Association of Lower Nutritional Status and Education Level with the Severity of Depression Symptoms in Older Adults—A Cross Sectional Survey
Abstract
1. Introduction
2. Materials and Methods
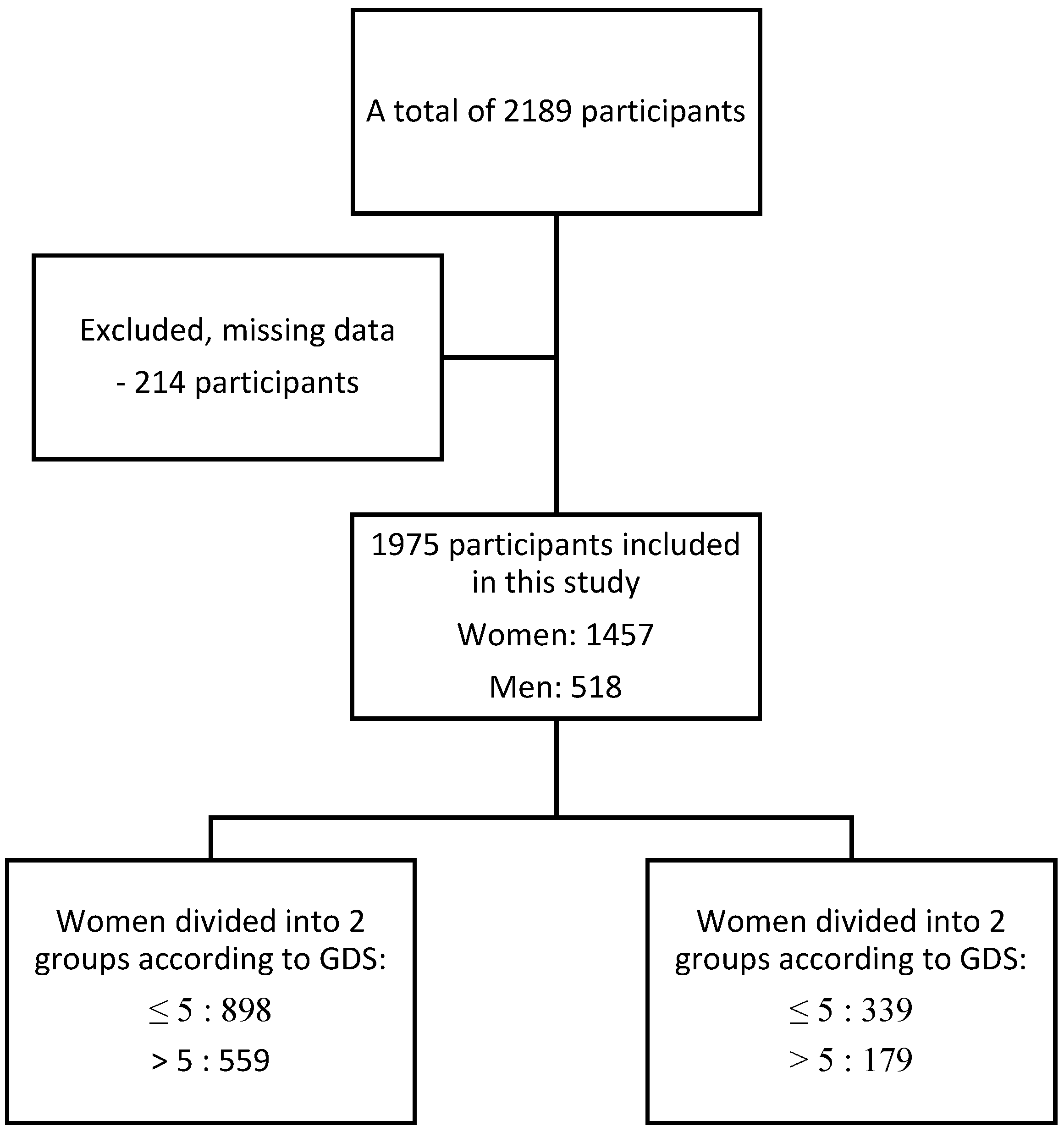
2.1. Design of the Study and Participants
2.2. Procedure
2.3. Statistical Analysis
2.4. Ethical Certification
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. Mental Health of Older Adults. Available online: https://www.who.int/news-room/fact-sheets/detail/mental-health-of-older-adults (accessed on 8 December 2020).
- World Health Organization. Depression. Available online: https://www.who.int/news-room/fact-sheets/detail/depression (accessed on 8 December 2020).
- Marx, W.; Moseley, G.; Berk, M.; Jacka, F. Nutritional psychiatry: The present state of the evidence. Proc. Nutr. Soc. 2017, 76, 427–436. [Google Scholar] [CrossRef] [PubMed]
- Molendijk, M.; Molero, P.; Ortuno Sanchez-Pedreno, F.; Van der Does, W.; Angel Martinez-Gonzalez, M. Diet quality and depression risk: A systematic review and dose-response meta-analysis of prospective studies. J. Affect. Disord 2018, 226, 346–354. [Google Scholar] [CrossRef]
- Tolkien, K.; Bradburn, S.; Murgatroyd, C. An anti-inflammatory diet as a potential intervention for depressive disorders: A systematic review and meta-analysis. Clin. Nutr. 2019, 38, 2045–2052. [Google Scholar] [CrossRef]
- Urquiza, M.; Fernandez, N.; Arrinda, I.; Sierra, I.; Irazusta, J.; Rodriguez Larrad, A. Nutritional Status Is Associated with Function, Physical Performance and Falls in Older Adults Admitted to Geriatric Rehabilitation: A Retrospective Cohort Study. Nutrients 2020, 12, 2855. [Google Scholar] [CrossRef] [PubMed]
- Dent, E.; Hoogendijk, E.O.; Visvanathan, R.; Wright, O.R.L. Malnutrition Screening and Assessment in Hospitalised Older People: A Review. J. Nutr. Health Aging 2019, 23, 431–441. [Google Scholar] [CrossRef] [PubMed]
- Favaro-Moreira, N.C.; Krausch-Hofmann, S.; Matthys, C.; Vereecken, C.; Vanhauwaert, E.; Declercq, A.; Bekkering, G.E.; Duyck, J. Risk Factors for Malnutrition in Older Adults: A Systematic Review of the Literature Based on Longitudinal Data. Adv. Nutr. 2016, 7, 507–522. [Google Scholar] [CrossRef]
- Naseer, M.; Forssell, H.; Fagerstrom, C. Malnutrition, functional ability and mortality among older people aged 60 years: A 7-year longitudinal study. Eur. J. Clin. Nutr. 2016, 70, 399–404. [Google Scholar] [CrossRef] [PubMed]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cereda, E.; Cruz-Jentoft, A.; Goisser, S.; de Groot, L.; Grosshauser, F.; Kiesswetter, E.; Norman, K.; et al. Management of Malnutrition in Older Patients-Current Approaches, Evidence and Open Questions. J. Clin. Med. 2019, 8, 974. [Google Scholar] [CrossRef] [PubMed]
- Cereda, E.; Veronese, N.; Caccialanza, R. The final word on nutritional screening and assessment in older persons. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 24–29. [Google Scholar] [CrossRef]
- Leij-Halfwerk, S.; Verwijs, M.H.; van Houdt, S.; Borkent, J.W.; Guaitoli, P.R.; Pelgrim, T.; Heymans, M.W.; Power, L.; Visser, M.; Corish, C.A.; et al. Prevalence of protein-energy malnutrition risk in European older adults in community, residential and hospital settings, according to 22 malnutrition screening tools validated for use in adults ≥65 years: A systematic review and meta-analysis. Maturitas 2019, 126, 80–89. [Google Scholar] [CrossRef] [PubMed]
- Wysokinski, A.; Sobow, T.; Kloszewska, I.; Kostka, T. Mechanisms of the anorexia of aging-a review. Age (Dordr) 2015, 37, 9821. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, S.; Baral, B.K.; Pokhrel, B.R.; Pokhrel, A.; Acharya, A.; Amatya, D.; Amatya, P.; Mishra, S.R. Depression, malnutrition, and health-related quality of life among Nepali older patients. BMC Geriatr. 2018, 18, 191. [Google Scholar] [CrossRef]
- Wei, J.; Fan, L.; Zhang, Y.; Li, S.; Partridge, J.; Claytor, L.; Sulo, S. Association Between Malnutrition and Depression Among Community-Dwelling Older Chinese Adults. Asia Pac. J. Public Health 2018, 30, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Kwan, R.Y.C.; Leung, A.Y.M.; Yee, A.; Lau, L.T.; Xu, X.Y.; Dai, D.L.K. Cognitive Frailty and Its Association with Nutrition and Depression in Community-Dwelling Older People. J. Nutr. Health Aging 2019, 23, 943–948. [Google Scholar] [CrossRef]
- Poland Statistics. The Situation of Older People in Poland in 2018. Statistical Office in Białystok. 2018. Available online: https://stat.gov.pl/en/topics/older-people/older-people/the-situation-of-older-people-in-poland-in-2018,1,1.html (accessed on 8 December 2020).
- Mundi Index. Poland Demographics Profile. Available online: https://www.indexmundi.com/poland/demographics_profile.html (accessed on 26 January 2021).
- Bledowski, P.; Mossakowska, M.; Chudek, J.; Grodzicki, T.; Milewicz, A.; Szybalska, A.; Wieczorowska-Tobis, K.; Wiecek, A.; Bartoszek, A.; Dabrowski, A.; et al. Medical, psychological and socioeconomic aspects of aging in Poland: Assumptions and objectives of the PolSenior project. Exp. Gerontol. 2011, 46, 1003–1009. [Google Scholar] [CrossRef] [PubMed]
- Pocklington, C.; Gilbody, S.; Manea, L.; McMillan, D. The diagnostic accuracy of brief versions of the Geriatric Depression Scale: A systematic review and meta-analysis. Int. J. Geriatr. Psychiatry 2016, 31, 837–857. [Google Scholar] [CrossRef]
- Yesavage, J.A.; Brink, T.L.; Rose, T.L.; Lum, O.; Huang, V.; Adey, M.; Leirer, V.O. Development and validation of a geriatric depression screening scale: A preliminary report. J. Psychiatr. Res. 1982, 17, 37–49. [Google Scholar] [CrossRef]
- Guigoz, Y.; Vellas, B.; Garry, P.J. Assessing the nutritional status of the elderly: The Mini Nutritional Assessment as part of the geriatric evaluation. Nutr. Rev. 1996, 54, 59–65. [Google Scholar] [CrossRef]
- Kessler, R.C.; Birnbaum, H.; Bromet, E.; Hwang, I.; Sampson, N.; Shahly, V. Age differences in major depression: Results from the National Comorbidity Survey Replication (NCS-R). Psychol. Med. 2010, 40, 225–237. [Google Scholar] [CrossRef]
- Klimova, B.; Novotny, M.; Valis, M. The Impact of Nutrition and Intestinal Microbiome on Elderly Depression—A Systematic Review. Nutrients 2020, 12, 710. [Google Scholar] [CrossRef]
- Vafaei, Z.; Mokhtari, H.; Sadooghi, Z.; Meamar, R.; Chitsaz, A.; Moeini, M. Malnutrition is associated with depression in rural elderly population. J. Res. Med. Sci. 2013, 18, S15–S19. [Google Scholar] [PubMed]
- Ahmadi, S.M.; Mohammadi, M.R.; Mostafavi, S.A.; Keshavarzi, S.; Kooshesh, S.M.; Joulaei, H.; Sarikhani, Y.; Peimani, P.; Heydari, S.T.; Lankarani, K.B. Dependence of the geriatric depression on nutritional status and anthropometric indices in elderly population. Iran. J. Psychiatry 2013, 8, 92–96. [Google Scholar] [PubMed]
- Mantzorou, M.; Vadikolias, K.; Pavlidou, E.; Serdari, A.; Vasios, G.; Tryfonos, C.; Giaginis, C. Nutritional status is associated with the degree of cognitive impairment and depressive symptoms in a Greek elderly population. Nutr. Neurosci. 2020, 23, 201–209. [Google Scholar] [CrossRef] [PubMed]
- Polyzos, S.A.; Margioris, A.N. Sarcopenic obesity. Hormones (Athens) 2018, 17, 321–331. [Google Scholar] [CrossRef] [PubMed]
- Santanasto, A.J.; Goodpaster, B.H.; Kritchevsky, S.B.; Miljkovic, I.; Satterfield, S.; Schwartz, A.V.; Cummings, S.R.; Boudreau, R.M.; Harris, T.B.; Newman, A.B. Body Composition Remodeling and Mortality: The Health Aging and Body Composition Study. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 513–519. [Google Scholar] [CrossRef]
- Ishida, Y.; Maeda, K.; Nonogaki, T.; Shimizu, A.; Yamanaka, Y.; Matsuyama, R.; Kato, R.; Mori, N. Impact of edema on length of calf circumference in older adults. Geriatr. Gerontol. Int. 2019, 19, 993–998. [Google Scholar] [CrossRef]
- Wada, O.; Kurita, N.; Kamitani, T.; Mizuno, K. Implications of evaluating leg muscle mass and fat mass separately for quadriceps strength in knee osteoarthritis: The SPSS-OK study. Clin. Rheumatol. 2020, 39, 1655–1661. [Google Scholar] [CrossRef]
- Verbrugge, F.H.; Bertrand, P.B.; Willems, E.; Gielen, E.; Mullens, W.; Giri, S.; Tang, W.H.W.; Raman, S.V.; Verhaert, D. Global myocardial oedema in advanced decompensated heart failure. Eur. Heart J. Cardiovasc. Imaging 2017, 18, 787–794. [Google Scholar] [CrossRef]
- Sanada, K.; Chen, R.; Willcox, B.; Ohara, T.; Wen, A.; Takenaka, C.; Masaki, K. Association of sarcopenic obesity predicted by anthropometric measurements and 24-y all-cause mortality in elderly men: The Kuakini Honolulu Heart Program. Nutrition 2018, 46, 97–102. [Google Scholar] [CrossRef]
- Huang, C.Q.; Dong, B.R.; Lu, Z.C.; Yue, J.R.; Liu, Q.X. Chronic diseases and risk for depression in old age: A meta-analysis of published literature. Ageing Res. Rev. 2010, 9, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Engel, J.H.; Siewerdt, F.; Jackson, R.; Akobundu, U.; Wait, C.; Sahyoun, N. Hardiness, depression, and emotional well-being and their association with appetite in older adults. J. Am. Geriatr Soc. 2011, 59, 482–487. [Google Scholar] [CrossRef]
- World Health Organization. Obesity: Preventing and managing the global epidemic. In Report of a WHO consultation; WHO Technical Report Series 894; WHO: Geneva, Switzerland, 2000; 252p. [Google Scholar]
- Savva, S.C.; Lamnisos, D.; Kafatos, A.G. Predicting cardiometabolic risk: Waist-to-height ratio or BMI. A meta-analysis. Diabetes Metab. Syndr. Obes. 2013, 6, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Punyanitya, M.; Chen, J.; Gallagher, D.; Albu, J.; Pi-Sunyer, X.; Lewis, C.E.; Grunfeld, C.; Heshka, S.; Heymsfield, S.B. Waist circumference correlates with metabolic syndrome indicators better than percentage fat. Obesity (Silver Spring) 2006, 14, 727–736. [Google Scholar] [CrossRef]
- Zhao, G.; Ford, E.S.; Li, C.; Tsai, J.; Dhingra, S.; Balluz, L.S. Waist circumference, abdominal obesity, and depression among overweight and obese U.S. adults: National Health and Nutrition Examination Survey 2005–2006. BMC Psychiatry 2011, 11, 130. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Anderson, D.; Lurie-Beck, J. The relationship between abdominal obesity and depression in the general population: A systematic review and meta-analysis. Obes. Res. Clin. Pract. 2011, 5, e267–e360. [Google Scholar] [CrossRef] [PubMed]
- Hirani, V. Generalised and abdominal adiposity are important risk factors for chronic disease in older people: Results from a nationally representative survey. J. Nutr. Health Aging 2011, 15, 469–478. [Google Scholar] [CrossRef]
- Nasimi, N.; Dabbaghmanesh, M.H.; Sohrabi, Z. Nutritional status and body fat mass: Determinants of sarcopenia in community-dwelling older adults. Exp. Gerontol. 2019, 122, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Landi, F.; Onder, G.; Russo, A.; Liperoti, R.; Tosato, M.; Martone, A.M.; Capoluongo, E.; Bernabei, R. Calf circumference, frailty and physical performance among older adults living in the community. Clin. Nutr. 2014, 33, 539–544. [Google Scholar] [CrossRef]
- Bonnefoy, M.; Jauffret, M.; Kostka, T.; Jusot, J.F. Usefulness of calf circumference measurement in assessing the nutritional state of hospitalized elderly people. Gerontology 2002, 48, 162–169. [Google Scholar] [CrossRef]
- Silva, D.F.O.; Lima, S.; Sena-Evangelista, K.C.M.; Marchioni, D.M.; Cobucci, R.N.; Andrade, F.B. Nutritional Risk Screening Tools for Older Adults with COVID-19: A Systematic Review. Nutrients 2020, 12, 2956. [Google Scholar] [CrossRef] [PubMed]
- Chang-Quan, H.; Zheng-Rong, W.; Yong-Hong, L.; Yi-Zhou, X.; Qing-Xiu, L. Education and risk for late life depression: A meta-analysis of published literature. Int. J. Psychiatry Med. 2010, 40, 109–124. [Google Scholar] [CrossRef] [PubMed]
- Da Costa Dias, F.L.; Teixeira, A.L.; Guimaraes, H.C.; Santos, A.P.B.; Resende, E.P.F.; Machado, J.C.B.; Barbosa, M.T.; Caramelli, P. The influence of age, sex and education on the phenomenology of depressive symptoms in a population-based sample aged 75+ years with major depression: The Pieta Study. Aging Ment. Health 2019. Online ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Jung, S.E.; Kim, S.; Bishop, A.; Hermann, J. Poor Nutritional Status among Low-Income Older Adults: Examining the Interconnection between Self-Care Capacity, Food Insecurity, and Depression. J. Acad. Nutr. Diet. 2019, 119, 1687–1694. [Google Scholar] [CrossRef]
- Chrzastek, Z.; Guligowska, A.; Piglowska, M.; Soltysik, B.; Kostka, T. Association between sucrose and fiber intake and symptoms of depression in older people. Nutr. Neurosci. 2020, 1–12. [Google Scholar] [CrossRef]
- Schefft, C.; Kilarski, L.L.; Bschor, T.; Kohler, S. Efficacy of adding nutritional supplements in unipolar depression: A systematic review and meta-analysis. Eur. Neuropsychopharmacol. 2017, 27, 1090–1109. [Google Scholar] [CrossRef]
- Velazquez-Alva, M.C.; Irigoyen-Camacho, M.E.; Cabrer-Rosales, M.F.; Lazarevich, I.; Arrieta-Cruz, I.; Gutierrez-Juarez, R.; Zepeda-Zepeda, M.A. Prevalence of Malnutrition and Depression in Older Adults Living in Nursing Homes in Mexico City. Nutrients 2020, 12, 2429. [Google Scholar] [CrossRef]
- Piglowska, M.; Guligowska, A.; Kostka, T. Nutritional Status Plays More Important Role in Determining Functional State in Older People Living in the Community than in Nursing Home Residents. Nutrients 2020, 12, 2042. [Google Scholar] [CrossRef]
| Variable | All (n = 1975) | Women (n = 1457) | Men (n = 518) | p-Value |
|---|---|---|---|---|
| Age [years] | 75 (67–80) | 75 (67–81) | 73 (66–78) | <0.001 a |
| Education [years] | 11 (7–14) | 11 (7–13) | 11 (7–15) | 0.023 a |
| BMI [kg/m2] | 26.9 (24.1–30.1) | 26.9 (23.9–30.5) | 26.7 (24.5–29.5) | ns a |
| Waist circumference [cm] | 93 (84–102) | 91 (82–100) | 99 (92–107) | <0.001 a |
| Hips circumference [cm] | 104 (98–111) | 105 (98–112) | 102 (98–107) | <0.001 a |
| Calf circumference [cm] | 36 (33–38) | 35 (33–38) | 36 (34–39) | <0.001 a |
| WHtR | 57.9 (52.6–63.6) | 58.1 (52.2–63.7) | 57.7 (53.9–62.9) | ns a |
| WHR | 0.89 (0.83–0.94) | 0.86 (0.81–0.91) | 0.97 (0.93–1.01) | <0.001 a |
| MNA | 25.5 (22.5–27.5) | 25.5 (22.5–27.5) | 25.7 (23.5–27.5) | ns a |
| Depression [n (%)] | 241 (12.2) | 196 (13.4) | 45 (8.7) | 0.004 b |
| Hypertension [n (%)] | 1330 (67.3) | 995 (68.2) | 335 (64.7) | ns b |
| Stroke [n (%)] | 233 (11.8) | 167 (11.5) | 66 (12.7) | ns b |
| Cancer [n (%)] | 165 (8.4) | 118 (8.1) | 47 (9.1) | ns b |
| Osteoporosis [n (%)] | 485 (24.6) | 403 (27.7) | 82 (15.8) | <0.001 b |
| COPD [n (%)] | 114 (5.8) | 86 (5.9) | 28 (5.4) | ns b |
| Congestive heart failure [n (%)] | 745 (37.7) | 533 (36.6) | 212 (40.9) | ns b |
| Diabetes [n (%)] | 376 (19.1) | 266 (18.3) | 110 (21.3) | ns b |
| Myocardial infarction [n (%)] | 214 (10.8) | 134 (9.2) | 80 (15.4) | <0.001 b |
| Variable | Women GDS ≤ 5 (n = 898) Median (Quartiles) | Women GDS > 5 (n = 559) Median (Quartiles) | Men GDS ≤ 5 (n = 339) Median (Quartiles) | Men GDS > 5 (n = 179) Median (Quartiles) |
|---|---|---|---|---|
| Age [years] a | 74 (66–79) | 77 (70–83) *** | 74 (66–79) | 72 (68–77) |
| Education [years] a | 12 (8–16) | 7 (7–12) *** | 12 (9.5–16) | 10 (7–12) *** |
| BMI [kg/m2] a | 27 (24.2–30.1) | 26.7 (23.4–30.1) | 27 (24.6–29.7) | 26.5 (24.1–29.4) |
| Waist circumference [cm] a | 90 (82–99) | 91 (82–102) | 99 (93–106) | 100 (90–108) |
| Hips circumference [cm] a | 105 (99–112) | 105 (98–114) | 102 (98–107) | 102 (96–107) |
| Calf circumference [cm] a | 36 (33–38) | 34 (32–37) *** | 57.2 (54–62) | 36 (33–38) * |
| WHtR a | 57.4 (52.3–62.9) | 58.8 (52.1–65.6) * | 57.2 (54–62) | 58.7 (53–63.8) |
| WHR a | 0.86 (0.81–0.91) | 0.87 (0.82–0.91) | 0.97 (0.93–1) | 0.96 (0.93–1) |
| MNA a | 26.5 (24–28) | 23 (20.5–26) *** | 26.5 (24.5–28) | 24 (20.5–26.5) *** |
| Depression [n (%)] b | 107 (11.9) | 89 (15.9) * | 17 (5) | 28 (15.6) *** |
| Hypertension [n (%)] b | 583 (64.9) | 412 (73.7) *** | 218 (64.3) | 117 (65.4) |
| Stroke [n (%)] b | 70 (7.8) | 97 (17.3) *** | 31 (9.1) | 35 (19.5) *** |
| Cancer [n (%)] b | 76 (8.5) | 42 (7.5) | 29 (8.5) | 18 (10.1) |
| Osteoporosis [n (%)] b | 243 (27.1) | 160 (28.6) | 57 (16.8) | 25 (14) |
| COPD [n (%)] b | 62 (6.9) | 24 (4.3) * | 17 (5) | 11 (6.2) |
| Congestive heart failure [n (%)] b | 291 (32.4) | 242 (43.3) *** | 122 (36) | 90 (50.3) ** |
| Diabetes [n (%)] b | 141 (15.7) | 125 (22.4) *** | 72 (21.2) | 38 (21.3) |
| Myocardial infarction [n (%)] b | 71 (7.9) | 63 (11.3) * | 50 (14.7) | 30 (16.8) |
| Parameters | Women | Men |
|---|---|---|
| rS (rP) | rS (rP) | |
| Age [years] | 0.25 *** (0.22 ***) | −0.02 (−0.04) |
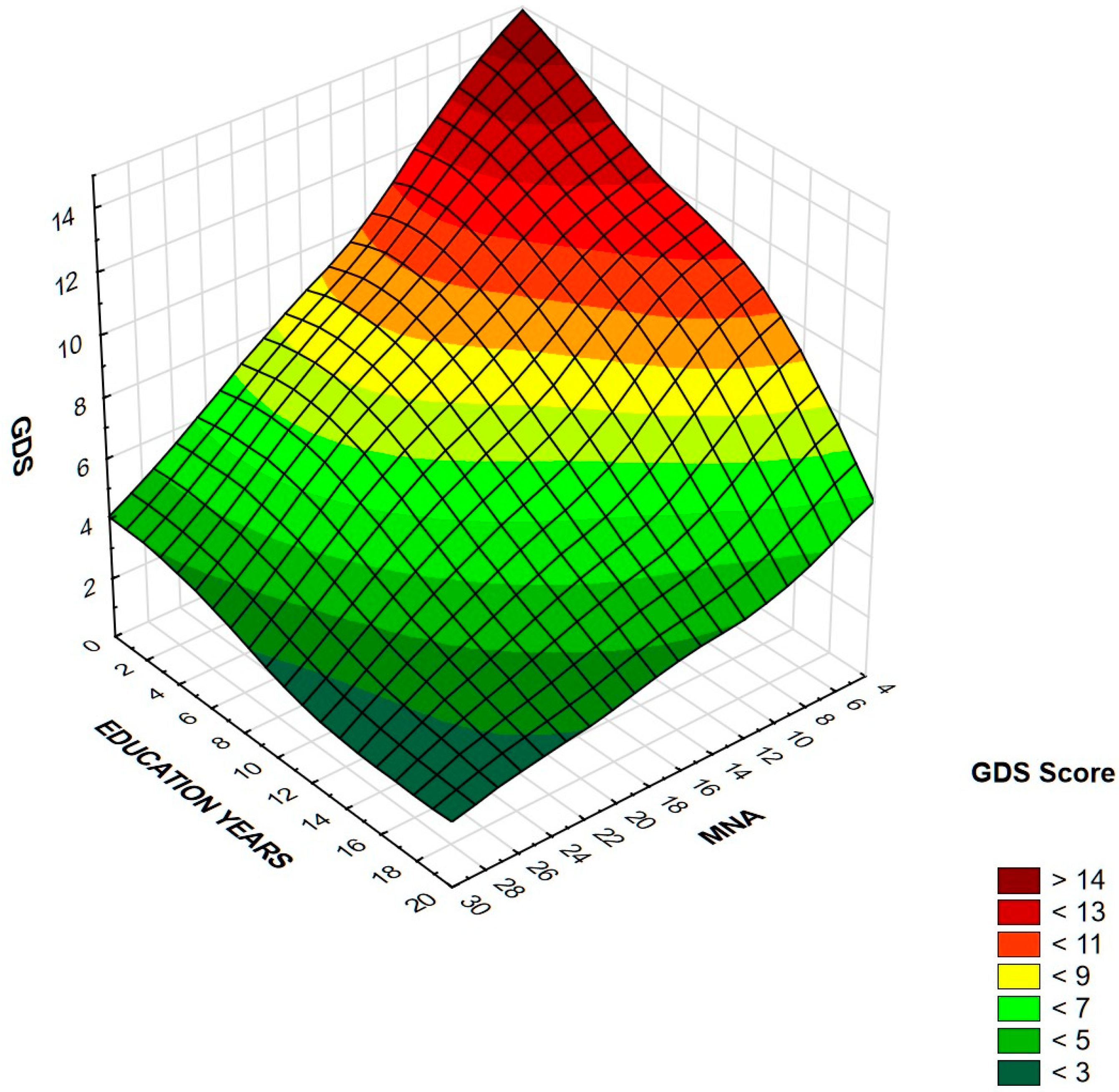
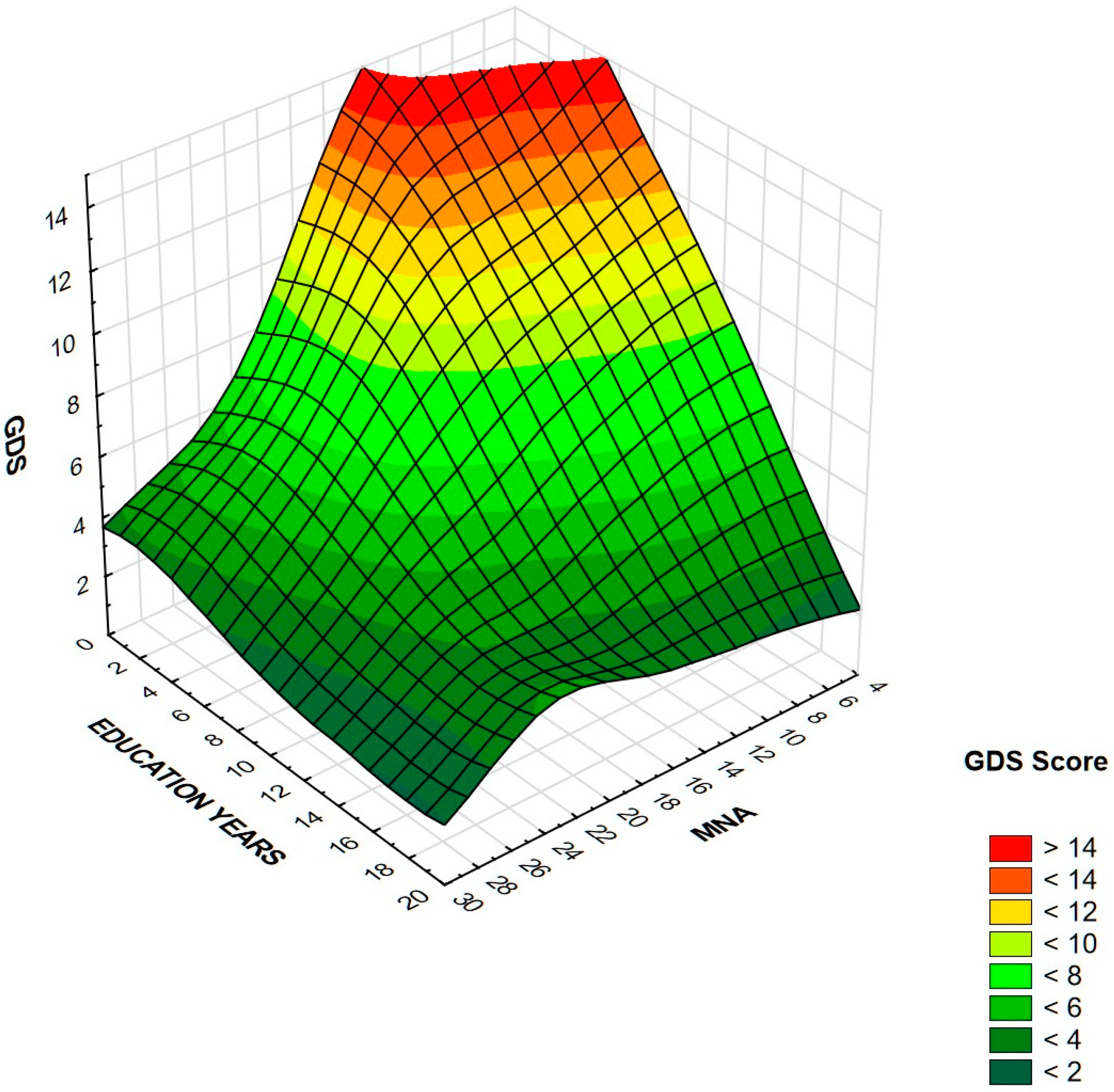
| Education [years] | −0.37 *** (−0.36 ***) | −0.31 *** (−0.30 ***) |
| BMI [kg/m2] | −0.03 (0.00) | −0.06 (−0.06) |
| Waist circumference [cm] | 0.04 (0.04) | −0.001 (−0.04) |
| Hips circumference [cm] | 0.04 (0.05) | −0.004 (0.003) |
| Calf circumference [cm] | −0.17 *** (−0.13 ***) | −0.13 ** (−0.11 **) |
| WHR | 0.03 (−0.01) | −0.03 (−0.07) |
| WHtR | 0.06 * (0.05 *) | −0.02 (0.002) |
| MNA | −0.42 *** (−0.38 ***) | −0.40 *** (−0.45 ***) |
| Variable | Women | Men | ||||
|---|---|---|---|---|---|---|
| OR | 95%CI | p | OR | 95%CI | p | |
| Education [years] | 0.87 | 0.84–0.90 | <0.001 | 0.9 | 0.85–0.95 | <0.001 |
| MNA | 0.83 | 0.80–0.86 | <0.001 | 0.81 | 0.75–0.87 | <0.001 |
| WHtR | 1.02 | 1.002–1.03 | 0.03 | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chrzastek, Z.; Guligowska, A.; Soltysik, B.; Pigłowska, M.; Borowiak, E.; Kostka, J.; Kostka, T. Association of Lower Nutritional Status and Education Level with the Severity of Depression Symptoms in Older Adults—A Cross Sectional Survey. Nutrients 2021, 13, 515. https://doi.org/10.3390/nu13020515
Chrzastek Z, Guligowska A, Soltysik B, Pigłowska M, Borowiak E, Kostka J, Kostka T. Association of Lower Nutritional Status and Education Level with the Severity of Depression Symptoms in Older Adults—A Cross Sectional Survey. Nutrients. 2021; 13(2):515. https://doi.org/10.3390/nu13020515
Chicago/Turabian StyleChrzastek, Zuzanna, Agnieszka Guligowska, Bartlomiej Soltysik, Malgorzata Pigłowska, Ewa Borowiak, Joanna Kostka, and Tomasz Kostka. 2021. "Association of Lower Nutritional Status and Education Level with the Severity of Depression Symptoms in Older Adults—A Cross Sectional Survey" Nutrients 13, no. 2: 515. https://doi.org/10.3390/nu13020515
APA StyleChrzastek, Z., Guligowska, A., Soltysik, B., Pigłowska, M., Borowiak, E., Kostka, J., & Kostka, T. (2021). Association of Lower Nutritional Status and Education Level with the Severity of Depression Symptoms in Older Adults—A Cross Sectional Survey. Nutrients, 13(2), 515. https://doi.org/10.3390/nu13020515







