Potential Effects of a Modified Mediterranean Diet on Body Composition in Lipoedema
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Subjects
2.2. Anthropometrics and Body Composition
2.3. Indirect Calorimetry
2.4. Dietary Assessments
2.5. Dietary Intervention
2.6. Quality of Life
2.7. Fibromyalgia Assessment Status
2.8. Statistical Analysis
3. Results
3.1. Dietary Components
3.2. Anthropometry and Body Composition
3.3. Quality of Life and Fibromyalgia
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| BCM | Body Cell Mass |
| BCMI | Body Cell Mass Index |
| BIA | Bioelectrical impedance analysis |
| BMI | Body Mass Index |
| CTRL | Control Group |
| DHA | Docosahexaenoic Acid |
| DXA | Dual-Energy X-Ray Absorptiometry |
| ECW | Extracellular Water |
| EPA | Eicosapentaenoic Acid |
| Fe | Iron |
| FFM | Free Fat Mass |
| FM | Fat Mass |
| IMAT | Intermuscular Adipose Tissue |
| K | Potassium |
| LIPPY | Lipoedema Group |
| LM | Lean Mass |
| MAI | Mediterranean Adequacy Index |
| mMeD | Modified Mediterranean Diet Therapy |
| Mg | Magnesium |
| MREE | Measured Resting Energy Expenditure |
| MUFA | Monounsaturated Fatty Acid |
| Na | Sodium |
| ORAC | Oxygen Radical Absorbance Capacity |
| PA | Phase Angle |
| PUFA | Polyunsaturated Fatty Acid |
| Rz | Resistance |
| Xc | Reactance |
| SFA | Saturated Fatty Acid |
| TBW | Total Body Water |
| VCO2 | Volumes of Carbon Dioxide |
| Vit. | Vitamin |
| VO2 | Volumes of Oxygen |
| WHR | Waist-to-Hip Ratio |
Appendix A. Medium-Length FFQs
MEDIUM-LENGTH FOOD FREQUENCY QUESTIONNAIRE
| A. DRINKS | ||||||
| 1. Do you drink COFFEE? Yes ☐ No, never. ☐ | ||||||
| How many times per day? ……… | ||||||
| 2. Do you drink ALCOHOL DRINKS? Yes ☐ No, never. ☐ | ||||||
| How much? | Wine: | 0.15 L ☐ | 0.20 L ☐ | 0.25 L ☐ | ||
| Beer: | 0.23 L ☐ | 0.33 L ☐ | 0.45 L ☐ | 0.66 L ☐ | ||
| Strong alcohol: | 0.03 L ☐ | 0.13 L ☐ | 0.20 L ☐ | 0.30 L ☐ | ||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 3. Do you drink SOFT DRINKS (coke, soda…) ? Yes ☐ No, never. ☐ | ||||||
| How many times per day? ……… | ||||||
| B. MILK and DAIRY PRODUCTS | ||||||
| 4. Do you drink MILK? Yes ☐ No, never. ☐ | ||||||
| Which kind? | Whole ☐ | Semi-skimmed ☐ | Skimmed ☐ | |||
| How much? | Small (120 mL) ☐ | Medium (200 mL) ☐ | Large (300 mL) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 5. Do you eat Yogurt? Yes ☐ No, never. ☐ | ||||||
| How much? | Small (125 g) ☐ | Medium (150 g) ☐ | Large (180 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 6. Do you eat CHEESE? Yes ☐ No, never. ☐ | ||||||
| How many times per week? ……… Less than once a week. | ||||||
| 6.1. HARD CHEESE(e.g., parmesan, sheep’s milk, Swiss) ? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (30 g) ☐ | Medium serving (50 g) ☐ | Large serving (70 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 6.2. SOFT CHEESE? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (50 g) ☐ | Medium serving (70 g) ☐ | Large serving (100 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 6.3. MOZZARELLA? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (60 g) ☐ | Medium serving (125 g) ☐ | Large serving (200 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 6.4. COTTAGE CHEESE? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (100 g) ☐ | Medium serving (150 g) ☐ | Large serving (200 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| C. MEAT, FISH and EGGS | ||||||
| 7. Do you eat RED MEAT? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (80 g) ☐ | Medium serving (120 g) ☐ | Large serving (200 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 8. Do you eat POULTRY? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (80 g) ☐ | Medium serving (120 g) ☐ | Large serving (160 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 9. Do you eat PORK? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (50 g) ☐ | Medium serving (100 g) ☐ | Large serving (150 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 10. Do you eat FISH? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (100 g) ☐ | Medium serving (150 g) ☐ | Large serving (200 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 11. Do you eat CURED MEATS AND SALAMI? Yes ☐ No, never. ☐ | ||||||
| How much? | ||||||
| CURED MEAT: | Small serving (20 g) ☐ | Medium serving (40 g) ☐ | Large serving (80 g) ☐ | |||
| SALAMI: | Small serving (15 g) ☐ | Medium serving (30 g) ☐ | Large serving (50 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 12. Do you eat EGGS? Yes ☐ No, never. ☐ | ||||||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| D. CEREALS | ||||||
| 13. Do you eat PASTA or RICE? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (40–50 g) ☐ | Medium serving (60–80 g) ☐ | Large serving (100–120 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 14. Do you eat BREAD? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (30 g) ☐ | Medium serving (50 g) ☐ | Large serving (100 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 15. Do you eat BAKERY PRODUCTS? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (15 g) ☐ | Medium serving (30 g) ☐ | Large serving (45 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 16. Do you eat POTATOES? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (100 g) ☐ | Medium serving (150 g) ☐ | Large serving (200 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 17. Do you eat PIZZA? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (100 g) ☐ | Medium serving (150 g) ☐ | Large serving (300 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| E. VEGETABLES, LEGUMES and FRUIT | ||||||
| 18. Do you eat VEGETABLES or GREENS? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (100 g) ☐ | Medium serving (150 g) ☐ | Large serving (300 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 19. Do you eat LEGUMES (beans, peas) ? Yes ☐ No, never. ☐ | ||||||
| How much? | ||||||
| DRIED: | Small serving (30 g) ☐ | Medium serving (50 g) ☐ | Large serving (70 g) ☐ | |||
| FRESH: | Small serving (75 g) ☐ | Medium serving (125g) ☐ | Large serving (175 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 20. Do you eat FRESH FRUIT? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (100 g) ☐ | Medium serving (150 g) ☐ | Large serving (200 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| F. FATTY DRESSINGS | ||||||
| 21. Do you eat OLIVE OIL? Yes ☐ No, never. ☐ | ||||||
| How much? | 1 teaspoon (5 g) ☐ | 2 teaspoons (10 g) ☐ | 2 teaspoons (15 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 22. Do you eat SEED OIL? Yes ☐ No, never. ☐ | ||||||
| How much? | 1 teaspoon (5 g) ☐ | 2 teaspoons (10 g) ☐ | 2 teaspoons (15 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 23. Do you eat BUTTER? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (5 g) ☐ | Medium serving (10 g) ☐ | Large serving (15 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 24. Do you eat MARGARINE? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (5 g) ☐ | Medium serving (10 g) ☐ | Large serving (15 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| G. OTHER | ||||||
| 25. Do you eat SWEETS? Yes ☐ No, never. ☐ | ||||||
| How much? | Small serving (30 g) ☐ | Medium serving (50 g) ☐ | Large serving (70 g) ☐ | |||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 26. Do you eat FRIED FOODS? Yes ☐ No, never. ☐ | ||||||
| How many times per week? ……… Less than once a week. ☐ | ||||||
| 27. Do you eat FAST FOOD? Yes ☐ No, never. ☐ | ||||||
| Which kind? | ||||||
| HAMBERGER: | Medium serving (120 g) ☐ | Large serving (210 g) ☐ | ||||
| Hot Dog: | Medium serving (120 g) ☐ | Large serving (210 g) ☐ | ||||
| LOCAL STREET FOOD: | ||||||
| Panelle sandwich | Medium serving (200 g) ☐ | |||||
| Spleen sandwich | Medium serving (250 g) ☐ | |||||
| How many times per week? ……… Less than once a week. ☐ | ||||||
Appendix B
| Categories | Items | Scotti-Bassani Atlas Reference (Table Number) | MetaDieta Software Selection |
|---|---|---|---|
| Drinks | Coffee | Cup (n.99) | Espresso |
| Wine | Glasses 2 (n.94) | Red wine | |
| Beer | Bottles and can 1 (n.95) | Lager beer | |
| Strong drinks | Glasses 1 (n.93) | Vodka | |
| Soft drinks | Bottles and can 2 (n.95) | Coke | |
| Milk and dairy products | Milk | Milk (n.33) | Semi-skimmed milk |
| Yogurt | Yogurt (n.35) | Whole milk yogurt | |
| Hard cheese | Cheese 2 (n.31) | Standard hard cheese serving | |
| Soft cheese | Cheese 1 (n.30) | Standard soft cheese serving | |
| Mozzarella | Mozzarella (n.34) | Mozzarella | |
| Cottage cheese | Cottage cheese (n.32) | Cottage cheese | |
| Meat, fish and eggs | Red meat | Beef steak (n.40) | Semi-fat beef |
| Poultry | Chicken breast (n.42) | Chicken breast | |
| Pork | Pork steak (n.40) | Pork Sirloin Chop | |
| Fish | Cod fillet (n.54) | Cod fillet | |
| Cured meat | Ham (n.46) | Ham | |
| Salami | Salami (n.48) | Salami | |
| Eggs | Omelette (n.36) | Chicken eggs | |
| Cereals | Pasta/rice | Pasta and tomato sauce (n.22) | Semolina pasta |
| Bread | Bread (n.4) | Bread Bread (n.4) Bread | |
| Bakery products | Crackers (n.1)Bread sticks (n.3) | Crackers, bread sticks | |
| Potatoes | Boiled potatoes (n.28) | Boiled potatoes | |
| Pizza | Pizza (n. 8) | Pizza tomato and mozzarella | |
| Vegetables, legumes and fruits | Vegetables/greenS | Vegetable and greens (n. 61–71) | Lettuce, spinach, eggplant, zucchini, carrots |
| Dried legumes | Beans (n.26) | Beans | |
| Fresh legumes | Peas (n.29) | Peas | |
| Fresh Fruits | Fresh fruit (n.72–80) | Seasonal fresh fruit | |
| Fatty dressings | Olive oil | Spoons, spoons and ladles (n.98) | Extra-virgin olive oil |
| Seed oil | Spoons, spoons and ladles (n.98) | Sunflower seed oil | |
| Butter | Fat dressings (n.92) | Butter | |
| Margarine | Fat dressings (n.92) | Margarine | |
| Other | Sweets | Sweets (n.87, 88) | Standard sweets serving |
| Fried foods | French fries (n.27) | French fries | |
| Fast food | |||
| Hamburger | Hamburger sandwich (n.82) | Hamburger sandwich | |
| Hot dog | Hot dog | ||
| Local street food | Panelle sandwich and spleen sandwich |
References
- Herbst, K.L. Subcutaneous Adipose Tissue Diseases: Dercum Disease, Lipoedema, Familial Multiple Lipomatosis, and Madelung Disease. In Endotext; Feingold, K.R., Anawalt, B., Boyce, A., Chrousos, G., de Herder, W.W., Dungan, K., Grossman, A., Hershman, J.M., Hofland, H.J., Kaltsas, G., et al., Eds.; MDText.com, Inc.: South Dartmouth, MA, USA, 2000. [Google Scholar]
- Child, A.H.; Gordon, K.D.; Sharpe, P.; Brice, G.; Ostergaard, P.; Jeffery, S.; Mortimer, P.S. Lipoedema: An Inherited Condition. Am. J. Med. Genet. Part A 2010, 152, 970–976. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-G.; Hsu, S.-D.; Chen, T.-M.; Wang, H.-J. Painful Fat Syndrome in a Male Patient. Br. J. Plast. Surg. 2004, 57, 282–286. [Google Scholar] [CrossRef]
- Földi, M.; Földi, E.; Kubik, S. Földi’s Textbook of Lymphology; Elsevier: Amsterdam, The Netherlands, 2003. [Google Scholar]
- Herbst, K.L. Lipoedema Fat and Signs and Symptoms of Illness, Increase with Advancing Stages. 2014. Available online: https://www.archivesofmedicine.com/medicine/lipoedema-fat-andsigns-and-symptoms-of-illness-increase-with-advancing-stagephp (accessed on 25 March 2020).
- Al-Ghadban, S.; Cromer, W.; Allen, M.; Ussery, C.; Badowski, M.; Harris, D.; Herbst, K. Dilated Blood and Lymphatic Microvessels, Angiogenesis, Increased Macrophages, and Adipocyte Hypertrophy in Lipoedema Thigh Skin and Fat Tissue. J. Obes. 2019, 2019, 1–10. [Google Scholar] [CrossRef]
- Felmerer, G.; Stylianaki, A.; Hollmén, M.; Ströbel, P.; Stepniewski, A.; Wang, A.; Frueh, F.S.; Kim, B.-S.; Giovanoli, P.; Lindenblatt, N.; et al. Increased Levels of VEGF-C and Macrophage Infiltration in Lipoedema Patients without Changes in Lymphatic Vascular Morphology. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Buck, D.W., II.; Herbst, K.L. Lipoedema: A Relatively Common Disease with Extremely Common Misconceptions. Plast. Reconstr. Surg. Glob. Open 2016, 4, e1043. [Google Scholar] [CrossRef]
- Michelini, S.; Chiurazzi, P.; Marino, V.; Dell’Orco, D.; Manara, E.; Baglivo, M.; Fiorentino, A.; Maltese, P.E.; Pinelli, M.; Herbst, K.L. Aldo-Keto Reductase 1C1 (AKR1C1) as the First Mutated Gene in a Family with Nonsyndromic Primary Lipoedema. Int. J. Mol. Sci. 2020, 21, 6264. [Google Scholar] [CrossRef]
- De Lorenzo, A.; Gratteri, S.; Gualtieri, P.; Cammarano, A.; Bertucci, P.; Di Renzo, L. Why Primary Obesity Is a Disease? J. Transl. Med. 2019, 17, 169. [Google Scholar] [CrossRef]
- Di Renzo, L.; Gualtieri, P.; Alwardat, N.; De Santis, G.; Zomparelli, S.; Romano, L.; Marchetti, M.; Michelin, S.; Capacci, A.; Piccioni, A.; et al. The Role of IL-6 Gene Polymorphisms in the Risk of Lipoedema. Eur. Rev. Med. Pharmacol. Sci. 2020, 24, 3236–3244. [Google Scholar] [CrossRef]
- Dietzel, R.; Reisshauer, A.; Jahr, S.; Calafiore, D.; Armbrecht, G. Body Composition in Lipoedema of the Legs Using Dual-Energy X-Ray Absorptiometry: A Case–Control Study. Br. J. Dermatol. 2015, 173, 594–596. [Google Scholar] [CrossRef]
- Herbst, K.L. Rare Adipose Disorders (RADs) Masquerading as Obesity. Acta Pharmacol. Sin. 2012, 33, 155–172. [Google Scholar] [CrossRef]
- Hardy, D.; Williams, A. Best Practice Guidelines for the Management of Lipoedema. Br. J. Community Nurs. 2017, 22, S44–S48. [Google Scholar] [CrossRef]
- Ehrlich, C.; Iker, E.; Herbst, K.L.; Kahn, L.-A.; Sears, D.D.; Kenyon, M.; McMahon, E. Lymphedema and Lipoedema Nutrition Guide. Foods, Vitamins, Minerals, and Supplements; Lymph Notes: San Francisco, CA, USA, 2015. [Google Scholar]
- Dayan, E.; Kim, J.N.; Smith, M.L.; Seo, C.A.; Damstra, R.J.; Schmeller, W. Lipoedema—The Disease They Call FAT: An Overview for Clinicians; The Lipoedema Project at The Friedman Center for Lymphedema Research and Treatment in collaboration with Lymphatic Education & Research Network: Boston, MA, USA, 2017. [Google Scholar]
- Dudek, J.E.; Białaszek, W.; Ostaszewski, P. Quality of Life in Women with Lipoedema: A Contextual Behavioral Approach. Qual. Life Res. 2016, 25, 401–408. [Google Scholar] [CrossRef]
- Reich-Schupke, S.; Schmeller, W.; Brauer, W.J.; Cornely, M.E.; Faerber, G.; Ludwig, M.; Lulay, G.; Miller, A.; Rapprich, S.; Richter, D.F.; et al. S1 Guidelines: Lipoedema. J. Dtsch. Dermatol. Ges. 2017, 15, 758–767. [Google Scholar] [CrossRef]
- Keith, L.; Seo, C.A.; Rowsemitt, C.; Pfeffer, M.; Wahi, M.; Staggs, M.; Dudek, J.; Gower, B.; Carmody, M. Ketogenic Diet as a Potential Intervention for Lipoedema. Med. Hypotheses 2020, 110435. [Google Scholar] [CrossRef]
- De Lorenzo, A.; Bernardini, S.; Gualtieri, P.; Cabibbo, A.; Perrone, M.A.; Giambini, I.; Di Renzo, L. Mediterranean Meal versus Western Meal Effects on Postprandial Ox-LDL, Oxidative and Inflammatory Gene Expression in Healthy Subjects: A Randomized Controlled Trial for Nutrigenomic Approach in Cardiometabolic Risk. Acta Diabetol. 2017, 54, 141–149. [Google Scholar] [CrossRef]
- Di Renzo, L.; Rizzo, M.; Iacopino, L.; Sarlo, F.; Domino, E.; Jacoangeli, F.; Colica, C.; Sergi, D.; De Lorenzo, A. Body Composition Phenotype: Italian Mediterranean Diet and C677T MTHFR Gene Polymorphism Interaction. Eur. Rev. Med. Pharmacol. Sci. 2013, 17, 2555–2565. [Google Scholar]
- Di Renzo, L.; Di Pierro, D.; Bigioni, M.; Sodi, V.; Galvano, F.; Cianci, R.; La Fauci, L.; De Lorenzo, A. Is Antioxidant Plasma Status in Humans a Consequence of the Antioxidant Food Content Influence? Eur. Rev. Med. Pharmacol. Sci. 2007, 11, 185–192. [Google Scholar]
- Di Renzo, L.; Marsella, L.T.; Carraro, A.; Valente, R.; Gualtieri, P.; Gratteri, S.; Tomasi, D.; Gaiotti, F.; De Lorenzo, A. Changes in LDL Oxidative Status and Oxidative and Inflammatory Gene Expression after Red Wine Intake in Healthy People: A Randomized Trial. Mediat. Inflamm. 2015, 2015, 1–13. [Google Scholar] [CrossRef]
- World Health Organization. Obesity: Preventing and Managing the Global Epidemic. Report of a WHO Consultation; WHO Technical Report Series; World Health Organization: Geneva, Switzerland, 2000; p. 252. [Google Scholar]
- De Lorenzo, A.; Romano, L.; Di Renzo, L.; Di Lorenzo, N.; Cenname, G.; Gualtieri, P. Obesity: A Preventable, Treatable, but Relapsing Disease. Nutrition 2020, 71, 110615. [Google Scholar] [CrossRef]
- Colica, C.; Merra, G.; Gasbarrini, A.; De Lorenzo, A.; Cioccoloni, G.; Gualtieri, P.; Perrone, M.A.; Bernardini, S.; Bernardo, V.; Di Renzo, L. Efficacy and Safety of Very-Low-Calorie Ketogenic Diet: A Double Blind Randomized Crossover Study. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2274–2289. [Google Scholar]
- De Lorenzo, A.; Di Renzo, L.; Morini, P.; de Miranda, R.C.; Romano, L.; Colica, C. New Equations to Estimate Resting Energy Expenditure in Obese Adults from Body Composition. Acta Diabetol. 2018, 55, 59–66. [Google Scholar] [CrossRef] [PubMed]
- De Weir, J.V. New Methods for Calculating Metabolic Rate with Special Reference to Protein Metabolism. J. Physiol. 1949, 109, 1. [Google Scholar] [CrossRef] [PubMed]
- United Nations University; World Health Organization. Human Energy Requirements: Report of a Joint FAO/WHO/UNU Expert Consultation: Rome, 17–24 October 2001; Food & Agriculture Organization of the United Nations: Rome, Italy, 2004; Volume 1. [Google Scholar]
- Buscemi, S.; Rosafio, G.; Vasto, S.; Massenti, F.M.; Grosso, G.; Galvano, F.; Rini, N.; Barile, A.M.; Maniaci, V.; Cosentino, L. Validation of a Food Frequency Questionnaire for Use in Italian Adults Living in Sicily. Int. J. Food Sci. Nutr. 2015, 66, 426–438. [Google Scholar] [CrossRef]
- Alberti-Fidanza, A.; Fidanza, F. Mediterranean Adequacy Index of Italian Diets. Public Health Nutr. 2004, 7, 937–941. [Google Scholar] [CrossRef]
- De Lorenzo, A.; Noce, A.; Bigioni, M.; Calabrese, V.; Della Rocca, D.G.; Daniele, N.D.; Tozzo, C.; Renzo, L.D. The Effects of Italian Mediterranean Organic Diet (IMOD) on Health Status. Curr. Pharm. Des. 2010, 16, 814–824. [Google Scholar] [CrossRef]
- The Measurement and Valuation of Health Status Using EQ-5D: A European Perspective: Evidence from the EuroQol BIOMED Research Programme; Brooks, R., Rabin, R., de Charro, F., Eds.; Springer: Dordrecht, The Netherlands, 2003; ISBN 9789401702331. [Google Scholar]
- Eurool ResearchRabinEuroQol Research Foundation. EQ-5D-3L User Guide Version 2018. Available online: https://euroqol.org/publications/user-guides (accessed on 25 March 2020).
- Salaffi, F.; Sarzi-Puttini, P.; Girolimetti, R.; Gasparini, S.; Atzeni, F.; Grassi, W. Development and Validation of the Self-Administered Fibromyalgia Assessment Status: A Disease-Specific Composite Measure for Evaluating Treatment Effect. Arthritis Res. Ther. 2009, 11, R125. [Google Scholar] [CrossRef]
- la Torre, Y.S.-D.; Wadeea, R.; Rosas, V.; Herbst, K.L. Lipoedema: Friend and Foe. Horm. Mol. Biol. Clin. Investig. 2018, 33. [Google Scholar] [CrossRef]
- Alwardat, N.; Di Renzo, L.; Alwardat, M.; Romano, L.; De Santis, G.L.; Gualtieri, P.; Carrano, E.; Nocerino, P.; De Lorenzo, A. The Effect of Lipoedema on Health-Related Quality of Life and Psychological Status: A Narrative Review of the Literature. Eat. Weight Disord. Stud. Anorex. Bulim. Obes. 2020, 25, 851–856. [Google Scholar] [CrossRef]
- Marshall, M.N. Sampling for Qualitative Research. Fam. Pract. 1996, 13, 522–525. [Google Scholar] [CrossRef]
- Runkel, N.; Colombo-Benkmann, M.; Hüttl, T.P.; Tigges, H.; Mann, O.; Flade-Kuthe, R.; Shang, E.; Susewind, M.; Wolff, S.; Wunder, R.; et al. Evidence-Based German Guidelines for Surgery for Obesity. Int. J. Colorectal Dis. 2011, 26, 397–404. [Google Scholar] [CrossRef]
- Seifried, H.E.; Anderson, D.E.; Fisher, E.I.; Milner, J.A. A Review of the Interaction among Dietary Antioxidants and Reactive Oxygen Species. J. Nutr. Biochem. 2007, 18, 567–579. [Google Scholar] [CrossRef]
- Tufts, H.R.; Harris, C.S.; Bukania, Z.N.; Johns, T. Antioxidant and Anti-Inflammatory Activities of Kenyan Leafy Green Vegetables, Wild Fruits, and Medicinal Plants with Potential Relevance for Kwashiorkor. Evid. Based Complementary Altern. Med. 2015, 2015, 807158. [Google Scholar] [CrossRef]
- Sergeev, I.N. Vitamin D Status and Vitamin D-Dependent Apoptosis in Obesity. Nutrients 2020, 12, 1392. [Google Scholar] [CrossRef]
- Butovskaya, M.; Sorokowska, A.; Karwowski, M.; Sabiniewicz, A.; Fedenok, J.; Dronova, D.; Negasheva, M.; Selivanova, E.; Sorokowski, P. Waist-to-Hip Ratio, Body-Mass Index, Age and Number of Children in Seven Traditional Societies. Sci. Rep. 2017, 7. [Google Scholar] [CrossRef]
- Kyle, U.G.; Bosaeus, I.; Lorenzo, A.D.D.; Deurenberg, P.; Elia, M.; Gómez, J.M.; Heitmann, B.L.; Kent-Smith, L.; Melchior, J.-C.; Pirlich, M.; et al. Bioelectrical Impedance Analysis—Part II: Utilization in Clinical Practice. Clin. Nutr. 2004, 23, 1430–1453. [Google Scholar] [CrossRef]
- Boghossian, N.S.; Yeung, E.H.; Mumford, S.L.; Zhang, C.; Gaskins, A.J.; Wactawski-Wende, J.; Schisterman, E.F. Adherence to the Mediterranean Diet and Body Fat Distribution in Reproductive Aged Women. Eur. J. Clin. Nutr. 2013, 67, 289–294. [Google Scholar] [CrossRef]
- Wold, L.E.; Hines, E.A.; Allen, E.V. Lipoedema of the Legs; a Syndrome Characterized by Fat Legs and Edema. Ann. Intern. Med. 1951, 34, 1243–1250. [Google Scholar] [CrossRef]
- Ebbeling, C.B.; Swain, J.F.; Feldman, H.A.; Wong, W.W.; Hachey, D.L.; Garcia-Lago, E.; Ludwig, D.S. Effects of Dietary Composition on Energy Expenditure during Weight-Loss Maintenance. JAMA 2012, 307, 2627–2634. [Google Scholar] [CrossRef]
- Di Renzo, L.; Cinelli, G.; Dri, M.; Gualtieri, P.; Attinà, A.; Leggeri, C.; Cenname, G.; Esposito, E.; Pujia, A.; Chiricolo, G.; et al. Mediterranean Personalized Diet Combined with Physical Activity Therapy for the Prevention of Cardiovascular Diseases in Italian Women. Nutrients 2020, 12, 3456. [Google Scholar] [CrossRef]
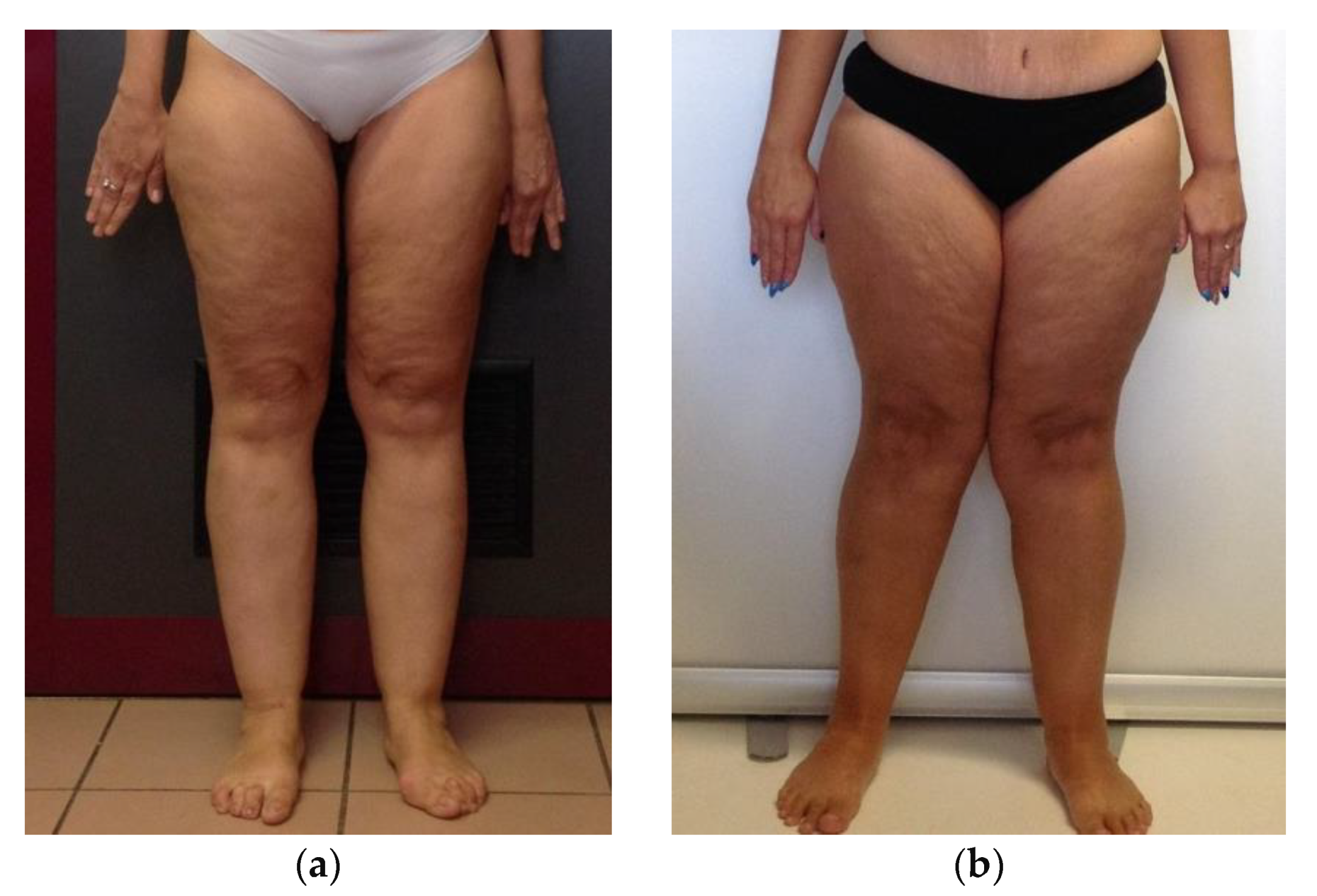
| LIPPY | CTRL | |||
|---|---|---|---|---|
| Baseline | mMeD | Baseline | mMeD | |
| Mean ± SD | Mean ± SD | Mean ± SD | Mean ± SD | |
| Energy (Kcal) | 1536.0 ± 320.8 | 1370.4 ± 222.8 * | 1570.7 ± 301.7 | 1494.1 ± 171.5 |
| Proteins (% Kcal) | 17.6 ± 3.8 | 27.2 ± 4.4 ** | 20 ± 3.7 | 22.5 ± 2.04 |
| Vegetable proteins (% Kcal) | 28.2 ± 9.4 | 30.2 ± 4.6 | 28.5 ± 8 | 32 ± 8 |
| Animal proteins (% Kcal) | 71.8 ± 9.4 | 62 ± 5.3 * | 67.4 ± 8.9 | 52.6 ± 5.2 ** |
| Carbohydrates (% Kcal) | 37.5 ± 11.8 | 40.2 ± 1.6 | 43.9 ± 7.1 | 43.8 ± 3 |
| Sugars (% Kcal) | 13.6 ± 4 | 14.6 ± 1.17 | 15.9 ± 4.6 | 16.3 ± 2 |
| Total fiber (g) | 13.8 ± 6 | 26.4 ± 4 ** | 19.7 ± 4.3 | 28.9 ± 3.6 ** |
| Lipids (% Kcal) | 44.4 ± 9.6 | 31.9 ± 3.6 * | 36.1 ± 7 | 33.5 ± 3.1 |
| SFA (g) | 18.2 ± 6.9 | 9.4 ± 2.2 * | 17.5 ± 7.9 | 12.6 ± 3 * |
| PUFA (g) | 8.6 ± 3.4 | 9.05 ± 4 | 6.9 ± 1.7 | 8.2 ± 1.5 * |
| PUFA/SFA | 0.47 ± 0.2 | 0.93 ± 0.2 ** | 1.7 ± 3 | 0.64 ± 0.2 |
| MUFA (g) | 36.1 ± 13.4 | 23.4 ± 5.9 * | 32 ± 9.7 | 28.1 ± 4.9 |
| EPA (g) | 0.13 ± 0.21 | 0.53 ± 0.27 * | 0.10 ± 0.20 | 0.40 ± 0.11 * |
| DHA (g) | 0.20 ± 0.40 | 1.1 ± 0.46 ** | 0.22 ± 0.54 | 0.62 ± 0.24 * |
| ω6/ω3 | 8.04 ± 3.2 | 2.6 ± 0.54 ** | 6.1 ± 1.8 | 3.5 ± 1 ** |
| Oleic acid (g) | 29.7 ± 12 | 23.3 ± 5.5 | 30.7 ± 9.3 | 27 ± 4.7 |
| Na (mg) | 1210 ± 996.7 | 957.4 ± 236.1 | 1435.5 ± 677.4 | 1278.8 ± 203.7 |
| K (mg) | 2270.7 ± 655.5 | 3549.1 ± 342.1 ** | 2871.8 ± 631.1 | 3637.1 ± 390.2 * |
| Fe (mg) | 9.7 ± 3.2 | 15.1 ± 2.17 ** | 10.5 ± 2 | 13.6 ± 1.81 * |
| Mg (mg) | 162.7 ± 75.5 | 295.8 ± 37.2 ** | 227.5 ± 94 | 304.1 ± 48.3 * |
| Vit. A (µcg) | 764.3 ± 329.7 | 1091.3 ± 71.2 * | 930.6 ± 306 | 1281.2 ± 455.9 * |
| Vit. C (mg) | 112 ± 48.5 | 190.2 ± 14.7 ** | 145.4 ± 62.20 | 224.5 ± 38.2 * |
| Vit. D (µcg) | 4.3 ± 4.4 | 7.63 ± 2.5 * | 2.3 ± 3.8 | 5.3 ± 2 * |
| Vit. E (mg) | 11.7 ± 2.2 | 12.6 ± 2 | 5.3 ± 11.8 | 14.7 ± 1.6 |
| ORAC (µmol) | 2423.0 ± 1814.0 | 13538.0 ± 1517.0 ** | 11759.0 ± 6910.0 | 14105.0 ± 1584.0 |
| MAI | 1.4 ± 0.7 | 14.7 ± 0.7 ** | 1.45 ± 1.07 | 14.06 ± 1.9 ** |
| Parameters | CTRL | LIPPY | Δ% Baseline–4 Weeks | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | 4 Weeks | Baseline | 4 Weeks | CTRL | LIPPY | ||||
| Mean ± SD | Mean ± SD | p | Mean ± SD | Mean ± SD | p | Mean ± SD | Mean ± SD | p | |
| Weight (kg) | 73.45 ± 14.50 | 70.21 ± 14.09 | 0.001 **† | 91.06 ± 28.63 | 88.1 ± 27.7 | 0.025 *† | −4.37 ± 3.94 | −3.04 ± 4.75 | 0.418 |
| BMI (kg/m2) | 27.52 ± 5.22 | 26.38 ± 4.99 | 0.002 **† | 35.50 ± 12.17 | 34.36 ± 11.84 | 0.021 *† | −4.05 ± 4.03 | −3.04 ± 4.75 | 0.540 |
| Neck C. (cm) | 35.50 ± 3.16 | 34.77 ± 3.08 | 0.012 * | 37.21 ± 3.59 | 36.79 ± 2.96 | 0.443 | −2.12 ± 2.45 | −0.89 ± 5.32 | 0.469 |
| Waist C. (cm) | 85.12 ± 13.37 | 80.24 ± 10.67 | 0.003 ** | 87.18 ± 16.08 | 85.44 ± 14.44 | 0.115 | −5.43 ± 4.33 | −1.66 ± 3.84 | 0.022 * |
| Hip C. (cm) | 108.08 ± 9.87 | 105.19 ± 9.06 | 0.002 **† | 122.11 ± 23.31 | 120.68 ± 21.76 | 0.250† | −2.59 ± 2.55 | −0.95 ± 3.89 | 0.198 |
| WHR | 0.78 ± 0.08 | 0.76 ± 0.07 | 0.040 * | 0.72 ± 0.07 | 0.71 ± 0.06 | 0.612 | −3.00 ± 4.36 | −0.60 ± 5.15 | 0.194 |
| VO2 (ml/min) | 218.87 ± 31.61 | 213.57 ± 36.55 | 0.323 | 217.14 ± 46.08 | 213.17 ± 40.93 | 0.515 | −2.02 ± 7.01 | −1.57 ± 14.29 | 0.919 |
| VCO2 (ml/min) | 182.13 ± 29.55 | 181.36 ± 31.38 | 0.752 | 173.57 ± 41.52 | 172.75 ± 32.64 | 0.972 | 0.13 ± 2.51 | 2.36 ± 20.9 | 0.694 |
| MREE (kcal) | 1508.00 ± 247.11 | 1502.79 ± 260.49 | 0.677 | 1479.21 ± 321.51 | 1454.83 ± 281.4 | 0.633 | 0.24 ± 2.68 | −0.88 ± 15.18 | 0.789 |
| Rz | 561.60 ± 75.80 | 569.33 ± 77.56 | 0.320 | 507.64 ± 86.63 | 510 ± 94.72 | 0.779 | 1.53 ± 5.02 | 0.43 ± 6.25 | 0.603 |
| Xc | 59.87 ± 9.62 | 58.27 ± 9.60 | 0.411 | 46.64 ± 9.37 | 49.71 ± 14.62 | 0.344 | −2.21 ± 11.29 | 7.07 ± 24.27 | 0.193 |
| TBW (L) | 34.89 ± 4.66 | 34.37 ± 4.53 | 0.800† | 37.99 ± 5.84 | 37.79 ± 5.97 | 0.396 † | −1.43 ± 2.82 | −0.54 ± 3.7 | 0.469 |
| ECW (L) | 15.80 ± 2.08 | 16.00 ± 2.22 | 0.522 | 18.88 ± 3.37 | 18.35 ± 3.73 | 0.433 | 1.48 ± 8.03 | −2.49 ± 13.00 | 0.328 |
| BCM (kg) | 25.93 ± 4.98 | 24.82 ± 3.65 | 0.110 | 25.12 ± 3.66 | 25.68 ± 4.85 | 0.559 | −3.45 ± 7.77 | 2.39 ± 14.53 | 0.184 |
| PA (°) | 6.13 ± 1.08 | 5.85 ± 0.61 | 0.008 **† | 5.24 ± 0.59 | 5.54 ± 1.24 | 0.683 † | −3.01 ± 11.44 | 6.15 ± 22.28 | 0.171 |
| BCMI (kg/m2) | 9.73 ± 1.78 | 9.31 ± 1.24 | 0.153 † | 9.76 ± 1.71 | 10.08 ± 2.31 | 0.875 † | −3.48 ± 7.62 | 3.29 ± 16.70 | 0.166 |
| Parameters | CTRL | LIPPY | Δ% Baseline−4 Weeks | ||||||
|---|---|---|---|---|---|---|---|---|---|
| Baseline | 4 Weeks | Baseline | 4 Weeks | CTRL | LIPPY | ||||
| Mean ± SD | Mean ± SD | p | Mean ± SD | Mean ± SD | p | Mean ± SD | Mean ± SD | p | |
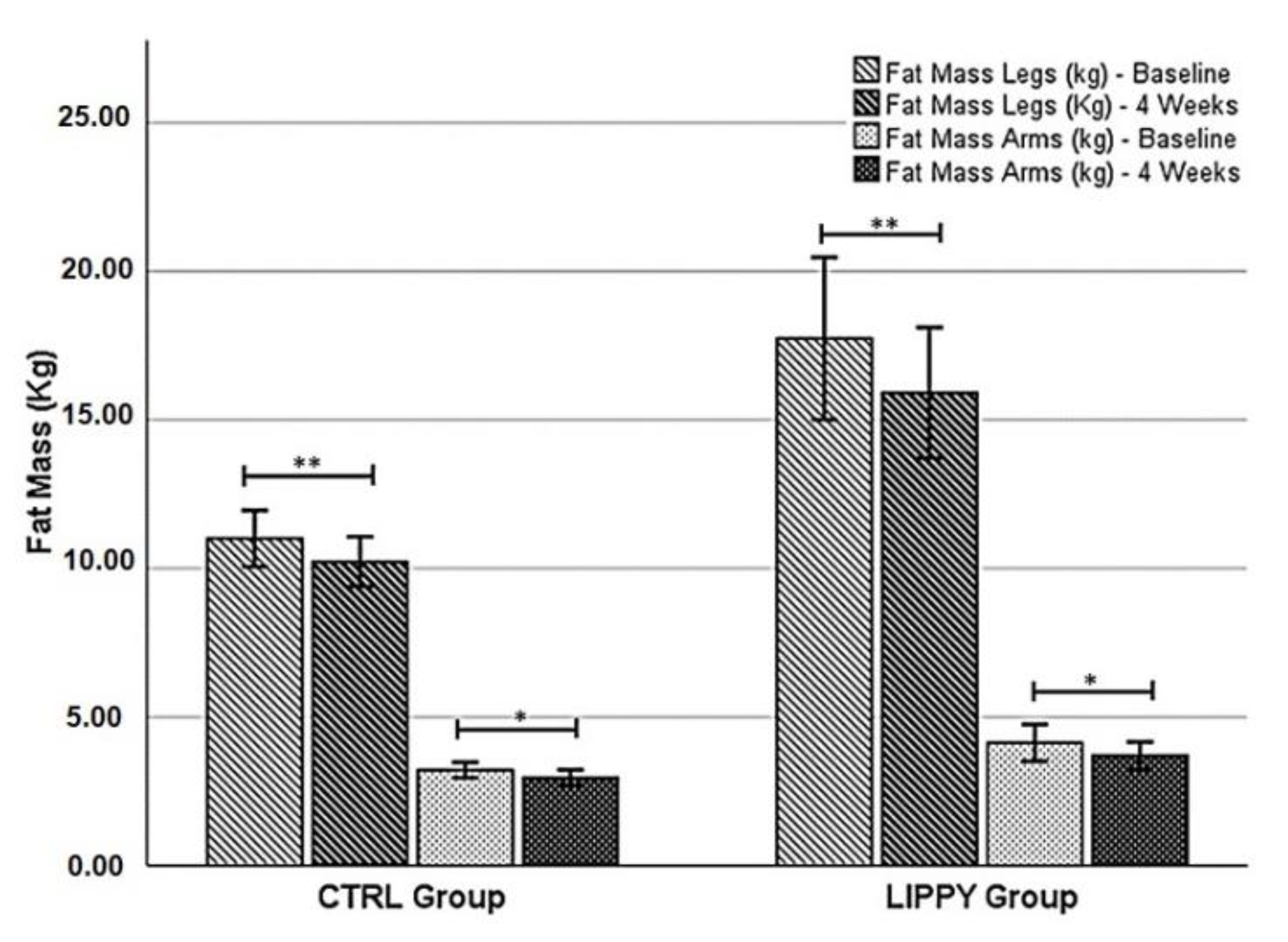
| Fat mass, trunk (kg) | 14.09 ± 5.97 | 12.39 ± 6.03 | 0.001 ** | 18.66 ± 11.71 | 17.64 ± 11.23 | 0.220 | −13.77 ± 12.44 | −0.81 ± 10.17 | 0.012 * |
| Fat mass, android (kg) | 2.26 ± 1.14 | 2.05 ± 1.14 | 0.003 ** | 3.08 ± 2.06 | 3.08 ± 2.47 | 0.717 | −11.95 ± 13.31 | −1.14 ± 17.68 | 0.094 |
| Fat mass, gynoid (kg) | 5.43 ± 1.74 | 4.92 ± 1.67 | 0.001 **† | 7.81 ± 4.33 | 7.16 ± 3.68 | 0.074† | −9.88 ± 7.44 | −4.27 ± 8.49 | 0.094 |
| Fat mass, total body (kg) | 29.2 ± 9.8 | 26.4 ± 9.49 | 0.001 **† | 42.02 ± 21.94 | 39.25 ± 20.24 | 0.114† | −9.94 ± 8.86 | −3.10 ± 5.65 | 0.042 * |
| Lean mass, arms (kg) | 4.49 ± 0.97 | 4.4 0± 1.02 | 0.117 † | 4.41 ± 0.78 | 4.06 ± 0.68 | 0.225† | −2.16 ± 4.87 | −4.52 ± 11.13 | 0.473 |
| Lean mass, legs (kg) | 14.82 ± 2.58 | 14.46 ± 2.64 | 0.023 * | 17.13 ± 2.83 | 15.87 ± 2.81 | 0.083 | −2.49 ± 3.81 | −4.45 ± 6.96 | 0.373 |
| Lean mass, trunk (kg) | 19.31 ± 2.88 | 19.47 ± 2.95 | 0.516 | 21.42 ± 4.22 | 21.42 ± 4.26 | 0.126 | 0.90 ± 5.00 | 3.47 ± 6.13 | 0.263 |
| Lean mass, total body (kg) | 41.74 ± 6.27 | 41.38 ± 6.32 | 0.367 | 46.65 ± 7.99 | 45.74 ± 7.43 | 0.366 | −0.85 ± 3.52 | 1.89 ± 4.43 | 0.099 |
| Total mass, arms (kg) | 8.01 ± 1.82 | 7.65 ± 1.89 | 0.043 * | 8.74 ± 2.54 | 8.02 ± 1.93 | 0.083 | −4.31 ± 7.90 | −5.94 ± 8.70 | 0.632 |
| Total mass, legs (kg) | 26.65 ± 5.58 | 26.18 ± 5.06 | 0.485 | 36.15 ± 10.62 | 32.64 ± 9.53 | 0.011 * | −1.01 ± 11.38 | −6.87 ± 5.54 | 0.145 |
| Total mass, trunk (kg) | 34.13 ± 7.73 | 32.58 ± 7.66 | 0.006 ** | 40.86 ± 15.33 | 39.93 ± 14.93 | 0.917 | −4.55 ± 5.44 | 1.12 ± 6.40 | 0.026 * |
| Total mass, android (kg) | 5.12 ± 1.45 | 4.93 ± 1.45 | 0.028 * | 6.38 ± 2.63 | 6.53 ± 3.69 | 0.456 | −4.00 ± 6.14 | 3.29 ± 17.09 | 0.141 |
| Total mass, gynoid (kg) | 12.12 ± 2.4 | 11.49 ± 2.36 | 0.001 ** | 15.27 ± 5.58 | 14.27 ± 4.78 | 0.293 | −5.17 ± 4.81 | −2.85 ± 8.14 | 0.380 |
| Fat region %, arms | 39.75 ± 5.61 | 38.06 ± 6.79 | 0.106 | 44.16 ± 10.39 | 44.61 ± 9.55 | 0.467 | −4.27 ± 9.87 | −0.72 ± 6.35 | 0.326 |
| Fat region %, legs | 40.6 ± 6.32 | 39.44 ± 6.20 | 0.029 * | 48.19 ± 9.48 | 47.3 ± 8.43 | 0.183 | −2.86 ± 4.67 | −1.57 ± 4.36 | 0.492 |
| Fat region %, trunk | 39.92 ± 9.40 | 36.41 ± 10.6 | 0.001 ** | 41.61 ± 13.91 | 40.78 ± 12.74 | 0.123 | −9.92 ± 9.84 | −2.03 ± 4.90 | 0.029 * |
| Fat region %, android | 42.08 ± 11.31 | 39.22 ± 12.72 | 0.001 ** | 43.04 ± 16.63 | 41.8 ± 14.67 | 0.071 | −8.6 ± 10.37 | −4.21 ± 8.18 | 0.273 |
| Fat region %, gynoid | 43.95 ± 6.90 | 41.88 ± 7.39 | 0.003 ** | 48.20 ± 11.03 | 47.65 ± 9.34 | 0.121 | −4.98 ± 5.93 | −1.84 ± 4.29 | 0.164 |
| Fat region %, total body | 39.02 ± 7.13 | 36.78 ± 7.57 | 0.002 ** | 42.78 ± 11.63 | 42.5 ± 10.08 | 0.796 | −6.07 ± 6.45 | 1.15 ± 11.92 | 0.061 |
| IMAT | 1.14 ± 0.43 | 1.02 ± 0.45 | 0.004 ** | 1.37 ± 0.54 | 1.35 ± 0.54 | 0.674 | −11.57 ± 11.88 | 0.74 ± 5.38 | 0.006 ** |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Renzo, L.; Cinelli, G.; Romano, L.; Zomparelli, S.; Lou De Santis, G.; Nocerino, P.; Bigioni, G.; Arsini, L.; Cenname, G.; Pujia, A.; et al. Potential Effects of a Modified Mediterranean Diet on Body Composition in Lipoedema. Nutrients 2021, 13, 358. https://doi.org/10.3390/nu13020358
Di Renzo L, Cinelli G, Romano L, Zomparelli S, Lou De Santis G, Nocerino P, Bigioni G, Arsini L, Cenname G, Pujia A, et al. Potential Effects of a Modified Mediterranean Diet on Body Composition in Lipoedema. Nutrients. 2021; 13(2):358. https://doi.org/10.3390/nu13020358
Chicago/Turabian StyleDi Renzo, Laura, Giulia Cinelli, Lorenzo Romano, Samanta Zomparelli, Gemma Lou De Santis, Petronilla Nocerino, Giulia Bigioni, Lorenzo Arsini, Giuseppe Cenname, Alberto Pujia, and et al. 2021. "Potential Effects of a Modified Mediterranean Diet on Body Composition in Lipoedema" Nutrients 13, no. 2: 358. https://doi.org/10.3390/nu13020358
APA StyleDi Renzo, L., Cinelli, G., Romano, L., Zomparelli, S., Lou De Santis, G., Nocerino, P., Bigioni, G., Arsini, L., Cenname, G., Pujia, A., Chiricolo, G., & De Lorenzo, A. (2021). Potential Effects of a Modified Mediterranean Diet on Body Composition in Lipoedema. Nutrients, 13(2), 358. https://doi.org/10.3390/nu13020358







