Using a Paleo Ratio to Assess Adherence to Paleolithic Dietary Recommendations in a Randomized Controlled Trial of Individuals with Type 2 Diabetes
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Study Population
2.3. Dietary Intervention
2.4. Food Records
2.5. Calculation of the Paleo Ratio and Other Ratios
2.6. Body Composition, Anthropometric, and Biochemical Measurements
2.7. Observational Group
2.8. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Global Report on Diabetes. 2016. Available online: https://apps.who.int/iris/bitstream/handle/10665/204871/9789241565257_eng.pdf (accessed on 27 December 2020).
- Gregg, E.W.; Cheng, Y.J.; Saydah, S.; Cowie, C.; Garfield, S.; Geiss, L.; Barker, L. Trends in Death Rates among U.S. Adults With and Without Diabetes Between 1997 and 2006. Find. Natl. Health Interview Surv. 2012, 35, 1252–1257. [Google Scholar]
- Nield, L.; Moore, H.; Hooper, L.; Cruickshank, K.; Vyas, A.; Whittaker, V.; Summerbell, C.D. Dietary advice for treatment of type 2 diabetes mellitus in adults. Cochrane Database Syst. Rev. 2007, CD004097. [Google Scholar] [CrossRef] [PubMed]
- Esposito, K.; Maiorino, M.I.; Bellastella, G.; Chiodini, P.; Panagiotakos, D.; Giugliano, D. A journey into a Mediterranean diet and type 2 diabetes: A systematic review with meta-analyses. BMJ Open 2015, 5, e008222. [Google Scholar] [CrossRef] [PubMed]
- Bolla, A.M.; Caretto, A.; Laurenzi, A.; Scavini, M.; Piemonti, L. Low-carb and ketogenic diets in type 1 and type 2 diabetes. Nutrients 2019, 11, 962. [Google Scholar] [CrossRef]
- Dietary Treatment of Diabetes; SBU report no 201; Swedish Council on Health Technology Assessment (SBU): Stockholm, Swedish, 2010. (In Swedish)
- Fats and Fatty Acids in Human Nutrition: Report of an Expert Consultation. FAO Food and Nutrition Paper 91; Food and Agriculture Organization of the United Nations: Rome, Italy, 2010; Available online: http://www.fao.org/3/a-i1953e.pdf (accessed on 27 December 2020).
- World Health Organization. Guideline: Sugars Intake for Adults and Children; World Health Organization: Geneva, Switzerland, 2015; Available online: https://apps.who.int/iris/bitstream/handle/10665/149782/9789241549028_eng.pdf?sequence=1 (accessed on 27 December 2020).
- Morin, E.; Meier, J.; El Guennouni, K.; Moigne, A.M.; Lebreton, L.; Rusch, L.; Valensi, P.; Conolly, J.; Cochard, D. New evidence of broader diets for archaic. Sci. Adv. 2019, 5, eaav9106. [Google Scholar] [CrossRef]
- Hardy, K. Paleomedicine and the use of plant secondary compounds in the Paleolithic and Early Neolithic. Evol. Anthropol. 2019, 28, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Eaton, S.B.; Konner, M. Paleolithic nutrition. A consideration of its nature and current implications. N. Engl. J. Med. 1985, 312, 283–289. [Google Scholar] [CrossRef] [PubMed]
- Lindeberg, S.; Cordain, L.; Eaton, S.B. Biological and Clinical Potential of a Palaeolithic Diet. J. Nutr. Environ. Med. 2003, 13, 149–160. [Google Scholar] [CrossRef]
- Cordain, L.; Miller, J.B.; Eaton, S.B.; Mann, N.; Holt, S.H.; Speth, J.D. Plant-animal subsistence ratios and macronutrient energy estimations in worldwide hunter-gatherer diets. Am. J. Clin. Nutr. 2000, 71, 682–692. [Google Scholar] [CrossRef] [PubMed]
- Lindeberg, S.; Jönsson, T.; Granfeldt, Y.; Borgstrand, E.; Soffman, J.; Sjöström, K.; Ahrén, B. A Palaeolithic diet improves glucose tolerance more than a Mediterranean-like diet in individuals with ischaemic heart disease. Diabetologia 2007, 50, 1795–1807. [Google Scholar] [CrossRef]
- Frassetto, L.A.; Schloetter, M.; Mietus-Synder, M.; Morris, R.C.; Sebastian, A. Metabolic and physiologic improvements from consuming a paleolithic, hunter-gatherer type diet. Eur. J. Clin. Nutr. 2009, 63, 947–955. [Google Scholar] [CrossRef] [PubMed]
- Mellberg, C.; Sandberg, S.; Ryberg, M.; Eriksson, M.; Brage, S.; Larsson, C.; Olsson, T.; Lindahl, B. Long-term effects of a Palaeolithic-type diet in obese postmenopausal women: A 2-year randomized trial. Eur. J. Clin. Nutr. 2014, 68, 350–357. [Google Scholar] [CrossRef] [PubMed]
- Jönsson, T.; Granfeldt, Y.; Ahrén, B.; Branell, U.C.; Pålsson, G.; Hansson, A.; Söderström, M.; Lindeberg, S. Beneficial effects of a Paleolithic diet on cardiovascular risk factors in type 2 diabetes: A randomized cross-over pilot study. Cardiovasc. Diabetol. 2009, 8, 35. [Google Scholar] [CrossRef]
- Masharani, U.; Sherchan, P.; Schloetter, M.; Stratford, S.; Xiao, A.; Sebastian, A.; Kennedy, M.N.; Frassetto, L. Metabolic and physiologic effects from consuming a hunter-gatherer (Paleolithic)-type diet in type 2 diabetes. Eur. J. Clin. Nutr. 2015, 69, 944–948. [Google Scholar] [CrossRef] [PubMed]
- Österdahl, M.; Kocturk, T.; Koochek, A.; Wändell, P.E. Effects of a short-term intervention with a paleolithic diet in healthy volunteers. Eur. J. Clin. Nutr. 2008, 62, 682–685. [Google Scholar] [CrossRef]
- Ghaedi, E.; Mohammadi, M.; Mohammadi, H.; Ramezani-Jolfaie, N.; Malekzadeh, J.; Hosseinzadeh, M.; Salehi-Abargouei, A. Effects of a Paleolithic Diet on Cardiovascular Disease Risk Factors: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2019, 10, 634–646. [Google Scholar] [CrossRef] [PubMed]
- Feng, Q.; Fan, S.; Wu, Y.; Zhou, D.; Zhao, R.; Liu, M.; Song, Y. Adherence to the dietary approaches to stop hypertension diet and risk of stroke: A meta-analysis of prospective studies. Medicine 2018, 97, e12450. [Google Scholar] [CrossRef] [PubMed]
- Otten, J.; Stomby, A.; Waling, M.; Isaksson, A.; Tellström, A.; Lundin-Olsson, L.; Brage, S.; Ryberg, M.; Svensson, M.; Olsson, T. Benefits of a Paleolithic diet with and without supervised exercise on fat mass, insulin sensitivity, and glycemic control: A randomized controlled trial in individuals with type 2 diabetes. Diabetes Metab. Res. Rev. 2017, 33, e2828. [Google Scholar] [CrossRef]
- Livsmedelsverket. Portionsguide; Livsmedelsverket: Uppsala, Uppsala Lan, Sweden, 2009; ISBN 9177141911. [Google Scholar]
- Jacobs, D.R.; Tapsell, L.C. Food, not nutrients, is the fundamental unit in nutrition. Nutr. Rev. 2007, 65, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Amcoff, E.; Edberg, A.; Barbieri, H.E.; Lindroos, A.K.; Nälsén, C.; Pearson, M. Riksmaten vuxna 2010-11. Livsmedels- och näringsintag Bland Vuxna i Sverige. Livsmedelsverket. 2012. Available online: https://www.livsmedelsverket.se/globalassets/publikationsdatabas/rapporter/2014/riksmaten-vuxna-2010-11-metodrapport--rapport-8---2014.pdf (accessed on 27 December 2020).
- Subar, A.F.; Freedman, L.S.; Tooze, J.A.; Kirkpatrick, S.I.; Boushey, C.; Neuhouser, M.L.; Thompson, F.E.; Potischman, N.; Guenther, P.M.; Tarasuk, V.; et al. Addressing Current Criticism Regarding the Value of Self-Report Dietary Data. J. Nutr. 2015, 145, 2639–2645. [Google Scholar] [CrossRef] [PubMed]
- Blomquist, C.; Chorell, E.; Ryberg, M.; Mellberg, C.; Worrsjö, E.; Makoveichuk, E.; Larsson, C.; Lindahl, B.; Olivecrona, G.; Olsson, T. Decreased lipogenesis-promoting factors in adipose tissue in postmenopausal women with overweight on a Paleolithic-type diet. Eur. J. Nutr. 2018, 57, 2877–2886. [Google Scholar] [CrossRef]
- Bradberry, J.C.; Hilleman, D.E. Overview of omega-3 Fatty Acid therapies. J. Formul. Manag. 2013, 38, 681–691. [Google Scholar]
- Bang, H.O.; Dyerberg, J.; Sinclair, H.M. The composition of the Eskimo food in north western Greenland. Am. J. Clin. Nutr. 1980, 33, 2657–2661. [Google Scholar] [CrossRef] [PubMed]
- Cordain, L.; Eaton, S.B.; Miller, J.B.; Mann, N.; Hill, K. The paradoxical nature of hunter-gatherer diets: Meat-based, yet non-atherogenic. Eur. J. Clin. Nutr. 2002, 56, S42–S52. [Google Scholar] [CrossRef] [PubMed]
- Fechner, E.; Smeets, E.T.; Schrauwen, P.; Mensink, R.P. The Effects of Different Degrees of Carbohydrate Restriction and Carbohydrate Replacement on Cardiometabolic Risk Markers in Humans-A Systematic Review and Meta-Analysis. Nutrients 2020, 12, 991. [Google Scholar] [CrossRef] [PubMed]
- Ryberg, M.; Sandberg, S.; Mellberg, C.; Stegle, O.; Lindahl, B.; Larsson, C.; Hauksson, J.; Olsson, T. A Palaeolithic-type diet causes strong tissue-specific effects on ectopic fat deposition in obese postmenopausal women. J. Intern. Med. 2013, 274, 67–76. [Google Scholar] [CrossRef] [PubMed]
- Boers, I.; Muskiet, F.A.; Berkelaar, E.; Schut, E.; Penders, R.; Hoenderdos, K.; Wichers, H.J.; Jong, M.C. Favourable effects of consuming a Palaeolithic-type diet on characteristics of the metabolic syndrome: A randomized controlled pilot-study. Lipids Health Dis. 2014, 13, 160. [Google Scholar] [CrossRef]
- O’dea, K. Marked improvement in carbohydrate and lipid metabolism in diabetic Australian aborigines after temporary reversion to traditional lifestyle. Diabetes 1984, 33, 596–603. [Google Scholar] [CrossRef]
- Goldberg, I.J. Clinical review 124: Diabetic dyslipidemia: Causes and consequences. J. Clin. Endocrinol. Metab. 2001, 86, 965–971. [Google Scholar] [CrossRef] [PubMed]
- McTaggart, F.; Jones, P. Effects of statins on high-density lipoproteins: A potential contribution to cardiovascular benefit. Cardiovasc. Drugs Ther. 2008, 22, 321–338. [Google Scholar] [CrossRef] [PubMed]
- Blumenthal, J.A.; Babyak, M.A.; Hinderliter, A.; Watkins, L.L.; Craighead, L.; Lin, P.H.; Caccia, C.; Johnson, J.; Waugh, R.; Sherwood, A. Effects of the DASH diet alone and in combination with exercise and weight loss on blood pressure and cardiovascular biomarkers in men and women with high blood pressure: The ENCORE study. Arch. Intern. Med. 2010, 170, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Appel, L.J.; Champagne, C.M.; Harsha, D.W.; Cooper, L.S.; Obarzanek, E.; Elmer, P.J.; Stevens, V.J.; Vollmer, W.M.; Lin, P.H.; Svetkey, L.P.; et al. Effects of comprehensive lifestyle modification on blood pressure control: Main results of the PREMIER clinical trial. JAMA 2003, 289, 2083–2093. [Google Scholar] [PubMed]
- Appel, L.J.; Moore, T.J.; Obarzanek, E.; Vollmer, W.M.; Svetkey, L.P.; Sacks, F.M.; Bray, G.A.; Vogt, T.M.; Cutler, J.A.; Windhauser, M.M.; et al. A clinical trial of the effects of dietary patterns on blood pressure. DASH Collaborative Research Group. N. Engl. J. Med. 1997, 336, 1117–1124. [Google Scholar] [CrossRef] [PubMed]
- Yamaoka, K.; Tango, T. Efficacy of lifestyle education to prevent type 2 diabetes: A meta-analysis of randomized controlled trials. Diabetes Care 2005, 28, 2780–2786. [Google Scholar] [CrossRef]
- Hamman, R.F.; Wing, R.R.; Edelstein, S.L.; Lachin, J.M.; Bray, G.A.; Delahanty, L.; Hoskin, M.; Kriska, A.M.; Mayer-Davis, E.J.; Pi-Sunyer, X.; et al. Effect of weight loss with lifestyle intervention on risk of diabetes. Diabetes Care 2006, 29, 2102–2107. [Google Scholar] [CrossRef] [PubMed]
- Trichopoulou, A.; Costacou, T.; Bamia, C.; Trichopoulos, D. Adherence to a Mediterranean diet and survival in a Greek population. N. Engl. J. Med. 2003, 348, 2599–2608. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.M.M.; Fung, T.T.; Steck, S.E.; Zhang, J.; Hazlett, L.J.; Han, K.; Lee, S.H.; Merchant, A.T. Diet Quality and Mortality Risk in Metabolically Obese Normal-Weight Adults. Mayo Clin. Proc. 2016, 91, 1372–1383. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Bogensberger, B.; Hoffmann, G. Diet Quality as Assessed by the Healthy Eating Index, Alternate Healthy Eating Index, Dietary Approaches to Stop Hypertension Score, and Health Outcomes: An Updated Systematic Review and Meta-Analysis of Cohort Studies. J. Acad. Nutr. Diet. 2018, 118, 74–100.e11. [Google Scholar] [CrossRef]
- Milà-Villarroel, R.; Bach-Faig, A.; Puig, J.; Puchal, A.; Farran, A.; Serra-Majem, L.; Carrasco, J.L. Comparison and evaluation of the reliability of indexes of adherence to the Mediterranean diet. Public Health Nutr. 2011, 14, 2338–2345. [Google Scholar] [CrossRef]
- Rutishauser, I.H. Dietary intake measurements. Public Health Nutr. 2005, 8, 1100–1107. [Google Scholar] [CrossRef] [PubMed]
- Ishii, Y.; Ishihara, J.; Takachi, R.; Shinozawa, Y.; Imaeda, N.; Goto, C.; Wakai, K.; Takahashi, T.; Iso, H.; Nakamura, K.; et al. Comparison of weighed food record procedures for the reference methods in two validation studies of food frequency questionnaires. J. Epidemiol. 2017, 27, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Ortega, R.M.; Pérez-Rodrigo, C.; López-Sobaler, A.M. Dietary assessment methods: Dietary records. Nutr. Hosp. 2015, 31 (Suppl. 3), 38–45. [Google Scholar] [PubMed]
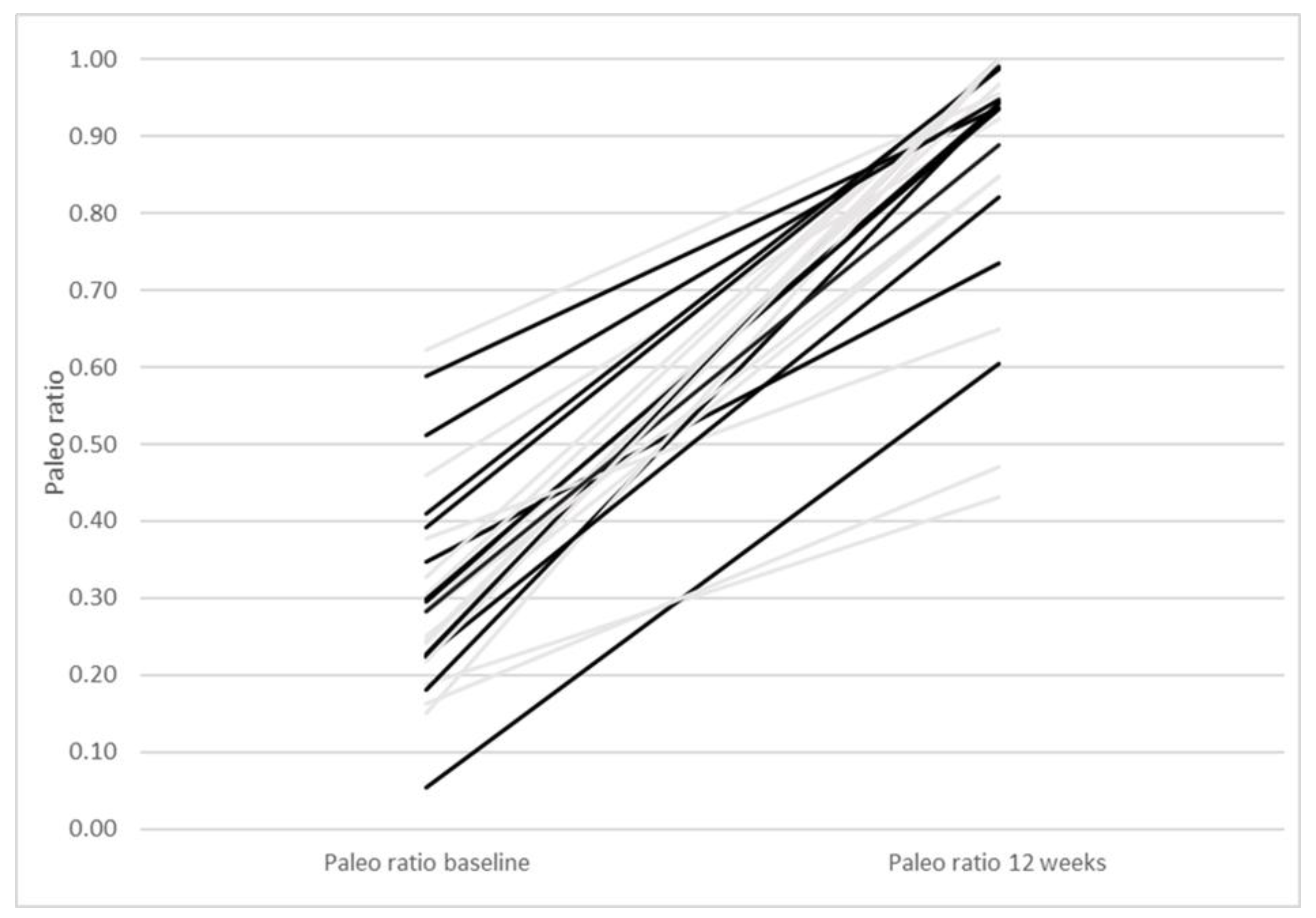
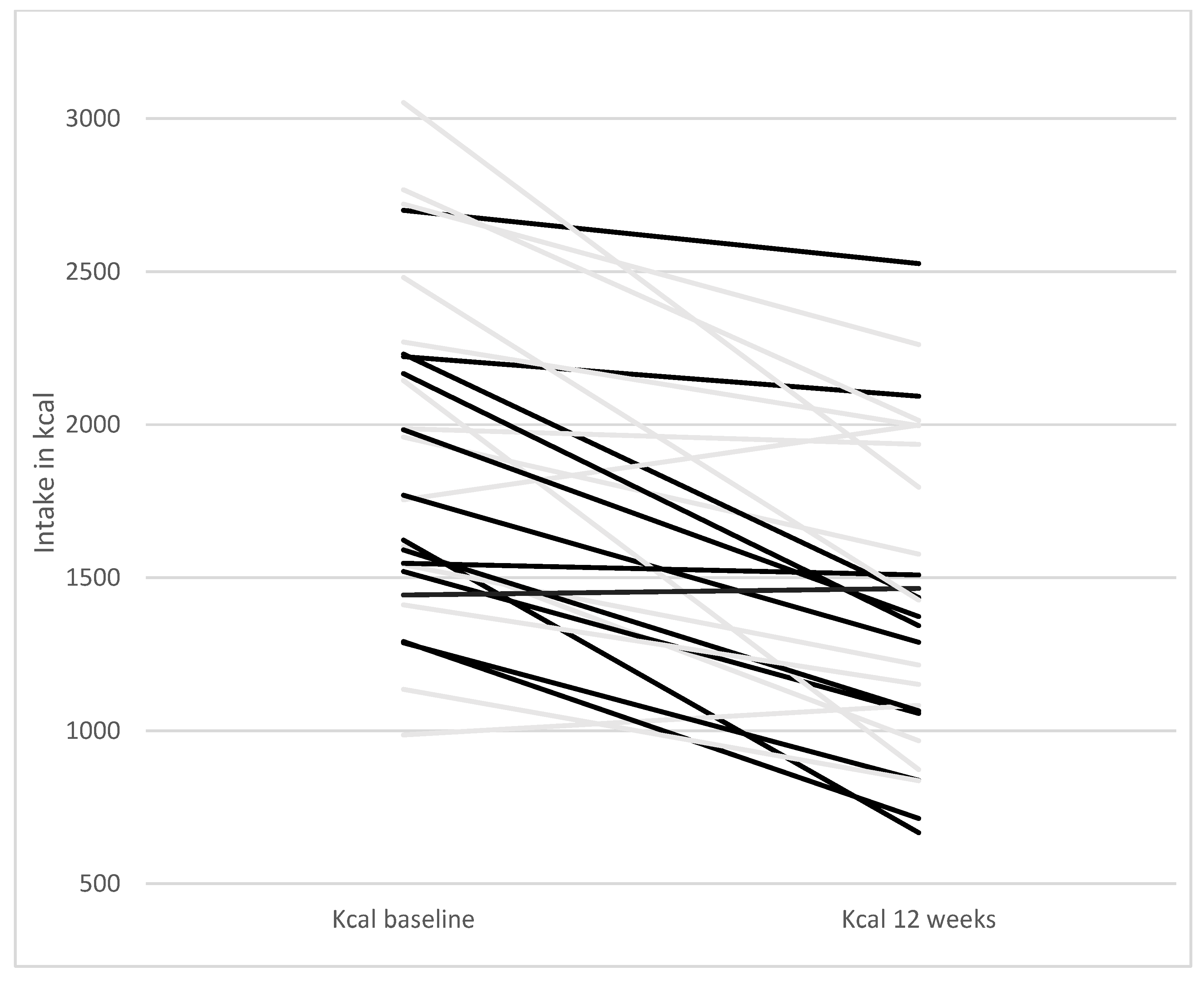
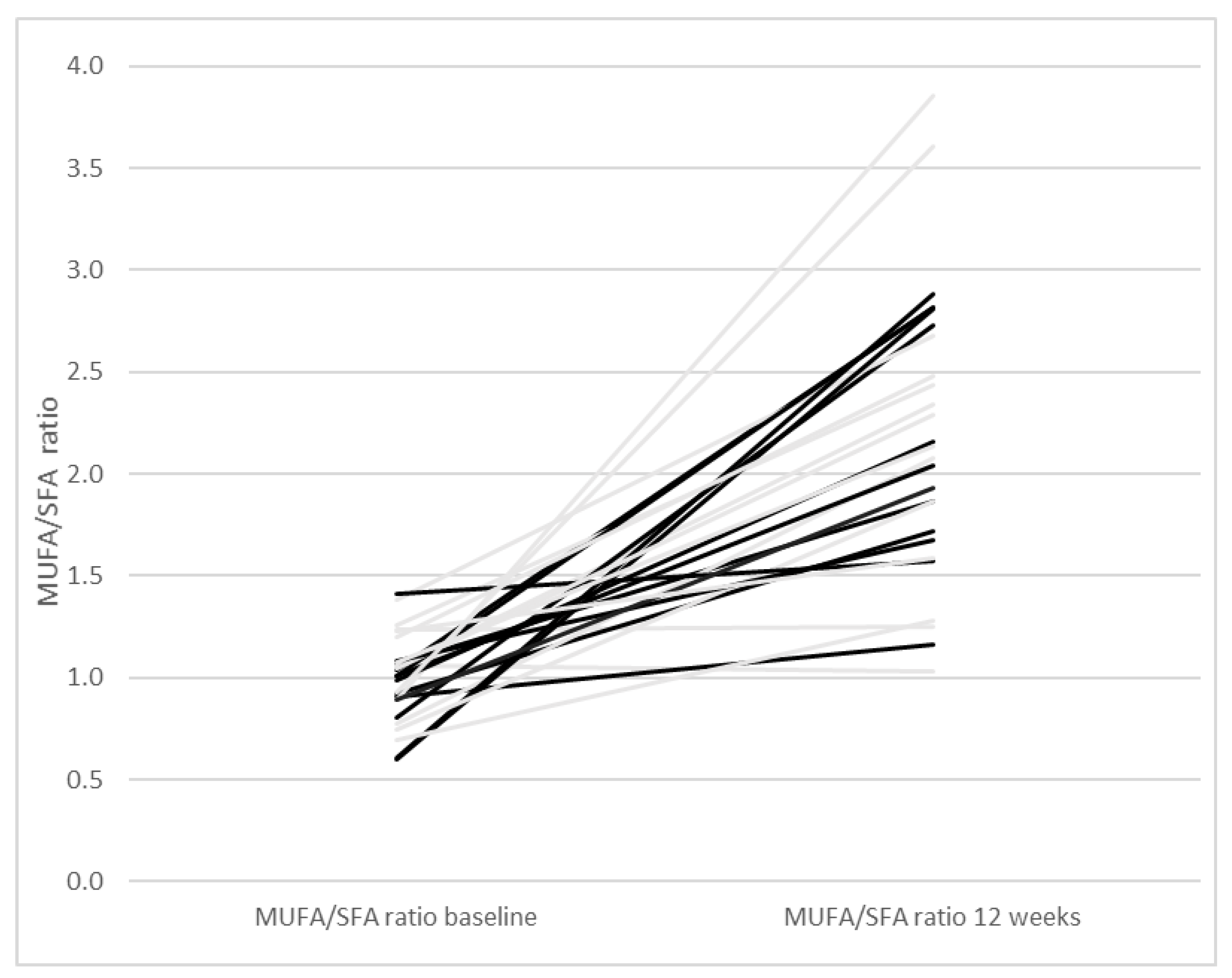
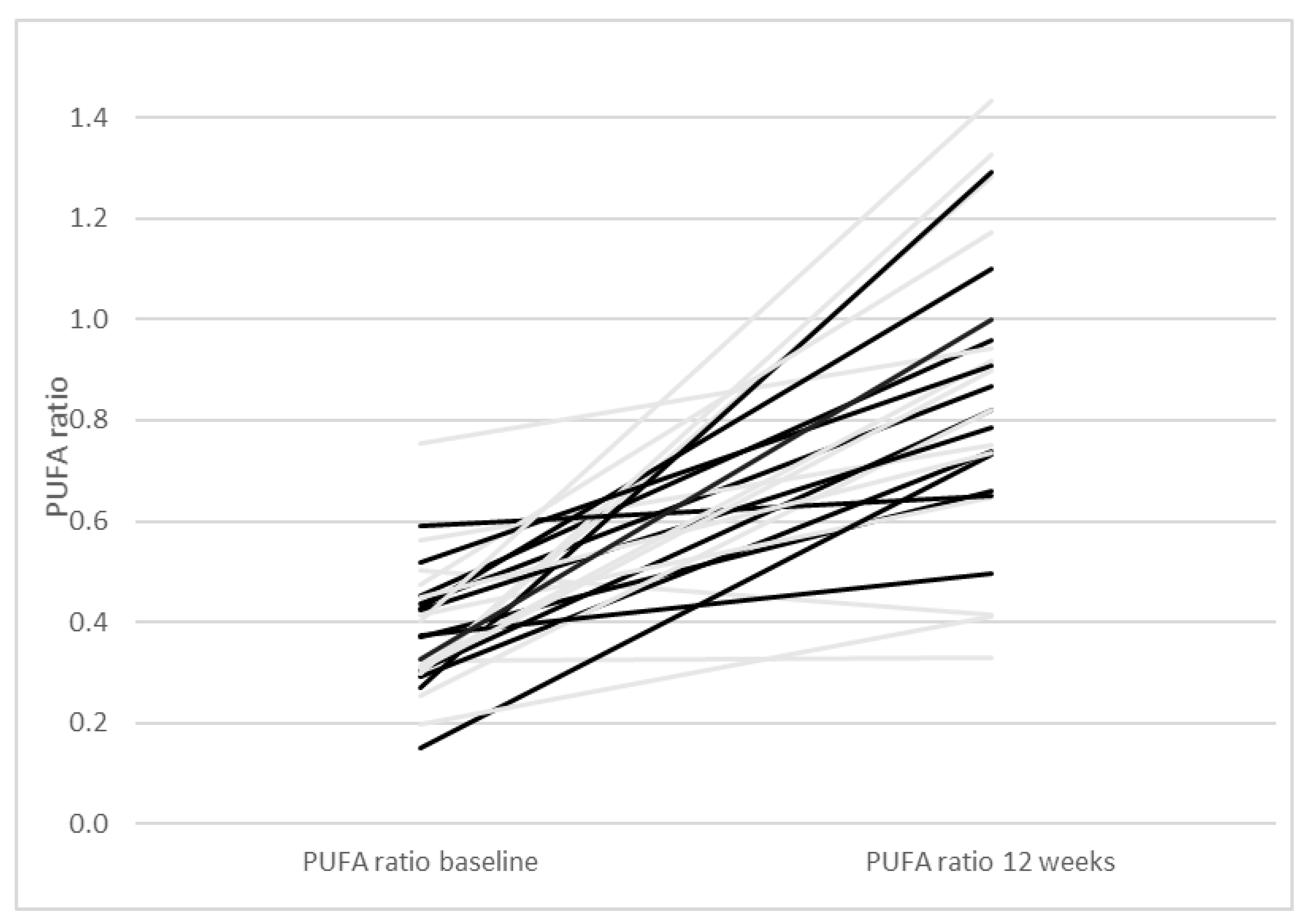
| Paleolithic Diet (Only Diet) (n = 14) | Paleolithic Diet (Diet + Exercise) (n = 13) | Paleolithic Diet (All Participants) (n = 27) | |
|---|---|---|---|
| Age (years) | 61 (52–64) | 61 (58–67) | 61 (57–65) |
| Women/Men (n) | 5/9 | 4/9 | 9/18 |
| Diabetes duration (years) | 3 (1–6) | 6 (1–8) | 3 (1–8) |
| BMI (kg/m2) | 31 (29–34) | 31 (29–35) | 31 (29–33) |
| Hba1c (mmol/mol, n = 26) | 54 (48–57) | 57 (50–66) | 55 (48–58) |
| Diabetes treatment (n) | |||
| Metformin | 10 | 9 | 19 |
| No medication | 4 | 4 | 8 |
| Other treatments (n) | |||
| ACEI or ARB | 9 | 9 | 18 |
| Diuretics | 6 | 4 | 10 |
| Calcium channel blocker | 4 | 4 | 8 |
| Statin | 6 | 7 | 13 |
| Antiplatelet drug | 2 | 3 | 5 |
| Other | 7 | 2 | 9 |
| Paleolithic Food Groups | Non-Paleolithic Food Groups |
|---|---|
| Berries | Beer |
| Cacao | Bouillon and salt |
| Fish/shellfish | Butter |
| Fresh juice | Bread |
| Fruits | Cheese |
| Honey | Fish/shellfish-based products (Ex; breaded fish) |
| Meats with added salt | Grains |
| Meats without added salt | Ice cream |
| Mushroom protein | Legumes |
| Mushrooms | Meat based products |
| Poultry | Milk/cream |
| Root vegetables (excluding potatoes) | Organ meats |
| Sauces (such as homemade mayonnaise) | Pasta |
| Vegetable products (including only items from Paleolithic food groups) | Preserves |
| Vegetables | Products containing sugar |
| Vinegar | Sauces (such as bearnaise) |
| Sausages | |
| Snacks | |
| Soup | |
| Spirits | |
| Sweet drinks | |
| Vegetable dishes(e.g., vegetable dishes containing non-paleolithic food items) | |
| Vegetable products (e.g., salads with dairy) | |
| Yoghurt | |
| Paleolithic Food Included with Restrictions | Food Excluded |
| Wine, one glass/week (150 g) | Wine over restricted amount |
| Rapeseed or olive oil, maximum 15 g/day | Rapeseed/olive oil over restricted amount |
| Potatoes, 1 medium sized/day | Potato-based products and potatoes exceeding restricted amount |
| Nuts, 60 g/day | Nuts above restricted amount |
| Eggs, 1–2/day, max 5/week | Egg based products and eggs exceeding restricted amount |
| Dried fruit, 130 g/day | Dried fruit exceeding restricted amount |
| Coffee/tea, 300 g/day | Instant coffee, or coffee exceeding restricted amount. |
| MUFA/SFA Ratio | PUFA/SFA Ratio | |||
|---|---|---|---|---|
| B (95% CI) | p-Value | B (95% CI) | p-Value | |
| Anthropometry | ||||
| Fat mass, kg | −1.2 (−2.5; 0.1) | 0.075 | −1.7 (−5.0; 1.6) | 0.287 |
| BMI, kg/m2 | −0.6 (−1.1; 0.1) | 0.080 | −0.9 (−2.5; 0.7) | 0.237 |
| Waist circumference, cm | −1.9 (−4.2; 0.4) | 0.108 | −3.9 (−9.8; 1.9) | 0.178 |
| Lean mass, kg | −1.5 (−3.4; 0.5) | 0.129 | −2.7 (−7.5; 2.1) | 0.260 |
| Blood pressure | ||||
| SBP, mmHg | −4 (−10; 1) | 0.099 | −9 (−22; 4) | 0.156 |
| DBP, mmHg | 0 (−3; 3) | 0.931 | −5 (−13; 3) | 0.191 |
| Blood lipids | ||||
| Triglycerides, mmol/L | −0.5 (−0.8; −0.2) | 0.006 | −1.1 (−1.9; −0.3) | 0.011 |
| HDL, mmol/L | 0.0 (0.0; 0.1) | 0.198 | 0.0 (−0.1; 0.2) | 0.767 |
| LDL, mmol/L | −0.1 (−0.4; 0.2) | 0.630 | 0.1 (−0.6; 0.9) | 0.717 |
| Glucose homeostasis | ||||
| HbA1c, mmol/mol | −11 −7; 5) | 0.788 | −22 (−17; 13) | 0.824 |
| Fasting P-insulin, mlU/L | −1.8 (−4.4; 0.7) | 0.154 | −5.6 (−11.8; 0.5) | 0.072 |
| Fasting glucose, mmol/L | −0.2 (−1.2; 0.8) | 0.691 | −0.7 (−3.2; 1.9) | 0.603 |
| Paleo Ratio—Model 1 | Paleo Ratio—Model 2 | Paleo Ratio—Model 3 | ||||
|---|---|---|---|---|---|---|
| B (95% CI) | p-Value | B (95% CI) | p-Value | B (95% CI) | p-Value | |
| Anthropometry | ||||||
| Fat mass, kg | −5.9 (−11.4; −0.5) | 0.034 | −5.5 (−11.0; −0.04) | 0.048 | ||
| BMI, kg/m2 | −2.6 (−5.2; 0.04) | 0.053 | −2.7 (−5.4; −0.02) | 0.049 | ||
| Waist circumference, cm | −9.2 (−18.9; 0.5) | 0.061 | −9.8 (−19.6; −0.02) | 0.0496 | ||
| Lean mass, kg | −0.8 (−6.5; 5.0) | 0.786 | −1.5 (−6.7; 3.7) | 0.561 | ||
| Blood pressure | ||||||
| SBP, mmHg | −23 (−44; −3) | 0.029 | −24 (−46; −3) | 0.028 | −15 (−35; 4) | 0.119 |
| DBP, mmHg | −1 (−14; 12) | 0.865 | 0 (−14; 14) | 0.958 | ||
| Blood lipids | ||||||
| Triglycerides, mmol/L | −1.8 (−3.2; −0.4) | 0.016 | −1.7 (−3.2; −0.03) | 0.022 | −1.6 (−3.1; −0.03) | 0.046 |
| HDL, mmol/L | 0.0 (−0.2; 0.3) | 0.737 | 0.0 (−0.2; 0.3) | 0.754 | ||
| LDL, mmol/L | −0.9 (−2.2; 0.4) | 0.170 | −0.9 (−2.2; 0.4) | 0.183 | ||
| Glucose homeostasis | ||||||
| HbA1c, mmol/mol | −6 (−30; 19) | 0.645 | −5 (−31; 20) | 0.671 | ||
| Fasting P-insulin, mlU/L | −3.4 (−15.5; 8.8) | 0.569 | −3.2 (−15.5; 9.1) | 0.594 | ||
| Fasting glucose, mmol/L | 0.4 (−3.8; 4.7) | 0.833 | 0.5 (−3.9; 4.9) | 0.814 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mårtensson, A.; Stomby, A.; Tellström, A.; Ryberg, M.; Waling, M.; Otten, J. Using a Paleo Ratio to Assess Adherence to Paleolithic Dietary Recommendations in a Randomized Controlled Trial of Individuals with Type 2 Diabetes. Nutrients 2021, 13, 969. https://doi.org/10.3390/nu13030969
Mårtensson A, Stomby A, Tellström A, Ryberg M, Waling M, Otten J. Using a Paleo Ratio to Assess Adherence to Paleolithic Dietary Recommendations in a Randomized Controlled Trial of Individuals with Type 2 Diabetes. Nutrients. 2021; 13(3):969. https://doi.org/10.3390/nu13030969
Chicago/Turabian StyleMårtensson, Alexander, Andreas Stomby, Anna Tellström, Mats Ryberg, Maria Waling, and Julia Otten. 2021. "Using a Paleo Ratio to Assess Adherence to Paleolithic Dietary Recommendations in a Randomized Controlled Trial of Individuals with Type 2 Diabetes" Nutrients 13, no. 3: 969. https://doi.org/10.3390/nu13030969
APA StyleMårtensson, A., Stomby, A., Tellström, A., Ryberg, M., Waling, M., & Otten, J. (2021). Using a Paleo Ratio to Assess Adherence to Paleolithic Dietary Recommendations in a Randomized Controlled Trial of Individuals with Type 2 Diabetes. Nutrients, 13(3), 969. https://doi.org/10.3390/nu13030969








