Neurodevelopmental Outcomes and Gut Bifidobacteria in Term Infants Fed an Infant Formula Containing High sn-2 Palmitate: A Cluster Randomized Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Participants
2.3. Study Feedings
2.4. Data Collection
2.4.1. General Information
2.4.2. Ages and Stages Questionnaire
2.4.3. Anthropometry
2.4.4. Fecal Bifidobacteria
2.4.5. Digestive Tolerance
2.5. Statistical Analysis
3. Results
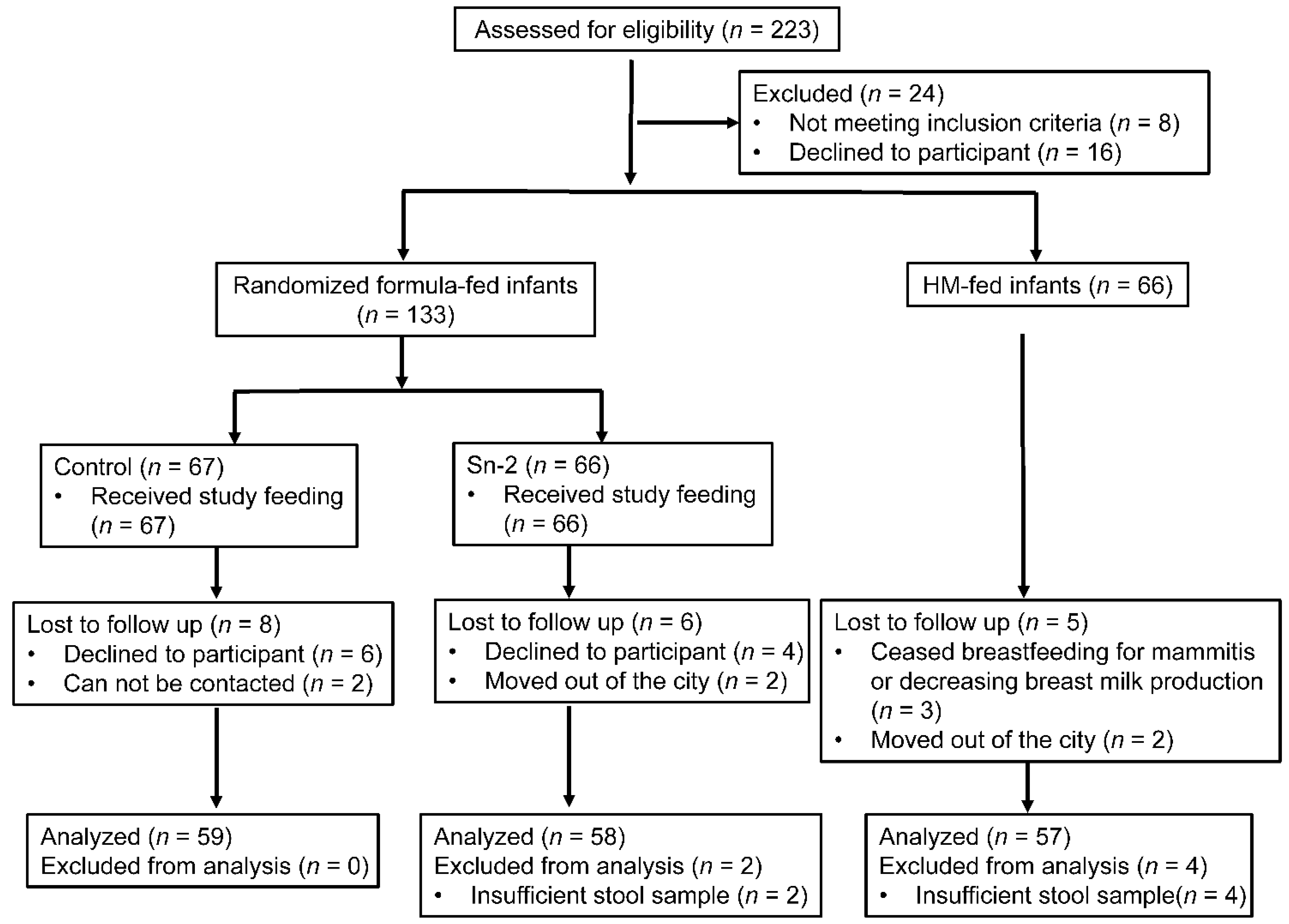
3.1. Study Population
3.2. Anthropometric Data
3.3. ASQ-3 Scores
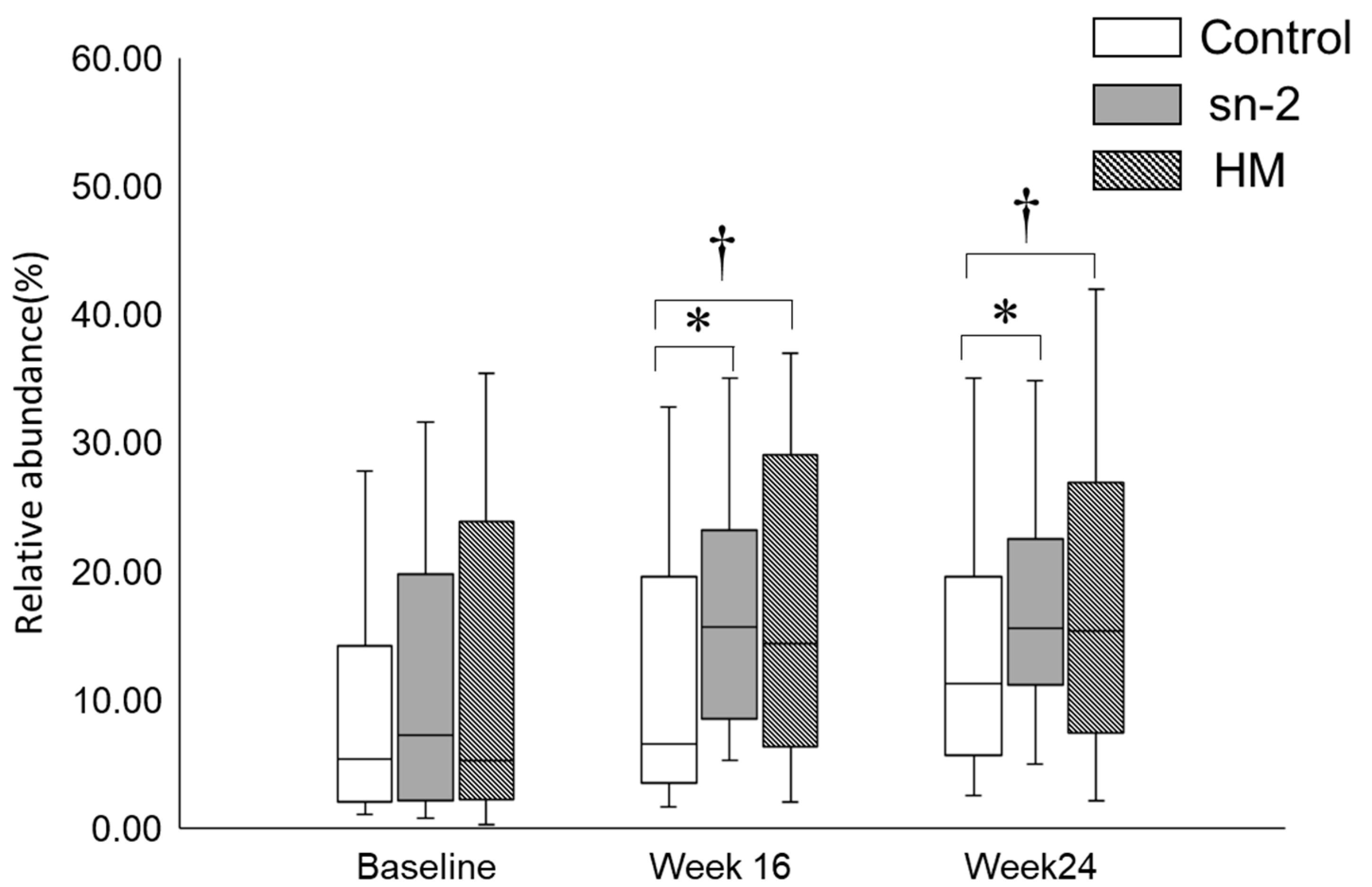
3.4. Fecal Bifidobacteria
3.5. Digestive Tolerance and Allergic Diseases
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mazzocchi, A.; D’Oria, V.; Cosmi, V.D.; Bettocchi, S.; Milani, G.P.; Silano, M.; Agostoni, C. The Role of Lipids in Human Milk and Infant Formulae. Nutrients 2018, 10, 567. [Google Scholar] [CrossRef] [PubMed]
- Jiang, T.; Liu, B.; Li, J.; Dong, X.; Lin, M.; Zhang, M.; Zhao, J.; Dai, Y.; Chen, L. Association between sn-2 fatty acid profiles of breast milk and development of the infant intestinal microbiome. Food Funct. 2018, 9, 1028–1037. [Google Scholar] [CrossRef]
- Mu, H.; Høy, C.-E. The digestion of dietary triacylglycerols. Prog. Lipid Res. 2004, 43, 105–133. [Google Scholar] [CrossRef]
- Bar-Yoseph, F.; Lifshitz, Y.; Cohen, T.; Malard, P.; Xu, C. SN2-Palmitate Reduces Fatty Acid Excretion in Chinese Formula-fed Infants. J. Pediatr. Gastroenterol. Nutr. 2016, 62, 341–347. [Google Scholar] [CrossRef]
- Innis, S.M. Dietary triacylglycerol structure and its role in infant nutrition. Adv. Nutr. 2011, 2, 275–283. [Google Scholar] [CrossRef]
- Kennedy, K.; Fewtrell, M.S.; Morley, R.; Abbott, R.; Quinlan, P.T.; Wells, J.C.; Bindels, J.G.; Lucas, A. Double-blind, randomized trial of a synthetic triacylglycerol in formula-fed term infants: Effects on stool biochemistry, stool characteristics, and bone mineralization. Am. J. Clin. Nutr. 1999, 70, 920–927. [Google Scholar] [CrossRef] [PubMed]
- Yao, M.; Lien, E.L.; Capeding, M.R.Z.; Fitzgerald, M.; Ramanujam, K.; Yuhas, R.; Northington, R.; Lebumfacil, J.; Wang, L.; DeRusso, P.A. Effects of term infant formulas containing high sn-2 palmitate with and without oligofructose on stool composition, stool characteristics, and bifidogenicity. J. Pediatr. Gastroenterol. Nutr. 2014, 59, 440–448. [Google Scholar] [CrossRef]
- Nowacki, J.; Lee, H.-C.; Lien, R.; Cheng, S.-W.; Li, S.-T.; Yao, M.; Northington, R.; Jan, I.; Mutungi, G. Stool fatty acid soaps, stool consistency and gastrointestinal tolerance in term infants fed infant formulas containing high sn-2 palmitate with or without oligofructose: A double-blind, randomized clinical trial. Nutr. J. 2014, 13, 1–11. [Google Scholar] [CrossRef]
- Yaron, S.; Shachar, D.; Abramas, L.; Riskin, A.; Bader, D.; Litmanovitz, Z.; Bar-Yoseph, F.; Cohen, T.; Levi, L.; Lifshitz, Y.; et al. Effect of high β-palmitate content in infant formula on the intestinal microbiota of term infants. J. Pediatr. Gastroenterol. Nutr. 2013, 56, 376–381. [Google Scholar] [CrossRef]
- Civardi, E.; Garofoli, F.; Mazzucchelli, I.; Angelini, M.; Manzoni, P.; Stronati, M. Enteral nutrition and infections: The role of human milk. Early Hum. Dev. 2014, 90 (Suppl. 1), S57–S59. [Google Scholar] [CrossRef]
- Bezirtzoglou, E.; Stavropoulou, E. Immunology and probiotic impact of the newborn and young children intestinal microflora. Anaerobe 2011, 17, 369–374. [Google Scholar] [CrossRef]
- Sjögren, Y.M.; Jenmalm, M.C.; Böttcher, M.F.; Björkstén, B.; Sverremark-Ekström, E. Altered early infant gut microbiota in children developing allergy up to 5 years of age. Clin. Exp. Allergy 2009, 39, 518–526. [Google Scholar] [CrossRef]
- Desbonnet, L.; Clarke, G.; Shanahan, F.; Dinan, T.G.; Cryan, J.F. Microbiota is essential for social development in the mouse. Mol. Psychiatry 2014, 19, 146–148. [Google Scholar] [CrossRef] [PubMed]
- Clarke, G.; Grenham, S.; Scully, P.; Fitzgerald, P.; Moloney, R.D.; Shanahan, F.; Dinan, T.G.; Cryan, J.F. The microbiome-gut-brain axis during early life regulates the hippocampal serotonergic system in a sex-dependent manner. Mol. Psychiatry 2013, 18, 666–673. [Google Scholar] [CrossRef]
- Tremlett, H.; Fadrosh, D.W.; Faruqi, A.A.; Zhu, F.; Hart, J.; Roalstad, S.; Graves, J.; Lynch, S.; Waubant, E.; US Network of Pediatric MS Centers. Gut microbiota in early pediatric multiple sclerosis: A case-control study. Eur. J. Neurol. 2016, 23, 1308–1321. [Google Scholar] [CrossRef] [PubMed]
- Rose, D.R.; Yang, H.; Serena, G.; Sturgeon, C.; Ma, B.; Careaga, M.; Hughes, H.K.; Angkustsiri, K.; Rose, M.; Hertz-Picciotto, I.; et al. Differential immune responses and microbiota profiles in children with autism spectrum disorders and co-morbid gastrointestinal symptoms. Brain Behav. Immun. 2018, 70, 354–368. [Google Scholar] [CrossRef] [PubMed]
- Zyoud, S.H.; Smale, S.; Waring, W.S.; Sweileh, W.M.; Al-Jabi, S.W. Global research trends in microbiome-gut-brain axis during 2009-2018: A bibliometric and visualized study. BMC Gastroenterol. 2019, 19, 158. [Google Scholar] [CrossRef]
- Kałużna-Czaplińska, J.; Błaszczyk, S. The level of arabinitol in autistic children after probiotic therapy. Nutrition 2012, 28, 124–126. [Google Scholar] [CrossRef] [PubMed]
- Kazemi, A.; Noorbala, A.A.; Azam, K.; Eskandari, M.H.; Djafarian, K. Effect of probiotic and prebiotic vs placebo on psychological outcomes in patients with major depressive disorder: A randomized clinical trial. Clin. Nutr. 2019, 38, 522–528. [Google Scholar] [CrossRef]
- The Ministry of Health of the People’s Republic of China. National Food Safety Standard on Infant Formula; The Ministry of Health of the People’s Republic of China: Beijing, China, 2010.
- Squires, J.; Twombly, E.; Bricker, D.; Potter, L. ASQ-3 User’s Guide; Paul H Brookes Publishing Co: Baltimore, MD, USA, 2009. [Google Scholar]
- Wei, M.; Bai, X.; Squires, J.; Yao, G.; Wang, X.; Xie, H.; Song, W.; Lu, J. Studied of the norm and psychometrical properties of the ages and stages questionnaires, third edition, with a Chinese national sample. Chin. J. Pediatr. 2015, 53, 913. [Google Scholar]
- Bellinger, D.C. What is an adverse effect? A possible resolution of clinical and epidemiological perspectives on neurobehavioral toxicity. Environ. Res. 2004, 95, 394–405. [Google Scholar] [CrossRef]
- Tang, K.; Wang, H.; Tan, S.H.; Xin, T.; Qu, X.; Tang, T.; Wang, Y.; Liu, Y.; Gaoshan, J. Association between maternal education and breast feeding practices in China: A population-based cross-sectional study. BMJ Open 2019, 9, e028485. [Google Scholar] [CrossRef] [PubMed]
- Sarki, M.; Parlesak, A.; Robertson, A. Comparison of national cross-sectional breast-feeding surveys by maternal education in Europe (2006–2016). Public Health Nutr. 2019, 22, 848–861. [Google Scholar] [CrossRef]
- Sordillo, J.E.; Korrick, S.; Laranjo, N.; Carey, V.; Weinstock, G.M.; Gold, D.R.; O’Connor, G.; Sandel, M.; Bacharier, L.B.; Beigelman, A.; et al. Association of the Infant Gut Microbiome With Early Childhood Neurodevelopmental Outcomes: An Ancillary Study to the VDAART Randomized Clinical Trial. JAMA Netw. Open 2019, 2, e190905. [Google Scholar] [CrossRef] [PubMed]
- Davis, E.E.; Pitchford, N.J.; Limback, E. The interrelation between cognitive and motor development in typically developing children aged 4–11 years is underpinned by visual processing and fine manual control. Br. J. Psychol. (Lond. Engl. 1953) 2011, 102, 569–584. [Google Scholar] [CrossRef]
- Guesnet, P.; Alessandri, J.M. Docosahexaenoic acid (DHA) and the developing central nervous system (CNS)—Implications for dietary recommendations. Biochimie 2011, 93, 7–12. [Google Scholar] [CrossRef] [PubMed]
- Carlson, A.L.; Xia, K.; Azcarate-Peril, M.A.; Goldman, B.D.; Ahn, M.; Styner, M.A.; Thompson, A.L.; Geng, X.; Gilmore, J.H.; Knickmeyer, R.C. Infant Gut Microbiome Associated With Cognitive Development. Biol. Psychiatry 2018, 83, 148–159. [Google Scholar] [CrossRef]
- Yatsunenko, T.; Rey, F.E.; Manary, M.J.; Trehan, I.; Dominguez-Bello, M.G.; Contreras, M.; Magris, M.; Hidalgo, G.; Baldassano, R.N.; Anokhin, A.P.; et al. Human gut microbiome viewed across age and geography. Nature 2012, 486, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Stiles, J.; Jernigan, T.L. The basics of brain development. Neuropsychol. Rev. 2010, 20, 327–348. [Google Scholar] [CrossRef]
- Tau, G.Z.; Peterson, B.S. Normal development of brain circuits. Neuropsychopharmacology 2010, 35, 147–168. [Google Scholar] [CrossRef] [PubMed]
- Quigley, E.M.M. Microbiota-Brain-Gut Axis and Neurodegenerative Diseases. Curr. Neurol. Neurosci. Rep. 2017, 17, 94. [Google Scholar] [CrossRef] [PubMed]
- Schirmer, M.; Smeekens, S.P.; Vlamakis, H.; Jaeger, M.; Oosting, M.; Franzosa, E.A.; Horst, R.T.; Jansen, T.; Jacobs, L.; Bonder, M.J.; et al. Linking the Human Gut Microbiome to Inflammatory Cytokine Production Capacity. Cell 2016, 167, 1897. [Google Scholar] [CrossRef]
- Vuong, H.E.; Yano, J.M.; Fung, T.C.; Hsiao, E.Y. The Microbiome and Host Behavior. Annu. Rev. Neurosci. 2017, 40, 21–49. [Google Scholar] [CrossRef]
- Choi, J.; Lee, S.; Won, J.; Jin, Y.; Hong, Y.; Hur, T.-Y.; Kim, J.-H.; Lee, S.-R.; Hong, Y. Pathophysiological and neurobehavioral characteristics of a propionic acid-mediated autism-like rat model. PLoS ONE 2018, 13, e0192925. [Google Scholar] [CrossRef] [PubMed]
- O’Mahony, S.M.; Clarke, G.; Borre, Y.E.; Dinan, T.G.; Cryan, J.F. Serotonin, tryptophan metabolism and the brain-gut-microbiome axis. Behav. Brain Res. 2015, 277, 32–48. [Google Scholar] [CrossRef] [PubMed]
- Sharon, G.; Sampson, T.R.; Geschwind, D.H.; Mazmanian, S.K. The Central Nervous System and the Gut Microbiome. Cell 2016, 167, 915–932. [Google Scholar] [CrossRef]
- Shaaban, S.Y.; Gendy, Y.G.E.; Mehanna, N.S.; El-Senousy, W.M.; El-Feki, H.S.A.; Saad, K.; El-Asheer, O.M. The role of probiotics in children with autism spectrum disorder: A prospective, open-label study. Nutr. Neurosci. 2018, 21, 676–681. [Google Scholar] [CrossRef]
| Composition | sn-2 | Control |
|---|---|---|
| Energy, KJ/100 g | 2130.3 | 2102.0 |
| Protein, g/100 g | 11.7 | 10.4 |
| Fat, g/100 g | 26.8 | 27.2 |
| Carbohydrate, g/100 g | 53.7 | 57.8 |
| Magnesium, mg/100 g | 30.0 | 38.0 |
| Calcium, mg/100 g | 349.4 | 380.0 |
| Phosphorus, mg/100 g | 223.7 | 230.0 |
| Vitamin B6, μg/100 g | 420.0 | 439.0 |
| Vitamin B12, μg/100 g | 1.5 | 1.5 |
| Folic acid, μg/100 g | 65.0 | 81.5 |
| Pantothenic acid, μg/100 g | 2850.0 | 2871.0 |
| Biotin, μg/100 g | 16.0 | 15.1 |
| Galacto-oligosaccharide, g/100 g | 1.5 | 1.5 |
| Fatty acid (%) 1 | ||
| C10 | 0.5 | 0.8 |
| C12 | 0.7 | 0.7 |
| C14 | 2.1 | 2.5 |
| C15 | 0.2 | 0.1 |
| C16:0 | 25.8 | 24.6 |
| C16:0 in sn-2 position 2 | 46.3 | 10.3 |
| C16:1 | 0.2 | 0.1 |
| C18 | 4.4 | 4.4 |
| C18:1n9 | 35.2 | 37.1 |
| C18:2n6 | 20.6 | 19.5 |
| C18:3n6 | 0.1 | 0.1 |
| C18:3n3 | 2.6 | 1.9 |
| C20 | 0.3 | 0.4 |
| C20:4n6 | 0.4 | 0.2 |
| C22 | 0.2 | 0.3 |
| C24 | 0.2 | 0.1 |
| Characteristic | Feeding Group | |||
|---|---|---|---|---|
| Control | sn-2 | BF | ||
| n | 59 | 58 | 57 | |
| Infant | ||||
| Female (%) | 30 (50.9) | 25 (43.1) | 21 (36.8) | |
| Age, days | 11 ± 1.7 | 12 ± 1.2 | 9 ± 2.5 | |
| Gestational age, weeks | 38.6 ± 1.3 | 38.8 ± 1.0 | 39.2 ± 0.9 | |
| Vaginal delivery (%) 2 | 15 (25.4) | 23 (37.5) | 38 (66.7) | |
| Weight, kg | 3.4 (3.1, 3.6) | 3.3 (3.1, 3.6) | 3.4 (3.1, 3.6) | |
| Length, cm | 50.0 (50.0, 51.0) | 50.0 (50.0, 51.0) | 50.0 (50.0, 51.0) | |
| Mother | ||||
| Age, years | 29 (27, 32) | 31 (28, 35) | 30 (28, 31) | |
| Education level (%) 3 bachelor’s degree or above | 12 (20.3) | 15 (25.9) | 31 (54.4) | |
| Occupational status (%) working | 34 (57.6) | 31 (53.4) | 40 (70.2) | |
| BMI before pregnancy | ≤18.0 | 11 (18.6) | 7 (12.1) | 7 (12.3) |
| ≤24.0 | 35 (59.3) | 37 (63.8) | 45 (78.9) | |
| >24.0 | 13 (22.0) | 14 (24.1) | 5 (8.8) | |
| Smoking during pregnancy | 1 (1.7) | 1 (1.7) | 1 (1.8) | |
| Drinking during pregnancy | 0 (0.0) | 0 (0.0) | (0.0) | |
| Developmental Domain | Compared with the Typical Development Threshold 1 | Control 59 | sn-2 58 | BF 57 | p | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| sn-2 vs. Control | sn-2 vs. BF | Control vs. BF | ||||||||
| Unadjusted 2 | Adjusted 3 | Unadjusted 2 | Adjusted 3 | Unadjusted 2 | Adjusted 3 | |||||
| Communication | Above | 50 (84.8) | 49 (84.5) | 52 (91.2) | Reference | Reference | Reference | |||
| Close to | 5 (8.5) | 7 (12.1) | 4 (7.0) | 0.543 | 0.574 | 0.331 | 0.156 | 0.708 | 0.307 | |
| Below | 4 (6.8) | 2 (3.4) | 1 (1.8) | 0.463 | 0.474 | 0.533 | 0.258 | 0.161 | ||
| Gross motor | Above | 53 (89.8) | 56 (96.6) | 56 (98.3) | Reference | Reference | Reference | |||
| Close to | 3 (5.1) | 2 (3.4) | 1 (1.8) | 0.635 | 0.687 | 0.566 | 0.304 | 0.324 | 0.127 | |
| Below | 3 (5.1) | 0 (0.0) | 0 (0.0) | 0.954 | 0.950 | — | — | 0.954 | 0.950 | |
| Fine motor | Above | 46 (78.0) | 53 (91.4) | 50 (87.7) | Reference | Reference | Reference | |||
| Close to | 11 (18.6) | 3 (5.2) | 7 (12.3) | 0.037 | 0.036 | 0.217 | 0.513 | 0.308 | 0.283 | |
| Below | 2 (3.4) | 2 (3.4) | 0 (0) | 0.904 | 0.882 | 0.984 | 0.953 | 0.943 | 0.943 | |
| Problem-solving | Above | 50 (84.8) | 53 (91.4) | 51 (89.5) | Reference | Reference | Reference | |||
| Close to | 6 (10.2) | 3 (5.2) | 6 (10.5) | 0.319 | 0.247 | 0.332 | 0.395 | 0.974 | 0.944 | |
| Below | 3 (5.1) | 2 (3.4) | 0 (0) | 0.634 | 0.509 | 0.984 | 0.954 | 0.954 | 0.955 | |
| Personal and social | Above | 51 (86.4) | 45 (77.6) | 52 (91.2) | Reference | Reference | Reference | |||
| Close to | 7 (11.9) | 10 (17.2) | 3 (5.3) | 0.345 | 0.384 | 0.047 | 0.077 | 0.227 | 0.188 | |
| Below | 1 (1.7) | 3 (5.2) | 2 (3.5) | 0.288 | 0.301 | 0.541 | 0.511 | 0.587 | 0.735 | |
| Number of domain scoring close to the threshold | 0 | 37 (62.7) | 40 (69.0) | 44 (77.2) | Reference | Reference | Reference | |||
| 1 | 14 (23.7) | 11 (19.0) | 5 (8.8) | 0.526 | 0.481 | 0.119 | 0.058 | 0.034 | 0.522 | |
| ≥2 | 8 (13.6) | 7 (12.1) | 8 (14.0) | 0.742 | 0.661 | 0.982 | 0.741 | 0.752 | 0.456 | |
| Number of domain scoring below the threshold | 0 | 52 (88.1) | 54 (93.1) | 54 (94.7) | Reference | Reference | Reference | |||
| 1 | 3 (5.1) | 1 (1.7) | 3 (5.3) | 0.340 | 0.338 | 0.356 | 0.427 | 0.964 | 0.786 | |
| ≥2 | 4 (6.8) | 3 (5.2) | 0 (0.0) | 0.697 | 0.622 | 0.954 | 0.938 | 0.947 | 0.947 | |
| Developmental Domain | Compared with the Typical Development Threshold | OR (95%CI) 1 | p |
|---|---|---|---|
| Communication | Above | Reference | |
| Close to | 1.003 (0.961–1.047) | 0.888 | |
| Below | 0.913 (0.798–1.045) | 0.186 | |
| Gross motor | Above | Reference | |
| Close to | 0.989 (0.925–1.057) | 0.74 | |
| Below | 0.622 (0.287–1.348) | 0.229 | |
| Fine motor | Above | Reference | |
| Close to | 0.997 (0.960–1.036) | 0.889 | |
| Below | 0.839 (0.654–1.077) | 0.168 | |
| Problem–solving | Above | Reference | |
| Close to | 0.952 (0.880–1.030) | 0.218 | |
| Below | 0.978 (0.900–1.062) | 0.595 | |
| Personal and social | Above | Reference | |
| Close to | 0.982 (0.938–1.029) | 0.452 | |
| Below | 1.051 (0.988–1.117) | 0.116 | |
| Number of domain scoring close to the threshold | 0 | Reference | |
| 1 | 0.947 (0.901–0.996) | 0.034 | |
| ≥2 | 1.001 (0.964–1.039) | 0.962 | |
| Number of domain scoring below the threshold | 0 | Reference | |
| 1 | 1.017 (0.962–1.075) | 0.554 | |
| ≥2 | 0.961 (0.884–1.045) | 0.352 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, W.; Zhao, A.; Liu, B.; Ye, W.-H.; Su, H.-W.; Li, J.; Zhang, Y.-M. Neurodevelopmental Outcomes and Gut Bifidobacteria in Term Infants Fed an Infant Formula Containing High sn-2 Palmitate: A Cluster Randomized Clinical Trial. Nutrients 2021, 13, 693. https://doi.org/10.3390/nu13020693
Wu W, Zhao A, Liu B, Ye W-H, Su H-W, Li J, Zhang Y-M. Neurodevelopmental Outcomes and Gut Bifidobacteria in Term Infants Fed an Infant Formula Containing High sn-2 Palmitate: A Cluster Randomized Clinical Trial. Nutrients. 2021; 13(2):693. https://doi.org/10.3390/nu13020693
Chicago/Turabian StyleWu, Wei, Ai Zhao, Biao Liu, Wen-Hui Ye, Hong-Wen Su, Jing Li, and Yu-Mei Zhang. 2021. "Neurodevelopmental Outcomes and Gut Bifidobacteria in Term Infants Fed an Infant Formula Containing High sn-2 Palmitate: A Cluster Randomized Clinical Trial" Nutrients 13, no. 2: 693. https://doi.org/10.3390/nu13020693
APA StyleWu, W., Zhao, A., Liu, B., Ye, W.-H., Su, H.-W., Li, J., & Zhang, Y.-M. (2021). Neurodevelopmental Outcomes and Gut Bifidobacteria in Term Infants Fed an Infant Formula Containing High sn-2 Palmitate: A Cluster Randomized Clinical Trial. Nutrients, 13(2), 693. https://doi.org/10.3390/nu13020693






