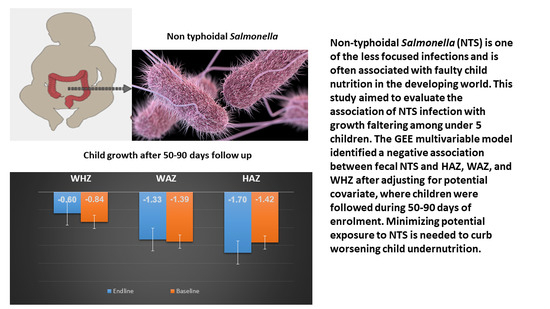
Association between Non-Typhoidal Salmonella Infection and Growth in Children under 5 Years of Age: Analyzing Data from the Global Enteric Multicenter Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Site
2.2. Study Design and Study Participants
2.3. Stool Sample Collection and Fecal Microbiology
2.4. Outcome Variable
2.5. Variables of Interest
2.5.1. Anthropometry Measurements
2.5.2. Diarrhea
2.5.3. Vomiting and Fever
2.5.4. Breastfeeding Status
2.5.5. Sociodemographic Information
2.5.6. Wealth Index
2.5.7. Household Follow-up Visit
2.6. Statistical Analysis
2.7. Ethical Consideration
3. Results
3.1. Characteristics of the Study Population
3.2. Child Growth and Fecal NTS
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kariuki, S.; Mbae, C.; Van Puyvelde, S.; Onsare, R.; Kavai, S.; Wairimu, C.; Ngetich, R.; Clemens, J.; Dougan, G. High relatedness of invasive multi-drug resistant non-typhoidal Salmonella genotypes among patients and asymptomatic carriers in endemic informal settlements in Kenya. PLoS Negl. Trop. Dis. 2020, 14, e0008440. [Google Scholar] [CrossRef] [PubMed]
- Majowicz, S.E.; Musto, J.; Scallan, E.; Angulo, F.J.; Kirk, M.; O’Brien, S.J.; Jones, T.F.; Fazil, A.; Hoekstra, R.M. The global burden of nontyphoidal Salmonella gastroenteritis. Clin. Infect. Dis. 2010, 50, 882–889. [Google Scholar] [CrossRef] [PubMed]
- Murray, C.J.; Rosenfeld, L.C.; Lim, S.S.; Andrews, K.G.; Foreman, K.J.; Haring, D.; Fullman, N.; Naghavi, M.; Lozano, P.R.; Lopez, P.A.D. Global malaria mortality between 1980 and 2010: A systematic analysis. Lancet 2012, 379, 413–431. [Google Scholar] [CrossRef]
- Murray, C.J.; Vos, T.; Lozano, R.; Naghavi, M.; Flaxman, A.D.; Michaud, C.; Ezzati, P.M.; Shibuya, P.K.; Salomon, P.J.A.; Abdalla, S.; et al. Disability-adjusted life years (DALYs) for 291 diseases and injuries in 21 regions, 1990-2010: A systematic analysis for the Global Burden of Disease Study 2010. Lancet 2012, 380, 2197–2223. [Google Scholar] [CrossRef]
- Grisaru-Soen, G.; Wysoki, M.G.; Keller, N. Risk factors for development of nontyphoid Salmonella bacteremia. Clin. Pediatr. 2004, 43, 825–829. [Google Scholar] [CrossRef]
- Feasey, N.A.; Dougan, G.; Kingsley, R.A.; Heyderman, R.S.; Gordon, M.A. Invasive non-typhoidal salmonella disease: An emerging and neglected tropical disease in Africa. Lancet 2012, 379, 2489–2499. [Google Scholar] [CrossRef]
- Menezes, G.A.; Khan, M.A.; Harish, B.N.; Parija, S.C.; Goessens, W.; Vidyalakshmi, K.; Baliga, S.; Hays, J.P. Molecular characterization of antimicrobial resistance in non-typhoidal salmonellae associated with systemic manifestations from India. J. Med. Microbiol. 2010, 59, 1477–1483. [Google Scholar] [CrossRef]
- Jabeen, K.; Zafar, A.; Irfan, S.; Khan, E.; Mehraj, V.; Hasan, R. Increase in isolation of extended spectrum beta lactamase producing multidrug resistant non typhoidal Salmonellae in Pakistan. BMC Infect. Dis. 2010, 10, 101. [Google Scholar] [CrossRef]
- Kumar, Y.; Sharma, A.; Sehgal, R.; Kumar, S. Distribution trends of Salmonella serovars in India (2001–2005). Trans. R. Soc. Trop. Med. Hyg. 2009, 103, 390–394. [Google Scholar] [CrossRef]
- Nga, T.V.T.; Parry, C.M.; Le, T.; Lan, N.P.H.; Diep, T.S.; Campbell, J.I.; Hoang, N.V.; Dung, L.; Wain, J.; Dolecek, C.; et al. The decline of typhoid and the rise of non-typhoid salmonellae and fungal infections in a changing HIV landscape: Bloodstream infection trends over 15 years in southern Vietnam. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 26–34. [Google Scholar] [CrossRef]
- Kiratisin, P. Bacteraemia due to non-typhoidal Salmonella in Thailand: Clinical and microbiological analysis. Trans. R. Soc. Trop. Med. Hyg. 2008, 102, 384–388. [Google Scholar] [CrossRef]
- Acheson, D.; Hohmann, E.L. Nontyphoidal salmonellosis. Clin. Infect. Dis. 2001, 32, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Gorospe, E.C.; Oxentenko, A.S. Nutritional consequences of chronic diarrhoea. Best Pract. Res. Clin. Gastroenterol. 2012, 26, 663–675. [Google Scholar] [CrossRef] [PubMed]
- Shahunja, K.M.; Leung, D.T.; Ahmed, T.; Bardhan, P.K.; Ahmed, D.; Qadri, F.; Ryan, E.T.; Chisti, M.J. Factors Associated with Non-typhoidal Salmonella Bacteremia versus Typhoidal Salmonella Bacteremia in Patients Presenting for Care in an Urban Diarrheal Disease Hospital in Bangladesh. PLoS Negl. Trop. Dis. 2015, 9, e0004066. [Google Scholar] [CrossRef] [PubMed]
- Brent, A.J.; Oundo, J.O.; Mwangi, I.; Ochola, L.; Lowe, B.; Berkley, J.A. Salmonella bacteremia in Kenyan children. Pediatr. Infect. Dis. J. 2006, 25, 230–236. [Google Scholar] [CrossRef]
- Berkley, J.A.; Lowe, B.S.; Mwangi, I.; Williams, T.; Bauni, E.; Mwarumba, S.; Ngetsa, C.; Slack, M.P.E.; Njenga, S.; Hart, C.A.; et al. Bacteremia among children admitted to a rural hospital in Kenya. N. Engl. J. Med. 2005, 352, 39–47. [Google Scholar] [CrossRef]
- Farag, T.H.; Nasrin, D.; Wu, Y.; Muhsen, K.; Blackwelder, W.C.; Sommerfelt, H.; Panchalingam, S.; Nataro, J.P.; Kotloff, K.L.; Levine, M.M. Some epidemiologic, clinical, microbiologic, and organizational assumptions that influenced the design and performance of the Global Enteric Multicenter Study (GEMS). Clin. Infect. Dis. 2012, 55, S225–S231. [Google Scholar] [CrossRef]
- Kotloff, K.L.; Blackwelder, W.C.; Nasrin, D.; Nataro, J.P.; Farag, T.H.; Van Eijk, A.; Adegbola, R.A.; Alonso, P.L.; Breiman, R.F.; Faruque, A.S.G.; et al. The Global Enteric Multicenter Study (GEMS) of diarrheal disease in infants and young children in developing countries: Epidemiologic and clinical methods of the case/control study. Clin. Infect. Dis. 2012, 55, S232–S245. [Google Scholar] [CrossRef]
- Levine, M.M.; Kotloff, K.L.; Nataro, J.P.; Muhsen, K. The Global Enteric Multicenter Study (GEMS): Impetus, rationale, and genesis. Clin. Infect. Dis. 2012, 55, S215–S224. [Google Scholar] [CrossRef]
- Kotloff, K.L.; Nataro, J.P.; Blackwelder, W.C.; Nasrin, D.; Farag, T.H.; Panchalingam, S.; Wu, Y.; Sow, S.O.; Sur, D.; Breiman, R.F.; et al. Burden and aetiology of diarrhoeal disease in infants and young children in developing countries (the Global Enteric Multicenter Study, GEMS): A prospective, case-control study. Lancet 2013, 382, 209–222. [Google Scholar] [CrossRef]
- Livio, S.; Strockbine, N.A.; Panchalingam, S.; Tennant, S.M.; Barry, E.M.; Marohn, M.E.; Antonio, M.; Hossain, A.; Mandomando, I.; Ochieng, J.B.; et al. Shigella isolates from the global enteric multicenter study inform vaccine development. Clin. Infect. Dis. 2014, 59, 933–941. [Google Scholar] [CrossRef] [PubMed]
- Blackwelder, W.C.; Biswas, K.; Wu, Y.; Kotloff, K.L.; Farag, T.H.; Nasrin, D.; Graubard, B.I.; Sommerfelt, H.; Levine, M.M. Statistical methods in the Global Enteric Multicenter Study (GEMS). Clin. Infect. Dis. 2012, 55, S246–S253. [Google Scholar] [CrossRef] [PubMed]
- Sow, S.O.; Muhsen, K.; Nasrin, D.; Blackwelder, W.C.; Wu, Y.; Farag, T.H.; Panchalingam, S.; Sur, D.; Zaidi, A.K.M.; Faruque, A.S.G.; et al. The Burden of Cryptosporidium Diarrheal Disease among Children <24 Months of Age in Moderate/High Mortality Regions of Sub-Saharan Africa and South Asia, Utilizing Data from the Global Enteric Multicenter Study (GEMS). PLoS Negl. Trop. Dis. 2016, 10, e0004729. [Google Scholar] [CrossRef]
- Panchalingam, S.; Antonio, M.; Hossain, A.; Mandomando, I.; Ochieng, B.; Oundo, J.; Ramamurthy, T.; Tamboura, B.; Zaidi, A.K.M.; Petri, W.; et al. Diagnostic microbiologic methods in the GEMS-1 case/control study. Clin. Infect. Dis. 2012, 55, S294–S302. [Google Scholar] [CrossRef] [PubMed]
- Lindsay, B.; Oundo, J.; Hossain, M.A.; Antonio, M.; Tamboura, B.; Walker, A.; Paulson, J.N.; Parkhill, J.; Omore, R.; Faruque, A.S.G.; et al. Microbiota That Affect Risk for Shigellosis in Children in Low-Income Countries. Emerg. Infect. Dis. J. 2015, 21, 242. [Google Scholar] [CrossRef] [PubMed]
- Cogill, B. Anthropometric Indicators Measurement Guide; Food and Nutrition Technical Assistance Project, Academy for Educational Development: Washington, DC, USA, 2003. [Google Scholar]
- El Mouzan, M.I.; Foster, P.J.; Al Herbish, A.S.; Al Salloum, A.A.; Al Omar, A.A.; Qurachi, M.M. Prevalence of malnutrition in Saudi children: A community-based study. Ann. Saudi Med. 2010, 30, 381–385. [Google Scholar] [CrossRef]
- World Health Organization. WHO Child Growth Standards: Length/Height-for-Age, Weight-for-Age, Weight-for-Length, Weight -for-Height and Body Mass Index-for-Age: Methods and Development; World Health Organization: Geneva, Switzerland, 2006. [Google Scholar]
- World Health Organization. WHO Child Growth Standards, SAS Macro, version 3.2.2; World Health Organization: Geneva, Switzerland, 2012. [Google Scholar]
- Baqui, A.H.; Black, R.E.; Yunus, M.D.; Hoque, A.R.A.; Chowdhury, H.R.; Sack, R.B. Methodological issues in diarrhoeal diseases epidemiology: Definition of diarrhoeal episodes. Int. J. Epidemiol. 1991, 20, 1057–1063. [Google Scholar] [CrossRef]
- Filmer, D.; Pritchett, L.H. Estimating wealth effects without expenditure data—Or tears: An application to educational enrollments in states of India. Demography 2001, 38, 115–132. [Google Scholar]
- Diggle, P.; Liang, K.-Y.; Zeger, S.L. Longitudinal Data Analysis; Oxford University Press: New York, NY, USA, 1994; Volume 5, p. 13. [Google Scholar]
- Das, S.; Alam, M.A.; Mahfuz, M.; Arifeen, S.E.; Ahmed, T. Relative contributions of the correlates of stunting in explaining the mean length-for-age z-score difference between 24-month-old stunted and non-stunted children living in a slum of Dhaka, Bangladesh: Results from a decomposition analysis. BMJ Open 2019, 9, e025439. [Google Scholar] [CrossRef]
- Mandomando, I.; Macete, E.; Sigaúque, B.; Morais, L.; Quintó, L.; Sacarlal, J.; Espasa, M.; Vallès, X.; Bassat, Q.; Aide, P.; et al. Invasive non-typhoidal Salmonella in Mozambican children. Trop. Med. Int. Health 2009, 14, 1467–1474. [Google Scholar] [CrossRef]
- Prando, C.; Samarina, A.; Bustamante, J.; Boisson-Dupuis, S.; Cobat, A.; Picard, C.; AlSum, Z.; Al-Jumaah, S.; Al-Hajjar, S.; Frayha, H.; et al. Inherited IL-12p40 Deficiency: Genetic, Immunologic, and Clinical Features of 49 Patients From 30 Kindreds: Erratum. Medicine 2013, 92, 109–122. [Google Scholar] [CrossRef]
- González-Torres, C.; González-Martínez, H.; Miliar, A.; Nájera, O.; Graniel, J.; Firo, V.; Alvarez, C.; Bonilla, E.; Rodríguez, L. Effect of malnutrition on the expression of cytokines involved in Th1 cell differentiation. Nutrients 2013, 5, 579–593. [Google Scholar] [CrossRef]
- Rout, W.R.; Formal, S.B.; Dammin, G.J.; Giannella, R.A. Pathophysiology of Salmonella Diarrhea in the Rhesus Monkey: Intestinal Transport, Morphological and Bacteriological Studies. Gastroenterology 1974, 67, 59–70. [Google Scholar] [CrossRef]
- Kobayashi, A.; Adachi, Y.; Iwata, Y.; Sakai, Y.; Shigemitu, K.; Todoroki, M.; M, I. Abdominal ultrasonographic findings in typhoid fever: A comparison between typhoid patients and those with non-typhoidal Salmonella and Campylobacter jejuni enterocolitis. Southeast Asian J. Trop. Med. Public Health 2012, 43, 423–430. [Google Scholar] [PubMed]
- Prado, M.d.S.; Cairncross, S.; Strina, A.; Barreto, M.L.; Oliveira-Assis, A.M.; Rego, S. Asymptomatic giardiasis and growth in young children; a longitudinal study in Salvador, Brazil. Parasitology 2005, 131, 51–56. [Google Scholar] [CrossRef] [PubMed]
- Simsek, Z.; Zeyrek, F.Y.; Kurcer, M.A. Effect of Giardia infection on growth and psychomotor development of children aged 0–5 years. J. Trop. Pediatr. 2004, 50, 90–93. [Google Scholar] [CrossRef][Green Version]
- Haque, M.A.; Platts-Mills, J.A.; Mduma, E.; Bodhidatta, L.; Bessong, P.; Shakoor, S.; Kang, G.; Kosek, M.N.; Lima, A.A.M.; Shrestha, S.K.; et al. Determinants of Campylobacter infection and association with growth and enteric inflammation in children under 2 years of age in low-resource settings. Sci. Rep. 2019, 9, 17124. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Bangladesh | India | Pakistan | The Gambia | Mali | Mozambique | Kenya | Overall |
|---|---|---|---|---|---|---|---|---|
| n (%) | n = 284 | n = 72 | n = 365 | n = 168 | n = 8 | n = 28 | n = 584 | n = 1512 |
| Non-typhoidal Salmonella (+) | 71 (18.8) | 18 (4.8) | 92 (24.3) | 42 (11.1) | 2 (0.5) | 7 (1.9) | 146 (38.6) | 378 (25) |
| Age group | ||||||||
| 0–11 months | 41 (57.8) | 11(61.1) | 39 (42.4) | 19 (45.2) | - | 6 (85.7) | 68 (46.6) | 184 (48.7) |
| 12–23 months | 16 (22.5) | 3 (16.7) | 33 (35.9) | 19 (45.2) | 1 (50) | 1 (14.3) | 46 (31.5) | 119 (38.5) |
| 24–59 months | 14 (19.7) | 4 (22.2) | 20 (21.7) | 4 (9.5) | 1 (50) | - | 32 (21.9) | 75 (19.8) |
| Gender (Female) | 29 (40.9) | 3 (16.7) | 36 (39.1) | 24 (57.1) | 1 (50) | 2 (28.6) | 57 (3.0) | 152 (40.2) |
| Breastfed | 60 (84.5) | 17(94.4) | 49 (53.3) | 33 (78.6) | - | 6 (85.7) | 105 (71.9) | 270 (71.4) |
| Baseline Anthropometry | ||||||||
| MUAC, median ¶ | 13.8 ± 1.3 | 14.2 ± 1.3 | 13.1 ± 1.4 | 13.8 ± 1.4 | 14.6 ± 2.4 | 13.2 ± 1.5 | 14.0 ± 1.6 | 13.7 ± 1.5 |
| HAZ ¶ | −1.09 ± 1.1 | −1.09 ± 1.2 | −2.18 ± 1.4 | −1.41 ± 1.8 | −1.47 ± 2.4 | −1.95 ± 1.6 | −1.39 ± 1.4 | −1.52 ± 1.4 |
| WAZ ¶ | −1.32 ± 1.3 | −1.05 ± 1.4 | −2.15 ± 1.3 | −1.36 ± 1.5 | −1.87 ± 2.0 | −1.87 ± 1.5 | −1.18 ± 1.4 | −1.47 ± 1.4 |
| WHZ ¶ | −0.95 ± 1.3 | −0.63 ± 1.2 | −1.33 ± 1.3 | −0.77 ± 1.8 | −1.69 ± 1.3 | −0.93 ± 1.0 | −0.58 ± 1.4 | −0.87 ± 1.4 |
| Clinical features | ||||||||
| Diarrhea | 42 (59.2) | 4 (22.2) | 39 (42.4) | 17 (40.5) | 2 (100) | 5 (71.4) | 81(55.5) | 190 (50.3) |
| Visible blood in stool | 34 (47.9) | 1 (5.6) | 9 (9.9) | 2 (4.8) | - | 1 (14.3) | 8 (5.5) | 55 (14.6) |
| Fever | 33 (46.5) | 7 (38.9) | 34 (36.9) | 9 (21.4) | 1 (50) | 3 (42.7) | 103(70.6) | 190 (50.3) |
| Vomiting ≥3 times/day | 6 (8.5) | 3 (16.7) | 17 (18.5) | 10 (23.8) | 1 (50) | - | 47 (32.2) | 84 (22.2) |
| Socio-demographic characteristics | ||||||||
| Primary caretaker mother | 71 (100) | 18 (100) | 88 (95.7) | 40 (95.2) | 2 (100) | 7 (100) | 141 (96.6) | 367 (97.1) |
| Mother’s education (literate) | 66 (92.9) | 14 (77.8) | 30 (32.6) | 38 (90.5) | 1 (50) | 7 (100) | 144 (98.6) | 266 (70.4) |
| People regularly sleep in the house ¶ | 5.3 ± 2.1 | 4.9 ± 2.1 | 8.7 ± 4.8 | 24.7 ± 18.3 | 16.5 ± 0.7 | 5.8 ± 2.9 | 4.7 ± 1.7 | 8.1 ±9.1 |
| Under 5 children in the house ¶ | 0.7 ± 0.6 | 0.3 ± 0.6 | 0.9 ± 1.3 | 1.9 ± 2.9 | 1.5 ± 0.7 | 0.9 ± 0.7 | 0.9 ± 0.9 | 0.9 ±1.4 |
| Predominant floor (cement) | 14 (19.7) | 17 (94.4) | 63 (68.5) | 29 (69.1) | 2 (100) | 7 (100) | 31 (21.2) | 163 (43.1) |
| Wealth index | ||||||||
| Poor | 14 (19.7) | 5 (27.8) | 10 (10.9) | 12 (28.6) | - | 1 (14.3) | 20 (13.7) | 62 (16.4) |
| Lower middle | 13 (18.3) | 3 (16.7) | 25 (27.2) | 8 (19.1) | - | 2 (28.6) | 33 (22.6) | 84 (22.2) |
| Middle | 16 (22.5) | 3 (16.7) | 24 (26.1) | 7 (16.7) | - | 2 (28.6) | 37 (25.3) | 89 (23.5) |
| Upper middle | 14 (19.7) | 5 (27.8) | 15 (16.3) | 9 (21.4) | 1 (50) | 1 (14.3) | 18 (12.3) | 63 (16.7) |
| Richest | 14 (19.7) | 2 (11.1) | 18 (19.6) | 6 (14.3) | 1 (50) | 1 (14.3) | 38 (26.0) | 80 (21.2) |
| Animal present at the house | ||||||||
| Sheep | 2 (2.8) | - | - | 31 (73.8) | 1 (50) | - | 46 (31.5) | 80 (21.2) |
| Goat | 11 (15.5) | 7 (38.9) | 8 (8.7) | 30 (71.4) | - | 1 (14.3) | 88 (60.3) | 145 (38.4) |
| Cow | 39 (54.9) | 1 (5.6) | 4 (4.4) | 16 (38.1) | - | - | 107 (23.3) | 167 (44.2) |
| Fowl | 41 (57.8) | 9 (50) | 13 (14.1) | 38 (90.5) | - | 4 (57.1) | 141 (96.6) | 246 (65.1) |
| Cat | 63 (88.7) | 17 (94.4) | 8 (8.7) | 7 (16.7) | - | - | 101 (69.2) | 196 (51.9) |
| Dog | 60 (84.5) | 18 (100) | 3 (3.3) | 13 (30.9) | - | - | 103 (70.6) | 197 (52.1) |
| WASH | ||||||||
| Main source of drinking water (tube well) | 68 (95.8) | - | - | 8 (19.0) | - | - | 5 (3.4) | 81(21.4) |
| Use a water treatment method | 1 (1.5) | 11 (61.1) | 31 (36.1) | 13 (36.1) | - | - | 88 (61.9) | 144 (40.1) |
| Toilet facility available in the house | 65 (91.6) | 18 (100) | 89 (96.7) | 42 (100) | 2 (100) | 7 (100) | 107 (73.3) | 330 (87.3) |
| Handwash with water and soap | 52 (73.2) | 7 (38.9) | 60 (65.2) | 32 (76.2) | 2 (100) | 6 (85.7) | 139 (95.2) | 298 (78.8) |
| Practice handwashing | ||||||||
| Before nursing a child | 22 (30.9) | 14 (77.8) | 32 (34.8) | 14 (33.3) | 1 (50) | 5 (71.4) | 40 (27.4) | 128 (33.9) |
| After handling animal | 22 (30.9) | - | 8 (8.7) | 8 (19.1) | - | - | 11 (7.5) | 49 (12.9) |
| After cleaning the child | 22 (30.9) | 15 (83.3) | 54 (58.7) | 36 (85.7) | - | 1 (14.3) | 38 (26.0) | 166 (43.9) |
| Toilet facility available | 65 (91.6) | 18 (100) | 89 (96.7) | 42 (100) | 2 (100) | 7 (100) | 107 (73.3) | 330 (87.3) |
| Co pathogens isolated in stool | ||||||||
| Campylobacter | 16 (22.5) | 1 (5.6) | 32 (34.8) | 1 (2.4) | - | - | 15 (10.3) | 65 (17.2) |
| Giardia | 5 (7.0) | 4 (22.2) | 18 (19.6) | 3 (7.1) | 1 (50) | 1 (14.3) | 19 (13.0) | 51 (13.6) |
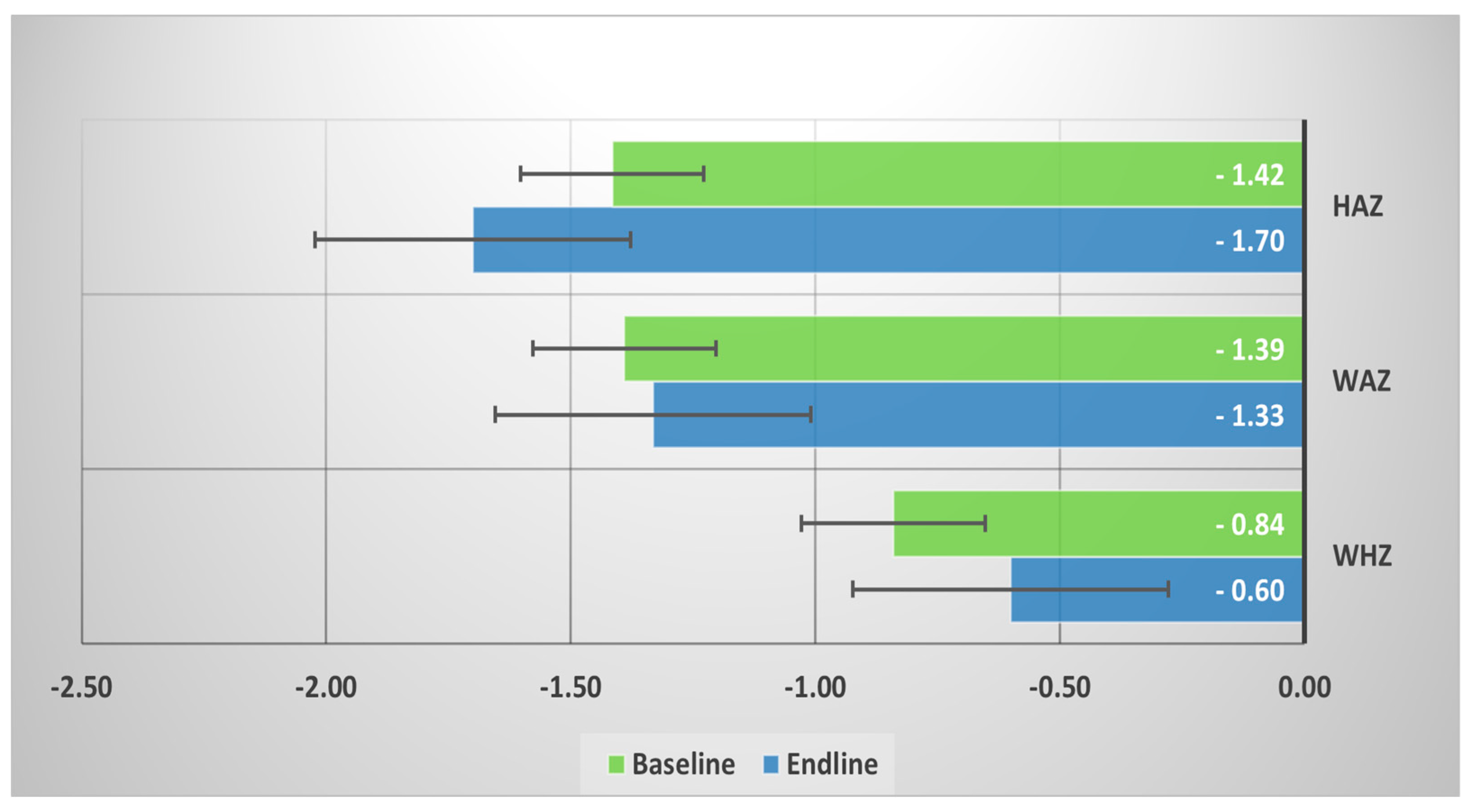
| Z- Score | Age Group | Mean at Baseline (95% CI) | Mean at Endline (95% CI) | * Mean Diff (95% CI) | p Value |
|---|---|---|---|---|---|
| HAZ | Overall | −1.44 (−1.51, −1.38) | −1.61 (−1.68, −1.55) | −0.17 (0.14, 0.19) | <0.001 |
| 0–11 m | −1.11 (−1.21, −1.01) | −1.36 (−1.46, −1.26) | −0.25 (−0.30, −0.20) | <0.001 | |
| 12–23 m | −1.61 (−1.73, −1.49) | −1.79 (−1.91, −1.67) | 0.18 (−0.22, −0.14) | <0.001 | |
| 24–59 m | −1.77 (−1.90, −1.65) | −1.79 (−1.92, −1.67) | −0.02 (−0.06, 0.02) | 0.359 | |
| WAZ | Overall | −1.26 (−1.33, −1.19) | −1.23 (−1.29, −1.16) | 0.03 (−0.07, 0.01) | 0.131 |
| 0–11 m | −1.09 (−1.21, −0.98) | −1.12 ( −1.23, −1.01) | −0.02 (−0.09, 0.04) | 0.458 | |
| 12–23 m | −1.33 (−1.45, −1.20) | −1.32 (−1.44, −1.19) | 0.01 (0.05, 0.07) | 0.788 | |
| 24–59 m | −1.43 (−1.55, −1.31) | −1.28 (−1.40, −1.17) | 0.14 (0.08, 0.21) | <0.001 | |
| WHZ | Overall | −0.63 (−0.69, −0.55) | −0.49 (−0.56, −0.43) | 0.13 (0.08, 0.19) | <0.001 |
| 0–11 m | −0.51 (−0.62, −0.39) | −1.45 (−0.56, −0.33) | 0.60 (−0.03, 0.16) | <0.001 | |
| 12–23 m | −0.77 (−0.89, −0.65) | −0.63 (0.75, 0.51) | 0.14 (0.06, 0.23) | <0.001 | |
| 24–59 m | −0.64 (−0.77, −0.51) | −0.39 (−0.51, −0.28) | 0.24 (0.14, 0.35) | <0.001 |
| Z-Score | Non-Typhoidal Salmonella | |||
|---|---|---|---|---|
| Unadjusted | Adjusted * | |||
| Coef. (95% CI) | p Value | Coef. (95% CI) | p Value | |
| Height-age z-score | −0.90 (−0.24, 0.05) | 0.216 | −0.13 (−0.27, 0.01) | 0.073 |
| Weight-age z-score | −0.18 (−0.33, −0.03) | 0.021 | −0.19 (−0.33, −0.04) | 0.010 |
| Weight-height z-score | −0.19 (−0.34, −0.05) | 0.010 | −0.19 (−0.34, −0.04) | 0.007 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, R.; Haque, M.A.; Chisti, M.J.; Faruque, A.S.G.; Ahmed, T. Association between Non-Typhoidal Salmonella Infection and Growth in Children under 5 Years of Age: Analyzing Data from the Global Enteric Multicenter Study. Nutrients 2021, 13, 392. https://doi.org/10.3390/nu13020392
Das R, Haque MA, Chisti MJ, Faruque ASG, Ahmed T. Association between Non-Typhoidal Salmonella Infection and Growth in Children under 5 Years of Age: Analyzing Data from the Global Enteric Multicenter Study. Nutrients. 2021; 13(2):392. https://doi.org/10.3390/nu13020392
Chicago/Turabian StyleDas, Rina, Md. Ahshanul Haque, Mohammod Jobayer Chisti, Abu Sayed Golam Faruque, and Tahmeed Ahmed. 2021. "Association between Non-Typhoidal Salmonella Infection and Growth in Children under 5 Years of Age: Analyzing Data from the Global Enteric Multicenter Study" Nutrients 13, no. 2: 392. https://doi.org/10.3390/nu13020392
APA StyleDas, R., Haque, M. A., Chisti, M. J., Faruque, A. S. G., & Ahmed, T. (2021). Association between Non-Typhoidal Salmonella Infection and Growth in Children under 5 Years of Age: Analyzing Data from the Global Enteric Multicenter Study. Nutrients, 13(2), 392. https://doi.org/10.3390/nu13020392