Cloudy Apple Juice Fermented by Lactobacillus Prevents Obesity via Modulating Gut Microbiota and Protecting Intestinal Tract Health
Abstract
:1. Introduction
2. Materials and Methods
2.1. Microorganisms and Apples
2.2. Methods
Preparation of Three Cloudy Apple Juices
2.3. Animal Experiment Design
2.3.1. Animals and Diet
2.3.2. Serum and Hepatic Parameter Analyses
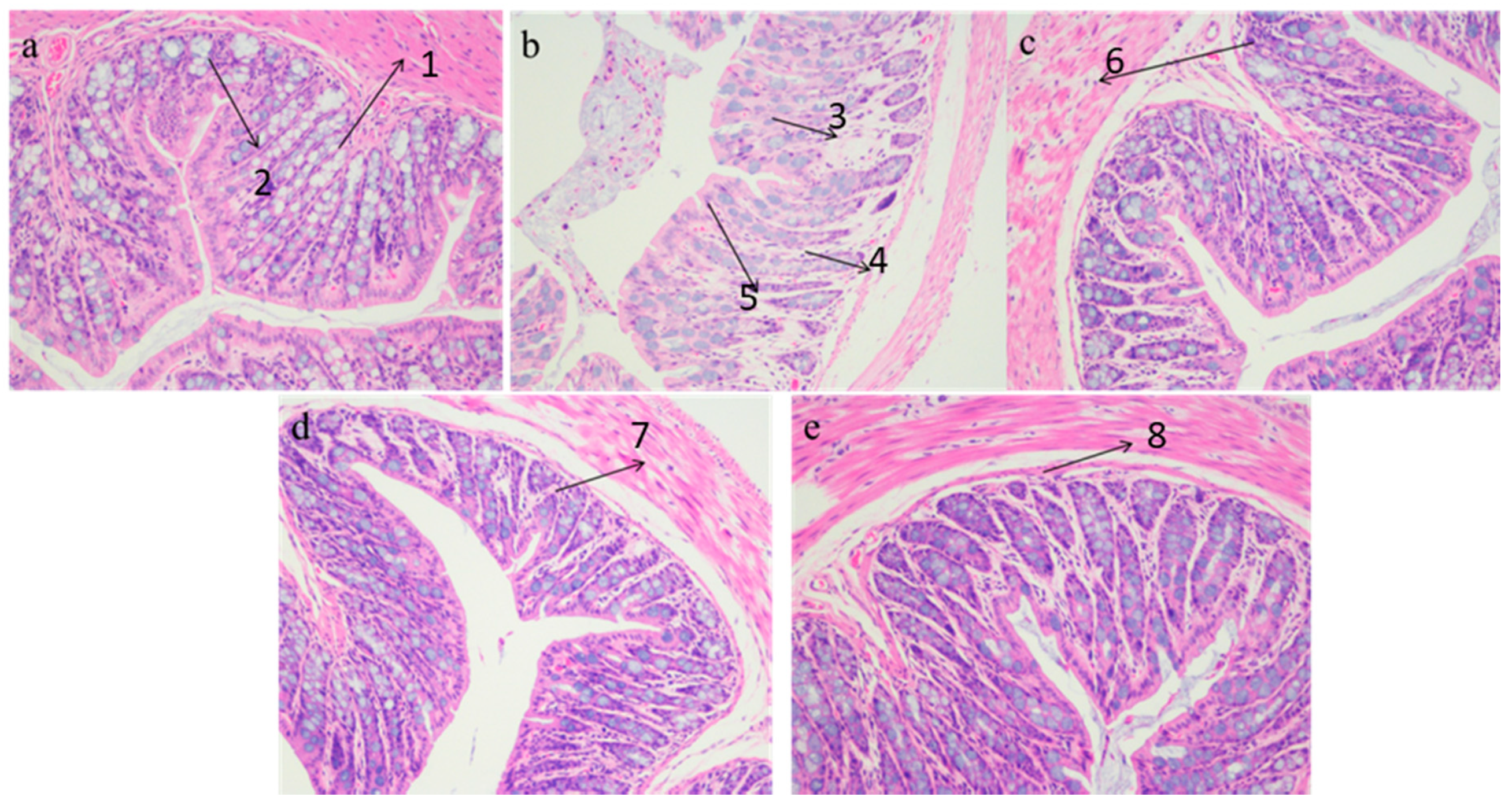
2.3.3. Histopathological Analysis
2.3.4. Sequencing of Fecal Microbiota
2.3.5. Quantification of Short-Chain Fatty Acids (SCFAs) in Cecal Contents
2.3.6. Evaluation of Intestinal Permeability and Intestinal Inflammation
2.4. Statistical Analysis
3. Results
3.1. Effects of Three Cloudy Apple Juices on Weight Lipid Levels and Oxidative Damage
3.2. Liver and Epididymal Adipose Tissue Histology Analysis
3.3. The Richness and Diversity of the Microbial Community
3.4. Gut Microbiota Composition at the Phylum and Genus Levels
3.5. Differences in the Dominant Gut Microbiota
3.6. Effect of Three Cloudy Apple Juices on SCFAs Concentrations
3.7. Effects of Three Cloudy Apple Juices on Intestinal Permeability, Intestinal Inflammation and Immunological Barrier
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Li, L.; Li, R.; Zhu, R.; Chen, B.; Tian, Y.; Zhang, H.; Xia, B.; Jia, Q.; Wang, L.; Zhao, D.; et al. Salvianolic acid B prevents body weight gain and regulates gut microbiota and LPS/TLR4 signaling pathway in high-fat diet-induced obese mice. Food Funct. 2020, 11, 8743–8756. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Han, M.; Zhang, M.; Wang, Y.; Ren, Y.; Yue, T.; Gao, Z. In vitro evaluation of the hypoglycemic properties of lactic acid bacteria and its fermentation adaptability in apple juice. LWT 2021, 136, 110363. [Google Scholar] [CrossRef]
- Liu, D.; Ji, Y.; Zhao, J.; Wang, H.; Guo, Y.; Wang, H. Black rice (Oryza sativa L.) reduces obesity and improves lipid metabolism in C57BL/6J mice fed a high-fat diet. J. Funct. Foods 2020, 64, 103605. [Google Scholar] [CrossRef]
- Li, H. Hypoglycemic and Hypolipidemic Mechanism of Tea Polysaccharides on Type 2 Diabetic Rats via Gut Microbiota and Metabolism Alteration. J. Agric. Food Chem. 2020, 68, 10015–10028. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Liu, M.; Zhang, P.; Fan, S.; Huang, J.; Yu, S.; Zhang, C.; Li, H. Fucoidan and galactooligosaccharides ameliorate high-fat diet–induced dyslipidemia in rats by modulating the gut microbiota and bile acid metabolism. Nutrition 2019, 65, 50–59. [Google Scholar] [CrossRef]
- Wang, X.; Chen, D.; Li, Y.; Zhao, S.; Chen, C.; Ning, D. Alleviating effects of walnut green husk extract on disorders of lipid levels and gut bacteria flora in high fat diet-induced obesity rats. J. Funct. Foods 2019, 52, 576–586. [Google Scholar] [CrossRef]
- Wei, T.; Dang, Y.; Cao, J.; Wu, Z.; He, J.; Sun, Y.; Pan, D.; Tian, Z. Different duck products protein on rat physiology and gut microbiota. J. Proteom. 2019, 206, 103436. [Google Scholar] [CrossRef] [PubMed]
- Tamura, Y.; Tomiya, S.; Takegaki, J.; Kouzaki, K.; Tsutaki, A.; Nakazato, K. Apple polyphenols induce browning of white adipose tissue. J. Nutr. Biochem. 2020, 77, 108299. [Google Scholar] [CrossRef] [PubMed]
- Gowd, V.; Karim, N.; Shishir, M.R.I.; Xie, L.; Chen, W. Dietary polyphenols to combat the metabolic diseases via altering gut microbiota. Trends Food Sci. Technol. 2019, 93, 81–93. [Google Scholar] [CrossRef]
- Li, X.; Huang, Y.; Song, L.; Xiao, Y.; Lu, S.; Xu, J.; Li, J.; Ren, Z. Lactobacillus plantarum prevents obesity via modulation of gut microbiota and metabolites in high-fat feeding mice. J. Funct. Foods 2020, 73, 104103. [Google Scholar] [CrossRef]
- Wang, M.; Zhang, B.; Hu, J.; Nie, S.; Xiong, T.; Xie, M. Intervention of five strains of Lactobacillus on obesity in mice induced by high-fat diet. J. Funct. Foods 2020, 72, 104078. [Google Scholar] [CrossRef]
- Peng, W.; Meng, D.; Yue, T.; Wang, Z.; Gao, Z. Effect of the apple cultivar on cloudy apple juice fermented by a mixture of Lactobacillus acidophilus, Lactobacillus plantarum, and Lactobacillus fermentum. Food Chem. 2020, 340, 127922. [Google Scholar] [CrossRef]
- Li, T.; Jiang, T.; Liu, N.; Wu, C.; Xu, H.; Lei, H. Biotransformation of phenolic profiles and improvement of antioxidant capacities in jujube juice by select lactic acid bacteria. Food Chem. 2021, 339, 127859. [Google Scholar] [CrossRef] [PubMed]
- Wen, J.; Ma, L.; Xu, Y.; Wu, J.; Yu, Y.; Peng, J.; Tang, D.; Zou, B.; Li, L. Effects of probiotic litchi juice on immunomodulatory function and gut microbiota in mice. Food Res. Int. 2020, 137, 109433. [Google Scholar] [CrossRef] [PubMed]
- Mi, Y. EGCG ameliorates high-fat- and high-fructose-induced cognitive defects by regulating the IRS/AKT and ERK/CREB/BDNF signaling pathways in the CNS. FASEB J. 2017, 31, 4998–5011. [Google Scholar] [CrossRef] [Green Version]
- Zhao, J.; Yu, L.; Zhai, Q.; Tian, F.; Zhang, H.; Chen, W. Effects of probiotic administration on hepatic antioxidative parameters depending on oxidative stress models: A meta-analysis of animal experiments. J. Funct. Foods 2020, 71, 103936. [Google Scholar] [CrossRef]
- Joseph, N.; Vasodavan, K.; Saipudin, N.A.; Yusof, B.N.M.; Kumar, S.; Nordin, S.A. Gut microbiota and short-chain fatty acids (SCFAs) profiles of normal and overweight school children in Selangor after probiotics administration. J. Funct. Foods 2019, 57, 103–111. [Google Scholar] [CrossRef]
- Xia, Y.-G.; Wang, T.-L.; Yu, S.-M.; Liang, J.; Kuang, H.-X. Structural characteristics and hepatoprotective potential of Aralia elata root bark polysaccharides and their effects on SCFAs produced by intestinal flora metabolism. Carbohydr. Polym. 2019, 207, 256–265. [Google Scholar] [CrossRef]
- Li, H.; Li, T.; Beasley, D.E.; Heděnec, P.; Xiao, Z.; Zhang, S.; Li, J.; Lin, Q.; Li, X. Diet Diversity Is Associated with Beta but not Alpha Diversity of Pika Gut Microbiota. Front. Microbiol. 2016, 7, 1169. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barlow, G.M.; Yu, A.; Mathur, R. Role of the Gut Microbiome in Obesity and Diabetes Mellitus. Nutr. Clin. Pr. 2015, 30, 787–797. [Google Scholar] [CrossRef]
- Chen, X.; Hou, F.; Wu, Y.; Cheng, Y. Bacterial and Fungal Community Structures in Loess Plateau Grasslands with Different Grazing Intensities. Front. Microbiol. 2017, 8, 606. [Google Scholar]
- Zhong, H. Probiotic-fermented blueberry juice prevents obesity and hyperglycemia in high fat diet-fed mice in association with modulating the gut microbiota. Food Funct. 2020, 11, 9192–9207. [Google Scholar] [CrossRef] [PubMed]
- Park, S. Effects of Cabbage-Apple Juice Fermented by Lactobacillus plantarum EM on Lipid Profile Improvement and Obesity Amelioration in Rats. Nutrients 2020, 12, 1135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dietary Methionine Restriction Ameliorated Fat Accumulation, Systemic Inflammation, and Increased Energy Metabolism by Altering Gut Microbiota in Middle-Aged Mice Administered Different Fat Diets. J. Agric. Food Chem. 2020, 68, 7745–7756. [CrossRef] [PubMed]
- Cao, S.-Y.; Zhao, C.-N.; Xu, X.-Y.; Tang, G.-Y.; Corke, H.; Gan, R.-Y.; Li, H.-B. Dietary plants, gut microbiota, and obesity: Effects and mechanisms. Trends Food Sci. Technol. 2019, 92, 194–204. [Google Scholar] [CrossRef]
- Liu, D.; Ji, Y.; Guo, Y.; Wang, H.; Wu, Z.; Li, H.; Wang, H. Dietary Supplementation of Apple Phlorizin Attenuates the Redox State Related to Gut Microbiota Homeostasis in C57BL/6J Mice Fed with a High-Fat Diet. J. Agric. Food Chem. 2021, 69, 198–211. [Google Scholar] [CrossRef]
- Mohamad, N.E.; Yeap, S.K.; Ky, H.; Ho, W.Y.; Boo, S.Y.; Chua, J.; Beh, B.-K.; Sharifuddin, S.A.; Long, K.; Alitheen, N.B. Dietary coconut water vinegar for improvement of obesity-associated inflammation in high-fat-diet-treated mice. Food Nutr. Res. 2017, 61, 1368322. [Google Scholar] [CrossRef]
- Lau, E. The role of I-FABP as a biomarker of intestinal barrier dysfunction driven by gut microbiota changes in obesity. Nutr. Metab. 2016, 13, 1–7. [Google Scholar] [CrossRef] [Green Version]
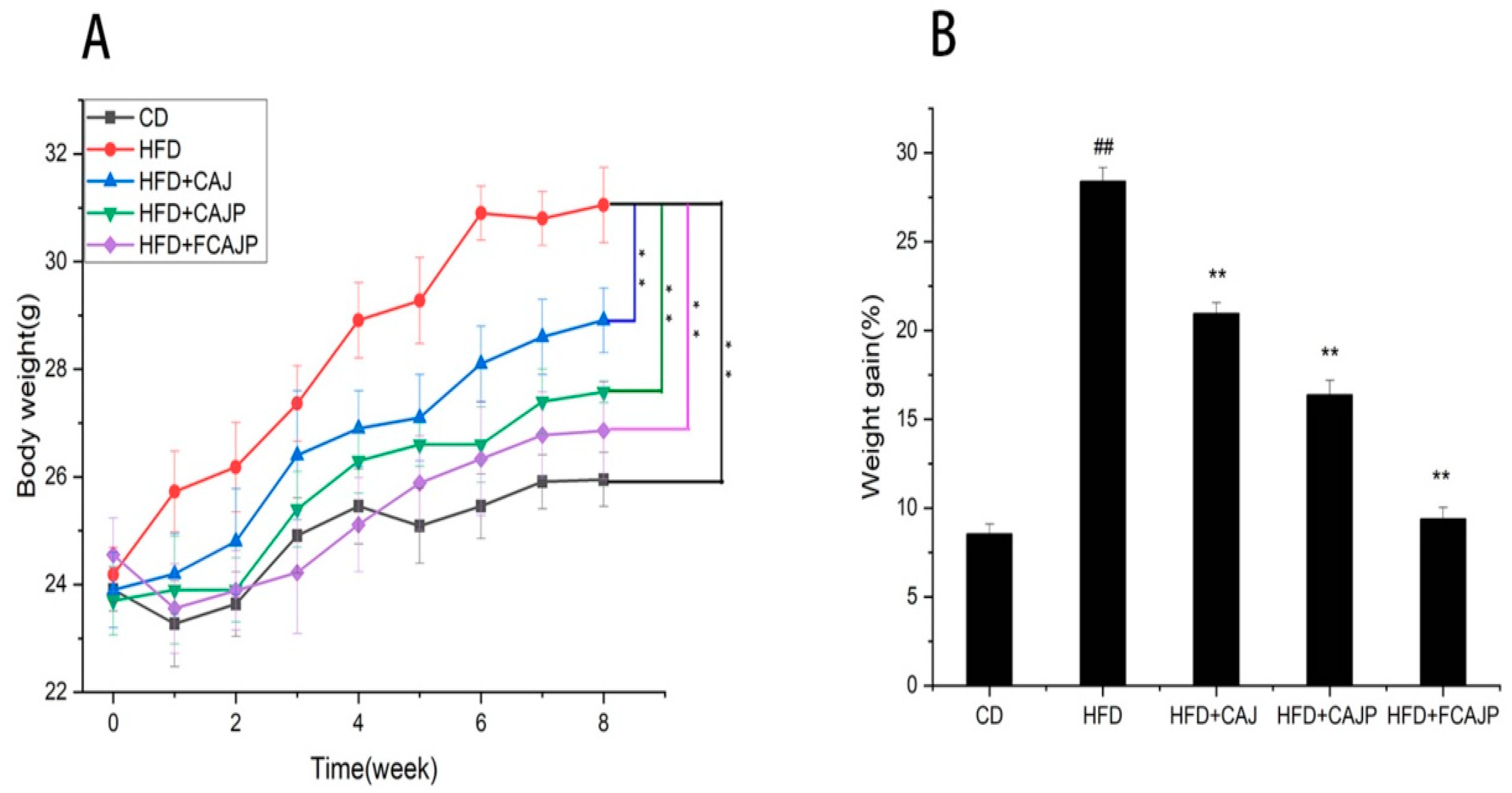
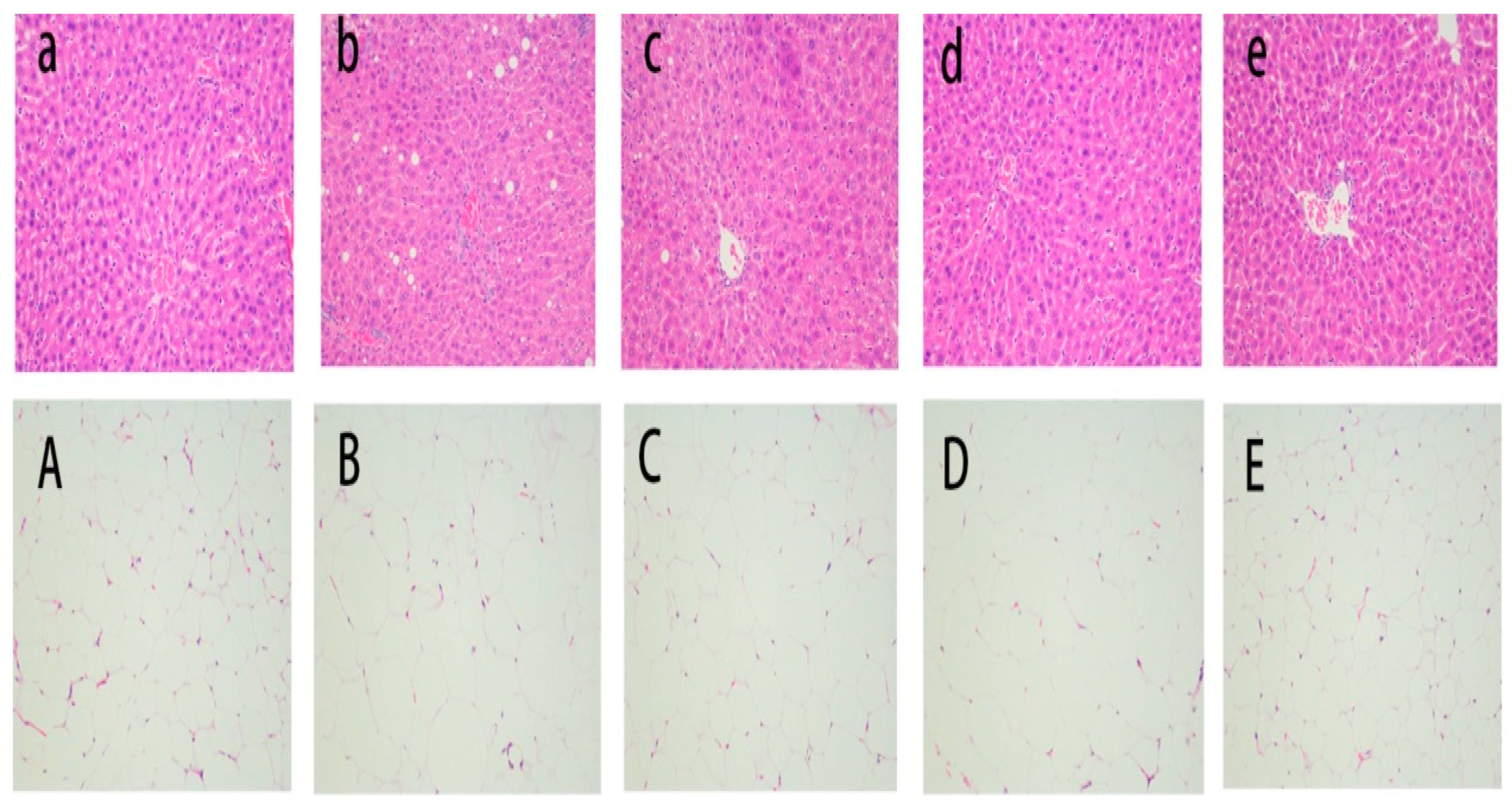
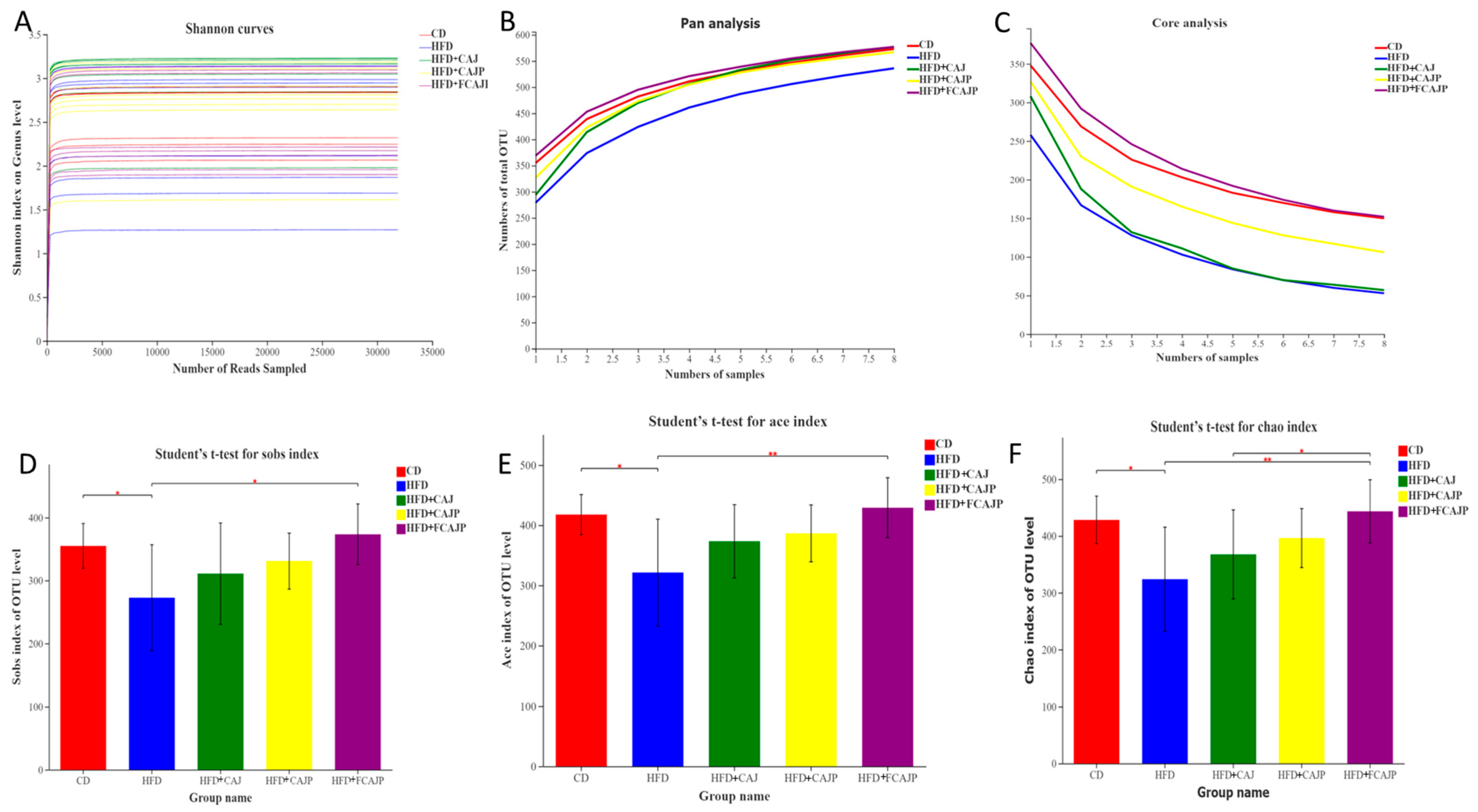
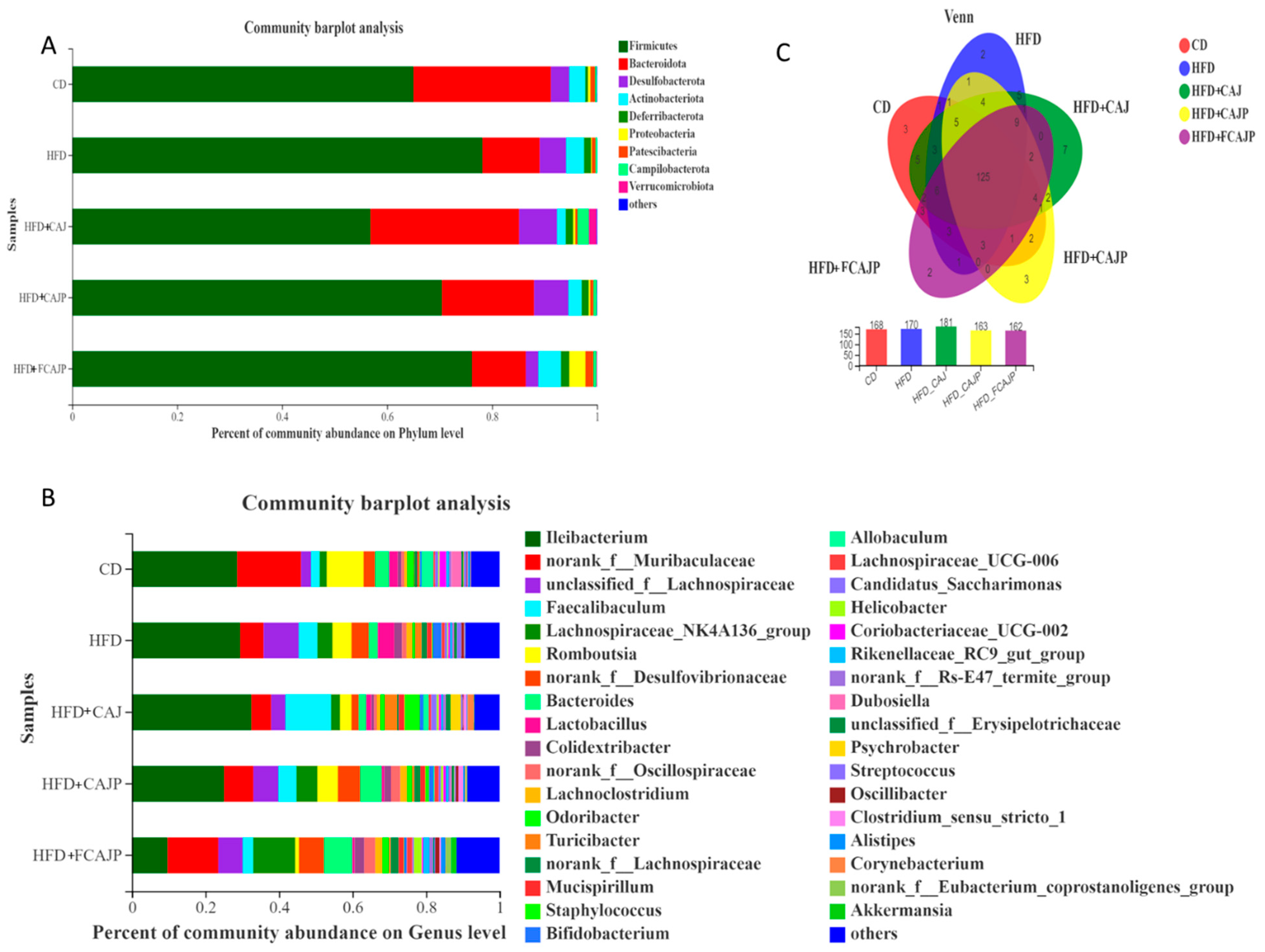
| CD | HFD | HFD + CAJ | HFD + CAJP | HFD + FCAJP | |
|---|---|---|---|---|---|
| Food intake (g/day) | 2.09 ± 0.20 a | 2.15 ± 0.15 a | 2.01 ± 0.23 a | 1.99 ± 0.14 a | 2.05 ± 0.16 a |
| Water intake (mL) | 3.25 ± 0.31 a | 3.21 ± 0.32 a | 3.43 ± 0.22 a | 3.39 ± 0.41 a | 3.22 ± 0.31 a |
| Epididymal fat (g) | 0.753 ± 0.13 a | 2.01 ± 0.94 b | 1.44 ± 0.16 c | 1.304 ± 0.22 c | 0.84 ± 0.56 a |
| Liver index (%) | 3.24 ± 0.34 a | 3.76 ± 0.18 b | 3.35 ± 0.18 a | 3.22 ± 0.28 a | 3.18 ± 0.32 a |
| TC (mmol/L) | 3.36 ± 0.39 a | 4.73 ± 0.33 b | 4.50 ± 0.61 b | 4.42 ± 1.00 b | 3.45 ± 0.37 a |
| TG (mmol/L) | 0.52 ± 0.03 a | 0.75 ± 0.07 c | 0.58 ± 0.08 ab | 0.55 ± 0.05 ab | 0.59 ± 0.07 ab |
| HDL-C(mmol/L) | 2.82 ± 0.23 b | 2.54 ± 0.20 a | 2.57 ± 0.28 a | 2.51 ± 0.44 a | 2.72 ± 0.14 b |
| LDL-C (mmol/L) | 0.33 ± 0.04 b | 0.52 ± 0.04 a | 0.45 ± 0.03 c | 0.44 ± 0.05 c | 0.35 ± 0.06 b |
| Liver GSH-Px | 555.68 ± 18.54 a | 548.31 ± 32.32 a | 629.48 ± 28.12 b | 690.83 ± 45.74 c | 612.37 ± 28.25 b |
| Liver SOD (U/mgprot) | 1100.96 ± 61.81 a | 1027.59 ± 69.32 a | 1093.68 ± 108.28 a | 1358.18 ± 67.77 b | 1303.89 ± 57.71 b |
| Liver MDA (nmol/mgprot) | 0.77 ± 0.06 a | 0.85 ± 0.22 b | 0.75 ± 0.06 a | 0.49 ± 0.11 c | 0.51 ± 0.06 c |
| CD | HFD | HFD + CAJ | HFD + CAJP | HFD + FCAJP | |
|---|---|---|---|---|---|
| Acetic acid | 687.74 ± 100.33 a | 489.94 ± 89.35 b | 550.39 ± 100.27 c | 570.39 ± 90.29 c | 620.67 ± 121,94 ac |
| Propionic acid | 101.02 ± 51.24 a | 70.44 ± 21.46 b | 79.33 ± 16.76 b | 77.98 ± 10.57 b | 87.35 ± 16.44 c |
| Isobutyric acid | 76.64 ± 16.54 a | 52.68 ± 14.41 b | 60.49 ± 13.11 c | 68.44 ± 10.47 ac | 74.98 ± 12.21 a |
| Butyric acid | 87.21 ± 13.72 a | 73.51 ± 19.22 b | 80.35 ± 11.53 ab | 82.42 ± 15.14 a | 90.21 ± 19.72 a |
| Isovaleric acid | 71.59 ± 11.65 a | 51.38 ± 10.45 b | 62.63 ± 10.15 c | 60.59 ± 15.65 c | 66.89 ± 11.25 ac |
| N-valeric acid | 114.14 ± 16.52 a | 94.79 ± 23.52 b | 99.14 ± 12.58 b | 107.34 ± 13.82 ac | 116.14 ± 20.12 ac |
| LBP(ng/mL) | 10.36 ± 1.53 a | 16.05 ± 1.93 b | 11.68 ± 1.75 c | 11.41 ± 1.39 c | 10.99 ± 0.86 ac |
| LPS(ng/mL) | 160.69 ± 15.93 a | 212.97 ± 20.11 b | 195.86 ± 21.11 c | 200.52 ± 17.43 c | 189.31 ± 10.44 c |
| FC (pg/g) | 51.01 ± 7.87 a | 57.6 ± 13.06 b | 54.25 ± 9.65 ab | 49.77 ± 13.35 ac | 44.77 ± 9.93 d |
| sIgA(μg/g) | 0.92 ± 0.21 a | 0.97 ± 0.38 b | 0.62 ± 0.31 c | 0.81 ± 0.42 d | 0.51 ± 0.27 e |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, M.; Zhang, M.; Wang, X.; Bai, X.; Yue, T.; Gao, Z. Cloudy Apple Juice Fermented by Lactobacillus Prevents Obesity via Modulating Gut Microbiota and Protecting Intestinal Tract Health. Nutrients 2021, 13, 971. https://doi.org/10.3390/nu13030971
Han M, Zhang M, Wang X, Bai X, Yue T, Gao Z. Cloudy Apple Juice Fermented by Lactobacillus Prevents Obesity via Modulating Gut Microbiota and Protecting Intestinal Tract Health. Nutrients. 2021; 13(3):971. https://doi.org/10.3390/nu13030971
Chicago/Turabian StyleHan, Mengzhen, Meina Zhang, Xiaowei Wang, Xue Bai, Tianli Yue, and Zhenpeng Gao. 2021. "Cloudy Apple Juice Fermented by Lactobacillus Prevents Obesity via Modulating Gut Microbiota and Protecting Intestinal Tract Health" Nutrients 13, no. 3: 971. https://doi.org/10.3390/nu13030971
APA StyleHan, M., Zhang, M., Wang, X., Bai, X., Yue, T., & Gao, Z. (2021). Cloudy Apple Juice Fermented by Lactobacillus Prevents Obesity via Modulating Gut Microbiota and Protecting Intestinal Tract Health. Nutrients, 13(3), 971. https://doi.org/10.3390/nu13030971





