Rehydration during Endurance Exercise: Challenges, Research, Options, Methods
Abstract
1. Introduction
2. Problem: Water and Salt Losses during Endurance Exercise
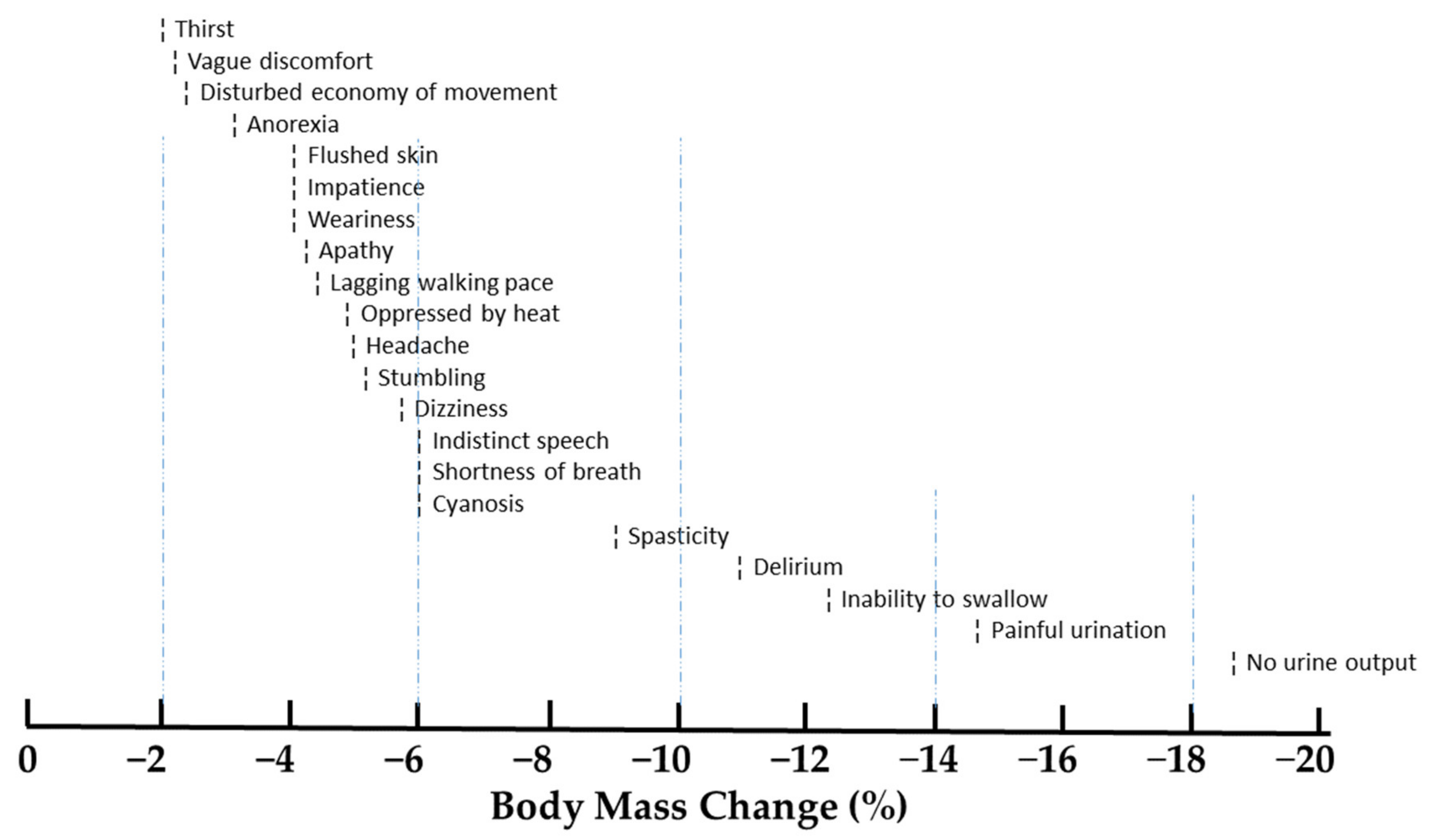
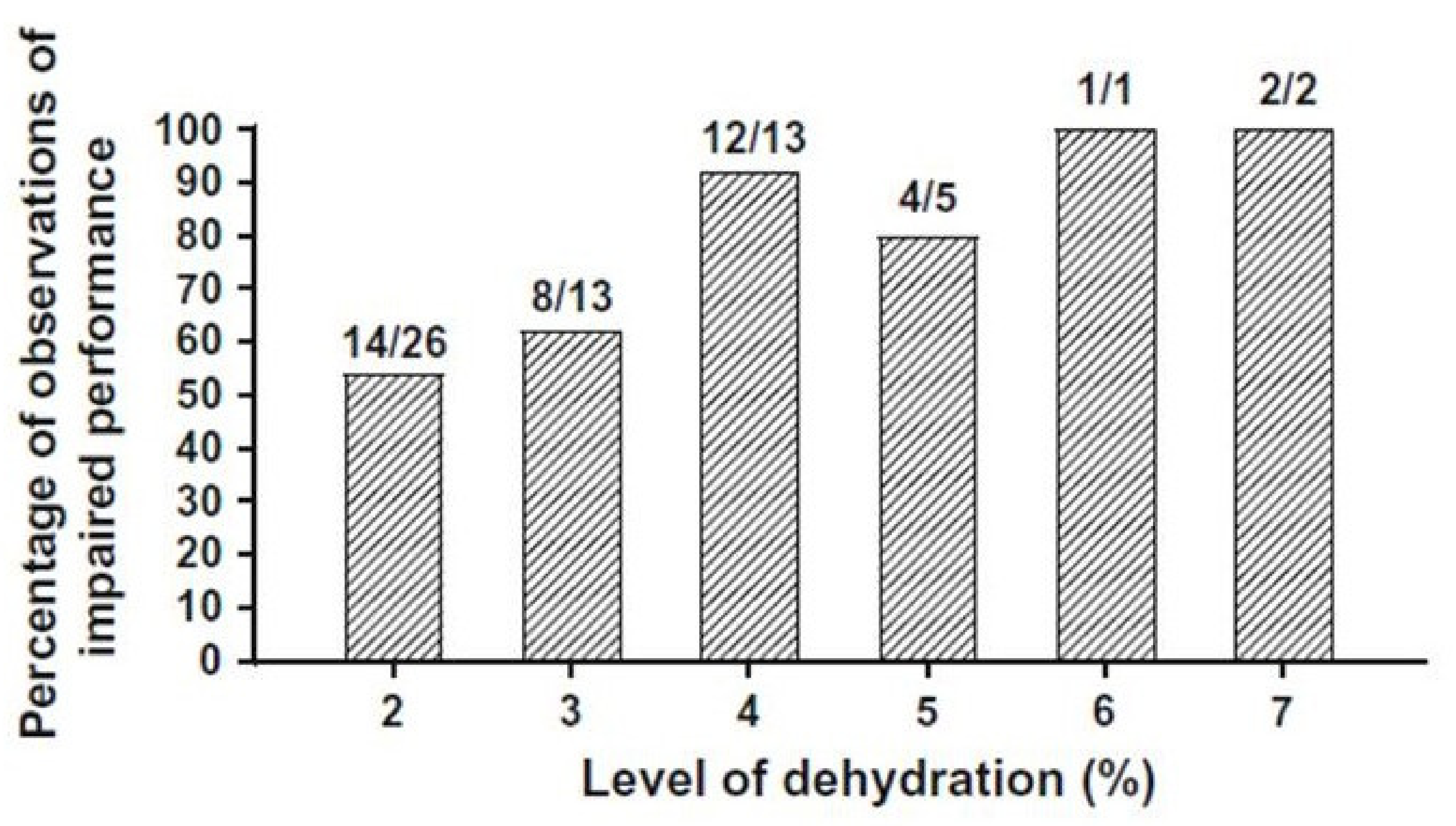
2.1. Effects of Dehydration on Endurance Exercise Performance
2.2. Effects of Dehydration on Symptomatology and Illness
2.2.1. Exercise Associated Collapse
2.2.2. Exertional Heat Illnesses
2.2.3. Kidney Dysfunction and Renal Stress
3. Problem: Overhydration during Endurance Exercise
3.1. Hyperhydration and Exercise Performance
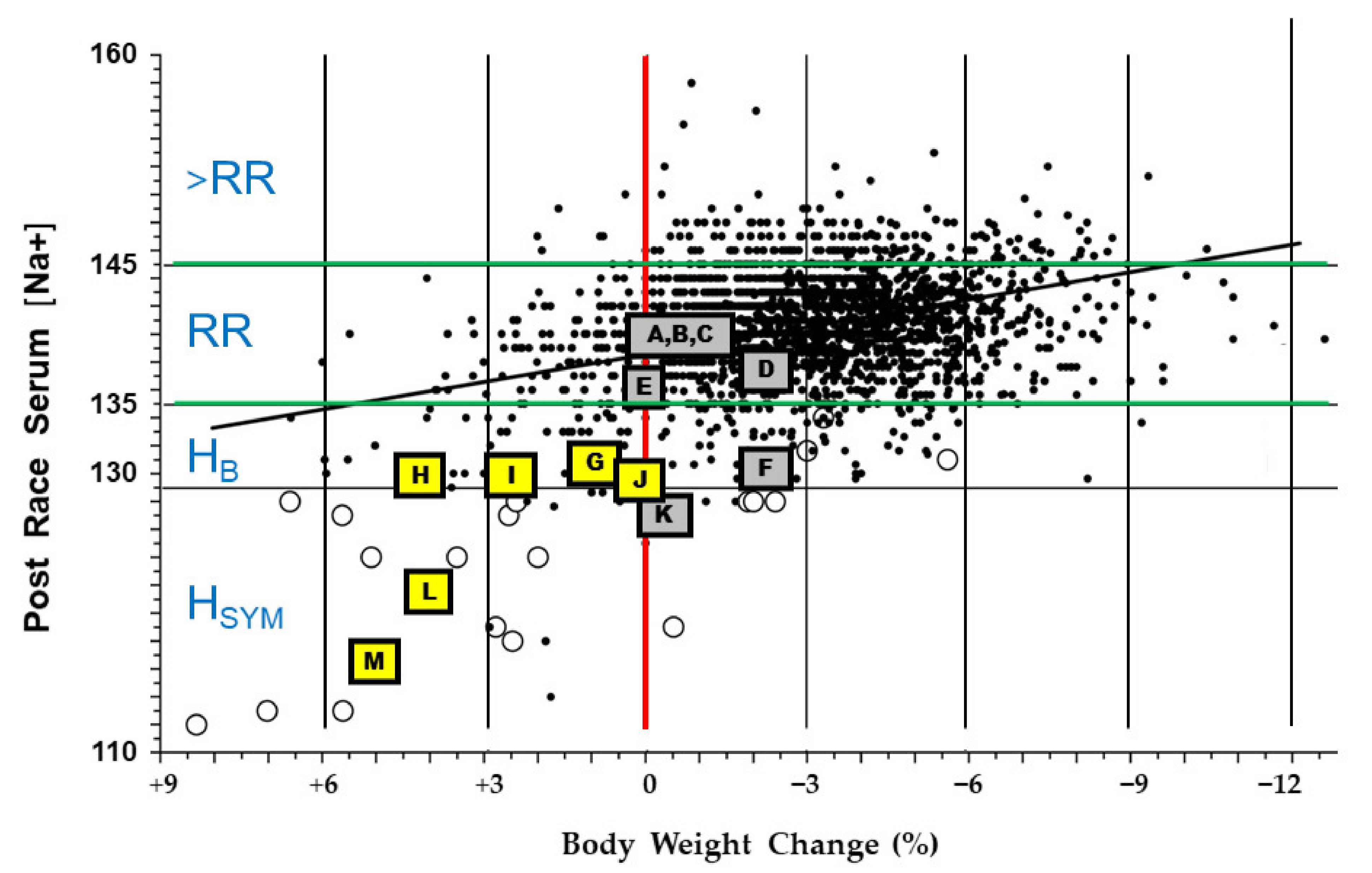
3.2. Exertional Hyponatremia (EHN): A Potential Medical Emergency
3.2.1. Predisposing Factors for EHN
3.2.2. EHN Etiologies
3.2.3. EHN Cases Involve Variable Vasopressin Responses
3.2.4. Evidence for an EHN Drinking Rate Threshold
3.2.5. Does Sodium Intake Counteract a Low Serum Na+?
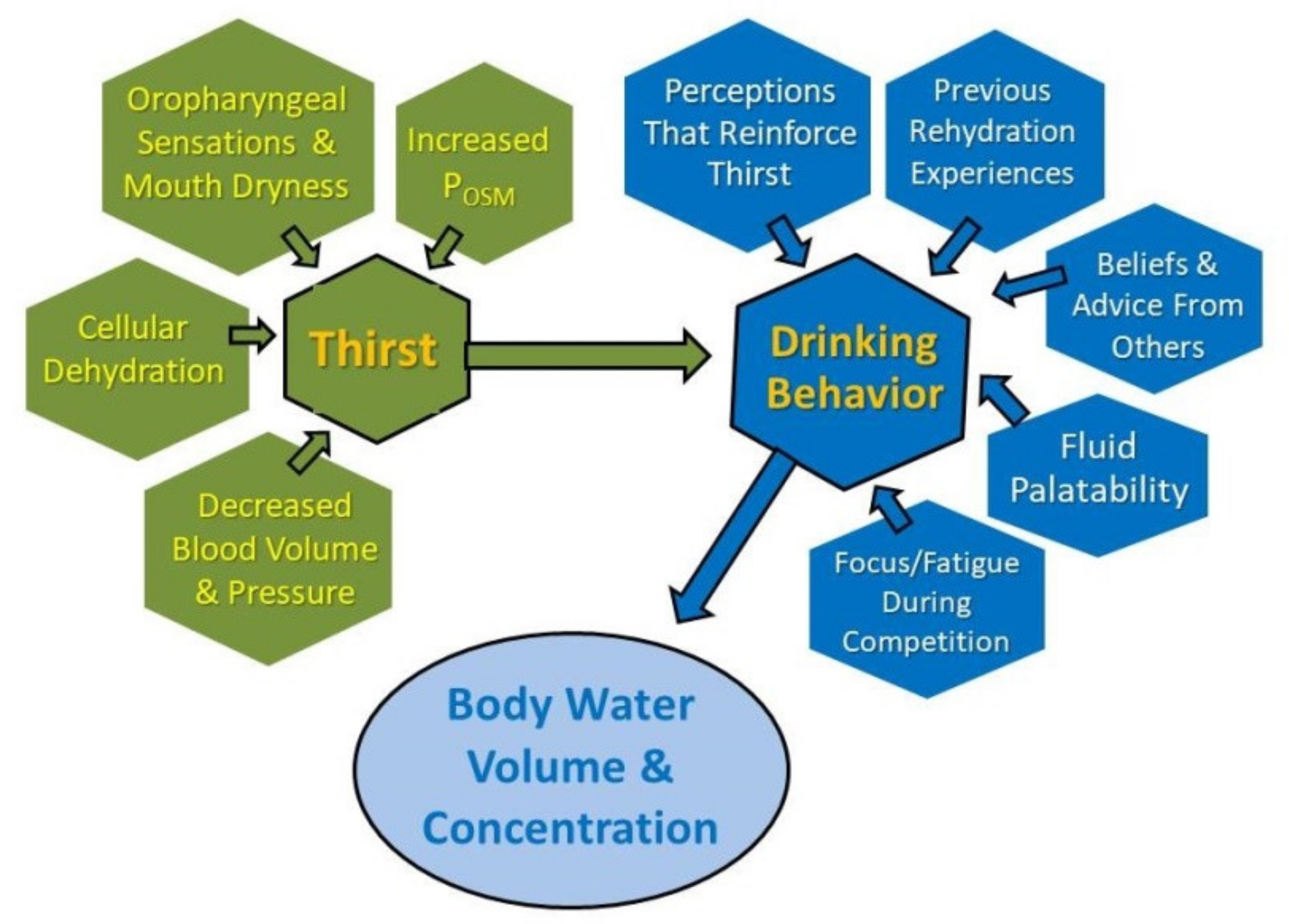
4. The Complexity of Thirst and Drinking
4.1. Multiple Factors Influence Drinking during Endurance Exercise
4.2. Inter-Individual Differences
4.3. Personal Beliefs and Sources of Rehydration Information
4.4. Unique Characteristics of Competitive Events
5. Rehydration Options
5.1. Options 1 and 2
5.2. Option 3
5.2.1. Determining Sweat Rate
5.2.2. Determining a Morning Baseline Body Mass
5.2.3. Interpreting Body Mass Changes
5.3. Options 4 and 5
6. Rehydration Recommendations for Endurance Athletes
- Measure body weight before and after exercise (Section 5.2.1). Change of body mass during exercise is a reasonable, albeit not perfect, surrogate measure of water gain or loss [15,77,119,122]. If body weight cannot be assessed on the day of endurance exercise, measure body weight on the morning after and compare this weight to a pre-determined baseline morning body weight [119]. Detailed methods are described above in Section 5.2.2 and Section 5.2.3.
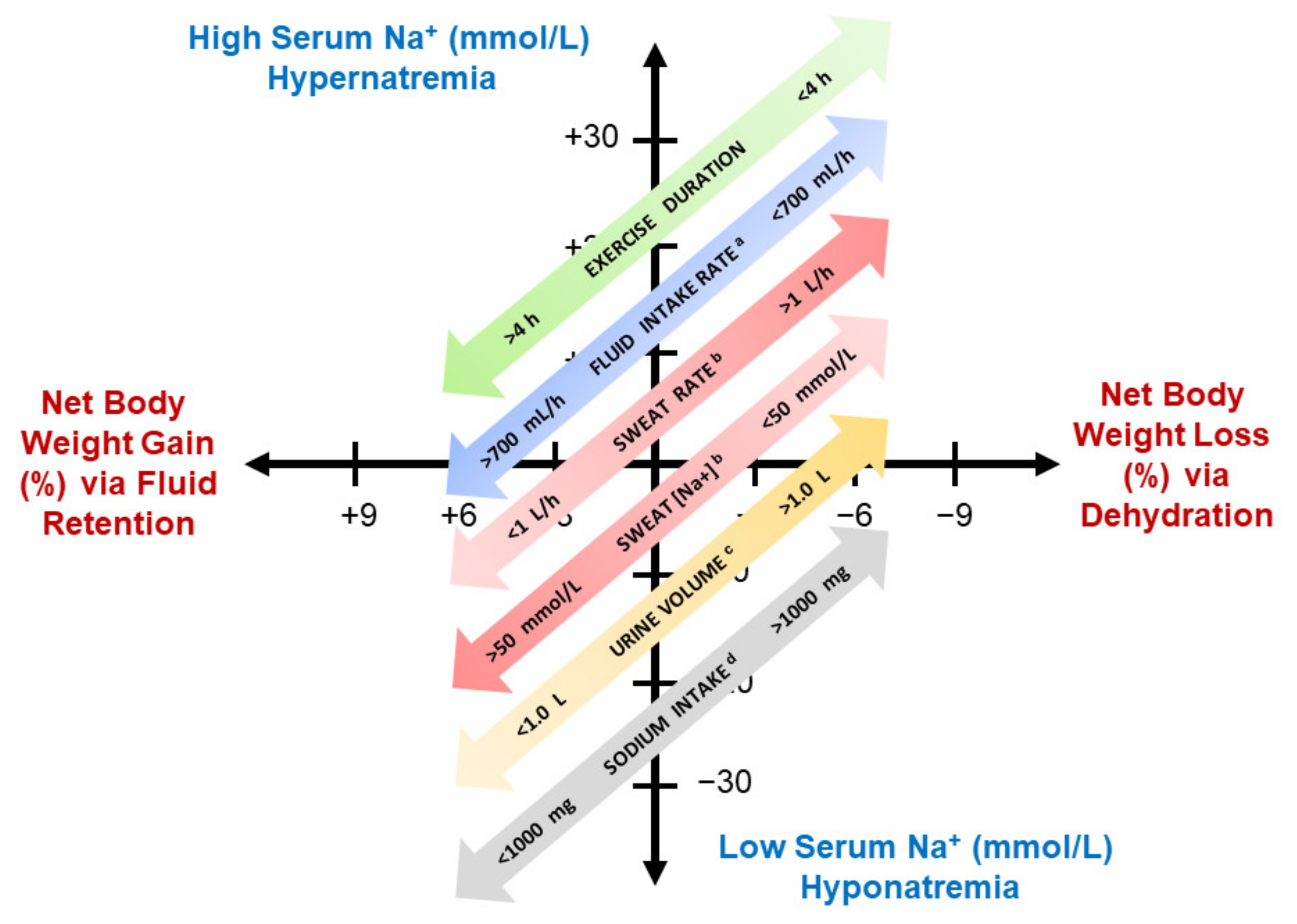
- Consume fluid at a rate less than 700 mL/h to reduce the risk of EHN. The proposed rationale for this recommendation is described in Section 3.2.4, Table 4 (column 8, symbols A–E), and Figure 2 (gray and yellow highlighted symbols). This recommendation is consistent with the 2001 guidelines of the International Marathon Medical Directors Association [84], and the 2007 fluid replacement position stand of the American College of Sports Medicine [24]. Both organizations recommend a 400 to 800 mL/h rate of fluid intake during endurance exercise.
- Be alert for physiologic and perceptual cues that discourage drinking. When stomach fullness, bloating, or vomiting are experienced, decrease fluid intake [84].
- After endurance exercise, white salt deposits on a shirt, jersey, or shorts indicate both a high sweat rate and a high sweat sodium concentration [124]. Consecutive days of profuse sweating (e.g., during lengthy training sessions) or a day-long utraendurance event in a hot environment may lead to whole-body salt deficiency [125,126] due to large sweat sodium losses, inadequate sodium intake, or both [46]. If salt depletion is suspected (e.g., increased salt appetite or salt craving), it is prudent to consider adding specific dietary food items to ensure that daily sodium intake replaces exercise-induced sodium loss. Refer to [97] to identify the amount of sodium in common food items. Sodium supplementation during meals should be guided by dietary recommendations for daily sodium intake [127], and by considering the potential negative health effects of chronic high dietary salt intake [128].
- Sodium consumption in solid food or capsules has a minor influence on serum Na+ and whole-body sodium balance during endurance exercise (Section 3.2.5) [88,89]. Athletes should be aware that sodium intake, while not discouraged, may provide little or no defense against EHN during prolonged exercise and the effects are unpredictable (see Table 3). This recommendation is supported by observations of ultramarathon runners [80]. Multiple regression analysis indicated that the amount of sodium consumed during a 161 km race accounted for only 6–8% of the variance in post-race serum Na+. This recommendation also is supported by the Wilderness Medical Society Clinical Practice Guidelines [123], which advise that sodium and/or salty snacks be consumed along with an appropriate fluid volume. Salt intake should not be combined with overdrinking, which increases the risk of EHN despite sodium consumption; see recommendations 2–4 above.
- Experiment with rehydration options (Table 5) during training sessions, before using them in competition or in hot environments.
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Thornton, S.N. Thirst and hydration: Physiology and consequences of dysfunction. Physiol. Behav. 2010, 100, 15–21. [Google Scholar] [CrossRef] [PubMed]
- Fitzsimons, J.T. The physiological basis of thirst. Kidney Int. 1976, 10, 3–11. [Google Scholar] [CrossRef][Green Version]
- Galanth, C.; Hus-Citharel, A.; Li, B.; Llorens-Cortes, C. Apelin in the control of body fluid homeostasis and cardiovascular functions. Curr. Pharm. Des. 2012, 18, 789–798. [Google Scholar] [CrossRef] [PubMed]
- Azizi, M.; Iturrioz, X.; Blanchard, A.; Peyrard, S.; De Mota, N.; Chartrel, N.; Vaudry, H.; Corvol, P.; Llorens-Cortes, C. Reciprocal regulation of plasma apelin and vasopressin by osmotic stimuli. J. Am. Soc. Nephrol. 2008, 19, 1015–1024. [Google Scholar] [CrossRef] [PubMed]
- Cheuvront, S.N.; Kenefick, R.W. Am I drinking enough? Yes, no, and maybe. J. Am. Coll. Nutr. 2016, 35, 185–192. [Google Scholar] [CrossRef] [PubMed]
- Greenleaf, J.E. Problem: Thirst, drinking behavior, and involuntary dehydration. Med. Sci. Sports Exerc. 1992, 24, 645–656. [Google Scholar] [CrossRef]
- Bartoli, W.P.; Davis, J.M.; Pate, R.R.; Ward, D.S.; Watson, P.D. Weekly variability in total body water using 2H2O dilution in college-age males. Med. Sci. Sports Exerc. 1993, 25, 1422–1428. [Google Scholar] [CrossRef]
- Armstrong, L.E. Assessing hydration status: The elusive gold standard. J. Am. Coll. Nutr. 2007, 26, 575S–584S. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Johnson, E.C. Water Intake, Water Balance, and the Elusive Daily Water Requirement. Nutrients 2018, 10, 1928. [Google Scholar] [CrossRef]
- Speedy, D.B.; Noakes, T.D.; Rogers, I.R.; Thompson, J.M.; Campbell, R.G.; Kuttner, J.A.; Boswell, D.R.; Wright, S.; Hamlin, M.A. Hyponatremia in ultradistance triathletes. Med. Sci. Sports Exerc. 1999, 31, 809–815. [Google Scholar] [CrossRef]
- Hew-Butler, T.; Collins, M.; Bosch, A.; Sharwood, K.; Wilson, G.; Armstrong, M.; Jennings, C.; Swart, J.; Noakes, T. Maintenance of plasma volume and serum sodium concentration despite body weight loss in ironman triathletes. Clin. J. Sport Med. 2007, 17, 116–122. [Google Scholar] [CrossRef]
- Adolph, E.F. Signs and symptoms of desert dehydration. In Physiology of Man in the Desert; Adolf, E.F., Ed.; Interscience Publishers: Cummings Park, MA, USA, 1947. [Google Scholar]
- Rehrer, N.J. Fluid and electrolyte balance in ultra-endurance sport. Sports Med. 2001, 31, 701–715. [Google Scholar] [CrossRef]
- Barr, S.I.; Costill, D.L. Water: Can the endurance athlete get too much of a good thing? J. Am. Diet. Assoc. 1989, 89, 1629. [Google Scholar]
- Armstrong, L.E.; Johnson, E.C.; Ganio, M.S.; Judelson, D.A.; Vingren, J.L.; Kupchak, B.R.; Kunces, L.J.; Muñoz, C.X.; McKenzie, A.L.; Williamson, K.H. Effective body water and body mass changes during summer ultra-endurance road cycling. J. Sports Sci. 2015, 33, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Knechtle, B.; Knechtle, P.; Wirth, A.; Rüst, C.A.; Rosemann, T. A faster running speed is associated with a greater body weight loss in 100-km ultramarathoners. J. Sports Sci. 2012, 30, 1131–1140. [Google Scholar] [CrossRef]
- Rehrer, N.J.; Burke, L.M. Sweat losses during various sports. Nutr. Diet. 1996, 53 (Suppl. S4), S13–S16. [Google Scholar]
- Barnes, K.A.; Anderson, M.L.; Stofan, J.R.; Dalrymple, K.J.; Reimel, A.J.; Roberts, T.J.; Randell, R.K.; Ungaro, C.T.; Baker, L.B. Normative data for sweating rate, sweat sodium concentration, and sweat sodium loss in athletes: An update and analysis by sport. J. Sports Sci. 2019, 37, 2356–2366. [Google Scholar] [CrossRef] [PubMed]
- Borg, G. Perceived exertion as an indicator of somatic stress. Scand. J. Rehabil. Med. 1970, 2, 92–98. [Google Scholar]
- Noakes, T.D.; Sharwood, K.; Speedy, D.; Hew, T.; Reid, S.; Dugas, J.; Almond, C.; Wharam, P.; Weschler, L. Three independent biological mechanisms cause exercise-associated hyponatremia: Evidence from 2135 weighed competitive athletic performances. Proc. Natl. Acad. Sci. USA 2005, 102, 18550–18555. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Hubbard, R.W.; Jones, B.; Daniels, J.T. Preparing Alberto Salazar for the Heat of the 1984 Olympic Marathon. Phys. Sportsmed. 1986, 14, 73–81. [Google Scholar] [CrossRef]
- Roberts, W.O.; Dorman, J.C.; Bergeron, M.F. Recurrent Heat Stroke in a Runner: Race Simulation Testing for Return to Activity. Med. Sci. Sports Exerc. 2016, 48, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, M.F. (SIVOTEC Analytics, Boca Raton, FL, USA). Personal communication, 2021.
- Sawka, M.N.; Burke, L.M.; Eichner, E.R.; Maughan, R.J.; Montain, S.J.; Stachenfeld, N.S. American College of Sports Medicine position stand. Exercise and fluid replacement. Med. Sci. Sports Exerc. 2007, 39, 377–390. [Google Scholar] [PubMed]
- Armstrong, L.E.; Casa, D.J.; Millard-Stafford, M.; Moran, D.S.; Pyne, S.W.; Roberts, W.O. Exertional heat illness during training and competition. Med. Sci. Sports Exerc. 2007, 39, 556–572. [Google Scholar] [CrossRef] [PubMed]
- McDermott, B.P.; Anderson, S.A.; Armstrong, L.E.; Casa, D.J.; Cheuvront, S.N.; Cooper, L.; Kenney, W.L.; O’Connor, F.G.; Roberts, W.O. National athletic trainers’ association position statement: Fluid replacement for the physically active. J. Athl. Train. 2017, 52, 877–895. [Google Scholar] [CrossRef]
- Casa, D.J.; DeMartini, J.K.; Bergeron, M.F.; Csillan, D.; Eichner, E.R.; Lopez, R.M.; Ferrara, M.S.; Miller, K.C.; O’Connor, F.; Sawka, M.N.; et al. National Athletic Trainers’ Association position statement: Exertional heat illnesses. J. Athl. Train. 2015, 50, 986–1000. [Google Scholar] [CrossRef]
- Collins, J.; Maughan, R.J.; Gleeson, M.; Bilsborough, J.; Jeukendrup, A.; Morton, J.P.; Phillips, S.M.; Armstrong, L.; Burke, L.M.; Close, G.L.; et al. UEFA expert group statement on nutrition in elite football. Current evidence to inform practical recommendations and guide future research. Br. J. Sports Med. 2020, 01961. [Google Scholar] [CrossRef]
- Cheuvront, S.N.; Kenefick, R.W. Dehydration: Physiology, assessment, and performance effects. Compr. Physiol. 2014, 4, 257–285. [Google Scholar]
- Judelson, D.A.; Maresh, C.M.; Anderson, J.M.; Armstrong, L.E.; Casa, D.J.; Kraemer, W.J.; Volek, J.S. Hydration and muscular performance: Does fluid balance affect strength, power and high-intensity endurance? Sports Med. 2007, 37, 907–921. [Google Scholar] [CrossRef]
- Sawka, M.N. Physiological consequences of hypohydration: Exercise performance and thermoregulation. Med. Sci. Sports Exerc. 1992, 24, 657–670. [Google Scholar] [CrossRef] [PubMed]
- Kenefick, R.W. Drinking strategies: Planned drinking versus drinking to thirst. Sports Med. 2018, 48, S31–S37. [Google Scholar] [CrossRef]
- Armstrong, L.; Costill, D.; Fink, W. Influence of diuretic-induced dehydration on competitive running performance. Med. Sci. Sports Exerc. 1985, 17, 456–461. [Google Scholar] [CrossRef]
- Bardis, C.N.; Kavouras, S.A.; Arnaoutis, G.; Panagiotakos, D.B.; Sidossis, L.S. Mild dehydration and cycling performance during 5-kilometer hill climbing. J. Athl. Train. 2013, 48, 741–747. [Google Scholar] [CrossRef]
- Bardis, C.N.; Kavouras, S.A.; Kosti, L.; Markousi, M.; Sidossis, L.S. Mild hypohydration decreases cycling performance in the heat. Med. Sci. Sports Exerc. 2013, 45, 1782–1789. [Google Scholar] [CrossRef]
- Walsh, R.M.; Noakes, T.D.; Hawley, J.A.; Dennis, S.C. Impaired High-Intensity Cycling Performance Time at Low Levels of Dehydration. Int. J. Sports Med. 1994, 15, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Goulet, E.D. Effect of exercise-induced dehydration on time-trial exercise performance: A meta-analysis. Br. J. Sports Med. 2011, 45, 1149–1156. [Google Scholar] [CrossRef] [PubMed]
- Sawka, M.N.; Pandolf, K.B. Effects of body water loss on physiological function and exercise performance. In Fluid Homeostasis during Exercise; Gisolfi, C.V., Lamb, D.R., Eds.; Benchmark Press: Carmel, IN, USA, 1990; pp. 1–38. ISBN 0-936157-52-6. [Google Scholar]
- Sawka, M.N.; Young, A.J.; Francesconi, R.P.; Muza, S.R.; Pandolf, K.B. Thermoregulatory and blood responses during exercise at graded hypohydration levels. J. Appl. Physiol. 1985, 59, 1394–1401. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Maresh, C.M.; Gabaree, C.V.; Hoffman, J.R.; Kavouras, S.A.; Kenefick, R.W.; Castellani, J.W.; Ahlquist, L.E. Thermal and circulatory responses during exercise: Effects of hypohydration, dehydration, and water intake. J. Appl. Physiol. 1997, 82, 2028–2035. [Google Scholar] [CrossRef]
- Cheuvront, S.N.; Carter, R.I.; Castellani, J.W.; Sawka, M.N. Hypohydration impairs endurance exercise performance in temperate but not cold air. J. Appl. Physiol. 2005, 99, 1972–1976. [Google Scholar] [CrossRef]
- Sawka, M.N.; Leon, L.R.; Montain, S.J.; Sonna, L.A. Integrated physiological mechanisms of exercise performance, adaptation, and maladaptation to heat stress. Compr. Physiol. 2011, 1, 1883–1928. [Google Scholar] [PubMed]
- Armstrong, L.E. Heat Exhaustion. In Exertional Heat Illness: A Clinical and Evidence-Based Guide; Adams, W.M., Jardine, J., Eds.; Springer Nature: New York, NY, USA, 2020; pp. 81–115. ISBN 978-3-030-27804-5. [Google Scholar]
- Kenefick, R.W.; Cheuvront, S.N.; Palombo, L.J.; Ely, B.R.; Sawka, M.N. Skin temperature modifies the impact of hypohydration on aerobic performance. J. Appl. Physiol. 2010, 109, 79–86. [Google Scholar] [CrossRef]
- Roberts, W.O. Exercise-associated collapse care matrix in the marathon. Sports Med. 2007, 37, 431–433. [Google Scholar] [CrossRef]
- Gaffin, S.L.; Hubbard, R.W. Pathophysiology of heatstroke. In Medical Aspects of Harsh Environments; Pandolf, K.B., Burr, R.E., Eds.; Office of the Surgeon General, U.S. Army: Washington, DC, USA, 2001; Volume 1, pp. 161–209. ISBN 978-0-160-51071-7. [Google Scholar]
- Lambert, G.P.; Lang, J.; Bull, A.; Pfeifer, P.C.; Eckerson, J.; Moore, G.; Lanspa, S.; O’Brien, J. Fluid restriction during running increases GI permeability. Int. J. Sports Med. 2008, 29, 194–198. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E.; Lee, E.C.; Armstrong, E.M. Interactions of gut microbiota, endotoxemia, immune function, and diet in exertional heatstroke. J. Sports Med. 2018, 5724575. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, M.F. Heat cramps: Fluid and electrolyte challenges during tennis in the heat. J. Sci. Med. Sport 2003, 6, 19–27. [Google Scholar] [CrossRef]
- King, B.A.; Barry, M.E. The physiological adaptations to heat-stress with a classification of heat illness and a description of the features of heat exhaustion. S. Afr. Med. J. 1962, 36, 451–455. [Google Scholar]
- Armstrong, L.E.; Hubbard, R.W.; Szlyk, P.C.; Sills, I.V.; Kraemer, W.J. Heat intolerance, heat exhaustion monitored: A case report. Aviat. Space Environ. Med. 1988, 59, 262–266. [Google Scholar]
- Sawka, M.N.; Young, A.J.; Latke, W.A.; Neufer, P.D.; Quigley, M.D.; Pandolf, K.B. Human tolerance to heat strain during exercise: Influence of hydration. J. Appl. Physiol. 1992, 73, 368–375. [Google Scholar] [CrossRef]
- Donoghue, A.M.; Sinclair, M.J.; Bates, G.P. Heat exhaustion in a deep underground metalliferous mine. Occup. Environ. Med. 2000, 57, 165–174. [Google Scholar] [CrossRef]
- Irving, R.A.; Noakes, T.D.; Irving, G.A.; Van Zyl-Smit, R. The immediate and delayed effects of marathon running on renal function. J. Urol. 1986, 136, 1176–1180. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Casa, D.J.; Emmanuel, H.; Ganio, M.S.; Klau, J.F.; Lee, E.C.; Maresh, C.M.; McDermott, B.P.; Stearns, R.L.; Vingren, J.L.; et al. Nutritional, Physiological and Perceptual Responses During a Summer Ultra-Endurance Cycling Event. J. Strength Cond. Res. 2012, 26, 307–318. [Google Scholar] [CrossRef]
- Hillman, A.R.; Turner, M.C.; Peart, D.J.; Bray, J.W.; Taylor, L.; McNaughton, L.R.; Siegler, J.C. A comparison of hyperhydration versus ad libitum fluid intake strategies on measures of oxidative stress, thermoregulation, and performance. Res. Sports Med. 2013, 21, 305–317. [Google Scholar] [CrossRef]
- Kavouras, S.A.; Armstrong, L.E.; Maresh, C.M.; Casa, D.J.; Herrera-Soto, J.A.; Scheett, T.P.; Stoppani, J.; Mack, G.W.; Kraemer, W.J. Rehydration with glycerol: Endocrine, cardiovascular, and thermoregulatory responses during exercise in the heat. J. Appl. Physiol. 2006, 100, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Van Rosendal, S.P.; Osborne, M.A.; Fassett, R.G.; Coombes, J.S. Guidelines for glycerol use in hyperhydration and rehydration associated with exercise. Sports Med. 2010, 40, 113–139. [Google Scholar] [CrossRef] [PubMed]
- Kerksick, C.M.; Wilborn, C.D.; Roberts, M.D.; Smith-Ryan, A.; Kleiner, S.M.; Jäger, R.; Collins, R.; Cooke, M.; Davis, J.N.; Galvan, E.; et al. ISSN Exercise & Sports Nutrition Review Update: Research & Recommendations. J. Int. Soc. Sports Nutr. 2018, 15, 1–57. [Google Scholar]
- Montain, S.J. Strategies to prevent hyponatremia during prolonged exercise. Curr. Sports Med. Rep. 2008, 7, S28–S35. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Curtis, W.C.; Hubbard, R.W.; Francesconi, R.P.; Moore, R.O.; Askew, E.W. Symptomatic hyponatremia during prolonged exercise in heat. Med. Sci. Sports Exerc. 1993, 25, 543–549. [Google Scholar] [CrossRef]
- Armstrong, L.E.; McDermott, B.P. Exertional hyponatremia. In Emergency Management for Sport and Physical Activity; Casa, D.J., Stearns, R., Eds.; Jones & Bartlett Learning: Burlington, MA, USA, 2015; pp. 169–180. ISBN 978-1-284-02216-2. [Google Scholar]
- Hew-Butler, T.; Rosner, M.H.; Fowkes-Godek, S.; Dugas, J.P.; Hoffman, M.D.; Lewis, D.P.; Maughan, R.J.; Miller, K.C.; Montain, S.J.; Rehrer, N.J.; et al. Statement of the Third International Exercise-Associated Hyponatremia Consensus Development Conference, Carlsbad, California, 2015. Clin. J. Sport Med. 2015, 25, 303–320. [Google Scholar] [CrossRef] [PubMed]
- Weschler, L.B. Exercise-associated hyponatraemia. Sports Med. 2005, 35, 899–922. [Google Scholar] [CrossRef] [PubMed]
- Kobrick, J.L.; Sampson, J.B. New inventory for the assessment of symptom occurrence and severity at high altitude. Aviat. Space Environ. Med. 1979, 50, 925–929. [Google Scholar]
- Armstrong, L.E.; Lee, E.C.; Casa, D.; Johnson, E.C.; Ganio, M.S.; McDermott, B.; Vingren, J.; Oh, H.M.; Williamson, K.H. Exertional Hyponatremia and Serum Sodium Change During Ultraendurance Cycling. Int. J. Sport Nutr. Exerc. Metab. 2017, 27, 139–147. [Google Scholar] [CrossRef]
- Armstrong, L.E. Exertional hyponatremia. In Exertional Heat Illnesses; Armstrong, L.E., Ed.; Human Kinetics: Champaign, IL, USA, 2003; pp. 103–136. ISBN 0-7360-3771-3. [Google Scholar]
- Hiller, W.D. Dehydration and hyponatremia during triathlons. Med. Sci. Sports Exerc. 1989, 21 (Suppl. S5), S219–S221. [Google Scholar]
- Armstrong, L.E.; Casa, D.J. Predisposing Factors for Exertional Heat Illnesses. In Exertional Heat Illnesses; Armstrong, L.E., Ed.; Human Kinetics: Champaign, IL, USA, 2003; pp. 162–167. ISBN 0-7360-3771-3. [Google Scholar]
- Seal, A.D.; Anastasiou, C.A.; Skenderi, K.P.; Echegaray, M.; Yiannakouris, N.; Tsekouras, Y.E.; Matalas, A.L.; Yannakoulia, M.; Pechlivani, F.; Kavouras, S.A. Incidence of hyponatremia during a continuous 246-km ultramarathon running race. Front. Nutr. 2019, 6, 161. [Google Scholar] [CrossRef]
- Rüst, C.A.; Knechtle, B.; Knechtle, P.; Wirth, A.; Rosemann, T. Body mass change and ultraendurance performance: A decrease in body mass is associated with an increased running speed in male 100-km ultramarathoners. J. Strength Cond. Res. 2012, 26, 1505–1516. [Google Scholar] [CrossRef] [PubMed]
- Arnaoutis, G.; Anastasiou, C.A.; Suh, H.; Maraki, M.; Tsekouras, Y.; Dimitroulis, E.; Echegaray, M.; Papamichalopoulou, D.; Methenitis, S.; Sidossis, L.S.; et al. Exercise-Associated Hyponatremia during the Olympus Marathon Ultra-Endurance Trail Run. Nutrients 2020, 12, 997. [Google Scholar] [CrossRef] [PubMed]
- Speedy, D.B.; Noakes, T.D.; Rogers, I.R.; Hellemans, I.; Kimber, N.E.; Boswell, D.R.; Campbell, R.; Kuttner, J.A. A prospective study of exercise-associated hyponatremia in two ultradistance triathletes. Clin. J. Sport Med. 2000, 10, 136–141. [Google Scholar] [CrossRef]
- Speedy, D.B.; Rogers, I.R.; Noakes, T.D.; Wright, S.; Thompson, J.M.; Campbell, R.; Hellemans, I.; Kimber, N.E.; Boswell, D.R.; Kuttner, J.A.; et al. Exercise-induced hyponatremia in ultradistance triathletes is caused by inappropriate fluid retention. Clin. J. Sport Med. 2000, 10, 272–278. [Google Scholar]
- Speedy, D.B.; Rogers, I.; Safih, S.; Foley, B. Hyponatremia and seizures in an ultradistance triathlete. J. Emerg. Med. 2000, 18, 41–44. [Google Scholar] [CrossRef]
- Montain, S.J.; Cheuvront, S.N.; Sawka, M.N. Exercise associated hyponatremia: Quantitative analysis for understand the aetiology. Br. J. Sports Med. 2006, 40, 98–106. [Google Scholar] [CrossRef]
- Hew-Butler, T.; Loi, V.; Pani, A.; Rosner, M.H. Exercise-associated hyponatremia: 2017 update. Front. Med. 2017, 4. [Google Scholar] [CrossRef]
- Noakes, T.D.; Goodwin, N.; Rayner, B.L.; Branken, T.R.; Taylor, R.K. Water intoxication: A possible complication during endurance exercise. Med. Sci. Sports Exerc. 1985, 17, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Frizzell, R.T.; Lang, G.H.; Lowance, D.C.; Lathan, S.R. Hyponatremia and ultramarathon running. JAMA 1986, 255, 772–774. [Google Scholar] [CrossRef]
- Hoffman, M.D.; Stuempfle, K.J. Sodium Supplementation and Exercise-Associated Hyponatremia during Prolonged Exercise. Med. Sci. Sports Exerc. 2015, 47, 1781–1787. [Google Scholar] [CrossRef] [PubMed]
- Bankir, L.; Bichet, D.G.; Morgenthaler, N.G. Vasopressin: Physiology, assessment and osmosensation. J. Intern. Med. 2017, 282, 284–297. [Google Scholar] [CrossRef] [PubMed]
- Hew-Butler, T. Arginine vasopressin, fluid balance and exercise. Sports Med. 2010, 40, 459–479. [Google Scholar] [CrossRef]
- Siegel, A.J.; Verbalis, J.G.; Clement, S.; Mendelson, J.H.; Mello, N.K.; Adner, M.; Shirey, T.; Glowacki, J.; Lee-Lewandrowski, E.; Lewandrowski, K.B. Hyponatremia in marathon runners due to inappropriate arginine vasopressin secretion. Am. J. Med. 2007, 120, 461.e11–461.e17. [Google Scholar] [CrossRef]
- Hew-Butler, T.; Verbalis, J.G.; Noakes, T.D. Updated fluid recommendation: Position statement from the International Marathon Medical Directors Association (IMMDA). Clin. J. Sport Med. 2006, 16, 283–292. [Google Scholar] [CrossRef] [PubMed]
- Speedy, D.B.; Thompson, J.M.; Rodgers, I.; Collins, M.; Sharwood, K. Oral salt supplementation during ultradistance exercise. Clin. J. Sport Med. 2002, 12, 279–284. [Google Scholar] [CrossRef]
- Hew-Butler, T.D.; Sharwood, K.; Collins, M.; Speedy, D.; Noakes, T. Sodium supplementation is not required to maintain serum sodium concentrations during an Ironman triathlon. Br. J. Sports Med. 2006, 40, 255–259. [Google Scholar] [CrossRef]
- Hoffman, M.D.; Myers, T.M. Symptomatic Exercise-Associated Hyponatremia in an Endurance Runner Despite Sodium Supplementation. Int. J. Sport Nutr. Exerc. Metab. 2015, 25, 603–606. [Google Scholar] [CrossRef]
- Barr, S.I.; Costill, D.L.; Fink, W.J. Fluid replacement during prolonged exercise: Effects of water, saline, or no fluid. Med. Sci. Sports Exerc. 1991, 23, 811–817. [Google Scholar] [CrossRef]
- Vrijens, D.M.J.; Rehrer, N.J. Sodium-free fluid ingestion decreases plasma sodium during exercise in the heat. J. Appl. Physiol. 1999, 86, 1847–1851. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Kavouras, S.A. Thirst and drinking paradigms: Evolution from single factor effects to brainwide dynamic networks. Nutrients 2019, 12, 2864. [Google Scholar] [CrossRef]
- Greenleaf, J.E.; Sargent, F. Voluntary dehydration in man. J. Appl. Physiol. 1965, 20, 719–724. [Google Scholar] [CrossRef]
- Allen, W.E.; Chen, M.Z.; Pichamoorthy, N.; Tien, R.H.; Pachitariu, M.; Luo, L.; Deisseroth, K. Thirst regulates motivated behavior through modulation of brainwide neural population dynamics. Science 2019, 364, 6437. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Giersch, G.E.W.; Dunn, L.; Fiol, A.; Muñoz, C.X.; Lee, E.C. Inputs to Thirst and Drinking during Water Restriction and Rehydration. Nutrients 2020, 12, 2554. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Giersch, G.E.; Colburn, A.T.; Lopez, V.; Sekiguchi, Y.; Muñoz, C.X.; Lee, E.C. Progression of human subjective perceptions during euhydration, mild dehydration, and drinking. Physiol. Behav. 2021, 229, 113211. [Google Scholar] [CrossRef] [PubMed]
- Fitzsimons, J.T. Angiotensin, thirst, and sodium appetite. Physiol. Rev. 1998, 78, 583–686. [Google Scholar] [CrossRef] [PubMed]
- Kratz, A.; Ferraro, M.; Sluss, P.M.; Lewandrowski, K.B. Laboratory Reference Values. N. Engl. J. Med. 2004, 35, 1548–1563. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Muňoz, C.X.; Armstrong, E.M. Distinguishing Low and High Water Consumers: A Paradigm of Disease Risk. Nutrients 2020, 12, 858. [Google Scholar] [CrossRef]
- Beis, L.Y.; Wright-Whyte, M.; Fudge, B.; Noakes, T.; Pitsiladis, Y.P. Drinking behaviors of elite male runners during marathon competition. Clin. J. Sport Med. 2012, 22, 254–261. [Google Scholar] [CrossRef] [PubMed]
- Winger, J.M.; Dugas, J.P.; Dugas, L.R. Beliefs about hydration and physiology drive drinking behaviours in runners. Br. J. Sports Med. 2011, 45, 646–649. [Google Scholar] [CrossRef] [PubMed]
- Wilson, P.B. Nutrition behaviors, perceptions, and beliefs of recent marathon finishers. Phys. Sportsmed. 2016, 44, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Cheuvront, S.N.; Haymes, E.M. Ad libitum fluid intakes and thermoregulatory responses of female distance runners in three environments. J. Sports Sci. 2001, 19, 845–854. [Google Scholar] [CrossRef]
- Cheuvront, S.N.; Haymes, E.M. Thermoregulation and marathon running: Biological and environmental influences. Sports Med. 2001, 31, 743–762. [Google Scholar] [CrossRef]
- Noakes, T.D. Fluid replacement during exercise. In Exercise and Sport Science Reviews, Holloszy, J., Ed.; Williams & Wilkins: Philadelphia, PA, USA, 1993; Volume 21, pp. 297–330. ISBN 978-0-683-00035-1. [Google Scholar]
- Hoffman, M.D.; Cotter, J.D.; Goulet, É.D.; Laursen, P.B. View: Is drinking to thirst adequate to appropriately maintain hydration status during prolonged endurance exercise? Yes. Wilderness Environ. Med. 2016, 27, 192–195. [Google Scholar] [CrossRef]
- Hoffman, M.D.; Cotter, J.D.; Goulet, É.D.; Laursen, P.B. Rebuttal from “Yes”. Wilderness Environ. Med. 2016, 27, 198–200. [Google Scholar] [CrossRef]
- Hoffman, M.D. Comment on “Drinking Strategies: Planned Drinking Versus Drinking to Thirst”. Sports Med. 2019, 49, 1133–1134. [Google Scholar] [CrossRef]
- Noakes, T.D. Hydration in the Marathon. Using Thirst to Gauge Safe Fluid Replacement. Sports Med. 2007, 37, 463–466. [Google Scholar] [CrossRef] [PubMed]
- Kenefick, R.W. Author’s Reply to Goulet: Comment on: “Drinking Strategies: Planned Drinking Versus Drinking to Thirst”. Sports Med. 2019, 49, 635–636. [Google Scholar] [CrossRef]
- Dion, T.; Savoie, F.A.; Asselin, A.; Gariepy, C.; Goulet, E.D. Half-marathon running performance is not improved by a rate of fluid intake above that dictated by thirst sensation in trained distance runners. Eur. J. Appl. Physiol. 2013, 113, 3011–3020. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Johnson, E.C.; Kunces, L.J.; Ganio, M.S.; Judelson, D.A.; Kupchak, B.R.; Vingren, J.L.; Munoz, C.X.; Huggins, R.A.; Hydren, J.R.; et al. Drinking to thirst versus drinking ad libitum during road cycling. J. Athl. Train. 2014, 49, 624–631. [Google Scholar] [CrossRef] [PubMed]
- Hubbard, R.W.; Szlyk, P.C.; Armstrong, L.E. Influence of thirst and fluid palatability on fluid ingestion during exercise. In Perspectives in Exercise Sciences and Sports Medicine. Fluid Homeostasis during Exercise; Gisolfi, C.V., Lamb, D.R., Eds.; Benchmark Press: Indianapolis, IN, USA, 1990; Volume 1, pp. 39–95. ISBN 978-0-697-14816-2. [Google Scholar]
- Armstrong, L.E.; Johnson, E.C.; Bergeron, M.F. Counterview: Is drinking to thirst adequate to appropriately maintain hydration status during prolonged endurance exercise? No. Wilderness Environ. Med. 2016, 27, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E.; Johnson, E.C.; Bergeron, M.F. Rebuttal from “No”. Wilderness Environ. Med. 2016, 27, 200–202. [Google Scholar] [CrossRef] [PubMed]
- Armstrong, L.E.; Johnson, E.C.; McKenzie, A.L.; Ellis, L.A.; Williamson, K.H. Ultraendurance Cycling in a Hot Environment: Thirst, Fluid Consumption and Water Balance. J. Strength Cond. Res. 2015, 29, 869–876. [Google Scholar] [CrossRef]
- Figaro, M.K.; Mack, G.W. Regulation of fluid intake in dehydrated humans: Role of oropharyngeal stimulation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 1997, 272, R1740–R1746. [Google Scholar] [CrossRef]
- Armstrong, L.E.; Casa, D.J. Methods to evaluate electrolyte and water turnover of athletes. Athl. Train. Sports Health Care 2009, 1, 169–179. [Google Scholar] [CrossRef]
- Casa, D.J. Proper hydration for distance running: Identifying individual fluid needs. Track Coach 2004, 167, 5321–5328. [Google Scholar]
- Armstrong, L.E. Hydration assessment techniques. Nutr. Rev. 2005, 63 Pt 2 (Suppl. S6), S40–S54. [Google Scholar] [CrossRef]
- Ganio, M.S.; Armstrong, L.E.; Kavouras, S.A. Hydration. In Sport and Physical Activity in the Heat; Casa, D.J., Ed.; Springer: Cham, Switzerland, 2018; pp. 83–100. ISBN 978-3-319-70216-2. [Google Scholar]
- Kavouras, S. Assessing hydration status. Curr. Opin. Clin. Nutr. Metab. Care. 2002, 5, 519–524. [Google Scholar] [CrossRef] [PubMed]
- Shirreffs, S.M. Markers of hydration status. Eur. J. Clin. Nutr. 2003, 57 (Suppl. S2), S6–S9. [Google Scholar] [CrossRef]
- Maughan, R.J.; Shirreffs, S.M.; Leiper, J.B. 2007. Errors in the estimation of sweat loss and changes in hydration status from changes in body mass during exercise. J. Sports Sci. 2007, 25, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Bennett, B.L.; Hew-Butler, T.; Rosner, M.H.; Myers, T.; Lipman, G.S. Wilderness Medical Society clinical practice guidelines for the management of exercise-associated hyponatremia: 2019 update. Wilderness Environ. Med. 2020, 31, 50–62. [Google Scholar] [CrossRef] [PubMed]
- Kavouras, S.A. Sodium balance during exercise and hyponatremia. In Fluid Balance, Hydration, and Athletic Performance; Meyer, F., Szygula, Z., Wilk, B., Eds.; CRC Press: Boca Raton, FL, USA, 2015; pp. 23–31. ISBN 978-1-4822-2328-6. [Google Scholar]
- Marriott, H. Water and salt depletion. Br. Med. J. 1942, 2, 285–290. [Google Scholar]
- Taylor, H.L.; Henschel, A.; Mickelsen, O.; Keys, A. The effect of the sodium chloride intake on the work performance of man during exposure to dry heat and experimental heat exhaustion. Am. J. Physiol. 1943, 140, 439–451. [Google Scholar] [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Dietary Reference Intakes for Sodium and Potassium; The National Academies Press: Washington, DC, USA, 2019. [Google Scholar] [CrossRef]
- Diet, Nutrition and the Prevention of Chronic Diseases: Report of a Joint WHO/FAO Expert Consultation. Geneva, 28 January–1 February 2002. Available online: https://www.who.int/publications-detail-redirect/924120916X (accessed on 10 February 2021).
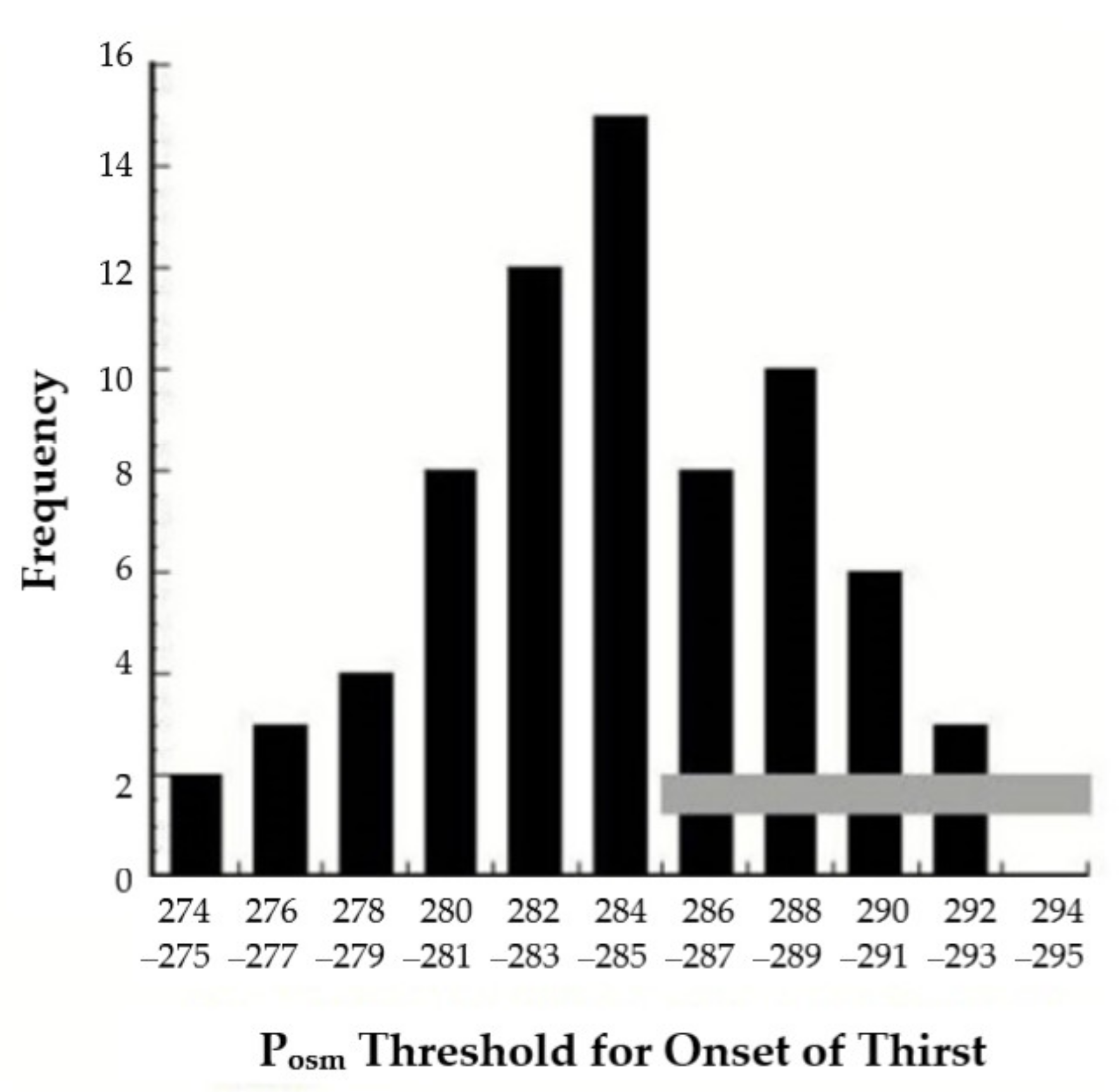
| Activity | Thirst & Drinking Behavior | Sweat Gland Secretion of Hypotonic Fluid | Kidney Regulation of Water & Electrolytes | Neuroendocrine Homeostatic Responses a | Effects on Water & Electrolyte Balance |
|---|---|---|---|---|---|
| Sedentary daily activities (16 h) | Basal b | Negligible | Basal b | Basal b | CNS responses are sufficient to maintain water and electrolyte homeostasis |
| Brief exercise (5–30 min) at moderate-to-high intensity | Minor | Minor-to-moderate | Minor | Minor, brief | Water and electrolyte losses are minor |
| Endurance exercise (0.5–5 h) at low-to-high intensity | Minor-to-large | Moderate-to-large | Minor-to-moderate | Minor-to-large, prolonged | Moderate-to-large turnover c due to sweating and drinking |
| Ultraendurance exercise (5–24 h) at low-to-moderate intensity | Moderate-to-large | Large | Moderate-to-large | Large, prolonged | Water and electrolyte losses in sweat and urine exceed 24 h dietary intake |
| Variables | Average Exercise Duration (h) a | ||
|---|---|---|---|
| 9.6 | 6.3 | 4.8 | |
| Pre-event body mass b (kg) | 81.90 | 82.05 | 82.55 |
| Number of male cyclists | 11 | 11 | 10 |
| Ground speed (km/h) | 17.2 d | 26.6 d | 34.0 d |
| Rating of perceived exertion at finish c | 16 | 16 | 16 |
| ad libitum total fluid intake e (g) | +6100 | +4500 | +3900 f |
| Rate of fluid intake (g/h) | +635 | +715 | +810 |
| Sweat secreted g (g) | −7700 | −7150 | −7000 |
| Sweat rate (g/h) | −800 d | −1135 | −1460 |
| Urine excreted g (g) | −1300 | −550 | −450 d |
| Solid food mass consumed e (g) | +423 | +355 | +350 |
| Body mass change b (g) | −1800 | −2300 | −2750 |
| Body mass change (%) | −2.0 | −2.9 | −3.4 |
| Cyclists | Total Fluid Intake (L) a,b | Total Fluid Intake (ml/kg) a,b | Sodium Intake (mg) a,b | Change of Serum Na+ (mmol/L) a | Pre-Event Body Mass (kg) | Body Mass Change (%) a | Urine Specific Gravity at Finish Line | Rating of Thirst at Finish Line c | Environmental Symptoms Questionnaire d Total Score at Finish Line |
|---|---|---|---|---|---|---|---|---|---|
| A | 3.7 | 42 | 356 | +6 | 88.6 | −4.6 | 1.021 | 4 | 13 |
| B | 5.3 | 75 | 194 | +4 | 71.0 | +1.4 | 1.024 | 8 | 10 |
| C | 3.0 | 48 | 328 | +3 | 61.8 | −4.2 | 1.030 | 6 | 11 |
| D | 4.7 | 62 | 149 | +1 | 75.2 | −1.2 | 1.026 | 8 | 27 |
| E | 10.9 | 139 | 1166 | +1 | 78.5 | −1.5 | 1.020 | 7 | 25 |
| F | 4.6 | 54 | 124 | −1 | 85.5 | +0.1 | 1.021 | 6 | 21 |
| G | 4.1 | 50 | 261 | −2 | 82.0 | −1.8 | 1.030 | 5 | 13 |
| H | 3.4 | 41 | 263 | −2 | 82.9 | −0.1 | 1.023 | 4 | 11 |
| I | 9.5 | 103 | 823 | −2 | 91.8 | −4.6 | 1.034 | 6 | 25 |
| J | 9.6 | 124 | 1259 | −3 | 77.2 | −1.9 | 1.016 | 4 | 17 |
| K | 10.5 | 101 | 1182 | −3 | 104.7 | +1.0 | 1.026 | 5 | 21 |
| L | 9.2 | 109 | 1601 | −6 | 84.7 | +1.1 | 1.003 | 5 | 12 |
| LC e | 13.7 | 191 | 1179 | −11 | 72.0 | +4.3 | 1.003 | 2 | 4 |
| AM e | 14.7 | 189 | 3292 | −11 | 77.5 | +0.1 | 1.010 | 2 | 11 |
| Symbols in Figure 2 | Men | Women | Scenario (Ambient Temperature, °C) | Final Serum Na+ (mmol/L) | Body Mass Change (%) | Exercise Duration (h) | Rate of Fluid Intake (ml/h) | Mean Initial Body Mass (kg) | Source |
|---|---|---|---|---|---|---|---|---|---|
| Background data points | a | a | 11 endurance events a | See Figure 3 | See Figure 3 | b | b | b | [20] |
| A | 42 | 164 km cycling (34.4) | 141 | −0.8 | 9.1 | 649 | 85.9 | [55] | |
| B | 31 | 164 km cycling (24.4–39.5) | 141 | −1.4 | 9.0 | 700 | 85.4 | [66] | |
| C | 6 | 164 km cycling (34.4) | 140 | −0.1 | 9.0 | 520 | 67.3 | [55] | |
| D | 50 | 100 km run (15.6–21.7) | 138 | −2.6 | 12.2 | 600 | 74.9 | [71] | |
| E | 7 | Treadmill walk (41.0) c | 136 | −0.1 | 4.0 c | 640 | 77.9 | [61] | |
| F | 5 | 44 km trail run (15–34) | 131 | −2.2 | 9.3 | 290 d | 81.9 | [72] | |
| G | 1 | Ironman triathlon (21.0) e,f | 131 | +0.9 | 13.3 | 733 | 57.5 | [73] | |
| H | 1 | 164 km cycling (24.4–39.5) g | 130 | +4.3 | 8.9 | 1,500 | 72.0 | [66] | |
| I | 1 | Ironman triathlon (21.0) e,f | 130 | +2.5 | 12.0 | 764 | 59.0 | [73] | |
| J | 1 | 164 km cycling (24.4–39.5) g | 130 | +0.1 | 10.6 | 1,400 | 77.5 | [66] | |
| K | 2 | 5 | Ironman triathlon e | 128 | −0.5 | 12.3 | b | 62.5 | [74] |
| L | 1 | Treadmill walk (41.0) c | 122 | +4.0 | 4.0 c | 2,061 h | 82.2 | [61] | |
| M | 1 | Ironman triathlon e | 116 | +5.0 | 14.0 | 1,642 | b | [75] |
| Description | Objective/Rationale | Relevant Publications |
|---|---|---|
| 1. Drink when thirsty. Fluid intake occurs only when thirst is sensed. | Primary focus: to prevent exertional hyponatremia. Secondary goal: to prevent a level of dehydration that impairs exercise performance. Proponents of this method assert that increased extracellular concentration triggers thirst to naturally protect athletes from the negative consequences of both fluid excess and severe dehydration. However, no randomized, controlled study confirms that drinking when thirsty successfully prevents exertional hyponatremia. Rationale: drinking when thirsty preserves serum Na+ and osmolality within the normal laboratory reference range. | [55,84,104,105,106,107,108,109,110] |
| 2. Ad libitum drinking. Consuming fluid whenever and in whatever volume desired, without specific focus on thirst. | Primary focus: to prevent exertional hyponatremia. Secondary goal: to prevent a level of dehydration that impairs exercise performance. Ad libitum drinking often is viewed as being identical to drinking when thirsty (above), however it is subtly different. See text for details. | [6,55,101,110,111] |
| 3. Individualized planned drinking. This involves drinking a predetermined fluid volume that is determined by measuring sweat rate. | Primary focus: to prevent excessive dehydration that impairs exercise performance and to prevent exertional hyponatremia. Secondary goals: to decrease the risk of heat illness (heat exhaustion, heat stroke), and reduce cardiovascular/thermoregulatory strain associated with dehydration. Rationale: because there is considerable inter-individual variability of sweat rate and sweat electrolyte concentration, a customized fluid replacement plan meets each athlete’s individual rehydration needs. | [24,26,32,66,108,112,113] |
| 4. Purposefully drink nothing during exercise. | No professional sport medicine or sport nutrition organization recommends this extreme option for prolonged endurance exercise. | |
| 5. Purposefully drink as much as possible, in excess of thirst. | No professional sport medicine or sport nutrition organization recommends this extreme option for prolonged endurance exercise. Nevertheless, a 2011 survey of runners (5 to 42.2 km finishers) determined that 8.9% plan to drink as much as possible during racing and training. | [99] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Armstrong, L.E. Rehydration during Endurance Exercise: Challenges, Research, Options, Methods. Nutrients 2021, 13, 887. https://doi.org/10.3390/nu13030887
Armstrong LE. Rehydration during Endurance Exercise: Challenges, Research, Options, Methods. Nutrients. 2021; 13(3):887. https://doi.org/10.3390/nu13030887
Chicago/Turabian StyleArmstrong, Lawrence E. 2021. "Rehydration during Endurance Exercise: Challenges, Research, Options, Methods" Nutrients 13, no. 3: 887. https://doi.org/10.3390/nu13030887
APA StyleArmstrong, L. E. (2021). Rehydration during Endurance Exercise: Challenges, Research, Options, Methods. Nutrients, 13(3), 887. https://doi.org/10.3390/nu13030887






