Patterson, A.J.; Ashton, L.M.; Hutchesson, M.J. Effectiveness of brief nutrition interventions on dietary behaviours in adults: A systematic review. Appetite 2018, 120, 335–347. [Google Scholar] [CrossRef]
- Ryan, K.; Dockray, S.; Linehan, C. A systematic review of tailored eHealth interventions for weight loss. Digit. Health 2019, 5, 2055207619826685. [Google Scholar] [CrossRef]
- Marzolini, S.; Balitsky, A.; Jagroop, D.; Corbett, D.; Brooks, D.; Grace, S.L.; Lawrence, D.; Oh, P.I. Factors Affecting Attendance at an Adapted Cardiac Rehabilitation Exercise Program for Individuals with Mobility Deficits Poststroke. J. Stroke Cerebrovasc. Dis. 2016, 25, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, S.L.; Donaghy, M.; Johnston, M.; Sniehotta, F.F.; Van Wijck, F.; Johnston, D.; Greig, C.; McMurdo, M.E.T.; Mead, G. A qualitative theory guided analysis of stroke survivors’ perceived barriers and facilitators to physical activity. Disabil. Rehabil. 2013, 36, 1857–1868. [Google Scholar] [CrossRef] [PubMed]
- Byaruhanga, J.; Atorkey, P.; McLaughlin, M.; Brown, A.; Byrnes, E.; Paul, C.; Wiggers, J.; Tzelepis, F. Effectiveness of Individual Real-Time Video Counseling on Smoking, Nutrition, Alcohol, Physical Activity, and Obesity Health Risks: Systematic Review. J. Med. Internet Res. 2020, 22, e18621. [Google Scholar] [CrossRef]
- Vilme, H.; Duke, N.N.; Muiruri, C.; Wordlaw, L.; Skinner, A.C. Using Telehealth to Disseminate Primary, Secondary, and Tertiary CVD Interventions to Rural Populations. Curr. Hypertens. Rep. 2019, 21, 92. [Google Scholar] [CrossRef] [PubMed]
- Kelly, J.T.; Reidlinger, D.P.; Hoffmann, T.C.; Campbell, K.L. Telehealth methods to deliver dietary interventions in adults with chronic disease: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2016, 104, 1693–1702. [Google Scholar] [CrossRef]
- Rigby, R.R.; Mitchell, L.J.; Hamilton, K.; Williams, L.T. The Use of Behavior Change Theories in Dietetics Practice in Primary Health Care: A Systematic Review of Randomized Controlled Trials. J. Acad. Nutr. Diet. 2020, 120, 1172–1197. [Google Scholar] [CrossRef]
- Samdal, G.B.; Eide, G.E.; Barth, T.; Williams, G.; Meland, E. Effective behaviour change techniques for physical activity and healthy eating in overweight and obese adults; systematic review and meta-regression analyses. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 1–14. [Google Scholar] [CrossRef]
- Michie, S.; Atkins, L.S.; West, R. The Behaviour Change Wheel: A Guide to Designing Interventions; Silverback Publishing: Sutton, UK, 2014. [Google Scholar]
- Stroke Association. Accessible Information Guidelines: Making Information Accesible for People with Aphasia; Stroke Association: London, UK, 2012. [Google Scholar]
- Schröder, H.; Fitó, M.; Estruch, R.; Martínez-González, M.A.; Corella, D.; Salas-Salvadó, J.; Lamuela-Raventós, R.; Ros, E.; Salaverría, I.; Fiol, M.; et al. A Short Screener Is Valid for Assessing Mediterranean Diet Adherence among Older Spanish Men and Women. J. Nutr. 2011, 141, 1140–1145. [Google Scholar] [CrossRef]
- Lawrence, W.; Black, C.; Tinati, T.; Cradock, S.; Begum, R.; Jarman, M.; Pease, A.; Margetts, B.; Davies, J.; Inskip, H.; et al. ‘Making every contact count’: Evaluation of the impact of an intervention to train health and social care practitioners in skills to support health behaviour change. J. Health Psychol. 2016, 21, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Harris, P.A.; Taylor, R.; Minor, B.L.; Elliott, V.; Fernandez, M.; O’Neal, L.; McLeod, L.; Delacqua, G.; Delacqua, F.; Kirby, J.; et al. The REDCap consortium: Building an international community of software platform partners. J. Biomed. Inform. 2019, 95, 103208. [Google Scholar] [CrossRef]
- Harris, P.A.; Taylor, R.; Thielke, R.; Payne, J.; Gonzalez, N.; Conde, J.G. Research electronic data capture (REDCap)—A metadata-driven methodology and workflow process for providing translational research informatics support. J. Biomed. Inform. 2009, 42, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Fryer, C.; Luker, J.; McDonnell, M.N.; Hillier, S.L. Self management programmes for quality of life in people with stroke. Cochrane Database Syst. Rev. 2016, 2016, CD010442. [Google Scholar] [CrossRef] [PubMed]
- Gagliardi, A.R.; Berta, W.; Kothari, A.; Boyko, J.; Urquhart, R. Integrated knowledge translation (IKT) in health care: A scoping review. Implement. Sci. 2015, 11, 1. [Google Scholar] [CrossRef] [PubMed]
- Choi-Kwon, S.; Kim, J.S. Poststroke Fatigue: An Emerging, Critical Issue in Stroke Medicine. Int. J. Stroke 2011, 6, 328–336. [Google Scholar] [CrossRef]
- Staub, F.; Bogousslavsky, J. Fatigue after Stroke: A Major but Neglected Issue. Cerebrovasc. Dis. 2001, 12, 75–81. [Google Scholar] [CrossRef]
- Ma, P.M.P.; Jørgensen, H.S.; Nakayama, H.; Raaschou, H.O.; Olsen, T.S. Aphasia in acute stroke: Incidence, determinants, and recovery. Ann. Neurol. 1995, 38, 659–666. [Google Scholar] [CrossRef]
- Azhar, A.; Maqbool, S.; Butt, G.A.; Iftikhar, S.; Iftikhar, G. Frequency of Aphasia and Its Symptoms in Stroke Patients. J. Speech Pathol. Ther. 2016, 2. [Google Scholar] [CrossRef]
- Economics, D.A. No postcode Untouched: Stroke in Australia 2017; Stroke Foundation: Sydney, NSW, Australia, 2017. [Google Scholar]
- Lawrence, E.S.; Coshall, C.; Dundas, R.; Stewart, J.; Rudd, A.G.; Howard, R.; Wolfe, C.D.A. Estimates of the Prevalence of Acute Stroke Impairments and Disability in a Multiethnic Population. Stroke 2001, 32, 1279–1284. [Google Scholar] [CrossRef]
- Adamson, J.; Beswick, A.; Ebrahim, S. Is stroke the most common cause of disability? J. Stroke Cerebrovasc. Dis. 2004, 13, 171–177. [Google Scholar] [CrossRef]
- Damasio, A.R. Aphasia. N. Engl. J. Med. 1992, 326, 531–539. [Google Scholar] [CrossRef]
- Borrelli, B. The assessment, monitoring, and enhancement of treatment fidelity in public health clinical trials. J. Public Health Dent. 2011, 71, S52–S63. [Google Scholar] [CrossRef] [PubMed]
- Towfighi, A.; Markovic, D.; Ovbiagele, B. Impact of a healthy lifestyle on all-cause and cardiovascular mortality after stroke in the USA. J. Neurol. Neurosurg. Psychiatry 2011, 83, 146–151. [Google Scholar] [CrossRef] [PubMed]
- Rees, K.; Takeda, A.; Martin, N.; Ellis, L.; Wijesekara, D.; Vepa, A.; Das, A.; Hartley, L.; Stranges, S. Mediterranean-style diet for the primary and secondary prevention of cardiovascular disease. Cochrane Database Syst. Rev. 2019, 3, CD009825. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).

