Efficacy of a Low-Dose Diosmin Therapy on Improving Symptoms and Quality of Life in Patients with Chronic Venous Disease: Randomized, Double-Blind, Placebo-Controlled Trial
Abstract
1. Introduction
2. Methods
2.1. Study Design
2.2. Patients
2.3. Primary Endpoints
2.4. Secondary Endpoints
- VAS scale for pain [24]. Using a ruler, the score is determined by measuring the distance (mm) on the 100 mm line between the “no pain” anchor and the patient’s mark, providing a range of scores from 0 to 100. A higher score indicates greater pain intensity. The following cut points on the pain VAS have been recommended: no pain (0–4 mm), mild pain (5–44 mm), moderate pain (45–74 mm), and severe pain (75–100 mm). At each visit, patients were asked to mark a vertical sign on the scale to assess the intensity of pain. For pain assessment, the investigator measured with a ruler the distance between point 0 and the marked line by the patient.
- The Venous Clinical Severity Score (VCSS) [25] includes 9 attributes of venous disease, each scored on a severity scale from 0 to 3. (Absent = 0, Mild = 1, Moderate = 2, Severe = 3). In order to generate a dynamic score, VCSS attributes are scored individually. These attributes include skin changes and pigmentation, inflammation and induration, and ulcers (including number, size, and duration), compression therapy, and pain. Total score (ranging between 0 and 27) was calculated by summing scores from all attributes. A decreasing score means a relief of symptoms.
- Investigator Global Assessment of Efficacy—Investigators made the global assessment regarding the treatment efficacy using a 4-point scale (1 = excellent, 2 = good, 3 = fair, 4 = poor), at visit 2 and 3.
- Patient Global Assessment of Efficacy—Patients made the global assessment regarding the treatment efficacy using a 5-point scale (1 = very satisfied, 2 = satisfied, 3 = adequate, 4 = unsatisfied, 5 = very unsatisfied), at visit 2 and 3.
- Continuation with investigational product—The evaluation of subject options about continuing with the investigational product after the study termination was assessed at week 8 (final visit) for both groups. This variable is treated as a categorical variable and described by percentages and frequencies for each allocation group.
- Evaluation of symptoms relief—Evaluation of symptoms relief was assessed by evaluating the percentage of subjects who experienced symptoms relief within the first week of intake, within 2 weeks of intake, or more than 2 weeks of intake.
- Investigator Global Assessment of Safety—Investigators made the global assessment regarding the safety of treatments using a 4-point scale (1 = very good safety, 2 = good safety, 3 = moderate safety and 4 = poor safety), at visit 2 and 3.
2.5. Statistical Analysis
3. Results
3.1. Patients
3.2. Efficacy of Study Treatments
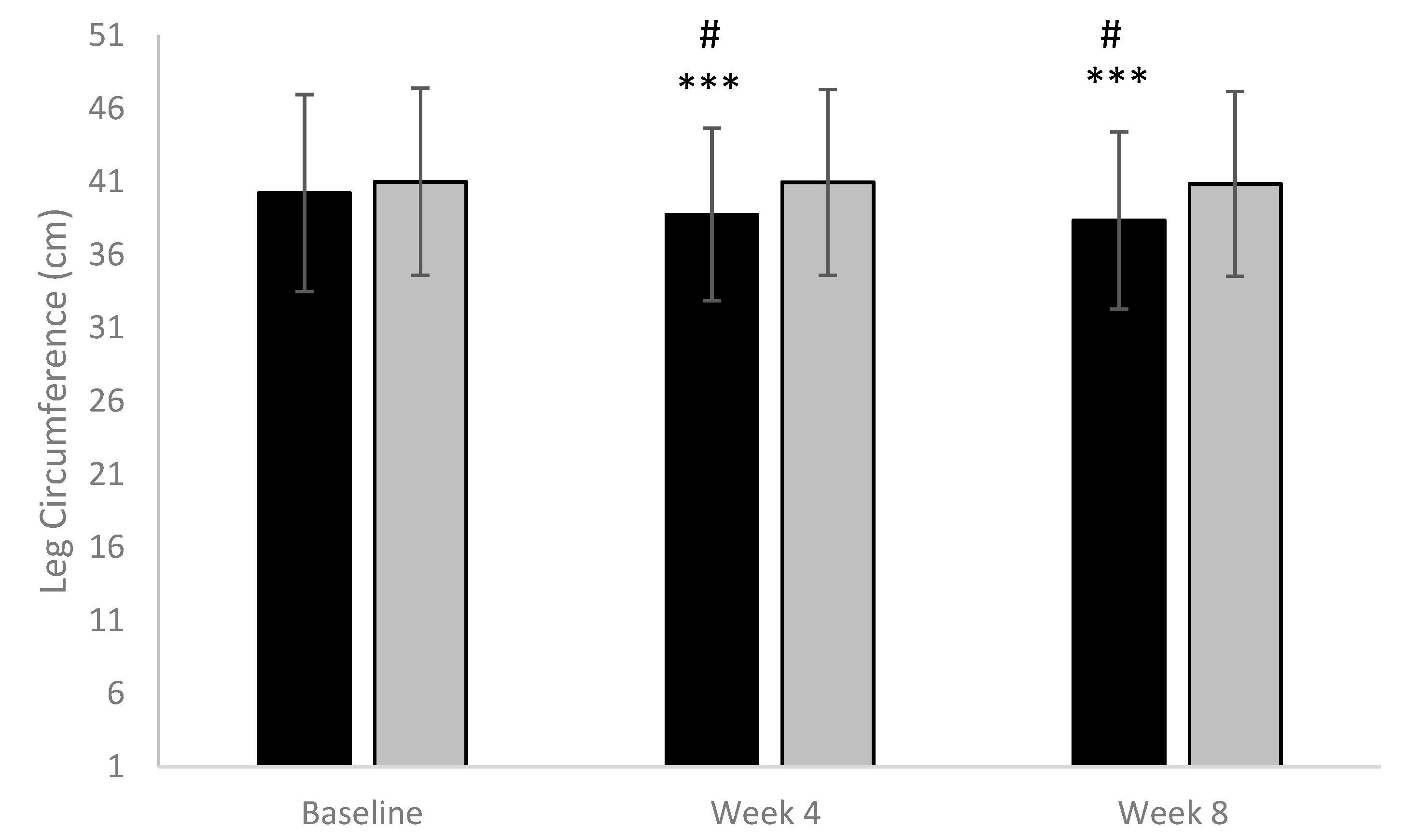
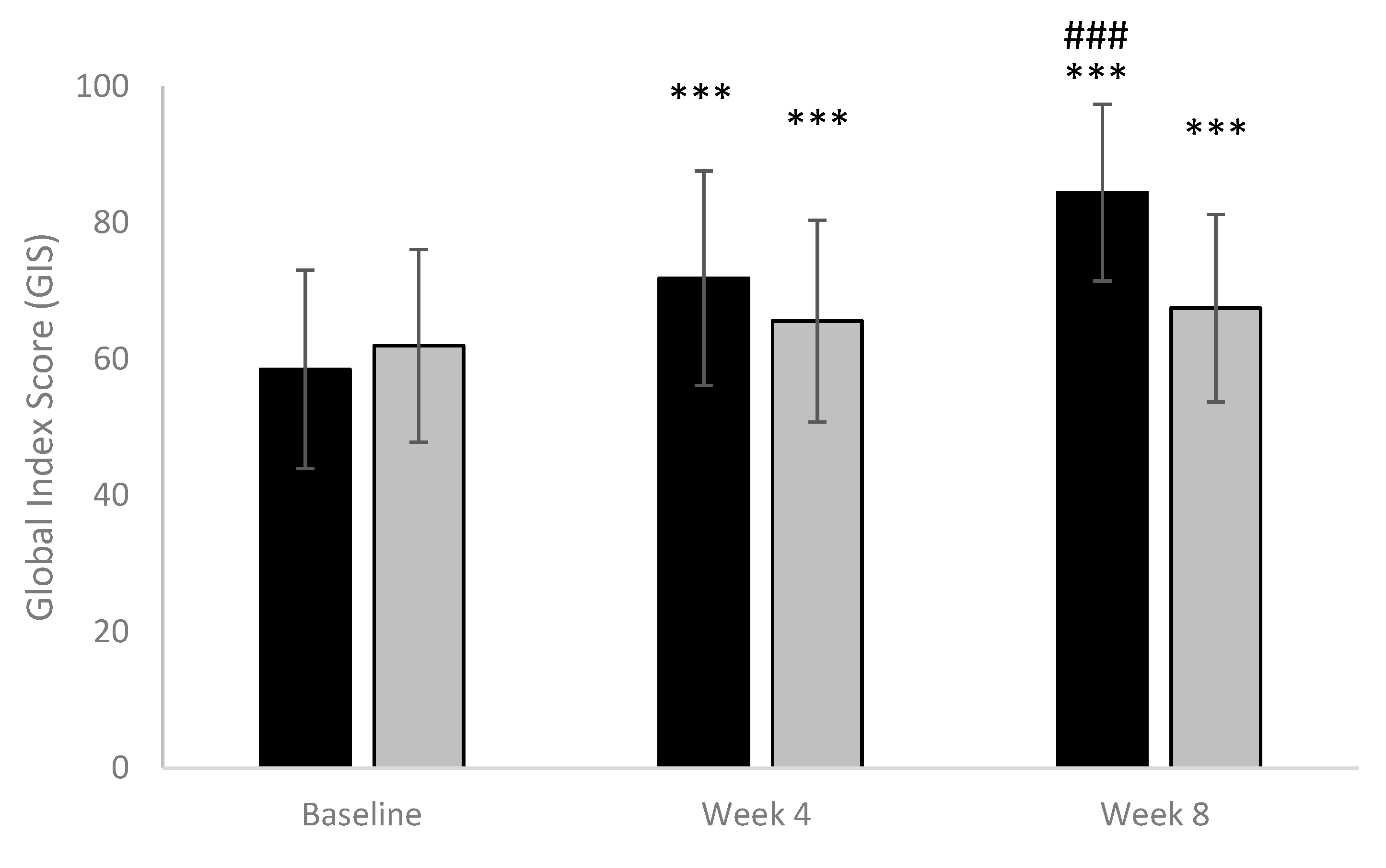
Primary Efficacy Outcomes: Circumference of Each Affected Leg (Calf) and Quality of Life Assessed by Global Index Score
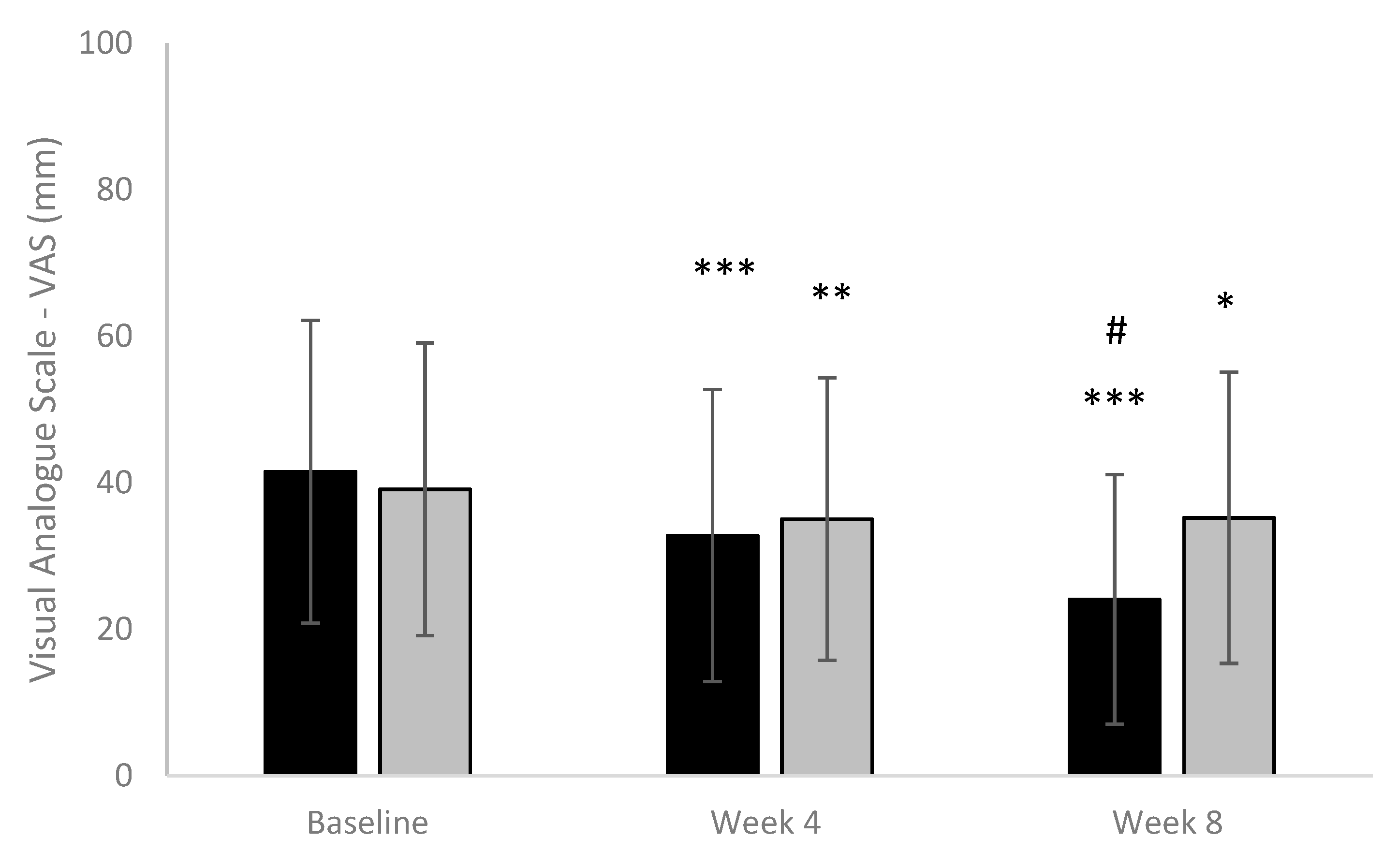
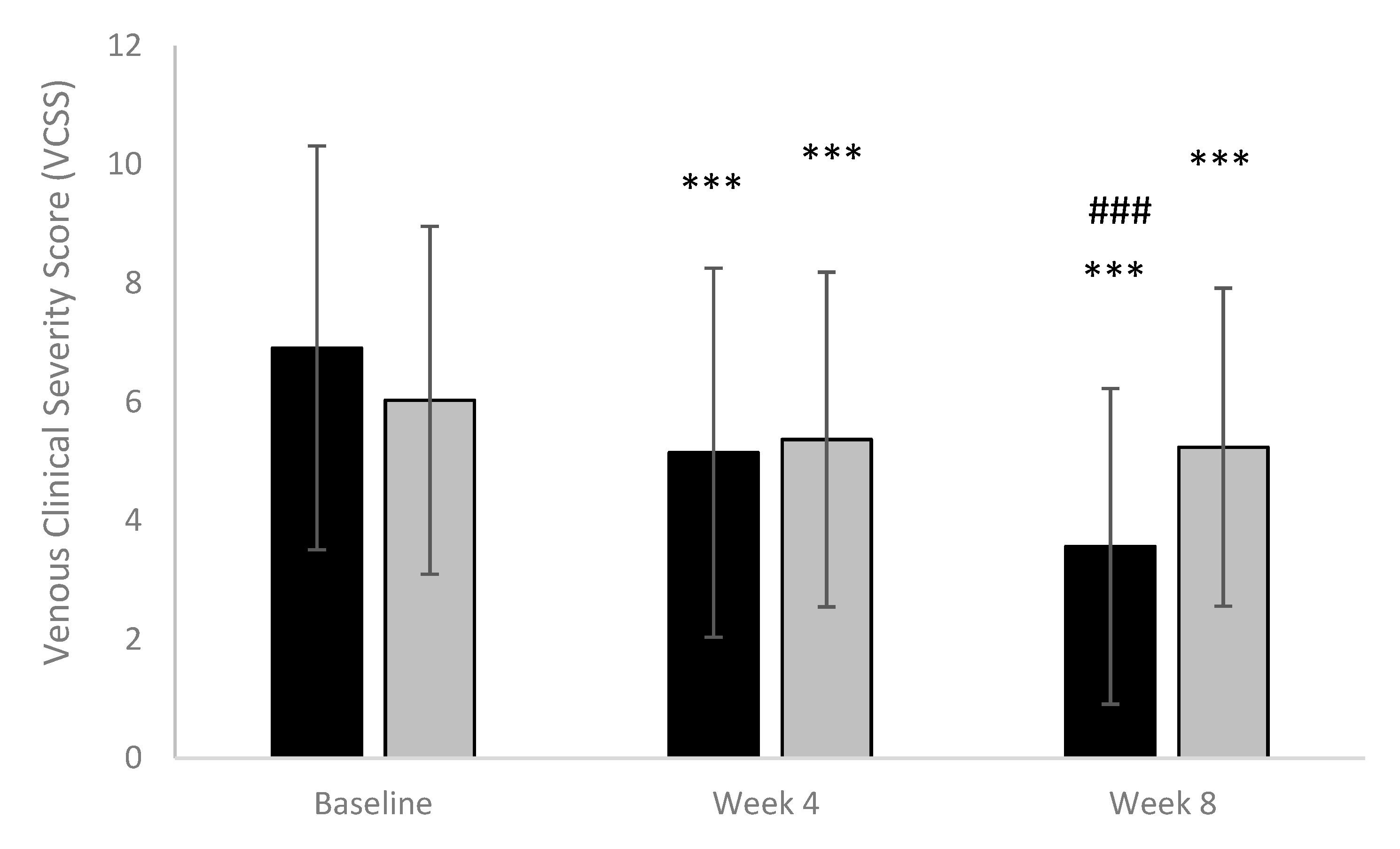
3.3. Secondary Efficacy Outcomes
3.4. Safety Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of variance |
| AE | Adverse Event |
| CEAP | Clinical-Etiology-Anatomy-Pathophysiology |
| IC | Informed Consent |
| IGAE | Investigator Global Assessment of Efficacy |
| IGAS | Investigator Global Assessment of Safety |
| IP | Investigational Product |
| ITT | Intention to treat |
| PGAE | Patient Global Assessment of Efficacy |
| QoL | Quality of Life |
| SAE | Serious Adverse Event |
| SD | Standard Deviation |
| VAS | Visual Analogue Scale |
References
- Serra, R.; Grande, R.; Butrico, L.; Fugetto, F.; de Franciscis, S. Epidemiology, diagnosis and treatment of chronic venous disease: A systematic review. Chirurgia 2016, 24, 34–45. [Google Scholar]
- Nicolaides, N.A.; Labropoulos, N. Burden and Suffering in Chronic Venous Disease. Adv. Ther. 2019, 36 (Suppl. 1), 1–4. [Google Scholar] [CrossRef]
- Serra, R.; Ssempijja, L.; Provenzano, M.; Andreucci, M. Genetic biomarkers in chronic venous disease. Biomark. Med. 2020, 14, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Kostas, T.I.; Ioannou, C.V.; Drygiannakis, I.; Georgakarakos, E.; Kounos, C.; Tsetis, D.; Katsamouris, A.N. Chronic venous disease progression and modification of predisposing factors. J. Vasc. Surg. 2010, 51, 900–907. [Google Scholar] [CrossRef]
- Labropoulos, N. How Does Chronic Venous Disease Progress from the First Symptoms to the Advanced Stages? A Review. Adv. Ther. 2019, 36 (Suppl. 1), 13–19. [Google Scholar] [CrossRef]
- Raffetto, J.D.; Mannello, F. Pathophysiology of chronic venous disease. Int. Angiol. 2014, 33, 212–221. [Google Scholar] [PubMed]
- Partsch, H. Varicose veins and chronic venous insufficiency. Vasa 2009, 38, 293–301. [Google Scholar] [CrossRef]
- Serra, R.; Ielapi, N.; Bevacqua, E.; Rizzuto, A.; De Caridi, G.; Massara, M.; Casella, F.; Di Mizio, G.; de Franciscis, S. Haemorrhage from varicose veins and varicose ulceration: A systematic review. Int. Wound J. 2018, 15, 829–833. [Google Scholar] [CrossRef] [PubMed]
- Davies, A.H. The Seriousness of Chronic Venous Disease: A Review of Real-World Evidence. Adv. Ther. 2019, 36 (Suppl. 1), 5–12. [Google Scholar] [CrossRef]
- Ruggiero, M.; Grande, R.; Naso, A.; Butrico, L.; Rubino, P.; Placida, G.D.; Cannistrà, M.; Serra, R. Symptoms in patients with skin changes due to chronic venous insufficiency often lead to emergency care service: An Italian observational study. Int. Wound J. 2016, 13, 967–971. [Google Scholar] [CrossRef]
- Nicolaides, A.; Kakkos, S.; Baekgaard, N.; Comerota, A.; de Maeseneer, M.; Eklof, B.; Giannoukas, A.D.; Lugli, M.; Maleti, O.; Myers, K.; et al. Management of chronic venous disorders of the lower limbs. Guidelines According to Scientific Evidence. Part I. Int. Angiol. 2018, 37, 181–254. [Google Scholar] [CrossRef] [PubMed]
- Mansilha, A. Early Stages of Chronic Venous Disease: Medical Treatment Alone or in Addition to Endovenous Treatments. Adv. Ther. 2020, 37 (Suppl. 1), 13–18. [Google Scholar] [CrossRef]
- Ayala-García, M.A.; Reyes, J.S.; Muñoz Montes, N.; Guaní-Guerra, E. Frequency of use of elastic compression stockings in patients with chronic venous disease of the lower extremities. Phlebology 2019, 34, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Serra, R.; Grande, R.; Butrico, L.; Buffone, G.; Caliò, F.G.; Squillace, A.; Rizzo, B.A.; Massara, M.; Spinelli, F.; Ferrarese, A.G.; et al. Effects of a new nutraceutical substance on clinical and molecular parameters in patients with chronic venous ulceration. Int. Wound J. 2016, 13, 88–96. [Google Scholar] [CrossRef] [PubMed]
- Mansilha, A.; Sousa, J. Pathophysiological Mechanisms of Chronic Venous Disease and Implications for Venoactive Drug Therapy. Int. J. Mol. Sci. 2018, 19, 1669. [Google Scholar] [CrossRef]
- Marinović Kulišić, S.; Lupi, D. Pharmacological treatment in patients with chronic venous disease. Acta Dermatovenerol. Croat. 2012, 20, 197–200. [Google Scholar]
- Perrin, M.; Ramelet, A.A. Pharmacological treatment of primary chronic venous disease: Rationale, results and unanswered questions. Eur. J. Vasc. Endovasc. Surg. 2011, 41, 117–125. [Google Scholar] [CrossRef]
- Andreozzi, G.M. Sulodexide in the treatment of chronic venous disease. Am. J. Cardiovasc. Drugs 2012, 12, 73–81. [Google Scholar] [CrossRef]
- Kakkos, S.K.; Nicolaides, A.N. Efficacy of micronized purified flavonoid fraction (Daflon®) on improving individual symptoms, signs and quality of life in patients with chronic venous disease: A systematic review and meta-analysis of randomized double-blind placebo-controlled trials. Int. Angiol. 2018, 37, 143–154. [Google Scholar]
- Russo, R.; Mancinelli, A.; Ciccone, M.; Terruzzi, F.; Pisano, C.; Severino, L. Pharmacokinetic Profile of µSMIN Plus™, a new Micronized Diosmin Formulation, after Oral Administration in Rats. Nat. Prod. Commun. 2015, 10, 1569–1572. [Google Scholar]
- Russo, R.; Chandradhara, D.; De Tommasi, N. Comparative Bioavailability of Two Diosmin Formulations after Oral Administration to Healthy Volunteers. Molecules 2018, 23, 2174. [Google Scholar] [CrossRef] [PubMed]
- Eklöf, B.; Rutherford, R.B.; Bergan, J.J.; Carpentier, P.H.; Gloviczki, P.; Kistner, R.L.; Meissner, M.H.; Moneta, G.L.; Myers, K.; Padberg, F.T.; et al. Revision of the CEAP classification for chronic venous disorders: Consensus statement. J. Vasc. Surg. 2004, 40, 1248–1252. [Google Scholar] [CrossRef] [PubMed]
- Biemans, A.A.; van der Velden, S.K.; Bruijninckx, C.M.; Buth, J.; Nijsten, T. Validation of the chronic venous insufficiency quality of life questionnaire in Dutch patients treated for varicose veins. Eur. J. Vasc. Endovasc. Surg. 2011, 42, 246–253. [Google Scholar] [CrossRef]
- Boonstra, A.M.; Schiphorst Preuper, H.R.; Reneman, M.; Posthumus, J.B.; Stewart, R.E. Reliability and validity of the visual analogue scale for disability in patients with chronic musculoskeletal pain. Int. J. Rehabil. Res. 2008, 31, 165–169. [Google Scholar] [CrossRef]
- Rutherford, R.B.; Padberg, F.T., Jr.; Comerota, A.J.; Kistner, R.L.; Meissner, M.H.; Moneta, G.L. Venous severity scoring: An adjunct to venous outcome assessment. J. Vasc. Surg. 2000, 31, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Casili, G.; Lanza, M.; Campolo, M.; Messina, S.; Scuderi, S.; Ardizzone, A.; Filippone, A.; Paterniti, I.; Cuzzocrea, S.; Esposito, E. Therapeutic potential of flavonoids in the treatment of chronic venous insufficiency. Vasc. Pharmacol. 2021, 137, 106825. [Google Scholar] [CrossRef] [PubMed]
- Pfisterer, L.; König, G.; Hecker, M.; Korff, T. Pathogenesis of varicose veins—Lessons from biomechanics. Vasa 2014, 43, 88–99. [Google Scholar] [CrossRef]
- Castro-Ferreira, R.; Cardoso, R.; Leite-Moreira, A.; Mansilha, A. The Role of Endothelial Dysfunction and Inflammation in Chronic Venous Disease. Ann. Vasc. Surg. 2018, 46, 380–393. [Google Scholar] [CrossRef]
- Nicolaides, A.N. Chronic venous disease and the leukocyte-endothelium interaction: From symptoms to ulceration. Angiology 2005, 56 (Suppl. 1), S11–S19. [Google Scholar] [CrossRef]
- Bergan, J. Molecular mechanisms in chronic venous insufficiency. Ann. Vasc. Surg. 2007, 21, 260–266. [Google Scholar] [CrossRef]
- Bergan, J.J.; Pascarella, L.; Schmid-Schönbein, G.W. Pathogenesis of primary chronic venous disease: Insights from animal models of venous hypertension. J. Vasc. Surg. 2008, 47, 183–192. [Google Scholar] [CrossRef] [PubMed]
- Pocock, E.S.; Alsaigh, T.; Mazor, R.; Schmid-Schönbein, G.W. Cellular and molecular basis of Venous insufficiency. Vasc. Cell 2014, 6, 24. [Google Scholar] [CrossRef] [PubMed]
- Feldo, M.; Wójciak-Kosior, M.; Sowa, I.; Kocki, J.; Bogucki, J.; Zubilewicz, T.; Kęsik, J.; Bogucka-Kocka, A. Effect of Diosmin Administration in Patients with Chronic Venous Disorders on Selected Factors Affecting Angiogenesis. Molecules 2019, 24, 3316. [Google Scholar] [CrossRef]
- Najmanová, I.; Pourová, J.; Vopršalová, M.; Pilařová, V.; Semecký, V.; Nováková, L.; Mladěnka, P. Flavonoid metabolite 3-(3-hydroxyphenyl)propionic acid formed by human microflora decreases arterial blood pressure in rats. Mol. Nutr. Food Res. 2016, 60, 981–991. [Google Scholar] [CrossRef] [PubMed]
- Cova, D.; De Angelis, L.; Giavarini, F.; Palladini, G.; Perego, R. Pharmacokinetics and metabolism of oral diosmin in healthy volunteers. Int. J. Clin. Pharmacol. Ther. Toxicol. 1992, 30, 29–33. [Google Scholar] [PubMed]
- Applová, L.; Karlíčková, J.; Warncke, P.; Macáková, K.; Hrubša, M.; Macháček, M.; Tvrdý, V.; Fischer, D.; Mladěnka, P. 4-Methylcatechol, a flavonoid metabolite with potent antiplatelet effects. Mol. Nutr. Food Res. 2019, 63, e1900261. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.H.; Jung, E.A.; Sohng, I.S.; Han, J.A.; Kim, T.H.; Han, M.J. Intestinal bacterial metabolism of flavonoids and its relation to some biological activities. Arch. Pharm. Res. 1998, 21, 17–23. [Google Scholar] [CrossRef]
- Perego, R.; Beccaglia, P.; Angelini, M.; Villa, P.; Cova, D. Pharmacokinetic studies of diosmin and diosmetin in perfused rat liver. Xenobiotica 1993, 23, 1345–1352. [Google Scholar] [CrossRef]
- Garner, R.C.; Garner, J.V.; Gregory, S.; Whattam, M.; Calam, A.; Leong, D. Comparison of the absorption of micronized (Daflon 500 mg) and nonmicronized 14C-diosmin tablets after oral administration to healthy volunteers by accelerator mass spectrometry and liquid scintillation counting. J. Pharm. Sci. 2002, 91, 32–40. [Google Scholar] [CrossRef]
- Silvestro, L.; Tarcomnicu, I.; Dulea, C.; Attili, N.R.; Ciuca, V.; Peru, D.; Rizea Savu, S. Confirmation of diosmetin 3-O-glucuronide as major metabolite of diosmin in humans, using micro-liquid-chromatography-mass spectrometry and ion mobility mass spectrometry. Anal. Bioanal. Chem. 2013, 405, 8295–8310. [Google Scholar] [CrossRef]
- Shoab, S.S.; Porter, J.B.; Scurr, J.H.; Coleridge-Smith, P.D. Effect of oral micronized purified flavonoid fraction treatment on leukocyte adhesion molecule expression in patients with chronic venous disease: A pilot study. J. Vasc. Surg. 2000, 31, 456–461. [Google Scholar] [CrossRef]
- Ramelet, A. Venoactive Drugs. In Sclerotherapy, 6th ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 426–434. [Google Scholar] [CrossRef]
- Sheikh, P.; Lohsiriwat, V.; Shelygin, Y. Micronized purified flavonoid fraction in hemorrhoid disease: A systematic review and meta-analysis. Adv. Ther. 2020, 37, 2792–2812. [Google Scholar] [CrossRef]
- Laliberté, F.; Bookhart, B.K.; Nelson, W.W.; Lefebvre, P.; Schein, J.R.; Rondeau-Leclaire, J.; Duh, M.S. Impact of once-daily versus twice-daily dosing frequency on adherence to chronic medications among patients with venous thromboembolism. Patient 2013, 6, 213–224. [Google Scholar] [CrossRef] [PubMed]
- Lamping, D.L.; Schroter, S.; Kurz, X.; Kahn, S.R.; Abenhaim, L. Evaluation of outcomes in chronic venous disorders of the leg: Development of a scientifically rigorous, patient-reported measure of symptoms and quality of life. J. Vasc. Surg. 2003, 37, 410–419. [Google Scholar] [CrossRef] [PubMed]
- Fokou, M.; Moifo, B.; Fongang, E.; Teyang, A.; Muna, W. Characteristics of patients and patterns of chronic venous disease of the lower limbs in a referral hospital in Cameroon. J. Vasc. Surg. Venous Lymphat. Disord. 2018, 6, 90–95. [Google Scholar] [CrossRef]
- Catarinella, F.S.; Nieman, F.H.; Wittens, C.H. An overview of the most commonly used venous quality of life and clinical outcome measurements. J. Vasc. Surg. Venous Lymphat. Disord. 2015, 3, 333–340. [Google Scholar] [CrossRef] [PubMed]
- Ulloa, J.H. Micronized purified flavonoid fraction (MPFF) for patients suffering from chronic venous disease: A review of new evidence. Adv. Ther. 2019, 36 (Suppl. 1), 20–25. [Google Scholar] [CrossRef] [PubMed]
- Jantet, G. Chronic venous insufficiency: Worldwide results of the RELIEF study. Reflux assEssment and quaLity of lIfe improvEment with micronized Flavonoids. Angiology 2002, 53, 245–256. [Google Scholar] [CrossRef]
| Variables | Treatment | n | Mean (SD) | Median | 1st Quartile | 3rd Quartile |
|---|---|---|---|---|---|---|
| Age (years) | µsmin® Plus | 37 | 53.32 (5.29) | 54 | 32 | 60 |
| Placebo | 35 | 52.63 (7.38) | 55 | 30 | 60 | |
| Weight (kg) | µsmin® Plus | 37 | 73.54 (10.42) | 72 | 50 | 97 |
| Placebo | 35 | 69.34 (7.91) | 69 | 50 | 81 | |
| Height (cm) | µsmin® Plus | 37 | 170.35 (8.35) | 170 | 156 | 190 |
| Placebo | 35 | 167.54 (6.46) | 167 | 155 | 183 | |
| BMI (kg/m2) | µsmin® Plus | 37 | 25.24 (2.16) | 25.06 | 20.2 | 29.30 |
| Placebo | 35 | 24.70 (2.50) | 24.45 | 19.03 | 29.38 | |
| Heart rate (bpm) | µsmin® Plus | 37 | 76.08 (7.29) | 75 | 60 | 90 |
| Placebo | 35 | 76.03 (7.25) | 76 | 60 | 90 | |
| Sex | µsmin® Plus | Males: 10 Females: 27 | - | - | - | - |
| Placebo | Males: 5 Females: 30 | - | - | - | - |
| Week | Treatment | n (%) | Excellent | Good | Fair | Poor |
|---|---|---|---|---|---|---|
| Week 4 | µsmin® Plus | 37 (100) | 6 (16.22) | 28 (75.68) | 3 (8.11) | 0 |
| Placebo | 35 (100) | 1 (2.86) | 3 (8.57) | 8 (22.86) | 23 (65.71) | |
| Week 8 | µsmin® Plus | 37 (100) | 19 (51.35) | 17 (45.95) | 1 (2.7) | 0 |
| Placebo | 35 (100) | 0 | 3 (8.57) | 3 (8.57) | 29 (82.86) |
| Week | Treatment | n (%) | Very Satisfied | Satisfied | Adequate | Unsatisfied | Very Unsatisfied |
|---|---|---|---|---|---|---|---|
| Week 4 | µsmin® Plus | 37 (100) | 4 (10.81) | 22 (59.46) | 11 (29.73) | 0 | 0 |
| Placebo | 35 (100) | 1 (2.86) | 2 (5.71) | 9 (25.71) | 21 (60.00) | 2 (5.71) | |
| Week 8 | µsmin® Plus | 37 (100) | 20 (54.05) | 17 (45.95) | 0 | 0 | 0 |
| Placebo | 35 (100) | 0 | 5 (14.29) | 2 (5.71) | 23 (65.71) | 5 (14.29) |
| Treatment | n (%) | Within the First Week of Intake | Within 2 Weeks of Intake | After More than 2 Weeks of Intake | No Improvement |
|---|---|---|---|---|---|
| µsmin® Plus | 37 (100) | 4 (10.81) | 25 (67.57) | 7 (18.92) | 1 (2.70) |
| Placebo | 35 (100) | 0 | 4 (11.43) | 6 (17.14) | 25 (71.43) |
| Week | Treatment | n (%) | Very Good | Good | Moderate | Poor |
|---|---|---|---|---|---|---|
| Week 4 | µsmin® Plus | 37 (100) | 36 (97.30) | 1 (2.70) | 0 | 0 |
| Placebo | 35 (100) | 24 (68.57) | 11 (31.43) | 0 | 0 | |
| Week 8 | µsmin® Plus | 37 (100) | 36 (97.30) | 1 (2.70) | 0 | 0 |
| Placebo | 35 (100) | 31 (88.57) | 4 (11.43) | 0 | 0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serra, R.; Ielapi, N.; Bitonti, A.; Candido, S.; Fregola, S.; Gallo, A.; Loria, A.; Muraca, L.; Raimondo, L.; Velcean, L.; et al. Efficacy of a Low-Dose Diosmin Therapy on Improving Symptoms and Quality of Life in Patients with Chronic Venous Disease: Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients 2021, 13, 999. https://doi.org/10.3390/nu13030999
Serra R, Ielapi N, Bitonti A, Candido S, Fregola S, Gallo A, Loria A, Muraca L, Raimondo L, Velcean L, et al. Efficacy of a Low-Dose Diosmin Therapy on Improving Symptoms and Quality of Life in Patients with Chronic Venous Disease: Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients. 2021; 13(3):999. https://doi.org/10.3390/nu13030999
Chicago/Turabian StyleSerra, Raffaele, Nicola Ielapi, Andrea Bitonti, Stefano Candido, Salvatore Fregola, Alessandro Gallo, Antonio Loria, Lucia Muraca, Luca Raimondo, Luminița Velcean, and et al. 2021. "Efficacy of a Low-Dose Diosmin Therapy on Improving Symptoms and Quality of Life in Patients with Chronic Venous Disease: Randomized, Double-Blind, Placebo-Controlled Trial" Nutrients 13, no. 3: 999. https://doi.org/10.3390/nu13030999
APA StyleSerra, R., Ielapi, N., Bitonti, A., Candido, S., Fregola, S., Gallo, A., Loria, A., Muraca, L., Raimondo, L., Velcean, L., Guadagna, S., & Gallelli, L. (2021). Efficacy of a Low-Dose Diosmin Therapy on Improving Symptoms and Quality of Life in Patients with Chronic Venous Disease: Randomized, Double-Blind, Placebo-Controlled Trial. Nutrients, 13(3), 999. https://doi.org/10.3390/nu13030999








