Current Evidence and Possible Future Applications of Creatine Supplementation for Older Adults
Abstract
1. Introduction
2. Creatine
3. Potential of Creatine Supplementation for Sarcopenia
4. Potential of Creatine Supplementation for Osteoporosis
5. Potential of Creatine Supplementation for Osteosarcopenia
6. Potential of Creatine Supplementation for Sarcopenic Obesity
7. Potential of Creatine Supplementation for Physical Frailty
8. Potential of Creatine Supplementation for Cachexia
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Writing Group for the European Working Group on Sarcopenia in Older People 2 (EWGSOP2), and the Extended Group for EWGSOP2 Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef]
- Shafiee, G.; Keshtkar, A.; Soltani, A.; Ahadi, Z.; Larijani, B.; Heshmat, R. Prevalence of sarcopenia in the world: A systematic review and meta- analysis of general population studies. J. Diabetes Metab. Disord. 2017, 16, 1–10. [Google Scholar] [CrossRef]
- Tournadre, A.; Vial, G.; Capel, F.; Soubrier, M.; Boirie, Y. Sarcopenia. Joint Bone Spine 2019, 86, 309–314. [Google Scholar] [CrossRef]
- Mitchell, W.K.; Williams, J.; Atherton, P.; Larvin, M.; Lund, J.; Narici, M. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front. Physiol. 2012, 3, 260. [Google Scholar] [CrossRef]
- Candow, D.G.; Forbes, S.C.; Chilibeck, P.D.; Cornish, S.M.; Antonio, J.; Kreider, R.B. Effectiveness of Creatine Supplementation on Aging Muscle and Bone: Focus on Falls Prevention and Inflammation. J. Clin. Med. 2019, 8, 488. [Google Scholar] [CrossRef]
- Chilibeck, P.D.; Kaviani, M.; Candow, D.G.; Zello, G.A. Effect of creatine supplementation during resistance training on lean tissue mass and muscular strength in older adults: A meta-analysis. Open Access J. Sports Med. 2017, 8, 213–226. [Google Scholar] [CrossRef]
- Forbes, S.C.; Chilibeck, P.D.; Candow, D.G. Creatine Supplementation During Resistance Training Does Not Lead to Greater Bone Mineral Density in Older Humans: A Brief Meta-Analysis. Front. Nutr. 2018, 5, 27. [Google Scholar] [CrossRef]
- Gualano, B.; Rawson, E.S.; Candow, D.G.; Chilibeck, P.D. Creatine supplementation in the aging population: Effects on skeletal muscle, bone and brain. Amino Acids 2016, 48, 1793–1805. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Wyss, M.; Kaddurah-Daouk, R. Creatine and creatinine metabolism. Physiol. Rev. 2000, 80, 1107–1213. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G. Sarcopenia: Current theories and the potential beneficial effect of creatine application strategies. Biogerontology 2011, 12, 273–281. [Google Scholar] [CrossRef]
- Candow, D.G.; Forbes, S.C.; Little, J.P.; Cornish, S.M.; Pinkoski, C.; Chilibeck, P.D. Effect of nutritional interventions and resistance exercise on aging muscle mass and strength. Biogerontology 2012, 13, 345–358. [Google Scholar] [CrossRef]
- Candow, D.G.; Forbes, S.C.; Chilibeck, P.D.; Cornish, S.M.; Antonio, J.; Kreider, R.B. Variables Influencing the Effectiveness of Creatine Supplementation as a Therapeutic Intervention for Sarcopenia. Front. Nutr. 2019, 6, 124. [Google Scholar] [CrossRef]
- Forbes, S.C.; Little, J.P.; Candow, D.G. Exercise and nutritional interventions for improving aging muscle health. Endocrine 2012, 42, 29–38. [Google Scholar] [CrossRef]
- Candow, D.G.; Chilibeck, P.D.; Forbes, S.C. Creatine supplementation and aging musculoskeletal health. Endocrine 2014, 45, 354–361. [Google Scholar] [CrossRef] [PubMed]
- Devries, M.C.; Phillips, S.M. Creatine supplementation during resistance training in older adults-a meta-analysis. Med. Sci. Sports Exerc. 2014, 46, 1194–1203. [Google Scholar] [CrossRef] [PubMed]
- Candow, D.G.; Chilibeck, P.D.; Chad, K.E.; Chrusch, M.J.; Davison, K.S.; Burke, D.G. Effect of ceasing creatine supplementation while maintaining resistance training in older men. J. Aging Phys. Act. 2004, 12, 219–231. [Google Scholar] [CrossRef] [PubMed]
- Aguiar, A.F.; Januario, R.S.; Junior, R.P.; Gerage, A.M.; Pina, F.L.; do Nascimento, M.A.; Padovani, C.R.; Cyrino, E.S. Long-term creatine supplementation improves muscular performance during resistance training in older women. Eur. J. Appl. Physiol. 2013, 113, 987–996. [Google Scholar] [CrossRef]
- Alves, C.R.; Merege Filho, C.A.; Benatti, F.B.; Brucki, S.; Pereira, R.M.; de Sa Pinto, A.L.; Lima, F.R.; Roschel, H.; Gualano, B. Creatine supplementation associated or not with strength training upon emotional and cognitive measures in older women: A randomized double-blind study. PLoS ONE 2013, 8, e76301. [Google Scholar] [CrossRef]
- Bemben, M.G.; Witten, M.S.; Carter, J.M.; Eliot, K.A.; Knehans, A.W.; Bemben, D.A. The effects of supplementation with creatine and protein on muscle strength following a traditional resistance training program in middle-aged and older men. J. Nutr. Health Aging 2010, 14, 155–159. [Google Scholar] [CrossRef]
- Eliot, K.A.; Knehans, A.W.; Bemben, D.A.; Witten, M.S.; Carter, J.; Bemben, M.G. The effects of creatine and whey protein supplementation on body composition in men aged 48 to 72 years during resistance training. J. Nutr. Health Aging 2008, 12, 208–212. [Google Scholar] [CrossRef]
- Bermon, S.; Venembre, P.; Sachet, C.; Valour, S.; Dolisi, C. Effects of creatine monohydrate ingestion in sedentary and weight-trained older adults. Acta Physiol. Scand. 1998, 164, 147–155. [Google Scholar] [CrossRef]
- Bernat, P.; Candow, D.G.; Gryzb, K.; Butchart, S.; Schoenfeld, B.J.; Bruno, P. Effects of high-velocity resistance training and creatine supplementation in untrained healthy aging males. Appl. Physiol. Nutr. Metab. 2019, 44, 1246–1253. [Google Scholar] [CrossRef] [PubMed]
- Brose, A.; Parise, G.; Tarnopolsky, M.A. Creatine supplementation enhances isometric strength and body composition improvements following strength exercise training in older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2003, 58, 11–19. [Google Scholar] [CrossRef]
- Candow, D.G.; Little, J.P.; Chilibeck, P.D.; Abeysekara, S.; Zello, G.A.; Kazachkov, M.; Cornish, S.M.; Yu, P.H. Low-dose creatine combined with protein during resistance training in older men. Med. Sci. Sports Exerc. 2008, 40, 1645–1652. [Google Scholar] [CrossRef]
- Candow, D.G.; Vogt, E.; Johannsmeyer, S.; Forbes, S.C.; Farthing, J.P. Strategic creatine supplementation and resistance training in healthy older adults. Appl. Physiol. Nutr. Metab. 2015, 40, 689–694. [Google Scholar] [CrossRef]
- Candow, D.G.; Chilibeck, P.D.; Gordon, J.; Vogt, E.; Landeryou, T.; Kaviani, M.; Paus-Jensen, L. Effect of 12 months of creatine supplementation and whole-body resistance training on measures of bone, muscle and strength in older males. Nutr. Health 2020, 260106020975247. [Google Scholar] [CrossRef]
- Chilibeck, P.D.; Candow, D.G.; Landeryou, T.; Kaviani, M.; Paus-Jenssen, L. Effects of Creatine and Resistance Training on Bone Health in Postmenopausal Women. Med. Sci. Sports Exerc. 2015, 47, 1587–1595. [Google Scholar] [CrossRef] [PubMed]
- Chrusch, M.J.; Chilibeck, P.D.; Chad, K.E.; Davison, K.S.; Burke, D.G. Creatine supplementation combined with resistance training in older men. Med. Sci. Sports Exerc. 2001, 33, 2111–2117. [Google Scholar] [CrossRef]
- Cooke, M.B.; Brabham, B.; Buford, T.W.; Shelmadine, B.D.; McPheeters, M.; Hudson, G.M.; Stathis, C.; Greenwood, M.; Kreider, R.; Willoughby, D.S. Creatine supplementation post-exercise does not enhance training-induced adaptations in middle to older aged males. Eur. J. Appl. Physiol. 2014, 114, 1321–1332. [Google Scholar] [CrossRef] [PubMed]
- Deacon, S.J.; Vincent, E.E.; Greenhaff, P.L.; Fox, J.; Steiner, M.C.; Singh, S.J.; Morgan, M.D. Randomized controlled trial of dietary creatine as an adjunct therapy to physical training in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 2008, 178, 233–239. [Google Scholar] [CrossRef]
- Eijnde, B.O.; Van Leemputte, M.; Goris, M.; Labarque, V.; Taes, Y.; Verbessem, P.; Vanhees, L.; Ramaekers, M.; Vanden Eynde, B.; Van Schuylenbergh, R.; et al. Effects of creatine supplementation and exercise training on fitness in men 55-75 yr old. J. Appl. Physiol. (1985) 2003, 95, 818–828. [Google Scholar] [CrossRef]
- Gualano, B.; de Salles Painelli, V.; Roschel, H.; Lugaresi, R.; Dorea, E.; Artioli, G.G.; Lima, F.R.; da Silva, M.E.; Cunha, M.R.; Seguro, A.C.; et al. Creatine supplementation does not impair kidney function in type 2 diabetic patients: A randomized, double-blind, placebo-controlled, clinical trial. Eur. J. Appl. Physiol. 2011, 111, 749–756. [Google Scholar] [CrossRef]
- Gualano, B.; Macedo, A.R.; Alves, C.R.; Roschel, H.; Benatti, F.B.; Takayama, L.; de Sa Pinto, A.L.; Lima, F.R.; Pereira, R.M. Creatine supplementation and resistance training in vulnerable older women: A randomized double- blind placebo-controlled clinical trial. Exp. Gerontol. 2014, 53, 7–15. [Google Scholar] [CrossRef] [PubMed]
- Johannsmeyer, S.; Candow, D.G.; Brahms, C.M.; Michel, D.; Zello, G.A. Effect of creatine supplementation and drop-set resistance training in untrained aging adults. Exp. Gerontol. 2016, 83, 112–119. [Google Scholar] [CrossRef]
- Neves, M.; Gualano, B.; Roschel, H.; Fuller, R.; Benatti, F.B.; Pinto, A.L.; Lima, F.R.; Pereira, R.M.; Lancha, A.H.; Bonfa, E. Beneficial effect of creatine supplementation in knee osteoarthritis. Med. Sci. Sports Exerc. 2011, 43, 1538–1543. [Google Scholar] [CrossRef] [PubMed]
- Pinto, C.L.; Botelho, P.B.; Carneiro, J.A.; Mota, J.F. Impact of creatine supplementation in combination with resistance training on lean mass in the elderly. J. Cachexia Sarcopenia Muscle 2016, 7, 413–421. [Google Scholar] [CrossRef]
- Smolarek, A.C.; McAnulty, S.R.; Ferreira, L.H.; Cordeiro, G.R.; Alessi, A.; Rebesco, D.B.; Honorato, I.C.; Laat, E.F.; Mascarenhas, L.P.; Souza-Junior, T.P. Effect of 16 weeks of strength training and creatine supplementation on strength and cognition in older adults: A pilot study. J. Exerc. Physiol. Online 2020, 23, 88–94. [Google Scholar]
- Baumgartner, R.N.; Koehler, K.M.; Gallagher, D.; Romero, L.; Heymsfield, S.B.; Ross, R.R.; Garry, P.J.; Lindeman, R.D. Epidemiology of sarcopenia among the elderly in New Mexico. Am. J. Epidemiol. 1998, 147, 755–763. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.B.; Kupelian, V.; Visser, M.; Simonsick, E.; Goodpaster, B.; Nevitt, M.; Kritchevsky, S.B.; Tylavsky, F.A.; Rubin, S.M.; Harris, T.B. Health ABC Study Investigators Sarcopenia: Alternative definitions and associations with lower extremity function. J. Am. Geriatr. Soc. 2003, 51, 1602–1609. [Google Scholar] [CrossRef] [PubMed]
- Macrae, P.G.; Lacourse, M.; Moldavon, R. Physical performance measures that predict faller status in community- dwelling older adults. J. Orthop. Sports Phys. Ther. 1992, 16, 123–128. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.C.; Candow, D.G.; Ferreira, L.H.B.; Souza-Junior, T.P. Effects of Creatine Supplementation on Properties of Muscle, Bone, and Brain Function in Older Adults: A Narrative Review. J. Diet. Suppl. 2021, 1–18. [Google Scholar] [CrossRef]
- Clynes, M.A.; Harvey, N.C.; Curtis, E.M.; Fuggle, N.R.; Dennison, E.M.; Cooper, C. The epidemiology of osteoporosis. Br. Med. Bull. 2020, 133, 105–117. [Google Scholar] [CrossRef]
- Reginster, J.Y.; Beaudart, C.; Buckinx, F.; Bruyere, O. Osteoporosis and sarcopenia: Two diseases or one? Curr. Opin. Clin. Nutr. Metab. Care 2016, 19, 31–36. [Google Scholar] [CrossRef] [PubMed]
- Chilibeck, P.D.; Chrusch, M.J.; Chad, K.E.; Shawn Davison, K.; Burke, D.G. Creatine monohydrate and resistance training increase bone mineral content and density in older men. J. Nutr. Health Aging 2005, 9, 352–353. [Google Scholar]
- Candow, D.G.; Forbes, S.C.; Vogt, E. Effect of pre-exercise and post-exercise creatine supplementation on bone mineral content and density in healthy aging adults. Exp. Gerontol. 2019, 119, 89–92. [Google Scholar] [CrossRef]
- Lobo, D.M.; Tritto, A.C.; da Silva, L.R.; de Oliveira, P.B.; Benatti, F.B.; Roschel, H.; Niess, B.; Gualano, B.; Pereira, M. Effects of long-term low-dose dietary creatine supplementation in older women. Exp. Gerontol. 2015, 70, 97–104. [Google Scholar] [CrossRef]
- Sales, L.P.; Pinto, A.J.; Rodrigues, S.F.; Alvarenga, J.C.; Goncalves, N.; Sampaio-Barros, M.M.; Benatti, F.B.; Gualano, B.; Rodrigues Pereira, R.M. Creatine Supplementation (3 g/d) and Bone Health in Older Women: A 2- Year, Randomized, Placebo-Controlled Trial. J. Gerontol. A Biol. Sci. Med. Sci. 2020, 75, 931–938. [Google Scholar] [CrossRef] [PubMed]
- Eriksen, E.F. Cellular mechanisms of bone remodeling. Rev. Endocr. Metab. Disord. 2010, 11, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Boskey, A.L.; Coleman, R. Aging and bone. J. Dent. Res. 2010, 89, 1333–1348. [Google Scholar] [CrossRef]
- Salech, F.; Marquez, C.; Lera, L.; Angel, B.; Saguez, R.; Albala, C. Osteosarcopenia Predicts Falls, Fractures, and Mortality in Chilean Community-Dwelling Older Adults. J. Am. Med. Dir. Assoc. 2020. [Google Scholar] [CrossRef] [PubMed]
- Kirk, B.; Miller, S.; Zanker, J.; Duque, G. A clinical guide to the pathophysiology, diagnosis and treatment of osteosarcopenia. Maturitas 2020, 140, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Kirk, B.; Zanker, J.; Duque, G. Osteosarcopenia: Epidemiology, diagnosis, and treatment-facts and numbers. J. Cachexia Sarcopenia Muscle 2020, 11, 609–618. [Google Scholar] [CrossRef]
- Atlihan, R.; Kirk, B.; Duque, G. Non-Pharmacological Interventions in Osteosarcopenia: A Systematic Review. J. Nutr. Health Aging 2021, 25, 25–32. [Google Scholar] [CrossRef]
- Antolic, A.; Roy, B.D.; Tarnopolsky, M.A.; Zernicke, R.F.; Wohl, G.R.; Shaughnessy, S.G.; Bourgeois, J.M. Creatine monohydrate increases bone mineral density in young Sprague-Dawley rats. Med. Sci. Sports Exerc. 2007, 39, 816–820. [Google Scholar] [CrossRef] [PubMed]
- Gerber, I.; ap Gwynn, I.; Alini, M.; Wallimann, T. Stimulatory effects of creatine on metabolic activity, differentiation and mineralization of primary osteoblast-like cells in monolayer and micromass cell cultures. Eur. Cell. Mater. 2005, 10, 8–22. [Google Scholar] [CrossRef] [PubMed]
- Kirk, B.; Feehan, J.; Lombardi, G.; Duque, G. Muscle, Bone, and Fat Crosstalk: The Biological Role of Myokines, Osteokines, and Adipokines. Curr. Osteoporos Rep. 2020, 18, 388–400. [Google Scholar] [CrossRef]
- Ho-Pham, L.T.; Nguyen, U.D.; Nguyen, T.V. Association between lean mass, fat mass, and bone mineral density: A meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 30–38. [Google Scholar] [CrossRef]
- Kirk, B.; Phu, S.; Brennan-Olsen, S.L.; Bani Hassan, E.; Duque, G. Associations between osteoporosis, the severity of sarcopenia and fragility fractures in community-dwelling older adults. Eur. Geriatr. Med. 2020, 11, 443–450. [Google Scholar] [CrossRef]
- Burke, D.G.; Candow, D.G.; Chilibeck, P.D.; MacNeil, L.G.; Roy, B.D.; Tarnopolsky, M.A.; Ziegenfuss, T. Effect of creatine supplementation and resistance-exercise training on muscle insulin-like growth factor in young adults. Int. J. Sport Nutr. Exerc. Metab. 2008, 18, 389–398. [Google Scholar] [CrossRef]
- Farshidfar, F.; Pinder, M.A.; Myrie, S.B. Creatine Supplementation and Skeletal Muscle Metabolism for Building Muscle Mass- Review of the Potential Mechanisms of Action. Curr. Protein Pept. Sci. 2017, 18, 1273–1287. [Google Scholar] [CrossRef]
- Saremi, A.; Gharakhanloo, R.; Sharghi, S.; Gharaati, M.R.; Larijani, B.; Omidfar, K. Effects of oral creatine and resistance training on serum myostatin and GASP-1. Mol. Cell. Endocrinol. 2010, 317, 25–30. [Google Scholar] [CrossRef]
- El-Kotob, R.; Ponzano, M.; Chaput, J.P.; Janssen, I.; Kho, M.E.; Poitras, V.J.; Ross, R.; Ross-White, A.; Saunders, T.J.; Giangregorio, L.M. Resistance training and health in adults: An overview of systematic reviews. Appl. Physiol. Nutr. Metab. 2020, 45, S165–S179. [Google Scholar] [CrossRef]
- Kirk, B.; Prokopidis, K.; Duque, G. Nutrients to mitigate osteosarcopenia: The role of protein, vitamin D and calcium. Curr. Opin. Clin. Nutr. Metab. Care 2021, 24, 25–32. [Google Scholar] [CrossRef]
- Jakicic, J.M.; Powell, K.E.; Campbell, W.W.; Dipietro, L.; Pate, R.R.; Pescatello, L.S.; Collins, K.A.; Bloodgood, B.; Piercy, K.L. 2018 physical activity guidelines advisory committee* Physical Activity and the Prevention of Weight Gain in Adults: A Systematic Review. Med. Sci. Sports Exerc. 2019, 51, 1262–1269. [Google Scholar] [CrossRef] [PubMed]
- Goodpaster, B.H.; Carlson, C.L.; Visser, M.; Kelley, D.E.; Scherzinger, A.; Harris, T.B.; Stamm, E.; Newman, A.B. Attenuation of skeletal muscle and strength in the elderly: The Health ABC Study. J. Appl. Physiol. 2001, 90, 2157–2165. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.S.; Lee, Y.; Chung, Y.S.; Lee, D.J.; Joo, N.S.; Hong, D.; Song, G.; Kim, H.J.; Choi, Y.J.; Kim, K.M. Prevalence of sarcopenia and sarcopenic obesity in the Korean population based on the Fourth Korean National Health and Nutritional Examination Surveys. J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 1107–1113. [Google Scholar] [CrossRef]
- Roh, E.; Choi, K.M. Health Consequences of Sarcopenic Obesity: A Narrative Review. Front. Endocrinol. (Lausanne) 2020, 11, 332. [Google Scholar] [CrossRef] [PubMed]
- Jee, S.H.; Sull, J.W.; Park, J.; Lee, S.Y.; Ohrr, H.; Guallar, E.; Samet, J.M. Body-mass index and mortality in Korean men and women. N. Engl. J. Med. 2006, 355, 779–787. [Google Scholar] [CrossRef]
- Despres, J.P. Abdominal obesity and cardiovascular disease: Is inflammation the missing link? Can. J. Cardiol. 2012, 28, 642–652. [Google Scholar] [CrossRef] [PubMed]
- Alberti, K.G.; Zimmet, P.; Shaw, J.; IDF Epidemiology Task Forcesensus Group. The metabolic syndrome—A new worldwide definition. Lancet 2005, 366, 1059–1062. [Google Scholar] [CrossRef]
- Lee, S.Y.; Park, H.S.; Kim, D.J.; Han, J.H.; Kim, S.M.; Cho, G.J.; Kim, D.Y.; Kwon, H.S.; Kim, S.R.; Lee, C.B.; et al. Appropriate waist circumference cutoff points for central obesity in Korean adults. Diabetes Res. Clin. Pract. 2007, 75, 72–80. [Google Scholar] [CrossRef] [PubMed]
- Garvey, W.T.; Mechanick, J.I.; Brett, E.M.; Garber, A.J.; Hurley, D.L.; Jastreboff, A.M.; Nadolsky, K.; Pessah- Pollack, R.; Plodkowski, R. Reviewers of the AACE/ACE Obesity Clinical Practice Guidelines American Association of Clinical Endocrinologists and American College of Endocrinology Comprehensive Clinical Practice Guidelines for Medical Care of Patients with Obesity. Endocr. Pract. 2016, 22, 1–203. [Google Scholar] [CrossRef]
- Batsis, J.A.; Mackenzie, T.A.; Lopez-Jimenez, F.; Bartels, S.J. Sarcopenia, sarcopenic obesity, and functional impairments in older adults: National Health and Nutrition Examination Surveys 1999–2004. Nutr. Res. 2015, 35, 1031–1039. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.; Candow, D.; Krentz, J.; Roberts, M.; Young, K. Body fat changes following creatine supplementation and resistance training in adults > 50 years of age: A meta-analysis. J. Funct. Morphol. Kinesiol. 2019, 4, 62. [Google Scholar] [CrossRef]
- Lee, N.; Kim, I.; Park, S.; Han, D.; Ha, S.; Kwon, M.; Kim, J.; Byun, S.H.; Oh, W.; Jeon, H.B.; et al. Creatine inhibits adipogenesis by downregulating insulin-induced activation of the phosphatidylinositol 3-kinase signaling pathway. Stem Cells Dev. 2015, 24, 983–994. [Google Scholar] [CrossRef]
- Chouchani, E.T.; Kazak, L.; Spiegelman, B.M. New Advances in Adaptive Thermogenesis: UCP1 and Beyond. Cell. Metab. 2019, 29, 27–37. [Google Scholar] [CrossRef] [PubMed]
- Wakatsuki, T.; Hirata, F.; Ohno, H.; Yamamoto, M.; Sato, Y.; Ohira, Y. Thermogenic responses to high-energy phosphate contents and/or hindlimb suspension in rats. Jpn. J. Physiol. 1996, 46, 171–175. [Google Scholar] [CrossRef]
- Kazak, L.; Chouchani, E.T.; Lu, G.Z.; Jedrychows, P.; Bare, C.J.; Mina, A.I.; Kumari, M.; Zhang, S.; Vuckovic, I.; Laznik-Bogoslavski, D.; et al. Genetic Depletion of Adipocyte Creatine Metabolism Inhibits Diet-Induced Thermogenesis and Drives Obesity. Cell. Metab. 2017, 26, 693. [Google Scholar] [CrossRef]
- Perna, M.K.; Kokenge, A.N.; Miles, K.N.; Udobi, K.C.; Clark, J.F.; Pyne-Geithman, G.J.; Khuchua, Z.; Skelton, M.R. Creatine transporter deficiency leads to increased whole body and cellular metabolism. Amino Acids 2016, 48, 2057–2065. [Google Scholar] [CrossRef]
- Kazak, L.; Rahbani, J.F.; Samborska, B.; Lu, G.Z.; Jedrychowski, M.P.; Lajoie, M.; Zhang, S.; Ramsay, L.C.; Dou, F.Y.; Tenen, D.; et al. Ablation of adipocyte creatine transport impairs thermogenesis and causes diet-induced obesity. Nat. Metab. 2019, 1, 360–370. [Google Scholar] [CrossRef]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Cardiovascular Health Study Collaborative Research Group Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, 146. [Google Scholar] [CrossRef]
- Song, X.; Mitnitski, A.; Rockwood, K. Prevalence and 10-year outcomes of frailty in older adults in relation to deficit accumulation. J. Am. Geriatr. Soc. 2010, 58, 681–687. [Google Scholar] [CrossRef] [PubMed]
- Rochat, S.; Cumming, R.G.; Blyth, F.; Creasey, H.; Handelsman, D.; Le Couteur, D.G.; Naganathan, V.; Sambrook, P.N.; Seibel, M.J.; Waite, L. Frailty and use of health and community services by community-dwelling older men: The Concord Health and Ageing in Men Project. Age Ageing 2010, 39, 228–233. [Google Scholar] [CrossRef] [PubMed]
- Thompson, M.Q.; Theou, O.; Karnon, J.; Adams, R.J.; Visvanathan, R. Frailty prevalence in Australia: Findings from four pooled Australian cohort studies. Australas. J. Ageing 2018, 37, 155–158. [Google Scholar] [CrossRef] [PubMed]
- Martin, F.C.; Ranhoff, A.H. Frailty and Sarcopenia; Falaschi, P., Marsh, D., Eds.; Orthogeriatrics: The Management of Older Patients with Fragility Fractures; Springer: Cham, Switzerland, 2021; pp. 53–65. [Google Scholar]
- Oktaviana, J.; Zanker, J.; Vogrin, S.; Duque, G. The Effect of beta-hydroxy-beta-methylbutyrate (HMB) on Sarcopenia and Functional Frailty in Older Persons: A Systematic Review. J. Nutr. Health Aging 2019, 23, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Rockwood, K.; Song, X.; MacKnight, C.; Bergman, H.; Hogan, D.B.; McDowell, I.; Mitnitski, A. A global clinical measure of fitness and frailty in elderly people. CMAJ 2005, 173, 489–495. [Google Scholar] [CrossRef]
- Collins, J.; Longhurst, G.; Roschel, H.; Gualano, B. Resistance Training and Co-supplementation with Creatine and Protein in Older Subjects with Frailty. J. Frailty Aging 2016, 5, 126–134. [Google Scholar] [PubMed]
- Riesberg, L.A.; Weed, S.A.; McDonald, T.L.; Eckerson, J.M.; Drescher, K.M. Beyond muscles: The untapped potential of creatine. Int. Immunopharmacol. 2016, 37, 31–42. [Google Scholar] [CrossRef]
- Dalle, S.; Rossmeislova, L.; Koppo, K. The Role of Inflammation in Age-Related Sarcopenia. Front. Physiol. 2017, 8, 1045. [Google Scholar] [CrossRef]
- Van Epps, P.; Oswald, D.; Higgins, P.A.; Hornick, T.R.; Aung, H.; Banks, R.E.; Wilson, B.M.; Burant, C.; Graventstein, S.; Canaday, D.H. Frailty has a stronger association with inflammation than age in older veterans. Immun. Ageing 2016, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Hassan, E.B.; Imani, M.; Duque, G. Is Physical Frailty a Neuromuscular Condition? J. Am. Med. Dir. Assoc. 2019, 20, 1556–1557. [Google Scholar] [CrossRef] [PubMed]
- Uchmanowicz, I. Oxidative Stress, Frailty and Cardiovascular Diseases: Current Evidence. Adv. Exp. Med. Biol. 2020, 1216, 65–77. [Google Scholar] [PubMed]
- Webster, J.M.; Kempen, L.J.A.P.; Hardy, R.S.; Langen, R.C.J. Inflammation and Skeletal Muscle Wasting During Cachexia. Front. Physiol. 2020, 11, 597675. [Google Scholar] [CrossRef] [PubMed]
- Wyart, E.; Bindels, L.B.; Mina, E.; Menga, A.; Stanga, S.; Porporato, P.E. Cachexia, a Systemic Disease beyond Muscle Atrophy. Int. J. Mol. Sci. 2020, 21, 8592. [Google Scholar] [CrossRef]
- Fairman, C.M.; Kendall, K.L.; Hart, N.H.; Taaffe, D.R.; Galvao, D.A.; Newton, R.U. The potential therapeutic effects of creatine supplementation on body composition and muscle function in cancer. Crit. Rev. Oncol. Hematol. 2019, 133, 46–57. [Google Scholar] [CrossRef]
- Jatoi, A.; Steen, P.D.; Atherton, P.J.; Moore, D.F.; Rowland, K.M.; Le-Lindqwister, N.A.; Adonizio, C.S.; Jaslowski, A.J.; Sloan, J.; Loprinzi, C. A double-blind, placebo-controlled randomized trial of creatine for the cancer anorexia/weight loss syndrome (N02C4): An Alliance trial. Ann. Oncol. 2017, 28, 1957–1963. [Google Scholar] [CrossRef]
- Bourgeois, J.M.; Nagel, K.; Pearce, E.; Wright, M.; Barr, R.D.; Tarnopolsky, M.A. Creatine monohydrate attenuates body fat accumulation in children with acute lymphoblastic leukemia during maintenance chemotherapy. Pediatr. Blood Cancer. 2008, 51, 183–187. [Google Scholar] [CrossRef]
- Norman, K.; Stubler, D.; Baier, P.; Schutz, T.; Ocran, K.; Holm, E.; Lochs, H.; Pirlich, M. Effects of creatine supplementation on nutritional status, muscle function and quality of life in patients with colorectal cancer—A double blind randomised controlled trial. Clin. Nutr. 2006, 25, 596–605. [Google Scholar] [CrossRef]
- Lonbro, S.; Dalgas, U.; Primdahl, H.; Overgaard, J.; Overgaard, K. Feasibility and efficacy of progressive resistance training and dietary supplements in radiotherapy treated head and neck cancer patients—The DAHANCA 25A study. Acta Oncol. 2013, 52, 310–318. [Google Scholar] [CrossRef]
| First Author, Year | Population | Supplement Dose | Resistance Training | Duration | Outcomes |
|---|---|---|---|---|---|
| Aguiar et al. 2013 [18] | N = 18; healthy women; Mean age = 65 y | CR (5 g/day), PLA | RT = 3 x/wk | 12 wks | CR ↑ gains in fat-free mass (+3.2%), muscle mass (+2.8%), 1 RM bench press, knee extension, and biceps curl compared to PLA |
| Alves et al. 2013 [19] | N = 47; healthy women, Mean age = 66.8 y (range: 60–80 y) | CR (20 g/day for 5 days, followed by 5 g/day thereafter), PLA with and without RT | RT = 2 x/wk | 24 wks | ↔1 RM strength compared to RT + PLA |
| Bemben et al. 2010 and Eliot et al. 2008 [20,21] | N = 42; healthy men; age = 48–72 y | CR (5 g/day), PRO (35 g/day), CR + PRO, PLA | RT = 3 x/wk | 14 wks | ↔lean tissue mass, 1 RM strength |
| Bermon et al. 1998 [22] | N = 32 (16 men, 16 women); healthy; age = 67–80 y | CR (20 g/day for 5 days followed by 3 g/day), PLA | RT = 3 x/wk | 7.4 wks (52 days) | ↔lower limb muscular volume, 1- and 12-repetitions maxima, and isometric intermittent endurance |
| Bernat et al. 2019 [23] | N = 24 healthy men; age = 59 ± 6 y | CR (0.1 g/kg/day), PLA | High-velocity RT = 2 x/wk | 8 wks | ↔muscle thickness, physical performance, upper-body muscle strength; CR ↑ leg press strength, total lower body strength |
| Brose et al. 2003 [24] | N = 28 (15 men, 13 women); healthy; age: men = 68.7, women = 70.8 y | CR (5 g/day), PLA | RT = 3 x/wk | 14 wks | CR ↑ gains in lean tissue mass and isometric knee extension strength; ↔ type 1, 2 a, 2 x muscle fibre area |
| Candow et al. 2008 [25] | N = 35; healthy men; age = 59–77 y | CR (0.1 g/kg/day), CR + PRO (PRO: 0.3 g/kg/day), PLA | RT = 3 x/wk | 10 wks | CR ↑ muscle thickness compared to PLA. CR ↑1 RM bench press ↔ 1 RM leg press |
| Candow et al. 2015 [26] | N = 39 (17 men, 22 women); healthy; age = 50–71 y | CR (0.1 g/kg) before RT, CR (0.1 g/kg) after RT, PLA | RT = 3 x/wk | 32 wks | CR after RT ↑ lean tissue mass, 1 RM leg press, 1 RM chest press compared to PLA |
| Candow et al. 2020 [27] | N = 38; healthy men; age = 49–67 y | CR (On training days: 0.05 g/kg before and 0.05 g/kg after exercise) + 0.1 g/kg/day on non-train-ing days (2 equal doses) or PLA | RT = 3 x/wk | 12 months | ↔lean tissue mass, muscle thickness, or muscle strength |
| Chilibeck et al. 2015 [28] | N = 33; healthy women; Mean age = 57 y | CR (0.1 g/kg/day), PLA | RT = 3 x/wk | 52 wks | ↔lean tissue mass and muscle thickness gains between groups; ↑ relative bench press strength compared to PLA. |
| Chrusch et al. 2001 [29] | N = 30; healthy men; age = 60–84 y | CR (0.3 g/kg/d for 5 days followed by 0.07 g/kg/day), PLA | RT = 3 x/wk | 12 wks | CR ↑ gains in lean tissue mass; CR ↑1 RM leg press, 1 RM knee extension, leg press endurance, and knee extension endurance; ↔ 1 RM bench press or bench press endurance. |
| Cooke et al. 2014 [30] | N = 20; healthy men; age = 55–70 y | CR (20 g/day for 7 days followed by 0.1 g/kg/day on training days) | RT = 3 x/wk | 12 wks | ↔lean tissue mass, 1 RM bench press, 1 RM leg press |
| Deacon et al. 2008 [31] | N = 80 (50 men, 30 women); COPD; age = 68.2 y | CR (22 g/day for 5 day followed by 3.76 g/day), PLA | RT = 3 x/wk | 7 wks | ↔lean tissue mass or muscle strength |
| Eijnde et al. 2003 [32] | N = 46; healthy men; age = 55–75 y | CR (5 g/day), PLA | Cardiorespiratory + RT = 2–3 x/wk | 26 wks | ↔lean tissue mass or isometric maximal strength |
| Gualano et al. 2011 [33] | N = 25 (9 men, 16 women); type 2 diabetes; age = 57 y | CR (5 g/day), PLA | RT = 3 x/wk | 12 wks | ↔lean tissue mass |
| Gualano et al. 2014 [34] | N = 30; "vulnera-ble" women; Mean age = 65.4 y | CR (20 g/day for 5 days; 5 g/day thereafter), PLA with and without RT | RT = 2 x/wk | 24 wks | CR + RT ↑ gains in 1RM bench press and appendicular lean mass compared to PLA + RT |
| Johannsmeyer et al. 2016 [35] | N = 31 (17 men, 14 women); healthy; age = 58 y | CR (0.1 g/kg/day), PLA | RT = 3 x/wk | 12 wks | CR ↑ gains in lean tissue mass; ↔ 1RM strength and endurance; CR attenuated magnitude increase in time to complete balance test compared to PLA |
| Neves et al. 2011 [36] | N = 24 (postmen-opausal women with Knee osteo-arthritis); Age = 55–65 y | CR (20 g/day for 1 week, followed by 5 g/day), PLA | RT = 3 x/wk | 12 wks | CR ↑ gains in limb lean mass. ↔ 1RM leg press |
| Pinto et al. 2016 [37] | N = 27 (men and women); healthy; age = 60–80 y | CR (5 g/day), PLA | RT = 3 x/wk | 12 wks | CR ↑ gains in lean tissue mass; ↔ 10 RM bench press or leg press strength |
| Smolarek et al. 2020 [38] | N = 26 (5 men, 21 women); long-term care residence; age = 68.9 ± 6.8 y | CR (5 g/day), PLA | RT = 2 x/wk | 16 wks | CR ↑ dominant and non-dominant handgrip strength |
| First Author, Year | Study Population | Intervention | Duration | Outcomes |
|---|---|---|---|---|
| Brose et al. 2003 [24] | N = 28; healthy (15 men, 13 women); age ≥ 65 y (men = 68.7 y, women = 70.8 y) | RCT; CR + RT, PLA + RT. CR = 5 g/day; RT = 3 x/wk | 14 wks | ↔on osteocalcin |
| Candow et al. 2008 [25] | N = 35; older men (age: 59–77 y) | RCT; CR + PRO + RT; CR + RT, PLA + RT; CR = 0.1 g/kg/day; RT = 3 x/wk | 10 wks | CR ↓ NTx |
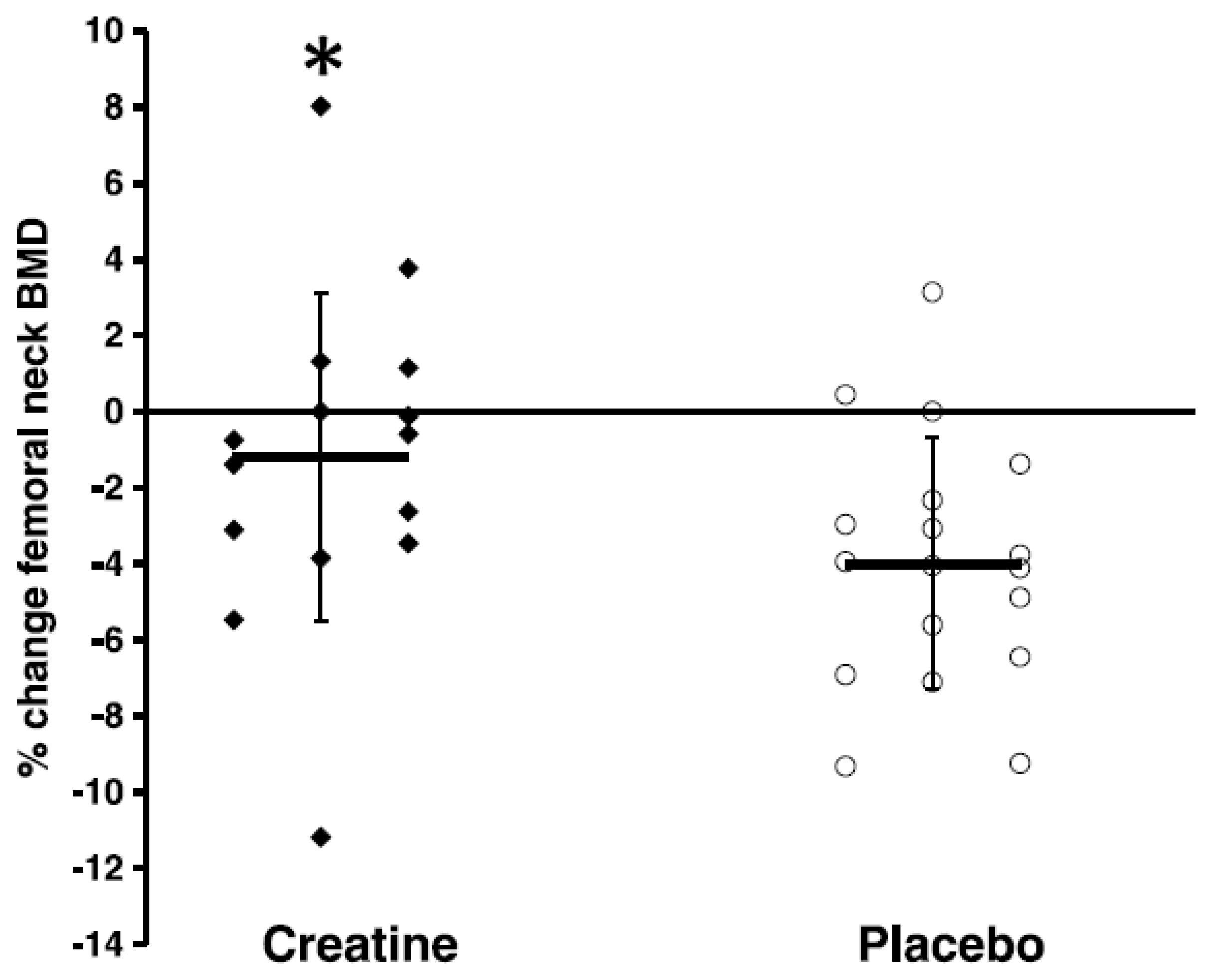
| Candow et al. 2019 [5] | N = 39; healthy (17 men; 22 women); age ≥ 50 y (mean ~55 y) | RCT; CR-Before + RT, CR-After + RT, PLA + RT; CR = 0.1 g/kg/day; RT = 3 x/wk | 8 mths | ↔BMD and BMC of the whole-body, limbs, femoral neck, lumbar spine, and total hip |
| Candow et al. 2020 [27] | N = 38; healthy men; age = 49–67 y | RCT; CR + RT, PLA + RT; CR = 0.1 g/kg/day; RT = 3 x/wk | 12 mths | ↔ BMD and geometry, bone speed of sound; CR ↑ (p = 0.06) section modulus of the narrow part of the femoral neck |
| Chilibeck et al. 2005 [45] | N = 29; older men (71 y). | RCT; CR + RT, PLA + RT; CR = 0.3 g/kg/day for 5 days followed by 0.07 g/kg/day for the remaining; RT = 3 x/wk | 12 wks | ↑ arm BMC greater in the CR group com-pared to PLA; ↔ between groups for whole-body and leg BMD |
| Chilibeck et al. 2015 [28] | N = 33; postmenopausal women; age: 57 ± 6 y | RCT; PLA + RT, CR + RT; CR = 0.1 g/kg/day (0.05 g/kg provided immediately before and 0.05 g/kg after training on training days and with two meals on non-training days); RT = 3 x/wk | 12 mths | CR attenuated rate of femoral neck BMD loss compared to PLA and CR ↑ femoral shaft subperiosteal width; ↔ between groups on all other outcome measures |
| Gualano et al. 2014 [34] | N = 60; older vulnerable women (age: 66 y) | RCT; PLA, CR, PLA + RT, CR + RT; CR = 20 g/day for 5 days followed by 5 g/day for the remaining; RT = 2 x/wk | 24 wks | ↔bone mineral and serum bone markers between groups |
| Pinto et al. 2016 [37] | N = 32; healthy, non-athletic men and women between 60–80 y | RCT; PLA + RT, CR + RT; CR = 5 g/day; RT = 3 x/wk. Muscle groups (i.e., upper and lower body) alternated between training days, 1.5 x/wk per muscle group | 12 wks | ↔BMD and BMC of all assessed sites between groups |
| Authors (Year) | Patients | Treatment Modality | Dosage | Protocol Du-RATION | Compliance | Exercise Program | Results | Adverse Effects Related to Supplementation |
|---|---|---|---|---|---|---|---|---|
| Jatoi et al. (2017) [98] | 263 cancer patients (65 ± 11 yrs.) with weight loss syndrome | 210 undergoing concurrent chemotherapy | 20 g/day for 5 days then 2 g/day | 39 weeks | nr | n/a | ↔body weight, appetite, QoL, frailty, grip strength | None reported |
| Bourgeois et al. (2008) [99] | 9 children (7.6 ± 3.8 yrs.) with ALL under-going chemotherapy | Maintenance phase of treatment on the Dann-Farber Cancer Institute protocol 2000–2001 | 0.1 g/kg/day | 2 × 16 weeks separated by 6-week wash-out period. | nr | n/a | ↓ BF% Cr, ↑ BF% NH, ↔BMD | None reported |
| Norman et al. (2006) [100] | 31 stage III/IV colorectal cancer patients (65.10 ± 12.55 yrs.) undergoing chemotherapy | n = 11: fluorouracil/folic acid (5-FU FA); n = 9: fluorouracil/folic acid + oxaliplatin (5-FU FA + O); n = 11: fluorouracil/folic acid + irinotecan (5-FU FA + I) | 20 g/day for 7 days then 5 g/day | 8 weeks | Cr: 84.55 ± 7.77%; PLA: 87.62 ± 5.90% | n/a | ↔ weight, capacitance, KE, HR, BCM, BF; ↑ HG 5-FU FA: ↑ phase angle, ECM/BCM ratio | None reported |
| Lonbro et al. (2013) [101] | 30 Head and neck patients treated with radiotherapy | Radiotherapy according to DA-HANCA guidelines (www.da-hanca.dk) + chemotherapy (n = 20: cisplatin, 40 mg/m2). N = 4 received Zalutumumab | 5 g/day + 30 g Pro/day | 12 weeks | 69% ingested all supplementation; 19% missed ≤ 3 supplementations; 12% terminated 4 weeks early | 3 days/wk., 3 × 10 total body | ↑LBM ProCr group, ns↑PLA ↔ muscle strength **, ↔ Physical function ** | No major adverse events reported; 2 participants stopped supplementation 4 weeks early due to muscle cramping and mucus production |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Candow, D.G.; Forbes, S.C.; Kirk, B.; Duque, G. Current Evidence and Possible Future Applications of Creatine Supplementation for Older Adults. Nutrients 2021, 13, 745. https://doi.org/10.3390/nu13030745
Candow DG, Forbes SC, Kirk B, Duque G. Current Evidence and Possible Future Applications of Creatine Supplementation for Older Adults. Nutrients. 2021; 13(3):745. https://doi.org/10.3390/nu13030745
Chicago/Turabian StyleCandow, Darren G., Scott C. Forbes, Ben Kirk, and Gustavo Duque. 2021. "Current Evidence and Possible Future Applications of Creatine Supplementation for Older Adults" Nutrients 13, no. 3: 745. https://doi.org/10.3390/nu13030745
APA StyleCandow, D. G., Forbes, S. C., Kirk, B., & Duque, G. (2021). Current Evidence and Possible Future Applications of Creatine Supplementation for Older Adults. Nutrients, 13(3), 745. https://doi.org/10.3390/nu13030745









