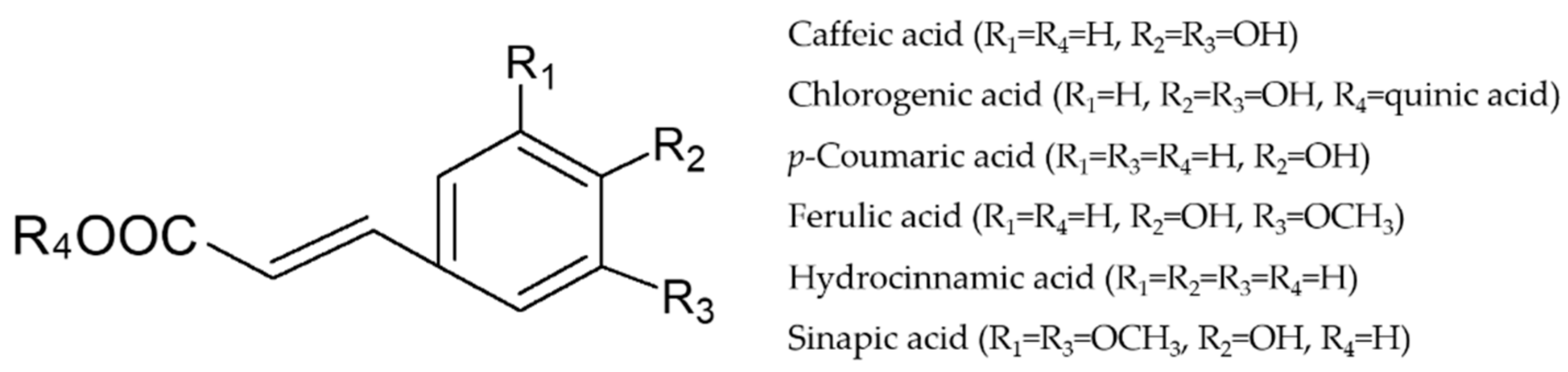
Caffeic Acid Modulates Processes Associated with Intestinal Inflammation
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Cell Culture Conditions
2.3. Cell Viability Assay
2.4. Measurement of PGE2 Biosynthesis in IL-1β-Stimulated Myofibroblasts by ELISA
2.5. Measurement of IL-1β-Induced COX-2 Expression by Western Blot
2.6. Measurement of Cytokine Production in IL-1β-Stimulated Myofibroblasts by ELISA
2.7. Inhibition of the Formation of the Advanced Glycation End Products (AGEs)
2.8. Angiotensin-I-Converting Enzyme Inhibitory Assay
2.9. Antioxidant Activity
2.10. Reducing Activity
2.11. Ferric-Reducing/Antioxidant Power (FRAP) Activity
2.12. Chelating Activity
2.13. Statistical Analysis
3. Results
3.1. Caffeic Acid Exerted No Cytotoxic Effects
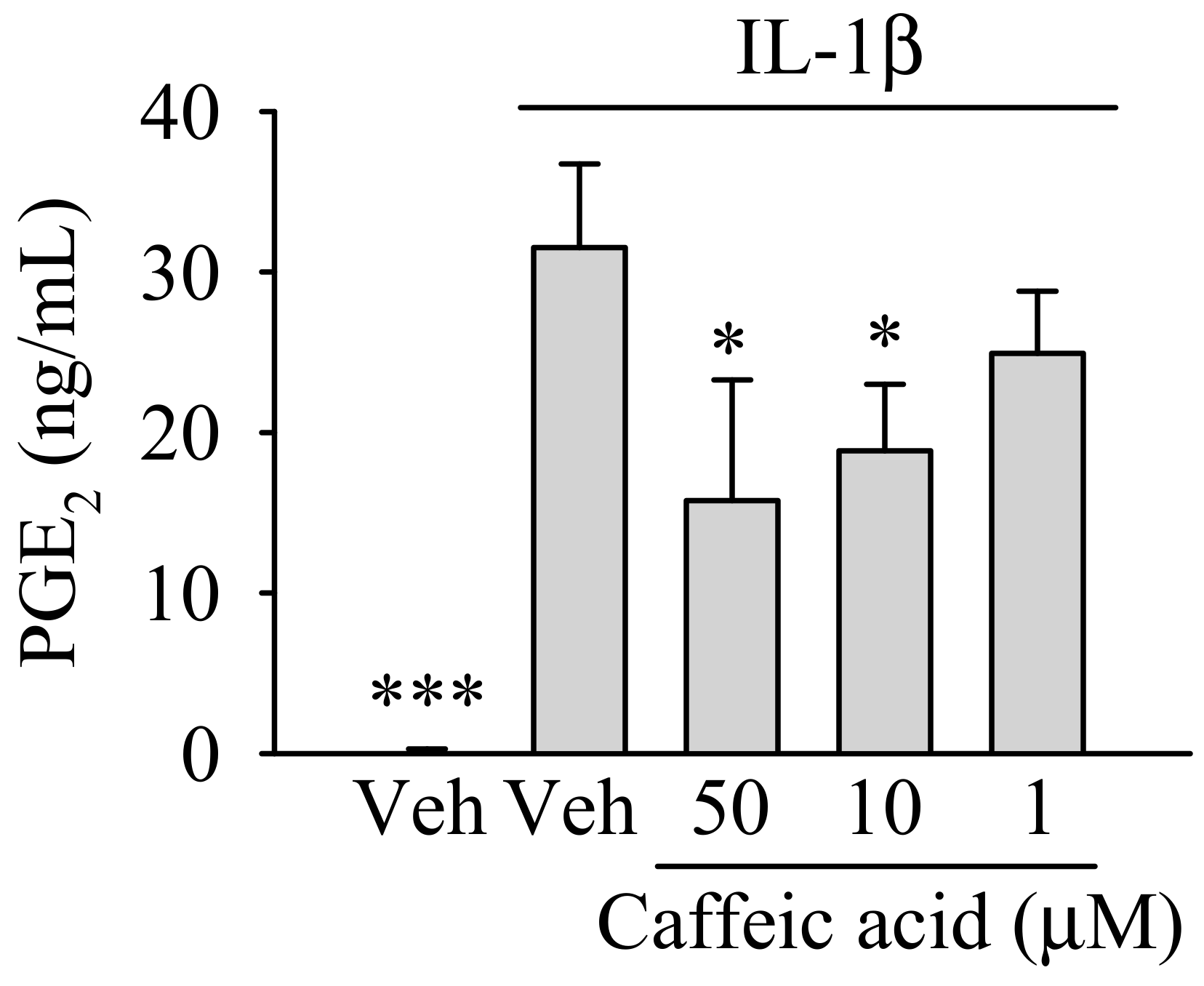
3.2. Caffeic Acid Ameliorates the Effect of IL-1β on the Biosynthesis of PGE in Myofibroblasts of the Colon, CCD-18Co
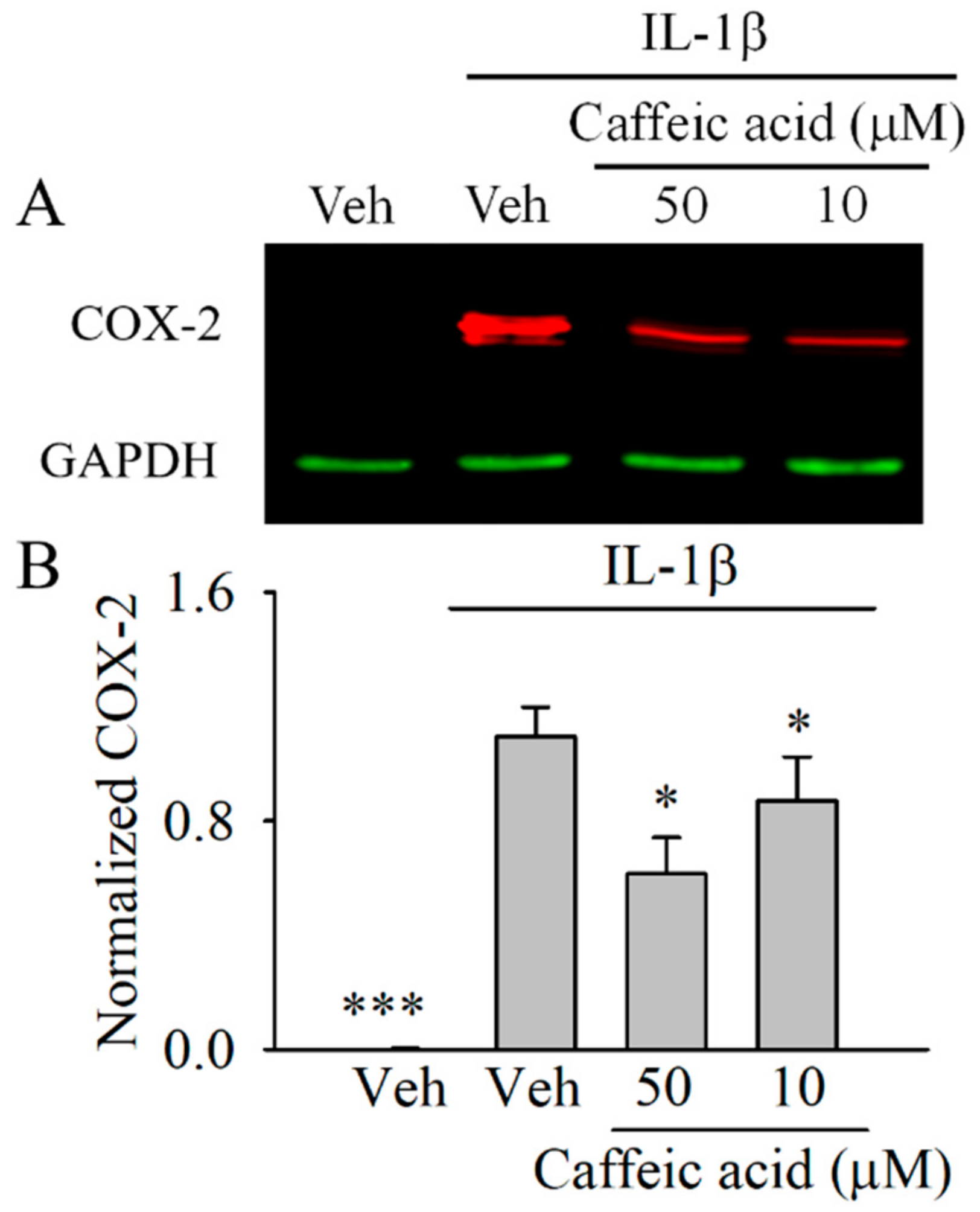
3.3. Caffeic Acid Downregulates the Expression of COX-2 in IL-1β-Stimulated Myofibroblasts of the Colon, CCD-18Co
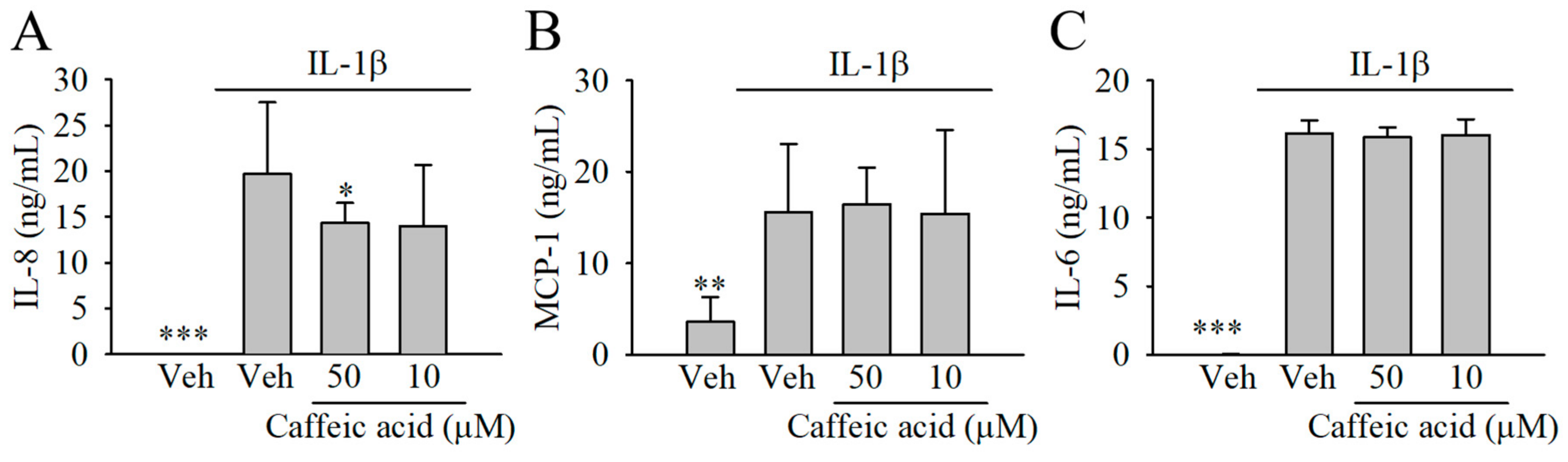
3.4. Caffeic Acid Lacked the Capacity to Modulate the Formation of Chemokines (IL-8 and MCP-1) and Cytokines (IL-6) in IL-1β-Stimulated Myofibroblasts of the Colon, CCD-18Co
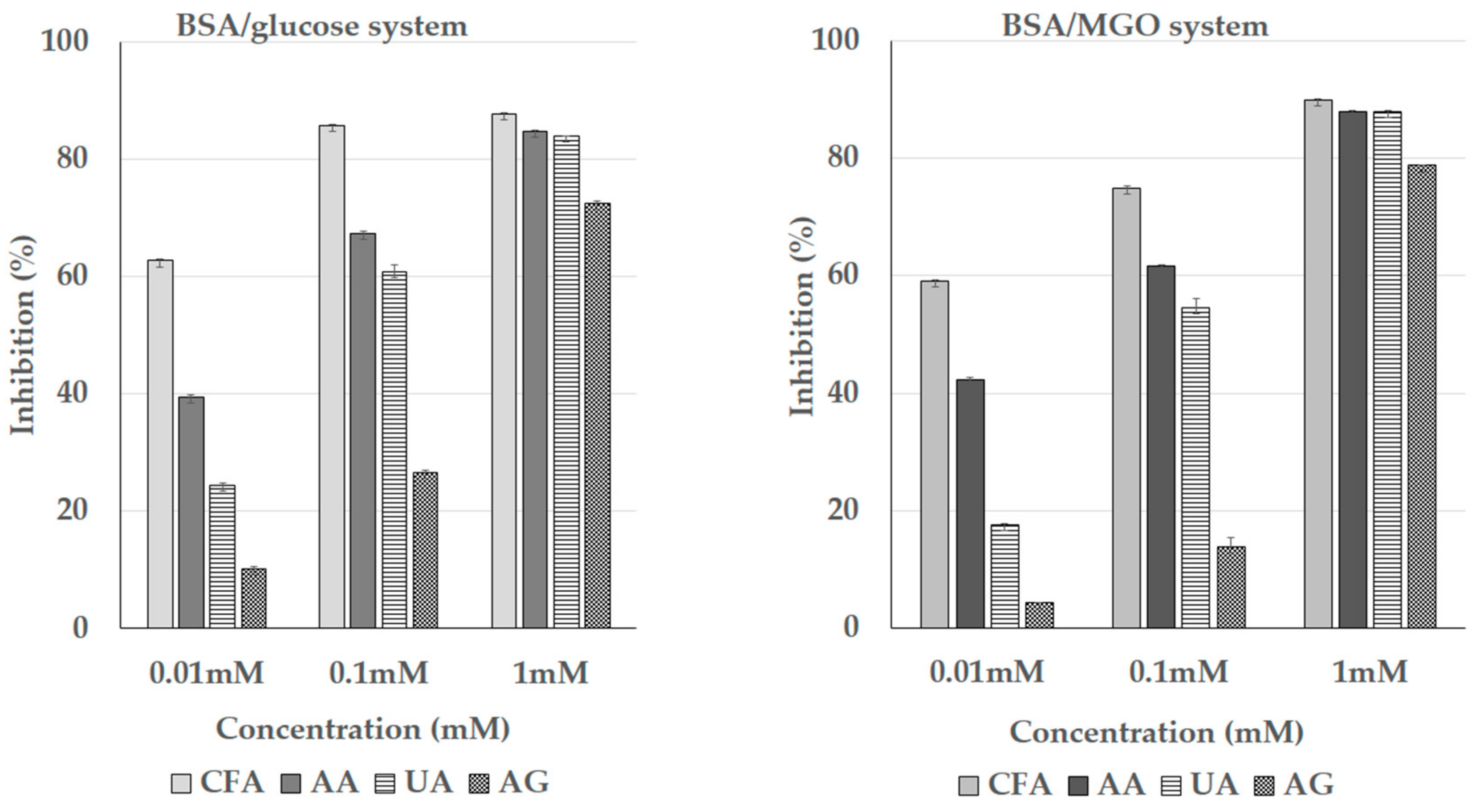
3.5. Inhibition of the Formation of the Advanced Glycation End Products (AGEs)
3.6. ACE Inhibitory Assay
3.7. The Antioxidant, Reducing, and Chelating Activity of Caffeic Acid
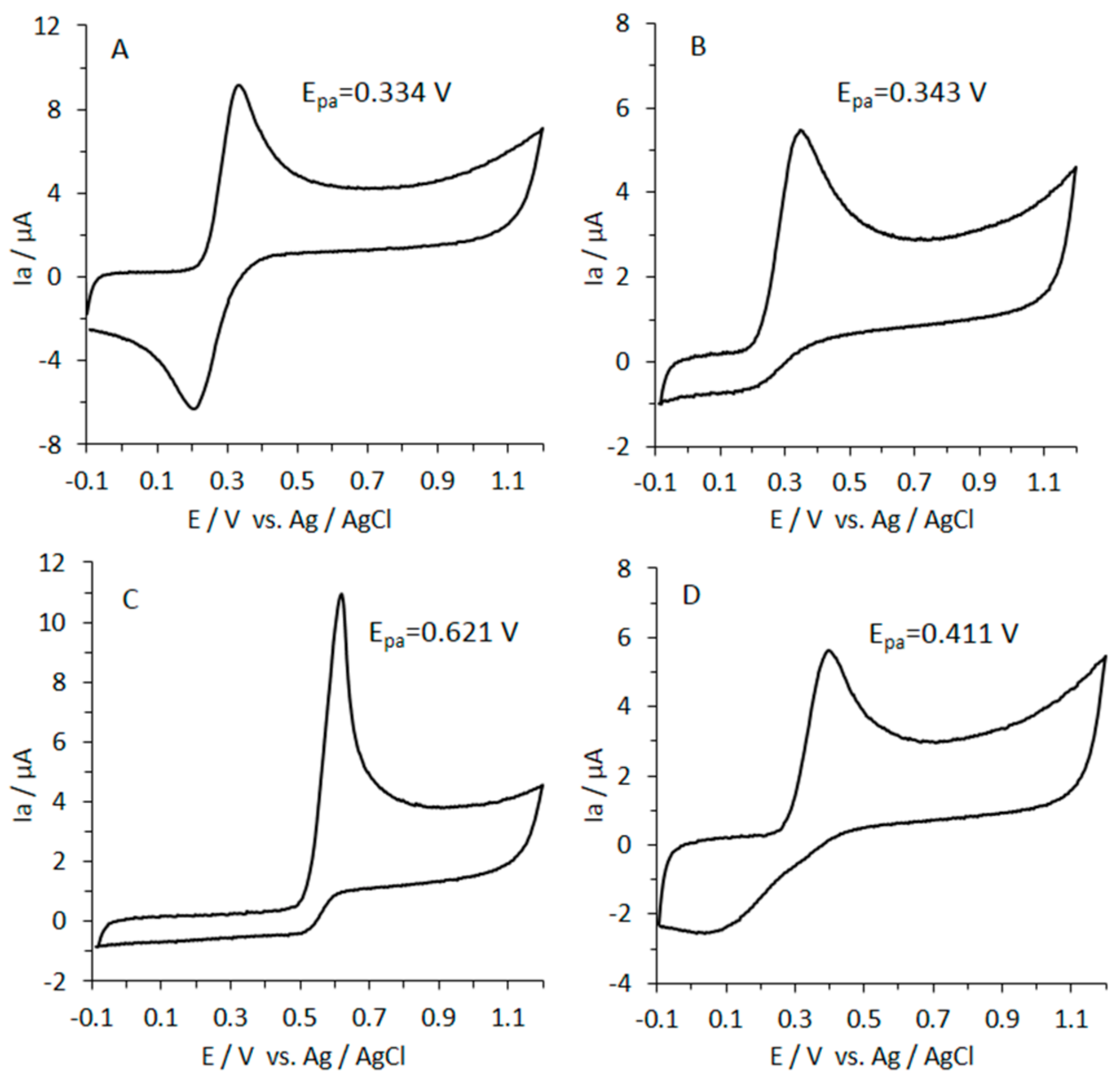
3.8. Reducing Power of Caffeic Acid by CV
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alatab, S.; Sepanlou, S.G.; Ikuta, K.; Vahedi, H.; Bisignano, C.; Safiri, S.; Sadeghi, A.; Nixon, M.R.; Abdoli, A.; Abolhassani, H.; et al. The Global, Regional, and National Burden of Inflammatory Bowel Disease in 195 Countries and Territories, 1990–2017: A Systematic Analysis for the Global Burden of Disease Study 2017. Lancet Gastroenterol. Hepatol. 2020, 5, 17–30. [Google Scholar] [CrossRef]
- Geicu, O.I.; Stanca, L.; Voicu, S.N.; Dinischiotu, A.; Bilteanu, L.; Serban, A.I.; Calu, V. Dietary AGEs Involvement in Colonic Inflammation and Cancer: Insights from an in Vitro Enterocyte Model. Sci. Rep. 2020, 10, 2754. [Google Scholar] [CrossRef]
- Neurath, M.F. Targeting Immune Cell Circuits and Trafficking in Inflammatory Bowel Disease. Nat. Immunol. 2019, 20, 970–979. [Google Scholar] [CrossRef]
- Speca, S.; Giusti, I.; Rieder, F.; Latella, G. Cellular and Molecular Mechanisms of Intestinal Fibrosis. World J. Gastroenterol. 2012, 18, 3635–3661. [Google Scholar] [CrossRef] [PubMed]
- Segal, J.P.; Htet, H.M.T.; Limdi, J.; Hayee, B. How to Manage IBD in the “Elderly”. Frontline Gastroenterol. 2020, 11, 468–477. [Google Scholar] [CrossRef]
- Xiao, H.-T.; Wen, B.; Shen, X.-C.; Bian, Z.-X. Potential of Plant-Sourced Phenols for Inflammatory Bowel Disease. Curr. Med. Chem. 2018, 25, 5191–5217. [Google Scholar] [CrossRef] [PubMed]
- El-Seedi, H.R.; El-Said, A.M.A.; Khalifa, S.A.M.; Göransson, U.; Bohlin, L.; Borg-Karlson, A.-K.; Verpoorte, R. Biosynthesis, Natural Sources, Dietary Intake, Pharmacokinetic Properties, and Biological Activities of Hydroxycinnamic Acids. J. Agric. Food Chem. 2012, 60, 10877–10895. [Google Scholar] [CrossRef]
- Tasson, L.; Canova, C.; Vettorato, M.G.; Savarino, E.; Zanotti, R. Influence of Diet on the Course of Inflammatory Bowel Disease. Dig. Dis. Sci. 2017, 62, 2087–2094. [Google Scholar] [CrossRef]
- Ng, S.C.; Tang, W.; Leong, R.W.; Chen, M.; Ko, Y.; Studd, C.; Niewiadomski, O.; Bell, S.; Kamm, M.A.; de Silva, H.J.; et al. Environmental Risk Factors in Inflammatory Bowel Disease: A Population-Based Case-Control Study in Asia-Pacific. Gut 2015, 64, 1063–1071. [Google Scholar] [CrossRef]
- Jenner, A.M.; Rafter, J.; Halliwell, B. Human Fecal Water Content of Phenolics: The Extent of Colonic Exposure to Aromatic Compounds. Free Radic. Biol. Med. 2005, 38, 763–772. [Google Scholar] [CrossRef]
- Clifford, M.N.; Kerimi, A.; Williamson, G. Bioavailability and Metabolism of Chlorogenic Acids (Acyl-Quinic Acids) in Humans. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1299–1352. [Google Scholar] [CrossRef]
- Ye, Z.; Liu, Z.; Henderson, A.; Lee, K.; Hostetter, J.; Wannemuehler, M.; Hendrich, S. Increased CYP4B1 MRNA Is Associated with the Inhibition of Dextran Sulfate Sodium-Induced Colitis by Caffeic Acid in Mice. Exp. Biol. Med. 2009, 234, 605–616. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Wu, X.; Cao, S.; Wang, L.; Wang, D.; Yang, H.; Feng, Y.; Wang, S.; Li, L. Caffeic Acid Ameliorates Colitis in Association with Increased Akkermansia Population in the Gut Microbiota of Mice. Oncotarget 2016, 7, 31790–31799. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi Ekbatan, S.; Li, X.-Q.; Ghorbani, M.; Azadi, B.; Kubow, S. Chlorogenic Acid and Its Microbial Metabolites Exert Anti-Proliferative Effects, S-Phase Cell-Cycle Arrest and Apoptosis in Human Colon Cancer Caco-2 Cells. Int. J. Mol. Sci. 2018, 19, 723. [Google Scholar] [CrossRef] [PubMed]
- Murad, L.D.; Da Costa Pereira Soares, N.; Brand, C.; Monteiro, M.C.; Teodoro, A.J. Effects of Caffeic and 5-Caffeoylquinic Acids on Cell Viability and Cellular Uptake in Human Colon Adenocarcinoma Cells. Nutr. Cancer 2015, 67, 532–542. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Shin, H.S.; Satsu, H.; Totsuka, M.; Shimizu, M. 5-Caffeoylquinic Acid and Caffeic Acid down-Regulate the Oxidative Stress- and TNF-Alpha-Induced Secretion of Interleukin-8 from Caco-2 Cells. J. Agric. Food Chem. 2008, 56, 3863–3868. [Google Scholar] [CrossRef] [PubMed]
- Shin, H.S.; Satsu, H.; Bae, M.-J.; Totsuka, M.; Shimizu, M. Catechol Groups Enable Reactive Oxygen Species Scavenging-Mediated Suppression of PKD-NFkappaB-IL-8 Signaling Pathway by Chlorogenic and Caffeic Acids in Human Intestinal Cells. Nutrients 2017, 9, 165. [Google Scholar] [CrossRef] [PubMed]
- Andoh, A.; Bamba, S.; Brittan, M.; Fujiyama, Y.; Wright, N.A. Role of Intestinal Subepithelial Myofibroblasts in Inflammation and Regenerative Response in the Gut. Pharmacol. Ther. 2007, 114, 94–106. [Google Scholar] [CrossRef]
- Giménez-Bastida, J.A.; Zielinski, H.; Piskula, M.; Zielinska, D.; Szawara-Nowak, D. Buckwheat Bioactive Compounds, Their Derived Phenolic Metabolites and Their Health Benefits. Mol. Nutr. Food Res. 2017, 61. [Google Scholar] [CrossRef] [PubMed]
- Zielinska, D.; Laparra-Llopis, J.M.; Zielinski, H.; Szawara-Nowak, D.; Giménez-Bastida, J.A. Role of Apple Phytochemicals, Phloretin and Phloridzin, in Modulating Processes Related to Intestinal Inflammation. Nutrients 2019, 11, 1173. [Google Scholar] [CrossRef]
- Giménez-Bastida, J.A.; Larrosa, M.; González-Sarrías, A.; Tomás-Barberán, F.; Espín, J.C.; García-Conesa, M.-T. Intestinal Ellagitannin Metabolites Ameliorate Cytokine-Induced Inflammation and Associated Molecular Markers in Human Colon Fibroblasts. J. Agric. Food Chem. 2012, 60, 8866–8876. [Google Scholar] [CrossRef]
- Thornalley, P.J. Use of Aminoguanidine (Pimagedine) to Prevent the Formation of Advanced Glycation Endproducts. Arch. Biochem. Biophys. 2003, 419, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Sentandreu, M.Á.; Toldrá, F. A Rapid, Simple and Sensitive Fluorescence Method for the Assay of Angiotensin-I Converting Enzyme. Food Chem. 2006, 97, 546–554. [Google Scholar] [CrossRef]
- Franke, A.A.; Custer, L.J.; Arakaki, C.; Murphy, S.P. Vitamin C and Flavonoid Levels of Fruits and Vegetables Consumed in Hawaii. J. Food Compos. Anal. 2004, 17, 1–35. [Google Scholar] [CrossRef]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C. Use of a Free Radical Method to Evaluate Antioxidant Activity. LWT Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Zielińska, D.; Zieliński, H. Antioxidant Activity of Flavone C-Glucosides Determined by Updated Analytical Strategies. Food Chem. 2011, 124, 672–678. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Strain, J.J. [2] Ferric reducing/antioxidant power assay: Direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. In Methods in Enzymology; Oxidants and Antioxidants Part A; Academic Press: Cambridge, MA, USA, 1999; Volume 299, pp. 15–27. [Google Scholar]
- Mladenka, P.; Zatloukalová, L.; Filipský, T.; Hrdina, R. Cardiovascular Effects of Flavonoids Are Not Caused Only by Direct Antioxidant Activity. Free Radic. Biol. Med. 2010, 49, 963–975. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Soendergaard, C.; Bergenheim, F.H.; Aronoff, D.M.; Milne, G.; Riis, L.B.; Seidelin, J.B.; Jensen, K.B.; Nielsen, O.H. COX-2-PGE2 Signaling Impairs Intestinal Epithelial Regeneration and Associates with TNF Inhibitor Responsiveness in Ulcerative Colitis. EBioMedicine 2018, 36, 497–507. [Google Scholar] [CrossRef] [PubMed]
- Ferrer, R.; Moreno, J.J. Role of Eicosanoids on Intestinal Epithelial Homeostasis. Biochem. Pharmacol. 2010, 80, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Drew, D.A.; Cao, Y.; Chan, A.T. Aspirin and Colorectal Cancer: The Promise of Precision Chemoprevention. Nat. Rev. Cancer 2016, 16, 173–186. [Google Scholar] [CrossRef]
- Shin, K.-M.; Kim, I.-T.; Park, Y.-M.; Ha, J.; Choi, J.-W.; Park, H.-J.; Lee, Y.S.; Lee, K.-T. Anti-Inflammatory Effect of Caffeic Acid Methyl Ester and Its Mode of Action through the Inhibition of Prostaglandin E2, Nitric Oxide and Tumor Necrosis Factor-Alpha Production. Biochem. Pharmacol. 2004, 68, 2327–2336. [Google Scholar] [CrossRef]
- Yang, W.S.; Jeong, D.; Yi, Y.-S.; Park, J.G.; Seo, H.; Moh, S.H.; Hong, S.; Cho, J.Y. IRAK1/4-Targeted Anti-Inflammatory Action of Caffeic Acid. Mediat. Inflamm. 2013, 2013, 518183. [Google Scholar] [CrossRef]
- González-Sarrías, A.; Larrosa, M.; Tomás-Barberán, F.A.; Dolara, P.; Espín, J.C. NF-KappaB-Dependent Anti-Inflammatory Activity of Urolithins, Gut Microbiota Ellagic Acid-Derived Metabolites, in Human Colonic Fibroblasts. Br. J. Nutr. 2010, 104, 503–512. [Google Scholar] [CrossRef] [PubMed]
- Búfalo, M.C.; Sforcin, J.M. The Modulatory Effects of Caffeic Acid on Human Monocytes and Its Involvement in Propolis Action. J. Pharm. Pharmacol. 2015, 67, 740–745. [Google Scholar] [CrossRef] [PubMed]
- Jun, Y.K.; Kwon, S.H.; Yoon, H.T.; Park, H.; Soh, H.; Lee, H.J.; Im, J.P.; Kim, J.S.; Kim, J.W.; Koh, S.-J. Toll-like Receptor 4 Regulates Intestinal Fibrosis via Cytokine Expression and Epithelial-Mesenchymal Transition. Sci. Rep. 2020, 10, 19867. [Google Scholar] [CrossRef]
- Cotton, J.A.; Platnich, J.M.; Muruve, D.A.; Jijon, H.B.; Buret, A.G.; Beck, P.L. Interleukin-8 in Gastrointestinal Inflammation and Malignancy: Induction and Clinical Consequences. Available online: https://www.dovepress.com/interleukin-8-in-gastrointestinal-inflammation-and-malignancy-inductio-peer-reviewed-article-IJICMR (accessed on 8 December 2020).
- Yu, Y.; Chadee, K. Prostaglandin E2 Stimulates IL-8 Gene Expression in Human Colonic Epithelial Cells by a Posttranscriptional Mechanism. J. Immunol. 1998, 161, 3746–3752. [Google Scholar]
- Cho, J.-S.; Han, I.-H.; Lee, H.R.; Lee, H.-M. Prostaglandin E2 Induces IL-6 and IL-8 Production by the EP Receptors/Akt/NF-ΚB Pathways in Nasal Polyp-Derived Fibroblasts. Allergy Asthma Immunol. Res. 2014, 6, 449–457. [Google Scholar] [CrossRef] [PubMed]
- Weber, L.; Hammoud Mahdi, D.; Jankuhn, S.; Lipowicz, B.; Vissiennon, C. Bioactive Plant Compounds in Coffee Charcoal (Coffeae Carbo) Extract Inhibit Cytokine Release from Activated Human THP-1 Macrophages. Molecules 2019, 24, 4263. [Google Scholar] [CrossRef] [PubMed]
- Sueyoshi, R.; Ignatoski, K.M.W.; Daignault, S.; Okawada, M.; Teitelbaum, D.H. Angiotensin Converting Enzyme-Inhibitor Reduces Colitis Severity in an IL-10 Knockout Model. Dig. Dis. Sci. 2013, 58, 3165–3177. [Google Scholar] [CrossRef]
- Jacobs, J.D.; Wagner, T.; Gulotta, G.; Liao, C.; Li, Y.C.; Bissonnette, M.; Pekow, J. Impact of Angiotensin II Signaling Blockade on Clinical Outcomes in Patients with Inflammatory Bowel Disease. Dig. Dis. Sci. 2019, 64, 1938–1944. [Google Scholar] [CrossRef] [PubMed]
- Actis-Goretta, L.; Ottaviani, J.I.; Fraga, C.G. Inhibition of Angiotensin Converting Enzyme Activity by Flavanol-Rich Foods. J. Agric. Food Chem. 2006, 54, 229–234. [Google Scholar] [CrossRef] [PubMed]
- Bhullar, K.S.; Lassalle-Claux, G.; Touaibia, M.; Rupasinghe, H.P.V. Antihypertensive Effect of Caffeic Acid and Its Analogs through Dual Renin-Angiotensin-Aldosterone System Inhibition. Eur. J. Pharmacol. 2014, 730, 125–132. [Google Scholar] [CrossRef]
- van der Lugt, T.; Opperhuizen, A.; Bast, A.; Vrolijk, M.F. Dietary Advanced Glycation Endproducts and the Gastrointestinal Tract. Nutrients 2020, 12, 2814. [Google Scholar] [CrossRef]
- Navarro, M.; Morales, F.J. In Vitro Investigation on the Antiglycative and Carbonyl Trapping Activities of Hydroxytyrosol. Eur. Food Res. Technol. 2016, 242, 1101–1110. [Google Scholar] [CrossRef]
- Navarro, M.; Morales, F.J. Mechanism of Reactive Carbonyl Species Trapping by Hydroxytyrosol under Simulated Physiological Conditions. Food Chem. 2015, 175, 92–99. [Google Scholar] [CrossRef] [PubMed]
- Genaro-Mattos, T.C.; Maurício, Â.Q.; Rettori, D.; Alonso, A.; Hermes-Lima, M. Antioxidant Activity of Caffeic Acid against Iron-Induced Free Radical Generation—A Chemical Approach. PLoS ONE 2015, 10, e0129963. [Google Scholar] [CrossRef]
- Silva, F.A.; Borges, F.; Guimarães, C.; Lima, J.L.; Matos, C.; Reis, S. Phenolic Acids and Derivatives: Studies on the Relationship among Structure, Radical Scavenging Activity, and Physicochemical Parameters. J. Agric. Food Chem. 2000, 48, 2122–2126. [Google Scholar] [CrossRef] [PubMed]
- Son, S.; Lewis, B.A. Free Radical Scavenging and Antioxidative Activity of Caffeic Acid Amide and Ester Analogues: Structure-Activity Relationship. J. Agric. Food Chem. 2002, 50, 468–472. [Google Scholar] [CrossRef]
- García, P.; Romero, C.; Brenes, M.; Garrido, A. Effect of Metal Cations on the Chemical Oxidation of Olive O-Diphenols in Model Systems. J. Agric. Food Chem. 1996, 44, 2101–2105. [Google Scholar] [CrossRef]
- Ávila-Gálvez, M.Á.; González-Sarrías, A.; Espín, J.C. In Vitro Research on Dietary Polyphenols and Health: A Call of Caution and a Guide on How To Proceed. J. Agric. Food Chem. 2018, 66, 7857–7858. [Google Scholar] [CrossRef]
- Koshihara, Y.; Neichi, T.; Murota, S.; Lao, A.; Fujimoto, Y.; Tatsuno, T. Caffeic Acid Is a Selective Inhibitor for Leukotriene Biosynthesis. Biochim. Biophys. Acta 1984, 792, 92–97. [Google Scholar] [PubMed]
- Giménez-Bastida, J.A.; Shibata, T.; Uchida, K.; Schneider, C. Roles of 5-Lipoxygenase and Cyclooxygenase-2 in the Biosynthesis of Hemiketals E2 and D2 by Activated Human Leukocytes. FASEB J. 2017, 31, 1867–1878. [Google Scholar] [CrossRef]
- Griesser, M.; Suzuki, T.; Tejera, N.; Mont, S.; Boeglin, W.E.; Pozzi, A.; Schneider, C. Biosynthesis of Hemiketal Eicosanoids by Cross-over of the 5-Lipoxygenase and Cyclooxygenase-2 Pathways. Proc. Natl. Acad. Sci. USA 2011, 108, 6945–6950. [Google Scholar] [CrossRef]
- Boer, R.E.; Giménez-Bastida, J.A.; Boutaud, O.; Jana, S.; Schneider, C.; Sulikowski, G.A. Total Synthesis and Biological Activity of the Arachidonic Acid Metabolite Hemiketal E2. Org. Lett. 2018, 20, 4020–4022. [Google Scholar] [CrossRef] [PubMed]
- Giménez-Bastida, J.A.; Suzuki, T.; Sprinkel, K.C.; Boeglin, W.E.; Schneider, C. Biomimetic Synthesis of Hemiketal Eicosanoids for Biological Testing. Prostaglandins Other Lipid Mediat. 2017, 132, 41–46. [Google Scholar] [CrossRef]
- Giménez-Bastida, J.A.; González-Sarrías, A.; Espín, J.C.; Schneider, C. Inhibition of 5-Lipoxygenase-Derived Leukotrienes and Hemiketals as a Novel Anti-Inflammatory Mechanism of Urolithins. Mol. Nutr. Food Res. 2020, e2000129. [Google Scholar] [CrossRef]
- Espín, J.C.; González-Sarrías, A.; Tomás-Barberán, F.A. The Gut Microbiota: A Key Factor in the Therapeutic Effects of (Poly)Phenols. Biochem. Pharmacol. 2017, 139, 82–93. [Google Scholar] [CrossRef]
- Cortés-Martín, A.; Selma, M.V.; Tomás-Barberán, F.A.; González-Sarrías, A.; Espín, J.C. Where to Look into the Puzzle of Polyphenols and Health? The Postbiotics and Gut Microbiota Associated with Human Metabotypes. Mol. Nutr. Food Res. 2020, 64, e1900952. [Google Scholar] [CrossRef] [PubMed]
| Standard | IC50 (µg/mL) | Linear Regression |
|---|---|---|
| Caffeic acid | 1616.78 ± 1.45 c | y = 0.021x + 15.401 |
| Captopril | 0.0013 ± 0.001 a | y = 23,075x + 20.606 |
| GSH | 12.79 ± 1.70 b | y = 0.435x + 44.431 |
| L-ascorbic acid | 14,320.09 ± 5.10 d | y = 0.003x + 4.176 |
| Compound/Assay | Antioxidant Activity (mM Trolox) | Reducing Activity (mM Trolox) | Chelating Activity (%) | |
|---|---|---|---|---|
| DPPH RSA | FRAP | CV | FZ | |
| Caffeic acid | 0.90 ± 0.01 a | 0.89 ± 0.01 a | 0.96 ± 0.01 a | 75.80 ± 1.07 c |
| L-ascorbic acid | 0.94 ± 0.01 a | 1.35 ± 0.02 b | 1.18 ± 0.02 b | 1.79 ± 0.36 b |
| Uric acid | 1.18 ± 0.01 b | 1.42 ± 0.03 c | 1.21 ± 0.02 b | 0.99 ± 0.11 a |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zielińska, D.; Zieliński, H.; Laparra-Llopis, J.M.; Szawara-Nowak, D.; Honke, J.; Giménez-Bastida, J.A. Caffeic Acid Modulates Processes Associated with Intestinal Inflammation. Nutrients 2021, 13, 554. https://doi.org/10.3390/nu13020554
Zielińska D, Zieliński H, Laparra-Llopis JM, Szawara-Nowak D, Honke J, Giménez-Bastida JA. Caffeic Acid Modulates Processes Associated with Intestinal Inflammation. Nutrients. 2021; 13(2):554. https://doi.org/10.3390/nu13020554
Chicago/Turabian StyleZielińska, Danuta, Henryk Zieliński, José Moisés Laparra-Llopis, Dorota Szawara-Nowak, Joanna Honke, and Juan Antonio Giménez-Bastida. 2021. "Caffeic Acid Modulates Processes Associated with Intestinal Inflammation" Nutrients 13, no. 2: 554. https://doi.org/10.3390/nu13020554
APA StyleZielińska, D., Zieliński, H., Laparra-Llopis, J. M., Szawara-Nowak, D., Honke, J., & Giménez-Bastida, J. A. (2021). Caffeic Acid Modulates Processes Associated with Intestinal Inflammation. Nutrients, 13(2), 554. https://doi.org/10.3390/nu13020554









