The Influence of Specific Bioactive Collagen Peptides on Knee Joint Discomfort in Young Physically Active Adults: A Randomized Controlled Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Participants
2.2. Efficacy Outcomes
2.3. Investigational Products
2.4. Statistical Analysis
3. Results
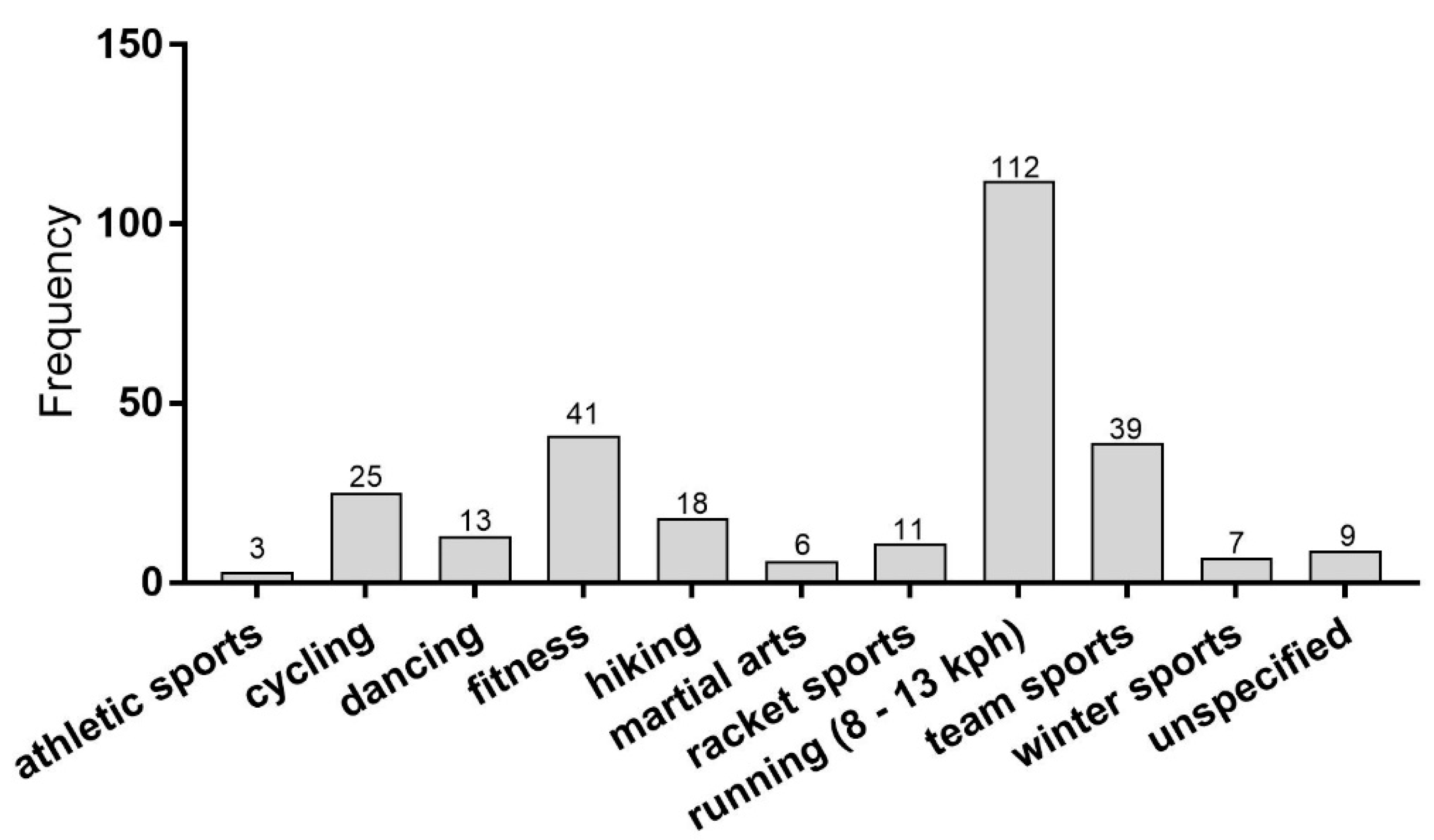
3.1. Subjects
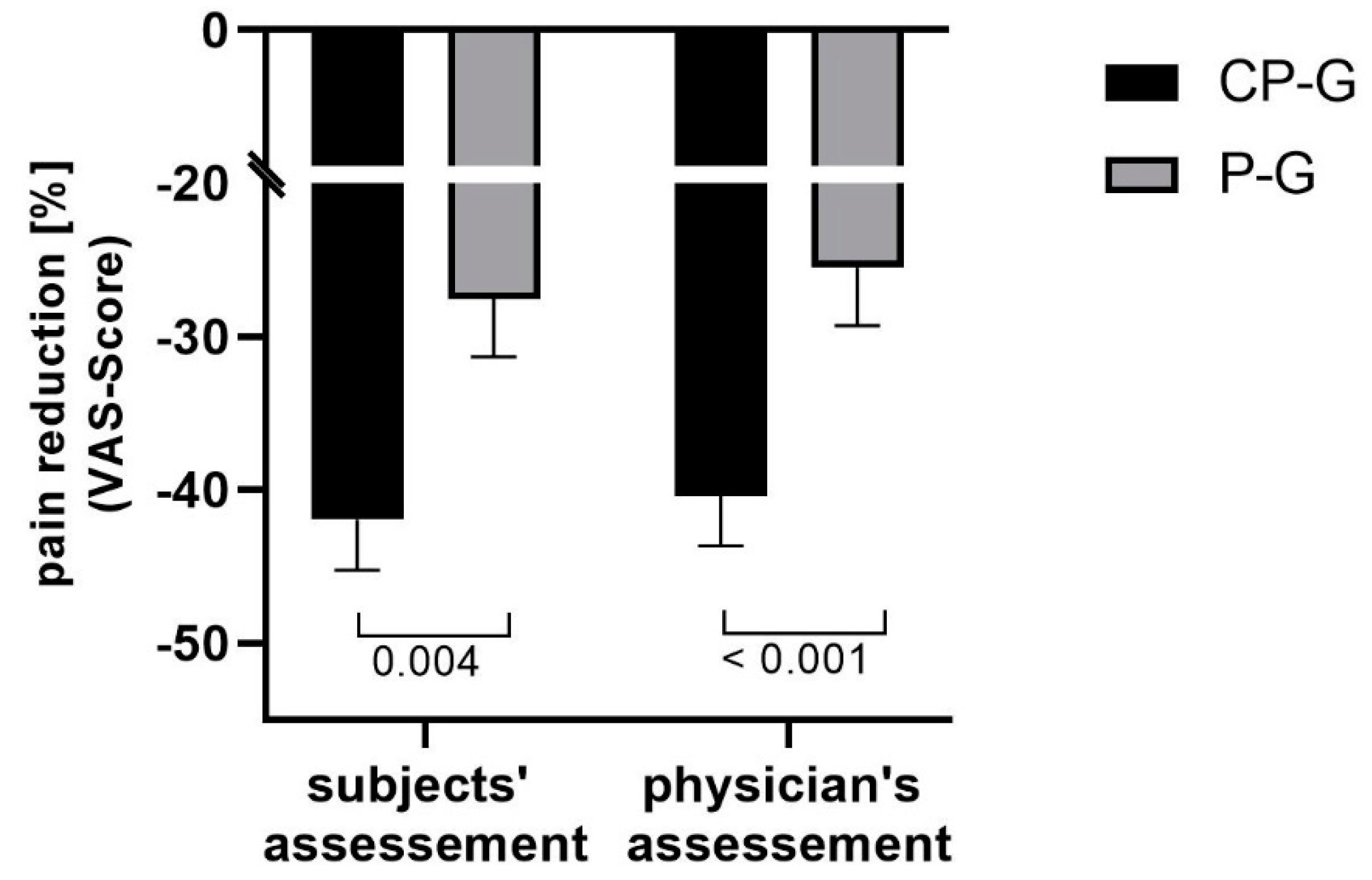
3.2. Change in Knee Pain and Range of Motion
3.3. Additional Treatment Options
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Briggs, A.M.; Cross, M.J.; Hoy, D.G.; Sànchez-Riera, L.; Blyth, F.M.; Woolf, A.D.; March, L. Musculoskeletal Health Conditions Represent a Global Threat to Healthy Aging: A Report for the 2015 World Health Organization World Report on Ageing and Health. Gerontologist 2016, 56, S243–S255. [Google Scholar] [CrossRef] [PubMed]
- IASP (International Association for the Study of Pain). Joint Pain Epidemiology. Available online: https://www.iasp-pain.org/ (accessed on 1 June 2017).
- United States Bone and Joint Initiative. The Burden of Musculoskeletal Diseases in the United States (BMUS), 3rd ed.; United States Bone and Joint Initiative: Rosemont, IL, USA, 2014. [Google Scholar]
- Gelse, K.; Pöschl, E.; Aigner, T. Collagens—Structure, function, and biosynthesis. Adv. Drug Deliv. Rev. 2003, 55, 1531–1546. [Google Scholar] [CrossRef]
- Buckwalter, J.A.; Mankin, H.J.; Grodzinsky, A.J. Articular cartilage and osteoarthritis. Instr. Course Lect. Am. Acad. Orthop. Surg. 2005, 54, 465–480. [Google Scholar]
- Goldring, M.B. Chondrogenesis, chondrocyte differentiation, and articular cartilage metabolism in health and osteoarthritis. Ther. Adv. Musculoskelet Dis. 2012, 4, 269–285. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves-Neto, J.; Witzel, S.S.; Teodoro, W.R.; Carvalho-Júnior, A.E.; Fernandes, T.D.; Yoshinari, H.H. Changes in collagen matrix composition in human posterior tibial tendon dysfunction. Jt. Bone Spine 2002, 69, 189–194. [Google Scholar] [CrossRef]
- Magnusson, S.P.; Langberg, H.; Kjaer, M. The pathogenesis of tendinopathy: Balancing the response to loading. Nat. Rev. Rheumatol. 2010, 6, 262–268. [Google Scholar] [CrossRef] [PubMed]
- Caine, D.; Maffulli, N.; Caine, C. Epidemiology of injury in child and adolescent sports: Injury rates, risk factors, and prevention. Clin. Sports Med. 2008, 27, 19–50. [Google Scholar] [CrossRef]
- DiFiori, J.P.; Benjamin, H.J.; Brenner, J.S.; Gregory, A.; Jayanthi, N.; Landry, G.L.; Luke, A. Overuse injuries and burnout in youth sports: A position statement from the American Medical Society for Sports Medicine. Br. J. Sports Med. 2014, 48, 287–288. [Google Scholar] [CrossRef]
- Peter Kriz, M.D. Overuse injuries in the young athlete. Med. Health R I 2011, 94, 203. [Google Scholar]
- Ramesh, R.; Arx, O.V.; Azzopardi, T.; Schranz, P.J. The risk of anterior cruciate ligament rupture with generalised joint laxity. Bone Jt. J. 2005, 87-B, 800–803. [Google Scholar] [CrossRef]
- Akhtar, M.A.; Robinson, C.M. Generalised ligament laxity and shoulder dislocations after sports injuries. Br. J. Sports Med. 2010, 44, i3. [Google Scholar] [CrossRef]
- Albers, I.S.; Zwerver, J.; Diercks, R.L.; Dekker, J.H.; Van den Akker-Scheek, I. Incidence and prevalence of lower extremity tendinopathy in a Dutch general practice population: A cross sectional study. BMC Musculoskelet Disord. 2016, 17, 16. [Google Scholar] [CrossRef]
- Holmes, J.C.; Pruitt, A.L.; Whalen, N.J. Iliotibial band syndrome in cyclists. Am. J. Sports Med. 1993, 21, 419–424. [Google Scholar] [CrossRef] [PubMed]
- Micheli, L.J.; Klein, J.D. Sports injuries in children and adolescents. Br. J. Sports Med. 1991, 25, 6–9. [Google Scholar] [CrossRef] [PubMed]
- D’Addona, A.; Maffulli, N.; Formisano, S.; Rosa, D. Inflammation in tendinopathy. Surgeon 2017, 15, 297–302. [Google Scholar] [CrossRef]
- Fredberg, U.; Bolvig, L. Jumper’s knee. Review of the literature. Scand, J. Med. Sci. Sports 1999, 9, 66–73. [Google Scholar] [CrossRef]
- Feng, M.; Betti, M. Transepithelial transport efficiency of bovine collagen hydrolysates in a human Caco-2 cell line model. Food Chem. 2017, 224, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Young, J.F.; Rasmussen, M.K.; Dalsgaard, T.K.; Lametsch, R.; Aluko, R.E.; Therkildsen, M. Angiotensin I-converting enzyme-inhibitory peptides from bovine collagen: Insights into inhibitory mechanism and transepithelial transport. Food Res. Int. 2016, 89, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Osawa, Y.; Mizushige, T.; Jinno, S.; Sugihara, F.; Inoue, N.; Tanaka, H.; Kabuyama, Y. Absorption and metabolism of orally administered collagen hydrolysates evaluated by the vascularly perfused rat intestine and liver in situ. Biomed. Res. 2018, 39, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Shimizu, K.; Sato, M.; Zhang, Y.; Kouguchi, T.; Takahata, Y.; Morimatsu, F.; Shimizu, M. Molecular size of collagen peptide reverses the permeability of Caco-2 cells. Biosci. Biotechnol. Biochem. 2010, 74, 1123–1125. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Taga, Y.; Kusubata, M.; Ogawa-Goto, K.; Hattori, S. Highly Accurate Quantification of Hydroxyproline-Containing Peptides in Blood Using a Protease Digest of Stable Isotope-Labeled Collagen. J. Agric. Food Chem. 2014, 62, 12096–12102. [Google Scholar] [CrossRef] [PubMed]
- Taga, Y.; Kusubata, M.; Ogawa-Goto, K.; Hattori, S. Efficient Absorption of X-Hydroxyproline (Hyp)-Gly after Oral Administration of a Novel Gelatin Hydrolysate Prepared Using Ginger Protease. J. Agric. Food Chem. 2016, 64, 2962–2970. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Wang, Q.; Liang, Q.; He, Y.; Wang, Z.; He, S.; Xu, J.; Ma, H. Determination of Bioavailability and Identification of Collagen Peptide in Blood after Oral Ingestion of Gelatin. J. Sci. Food Agric. 2015, 95, 2712–2717. [Google Scholar] [CrossRef]
- Nakatani, S.; Mano, H.; Sampei, C.; Shimizu, J.; Wada, M. Chondroprotective effect of the bioactive peptide prolyl-hydroxyproline in mouse articular cartilage in vitro and in vivo. Osteoarthr. Cartil. 2009, 17, 1620–1627. [Google Scholar] [CrossRef]
- Ng, K.W.; Saliman, J.D.; Lin, E.Y.; Statman, L.Y.; Kugler, L.E.; Lo, S.B.; Ateshian, G.A.; Hung, C.T. Culture duration modulates collagen hydrolysate-induced tissue remodeling in chondrocyte-seeded agarose hydrogels. Ann. Biomed. Eng. 2007, 35, 1914–1923. [Google Scholar] [CrossRef]
- Oesser, S.; Seifert, J. Stimulation of type II collagen biosynthesis and secretion in bovine chondrocytes cultured with degraded collagen. Cell Tissue Res. 2003, 311, 393–399. [Google Scholar] [CrossRef]
- Ohara, H.; Iida, H.; Ito, K.; Takeuchi, Y.; Nomura, Y. Effects of Pro-Hyp, a Collagen Hydrolysate-Derived Peptide, on Hyaluronic Acid Synthesis Using in Vitro Cultured Synovium Cells and Oral Ingestion of Collagen Hydrolysates in a Guinea Pig Model of Osteoarthritis. Biosci. Biotechnol. Biochem. 2010, 74, 2096–2099. [Google Scholar] [CrossRef]
- Isaka, S.; Someya, A.; Nakamura, S.; Naito, K.; Nozawa, M.; Inoue, N.; Sugihara, F.; Nagaoka, I.; Kaneko, K. Evaluation of the Effect of Oral Administration of Collagen Peptides on an Experimental Rat Osteoarthritis Model. Exp. Ther. Med. 2017, 13, 2699–2706. [Google Scholar] [CrossRef]
- McAlindon, T.E.; Nuite, M.; Krishnan, N.; Ruthazer, R.; Price, L.L.; Burstein, D.; Griffith, J.; Flechsenhar, K. Change in knee osteoarthritis cartilage detected by delayed gadolinium enhanced magnetic resonance imaging following treatment with collagen hydrolysate: A pilot randomized controlled trial. Osteoarthr. Cartil. 2011, 19, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Bruyère, O.; Zegels, B.; Leonori, L.; Rabenda, V.; Janssen, A.; Bourges, C.; Reginster, J.-Y. Effect of collagen hydrolysate in articular pain: A 6-month randomized, double-blind, placebo controlled study. Complement Ther. Med. 2012, 20, 124–130. [Google Scholar] [CrossRef]
- Jiang, I.-X.; Yu, S.; Huang, X.-L.; Zhang, C.-Q.; Zhou, J.-L.; Prawitt, J. Collagen Peptides Improve Knee Osteoarthritis in Elderly Women. Agro. Food Ind. Hi-Tech. 2014, 25, 19–23. [Google Scholar]
- Juher, T.F.; Pérez, E.B. An overview of the beneficial effects of hydrolysed collagen intake on joint and bone health and on skin ageing. Nutr. Hosp. 2015, 32 (Suppl. 1), 62–66. [Google Scholar] [CrossRef]
- García-Coronado, J.M.; Martínez-Olvera, L.; Elizondo-Omaña, R.E.; Acosta-Olivo, C.A.; Vilchez-Cavazos, F.; Simental-Mendía, L.E.; Simental-Mendía, M. Effect of collagen supplementation on osteoarthritis symptoms: A meta-analysis of randomized placebo-controlled trials. Int. Orthop. 2019, 43, 531–538. [Google Scholar] [CrossRef]
- Moskowitz, R.W. Role of collagen hydrolysate in bone and joint disease. Semin. Arthritis Rheum. 2000, 30, 87–99. [Google Scholar] [CrossRef]
- Benito-Ruiz, P.; Camacho-Zambrano, M.M.; Carrillo-Arcentales, J.N.; Mestanza-Peralta, M.A.; Vallejo-Flores, C.A.; Vargas-López, S.V.; Villacís-Tamayo, R.A.; Zurita-Gavilanes, L.A. A randomized controlled trial on the efficacy and safety of a food ingredient, collagen hydrolysate, for improving joint comfort. Int. J. Food Sci. Nutr. 2009, 60 (Suppl. 2), 99–113. [Google Scholar] [CrossRef]
- Clark, K.L.; Sebastianelli, W.; Flechsenhar, K.R.; Aukermann, D.F.; Meza, F.; Millard, R.L.; Deitch, J.R.; Sherbondy, P.S.; Albert, A. 24-Week study on the use of collagen hydrolysate as a dietary supplement in athletes with activity-related joint pain. Curr. Med. Res. Opin. 2008, 24, 1485–1496. [Google Scholar] [CrossRef] [PubMed]
- Zdzieblik, D.; Oesser, S.; Gollhofer, A.; König, D. Improvement of activity-related knee joint discomfort following supplementation of specific collagen peptides. Appl. Physiol. Nutr. Metab. 2017, 1–8. [Google Scholar] [CrossRef]
- Research Randomizer. Available online: https://www.randomizer.org/ (accessed on 2 March 2016).
- Hawker, G.A.; Mian, S.; Kendzerska, T.; French, M. Measures of adult pain: Visual Analog Scale for Pain (VAS Pain), Numeric Rating Scale for Pain (NRS Pain), McGill Pain Questionnaire (MPQ), Short-Form McGill Pain Questionnaire (SF-MPQ), Chronic Pain Grade Scale (CPGS), Short Form-36 Bodily Pain Scale (SF-36 BPS), and Measure of Intermittent and Constant Osteoarthritis Pain (ICOAP). Arthritis Care Res. (Hoboken) 2011, 63 (Suppl. 11), S240–S252. [Google Scholar] [CrossRef] [PubMed]
- McMurray, T.P. The semilunar cartilages. Br. J. Surg. 1942, 29, 407–414. [Google Scholar] [CrossRef]
- Steinmann, F. Referat über Meniskusverletzungen. Schweiz Med. Wochenschr. 1929, 10, 1355–1356. [Google Scholar]
- Butler, D.L.; Noyes, F.R.; Grood, E.S. Ligamentous restraints to anterior-posterior drawer in the human knee. A biomechanical study. J. Bone Jt. Surg. Am. 1980, 62, 259–270. [Google Scholar] [CrossRef]
- Aumüller, G.; Aust, G.; Doll, A.; Engele, J.; Kirsch, J.; Mense, S.; Reißig, D.; Salvetter, J.; Schmidt, W.; Schmitz, F.; et al. (Eds.) Duale Reihe Anatomie, 2nd ed.; Georg Thieme Verlag: Stuttgart, Germany, 2010; ISBN 978-3-13-136042-7. [Google Scholar]
- Flechsenhar, K.; Alf, D. Results of a Postmarketing Surveillance Study of Collagen Hydrolysate CH-Alpha. Orthopädische Praxis 2005, 9, 486–494. [Google Scholar]
- Osti, M.; Seil, R. Das schmerzhafte Kniegelenk im Kindes- und Jugendalter. Dtsch. Z. Sportmed. 2004, 55, 246–255. [Google Scholar]
- Jacobson, K.E. Jumper’s Knee. Hughston Health Alert 2001, 13, 5. Available online: https://hughston.com/wp-content/uploads/2020/01/vol13no4.pdf (accessed on 7 October 2016).
- Schilling, F. Das familiäre systematisierte Hypermobilitätssyndrom bei generalisierter Bindegewebsschwäche. Aktuelle Rheumatol. 2007, 32, 341–348. [Google Scholar] [CrossRef]
- Dressler, P.; Gehring, D.; Zdzieblik, D.; Oesser, S.; Gollhofer, A.; König, D. Improvement of Functional Ankle Properties Following Supplementation with Specific Collagen Peptides in Athletes with Chronic Ankle Instability. J. Sports Sci. Med. 2018, 17, 298–304. [Google Scholar] [CrossRef]
- Praet, S.F.E.; Purdam, C.R.; Welvaert, M.; Vlahovich, N.; Lovell, G.; Burke, L.M.; Gaida, J.E.; Manzanero, S.; Hughes, D.; Waddington, G. Oral Supplementation of Specific Collagen Peptides Combined with Calf-Strengthening Exercises Enhances Function and Reduces Pain in Achilles Tendinopathy Patients. Nutrients 2019, 11, 76. [Google Scholar] [CrossRef] [PubMed]
- Schunck, M.; Oesser, S. Specific collagen peptides benefit the biosynthesis of matrix molecules of tendons and ligaments. J. Int. Soc. Sports Nutr. 2013, 10, P23. [Google Scholar] [CrossRef]
- Hartog, A.; Cozijnsen, M.; de Vrij, G.; Garssen, J. Collagen hydrolysate inhibits zymosan-induced inflammation. Exp. Biol. Med. (Maywood) 2013, 238, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Dar, Q.-A.; Schott, E.M.; Catheline, S.E.; Maynard, R.D.; Liu, Z.; Kamal, F.; Farnsworth, C.W.; Ketz, J.P.; Mooney, R.A.; Hilton, M.J.; et al. Daily oral consumption of hydrolyzed type 1 collagen is chondroprotective and anti-inflammatory in murine posttraumatic osteoarthritis. PLoS ONE 2017, 12. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, X.; Wu, H.; Chen, S.; Zhu, H.; Zhang, J.; Hou, Y.; Hu, C.-A.A.; Zhang, G. Glycine enhances muscle protein mass associated with maintaining Akt-mTOR-FOXO1 signaling and suppressing TLR4 and NOD2 signaling in piglets challenged with LPS. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2016, 311, R365–R373. [Google Scholar] [CrossRef]
- Schaible, H.-G.; Schmelz, M.; Tegeder, I. Pathophysiology and treatment of pain in joint disease. Adv. Drug Deliv. Rev. 2006, 58, 323–342. [Google Scholar] [CrossRef] [PubMed]
- Goldring, M.B.; Otero, M.; Plumb, D.A.; Dragomir, C.; Favero, M.; El Hachem, K.; Hashimoto, K.; Roach, H.I.; Olivotto, E.; Borzì, R.M.; et al. Roles of inflammatory and anabolic cytokines in cartilage metabolism: Signals and multiple effectors converge upon MMP-13 regulation in osteoarthritis. Eur. Cell Mater. 2011, 21, 202–220. [Google Scholar] [CrossRef] [PubMed]
- Schadow, S.; Simons, V.S.; Lochnit, G.; Kordelle, J.; Gazova, Z.; Siebert, H.-C.; Steinmeyer, J. Metabolic Response of Human Osteoarthritic Cartilage to Biochemically Characterized Collagen Hydrolysates. Int. J. Mol. Sci. 2017, 18, 207. [Google Scholar] [CrossRef] [PubMed]
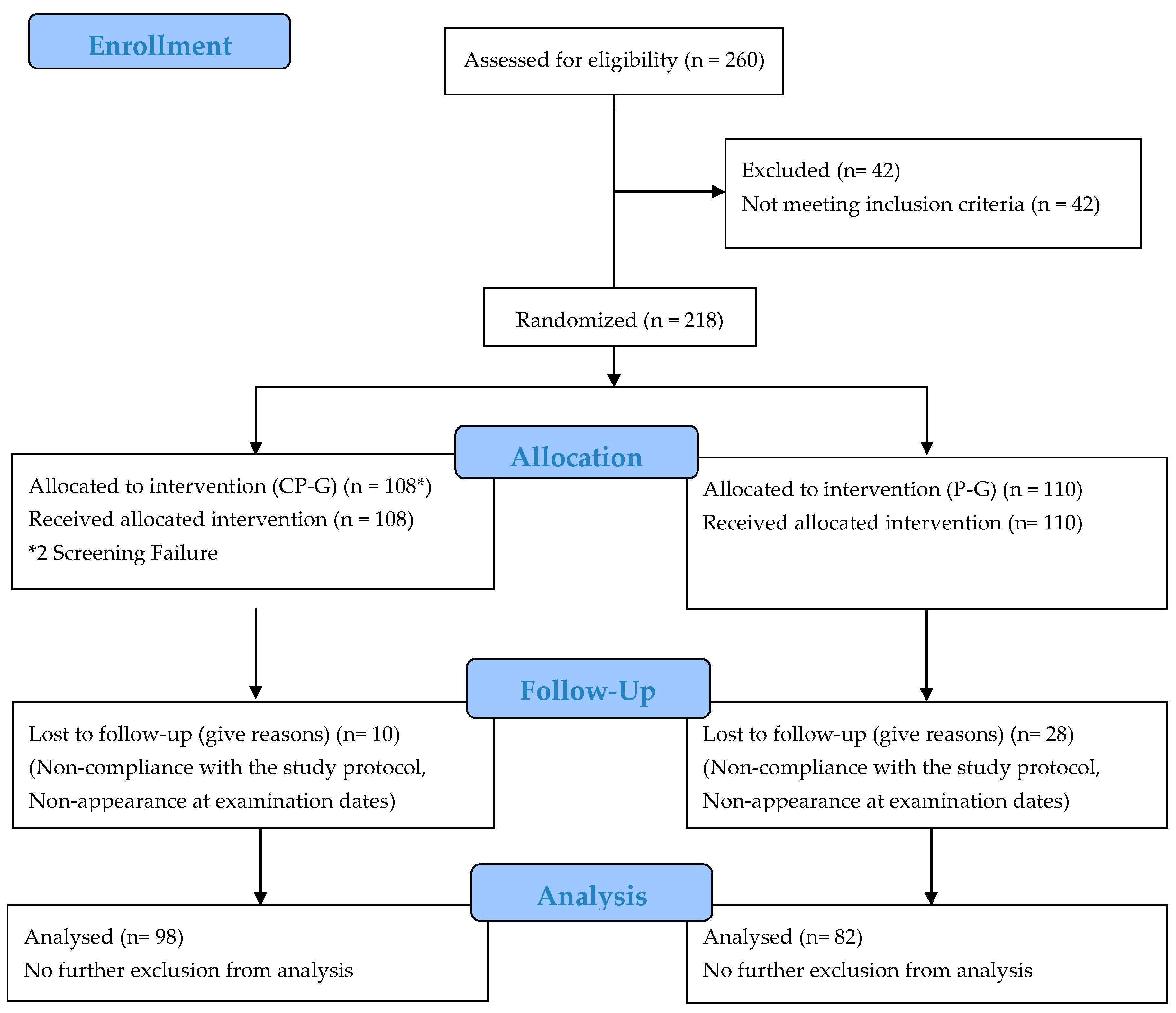
| Total (n = 180) | CP-G (n = 98) | P-G (n = 82) | p Value | |
|---|---|---|---|---|
| Age (years) | 23.9 ± 2.90 | 23.9 ± 3.07 | 23.8 ± 2.70 | 0.885 * |
| Gender (male/female) | 71/109 | 43/55 | 28/54 | 0.221 # |
| Height (m) | 1.74 ± 0.09 | 1.74 ± 0.09 | 1.73 ± 0.09 | 0.353 * |
| Body weight (kg) | 67.5 ± 10.8 | 68.1 ± 11.4 | 66.8 ± 10.0 | 0.578 * |
| BMI (kg/m2) | 22.3 ± 2.40 | 22.3 ± 2.53 | 22.2 ± 2.23 | 0.928 * |
| BP sys (mm Hg) | 121.1 ± 12.1 | 121.4 ± 12.2 | 120.8 ± 12.0 | 0.476 * |
| BP dia (mm Hg) | 74.2 ± 8.06 | 75.2 ± 8.71 | 73.1 ± 7.10 | 0.125 * |
| CP-G (n = 98) | P-G (n = 82) | p Value LMM | |||
|---|---|---|---|---|---|
| T0 | T12 | T0 | T12 | ||
| Pain during activity (subject) [mm] | 55.3 ± 15.8 | 33.4 ± 21.9 *** | 56.5 ± 15.0 | 40.8 ± 19.5 *** | 0.024 |
| Pain during activity (physician) [mm] | 55.5 ± 13.5 | 32.6 ± 20.3 *** | 55.4 ± 13.7 | 40.8 ± 18.1 *** | 0.003 |
| Pain during squat [mm] | 16.6 ± 21.5 | 7.80 ± 16.0 *** | 19.1 ± 21.7 | 10.1 ± 19.2 *** | 0.961 |
| Pain at rest [mm] | 13.5 ± 22.0 | 6.68 ± 17.5 ** | 18.3 ± 25.8 | 9.83 ± 19.3 ** | 0.628 |
| Extension [degree] | 5.82 ± 4.16 | 5.93 ± 3.78 | 6.12 ± 4.07 | 6.22 ± 3.55 | 0.979 |
| Flexion [degree] | 146.6 ± 7.68 | 146.4 ± 8.91 | 147.4 ± 6.62 | 148.2 ± 8.35 | 0.301 |
| Subgroup | CP-G (n = 98) | P-G (n = 82) | p Value LMM | |||
|---|---|---|---|---|---|---|
| T0 | T12 | T0 | T12 | |||
| Pain during activity (subject) [mm] | male | 51.6 ± 14.7 | 29.2 ± 21.3 *** | 54.0 ± 18.3 | 36.8 ± 22.9 *** | 0.028 |
| female | 58.0 ± 16.1 | 36.5 ± 22.0 *** | 57.7 ± 13.0 | 42.9 ± 17.3 *** | ||
| Pain during activity (physician) [mm] | male | 52.5 ± 13.1 | 29.3 ± 20.3 *** | 52.9 ± 15.6 | 35.9 ± 20.9 *** | 0.004 |
| female | 57.8 ± 13.4 | 35.0 ± 20.2 *** | 56.7 ± 12.6 | 43.3 ± 16.1 *** | ||
| Pain during squat [mm] | male | 15.6 ± 22.0 | 6.67 ± 16.4 ** | 18.1 ± 23.0 | 8.75 ± 20.3 ** | 0.954 |
| female | 17.4 ± 21.2 | 8.64 ± 15.8 ** | 19.6 ± 21.1 | 10.8 ± 18.8 ** | ||
| Pain at rest [mm] | male | 11.7 ± 21.8 | 1.62 ± 7.87 ** | 16.3 ± 23.3 | 10.8 ± 21.0 | 0.608 |
| female | 14.8 ± 22.3 | 10.5 ± 21.5 | 19.4 ± 27.1 | 9.33 ± 18.4 ** | ||
| Extension [degree] | male | 5.38 ± 4.01 | 6.02 ± 3.87 | 7.11 ± 3.46 | 7.50 ± 2.89 | 0.933 |
| female | 6.14 ± 4.28 | 5.86 ± 3.75 | 5.61 ± 4.29 | 5.56 ± 3.70 | ||
| Flexion [degree] | male | 146.8 ± 8.51 | 145.9 ± 8.57 | 146.4 ± 7.06 | 147.1 ± 8.28 | 0.341 |
| female | 146.4 ± 7.06 | 146.8 ± 9.21 | 147.9 ± 6.38 | 148.8 ± 8.40 | ||
| CP-G (n = 98) | P-G (n = 82) | |||
|---|---|---|---|---|
| T0 | T12 | T0 | T12 | |
| In total | 29 | 25 | 32 | 26 |
| Ointments and gels | 2 | 2 | 2 | 1 |
| Bandages and orthosis | 15 | 16 | 17 | 15 |
| Physiotherapy | 5 | 5 | 5 | 3 |
| various therapies 1 | 7 | 2 | 8 | 7 |
| p Value * | 0.022 | 0.012 | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zdzieblik, D.; Brame, J.; Oesser, S.; Gollhofer, A.; König, D. The Influence of Specific Bioactive Collagen Peptides on Knee Joint Discomfort in Young Physically Active Adults: A Randomized Controlled Trial. Nutrients 2021, 13, 523. https://doi.org/10.3390/nu13020523
Zdzieblik D, Brame J, Oesser S, Gollhofer A, König D. The Influence of Specific Bioactive Collagen Peptides on Knee Joint Discomfort in Young Physically Active Adults: A Randomized Controlled Trial. Nutrients. 2021; 13(2):523. https://doi.org/10.3390/nu13020523
Chicago/Turabian StyleZdzieblik, Denise, Judith Brame, Steffen Oesser, Albert Gollhofer, and Daniel König. 2021. "The Influence of Specific Bioactive Collagen Peptides on Knee Joint Discomfort in Young Physically Active Adults: A Randomized Controlled Trial" Nutrients 13, no. 2: 523. https://doi.org/10.3390/nu13020523
APA StyleZdzieblik, D., Brame, J., Oesser, S., Gollhofer, A., & König, D. (2021). The Influence of Specific Bioactive Collagen Peptides on Knee Joint Discomfort in Young Physically Active Adults: A Randomized Controlled Trial. Nutrients, 13(2), 523. https://doi.org/10.3390/nu13020523






