Validation of a Short Food Frequency Questionnaire to Measure Dietary Intake of a Selection of Micronutrients in Oncology Patients Undergoing Systemic Therapy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Population and Data Collected
2.2. Administration of FFQ and Diet History
2.3. Development of Food Frequency Questionnaire
2.4. Rationale for Use of Diet History as Reference Method
2.5. Databases and Data Entry Process
2.6. Statistical Analysis and Clinical Significance
3. Results
3.1. Participant Characteristics
3.2. Agreement between FFQ and Diet History
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global cancer statistics 2020: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Multhoff, G.; Molls, M.; Radons, J. Chronic Inflammation in Cancer Development. Front. Immunol. 2012, 2, 98. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colotta, F.; Allavena, P.; Sica, A.; Garlanda, C.; Mantovani, A. Cancer-related inflammation, the seventh hallmark of cancer: Links to genetic instability. Carcinogenesis 2009, 30, 1073–1081. [Google Scholar] [CrossRef] [Green Version]
- Nakamura, K.; Smyth, M.J. Targeting cancer-related inflammation in the era of immunotherapy. Immunol. Cell Biol. 2017, 95, 325–332. [Google Scholar] [CrossRef]
- Dunlop, R.J.; Campbell, C.W. Cytokines and Advanced Cancer. J. Pain Symptom Manag. 2000, 20, 214–232. [Google Scholar] [CrossRef]
- Bilen, M.A.; Martini, D.; Liu, Y.; Shabto, J.M.; Brown, J.T.; Williams, M.; Khan, A.I.; Speak, A.; Lewis, C.; Collins, H.; et al. Combined Effect of Sarcopenia and Systemic Inflammation on Survival in Patients with Advanced Stage Cancer Treated with Immunotherapy. Oncologist 2019, 25, e528–e535. [Google Scholar] [CrossRef] [Green Version]
- Köstek, O.; Demir, T. Sarcopenia and Inflammation with Immunotherapy. Oncologist 2020, 25, e875. [Google Scholar] [CrossRef] [Green Version]
- Ahluwalia, N.; Andreeva, V.; Kesse-Guyot, E.; Hercberg, S. Dietary patterns, inflammation and the metabolic syndrome. Diabetes Metab. 2013, 39, 99–110. [Google Scholar] [CrossRef]
- Key, T.J.; Bradbury, K.E.; Perez-Cornago, A.; Sinha, R.; Tsilidis, K.K.; Tsugane, S. Diet, nutrition, and cancer risk: What do we know and what is the way forward? BMJ 2020, 368, m996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz, R.B.; Hernández, P.S. Diet and cancer: Risk factors and epidemiological evidence. Maturitas 2014, 77, 202–208. [Google Scholar] [CrossRef]
- Bodén, S.; Myte, R.; Wennberg, M.; Harlid, S.; Johansson, I.; Shivappa, N.; Hébert, J.R.; Van Guelpen, B.; Nilsson, L.M. The inflammatory potential of diet in determining cancer risk; A prospective investigation of two dietary pattern scores. PLoS ONE 2019, 14, e0214551. [Google Scholar] [CrossRef] [PubMed]
- Block, K.I.; Koch, A.C.; Mead, M.N.; Tothy, P.K.; Newman, R.A.; Gyllenhaal, C. Impact of antioxidant supplementation on chemotherapeutic toxicity: A systematic review of the evidence from randomized controlled trials. Int. J. Cancer 2008, 123, 1227–1239. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, G.M. Use of Antioxidants During Chemotherapy and Radiotherapy Should Be Avoided. CA Cancer J. Clin. 2005, 55, 319–321. [Google Scholar] [CrossRef]
- Embracing cancer immunotherapy with vital micronutrients. World J. Clin. Oncol. 2021, 12, 712–724. [CrossRef]
- Wu, D.; Lewis, E.D.; Pae, M.; Meydani, S.N. Nutritional Modulation of Immune Function: Analysis of Evidence, Mechanisms, and Clinical Relevance. Front. Immunol. 2019, 9, 3160. [Google Scholar] [CrossRef] [PubMed]
- Singh, K.; Bhori, M.; Kasu, Y.A.; Bhat, G.; Marar, T. Antioxidants as precision weapons in war against cancer chemotherapy induced toxicity–Exploring the armoury of obscurity. Saudi Pharm. J. 2018, 26, 177–190. [Google Scholar] [CrossRef]
- Cade, J.; Thompson, R.; Burley, V.; Warm, D. Development, validation and utilisation of food-frequency questionnaires—A review. Public Health Nutr. 2002, 5, 567–587. [Google Scholar] [CrossRef] [Green Version]
- Moludi, J.; Qaisar, S.A.; Alizadeh, M.; Vayghan, H.J.; Naemi, M.; Rahimi, A.; Mousavi, R. The relationship between Dietary Inflammatory Index and disease severity and inflammatory status: A case–control study of COVID-19 patients. Br. J. Nutr. 2021, 1–9. [Google Scholar] [CrossRef]
- Qiu, Y.; Yang, X.; Wang, L.; Gao, K.; Jiang, Z. L-Arginine Inhibited Inflammatory Response and Oxidative Stress Induced by Lipopolysaccharide via Arginase-1 Signaling in IPEC-J2 Cells. Int. J. Mol. Sci. 2019, 20, 1800. [Google Scholar] [CrossRef] [Green Version]
- Raizel, R.; Leite, J.S.M.; Hypólito, T.M.; Coqueiro, A.Y.; Newsholme, P.; Cruzat, V.F.; Tirapegui, J. Determination of the anti-inflammatory and cytoprotective effects of l-glutamine and l-alanine, or dipeptide, supplementation in rats submitted to resistance exercise. Br. J. Nutr. 2016, 116, 470–479. [Google Scholar] [CrossRef] [Green Version]
- Shivappa, N.; Steck, S.E.; Hurley, T.G.; Hussey, J.R.; Hébert, J.R. Designing and developing a literature-derived, population-based dietary inflammatory index. Public Health Nutr. 2014, 17, 1689–1696. [Google Scholar] [CrossRef] [Green Version]
- Berthon, G. Is copper pro- or anti-inflammatory? A reconciling view and a novel approach for the use of copper in the control of inflammation. Inflamm. Res. 1993, 39, 210–217. [Google Scholar] [CrossRef] [PubMed]
- DiNicolantonio, J.J.; Mangan, D.; O’Keefe, J.H. Copper deficiency may be a leading cause of ischaemic heart disease. Open Heart 2018, 5, e000784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goodman, V.L.; Brewer, G.J.; Merajver, S.D. Copper deficiency as an anti-cancer strategy. Endocr.-Relat. Cancer 2004, 11, 255–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buonocore, D.; Negro, M.; Arcelli, E.; Marzatico, F. Anti-inflammatory Dietary Interventions and Supplements to Improve Performance during Athletic Training. J. Am. Coll. Nutr. 2015, 34, 62–67. [Google Scholar] [CrossRef]
- Zhenyukh, O.; González-Amor, M.; Díez, R.R.; Esteban, V.; Ruiz-Ortega, M.; Salaices, M.; Mas, S.; Briones, A.M.; Egido, J. Branched-chain amino acids promote endothelial dysfunction through increased reactive oxygen species generation and inflammation. J. Cell. Mol. Med. 2018, 22, 4948–4962. [Google Scholar] [CrossRef]
- García-Conesa, M.-T.; Philippou, E.; Pafilas, C.; Massaro, M.; Quarta, S.; Andrade, V.; Jorge, R.; Chervenkov, M.; Ivanova, T.; Dimitrova, D.; et al. Exploring the Validity of the 14-Item Mediterranean Diet Adherence Screener (MEDAS): A Cross-National Study in Seven European Countries around the Mediterranean Region. Nutrients 2020, 12, 2960. [Google Scholar] [CrossRef]
- Samman, S.; Herbert, J.; Petocz, P.; Lyons-Wall, P.M. Development and Validation of a Short Questionnaire for Estimating the Intake of Zinc. Biol. Trace Element Res. 2009, 134, 226–234. [Google Scholar] [CrossRef] [PubMed]
- Satia, J.A.; Watters, J.L.; Galanko, J.A. Validation of an Antioxidant Nutrient Questionnaire in Whites and African Americans. J. Am. Diet. Assoc. 2009, 109, 502–508.e6. [Google Scholar] [CrossRef] [Green Version]
- Sublette, M.E.; Segal-Isaacson, C.; Cooper, T.B.; Fekri, S.; Vanegas, N.; Galfalvy, H.C.; Oquendo, M.A.; Mann, J.J. Validation of a Food Frequency Questionnaire to Assess Intake of n-3 Polyunsaturated Fatty Acids in Subjects with and without Major Depressive Disorder. J. Am. Diet. Assoc. 2011, 111, 117–123.e2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Victoria, C.C. “Dietary Questionnaires”. Available online: https://www.cancervic.org.au/research/epidemiology/nutritional_assessment_services (accessed on 26 October 2021).
- Collins, B.J.; Delaney, C.L.; Boffo, J.E.; Miller, M.D. Development and Validation of a Short Food Frequency Questionnaire to Measure Dietary Intake of a Selection of Immune-Modulating Nutrients in Patients with Established Peripheral Arterial Disease. Nutrients 2021, 13, 3316. [Google Scholar] [CrossRef]
- AIWH. “Cancer Data in Australia”. Available online: https://www.aihw.gov.au/reports/cancer/cancer-data-in-australia/contents/cancer-summary-data-visualisation (accessed on 10 August 2021).
- ABS. “Australian Health Survey: Nutrition First Results-Foods and Nutrients”. Available online: https://www.abs.gov.au/statistics/health/health-conditions-and-risks/australian-health-survey-nutrition-first-results-foods-and-nutrients/latest-release (accessed on 10 August 2021).
- FSANZ. “Australian Food Composition Database”. Available online: https://www.foodstandards.gov.au/science/monitoringnutrients/afcd/Pages/default.aspx (accessed on 26 October 2021).
- Morán Fagúndez, L.J.; Rivera Torres, A.; González Sánchez, M.E.; de Torres Aured, M.L.; Pérez Rodrigo, C.; Irles Rocamora, J.A. Diet history: Method and applications. Nutr/ Hosp. 2015, 31 (Suppl. 3), 57–61. [Google Scholar] [CrossRef]
- Shim, J.-S.; Oh, K.; Kim, H.C. Dietary assessment methods in epidemiologic studies. Epidemiol. Health 2014, 36, e2014009. [Google Scholar] [CrossRef]
- Ralph, J.L.; Von Ah, D.; Scheett, A.J.; Hoverson, B.S.; Anderson, C.M. Diet Assessment Methods. Clin. J. Oncol. Nurs. 2011, 15, E114–E121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ludbrook, J. Confidence in Altman-Bland plots: A critical review of the method of differences. Clin. Exp. Pharmacol. Physiol. 2010, 37, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Bland, J.; Altman, D. Comparing methods of measurement: Why plotting difference against standard method is misleading. Lancet 1995, 346, 1085–1087. [Google Scholar] [CrossRef] [Green Version]
- Bilic-Zulle, L. Comparison of methods: Passing and Bablok regression. Biochem. Med. 2011, 21, 49–52. [Google Scholar] [CrossRef]
- Głabska, D.; Staniec, A.; Guzek, D. Assessment of Validity and Reproducibility of the Zinc-Specific Dietary Intake Questionnaire Conducted for Young Polish Female Respondents. Nutrients 2018, 10, 104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dhawan, D.K.; Chadha, V.D. Zinc: A promising agent in dietary chemoprevention of cancer. Indian J. Med. Res. 2010, 132, 676–682. [Google Scholar] [PubMed]
- Abiri, B.; Vafa, M. Vitamin C and Cancer: The Role of Vitamin C in Disease Progression and Quality of Life in Cancer Patients. Nutr. Cancer 2020, 73, 1–11. [Google Scholar] [CrossRef]
- Giavarina, D. Understanding Bland Altman analysis. Biochem. Med. 2015, 25, 141–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristics | Mean ± SD or n (%) |
|---|---|
| Age a | 64.53 ± 12.04 |
| Gender a | |
| Male | 55 (49.1%) |
| Female | 57 (50.9%) |
| Type of therapy a | |
| Chemotherapy | 68 (60.7%) |
| Immunotherapy | 23 (20.5%) |
| Chemotherapy and Immunotherapy | 16 (14.3%) |
| Targeted Therapy | 3 (2.7%) |
| Type of Cancer a | |
| Solid tumors | 111 (99.1%) |
| Hematological cancer | 1 (0.9%) |
| Living Situation a | |
| Home | 112 (100%) |
| Time taken to complete FFQ b | 9.96 ± 2.60 |
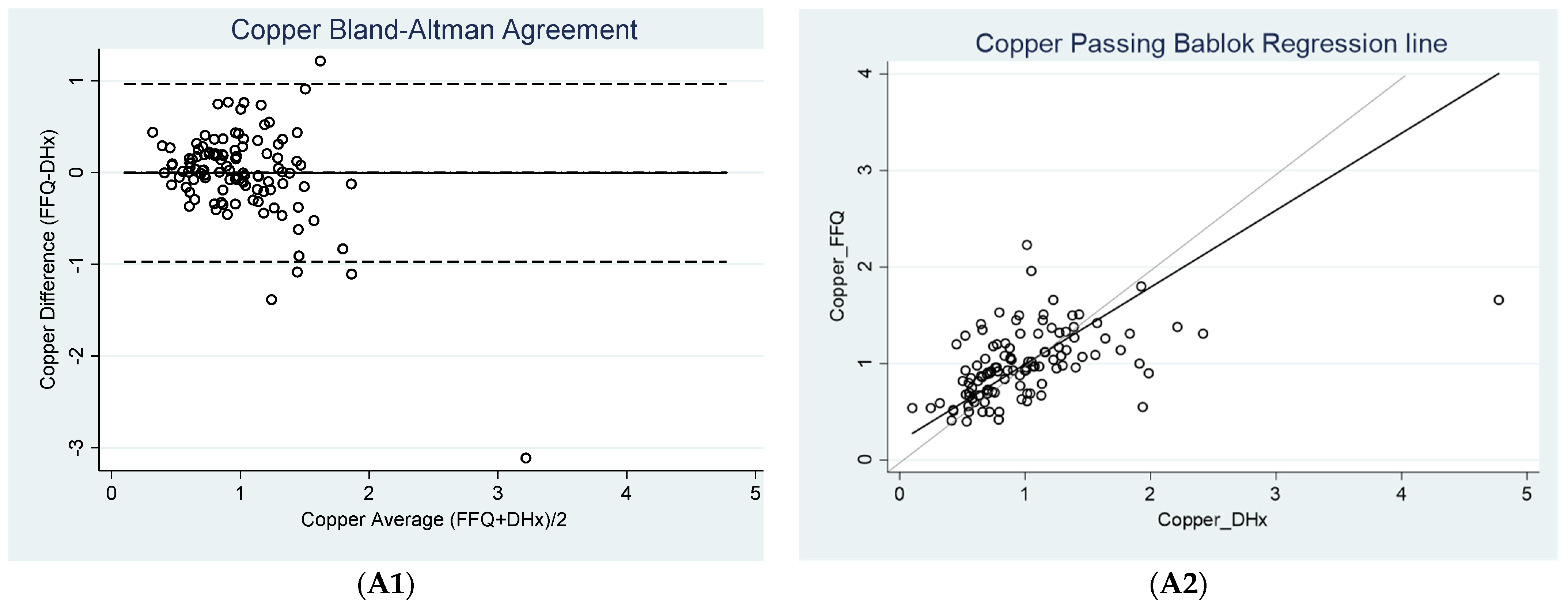
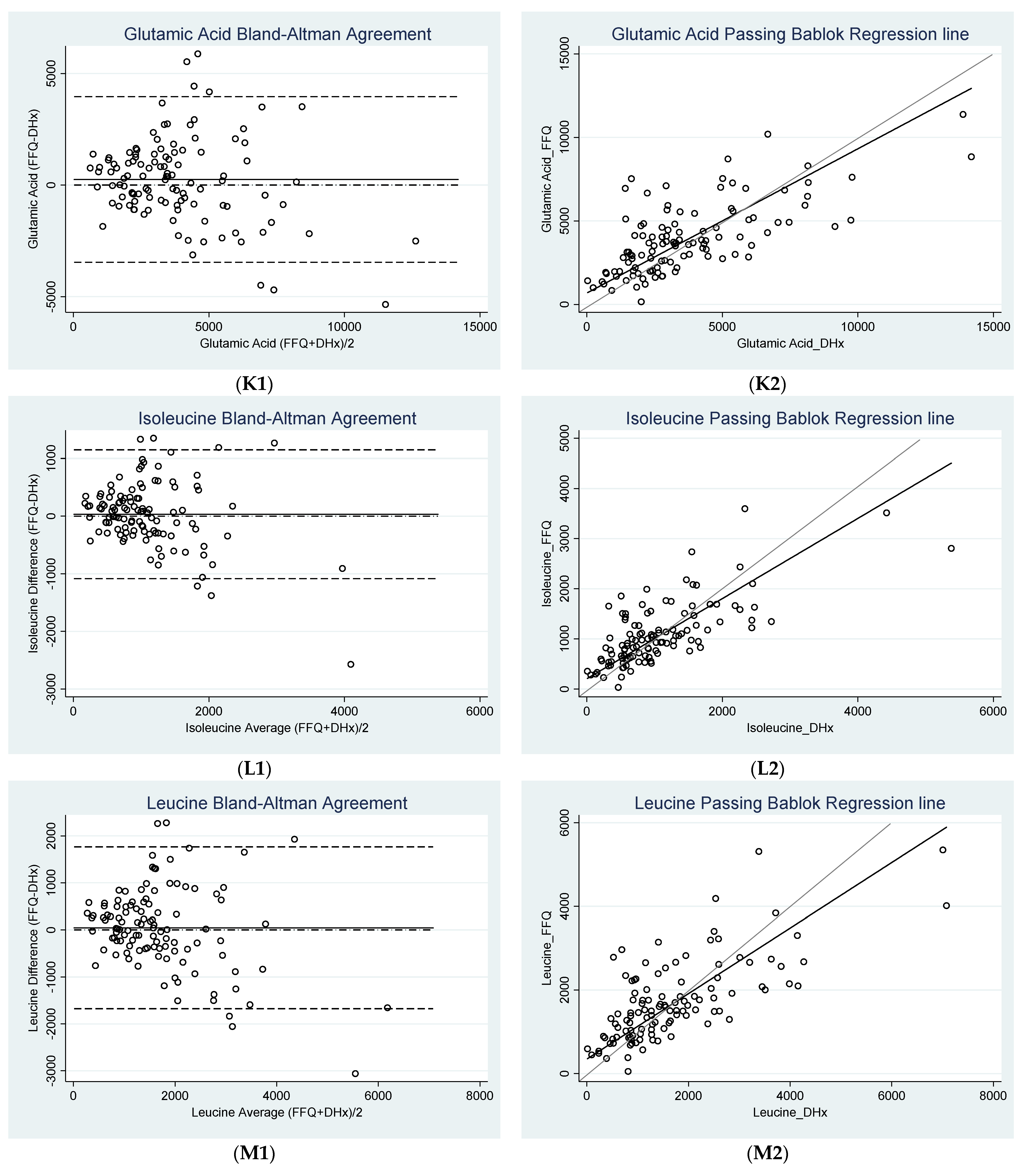
| Nutrient | Bias ± SD (95% CI) | Lower LOA (95% CI) | Upper LOA (95% CI) | Clinically Acceptable Bias (±) a | Clinically Acceptable LOA (±) b |
|---|---|---|---|---|---|
| Copper (mg) | 0.00 * ± 0.49 (−0.10, 0.09) | −0.97 c (−1.13, −0.81) | 0.96 c (0.80, 1.12) | 0.55 | 1.10 |
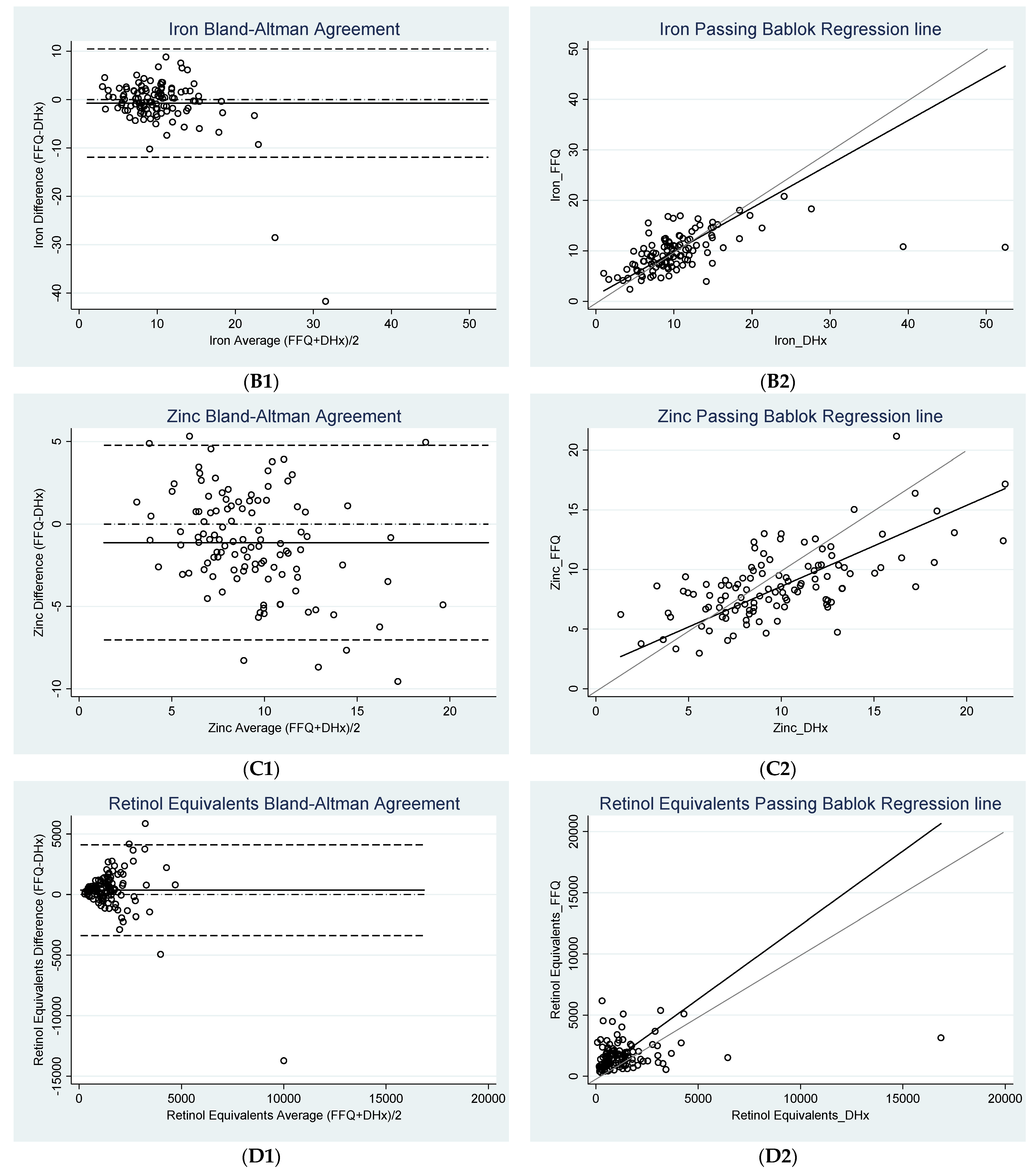
| Iron (mg) | −0.73 * ± 5.72 (−1.80, 0.34) | −11.93 (−13.79, −10.08) | 10.47 (8.62, 12.33) | 4.08 | 8.16 |
| Zinc (mg) | −1.13 * ± 3.01 (−1.69, −0.56) | −7.03 (−8.01, −6.05) | 4.78 (3.80, 5.75) | 2.10 | 4.20 |
| Retinol Equivalents (µg) | 361.52 * ± 1911.10 (3.69, 719.36) | −3384.16 c (−4003.95, −2764.38) | 4107.21 c (3487.42, 4727.00) | 2395.98 | 4791.95 |
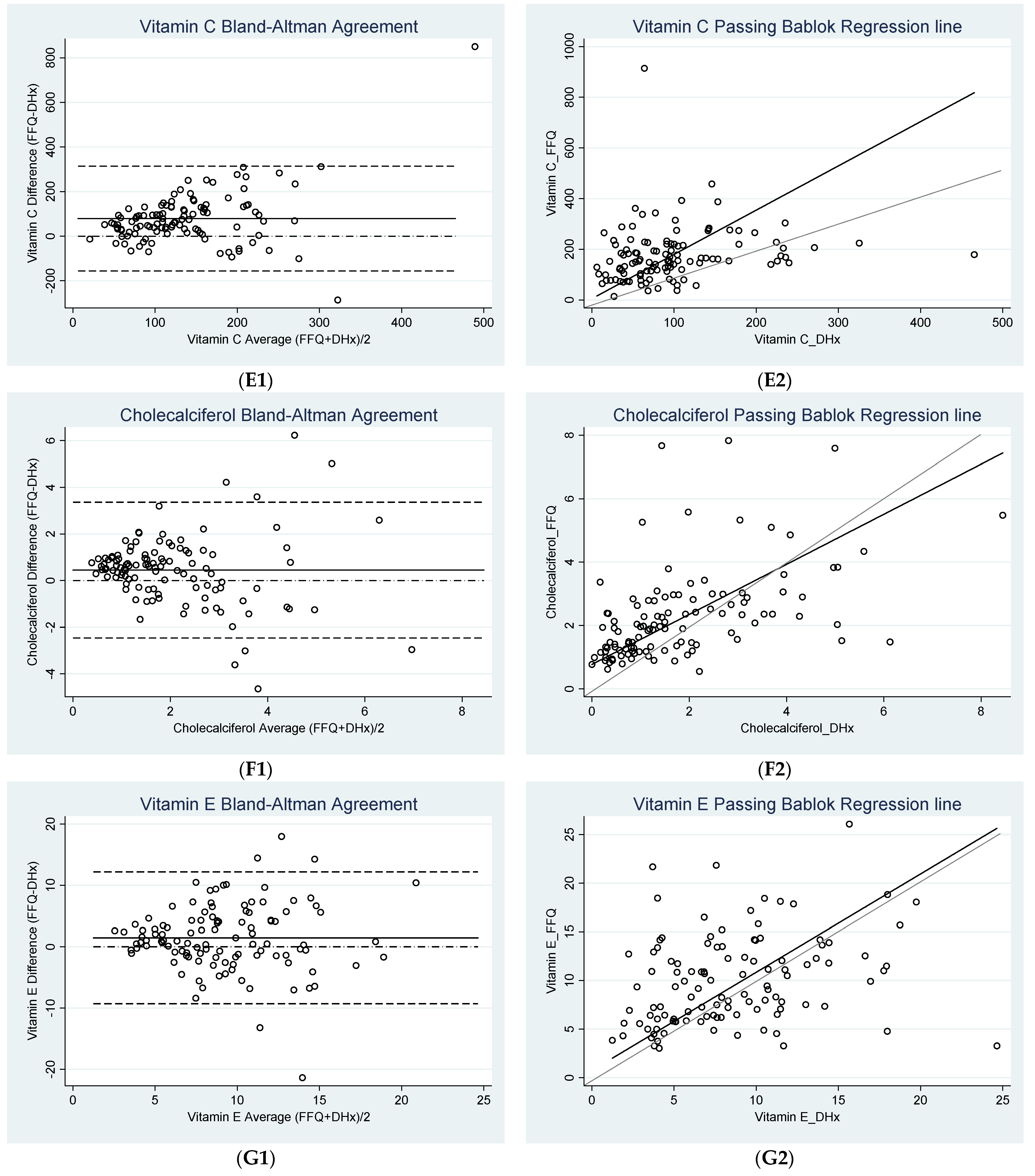
| Vitamin C (mg) | 79.23 ± 119.99 (56.76, 101.69) | −155.95 (−194.87, −117.04) | 314.40 (275.49,353.32) | 48.77 | 97.54 |
| Cholecalciferol (D3) (µg) | 0.45 * ± 1.49 (0.17, 0.73) | −2.46 c (−2.94, −1.98) | 3.37 c (2.88, 3.85) | 5.39 | 10.78 |
| Vitamin E (mg) | 1.45 * ± 5.48 (0.43, 2.48) | −9.29 c (−11.07, −7.51) | 12.19 (10.42, 13.97) | 4.77 | 9.54 |
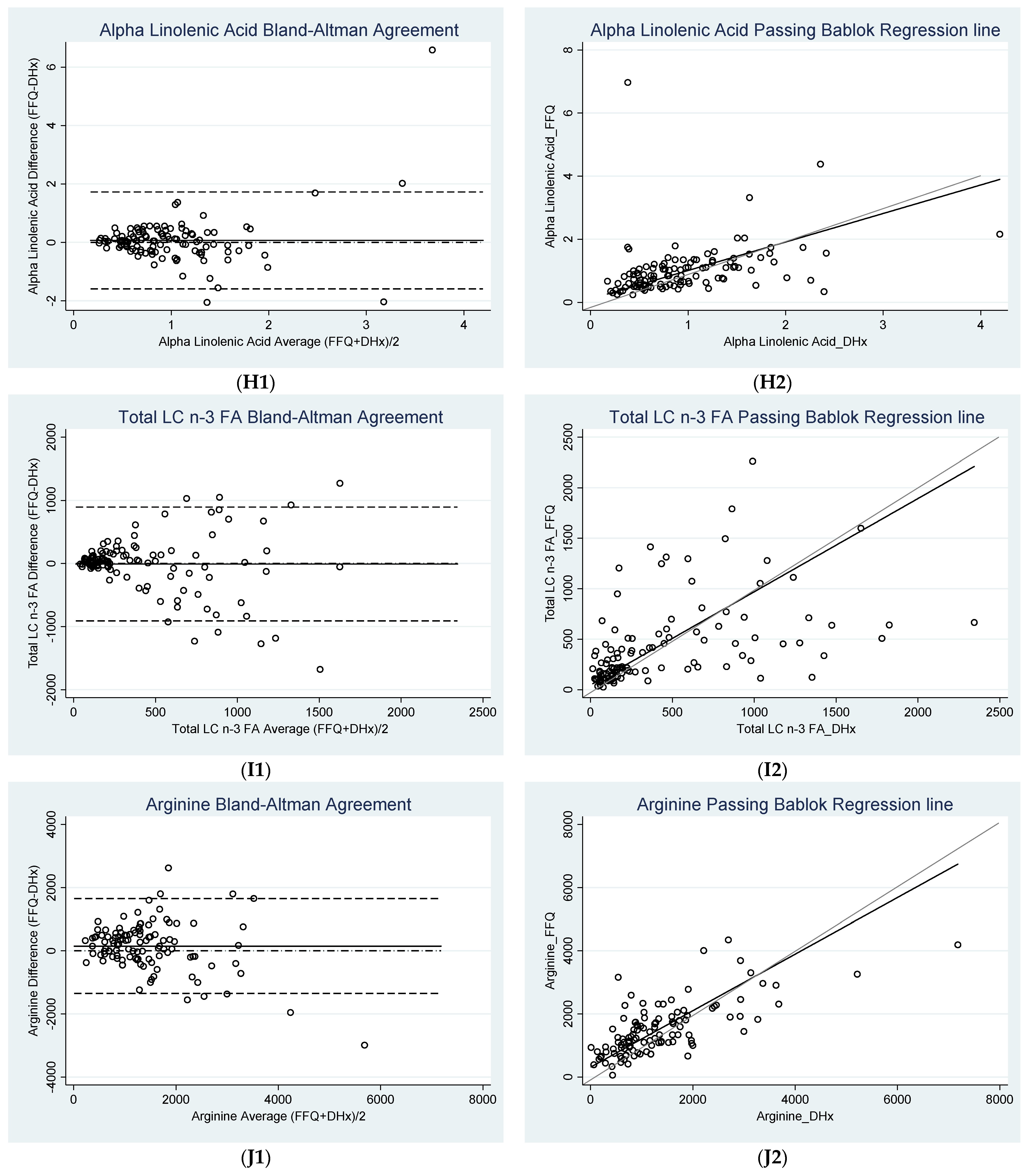
| Alpha Linolenic Acid (g) | 0.07 * ± 0.85 (−0.09, 0.22) | −1.59 (−1.87, −1.32) | 1.72 (1.45, 2.00) | 0.57 | 1.14 |
| Total LC n-3 FA (mg) | −8.59 * ± 460.46 (−94.81, 77.62) | −911.09 c (−1060.42, −761.75) | 893.90 c (744.57, 1043.23) | 1085.91 | 2171.82 |
| Arginine (mg) | 148.95 * ± 766.31 (5.47, 292.44) | −1352.99 c (−1601.52, −1104.47) | 1650.90 c (1402.38, 1899.42) | 1372.01 | 2744.03 |
| Glutamic Acid (mg) | 249.60 * ± 1892.85 (−104.82, 604.02) | −3460.32 c (−4074.19, −2846.45) | 3959.52 c (3345.65, 4573.39) | 2050.85 | 4101.70 |
| Isoleucine (mg) | 31.68 * ± 569.12 (−74.88,138.25) | −1083.78 c (−1268.35, −899.20) | 1147.14 c (962.57, 1331.71) | 994.26 | 1988.52 |
| Leucine (mg) | 43.45 * ± 878.65 (−121.07, 207.97) | −1678.67 c (−1963.63, −1393.72) | 1765.57 c (1480.62, 2050.53) | 1448.07 | 2896.13 |
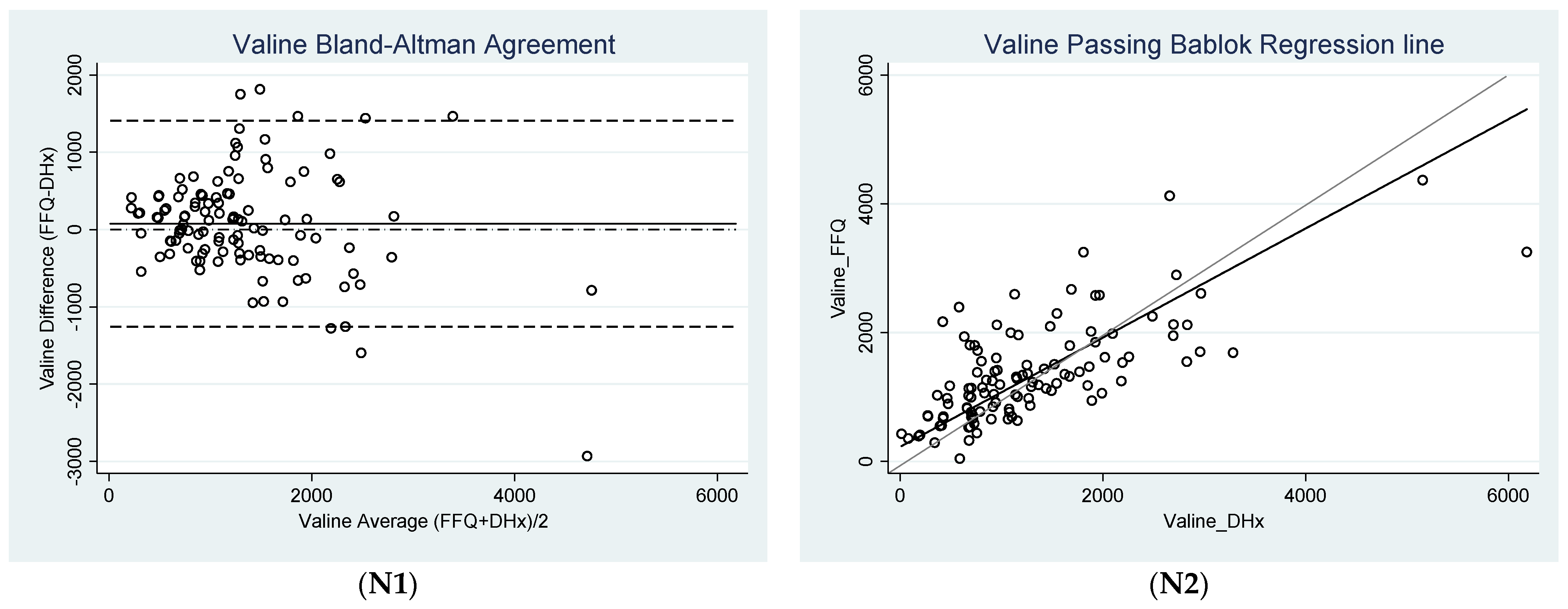
| Valine (mg) | 74.67 * ± 680.50 (−52.75, 202.08) | −1259.09 c (−1479.78, −1038.39) | 1408.42 c (1187.73, 1629.11) | 1151.57 | 2303.13 |
| Nutrient | Intercept (95% CI) | Slope (95% CI) | H Value | p-Value |
|---|---|---|---|---|
| Copper (mg) | 0.20 (0.04, 0.36) | 0.80 (0.64,0.98) | 0.93 | >0.20 c |
| Iron (mg) | 1.20 a (−0.52, 2.30) | 0.87 b (0.73, 1.04) | 1.04 | >0.05 c |
| Zinc (mg) | 1.78 (0.38, 3.07) | 0.68 (0.54, 0.83) | 0.53 | >0.20 c |
| Retinol Equivalents (µg) | 245.31 a (−89.92, 548.05) | 1.21 b (0.86, 1.72) | 0.79 | >0.20 c |
| Vitamin C (mg) | 5.05 a (−53.20, 48.12) | 1.75 (1.25, 2.52) | 1.46 | <0.05 |
| Cholecalciferol (D3) (µg) | 0.77 (0.52, 0.97) | 0.79 b (0.63, 1.04) | 1.19 | >0.10 c |
| Vitamin E (mg) | 0.75 a (−1.27, 2.76) | 1.01 b (0.78, 1.38) | 0.79 | >0.20 c |
| Alpha Linolenic Acid (g) | 0.11 a (−0.04, 0.26) | 0.90 b (0.72, 1.15) | 1.19 | >0.10 c |
| Total LC n-3 FA (mg) | 46.5 a (−4.49, 84.74) | 0.92 b (0.73, 1.17) | 1.19 | >0.10 c |
| Arginine (mg) | 311.54 (110.37, 486.65) | 0.90 b (0.73, 1.11) | 1.06 | >0.20 c |
| Glutamic Acid (mg) | 680.15 (46.56, 1129.52) | 0.86 b (0.72, 1.06) | 1.32 | >0.05 c |
| Isoleucine (mg) | 204.61 (104.47, 319.41) | 0.80 (0.67,0.97) | 1.46 | <0.05 |
| Leucine (mg) | 347.46 (76.69, 492.19) | 0.78 (0.65, 0.97) | 1.59 | <0.02 |
| Valine (mg) | 226.68 (67.68, 379.35) | 0.85 b (0.71, 1.03) | 1.46 | <0.05 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mukherjee, M.S.; Sukumaran, S.; Delaney, C.L.; Miller, M.D. Validation of a Short Food Frequency Questionnaire to Measure Dietary Intake of a Selection of Micronutrients in Oncology Patients Undergoing Systemic Therapy. Nutrients 2021, 13, 4557. https://doi.org/10.3390/nu13124557
Mukherjee MS, Sukumaran S, Delaney CL, Miller MD. Validation of a Short Food Frequency Questionnaire to Measure Dietary Intake of a Selection of Micronutrients in Oncology Patients Undergoing Systemic Therapy. Nutrients. 2021; 13(12):4557. https://doi.org/10.3390/nu13124557
Chicago/Turabian StyleMukherjee, Mitali S., Shawgi Sukumaran, Christopher L. Delaney, and Michelle D. Miller. 2021. "Validation of a Short Food Frequency Questionnaire to Measure Dietary Intake of a Selection of Micronutrients in Oncology Patients Undergoing Systemic Therapy" Nutrients 13, no. 12: 4557. https://doi.org/10.3390/nu13124557
APA StyleMukherjee, M. S., Sukumaran, S., Delaney, C. L., & Miller, M. D. (2021). Validation of a Short Food Frequency Questionnaire to Measure Dietary Intake of a Selection of Micronutrients in Oncology Patients Undergoing Systemic Therapy. Nutrients, 13(12), 4557. https://doi.org/10.3390/nu13124557






