A Systematic Review and Meta-Analysis Finds Increased Blood Levels of All Forms of Ghrelin in Both Restricting and Binge-Eating/Purging Subtypes of Anorexia Nervosa
Abstract
1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Search Strategy
2.3. Study Selection
2.4. Data Extraction and Synthesis
2.5. Risk of Bias in Individual Studies
2.6. Statistical Analysis
3. Results
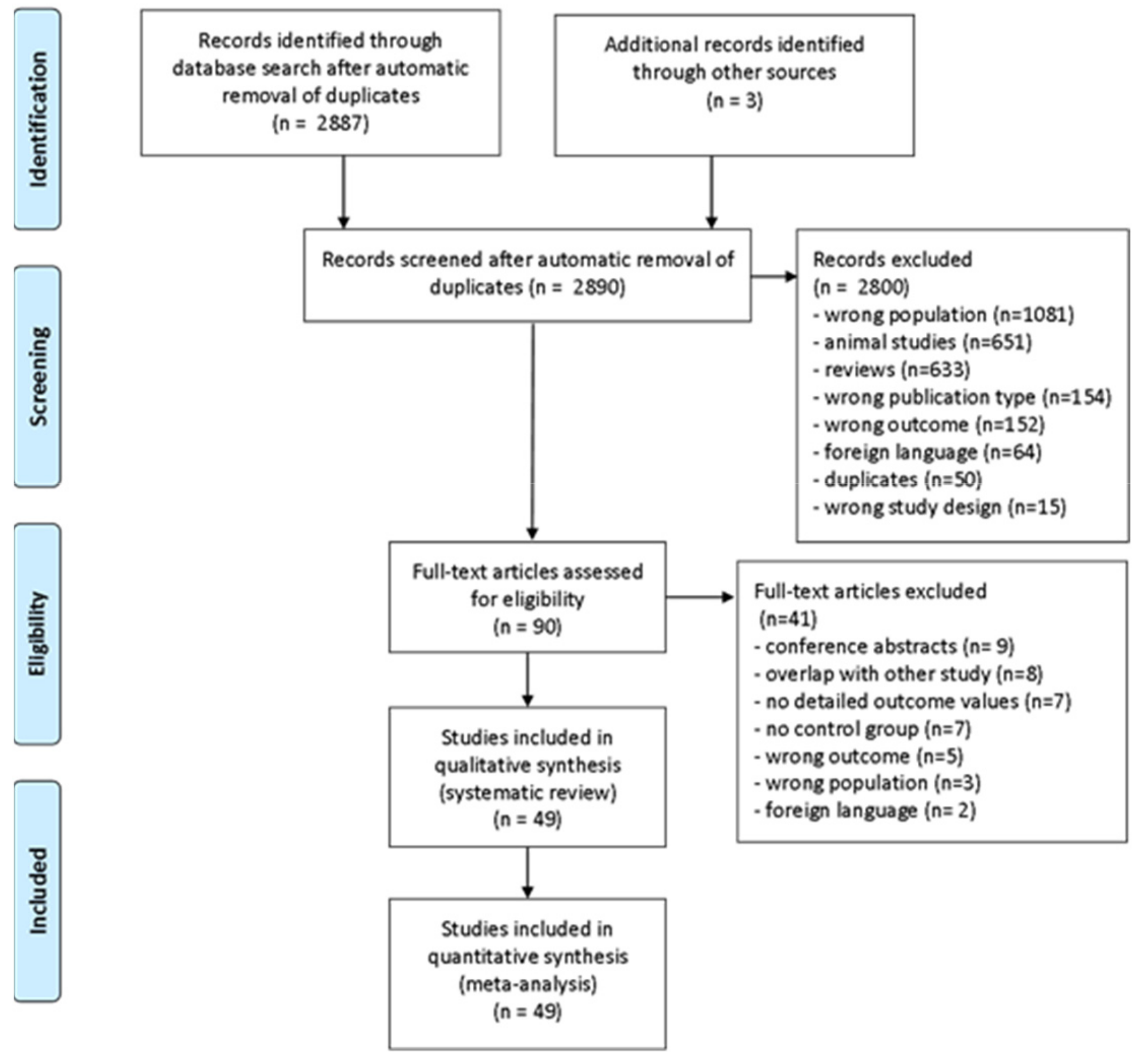
3.1. Study Search and Selection
3.2. Study Characteristics
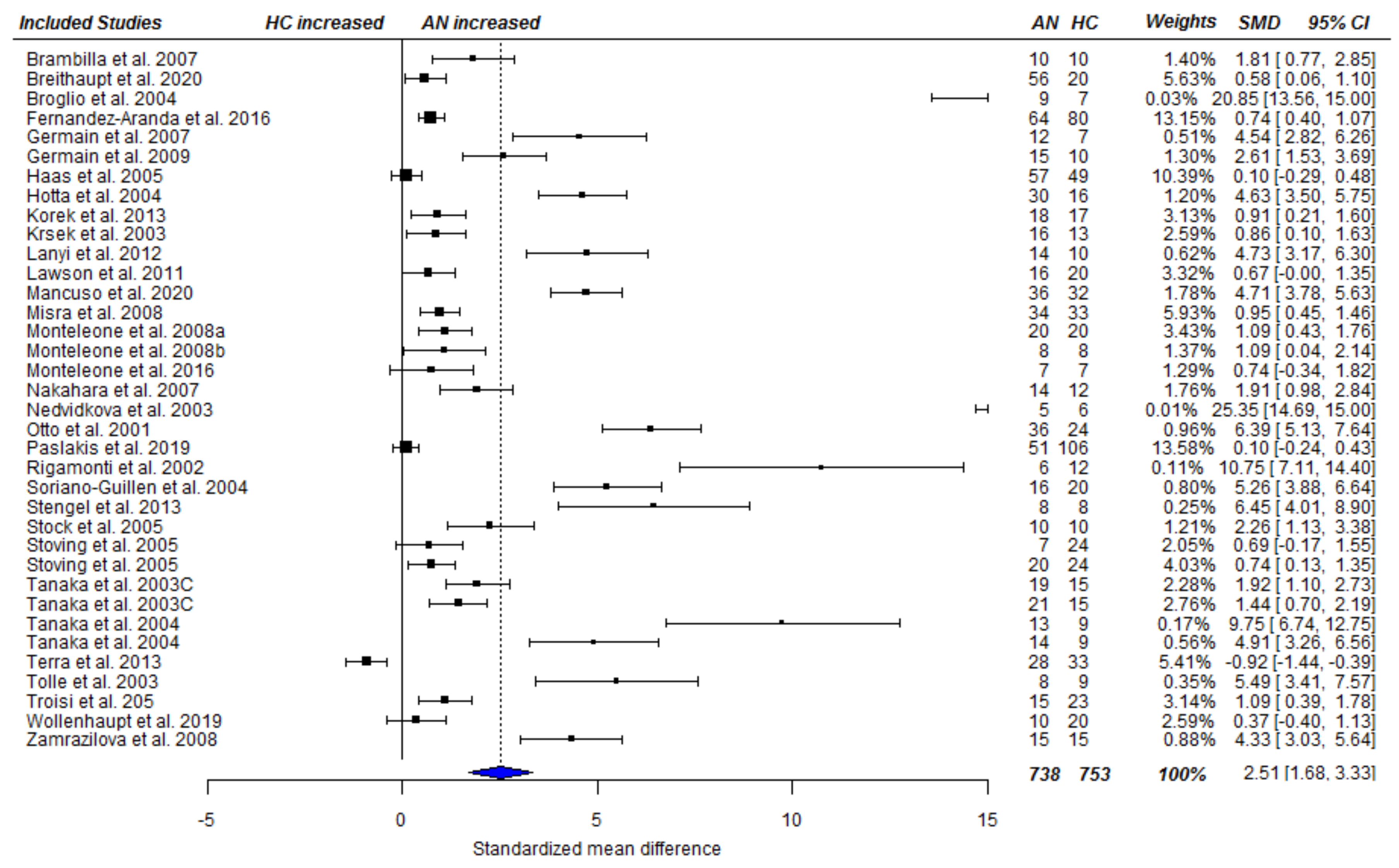
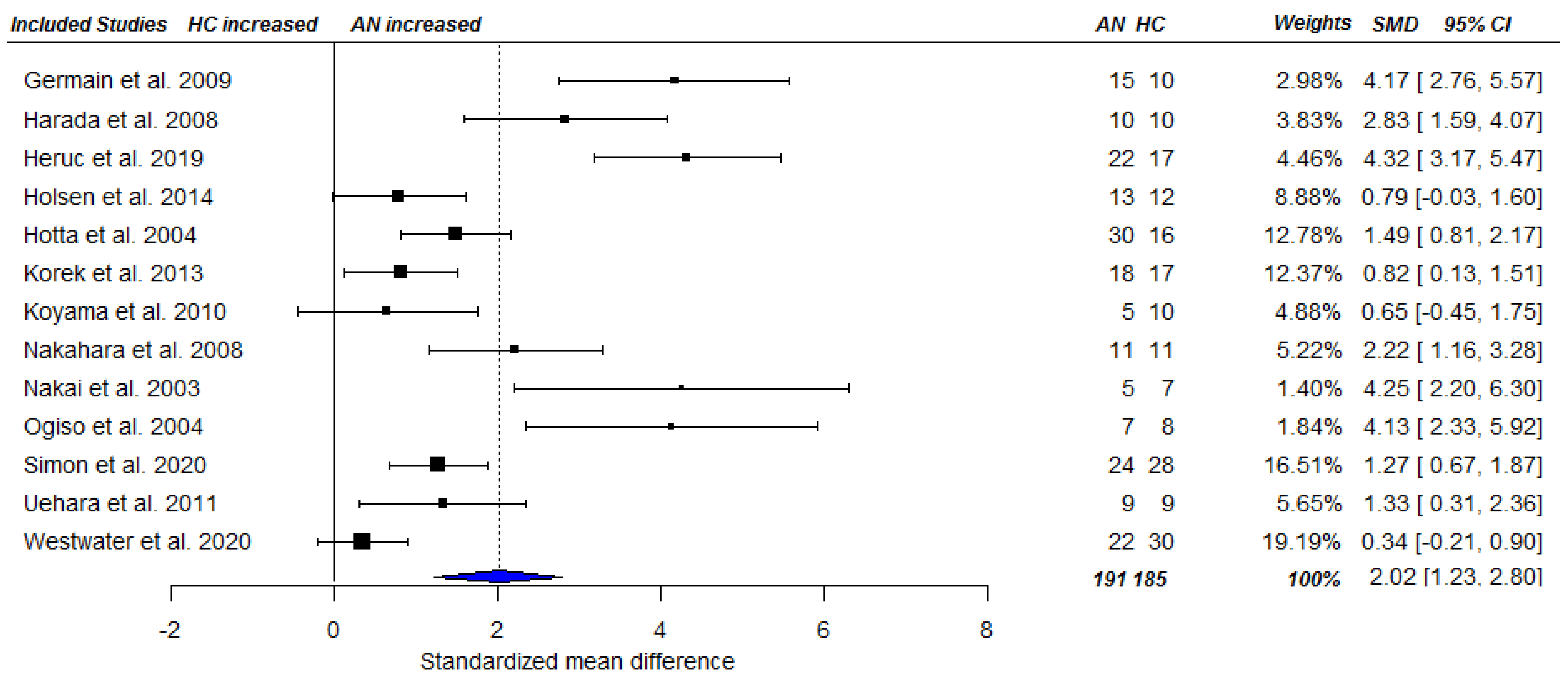
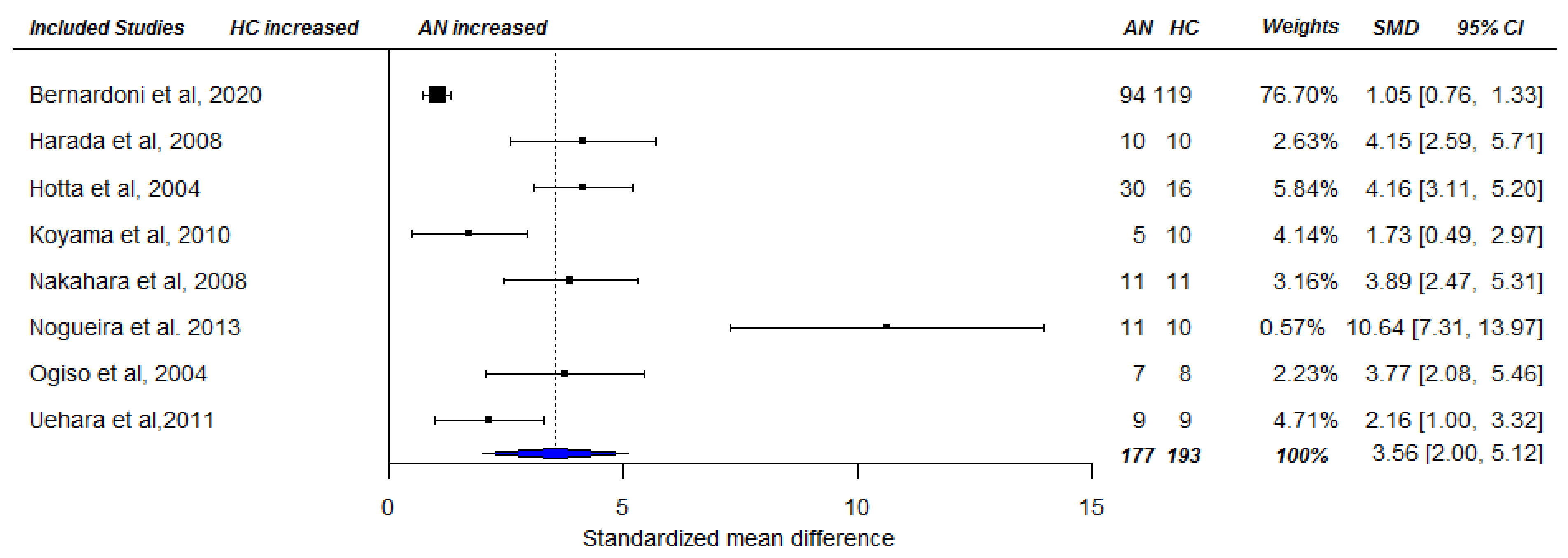
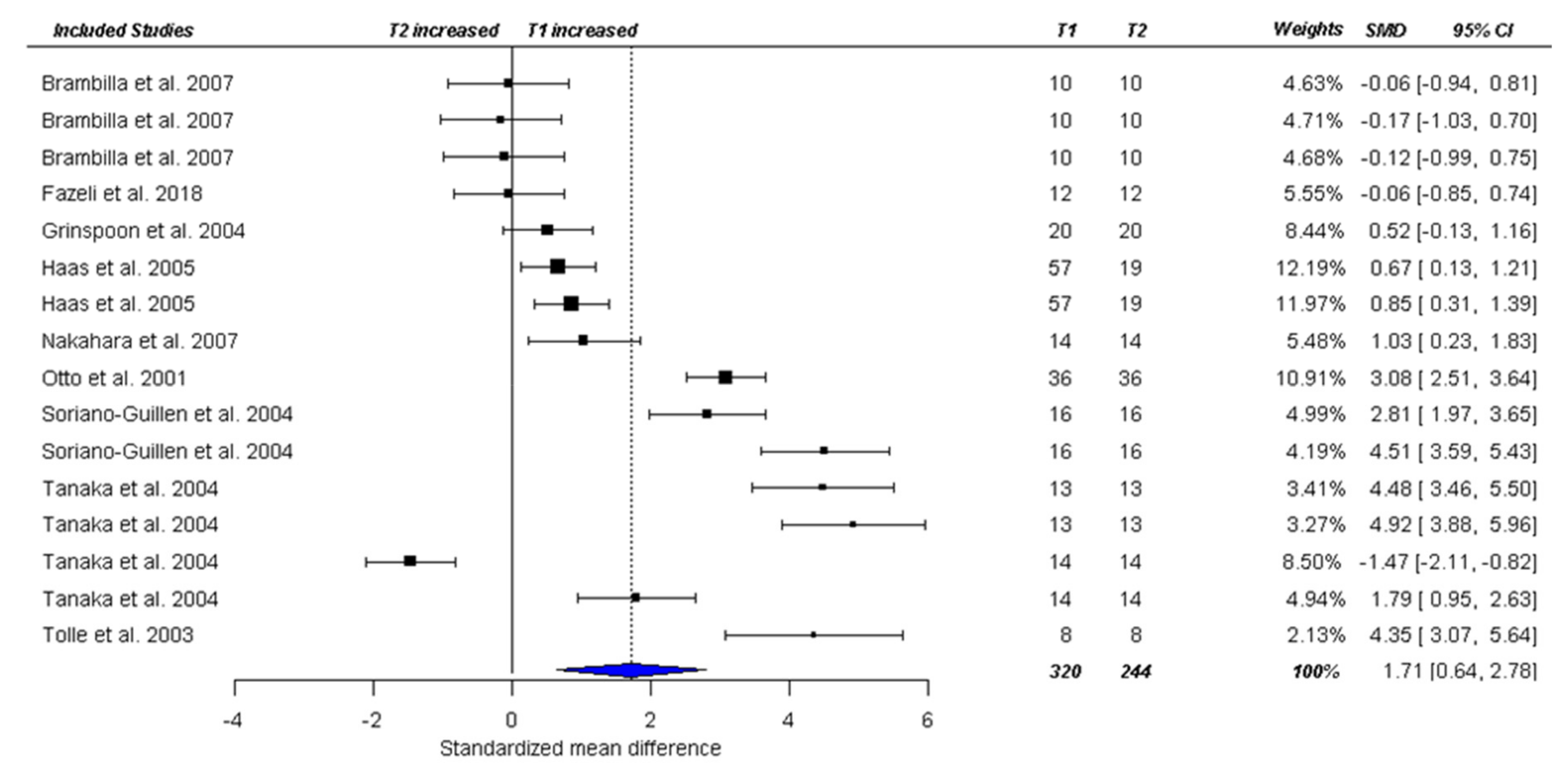
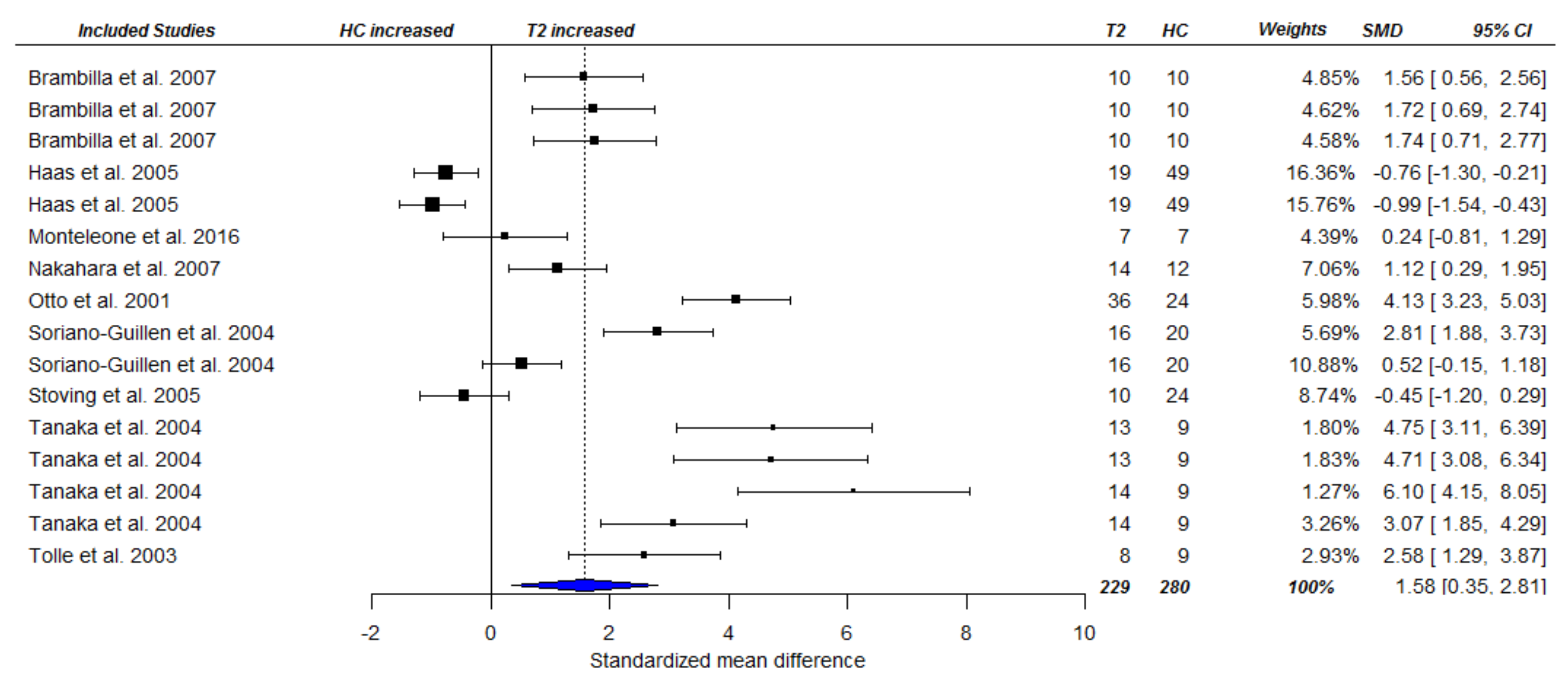
3.3. Meta-Analysis
3.3.1. Fasting Total Ghrelin Levels in Acute AN
3.3.2. Fasting Ghrelin Levels in AN in Longitudinal Studies
4. Discussion
4.1. Ghrelin at the Acute State
4.2. Ghrelin at Follow-Up
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 2013; ISBN 978-0-89042-555-8. [Google Scholar]
- Levine, R.L. Endocrine Aspects of Eating Disorders in Adolescents. Adolesc. Med. 2002, 13, 129–143. [Google Scholar]
- Singhal, V.; Misra, M.; Klibanski, A. Endocrinology of Anorexia Nervosa in Young People: Recent Insights. Curr. Opin. Endocrinol. Diabetes Obes. 2014, 21, 64–70. [Google Scholar] [CrossRef]
- Brown, C.; Mehler, P.S. Medical Complications of Anorexia Nervosa and Their Treatments: An Update on Some Critical Aspects. Eat. Weight Disord. 2015, 20, 419–425. [Google Scholar] [CrossRef] [PubMed]
- Kojima, M.; Hosoda, H.; Date, Y.; Nakazato, M.; Matsuo, H.; Kangawa, K. Ghrelin Is a Growth-Hormone-Releasing Acylated Peptide from Stomach. Nature 1999, 402, 656–660. [Google Scholar] [CrossRef] [PubMed]
- Warzecha, Z.; Dembiński, A.; Ceranowicz, P.; Dembiński, M.; Cieszkowski, J.; Bielański, W.; Pawlik, W.W.; Kuwahara, A.; Kato, I. Dual Age-Dependent Effect of Ghrelin Administration on Serum Level of Insulin-like Growth Factor-1 and Gastric Growth in Young Rats. Eur. J. Pharmacol. 2006, 529, 145–150. [Google Scholar] [CrossRef] [PubMed]
- Tschöp, M.; Smiley, D.L.; Heiman, M.L. Ghrelin Induces Adiposity in Rodents. Nature 2000, 407, 908–913. [Google Scholar] [CrossRef]
- Druce, M.R.; Wren, A.M.; Park, A.J.; Milton, J.E.; Patterson, M.; Frost, G.; Ghatei, M.A.; Small, C.; Bloom, S.R. Ghrelin Increases Food Intake in Obese as Well as Lean Subjects. Int. J. Obes. 2005, 29, 1130–1136. [Google Scholar] [CrossRef]
- Broglio, F.; Gianotti, L.; Destefanis, S.; Fassino, S.; Abbate Daga, G.; Mondelli, V.; Lanfranco, F.; Gottero, C.; Gauna, C.; Hofland, L.; et al. The Endocrine Response to Acute Ghrelin Administration Is Blunted in Patients with Anorexia Nervosa, a Ghrelin Hypersecretory State. Clin. Endocrinol. 2004, 60, 592–599. [Google Scholar] [CrossRef]
- Tack, J.; Depoortere, I.; Bisschops, R.; Delporte, C.; Coulie, B.; Meulemans, A.; Janssens, J.; Peeters, T. Influence of Ghrelin on Interdigestive Gastrointestinal Motility in Humans. Gut 2006, 55, 327–333. [Google Scholar] [CrossRef]
- Fernandez-Aranda, F.; Aguera, Z.; Fernandez-Garcia, J.C.; Garrido-Sanchez, L.; Alcaide-Torres, J.; Tinahones, F.J.; Giner-Bartolome, C.; Banos, R.M.; Botella, C.; Cebolla, A.; et al. Smell-Taste Dysfunctions in Extreme Weight/Eating Conditions: Analysis of Hormonal and Psychological Interactions. Endocrine 2016, 51, 256–267. [Google Scholar] [CrossRef]
- Inhoff, T.; Mönnikes, H.; Noetzel, S.; Stengel, A.; Goebel, M.; Dinh, Q.T.; Riedl, A.; Bannert, N.; Wisser, A.-S.; Wiedenmann, B. Desacyl Ghrelin Inhibits the Orexigenic Effect of Peripherally Injected Ghrelin in Rats. Peptides 2008, 29, 2159–2168. [Google Scholar] [CrossRef] [PubMed]
- Gauna, C.; Van de Zande, B.; Van Kerkwijk, A.; Themmen, A.P.; van der Lely, A.-J.; Delhanty, P.J. Unacylated Ghrelin Is Not a Functional Antagonist but a Full Agonist of the Type 1a Growth Hormone Secretagogue Receptor (GHS-R). Mol. Cell. Endocrinol. 2007, 274, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Heppner, K.M.; Piechowski, C.L.; Müller, A.; Ottaway, N.; Sisley, S.; Smiley, D.L.; Habegger, K.M.; Pfluger, P.T.; DiMarchi, R.; Biebermann, H. Both Acyl and Des-Acyl Ghrelin Regulate Adiposity and Glucose Metabolism via Central Nervous System Ghrelin Receptors. Diabetes 2014, 63, 122–131. [Google Scholar] [CrossRef]
- Wittekind, D.A.; Kluge, M. Ghrelin in Psychiatric Disorders—A Review. Psychoneuroendocrinology 2015, 52, 176–194. [Google Scholar] [CrossRef] [PubMed]
- Tolle, V.; Kadem, M.; Bluet-Pajot, M.T.; Frere, D.; Foulon, C.; Bossu, C.; Dardennes, R.; Mounier, C.; Zizzari, P.; Lang, F.; et al. Balance in Ghrelin and Leptin Plasma Levels in Anorexia Nervosa Patients and Constitutionally Thin Women. J. Clin. Endocrinol. Metab. 2003, 88, 109–116. [Google Scholar] [CrossRef]
- Hofmann, T.; Elbelt, U.; Haas, V.; Ahnis, A.; Klapp, B.F.; Rose, M.; Stengel, A. Plasma Kisspeptin and Ghrelin Levels Are Independently Correlated with Physical Activity in Patients with Anorexia Nervosa. Appetite 2017, 108, 141–150. [Google Scholar] [CrossRef]
- Mequinion, M.; Caron, E.; Zgheib, S.; Stievenard, A.; Zizzari, P.; Tolle, V.; Cortet, B.; Lucas, S.; Prevot, V.; Chauveau, C.; et al. Physical Activity: Benefit or Weakness in Metabolic Adaptations in a Mouse Model of Chronic Food Restriction? Am. J. Physiol. Endocrinol. Metab. 2015, 308, E241–E255. [Google Scholar] [CrossRef]
- Verhagen, L.A.; Egecioglu, E.; Luijendijk, M.C.; Hillebrand, J.J.; Adan, R.A.; Dickson, S.L. Acute and Chronic Suppression of the Central Ghrelin Signaling System Reveals a Role in Food Anticipatory Activity. Eur. Neuropsychopharmacol. 2011, 21, 384–392. [Google Scholar] [CrossRef]
- Tanaka, M.; Naruo, T.; Yasuhara, D.; Tatebe, Y.; Nagai, N.; Shiiya, T.; Masamitsu, N.; Shigeru, M.; Nozoe, S. Fasting Plasma Ghrelin Levels in Subtypes of Anorexia Nervosa. Psychoneuroendocrinology 2003, 28, 829–835. [Google Scholar] [CrossRef]
- Tanaka, M.; Naruo, T.; Nagai, N.; Kuroki, N.; Shiiya, T.; Nakazato, M.; Matsukura, S.; Nozoe, S. Habitual Binge/Purge Behavior Influences Circulating Ghrelin Levels in Eating Disorders. J. Psychiatr. Res. 2003, 37, 17–22. [Google Scholar] [CrossRef]
- Monteleone, P.; Serritella, C.; Martiadis, V.; Scognamiglio, P.; Maj, M. Plasma Obestatin, Ghrelin, and Ghrelin/Obestatin Ratio Are Increased in Underweight Patients with Anorexia Nervosa but Not in Symptomatic Patients with Bulimia Nervosa. J. Clin. Endocrinol. Metab. 2008, 93, 4418–4421. [Google Scholar] [CrossRef]
- Nakai, Y.; Hosoda, H.; Nin, K.; Ooya, C.; Hayashi, H.; Akamizu, T.; Kangawa, K. Plasma Levels of Active Form of Ghrelin during Oral Glucose Tolerance Test in Patients with Anorexia Nervosa. Eur. J. Endocrinol. 2003, 149, R1–R3. [Google Scholar] [CrossRef] [PubMed]
- Nakai, Y.; Hosoda, H.; Nin, K.; Ooya, C.; Hayashi, H.; Akamizu, T.; Kangawa, K. Short-Term Secretory Regulation of the Active Form of Ghrelin and Total Ghrelin during an Oral Glucose Tolerance Test in Patients with Anorexia Nervosa. Eur. J. Endocrinol. 2004, 150, 913–914. [Google Scholar] [CrossRef] [PubMed]
- Schalla, M.A.; Stengel, A. The Role of Ghrelin in Anorexia Nervosa. Int. J. Mol. Sci. 2018, 19, 2117. [Google Scholar] [CrossRef]
- Otto, B.; Cuntz, U.; Fruehauf, E.; Wawarta, R.; Folwaczny, C.; Riepl, R.L.; Heiman, M.L.; Lehnert, P.; Fichter, M.; Tschop, M. Weight Gain Decreases Elevated Plasma Ghrelin Concentrations of Patients with Anorexia Nervosa. Eur. J. Endocrinol. 2001, 145, R5–R9. [Google Scholar] [CrossRef]
- Nakahara, T.; Kojima, S.; Tanaka, M.; Yasuhara, D.; Harada, T.; Sagiyama, K.; Muranaga, T.; Nagai, N.; Nakazato, M.; Nozoe, S.; et al. Incomplete Restoration of the Secretion of Ghrelin and PYY Compared to Insulin after Food Ingestion Following Weight Gain in Anorexia Nervosa. J. Psychiatr. Res. 2007, 41, 814–820. [Google Scholar] [CrossRef]
- Brambilla, F.; Monteleone, P.; Maj, M. Olanzapine-Induced Weight Gain in Anorexia Nervosa: Involvement of Leptin and Ghrelin Secretion? Psychoneuroendocrinology 2007, 32, 402–406. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Nakahara, T.; Kojima, S.; Nakano, T.; Muranaga, T.; Nagai, N.; Ueno, H.; Nakazato, M.; Nozoe, S.; Naruo, T. Effect of Nutritional Rehabilitation on Circulating Ghrelin and Growth Hormone Levels in Patients with Anorexia Nervosa. Regul. Pept. 2004, 122, 163–168. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G. PRISMA Group the PRISMA Group Preferred Reporting Items for Systematic Reviews and Meta-Analyses. Prism. Statement BMJ 2009, 339, b2535. [Google Scholar]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—a Web and Mobile App for Systematic Reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; Text Revision; American Psychiatric Association: Washington, DC, USA, 2000; ISBN 978-0-89042-062-1. [Google Scholar]
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th ed.; American Psychiatric Association: Washington, DC, USA, 1994. [Google Scholar]
- World Health Organization. The ICD-10 Classification of Mental and Behavioural Disorders: Clinical Descriptions and Diagnostic Guidelines; World Health Organization: Geneva, Switzerland, 1992; ISBN 978-7-117-01957-6. [Google Scholar]
- Lundh, A.; Gøtzsche, P.C. Recommendations by Cochrane Review Groups for Assessment of the Risk of Bias in Studies. BMC Med. Res. Methodol. 2008, 8, 1–9. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’connell, D.; Peterson, J.; Welch, V.; Losos, M. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. 2009. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp (accessed on 21 February 2021).
- Hedges, L.V.; Tipton, E.; Johnson, M.C. Robust Variance Estimation in Meta-Regression with Dependent Effect Size Estimates. Res. Synth. Methods 2010, 1, 39–65. [Google Scholar] [CrossRef] [PubMed]
- Tanner-Smith, E.E.; Tipton, E.; Polanin, J.R. Handling Complex Meta-Analytic Data Structures Using Robust Variance Estimates: A Tutorial in R. J. Dev. Life-Course Criminol. 2016, 2, 85–112. [Google Scholar] [CrossRef]
- Tipton, E. Small Sample Adjustments for Robust Variance Estimation with Meta-Regression. Psychol. Methods 2015, 20, 375–393. [Google Scholar] [CrossRef]
- Fisher, Z.; Tipton, E. Robumeta: An R-Package for Robust Variance Estimation in Meta-Analysis. arXiv 2015, arXiv:1503.02220. [Google Scholar]
- Maria Monteleone, A.; Monteleone, P.; Dalle Grave, R.; Nigro, M.; El Ghoch, M.; Calugi, S.; Cimino, M.; Maj, M. Ghrelin Response to Hedonic Eating in Underweight and Short-Term Weight Restored Patients with Anorexia Nervosa. Psychiatry Res. 2016, 235, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Stengel, A.; Hofmann, T.; Goebel-Stengel, M.; Elbelt, U.; Kobelt, P.; Klapp, B.F. Circulating Levels of Irisin in Patients with Anorexia Nervosa and Different Stages of Obesity--Correlation with Body Mass Index. Peptides 2013, 39, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Korek, E.; Krauss, H.; Gibas-Dorna, M.; Kupsz, J.; Piatek, M.; Piatek, J. Fasting and Postprandial Levels of Ghrelin, Leptin and Insulin in Lean, Obese and Anorexic Subjects. Prz. Gastroenterol. 2013, 8, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Breithaupt, L.; Chunga-Iturry, N.; Lyall, A.E.; Cetin-Karayumak, S.; Becker, K.R.; Thomas, J.J.; Slattery, M.; Makris, N.; Plessow, F.; Pasternak, O. Developmental Stage-Dependent Relationships between Ghrelin Levels and Hippocampal White Matter Connections in Low-Weight Anorexia Nervosa and Atypical Anorexia Nervosa. Psychoneuroendocrinology 2020, 104722. [Google Scholar] [CrossRef]
- Germain, N.; Galusca, B.; Le Roux, C.W.; Bossu, C.; Ghatei, M.A.; Lang, F.; Bloom, S.R.; Estour, B. Constitutional Thinness and Lean Anorexia Nervosa Display Opposite Concentrations of Peptide YY, Glucagon-like Peptide 1, Ghrelin, and Leptin. Am. J. Clin. Nutr. 2007, 85, 967–971. [Google Scholar] [CrossRef]
- Germain, N.; Galusca, B.; Grouselle, D.; Frere, D.; Tolle, V.; Zizzari, P.; Lang, F.; Epelbaum, J.; Estour, B. Ghrelin/Obestatin Ratio in Two Populations with Low Bodyweight: Constitutional Thinness and Anorexia Nervosa. Psychoneuroendocrinology 2009, 34, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Haas, V.; Onur, S.; Paul, T.; Nutzinger, D.O.; Bosy-Westphal, A.; Hauer, M.; Brabant, G.; Klein, H.; Muller, M.J. Leptin and Body Weight Regulation in Patients with Anorexia Nervosa before and during Weight Recovery. Am. J. Clin. Nutr. 2005, 81, 889–896. [Google Scholar] [CrossRef] [PubMed]
- Hotta, M.; Ohwada, R.; Katakami, H.; Shibasaki, T.; Hizuka, N.; Takano, K. Plasma Levels of Intact and Degraded Ghrelin and Their Responses to Glucose Infusion in Anorexia Nervosa. J. Clin. Endocrinol. Metab. 2004, 89, 5707–5712. [Google Scholar] [CrossRef] [PubMed]
- Krsek, M.; Rosicka, M.; Papezova, H.; Krizova, J.; Kotrlikova, E.; Haluz’k, M.; Justova, V.; Lacinova, Z.; Jarkovska, Z. Plasma Ghrelin Levels and Malnutrition: A Comparison of Two Etiologies. Eat. Weight Disord. EWD 2003, 8, 207–211. [Google Scholar] [CrossRef]
- Lanyi, E.; Repasy, J.; Fenyvesi, I.; Stomfai, S.; Kovacs, E.; Gati, A.; Molnar, D. Ghrelin, Metabolic and Hormonal Parameters during OGTT in Patients with Obesity and Anorexia Nervosa. J. Pediatric Biochem. 2012, 2, 85–90. [Google Scholar] [CrossRef]
- Lawson, E.A.; Eddy, K.T.; Donoho, D.; Misra, M.; Miller, K.K.; Meenaghan, E.; Lydecker, J.; Herzog, D.; Klibanski, A. Appetite-Regulating Hormones Cortisol and Peptide YY Are Associated with Disordered Eating Psychopathology, Independent of Body Mass Index. Eur. J. Endocrinol. Suppl. 2011, 164, 253–261. [Google Scholar] [CrossRef]
- Mancuso, C.; Izquierdo, A.; Slattery, M.; Becker, K.R.; Plessow, F.; Thomas, J.J.; Eddy, K.T.; Lawson, E.A.; Misra, M. Changes in Appetite-Regulating Hormones Following Food Intake Are Associated with Changes in Reported Appetite and a Measure of Hedonic Eating in Girls and Young Women with Anorexia Nervosa. Psychoneuroendocrinology 2020, 113, 104556. [Google Scholar] [CrossRef]
- Misra, M.; Prabhakaran, R.; Miller, K.K.; Goldstein, M.A.; Mickley, D.; Clauss, L.; Lockhart, P.; Cord, J.; Herzog, D.B.; Katzman, D.K.; et al. Prognostic Indicators of Changes in Bone Density Measures in Adolescent Girls with Anorexia Nervosa-II. J. Clin. Endocrinol. Metab. 2008, 93, 1292–1297. [Google Scholar] [CrossRef]
- Nedvidkova, J.; Krykorkova, I.; Bartak, V.; Papezova, H.; Gold, P.W.; Alesci, S.; Pacak, K. Loss of Meal-Induced Decrease in Plasma Ghrelin Levels in Patients with Anorexia Nervosa. J. Clin. Endocrinol. Metab. 2003, 88, 1678–1682. [Google Scholar] [CrossRef] [PubMed]
- Paslakis, G.; Agüera, Z.; Granero, R.; Sánchez, I.; Riesco, N.; Jiménez-Murcia, S.; Fernández-García, J.C.; Garrido-Sánchez, L.; Tinahones, F.J.; Casanueva, F.F. Associations between Neuropsychological Performance and Appetite-Regulating Hormones in Anorexia Nervosa and Healthy Controls: Ghrelin’s Putative Role as a Mediator of Decision-Making. Mol. Cell. Endocrinol. 2019, 497, 110441. [Google Scholar] [CrossRef]
- Rigamonti, A.E.; Pincelli, A.I.; Corra, B.; Viarengo, R.; Bonomo, S.M.; Galimberti, D.; Scacchi, M.; Scarpini, E.; Cavagnini, F.; Muller, E.E. Plasma Ghrelin Concentrations in Elderly Subjects: Comparison with Anorexic and Obese Patients. J. Endocrinol. 2002, 175, R1–R5. [Google Scholar] [CrossRef]
- Soriano-Guillen, L.; Barrios, V.; Campos-Barros, A.; Argente, J. Ghrelin Levels in Obesity and Anorexia Nervosa: Effect of Weight Reduction or Recuperation. J. Pediatrics 2004, 144, 36–42. [Google Scholar] [CrossRef]
- Stock, S.; Leichner, P.; Wong, A.C.; Ghatei, M.A.; Kieffer, T.J.; Bloom, S.R.; Chanoine, J.P. Ghrelin, Peptide YY, Glucose-Dependent Insulinotropic Polypeptide, and Hunger Responses to a Mixed Meal in Anorexic, Obese, and Control Female Adolescents. J. Clin. Endocrinol. Metab. 2005, 90, 2161–2168. [Google Scholar] [CrossRef] [PubMed]
- Stoving, R.K.; Chen, J.W.; Glintborg, D.; Brixen, K.; Flyvbjerg, A.; Horder, K.; Frystyk, J. Bioactive Insulin-like Growth Factor (IGF) I and IGF-Binding Protein-1 in Anorexia Nervosa. J. Clin. Endocrinol. Metab. 2007, 92, 2323–2329. [Google Scholar] [CrossRef]
- Terra, X.; Auguet, T.; Agüera, Z.; Quesada, I.M.; Orellana-Gavaldà, J.M.; Aguilar, C.; Jiménez-Murcia, S.; Berlanga, A.; Guiu-Jurado, E.; Menchón, J.M.; et al. Adipocytokine Levels in Women with Anorexia Nervosa. Relationship with Weight Restoration and Disease Duration. Int. J. Eat. Disord. 2013, 46, 855–861. [Google Scholar] [CrossRef] [PubMed]
- Troisi, A.; Di Lorenzo, G.; Lega, I.; Tesauro, M.; Bertoli, A.; Leo, R.; Iantorno, M.; Pecchioli, C.; Rizza, S.; Turriziani, M.; et al. Plasma Ghrelin in Anorexia, Bulimia, and Binge-Eating Disorder: Relations with Eating Patterns and Circulating Concentrations of Cortisol and Thyroid Hormones. Neuroendocrinology 2005, 81, 259–266. [Google Scholar] [CrossRef] [PubMed]
- Wollenhaupt, C.; Wilke, L.; Erim, Y.; Rauh, M.; Steins-Loeber, S.; Paslakis, G. The Association of Leptin Secretion with Cognitive Performance in Patients with Eating Disorders. Psychiatry Res. 2019, 276, 269–277. [Google Scholar] [CrossRef]
- Zamrazilova, H.; Hainer, V.; Sedlackova, D.; Papezova, H.; Kunesova, M.; Bellisle, F.; Hill, M.; Nedvidkova, J. Plasma Obestatin Levels in Normal Weight, Obese and Anorectic Women. Physiol. Res. 2008, 57, S49–S55. [Google Scholar]
- Monteleone, P.; Serritella, C.; Martiadis, V.; Maj, M. Deranged Secretion of Ghrelin and Obestatin in the Cephalic Phase of Vagal Stimulation in Women with Anorexia Nervosa. Biol. Psychiatry 2008, 64, 1005–1008. [Google Scholar] [CrossRef]
- Harada, T.; Nakahara, T.; Yasuhara, D.; Kojima, S.; Sagiyama, K.; Amitani, H.; Laviano, A.; Naruo, T.; Inui, A. Obestatin, Acyl Ghrelin, and Des-Acyl Ghrelin Responses to an Oral Glucose Tolerance Test in the Restricting Type of Anorexia Nervosa. Biol. Psychiatry 2008, 63, 245–247. [Google Scholar] [CrossRef]
- Heruc, G.A.; Little, T.J.; Kohn, M.; Madden, S.; Clarke, S.; Horowitz, M.; Feinle-Bisset, C. Appetite Perceptions, Gastrointestinal Symptoms, Ghrelin, Peptide YY and State Anxiety Are Disturbed in Adolescent Females with Anorexia Nervosa and Only Partially Restored with Short-Term Refeeding. Nutrients 2019, 11, 59. [Google Scholar] [CrossRef]
- Holsen, L.M.; Lawson, E.A.; Christensen, K.; Klibanski, A.; Goldstein, J.M. Abnormal Relationships between the Neural Response to High- and Low-Calorie Foods and Endogenous Acylated Ghrelin in Women with Active and Weight-Recovered Anorexia Nervosa. Psychiatry Res. Neuroimaging 2014, 223, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Koyama, K.I.; Yasuhara, D.; Nakahara, T.; Harada, T.; Uehara, M.; Ushikai, M.; Asakawa, A.; Inui, A. Changes in Acyl Ghrelin, Des-Acyl Ghrelin, and Ratio of Acyl Ghrelin to Total Ghrelin with Short-Term Refeeding in Female Inpatients with Restricting-Type Anorexia Nervosa. Horm. Metab. Res. 2010, 42, 595–598. [Google Scholar] [CrossRef] [PubMed]
- Nakahara, T.; Harada, T.; Yasuhara, D.; Shimada, N.; Amitani, H.; Sakoguchi, T.; Kamiji, M.M.; Asakawa, A.; Inui, A. Plasma Obestatin Concentrations Are Negatively Correlated with Body Mass Index, Insulin Resistance Index, and Plasma Leptin Concentrations in Obesity and Anorexia Nervosa. Biol. Psychiatry 2008, 64, 252–255. [Google Scholar] [CrossRef] [PubMed]
- Ogiso, K.; Asakawa, A.; Amitani, H.; Nakahara, T.; Ushikai, M.; Haruta, I.; Koyama, K.; Amitani, M.; Harada, T.; Yasuhara, D.; et al. Plasma Nesfatin-1 Concentrations in Restricting-Type Anorexia Nervosa. Peptides 2011, 32. [Google Scholar] [CrossRef]
- Simon, J.J.; Stopyra, M.A.; Mönning, E.; Sailer, S.C.; Lavandier, N.; Kihm, L.; Bendszus, M.; Preissl, H.; Herzog, W.; Friederich, H.-C. Neuroimaging of Hypothalamic Mechanisms Related to Glucose Metabolism in Anorexia Nervosa and Obesity. J. Clin. Investig. 2020. [Google Scholar] [CrossRef]
- Westwater, M.L.; Mancini, F.; Shapleske, J.; Serfontein, J.; Ernst, M.; Ziauddeen, H.; Fletcher, P.C. Dissociable Hormonal Profiles for Psychopathology and Stress in Anorexia and Bulimia Nervosa. Psychol. Med. 2020, 1–11. [Google Scholar] [CrossRef]
- Uehara, M.; Yasuhara, D.; Nakahara, T.; Harada, T.; Koyama, K.I.; Ushikai, M.; Asakawa, A.; Inui, A. Increase in Energy Intake Leads to a Decrease in Obestatin in Restricting-Type of Anorexia Nervosa. Exp. Clin. Endocrinol. Diabetes 2011, 119, 536–539. [Google Scholar] [CrossRef]
- Bernardoni, F.; Bernhardt, N.; Pooseh, S.; King, J.A.; Geisler, D.; Ritschel, F.; Boehm, I.; Seidel, M.; Roessner, V.; Smolka, M.N.; et al. Metabolic State and Value-Based Decision-Making in Acute and Recovered Female Patients with Anorexia Nervosa. J. Psychiatry Neurosci. Jpn 2020, 45, 253–261. [Google Scholar] [CrossRef]
- Nogueira, J.P.; Valero, R.; Maraninchi, M.; Lorec, A.M.; Samuelian-Massat, C.; Begu-Le Corroller, A.; Nicolay, A.; Gaudart, J.; Portugal, H.; Vialettes, B. Growth Hormone Level at Admission and Its Evolution during Refeeding Are Predictive of Short-Term Outcome in Restrictive Anorexia Nervosa. Br. J. Nutr. 2013, 109, 2175–2181. [Google Scholar] [CrossRef]
- Grinspoon, S.; Miller, K.K.; Herzog, D.B.; Grieco, K.A.; Klibanski, A. Effects of Estrogen and Recombinant Human Insulin-like Growth Factor-I on Ghrelin Secretion in Severe Undernutrition. J. Clin. Endocrinol. Metab. 2004, 89, 3988–3993. [Google Scholar] [CrossRef][Green Version]
- Fazeli, P.K.; Lawson, E.A.; Faje, A.T.; Eddy, K.T.; Lee, H.; Fiedorek, F.T.; Breggia, A.; Gaal, I.M.; DeSanti, R.; Klibanski, A. Treatment with a Ghrelin Agonist in Outpatient Women with Anorexia Nervosa: A Randomized Clinical Trial. J. Clin. Psychiatry 2018, 79. [Google Scholar] [CrossRef] [PubMed]
- Duriez, P.; Robichon, L.; Dardennes, R.; Lavoisy, G.; Grouselle, D.; Epelbaum, J.; Ramoz, N.; Gorwood, P.; Tolle, V.; Viltart, O. Unexpected Association of Desacyl-Ghrelin with Physical Activity and Chronic Food Restriction: A Translational Study on Anorexia Nervosa. J. Clin. Med. 2020, 9. [Google Scholar] [CrossRef]
- Stojiljkovic-Drobnjak, S.; Fischer, S.; Arnold, M.; Langhans, W.; Ehlert, U. Menopause Is Associated with Decreased Postprandial Ghrelin, Whereas a History of Anorexia Nervosa Is Associated with Increased Total Ghrelin. J. Neuroendocrinol. 2019, 31, e12661. [Google Scholar] [CrossRef]
- Prince, A.C.; Brooks, S.J.; Stahl, D.; Treasure, J. Systematic Review and Meta-Analysis of the Baseline Concentrations and Physiologic Responses of Gut Hormones to Food in Eating Disorders. Am. J. Clin. Nutr. 2009, 89, 755–765. [Google Scholar] [CrossRef] [PubMed]
- Mequinion, M.; Langlet, F.; Zgheib, S.; Dickson, S.; Dehouck, B.; Chauveau, C.; Viltart, O. Ghrelin: Central and Peripheral Implications in Anorexia Nervosa. Front. Endocrinol. 2013, 4. [Google Scholar] [CrossRef]
- Ogiso, K.; Asakawa, A.; Amitani, H.; Inui, A. Ghrelin and Anorexia Nervosa: A Psychosomatic Perspective. Nutrition 2011, 27, 988–993. [Google Scholar] [CrossRef]
- Miljic, D.; Pekic, S.; Djurovic, M.; Doknic, M.; Milic, N.; Casanueva, F.F.; Ghatei, M.; Popovic, V. Ghrelin Has Partial or No Effect on Appetite, Growth Hormone, Prolactin, and Cortisol Release in Patients with Anorexia Nervosa. J. Clin. Endocrinol. Metab. 2006, 91, 1491–1495. [Google Scholar] [CrossRef] [PubMed]
- Miljic, D.; Djurovic, M.; Pekic, S.; Doknic, M.; Stojanovic, M.; Milic, N.; Casanueva, F.F.; Ghatei, M.; Popovic, V. Glucose Metabolism during Ghrelin Infusion in Patients with Anorexia Nervosa. J. Endocrinol. Investig. 2007, 30, 771–775. [Google Scholar] [CrossRef] [PubMed]
- Dardennes, R.M.; Zizzari, P.; Tolle, V.; Foulon, C.; Kipman, A.; Romo, L.; Iancu-Gontard, D.; Boni, C.; Sinet, P.M.; Therese Bluet, M.; et al. Family Trios Analysis of Common Polymorphisms in the Obestatin/Ghrelin, BDNF and AGRP Genes in Patients with Anorexia Nervosa: Association with Subtype, Body-Mass Index, Severity and Age of Onset. Psychoneuroendocrinology 2007, 32, 106–113. [Google Scholar] [CrossRef]
- Müller, T.D.; Tschöp, M.H.; Jarick, I.; Ehrlich, S.; Scherag, S.; Herpertz-Dahlmann, B.; Zipfel, S.; Herzog, W.; de Zwaan, M.; Burghardt, R.; et al. Genetic Variation of the Ghrelin Activator Gene Ghrelin O-Acyltransferase (GOAT) Is Associated with Anorexia Nervosa. J. Psychiatr. Res. 2011, 45, 706–711. [Google Scholar] [CrossRef] [PubMed]
- Ukkola, O.; Ravussin, E.; Jacobson, P.; Pérusse, L.; Rankinen, T.; Tschöp, M.; Heiman, M.L.; Leon, A.S.; Rao, D.C.; Skinner, J.S. Role of Ghrelin Polymorphisms in Obesity Based on Three Different Studies. Obes. Res. 2002, 10, 782–791. [Google Scholar] [CrossRef]
- Korbonits, M.; Goldstone, A.P.; Gueorguiev, M.; Grossman, A.B. Ghrelin—a Hormone with Multiple Functions. Front. Neuroendocrinol. 2004, 25, 27–68. [Google Scholar] [CrossRef]
- Schellekens, H.; Dinan, T.G.; Cryan, J.F. Lean Mean Fat Reducing “Ghrelin” Machine: Hypothalamic Ghrelin and Ghrelin Receptors as Therapeutic Targets in Obesity. Neuropharmacology 2010, 58, 2–16. [Google Scholar] [CrossRef]
- Stengel, A.; Taché, Y. Ghrelin–a Pleiotropic Hormone Secreted from Endocrine X/A-like Cells of the Stomach. Front. Neurosci. 2012, 6, 24. [Google Scholar] [CrossRef]
- Giannunzio, V.; Degortes, D.; Tenconi, E.; Collantoni, E.; Solmi, M.; Santonastaso, P.; Favaro, A. Decision-Making Impairment in Anorexia Nervosa: New Insights into the Role of Age and Decision-Making Style. Eur. Eat. Disord. Rev. 2018, 26, 302–314. [Google Scholar] [CrossRef] [PubMed]
- Stievenard, A.; Méquinion, M.; Andrews, Z.B.; Destée, A.; Chartier-Harlin, M.-C.; Viltart, O.; Vanbesien-Mailliot, C.C. Is There a Role for Ghrelin in Central Dopaminergic Systems? Focus on Nigrostriatal and Mesocorticolimbic Pathways. Neurosci. Biobehav. Rev. 2017, 73, 255–275. [Google Scholar] [CrossRef]
- Jerlhag, E.; Egecioglu, E.; Dickson, S.L.; Douhan, A.; Svensson, L.; Engel, J.A. Preclinical Study: Ghrelin Administration into Tegmental Areas Stimulates Locomotor Activity and Increases Extracellular Concentration of Dopamine in the Nucleus Accumbens. Addict. Biol. 2007, 12, 6–16. [Google Scholar] [CrossRef]
- Malik, S.; McGlone, F.; Bedrossian, D.; Dagher, A. Ghrelin Modulates Brain Activity in Areas That Control Appetitive Behavior. Cell Metab. 2008, 7, 400–409. [Google Scholar] [CrossRef]
- Kroemer, N.B.; Krebs, L.; Kobiella, A.; Grimm, O.; Pilhatsch, M.; Bidlingmaier, M.; Zimmermann, U.S.; Smolka, M.N. Fasting Levels of Ghrelin Covary with the Brain Response to Food Pictures: Ghrelin and Food-Cue Reactivity. Addict. Biol. 2013, 18, 855–862. [Google Scholar] [CrossRef]
- Dickson, S.L.; Egecioglu, E.; Landgren, S.; Skibicka, K.P.; Engel, J.A.; Jerlhag, E. The Role of the Central Ghrelin System in Reward from Food and Chemical Drugs. Mol. Cell. Endocrinol. 2011, 340, 80–87. [Google Scholar] [CrossRef]
- Anderberg, R.H.; Hansson, C.; Fenander, M.; Richard, J.E.; Dickson, S.L.; Nissbrandt, H.; Bergquist, F.; Skibicka, K.P. The Stomach-Derived Hormone Ghrelin Increases Impulsive Behavior. Neuropsychopharmacology 2016, 41, 1199–1209. [Google Scholar] [CrossRef]
- Farokhnia, M.; Grodin, E.N.; Lee, M.R.; Oot, E.N.; Blackburn, A.N.; Stangl, B.L.; Schwandt, M.L.; Farinelli, L.A.; Momenan, R.; Ramchandani, V.A. Exogenous Ghrelin Administration Increases Alcohol Self-Administration and Modulates Brain Functional Activity in Heavy-Drinking Alcohol-Dependent Individuals. Mol. Psychiatry 2018, 23, 2029–2038. [Google Scholar] [CrossRef]
- Ralevski, E.; Shanabrough, M.; Newcomb, J.; Gandelman, E.; Hayden, R.; Horvath, T.L.; Petrakis, I. Ghrelin Is Related to Personality Differences in Reward Sensitivity and Impulsivity. Alcohol Alcohol. 2018, 53, 52–56. [Google Scholar] [CrossRef]
- Bogdanov, V.B.; Bogdanova, O.V.; Dexpert, S.; Delgado, I.; Beyer, H.; Aubert, A.; Dilharreguy, B.; Beau, C.; Forestier, D.; Ledaguenel, P.; et al. Reward-Related Brain Activity and Behavior Are Associated with Peripheral Ghrelin Levels in Obesity. Psychoneuroendocrinology 2020, 112, 104520. [Google Scholar] [CrossRef]
- Fichter, M.M.; Quadflieg, N.; Crosby, R.D.; Koch, S. Long-Term Outcome of Anorexia Nervosa: Results from a Large Clinical Longitudinal Study. Int. J. Eat. Disord. 2017, 50, 1018–1030. [Google Scholar] [CrossRef] [PubMed]
- Franko, D.L.; Tabri, N.; Keshaviah, A.; Murray, H.B.; Herzog, D.B.; Thomas, J.J.; Coniglio, K.; Keel, P.K.; Eddy, K.T. Predictors of Long-Term Recovery in Anorexia Nervosa and Bulimia Nervosa: Data from a 22-Year Longitudinal Study. J. Psychiatr. Res. 2018, 96, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Treasure, J.; Zipfel, S.; Micali, N.; Wade, T.; Stice, E.; Claudino, A.; Schmidt, U.; Frank, G.K.; Bulik, C.M.; Wentz, E. Anorexia Nervosa. Nat. Rev. Dis. Primers 2015, 26, 15074. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, S.; Giel, K.E.; Bulik, C.M.; Hay, P.; Schmidt, U. Anorexia Nervosa: Aetiology, Assessment, and Treatment. Lancet Psychiatry 2015, 2, 1099–1111. [Google Scholar] [CrossRef]
- Steinglass, J.E.; Glasofer, D.R.; Dalack, M.; Attia, E. Between Wellness, Relapse, and Remission: Stages of Illness in Anorexia Nervosa. Int. J. Eat. Disord. 2020, 53, 1088–1096. [Google Scholar] [CrossRef]
- Fazeli, P.K.; Lawson, E.A.; Faje, A.T.; Eddy, K.T.; Gaal, I.; DeSanti, R.L.; Klibanski, A. Short-Term Treatment with a Ghrelin Agonist Significantly Improves Gastric Emptying in Anorexia Nervosa. In Proceedings of the Endocrine Society’s 98th Annual Meeting and Expo, Boston, MA, USA, 1–4 April 2016; Volume 37. [Google Scholar] [CrossRef]
- Hotta, M.; Ohwada, R.; Akamizu, T.; Shibasaki, T.; Takano, K.; Kangawa, K. Ghrelin Increases Hunger and Food Intake in Patients with Restricting-Type Anorexia Nervosa: A Pilot Study. Endocr. J. 2009, 56, 1119–1128. [Google Scholar] [CrossRef] [PubMed]
| Mean | SD | Min | Max | |
|---|---|---|---|---|
| Age at baseline | 22.56 | 3.72 | 17.00 | 28.9 |
| BMI at baseline | 14.94 | 1.49 | 12.40 | 17.8 |
| BMI delta | −1.71 | 1.24 | −0.1 | −4.40 |
| Duration to follow-up | 94.62 | 89.56 | 30 | 365 |
| Estimate | SE | p | 95% CI, L | 95%, CI, U | |
|---|---|---|---|---|---|
| Age | 0.19 | 0.37 | 0.64 | −1.13 | 1.52 |
| BMI baseline | −1.13 | 1.36 | 0.47 | −5.78 | 3.51 |
| BMI delta | 0.83 | 1.41 | 0.60 | −3.86 | 5.53 |
| Duration to follow-up | −0.01 | 0.01 | 0.74 | −0.07 | 0.06 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seidel, M.; Markmann Jensen, S.; Healy, D.; Dureja, A.; Watson, H.J.; Holst, B.; Bulik, C.M.; Sjögren, J.M. A Systematic Review and Meta-Analysis Finds Increased Blood Levels of All Forms of Ghrelin in Both Restricting and Binge-Eating/Purging Subtypes of Anorexia Nervosa. Nutrients 2021, 13, 709. https://doi.org/10.3390/nu13020709
Seidel M, Markmann Jensen S, Healy D, Dureja A, Watson HJ, Holst B, Bulik CM, Sjögren JM. A Systematic Review and Meta-Analysis Finds Increased Blood Levels of All Forms of Ghrelin in Both Restricting and Binge-Eating/Purging Subtypes of Anorexia Nervosa. Nutrients. 2021; 13(2):709. https://doi.org/10.3390/nu13020709
Chicago/Turabian StyleSeidel, Maria, Signe Markmann Jensen, Darren Healy, Aakriti Dureja, Hunna J. Watson, Birgitte Holst, Cynthia M. Bulik, and Jan Magnus Sjögren. 2021. "A Systematic Review and Meta-Analysis Finds Increased Blood Levels of All Forms of Ghrelin in Both Restricting and Binge-Eating/Purging Subtypes of Anorexia Nervosa" Nutrients 13, no. 2: 709. https://doi.org/10.3390/nu13020709
APA StyleSeidel, M., Markmann Jensen, S., Healy, D., Dureja, A., Watson, H. J., Holst, B., Bulik, C. M., & Sjögren, J. M. (2021). A Systematic Review and Meta-Analysis Finds Increased Blood Levels of All Forms of Ghrelin in Both Restricting and Binge-Eating/Purging Subtypes of Anorexia Nervosa. Nutrients, 13(2), 709. https://doi.org/10.3390/nu13020709







