Effect of Vitamin D Supplementation in Early Life on Children’s Growth and Body Composition: A Systematic Review and Meta-Analysis of Randomized Controlled Trials
Abstract
1. Introduction
2. Methods
2.1. Search Strategy
2.2. Study Selection
2.3. Quality Assessment
2.4. Data Extraction and Synthesis
3. Results
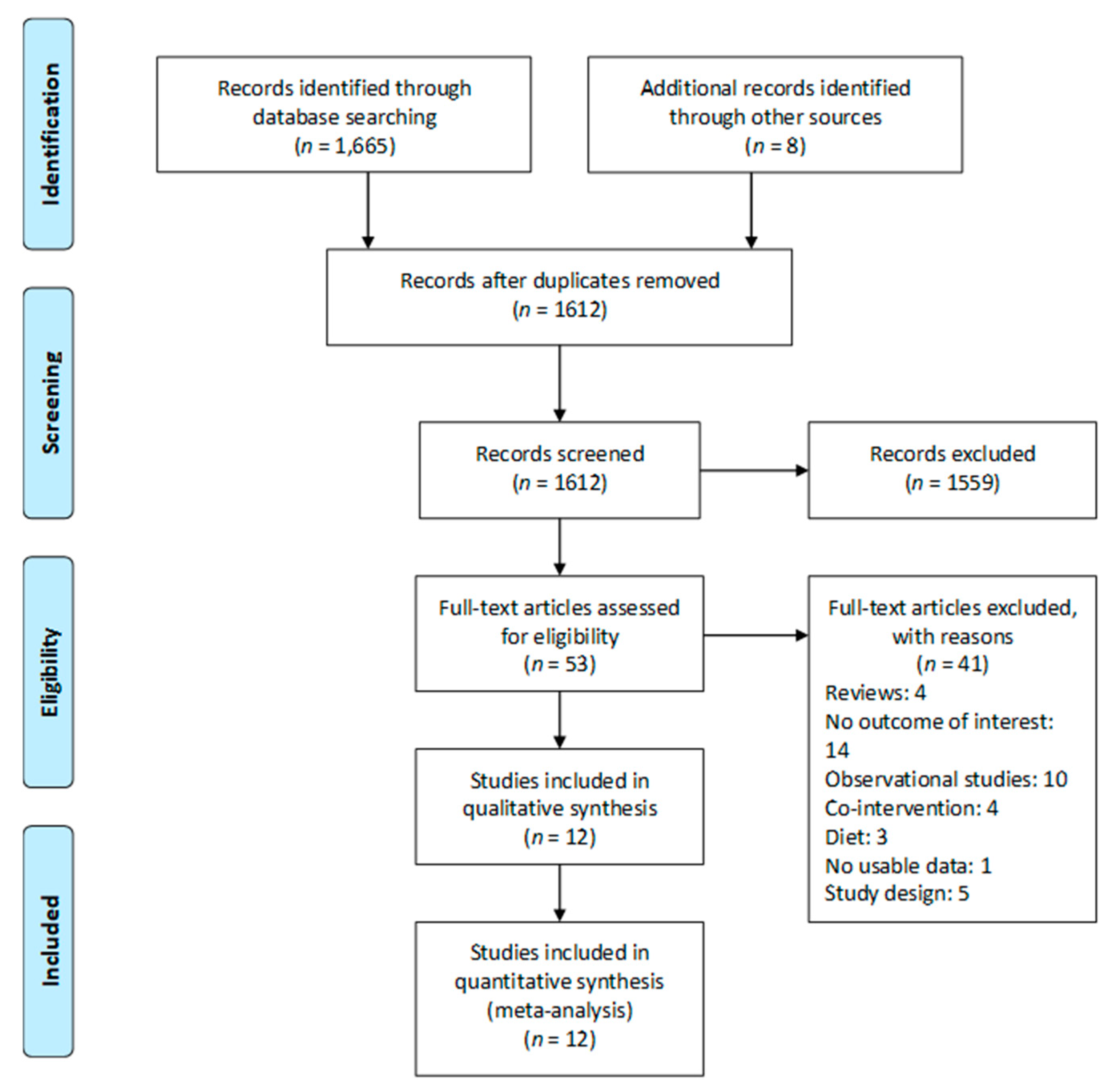
3.1. Study Selection
3.2. Characteristics of Included Trials
3.3. Risk of Bias of Included Clinical Trials
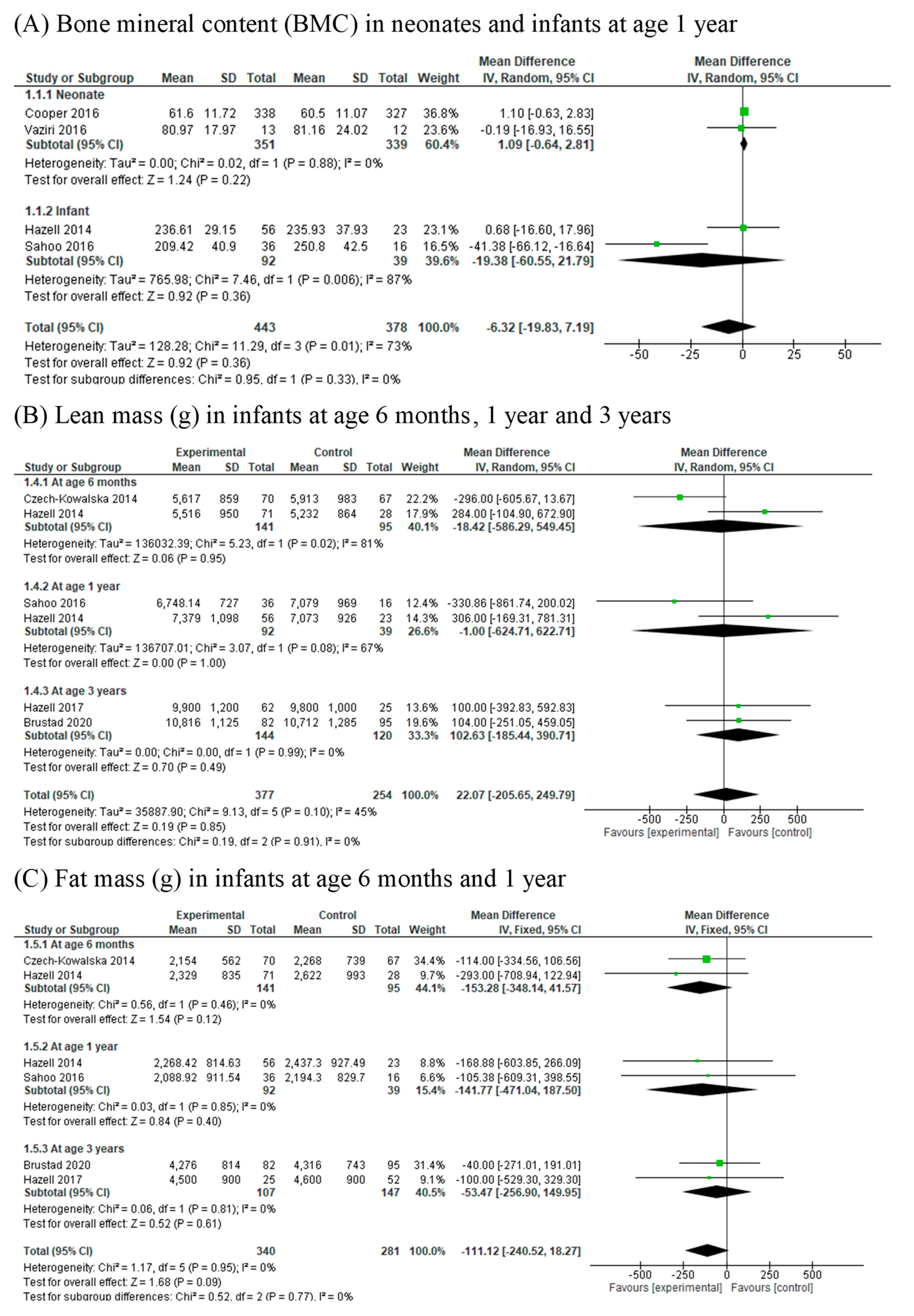
3.4. Bone Mineral Content (BMC)
3.5. Lean Mass (g) and Lean Mass Percentage (%)
3.6. Fat Mass (g) and Fat Mass Percentage (%)
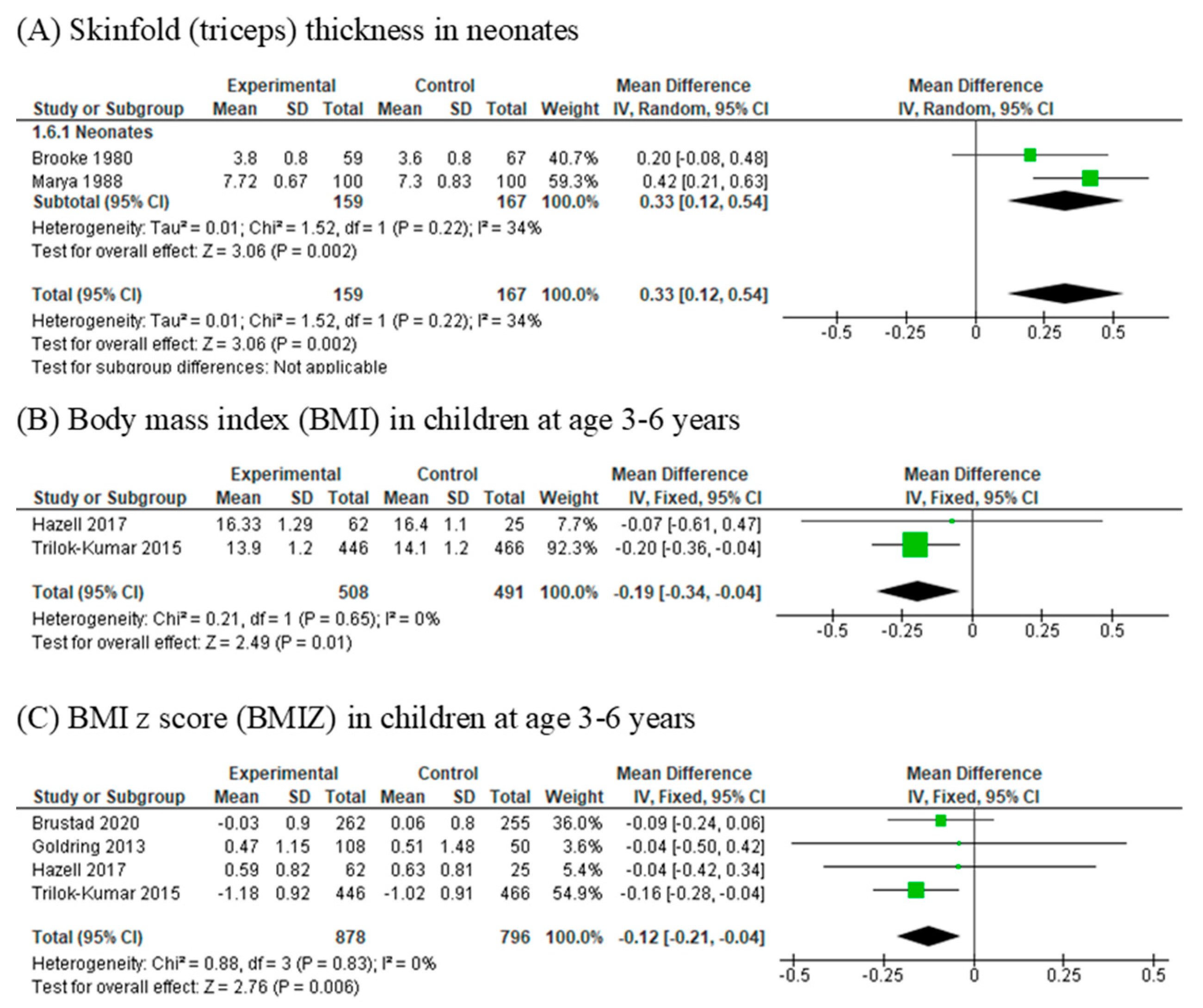
3.7. Skinfold Thickness
3.8. Body Mass Index (BMI)
3.9. Body Mass Index Z-Score (BMIZ)
3.10. Weight for Age Z-Score (WAZ) and Length for Age Z-Score (LAZ)
3.11. Head Circumference for Age Z-Score (HCAZ)
4. Discussion
4.1. Statement of Main Findings
4.2. Importance and Implications
4.3. Comparison with Previous Studies
4.4. Mechanisms
4.5. Strengths and Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Thorne-Lyman, A.; Fawzi, W.W. Vitamin D during pregnancy and maternal, neonatal and infant health outcomes: A systematic review and meta-analysis. Paediatr. Perinat. Epidemiol. 2012, 26 (Suppl. 1), 75–90. [Google Scholar] [CrossRef] [PubMed]
- Curtis, E.M.; Moon, R.J.; Harvey, N.C.; Cooper, C. Maternal vitamin D supplementation during pregnancy. Br. Med. Bull. 2018, 126, 57–77. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, F.; Dabbaghmanesh, M.H.; Samsami, A.; Nasiri, S.; Shirazi, P.T. Vitamin D supplementation during pregnancy on infant anthropometric measurements and bone mass of mother-infant pairs: A randomized placebo clinical trial. Early Hum. Dev. 2016, 103, 61–68. [Google Scholar] [CrossRef]
- Walsh, J.M.; McGowan, C.A.; Kilbane, M.; McKenna, M.J.; McAuliffe, F.M. The relationship between maternal and fetal vitamin D, insulin resistance, and fetal growth. Reprod. Sci. 2013, 20, 536–541. [Google Scholar] [CrossRef]
- Bischoff-Ferrari, H.A.; Borchers, M.; Gudat, F.; Durmuller, U.; Stahelin, H.B.; Dick, W. Vitamin D receptor expression in human muscle tissue decreases with age. J. Bone Miner. Res. 2004, 19, 265–269. [Google Scholar] [CrossRef]
- Harvey, N.C.; Moon, R.J.; Sayer, A.A.; Ntani, G.; Davies, J.H.; Javaid, M.K.; Robinson, S.M.; Godfrey, K.M.; Inskip, H.M.; Cooper, C.; et al. Maternal antenatal vitamin D status and offspring muscle development: Findings from the Southampton Women’s Survey. J. Clin. Endocrinol. Metab. 2014, 99, 330–337. [Google Scholar] [CrossRef] [PubMed]
- Crozier, S.R.; Harvey, N.C.; Inskip, H.M.; Godfrey, K.M.; Cooper, C.; Robinson, S.M.; Group, S.W.S.S. Maternal vitamin D status in pregnancy is associated with adiposity in the offspring: Findings from the southampton women’s survey. Am. J. Clin. Nutr. 2012, 96, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Javaid, M.K.; Crozier, S.R.; Harvey, N.C.; Gale, C.R.; Dennison, E.M.; Boucher, B.J.; Arden, N.K.; Godfrey, K.M.; Cooper, C.; Princess Anne Hospital Study Group. Maternal vitamin D status during pregnancy and childhood bone mass at age 9 years: A longitudinal study. Lancet 2006, 367, 36–43. [Google Scholar] [CrossRef]
- Sayers, A.; Tobias, J.H. Estimated maternal ultraviolet B exposure levels in pregnancy influence skeletal development of the child. J. Clin. Endocrinol. Metab. 2009, 94, 765–771. [Google Scholar] [CrossRef] [PubMed]
- Viljakainen, H.T.; Korhonen, T.; Hytinantti, T.; Laitinen, E.K.; Andersson, S.; Makitie, O.; Lamberg-Allardt, C. Maternal vitamin D status affects bone growth in early childhood—A prospective cohort study. Osteoporos. Int. 2011, 22, 883–891. [Google Scholar] [CrossRef]
- Santamaria, C.; Bi, W.G.; Leduc, L.; Tabatabaei, N.; Jantchou, P.; Luo, Z.C.; Audibert, F.; Nuyt, A.M.; Wei, S.Q. Prenatal vitamin D status and offspring’s growth, adiposity and metabolic health: A systematic review and meta-analysis. Br. J. Nutr. 2018, 119, 310–319. [Google Scholar] [CrossRef]
- Krishnaveni, G.V.; Veena, S.R.; Winder, N.R.; Hill, J.C.; Noonan, K.; Boucher, B.J.; Karat, S.C.; Fall, C.H. Maternal vitamin D status during pregnancy and body composition and cardiovascular risk markers in Indian children: The Mysore parthenon study. Am. J. Clin. Nutr. 2011, 93, 628–635. [Google Scholar] [CrossRef]
- Hyde, N.K.; Brennan-Olsen, S.L.; Wark, J.D.; Hosking, S.M.; Holloway-Kew, K.L.; Pasco, J.A. Vitamin D during pregnancy and offspring body composition: A prospective cohort study. Pediatr. Obes. 2018, 13, 514–521. [Google Scholar] [CrossRef]
- Daraki, V.; Roumeliotaki, T.; Chalkiadaki, G.; Katrinaki, M.; Karachaliou, M.; Leventakou, V.; Vafeiadi, M.; Sarri, K.; Vassilaki, M.; Papavasiliou, S.; et al. Low maternal vitamin D status in pregnancy increases the risk of childhood obesity. Pediatr. Obes. 2018, 13, 467–475. [Google Scholar] [CrossRef] [PubMed]
- Morales, E.; Rodriguez, A.; Valvi, D.; Iñiguez, C.; Esplugues, A.; Vioque, J.; Marina, L.S.; Jiménez, A.; Espada, M.; Dehli, C.R.; et al. Deficit of vitamin D in pregnancy and growth and overweight in the offspring. Int. J. Obes. 2015, 39, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Anatoliotaki, M.; Tsilimigaki, A.; Tsekoura, T.; Schinaki, A.; Stefanaki, S.; Nicolaidou, P. Congenital rickets due to maternal vitamin D deficiency in a sunny island of Greece. Acta Paediatr. 2003, 92, 389–391. [Google Scholar] [CrossRef] [PubMed]
- Innes, A.M.; Seshia, M.M.; Prasad, C.; Al Saif, S.; Friesen, F.R.; Chudley, A.E.; Reed, M.; Dilling, L.A.; Haworth, J.C.; Greenberg, C.R. Congenital rickets caused by maternal vitamin D deficiency. Paediatr. Child Health 2002, 7, 455–458. [Google Scholar] [CrossRef][Green Version]
- Orbak, Z.; Karacan, M.; Doneray, H.; Karakelleoglu, C. Congenital rickets presenting with hypocalcaemic seizures. West Indian Med. J. 2007, 56, 364–367. [Google Scholar]
- Zhu, K.; Whitehouse, A.J.; Hart, P.H.; Kusel, M.; Mountain, J.; Lye, S.; Pennell, C.; Walsh, J.P. Maternal vitamin D status during pregnancy and bone mass in offspring at 20 years of age: A prospective cohort study. J. Bone Miner. Res. 2014, 29, 1088–1095. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Yilmaz, B.; Aygun, C.; Cetinoglu, E. Vitamin D levels in newborns and association with neonatal hypocalcemia. J. Matern. Fetal. Neonatal. Med. 2018, 31, 1889–1893. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Ke, H.J.; Liu, Y.; Fu, M.; Ning, J.; Yu, L.; Xiao, Y.; Che, D.; Chen, X.Y.; Deng, Y.H.; et al. Prevalence of vitamin D insufficiency among children in southern china: A cross-sectional survey. Medicine 2018, 97, e11030. [Google Scholar] [CrossRef]
- Nair, R.; Maseeh, A. Vitamin D: The “sunshine” vitamin. J. Pharmacol. Pharmacother. 2012, 3, 118–126. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Syst. Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Harvey, N.C.; Holroyd, C.; Ntani, G.; Javaid, K.; Cooper, P.; Moon, R.; Cole, Z.; Tinati, T.; Godfrey, K.; Dennison, E.; et al. Vitamin D supplementation in pregnancy: A systematic review. Health Technol. Assess. 2014, 18, 1–190. [Google Scholar] [CrossRef] [PubMed]
- Cooper, C.; Harvey, N.C.; Bishop, N.J.; Kennedy, S.; Papageorghiou, A.T.; Schoenmakers, I.; Fraser, R.; Gandhi, S.V.; Carr, A.; D’Angelo, S.; et al. Maternal gestational vitamin D supplementation and offspring bone health (MAVIDOS): A multicentre, double-blind, randomised placebo-controlled trial. Lancet Diabetes Endocrinol. 2016, 4, 393–402. [Google Scholar] [CrossRef]
- Sahoo, S.K.; Katam, K.K.; Das, V.; Agarwal, A.; Bhatia, V. Maternal vitamin D supplementation in pregnancy and offspring outcomes: A double-blind randomized placebo-controlled trial. J. Bone Miner. Metab. 2017, 35, 464–471. [Google Scholar] [CrossRef]
- Perez-Lopez, F.R.; Pasupuleti, V.; Mezones-Holguin, E.; Benites-Zapata, V.A.; Thota, P.; Deshpande, A.; Hernandez, A.V. Effect of vitamin D supplementation during pregnancy on maternal and neonatal outcomes: A systematic review and meta-analysis of randomized controlled trials. Fertil. Steril. 2015, 103, 1278–1288.e4. [Google Scholar] [CrossRef]
- Higgins, J.P.; Altman, D.G.; Gøtzsche, P.C.; Jüni, P.; Moher, D.; Oxman, A.D.; Savović, J.; Schulz, K.F.; Weeks, L.; Sterne, J.A. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ 2011, 343, d5928. [Google Scholar] [CrossRef]
- Czech-Kowalska, J.; Latka-Grot, J.; Bulsiewicz, D.; Jaworski, M.; Pludowski, P.; Wygledowska, G.; Chazan, B.; Pawlus, B.; Zochowska, A.; Borszewska-Kornacka, M.K.; et al. Impact of vitamin D supplementation during lactation on vitamin D status and body composition of mother-infant pairs: A MAVID randomized controlled trial. PLoS ONE 2014, 9, e107708. [Google Scholar] [CrossRef]
- Goldring, S.T.; Griffiths, C.J.; Martineau, A.R.; Robinson, S.; Yu, C.; Poulton, S.; Kirkby, J.C.; Stocks, J.; Hooper, R.; Shaheen, S.O.; et al. Prenatal vitamin d supplementation and child respiratory health: A randomised controlled trial. PLoS ONE 2013, 8, e66627. [Google Scholar] [CrossRef] [PubMed]
- Gallo, S.; Comeau, K.; Vanstone, C.; Agellon, S.; Sharma, A.; Jones, G.; L’Abbe, M.; Khamessan, A.; Rodd, C.; Weiler, H. Effect of different dosages of oral vitamin D supplementation on vitamin D status in healthy, breastfed infants: A randomized trial. JAMA 2013, 309, 1785–1792. [Google Scholar] [CrossRef]
- Hazell, T.J.; Gallo, S.; Berzina, I.; Vanstone, C.A.; Rodd, C.; Weiler, H.A. Plasma 25-hydroxyvitamin D, more so than its epimer, has a linear relationship to leaner body composition across infancy in healthy term infants. Appl. Physiol. Nutr. Metab. 2014, 39, 1137–1143. [Google Scholar] [CrossRef]
- Hazell, T.J.; Gallo, S.; Vanstone, C.A.; Agellon, S.; Rodd, C.; Weiler, H.A. Vitamin D supplementation trial in infancy: Body composition effects at 3 years of age in a prospective follow-up study from Montreal. Pediatr. Obes. 2017, 12, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Marya, R.K.; Rathee, S.; Dua, V.; Sangwan, K. Effect of vitamin D supplementation during pregnancy on foetal growth. Indian J. Med. Res. 1988, 88, 488–492. [Google Scholar]
- Roth, D.E.; Perumal, N.; Al Mahmud, A.; Baqui, A.H. Maternal vitamin D3 supplementation during the third trimester of pregnancy: Effects on infant growth in a longitudinal follow-up study in Bangladesh. J. Pediatr. 2013, 163, 1605–1611.e3. [Google Scholar] [CrossRef] [PubMed]
- Roth, D.E.; Morris, S.K.; Zlotkin, S.; Gernand, A.D.; Ahmed, T.; Shanta, S.S.; Papp, E.; Korsiak, J.; Shi, J.; Islam, M.M.; et al. Vitamin D supplementation in pregnancy and lactation and infant growth. N. Engl. J. Med. 2018, 379, 535–546. [Google Scholar] [CrossRef]
- Trilok-Kumar, G.; Kaur, M.; Rehman, A.M.; Arora, H.; Rajput, M.M.; Chugh, R.; Kurpad, A.; Sachdev, H.S.; Filteau, S. Effects of vitamin D supplementation in infancy on growth, bone parameters, body composition and gross motor development at age 3-6 years: Follow-up of a randomized controlled trial. Int. J. Epidemiol. 2015, 44, 894–905. [Google Scholar] [CrossRef] [PubMed]
- Brooke, O.G.; Brown, I.R.; Bone, C.D.; Carter, N.D.; Cleeve, H.J.; Maxwell, J.D.; Robinson, V.P.; Winder, S.M. Vitamin D supplements in pregnant Asian women: Effects on calcium status and fetal growth. Br. Med. J. 1980, 280, 751–754. [Google Scholar] [CrossRef] [PubMed]
- Brustad, N.; Garland, J.; Thorsen, J.; Sevelsted, A.; Krakauer, M.; Vinding, R.K.; Stokholm, J.; Bønnelykke, K.; Bisgaard, H.; Chawes, B.L. Effect of High-Dose vs Standard-Dose Vitamin D Supplementation in Pregnancy on Bone Mineralization in Offspring Until Age 6 Years: A Prespecified Secondary Analysis of a Double-Blinded, Randomized Clinical Trial. JAMA Pediatr. 2020, 174, 419–427. [Google Scholar] [CrossRef]
- Karras, S.N.; Fakhoury, H.; Muscogiuri, G.; Grant, W.B.; van den Ouweland, J.M.; Colao, A.M.; Kotsa, K. Maternal vitamin D levels during pregnancy and neonatal health: Evidence to date and clinical implications. Ther. Adv. Musculoskelet. Dis. 2016, 8, 124–135. [Google Scholar] [CrossRef] [PubMed]
- Moon, R.J.; Harvey, N.C.; Cooper, C. Endocrinology in pregnancy: Influence of maternal vitamin D status on obstetric outcomes and the fetal skeleton. Eur. J. Endocrinol. 2015, 173, R69–R83. [Google Scholar] [CrossRef] [PubMed]
- Bi, W.G.; Nuyt, A.M.; Weiler, H.; Leduc, L.; Santamaria, C.; Wei, S.Q. Association between vitamin D supplementation during pregnancy and offspring growth, morbidity, and mortality: A systematic review and Meta-analysis. JAMA Pediatr. 2018, 172, 635–645. [Google Scholar] [CrossRef] [PubMed]
- Maugeri, A.; Barchitta, M.; Blanco, I.; Agodi, A. Effects of vitamin D supplementation during pregnancy on birth size: A systematic review and Meta-Analysis of randomized controlled trials. Nutrients 2019, 11, 442. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; Kostiuk, L.K.; Pena-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019, 7, CD008873. [Google Scholar] [CrossRef]
- Moon, R.J.; Harvey, N.C.; Davies, J.H.; Cooper, C. Vitamin D and skeletal health in infancy and childhood. Osteoporos. Int. 2014, 25, 2673–2684. [Google Scholar] [CrossRef]
- Salles, J.; Chanet, A.; Giraudet, C.; Patrac, V.; Pierre, P.; Jourdan, M.; Luiking, Y.C.; Verlaan, S.; Migne, C.; Boirie, Y.; et al. 1,25(OH)2-vitamin D3 enhances the stimulating effect of leucine and insulin on protein synthesis rate through Akt/PKB and mTOR mediated pathways in murine C2C12 skeletal myotubes. Mol. Nutr. Food Res. 2013, 57, 2137–2146. [Google Scholar] [CrossRef] [PubMed]
- Plachta-Danielzik, S.; Bosy-Westphal, A.; Kehden, B.; Gehrke, M.I.; Kromeyer-Hauschild, K.; Grillenberger, M.; Willhoft, C.; Heymsfield, S.B.; Muller, M.J. Adiposity rebound is misclassified by BMI rebound. Eur. J. Clin. Nutr. 2013, 67, 984–989. [Google Scholar] [CrossRef]
- Belenchia, A.M.; Johnson, S.A.; Ellersieck, M.R.; Rosenfeld, C.S.; Peterson, C.A. In utero vitamin D deficiency predisposes offspring to long-term adverse adipose tissue effects. J. Endocrinol. 2017, 234, 301–313. [Google Scholar] [CrossRef] [PubMed]
- Rosen, E.D.; Spiegelman, B.M. Molecular regulation of adipogenesis. Ann. Rev. Cell Dev. Biol. 2000, 16, 145–171. [Google Scholar] [CrossRef] [PubMed]
- Spiegelman, B.M.; Hu, E.; Kim, J.B.; Brun, R. PPAR gamma and the control of adipogenesis. Biochimie 1997, 79, 111–112. [Google Scholar] [CrossRef]
- Lazar, M.A. Becoming fat. Genes Dev. 2002, 16, 1–5. [Google Scholar] [CrossRef] [PubMed]
| Study | Country | Study Design | Total Sample Size (n) | Participants | Baseline 25(OH)D (nmol/L) | Initiation and Duration of Supplementation | Interventions | Compliance (% of Dosages Taken) | Outcomes |
|---|---|---|---|---|---|---|---|---|---|
| Brooke 1980 [39] | United Kingdom | RCT | 126 | Pregnant Asian women | NA | Third trimester | Ergocalciferol, 1000 IU/day vs. placebo | NA | Neonatal anthropometry (triceps skinfold thickness) |
| Brustad 2020 [40] | Denmark | RCT | 517 | Pregnant women | 77.5 nmol/L | 24 weeks of gestation to 1 week postpartum | Cholecalciferol 2800 IU/day vs. 400 IU/day | 74% | BMC, fat and lean mass, and BMIZ in children at 3 years of age |
| Cooper 2016 [26] | United Kingdom | RCT | 965 | Pregnant women gestation age less than 17 weeks, serum 25(OH)D at 25–100 nmol/L at 10–17 weeks’ gestation | Mean: 45.8 nmol/L <50 nmol/L:42.2% | 14 weeks of gestation or before 17 weeks of gestation until delivery | Cholecalciferol, 1000 IU/day vs. placebo | 70% | Neonatal whole-body BMC, fat mass and lean mass by DXA |
| Czech-Kowalska 2014 [30] | Poland | RCT | 137 | Healthy women who delivered at term, a single neonate and breastfed for the next 6 months | Mean: 37.9 nmol/L <50 nmol/L:68% | At delivery for 6 months | Cholecalciferol 1200 IU/day vs. 400 IU/day | 82% | Fat mass and lean body mass by DXA in infants at 6 months |
| Goldring 2013 [31] | United Kingdom | RCT | 158 | Women presenting at 27 weeks gestation | <25 nmol/L:44.9% | 27 weeks of gestation until delivery or single dose | Ergocalciferol 800 IU/day or cholecalciferol single oral dose of 200,000 IU vs. no treatment (control) | NA | BMIZ in children at 3 years of age |
| Gallo 2013 [32] Hazell 2014 [33] Hazell 2017 [34] | Canada | RCT | 132 | 1 month old healthy, breastfed infants | Mean: 62.1 nmol/L | 1 month old to 12-months (for 11 months) | Cholecalciferol of 800, 1200 or 1600 IU/day vs. 400 IU/day | 84% | Anthropometry (WAZ, HAZ), BMI, BMIZ, body composition (BMC, lean mass and fat mass) by DXA at 1 and 3 years of age |
| Marya 1988 [35] | India | RCT | 200 | Pregnant women aged 22–35 years without complications | NA | Administration of 2 doses, one at the 7th and the other at the 8th month of gestation | Cholecalciferol 600,000 IU/dose vs. unsupplemented control | NA | Neonatal anthropometry (triceps skinfold) |
| Roth 2013 [36] | Bangladesh | RCT | 134 | Pregnant women at 26 to 29 weeks gestation | Mean: 41.1 nmol/L | Third trimester | Cholecalciferol 35,000 IU/week vs. placebo | NA | Anthropometry (WAZ, LAZ, HCAZ) at 1 year of age |
| Roth 2018 [37] | Bangladesh | RCT | 1164 | Pregnant women and their infants | Mean: 27.5 nmol/L | Supplementation of pregnant women from 17 to 24 weeks of gestation until birth and, in one subgroup, 26 weeks of postnatal supplementation in infants | Cholecalciferol 4200 IU/week vs. 16,800 IU/week vs. 28,800 IU/week vs. placebo | At least 90% | Anthropometry (WAZ, LAZ, HCAZ, BMIZ) at birth and at 1 year of age |
| Sahoo 2016 [27] | India | RCT | 52 | Healthy pregnant women less than 20 weeks of gestation | Mean: 28.2 nmol/L <50 nmol/L: 87% | 14–20 weeks of gestation until delivery | Cholecalciferol 60,000 IU/4 weeks or 60,000 IU/8 weeks vs. 400 IU/day | NA | Anthropometry (WAZ, LAZ, HCAZ), whole body BMC, lean mass and fat mass by DXA at 1 year of age |
| Trilok-Kumar 2015 [38] | India | RCT | 912 | Healthy neonates | Mean: 36 nmol/L <50 nmol/L: 73% | Infants at 7 days of age for 6 months | Cholecalciferol 1500 IU/week vs. placebo | NA | Anthropometry (WAZ, HAZ, triceps skinfold), BMI, BMIZ, bone structure and strength, deuterium dilution test of body composition on a subset (n = 229) at age 3–6 years |
| Vaziri 2016 [3] | Iran | RCT | 25 | Healthy pregnant women | Mean: 30.2 nmol/L | 26–28 weeks of gestation until childbirth | Cholecalciferol 2000 IU/day vs. placebo | NA | Whole-body: BMC by DXA in offspring at birth, 4th and 8th weeks of age |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, K.; Wei, S.Q.; Bi, W.G.; Weiler, H.A.; Wen, S.W. Effect of Vitamin D Supplementation in Early Life on Children’s Growth and Body Composition: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2021, 13, 524. https://doi.org/10.3390/nu13020524
Ma K, Wei SQ, Bi WG, Weiler HA, Wen SW. Effect of Vitamin D Supplementation in Early Life on Children’s Growth and Body Composition: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients. 2021; 13(2):524. https://doi.org/10.3390/nu13020524
Chicago/Turabian StyleMa, Kristine, Shu Qin Wei, Wei Guang Bi, Hope A. Weiler, and Shi Wu Wen. 2021. "Effect of Vitamin D Supplementation in Early Life on Children’s Growth and Body Composition: A Systematic Review and Meta-Analysis of Randomized Controlled Trials" Nutrients 13, no. 2: 524. https://doi.org/10.3390/nu13020524
APA StyleMa, K., Wei, S. Q., Bi, W. G., Weiler, H. A., & Wen, S. W. (2021). Effect of Vitamin D Supplementation in Early Life on Children’s Growth and Body Composition: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients, 13(2), 524. https://doi.org/10.3390/nu13020524






