Creatine Supplementation and Brain Health
Abstract
1. Introduction
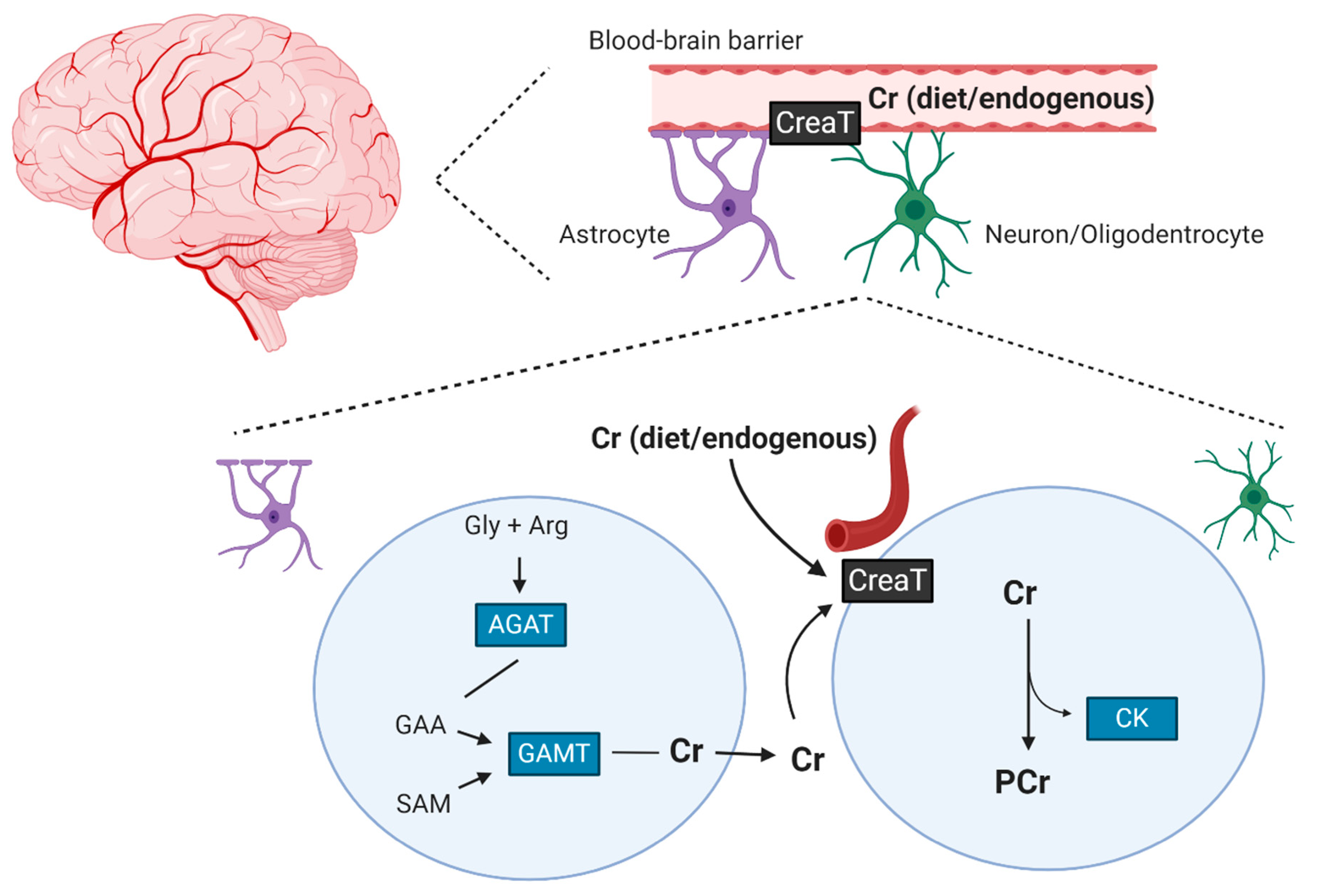
2. The Effects of Creatine Supplementation on Brain Creatine Levels
3. Creatine Supplementation and Cognition
4. Creatine Supplementation and Brain Injury, Concussion, and Hypoxia
5. Conclusions and Future Directions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gualano, B.; Roschel, H.; Lancha, A.H.; Brightbill, C.E.; Rawson, E.S.; Junior, A.H.L. In sickness and in health: The widespread application of creatine supplementation. Amino Acids 2011, 43, 519–529. [Google Scholar] [CrossRef]
- Dolan, E.; Artioli, G.G.; Pereira, R.M.R.; Gualano, B. Muscular Atrophy and Sarcopenia in the Elderly: Is There a Role for Creatine Supplementation? Biomolecules 2019, 9, 642. [Google Scholar] [CrossRef]
- Dolan, E.; Gualano, B.; Rawson, E.S. Beyond muscle: The effects of creatine supplementation on brain creatine, cognitive processing, and traumatic brain injury. Eur. J. Sport Sci. 2019, 19, 1–14. [Google Scholar] [CrossRef]
- Wallimann, T.; Wyss, M.; Brdiczka, D.; Nicolay, K.; Eppenberger, H.M. Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: The ‘phosphocreatine circuit’ for cellular energy homeostasis. Biochem. J. 1992, 281, 21–40. [Google Scholar] [CrossRef]
- Wallimann, T.; Turner, D.C.; Eppenberger, H.M. Localization of creatine kinase isoenzymes in myofibrils. I. Chicken skeletal muscle. J. Cell Biol. 1977, 75, 297–317. [Google Scholar] [CrossRef]
- Sahlin, K.; Harris, R.C. The creatine kinase reaction: A simple reaction with functional complexity. Amino Acids 2011, 40, 1363–1367. [Google Scholar] [CrossRef]
- Sestili, P.; Martinelli, C.; Colombo, E.; Barbieri, E.; Potenza, L.; Sartini, S.; Fimognari, C. Creatine as an antioxidant. Amino Acids 2011, 40, 1385–1396. [Google Scholar] [CrossRef] [PubMed]
- Beal, M.F. Neuroprotective effects of creatine. Amino Acids 2011, 40, 1305–1313. [Google Scholar] [CrossRef]
- Gualano, B.; Artioli, G.G.; Poortmans, J.R.; Junior, A.H.L. Exploring the therapeutic role of creatine supplementation. Amino Acids 2009, 38, 31–44. [Google Scholar] [CrossRef] [PubMed]
- Turner, C.E.; Byblow, W.D.; Gant, N. Creatine Supplementation Enhances Corticomotor Excitability and Cognitive Performance during Oxygen Deprivation. J. Neurosci. 2015, 35, 1773–1780. [Google Scholar] [CrossRef] [PubMed]
- Kaldis, P.; Hemmer, W.; Zanolla, E.; Holtzman, D.; Wallimann, T. ’Hot Spots’ of Creatine Kinase Localization in Brain: Cerebellum, Hippocampus and Choroid Plexus. Dev. Neurosci. 1996, 18, 542–554. [Google Scholar] [CrossRef] [PubMed]
- Salomons, G.S.; Van Dooren, S.J.M.; Verhoeven, N.M.; Marsden, D.; Schwartz, C.; Cecil, K.M.; Degrauw, T.J.; Jakobs, C. X-linked creatine transporter defect: An overview. J. Inherit. Metab. Dis. 2003, 26, 309–318. [Google Scholar] [CrossRef]
- Stockler, S.; Holzbach, U.; Hanefeld, F.; Marquardt, I.; Helms, G.; Requart, M.; Hanicke, W.; Frahm, J. Creatine Deficiency in the Brain: A New, Treatable Inborn Error of Metabolism. Pediatr. Res. 1994, 36, 409–413. [Google Scholar] [CrossRef]
- Stockler, S.; Schutz, P.W.; Salomons, G.S. Cerebral creatine deficiency syndromes: Clinical aspects, treatment and pathophysiology. Alzheimer’s Dis. 2007, 46, 149–166. [Google Scholar] [CrossRef]
- Benton, D.; Donohoe, R. The influence of creatine supplementation on the cognitive functioning of vegetarians and omnivores. Br. J. Nutr. 2010, 105, 1100–1105. [Google Scholar] [CrossRef] [PubMed]
- McMorris, T.; Mielcarz, G.; Harris, R.C.; Swain, J.P.; Howard, A.N. Creatine Supplementation and Cognitive Performance in Elderly Individuals. Aging Neuropsychol. Cogn. 2007, 14, 517–528. [Google Scholar] [CrossRef] [PubMed]
- Rae, C.D.; Bröer, S. Creatine as a booster for human brain function. How might it work? Neurochem. Int. 2015, 89, 249–259. [Google Scholar] [CrossRef] [PubMed]
- Avgerinos, K.I.; Spyrou, N.; Bougioukas, K.I.; Kapogiannis, D. Effects of creatine supplementation on cognitive function of healthy individuals: A systematic review of randomized controlled trials. Exp. Gerontol. 2018, 108, 166–173. [Google Scholar] [CrossRef]
- Walker, J.B. Creatine: Biosynthesis, regulation, and function. Adv. Enzymol. Relat. Areas Mol. Biol. 1979, 50, 177–242. [Google Scholar] [PubMed]
- Braissant, O.; Bachmann, C.; Henry, H. Expression and function of AGAT, GAMT and CT1 in the mammalian brain. Alzheimer’s Dis. 2007, 46, 67–81. [Google Scholar] [CrossRef]
- Solis, M.Y.; Painelli, V.D.S.; Artioli, G.G.; Roschel, H.; Otaduy, M.C.; Gualano, B. Brain creatine depletion in vegetarians? A cross-sectional 1H-magnetic resonance spectroscopy (1H-MRS) study. Br. J. Nutr. 2013, 111, 1272–1274. [Google Scholar] [CrossRef]
- Stockler-Ipsiroglu, S.; Van Karnebeek, C.D.M.; Longo, N.; Korenke, G.C.; Mercimek-Mahmutoglu, S.; Marquart, I.; Barshop, B.; Grolik, C.; Schlune, A.; Angle, B.; et al. Guanidinoacetate methyltransferase (GAMT) deficiency: Outcomes in 48 individuals and recommendations for diagnosis, treatment and monitoring. Mol. Genet. Metab. 2014, 111, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Laakso, M.P.; Hiltunen, Y.; Könönen, M.; Kivipelto, M.; Koivisto, A.; Hallikainen, M.; Soininen, H. Decreased brain creatine levels in elderly apolipoprotein E epsilon 4 carriers. J. Neural Transm. 2003, 110, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Solis, M.Y.; Artioli, G.G.; Otaduy, M.C.G.; Leite, C.D.C.; Arruda, W.; Veiga, R.R.; Gualano, B. Effect of age, diet, and tissue type on PCr response to creatine supplementation. J. Appl. Physiol. 2017, 123, 407–414. [Google Scholar] [CrossRef]
- Rawson, E.S.; Venezia, A.C. Use of creatine in the elderly and evidence for effects on cognitive function in young and old. Amino Acids 2011, 40, 1349–1362. [Google Scholar] [CrossRef] [PubMed]
- Kreider, R.B.; Kalman, D.S.; Antonio, J.; Ziegenfuss, T.N.; Wildman, R.; Collins, R.; Candow, D.G.; Kleiner, S.M.; Almada, A.L.; Lopez, H.L. International Society of Sports Nutrition position stand: Safety and efficacy of creatine supplementation in exercise, sport, and medicine. J. Int. Soc. Sports Nutr. 2017, 14, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Dechent, P.; Pouwels, P.J.W.; Wilken, B.; Hanefeld, F.; Frahm, J. Increase of total creatine in human brain after oral supplementation of creatine-monohydrate. Am. J. Physiol. Content 1999, 277, R698–R704. [Google Scholar] [CrossRef]
- Merege-Filho, C.A.A.; Otaduy, M.C.G.; De Sá-Pinto, A.L.; De Oliveira, M.O.; Gonçalves, L.D.S.; Hayashi, A.P.T.; Roschel, H.; Pereira, R.M.R.; Silva, C.A.; Brucki, S.M.D.; et al. Does brain creatine content rely on exogenous creatine in healthy youth? A proof-of-principle study. Appl. Physiol. Nutr. Metab. 2017, 42, 128–134. [Google Scholar] [CrossRef] [PubMed]
- Béard, E.; Braissant, O. Synthesis and transport of creatine in the CNS: Importance for cerebral functions. J. Neurochem. 2010, 115, 297–313. [Google Scholar] [CrossRef]
- Wilkinson, I.D.; Mitchel, N.; Breivik, S.; Greenwood, P.; Griffiths, P.D.; Winter, E.M.; Van Beek, E.J.R. Effects of Creatine Supplementation on Cerebral White Matter in Competitive Sportsmen. Clin. J. Sport Med. 2006, 16, 63–67. [Google Scholar] [CrossRef]
- Ostojić, S.M.; Ostojić, J.; Drid, P.; Vraneš, M. Guanidinoacetic acid versus creatine for improved brain and muscle creatine levels: A superiority pilot trial in healthy men. Appl. Physiol. Nutr. Metab. 2016, 41, 1005–1007. [Google Scholar] [CrossRef]
- Tachikawa, M.; Hosoya, K.-I. Transport characteristics of guanidino compounds at the blood-brain barrier and blood-cerebrospinal fluid barrier: Relevance to neural disorders. Fluids Barriers CNS 2011, 8, 13. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.R.R.; Filho, C.A.A.M.; Benatti, F.B.; Brucki, S.M.D.; Pereira, R.M.R.; Pinto, A.L.D.S.; Lima, F.R.; Roschel, H.; Gualano, B. Creatine Supplementation Associated or Not with Strength Training upon Emotional and Cognitive Measures in Older Women: A Randomized Double-Blind Study. PLoS ONE 2013, 8, e76301. [Google Scholar] [CrossRef]
- Rawson, E.S.; Lieberman, H.R.; Walsh, T.M.; Zuber, S.M.; Harhart, J.M.; Matthews, T.C. Creatine supplementation does not improve cognitive function in young adults. Physiol. Behav. 2008, 95, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Hammett, S.T.; Wall, M.B.; Edwards, T.C.; Smith, A. Dietary supplementation of creatine monohydrate reduces the human fMRI BOLD signal. Neurosci. Lett. 2010, 479, 201–205. [Google Scholar] [CrossRef] [PubMed]
- Ling, J.; Kritikos, M.; Tiplady, B. Cognitive effects of creatine ethyl ester supplementation. Behav. Pharmacol. 2009, 20, 673–679. [Google Scholar] [CrossRef]
- McMorris, T.; Harris, R.; Howard, A.; Langridge, G.; Hall, B.; Corbett, J.; Dicks, M.; Hodgson, C. Creatine supplementation, sleep deprivation, cortisol, melatonin and behavior. Physiol. Behav. 2007, 90, 21–28. [Google Scholar] [CrossRef] [PubMed]
- McMorris, T.; Harris, R.C.; Swain, J.; Corbett, J.; Collard, K.; Dyson, R.J.; Dye, L.; Hodgson, C.; Draper, N. Effect of creatine supplementation and sleep deprivation, with mild exercise, on cognitive and psychomotor performance, mood state, and plasma concentrations of catecholamines and cortisol. Psychopharmacology 2006, 185, 93–103. [Google Scholar] [CrossRef] [PubMed]
- Rae, C.D.; Digney, A.L.; McEwan, S.R.; Bates, T.C. Oral creatine monohydrate supplementation improves brain performance: A double–blind, placebo–controlled, cross–over trial. Proc. R. Soc. B Biol. Sci. 2003, 270, 2147–2150. [Google Scholar] [CrossRef]
- Watanabe, A.; Kato, N.; Kato, T. Effects of creatine on mental fatigue and cerebral hemoglobin oxygenation. Neurosci. Res. 2002, 42, 279–285. [Google Scholar] [CrossRef]
- Borchio, L.; Machek, S.B.; Machado, M. Supplemental creatine monohydrate loading improves cognitive function in experienced mountain bikers. J. Sports Med. Phys. Fit. 2020, 60, 1168–1170. [Google Scholar] [CrossRef]
- Cook, C.J.; Crewther, B.T.; Kilduff, L.P.; Drawer, S.; Gaviglio, C.M. Skill execution and sleep deprivation: Effects of acute caffeine or creatine supplementation—A randomized placebo-controlled trial. J. Int. Soc. Sports Nutr. 2011, 8, 2. [Google Scholar] [CrossRef] [PubMed]
- Smolarek, A.C.; McAnulty, S.R.; Ferreira, L.H.; Cordeiro, G.R.; Alessi, A.; Rebesco, D.B.; Honorato, I.C.; Laat, E.F.; Mascarenhas, L.P.; Souza-Junior, T.P. Effect of 16 Weeks of Strength Training and Creatine Supplementation on Strength and Cognition in Older Adults: A Pilot Study. J. Exerc. Physiol. Online 2020, 23, 88–94. [Google Scholar]
- Van Cutsem, J.; Roelands, B.; Pluym, B.; Tassignon, B.; Verschueren, J.O.; De Pauw, K.; Meeusen, R. Can Creatine Combat the Mental Fatigue-associated Decrease in Visuomotor Skills? Med. Sci. Sports Exerc. 2020, 52, 120–130. [Google Scholar] [CrossRef]
- Meeusen, R. Exercise, Nutrition and the Brain. Sports Med. 2014, 44, 47–56. [Google Scholar] [CrossRef]
- Cox, G.; Mujika, I.; Tumilty, D.; Burke, L. Acute Creatine Supplementation and Performance during a Field Test Simulating Match Play in Elite Female Soccer Players. Int. J. Sport Nutr. Exerc. Metab. 2002, 12, 33–46. [Google Scholar] [CrossRef]
- Mohebbi, H.; Rahnama, N.; Moghadassi, M.; Ranjbar, K. Effect of Creatine Supplementation on Sprint and Skill Performance in Young Soccer Players. Middle-East J. Sci. Res. 2012, 12, 397–401. [Google Scholar]
- Dean, P.J.A.; Arikan, G.; Opitz, B.; Sterr, A. Potential for use of creatine supplementation following mild traumatic brain injury. Concussion 2017, 2, CNC34. [Google Scholar] [CrossRef]
- Vagnozzi, R.; Signoretti, S.; Floris, R.; Marziali, S.; Manara, M.; Amorini, A.M.; Belli, A.; Di Pietro, V.; D’Urso, S.; Pastore, F.S.; et al. Decrease in N-Acetylaspartate Following Concussion May Be Coupled to Decrease in Creatine. J. Head Trauma Rehabil. 2013, 28, 284–292. [Google Scholar] [CrossRef]
- Alosco, M.L.; Tripodis, Y.; Rowland, B.; Chua, A.S.; Liao, H.; Martin, B.; Jarnagin, J.; Chaisson, C.E.; Pasternak, O.; Karmacharya, S.; et al. A magnetic resonance spectroscopy investigation in symptomatic former NFL players. Brain Imaging Behav. 2020, 14, 1419–1429. [Google Scholar] [CrossRef] [PubMed]
- Barrett, E.C.; McBurney, M.I.; Ciappio, E.D. Omega-3 fatty acid supplementation as a potential therapeutic aid for the recovery from mild traumatic brain injury/concussion. Adv. Nutr 2014, 5, 268–277. [Google Scholar] [CrossRef]
- Sullivan, P.G.; Geiger, J.D.; Mattson, M.P.; Scheff, S.W. Dietary supplement creatine protects against traumatic brain injury. Ann. Neurol. 2000, 48, 723–729. [Google Scholar] [CrossRef]
- Ipsiroglu, O.S.; Stromberger, C.; Ilas, J.; Höger, H.; Mühl, A.; Stöckler-Ipsiroglu, S. Changes of tissue creatine concentrations upon oral supplementation of creatine-monohydrate in various animal species. Life Sci. 2001, 69, 1805–1815. [Google Scholar] [CrossRef]
- Sakellaris, G.; Kotsiou, M.; Tamiolaki, M.; Kalostos, G.; Tsapaki, E.; Spanaki, M.; Spilioti, M.; Charissis, G.; Evangeliou, A. Prevention of Complications Related to Traumatic Brain Injury in Children and Adolescents with Creatine Administration: An Open Label Randomized Pilot Study. J. Trauma: Inj. Infect. Crit. Care 2006, 61, 322–329. [Google Scholar] [CrossRef]
- Sakellaris, G.; Nasis, G.; Kotsiou, M.; Tamiolaki, M.; Charissis, G.; Evangeliou, A. Prevention of traumatic headache, dizziness and fatigue with creatine administration. A pilot study. Acta Paediatr. 2007, 97, 31–34. [Google Scholar] [CrossRef] [PubMed]
- Gualano, B.; Painelli, V.D.S.; Roschel, H.; Lugaresi, R.; Dorea, E.; Artioli, G.G.; Lima, F.R.; Da Silva, M.E.R.; Cunha, M.R.; Seguro, A.C.; et al. Creatine supplementation does not impair kidney function in type 2 diabetic patients: A randomized, double-blind, placebo-controlled, clinical trial. Graefe’s Arch. Clin. Exp. Ophthalmol. 2010, 111, 749–756. [Google Scholar] [CrossRef]
- Gualano, B.; Ferreira, D.C.; Sapienza, M.T.; Seguro, A.C.; Lancha, A.H. Effect of Short-term High-Dose Creatine Supplementation on Measured GFR in a Young Man with a Single Kidney. Am. J. Kidney Dis. 2010, 55, e7–e9. [Google Scholar] [CrossRef] [PubMed]
- Gualano, B.; Ugrinowitsch, C.; Novaes, R.B.; Artioli, G.G.; Shimizu, M.H.; Seguro, A.C.; Harris, R.C.; Lancha, A.H. Effects of creatine supplementation on renal function: A randomized, double-blind, placebo-controlled clinical trial. Graefe’s Arch. Clin. Exp. Ophthalmol. 2008, 103, 33–40. [Google Scholar] [CrossRef]
- Hayashi, A.P.; Solis, M.Y.; Sapienza, M.T.; Otaduy, M.C.G.; de Sa Pinto, A.L.; Silva, C.A.; Sallum, A.M.E.; Pereira, R.M.R.; Gualano, B. Efficacy and safety of creatine supplementation in childhood-onset systemic lupus erythematosus: A randomized, double-blind, placebo-controlled, crossover trial. Lupus 2014, 23, 1500–1511. [Google Scholar] [CrossRef]
- Lugaresi, R.; Leme, M.; Painelli, V.D.S.; Murai, I.H.; Roschel, H.; Sapienza, M.T.; Lancha, A.H.J.; Gualano, B. Does long-term creatine supplementation impair kidney function in resistance-trained individuals consuming a high-protein diet? J. Int. Soc. Sports Nutr. 2013, 10, 26. [Google Scholar] [CrossRef]
- Neves, M.; Gualano, B.; Roschel, H.; Lima, F.R.; De Sá-Pinto, A.L.; Seguro, A.C.; Shimizu, M.H.; Sapienza, M.T.; Fuller, R.; Lancha, A.H.; et al. Effect of creatine supplementation on measured glomerular filtration rate in postmenopausal women. Appl. Physiol. Nutr. Metab. 2011, 36, 419–422. [Google Scholar] [CrossRef]
- Rawson, E.S.; Clarkson, P.M.; Tarnopolsky, M.A. Perspectives on Exertional Rhabdomyolysis. Sports Med. 2017, 47, 33–49. [Google Scholar] [CrossRef] [PubMed]
- Solis, M.Y.; Hayashi, A.P.; Artioli, G.G.; Roschel, H.; Sapienza, M.T.; Otaduy, M.C.; De Sã Pinto, A.L.; Silva, C.A.; Sallum, A.M.E.; Pereira, R.M.R.; et al. Efficacy and safety of creatine supplementation in juvenile dermatomyositis: A randomized, double-blind, placebo-controlled crossover trial. Muscle Nerve 2015, 53, 58–66. [Google Scholar] [CrossRef] [PubMed]
| Population | Creatine Supplementation Protocol | Cognitive Tests (CT) Outcomes (O) | Reference |
|---|---|---|---|
| Healthy older women | 20 g/day + 5 g/day for 24 weeks | CT: Mini-mental state examination, stroop, trail making, digit span, delay recall test and the short version of the geriatric depression scale O: No change | Alves et al. (2013) [33] |
| Semiprofessional, non-vegetarian, male mountain bikers | 20 g/day for 7 days | CT: Simple and choice reaction time, differentiation task test, Eiksen flanker test and Corsi block test O: Creatine increased performance in choice reaction time, Eiksen flanker test and Corsi block test. | Borchio et al. (2020) [41] |
| Healthy young women (vegetarian and meat-eaters) | 20 g/day for 5 days | CT: Word recall, simple and choice reaction time, rapid visual information processing and controlled oral word association test O: Word recall test performance was reduced in meat-eater after creatine supplementation (within-group comparison). Post supplementation performance was higher in vegetarians than in meat-eaters. | Benton and Donohoe (2011) [15] |
| Professional male rugby players who were sleep-deprived (3–5 h) | 0.05 or 0.1 g/kg/bw for 1 day | CT: Rugby passing skill test O: Sleep deprivation reduced passing accuracy and creatine reversed this effect (trend for greater effect with larger dose). | Cook et al. (2011) [42] |
| Healthy young adults | 20 g/day for 5 days + 5 g/day for 2 days | CT: Backward digit span test and ravens advanced progressive matrices. O: Backward digit span performance was increased after creatine. | Hammett et al. (2010) [35] |
| Healthy young men and women | 5 g/day for 15 days | CT: Memory scanning, number-pair matching, sustained attention, arrow flankers and IQ test O: Aspect of improvement was reported in all the cognitive tests performed in the creatine group. | Ling et al. (2009) [36] |
| Healthy young men and women who were sleep-deprived (24 h) | 20 g/day for 7 days | CT: Random number generation, forward and backward recall, visual reaction time, static balance and mood state O: Performance reduction was attenuated in the creatine group for random movement generation, choice reaction time, balance and mood. | McMorris et al. (2006) [38] |
| Healthy elderly men and women | 20 g/day for 7 days | CT: Random number generation, forward and backward recall and long-term memory tests O: Forward number recall, forward and backward spatial recall and long-term memory performance were enhanced after creatine supplementation. | McMorris et al. (2007a) [16] |
| Healthy young men who were sleep-deprived (36 h) | 20 g/day for 7 days | CT: Random number generation, short-term number recall, visual reaction time, cognitive effort, dynamic balance test and mood state O: Performance on the random number generation test was improved following creatine. | McMorris et al. (2007b) [37] |
| Healthy male and female children | 0.3 g/kg/day for 7 days | CT: Stroop, Rey auditory verbal learning test, Raven progressive matrices and trail making test O: No change | Merege-Filho et al. (2017) [28] |
| Vegan and vegetarian healthy male and female young adults | 5 g/day for 6 weeks | CT: Ravens advanced progressive matrices and Wechsler auditory backward digit span task O: Creatine improved performance on the Raven’s test and the backward digit span task. | Rae and Broer (2015) [17] |
| Healthy male and female young adults | 0.03 g/kg/day for 6 weeks | CT: Automated neuropsychological assessment metrics O: No change | Rawson et al. (2008) [34] |
| Male and female institutionalized older adults (with full physical and mental capacities preserved) | 5 g/day for 16 weeks | CT: Montreal Cognitive Assessment (MoCA) questionnaire O: Creatine (plus resistance training) improved MoCA scores. | Smolarek et al. (2020) [43] |
| Healthy male and female young adults exposed to experimental hypoxia | 20 g/day for 7 days | CT: Neuropsychological test comprising verbal and visual memory, finger tapping, symbol digit coding stroop test, test of shifting attention, continuous performance test, alertness and peripheral and corticomotor excitability O: Creatine supplementation offset hypoxia-induced decrements in a number of cognitive tests. | Turner et al. (2015) [10] |
| Healthy male and female young adults exposed to mental fatigue (90 min Stoop task) | 20 g/day for 7 days | CT: Psychomotor performance (visuomotor task with Fitlight-hardware and software), strength endurance task, Flanker test, heart rate, blood glucose, success motivation and intrinsic motivation, mood, session ratings of perceived exertion and mental fatigue O: Accuracy throughout the 90 min Stroop task and strength endurance (in the non-dominant hand) were improved with creatine. No other effects of creatine supplementation were observed. | Van Cutsem et al. (2020) [44] |
| Healthy male and female young adults | 8 g/day for 5 days | CT: Serial calculation task (Uchida-Kraeplin) O: Both groups increased mean performance. Mental fatigue, assessed during the second half of the test, was increased in the creatine group only. | Watanabe et al. (2002) [40] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roschel, H.; Gualano, B.; Ostojic, S.M.; Rawson, E.S. Creatine Supplementation and Brain Health. Nutrients 2021, 13, 586. https://doi.org/10.3390/nu13020586
Roschel H, Gualano B, Ostojic SM, Rawson ES. Creatine Supplementation and Brain Health. Nutrients. 2021; 13(2):586. https://doi.org/10.3390/nu13020586
Chicago/Turabian StyleRoschel, Hamilton, Bruno Gualano, Sergej M. Ostojic, and Eric S. Rawson. 2021. "Creatine Supplementation and Brain Health" Nutrients 13, no. 2: 586. https://doi.org/10.3390/nu13020586
APA StyleRoschel, H., Gualano, B., Ostojic, S. M., & Rawson, E. S. (2021). Creatine Supplementation and Brain Health. Nutrients, 13(2), 586. https://doi.org/10.3390/nu13020586








