Classic Ketogenic Diet and Modified Atkins Diet in SLC2A1 Positive and Negative Patients with Suspected GLUT1 Deficiency Syndrome: A Single Center Analysis of 18 Cases
Abstract
1. Introduction
2. Materials and Methods
3. Results
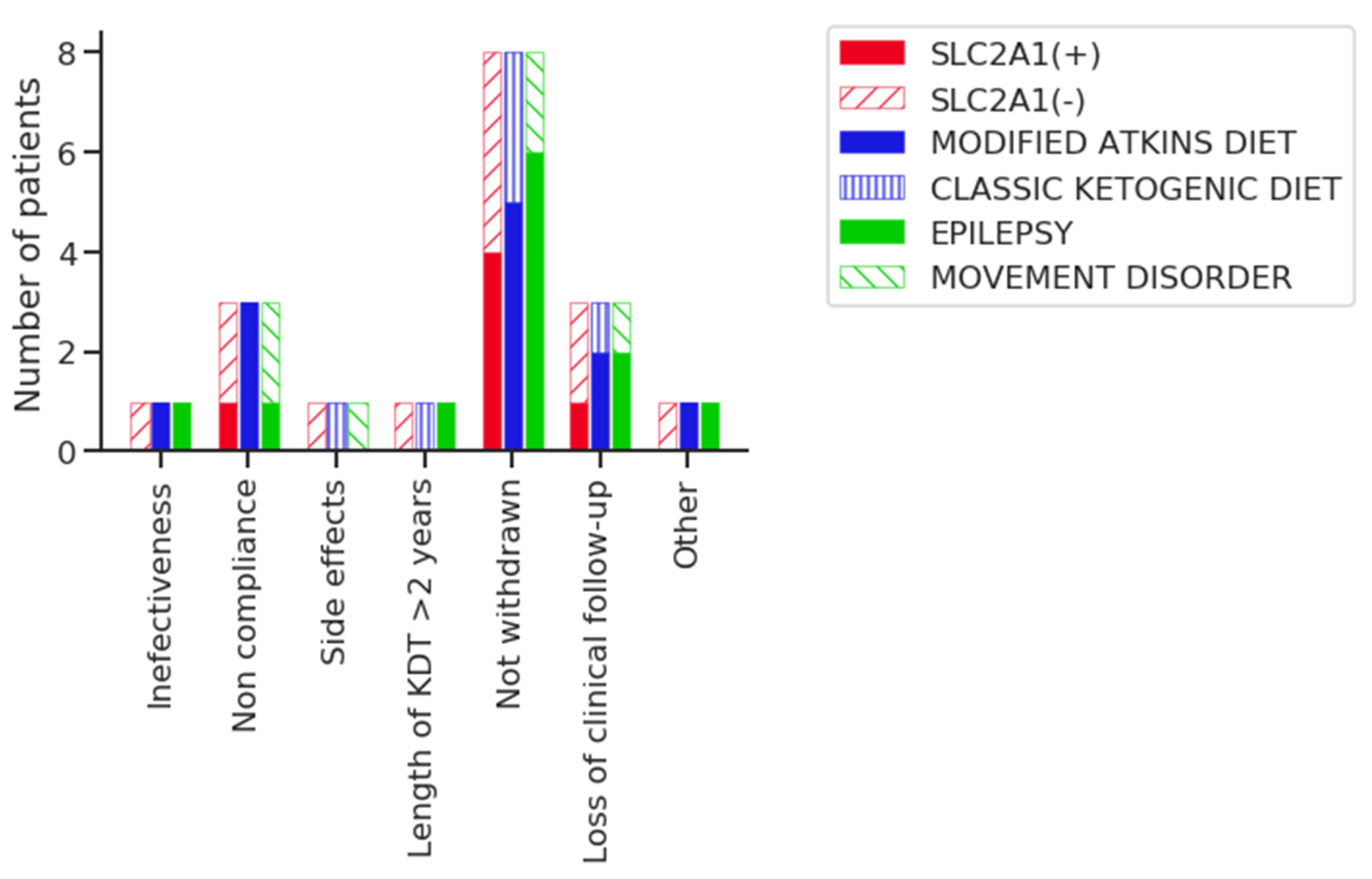
3.1. Outcome of the KDT
3.2. Side Effects
3.3. Nutritional Evolution
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Vivo, D.C.; Trifiletti, R.R.; Jacobson, R.I.; Ronen, G.M.; Behmand, R.A.; Harik, S.I. Defective glucose transport across the blood-brain barrier as a cause of persistent hypoglycorrhachia, seizures, and developmental delay. N. Engl. J. Med. 1991, 325, 703–709. [Google Scholar] [CrossRef]
- Seidner, G.; Alvarez, M.G.; Yeh, J.I.; O’Driscoll, K.R.; Klepper, J.; Stump, T.S.; Wang, D.; Spinner, N.B.; Birnbaum, M.J.; De Vivo, D.C. GLUT-1 deficiency syndrome caused by haploinsufficiency of the blood-brain barrier hexose carrier. Nat. Genet. 1998, 18, 188–191. [Google Scholar] [CrossRef]
- Leen, W.G.; Klepper, J.; Verbeek, M.M.; Leferink, M.; Hofste, T.; Van Engelen, B.G.; Wevers, R.A.; Arthur, T.; Bahi-Buisson, N.; Ballhausen, D.; et al. Glucose transporter-1 deficiency syndrome: The expanding clinical and genetic spectrum of a treatable disorder. Brain 2010, 133, 655–670. [Google Scholar] [CrossRef]
- De Giorgis, V.; Veggiotti, P. GLUT1 deficiency syndrome 2013: Current state of the art. Seizure 2013, 22, 803–811. [Google Scholar] [CrossRef]
- Klepper, J. Absence of SLC2A1 mutations does not exclude glut1 deficiency syndrome. Neuropediatrics 2013, 44, 235–236. [Google Scholar] [CrossRef] [PubMed]
- Klepper, J.; Akman, C.; Armeno, M.; Auvin, S.; Cervenka, M.; Cross, H.J.; De Giorgis, V.; Della Marina, A.; Engelstad, K.; Heussinger, N.; et al. Glut1 Deficiency Syndrome (Glut1DS): State of the art in 2020 and recommendations of the international Glut1DS study group. Epilepsia Open 2020, 5, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Kossoff, E.H. International consensus statement on clinical implementation of the ketogenic diet: Agreement, flexibility, and controversy. Epilepsia 2008, 49, 11–13. [Google Scholar] [CrossRef] [PubMed]
- Leen, W.G.; Mewasingh, L.; Verbeek, M.M.; Kamsteeg, E.J.; van de Warrenburg, B.P.; Willemsen, M.A. Movement disorders in GLUT1 deficiency syndrome respond to the modified Atkins diet. Mov. Disord. 2013, 28, 1439–1442. [Google Scholar] [CrossRef] [PubMed]
- Ohshiro-Sasaki, A.; Shimbo, H.; Takano, K.; Wada, T.; Osaka, H. A three-year-old boy with glucose transporter type 1 deficiency syndrome presenting with episodic ataxia. Pediatric Neurol. 2014, 50, 99–100. [Google Scholar] [CrossRef] [PubMed]
- Amalou, S.; Gras, D.; Ilea, A.; Greneche, M.-O.; Francois, L.; Bellavoine, V.; Delanoe, C.; Auvin, S. Use of modified Atkins diet in glucose transporter type 1 deficiency syndrome. Dev. Med. Child Neurol. 2016, 58, 1193–1199. [Google Scholar] [CrossRef]
- Holliday, M.A.; Segar, W.E. Maintenance need for water in parenteral fluid therapy. Pediatrics 1957, 19, 823–832. [Google Scholar] [CrossRef] [PubMed]
- Klepper, K.J. GLUT1 deficiency syndrome in clinical practice. Epilepsy Res. 2012, 100, 272–277. [Google Scholar] [CrossRef]
- Pong, A.W.; Geary, B.R.; Engelstad, K.M.; Natarajan, A.; Yang, H.; De Vivo, D.C. Glucose transporter type I deficiency syndrome: Epilepsy phenotypes and outcomes. Epilepsia 2012, 53, 1503–1510. [Google Scholar] [CrossRef]
- Leary, L.D.; Wang, D.; Nordli, D.R.; Engelstad, K.; De Vivo, D.C. Seizure characterization and electroencephalographic features in Glut-1 deficiency syndrome. Epilepsia 2003, 44, 701–707. [Google Scholar] [CrossRef]
- Wang, D.; Pascual, J.M.; Yang, H.; Engelstad, K.; Jhung, S.; Sun, R.P.; De Vivo, D.C. Glut-1 deficiency syndrome: Clinical, genetic, and therapeutic aspects. Ann. Neurol. 2004, 57, 111–118. [Google Scholar] [CrossRef]
- Kass, H.R.; Winesett, S.P.; Bessone, S.K.; Turner, Z.; Kossoff, E.H. Use of dietary therapies amongst patients with GLUT1 deficiency syndrome. Seizure 2016, 35, 83–87. [Google Scholar] [CrossRef] [PubMed]
- Ramm-Pettersen, A.; O’Nakken, K.; Skogseid, I.M.; Randby, H.; Skei, E.B.; Bindoff, A.L.; Selmer, K.K. Good outcome in patients with early dietary treatment of GLUT-1 deficiency syndrome: Results from a retrospective Norwegian study. Dev. Med. Child Neurol. 2013, 55, 440–447. [Google Scholar] [CrossRef] [PubMed]
- Legido, M.J.; Ledesma, C.C.; Cuesta, B.B.; Marín, L.L.; Extremera, V.C.; Pérez-Cerdá, C.; González, B.P.; Martín, E.L.; Gutiérrez-Solana, L.G. Estudio de pacientes pediátricos con fenotipo clínico y bioquímico de síndrome de déficit de transportador de glucosa cerebral (GLUT-1). Neurología 2019, in press. [Google Scholar] [CrossRef]
- Schwantje, M.; Verhagen, L.M.; van Hasselt, P.M.; Fuchs, S.A. Glucose transporter type 1 deficiency syndrome and the ketogenic diet. J. Inherit. Metab. Dis. 2020, 43, 216–222. [Google Scholar] [CrossRef] [PubMed]
- Tang, M.; Park, S.H.; De Vivo, D.C.; Monani, U.R. Therapeutic strategies for glucose transporter 1 deficiency syndrome. Ann. Clin. Transl. Neurol. 2019, 6, 1923–1932. [Google Scholar] [CrossRef]
- Haberlandt, E.; Karall, D.; Jud, V.; Baumgartner, S.S.; Zotter, S.; Rostasy, K.; Bauman, M.; Scholl-Buergi, S. Glucose transporter type 1 deficiency syndrome effectively treated with modified Atkins diet. Neuropediatrics 2014, 45, 117–119. [Google Scholar] [CrossRef]
- Ito, S.; Oguni, H.; Ito, Y.; Ishigaki, K.; Ohinata, J.; Osawa, M. Modified Atkins diet therapy for a case with glucose transporter type 1 deficiency syndrome. Brain Dev. 2008, 30, 226–228. [Google Scholar] [CrossRef]
- Fujii, T.; Ito, Y.; Takahashi, S.; Shimono, K.; Natsume, J.; Yanagihara, K.; Oguni, H. Outcome of ketogenic diets in GLUT1 deficiency syndrome in Japan: A nationwide survey. Brain Dev. 2016, 38, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Kossoff, E.H.; Zupec-Kania, B.A.; Auvin, S.; Ballaban-Gil, K.R.; Bergqvist, A.C.; Blackford, R.; Buchhalter, J.R.; Caraballo, R.H.; Cross, J.H.; Dahlin, M.G.; et al. Optimal clinical management of children receiving dietary therapies for epilepsy: Updated recommendations of the International Ketogenic Diet Study Group. Epilepsia Open 2018, 3, 175–192. [Google Scholar] [CrossRef] [PubMed]
- Lin, A.; Turner, Z.; Doerrer, S.C.; Stanfield, A.; Kossoff, E.H. Complications during ketogenic diet initiation: Prevalence, treatment, and influence on seizure outcomes. Pediatric Neurol. 2017, 68, 35–39. [Google Scholar] [CrossRef]
- Kang, H.C.; Chung, D.E.; Kim, D.W.; Kim, H.D. Early and late-onset complications of the ketogenic diet for intractable epilepsy. Epilepsia 2004, 45, 1116–1123. [Google Scholar] [CrossRef] [PubMed]
- Kossoff, E.H.; Pyzik, P.L.; Furth, S.L.; Hladky, H.D.; Freeman, J.M.; Vining, E.P. Kidney stones, carbonic anhydrase inhibitors, and the ketogenic diet. Epilepsia 2002, 43, 1168–1171. [Google Scholar] [CrossRef]
- Sampath, A.; Kossoff, E.H.; Furth, S.L.; Pyzik, P.L.; Vining, E.P. Kidney stones and the ketogenic diet: Risk factors and prevention. J. Child Neurol. 2007, 22, 375–378. [Google Scholar] [CrossRef]
- McNally, M.A.; Pyzik, P.L.; Rubenstein, J.E.; Hamdy, R.F.; Kossoff, E.H. Empiric use of potassium citrate reduces kidney-stone incidence with the ketogenic diet. Pediatrics 2009, 124, e300–e304. [Google Scholar] [CrossRef]
- Zamani, G.R.; Mohammadi, M.; Ashrafi, M.R.; Karimi, P.; Mahmoudi, M.; Badv, R.S.; Tavassoli, A.R.; Malamiri, R.A. The effects of classic ketogenic diet on serum lipid profile in children with refractory seizures. Acta Neurol. Belg. 2016, 116, 529–534. [Google Scholar] [CrossRef]
- Kwiterovich, P.O.; Vining, E.P.; Pyzik, P.; Skolasky, R.; Freeman, J.M. Effect of a high-fat ketogenic diet on plasma levels of lipids, lipoproteins, and apolipoproteins in children. JAMA 2003, 290, 912–920. [Google Scholar] [CrossRef]
- Bertoli, S.; Neri, I.G.; Trentani, C.; Ferraris, C.; De Amicis, R.S.; Battezzati, A.; Veggiotti, P.; De Giorgis, V.; Tagliabue, A. Short-term effects of ketogenic diet on anthropometric parameters, body fat distribution, and inflammatory cytokine production in GLUT1 deficiency syndrome. Nutrition 2015, 31, 981–987. [Google Scholar] [CrossRef]
- Heussinger, N.; Della Marina, A.; Beyerlein, A.; Leiendecker, B.; Hermann-Alves, S.; Pozza, R.D.; Klepper, J. 10 patients, 10 years–Long term follow-up of cardiovascular risk factors in Glut1 deficiency treated with ketogenic diet therapies: A prospective, multicenter case series. Clin. Nutr. 2018, 37, 2246–2251. [Google Scholar] [CrossRef]
- Christodoulides, S.S.; Neal, E.G.; Fitzsimmons, G.; Chaffe, H.M.; Jeanes, Y.M.; Aitkenhead, H.; Cross, J.H. The effect of the classical and medium chain triglyceride ketogenic diet on vitamin and mineral levels. J. Hum. Nutr. Diet. 2012, 25, 16–26. [Google Scholar] [CrossRef]
- Coppola, G.; Epifanio, G.; Auricchio, G.; Federico, R.R.; Resicato, G.; Pascotto, A. Plasma free carnitine in epilepsy children, adolescents and young adults treated with old and new antiepileptic drugs with or without ketogenic diet. Brain Dev. 2006, 28, 358–365. [Google Scholar] [CrossRef] [PubMed]
- Berry-Kravis, E.; Booth, G.; Sanchez, A.C.; Woodbury-Kolb, J. Carnitine levels and the ketogenic diet. Epilepsia 2001, 42, 1445–1451. [Google Scholar] [CrossRef] [PubMed]
- Cappuccio, G.; Pinelli, M.; Alagia, M.; Donti, T.; Day-Salvatore, D.-L.; Veggiotti, P.; De Giorgis, V.; Lunghi, S.; Vari, M.S.; Striano, P.; et al. Biochemical phenotyping unravels novel metabolic abnormalities and potential biomarkers associated with treatment of GLUT1 deficiency with ketogenic diet. PLoS ONE 2017, 12, e0184022. [Google Scholar] [CrossRef] [PubMed]
- Neal, E.G.; Chaffe, H.M.; Edwards, N.; Lawson, M.S.; Schwartz, R.H.; Cross, J.H. Growth of children on classical and medium chain triglyceride diets. Pediatrics 2008, 122, e334–e340. [Google Scholar] [CrossRef]
- Vining, E.P.; Pyzik, P.; McGrogan, J.; Hladky, H.; Anand, A.; Kriegler, S.; Freeman, J.M. Growth of children on the ketogenic diet. Dev. Med. Child Neurol. 2002, 44, 796–802. [Google Scholar] [CrossRef]
- Williams, S.; Basualdo-Hammond, C.; Curtis, R.; Schuller, R. Growth retardation in children with epilepsy on the ketogenic diet: A retrospective chart review. J. Am. Diet. Assoc. 2002, 102, 405–407. [Google Scholar] [CrossRef]
- Groleau, V.; Schall, J.I.; Stallings, V.A.; Bergqvist, C.A. Long-term impact of the ketogenic diet on growth and resting energy expenditure in children with intractable epilepsy. Dev. Med. Child. Neurol 2014, 56, 898–904. [Google Scholar] [CrossRef]
- Peterson, S.J.; Tangney, C.C.; Pimentel-Zablah, E.M.; Hjelmgren, B.; Booth, G.; Berry-Karvis, E. Changes in Growth and Seizure Reduction in Children on the Ketogenic Diet as a Treatment for Intractable Epilepsy. J. Am. Diet. Assoc. 2005, 105, 718–725. [Google Scholar] [CrossRef]
- Ferraris, C.; Guglielmetti, M.; Pasca, L.; De Giorgis, V.; Ferraro, O.E.; Brambilla, I.; Leone, A.; De Amicis, R.; Bertoli, S.; Veggiotti, P.; et al. Impact of the ketogenic diet on linear growth in children: A single-center retrospective analysis of 34 cases. Nutrients 2019, 11, 1442. [Google Scholar] [CrossRef] [PubMed]
- Wibisono, C.; Rowe, N.; Beavis, E.; Kepreotes, H.; Mackie, F.E.; Lawson, J.A.; Cardamone, M. Ten-year single-center experience of the ketogenic diet: Factors influencing efficacy, tolerability, and compliance. J. Pediatrics 2015, 166, 1030–1036. [Google Scholar] [CrossRef] [PubMed]
- Lambrechts, D.; Kinderen, R.; Vles, S.; Louw, A.; Aldenkamp, A.; Majoie, M. The MCT-ketogenic diet as a treatment option in refractory childhood epilepsy: A prospective study with 2-year follow-up. Epilepsy Behav. 2015, 51, 261–266. [Google Scholar] [CrossRef]
- Kim, J.T.; Kang, H.C.; Song, J.E.; Lee, M.J.; Lee, Y.J.; Lee, E.J.; Lee, J.S.; Kim, H.D. Catch-up growth after long-term implementation and weaning from ketogenic diet in pediatric epileptic patients. Clin. Nutr. 2013, 32, 98–103. [Google Scholar] [CrossRef] [PubMed]
- Ruiz Herrero, J.; Cañedo Villarroya, E.; García Peñas, J.J.; García Alcolea, B.; Gómez Fernández, B.; Puerta Macfarland, L.A.; Pedron Giner, C. Safety and effectiveness of the prolonged treatment of children with a ketogenic diet. Nutrients 2020, 12, 306. [Google Scholar] [CrossRef] [PubMed]
- Armeno, M.; Verini, A.; Del Pino, M.; Araujo, M.B.; Mestre, G.; Reyes, G.; Caraballo, R.H. A prospective study on changes in nutritional status and growth following two years of Ketogenic Diet (KD) therapy in children with refractory epilepsy. Nutrients 2019, 11, 1596. [Google Scholar] [CrossRef]


| Patients on a KDT (n = 18) | |
|---|---|
| Sex | Males, 8 (55.6%) Females, 10 (44.4%) |
| Median z-score of baseline head circumference (IQR) | −0.5 (−1.9–0.47) |
| Median age at the onset of symptoms (range) | 14 months (4 days to 7 years and 8 months) |
| Seizure (n = 13) | Myoclonic, 5 (41.6%) Typical absences, 3 (25%) Various, 2 (16.6%) Focal onset, 1 (8.3%) Generalized tonic-clonic, 1(8.3%) Clonic, 1 (8.3%) |
| Movements disorders (n = 5) | Paroxysmal exercise-induced dyskinesias, 1 Dystonia and nystagmus, 1 Dystonia and ataxia, 1 Paroxysmal eye movements, 1 Complex movement disorder with a predominance of intentional and postural tremor, 1 |
| Median age at the onset of KDT (range) | 5 years and 2 months (3.5 months to 17 years and 4 months) |
| Age at the onset of KDT | Infant, 2 (11%) 1–2 years, 2 (11%) 2–5 years, 5 (28%) 5–10 years, 6 (33%) >10 years, 3 (17%) |
| Median time from debut to the onset of KDT (range) | 3 year and 1 month (49 days to 13 years and 4 months) |
| Type of KDT | Classic ketogenic diet (CKD) 3:1, 6 (33.3%) Modified Atkins diet (MAD), 12 (66.7%) |
| Median time to reach ketosis (range) | 3.8 days (2–9 days) |
| Median length of KDT (IQR) | 463 days (170–1863 days) |
| Reasons for withdrawing KDT (n = 10) | Difficulties in compliance, 3 (16.6%) Loss of clinical follow-up, 3 (16.6%) Ineffectiveness, 1 (5.55%) Adverse effects, 1 (5.55%) Length of KDT >2 years, 1 (5.55%) Other, 1 (5.55%) |
| Time on the Diet | 3 Months (n = 17) | 6 Months (n = 13) | 12 Months (n = 10) | 24 Months (n = 8) |
|---|---|---|---|---|
| Total of patients suffering side effects | 10 | 5 | 7 | 5 |
| Constipation | 4 | 2 | 1 | 1 |
| Abdominal pain | - | - | - | 1 |
| Nausea/vomiting | 1 | - | - | - |
| Anorexia | 1 | - | - | - |
| Dehydration | 1 | - | - | - |
| Hypoglycemia | 1 | - | - | - |
| Hypercholesterolemia 1 | 4 | 2 | 3 | 2 |
| Hypertriglyceridemia 2 | 1 | - | 1 | - |
| Elevated AST, ALT, or GGT 3 | 1 | - | - | - |
| Hypercalciuria 4 | 3 | 2 | 3 | 1 |
| Hyperuricemia 5 | - | - | 1 | 3 |
| Metabolic acidosis 6 | 3 | - | - | - |
| Lab Reference Values | Mean ± SD | |||||
|---|---|---|---|---|---|---|
| Nutritional Markers in Blood | Baseline | 3 m | 6 m | 12 m | 24 m | |
| Prealbumin (mg/dL) | 15–40 | 17.7 ± 3.5 | 16.2 ± 3.5 | 15.7 ± 1.9 | 15.3 ± 2.9 | 17.1 ± 3.6 |
| Retinol binding protein (RBP) (mg/dL) | 2.5–6.9 | 3.4 ± 1.2 | 1.41 | 1.4 ± 0.07 | 2.5 ± 0.7 | 2.5 ± 0.7 |
| Ferritine (ng/mL) | 7–140 (<15 y) 15–175 (≥15 y) | 31 ± 19 | 48 ± 31 | 55 ± 31 | 52 ± 27 | 40 ± 21 |
| Micronutrients | ||||||
| Magnesium (mg/dL) | 1.5–2.5 | 2.2 ± 0.28 | 2.0 ± 0.11 | 1.9 ± 0.1 | 2.0 ± 0.08 | 1.85 ± 0.13 |
| Selenium (µg/L) | 70–120 | 73 ± 8.6 | - | 76.4 | 89 ± 6.8 | 84.4 ± 9.0 |
| Zinc (µg/dL) | 70–150 | 107 ± 4.0 | - | 97 | 103 ± 18.8 | 103 ± 10.3 |
| Total carnitine or Levocarnitine (µmol/L) | 21.5–64.5 20–50 | 52 ± 23 35 ± 2.5 | - - | 70.5 48.6 | 61.9 ± 40.3 34.8 ± 6.3 | 63 ± 25.4 41 ± 13.4 |
| Vitamin A (mg/L) | 0.2–0.6 | 0.31 ± 0.08 | - | 0.21 ± 0.03 | 0.28 ± 0.08 | 0.27 ± 0.07 |
| Vitamin E (mg/L) | 3–9 (<12 y) 5–20 (≥12 y) | 5.3 ± 1.2 | - | 3.53 | 6.0 ± 2.5 | 5.1 ± 1.4 |
| Folic acid (ng/mL) | 3.9–23.9 | 11 ± 5.2 | - | 18.1 ± 9.2 | 20.6 ± 7.8 | 17 ± 4.6 |
| Vitamin B12 (pg/mL) | 250–914 | 715 ± 545 | 1023 ± 291 | 612 ± 9 | 636 ± 353 | 859 ± 504 |
| 25-hydroxyvitamin D (ng/mL) | 20–80 | 33.3 ± 12.3 | - | 40 ± 6 | 36.5 ± 9.2 | 31 ± 10.0 |
| Calcium (mg/dL) | 8.8–10.8 | 9.6 ± 0.4 | 9.7 ± 0.4 | 9.5 ± 0.19 | 9.6 ± 0.2 | 9.6 ± 0.2 |
| Phosphorus (mg/dL) | 4.5–6.5 | 5.4 ± 0.9 | 5.0 ± 0.6 | 4.8 ± 0.5 | 4.8 ± 0.7 | 4.0 ± 0.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz Herrero, J.; Cañedo Villarroya, E.; González Gutiérrez-Solana, L.; García Alcolea, B.; Gómez Fernández, B.; Puerta Macfarland, L.A.; Pedrón-Giner, C. Classic Ketogenic Diet and Modified Atkins Diet in SLC2A1 Positive and Negative Patients with Suspected GLUT1 Deficiency Syndrome: A Single Center Analysis of 18 Cases. Nutrients 2021, 13, 840. https://doi.org/10.3390/nu13030840
Ruiz Herrero J, Cañedo Villarroya E, González Gutiérrez-Solana L, García Alcolea B, Gómez Fernández B, Puerta Macfarland LA, Pedrón-Giner C. Classic Ketogenic Diet and Modified Atkins Diet in SLC2A1 Positive and Negative Patients with Suspected GLUT1 Deficiency Syndrome: A Single Center Analysis of 18 Cases. Nutrients. 2021; 13(3):840. https://doi.org/10.3390/nu13030840
Chicago/Turabian StyleRuiz Herrero, Jana, Elvira Cañedo Villarroya, Luis González Gutiérrez-Solana, Beatriz García Alcolea, Begoña Gómez Fernández, Laura Andrea Puerta Macfarland, and Consuelo Pedrón-Giner. 2021. "Classic Ketogenic Diet and Modified Atkins Diet in SLC2A1 Positive and Negative Patients with Suspected GLUT1 Deficiency Syndrome: A Single Center Analysis of 18 Cases" Nutrients 13, no. 3: 840. https://doi.org/10.3390/nu13030840
APA StyleRuiz Herrero, J., Cañedo Villarroya, E., González Gutiérrez-Solana, L., García Alcolea, B., Gómez Fernández, B., Puerta Macfarland, L. A., & Pedrón-Giner, C. (2021). Classic Ketogenic Diet and Modified Atkins Diet in SLC2A1 Positive and Negative Patients with Suspected GLUT1 Deficiency Syndrome: A Single Center Analysis of 18 Cases. Nutrients, 13(3), 840. https://doi.org/10.3390/nu13030840






