Beyond Refeeding: The Effect of Including a Dietitian in Eating Disorder Treatment. A Systematic Review
Abstract
:1. Introduction
2. Materials and Methods
2.1. Search Strategy and Selection Criteria
2.2. Data Extraction and Analysis
2.3. Quality Appraisal and Risk of Bias
3. Results
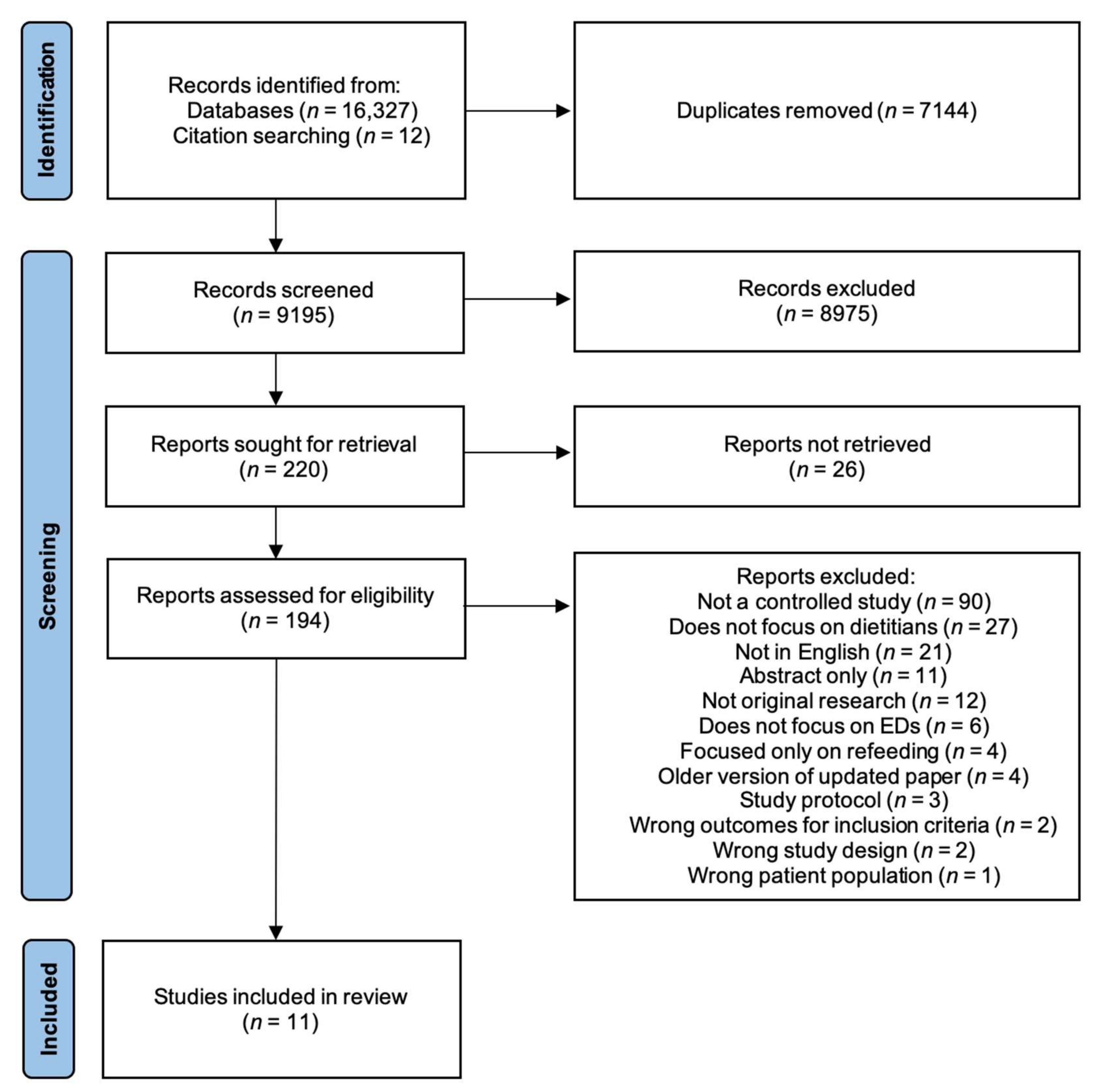
3.1. Study Selection
3.2. Study Characteristics
3.2.1. Participant Characteristics
3.2.2. Dietetic Intervention Characteristics
3.2.3. Comparator Intervention Characteristics
3.3. Study Outcomes
3.3.1. ED Psychopathology
3.3.2. Other Psychopathology
3.3.3. Level of Function and Quality of Life Measures
3.3.4. ED Behaviours/Weight
3.3.5. Diet Quality/Diet Adequacy
3.4. Quality Appraisal and Risk of Bias
4. Discussion
4.1. Summary of Findings
4.2. Intervention Components
4.3. Strengths and Limitations of Included Studies
4.4. Strengths and Limitations of This Review
4.5. Implications for Future Practice and Research
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Appendix A
| Words Relating to Eating Disorders | Search Term Used |
| Eating disorder Disordered eating Anorexia nervosa Anorexic Bulimia nervosa Binge eating disorder Binge eating Feeding disorder Orthorexia nervosa Muscle dysmorphia Rumination disorder * Purging disorder * Night eating syndrome Avoidant/restrictive food intake disorder (ARFID) Eating disorder not otherwise specified (EDNOS) Other specified feeding or eating disorder (OSFED) Unspecified eating or feeding disorder (UFED) Pica | Eating disorder * Disordered eating Anorexi * Bulimi * Binge Feeding disorder * Orthorexia Muscle dysmorphia Rumination disorder * Purging disorder * Night eating syndrome Intake disorder * ARFID EDNOS OSFED UFED Pica |
| Words Relating to Dietitians | Search Term Used |
| Dietitian Dietician Dietetic Nutritionist | Dieti#ian Dietetic * Nutrition * |
| Words Related to Role in Treatment | Search Term Used |
| Treatment Counselling Counsel Therapy Therapies Intervention Care Consultation Recommendation Plan Advice Management Education Prescription Support Role Function * | Treatment * Counsel * Therap * Intervention * Care Consult * Recommend * Plan Advice Management Educat * Prescri * Support Role * Function * |
Appendix B
| Population |
| People with an eating disorder as defined by the Diagnostic and Statistical Manual of Mental Health Disorders 5th edition (DSM-5) [57]; any type (including but not restricted to anorexia nervosa, bulimia nervosa, binge eating disorder, avoidant/restrictive food intake disorder (ARFID), and other specified feeding or eating disorder (OSFED)). Any age, any gender, any ethnicity, and any severity of ED. |
| Intervention |
| Specialist dietetic care (meal support, nutrition counselling, nutrition education etc.) as part of treatment for an ED. |
| Comparator |
| Any other ED treatment modality. |
| Outcome |
For each paper, outcomes measured by any validated instrument or questionnaire were selected for each of the following categories:
|
| Study Design |
| Controlled trials, randomised or non-randomised. |
References
- Hay, P.; Chinn, D.; Forbes, D.; Madden, S.; Newton, R.; Sugenor, L.; Touyz, S.; Ward, W. Royal Australian and New Zealand College of Psychiatrists clinical practice guidelines for the treatment of eating disorders. Aust. N. Z. J. Psychiatry 2014, 48, 977–1008. [Google Scholar] [CrossRef] [PubMed]
- Rock, C.L. Nutritional and Medical Assessment and Management of Eating Disorders. Nutr. Clin. Care 1999, 2, 332–343. [Google Scholar] [CrossRef]
- Le, L.K.-D.; Hay, P.; Mihalopoulos, C. A systematic review of cost-effectiveness studies of prevention and treatment for eating disorders. Aust. N. Z. J. Psychiatry 2018, 52, 328–338. [Google Scholar] [CrossRef] [PubMed]
- Hay, P.; Mitchison, D.; Collado, A.E.L.; González-Chica, D.A.; Stocks, N.; Touyz, S. Burden and health-related quality of life of eating disorders, including Avoidant/Restrictive Food Intake Disorder (ARFID), in the Australian population. J. Eat. Disord. 2017, 5, 21. [Google Scholar] [CrossRef] [PubMed]
- Santomauro, D.F.; Melen, S.; Mitchison, D.; Vos, T.; Whiteford, H.; Ferrari, A.J. The hidden burden of eating disorders: An extension of estimates from the Global Burden of Disease Study 2019. Lancet. Psychiatry 2021, 8, 320–328. [Google Scholar] [CrossRef]
- Galmiche, M.; Déchelotte, P.; Lambert, G.; Tavolacci, M.P. Prevalence of eating disorders over the 2000–2018 period: A systematic literature review. Am. J. Clin. Nutr. 2019, 109, 1402–1413. [Google Scholar] [CrossRef] [PubMed]
- NEDC. An Integrated Response to Complexity: National Eating Disorders Framework 2012; NEDC: Sydney, Australia, 2012. [Google Scholar]
- Association, A.P. Practice guideline for the treatment of patients with eating disorders. Am. J. Psychiatry 2006, 163, 4–54. [Google Scholar]
- NICE. Eating Disorders: Recognition and Treatment; NICE: London, UK, 2017. [Google Scholar]
- Fairburn, C.G. Cognitive Behavior Therapy and Eating Disorders; Guilford Publications: New York, NY, USA, 2008. [Google Scholar]
- Treasure, J.; Schmidt, U.; van Furth, E. Handbook of Eating Disorders; Wiley: Hoboken, NJ, USA, 2003. [Google Scholar]
- Wakefield, A.; Williams, H. (Eds.) Practice Recommendations for the Nutritional Management of Anorexia Nervosa in Adults; Dietitians Association of Australia: Deakin, Australia, 2009. [Google Scholar]
- Hart, S.; Russell, J.; Abraham, S. Nutrition and dietetic practice in eating disorder management. J. Hum. Nutr. Diet. 2011, 24, 144–153. [Google Scholar] [CrossRef]
- Ozier, A.D.; Henry, B.W. Position of the American Dietetic Association: Nutrition Intervention in the Treatment of Eating Disorders. J. Am. Diet. Assoc. 2011, 111, 1236–1241. [Google Scholar] [CrossRef]
- McMaster, C.M.; Wade, T.; Franklin, J.; Hart, S. A review of treatment manuals for adults with an eating disorder: Nutrition content and consistency with current dietetic evidence. Eat. Weight. Disord.-Stud. Anorex. Bulim. Obes. 2021, 26, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Royal College of Psychiatrists. Guidelines for the Nutritional Management of Anorexia Nervosa. Available online: https://www.rcpsych.ac.uk/docs/default-source/improving-care/better-mh-policy/college-reports/college-report-cr130.pdf?sfvrsn=c4aad5e3_6 (accessed on 9 November 2021).
- McMaster Caitlin, M.; Fong, M.; Franklin, J.; Hart, S. Dietetic intervention for adult outpatients with an eating disorder: A systematic review and assessment of evidence quality. Nutr. Rev. 2021, 5, 5. [Google Scholar] [CrossRef]
- Heruc, G.; Hart, S.; Stiles, G.; Fleming, K.; Casey, A.; Sutherland, F.; Jeffrey, S.; Roberton, M.; Hurst, K. ANZAED practice and training standards for dietitians providing eating disorder treatment. J. Eat. Disord. 2020, 8, 77. [Google Scholar] [CrossRef] [PubMed]
- McMaster, C.M. Dietetic Treatment for Adult Outpatients with an Eating Disorder: Evidence, Consensus and Barriers to Implementation. Ph.D. Thesis, University of Sydney, Sydney, Australia, 2021. [Google Scholar]
- Joy, E.A.; Wilson, C.; Varechok, S. The multidisciplinary team approach to the outpatient treatment of disordered eating. Curr. Sports Med. Rep. 2003, 2, 331–336. [Google Scholar] [CrossRef] [PubMed]
- Lian, B.; Forsberg, S.E.; Fitzpatrick, K.K. Adolescent Anorexia: Guiding Principles and Skills for the Dietetic Support of Family-Based Treatment. J. Acad. Nutr. Diet. 2019, 119, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Rienecke, R.D. Family-based treatment of eating disorders in adolescents: Current insights. Adolesc Health Med. Ther. 2017, 8, 69–79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sterling, W.; Crosbie, C.; Shaw, N.; Martin, S. The Use of the Plate-by-Plate Approach for Adolescents Undergoing Family-Based Treatment. J. Acad. Nutr. Diet. 2019, 119, 1075–1084. [Google Scholar] [CrossRef]
- Leonards, C.R.; Koenigs, L.M.P.; Norton, C. The Utilization of Nutrition Services in Family-Based Treatment by Members of the Society for Adolescent Health and Medicine in the care of Adolescents with Anorexia Nervosa. In N/A.; University of Massachusetts Amherst: Amherst, MA, USA, 2019; Volume 219. [Google Scholar]
- Lian, B.; Herrin, M.; Oliver, A. Dietitians Using Family Based Treatment (FBT): Strategies and Guidance (Workshop). Available online: https://iaedp.confex.com/iaedp/2019/webprogram/Session4144.html (accessed on 6 October 2021).
- Veritas Health Innovation. Covidence Systematic Review Software; Veritas Health Innovation Melbourne: Melbourne, Australia. Available online: https://www.covidence.org/ (accessed on 6 December 2021).
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders: DSM-5; American Psychiatric Association: Arlington, VA, USA, 2013. [Google Scholar]
- Hong, Q.N.; Fàbregues, S.; Bartlett, G.; Boardman, F.; Cargo, M.; Dagenais, P.; Gagnon, M.-P.; Griffiths, F.; Nicolau, B.; O’Cathain, A.; et al. The Mixed Methods Appraisal Tool (MMAT) Version 2018 for Information Professionals and Researchers. IOS Press: Amsterdam, The Netherlands, 2018. [Google Scholar]
- Bachar, E.; Latzer, Y.; Kreitler, S.; Berry, E.M. Empirical comparison of two psychological therapies. Self psychology and cognitive orientation in the treatment of anorexia and bulimia. J. Psychother. Pract. Res. 1999, 8, 115–128. [Google Scholar] [PubMed]
- Brambilla, F.; Samek, L.; Company, M.; Lovo, F.; Cioni, L.; Mellado, C. Multivariate therapeutic approach to binge-eating disorder: Combined nutritional, psychological and pharmacological treatment. Int. Clin. Psychopharmacol. 2009, 24, 312–317. [Google Scholar] [CrossRef]
- Compare, A.; Calugi, S.; Marchesini, G.; Shonin, E.; Grossi, E.; Molinari, E.; Dalle Grave, R. Emotionally focused group therapy and dietary counseling in binge eating disorder. Effect on eating disorder psychopathology and quality of life. Appetite 2013, 71, 361–368. [Google Scholar] [CrossRef]
- Compare, A.; Tasca, G.A. The Rate and Shape of Change in Binge Eating Episodes and Weight: An Effectiveness Trial of Emotionally Focused Group Therapy for Binge-Eating Disorder. Clin. Psychol. Psychother. 2016, 23, 24–34. [Google Scholar] [CrossRef]
- Hall, A.; Crisp, A.H. Brief psychotherapy in the treatment of anorexia nervosa. Outcome at one year. Br. J. Psychiatry 1987, 151, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Hsu, L.K.; Rand, W.; Sullivan, S.; Liu, D.W.; Mulliken, B.; McDonagh, B.; Kaye, W.H. Cognitive therapy, nutritional therapy and their combination in the treatment of bulimia nervosa. Psychol. Med. 2001, 31, 871–879. [Google Scholar] [CrossRef] [PubMed]
- Laessle, R.G.; Beumont, P.J.; Butow, P.; Lennerts, W.; O’Connor, M.; Pirke, K.M.; Touyz, S.W.; Waadt, S. A comparison of nutritional management with stress management in the treatment of bulimia nervosa. Br. J. Psychiatry 1991, 159, 250–261. [Google Scholar] [CrossRef] [PubMed]
- Ruggiero, G.M.; Mauri, M.C.; Omboni, A.C.; Volonteri, L.S.; Dipasquale, S.; Malvini, L.; Redaelli, G.; Pasqualinotto, L.; Cavagnini, F. Nutritional management of anorexic patients with and without fluoxetine: 1-year follow-up. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2003, 27, 425–430. [Google Scholar] [CrossRef]
- Serfaty, M.A.; Turkington, D.; Heap, M.; Ledsham, L.; Jolley, E. Cognitive therapy versus dietary counselling in the outpatient treatment of anorexia nervosa: Effects of the treatment phase. Eur. Eat. Disord. Rev. 1999, 7, 334–350. [Google Scholar] [CrossRef]
- Sundgot-Borgen, J.; Rosenvinge, J.H.; Bahr, R.; Schneider, L.S. The effect of exercise, cognitive therapy, and nutritional counseling in treating bulimia nervosa. Med. Sci. Sports Exerc. 2002, 34, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Ventura, M.; Bauer, B. Empowerment of women with purging-type bulimia nervosa through nutritional rehabilitation. Eat. Weight.Disord. EWD 1999, 4, 55–62. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Linardon, J.; Hindle, A.; Brennan, L. Dropout from cognitive-behavioral therapy for eating disorders: A meta-analysis of randomized, controlled trials. Int. J. Eat. Disord. 2018, 51, 381–391. [Google Scholar] [CrossRef]
- Probst, P.; Grummich, K.; Heger, P.; Zaschke, S.; Knebel, P.; Ulrich, A.; Büchler, M.W.; Diener, M.K. Blinding in randomized controlled trials in general and abdominal surgery: Protocol for a systematic review and empirical study. Syst. Rev. 2016, 5, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castro, J.; Deulofeu, R.; Gila, A.; Puig, J.; Toro, J. Persistence of nutritional deficiencies after short-term weight recovery in adolescents with anorexia nervosa. Int. J. Eat. Disord. 2004, 35, 169–178. [Google Scholar] [CrossRef]
- Schebendach, J.E.; Mayer, L.E.; Devlin, M.J.; Attia, E.; Contento, I.R.; Wolf, R.L.; Walsh, B.T. Food Choice and Diet Variety in Weight-Restored Patients with Anorexia Nervosa. J. Am. Diet. Assoc. 2011, 111, 732–736. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schebendach, J.; Mayer, L.E.S.; Devlin, M.J.; Attia, E.; Walsh, B.T. Dietary energy density and diet variety as risk factors for relapse in anorexia nervosa: A replication. Int. J. Eat. Disord. 2012, 45, 79–84. [Google Scholar] [CrossRef] [Green Version]
- Mayer, L.E.S.; Schebendach, J.; Bodell, L.P.; Shingleton, R.M.; Walsh, B.T. Eating behavior in anorexia nervosa: Before and after treatment. Int. J. Eat. Disord. 2012, 45, 290–293. [Google Scholar] [CrossRef] [Green Version]
- Heruc, G.; Hurst, K.; Casey, A.; Fleming, K.; Freeman, J.; Fursland, A.; Hart, S.; Jeffrey, S.; Knight, R.; Roberton, M.; et al. ANZAED eating disorder treatment principles and general clinical practice and training standards. J. Eat. Disord. 2020, 8, 63. [Google Scholar] [CrossRef] [PubMed]
- Fairburn, C.G.; Jones, R.; Peveler, R.C.; Hope, R.A.; O’Connor, M. Psychotherapy and Bulimia Nervosa: Longer-term Effects of Interpersonal Psychotherapy, Behavior Therapy, and Cognitive Behavior Therapy. Arch. Gen. Psychiatry 1993, 50, 419–428. [Google Scholar] [CrossRef] [PubMed]
- Jahan, N.; Naveed, S.; Zeshan, M.; Tahir, M.A. How to Conduct a Systematic Review: A Narrative Literature Review. Cureus 2016, 8, e864. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mond, J.M.; Hay, P.J. Public perceptions of binge eating and its treatment. Int. J. Eat. Disord. 2008, 41, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Mond, J.M.; Hay, P.J.; Rodgers, B.; Owen, C.; Beumont, P.J.V. Beliefs of the public concerning the helpfulness of interventions for bulimia nervosa. Int. J. Eat. Disord. 2004, 36, 62–68. [Google Scholar] [CrossRef] [PubMed]
- Nishizono-Maher, A.; Escobar-Koch, T.; Ringwood, S.; Banker, J.; van Furth, E.; Schmidt, U. What are the top five essential features of a high quality eating disorder service? A comparison of the views of US and UK eating disorder sufferers, carers and health professionals. Eur. Eat. Disord. Rev. 2011, 19, 411–416. [Google Scholar] [CrossRef]
- Page, S.J.; Persch, A.C. Recruitment, retention, and blinding in clinical trials. Am. J. Occup. Ther. 2013, 67, 154–161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McMaster, C.M.; Wade, T.; Basten, C.; Franklin, J.; Ross, J.; Hart, S. Rationale and development of a manualised dietetic intervention for adults undergoing psychological treatment for an eating disorder. Eat. Weight. Disord.-Stud. Anorex. Bulim. Obes. 2021, 26, 1467–1481. [Google Scholar] [CrossRef] [PubMed]
- Collins, C.E.; Watson, J.F.; Guest, M.; Boggess, M.M.; Duncanson, K.; Pezdirc, K.; Rollo, M.; Hutchesson, M.J.; Burrows, T.L. Reproducibility and comparative validity of a food frequency questionnaire for adults. Clin. Nutr. 2014, 33, 906–914. [Google Scholar] [CrossRef]
- Giles, G.G.; Ireland, P.D. Dietary Questionnaire for Epidemiological Studies (Version 3.2); Cancer Council Victoria: Melbourne, Australia, 1996. [Google Scholar]
- Schebendach, J.E.; Porter, K.J.; Wolper, C.; Walsh, B.T.; Mayer, L.E.S. Accuracy of self-reported energy intake in weight-restored patients with anorexia nervosa compared with obese and normal weight individuals. Int. J. Eat. Disord. 2012, 45, 570–574. [Google Scholar] [CrossRef] [PubMed]
- Hadigan, C.M.; Anderson, E.J.; Miller, K.K.; Hubbard, J.L.; Herzog, D.B.; Klibanski, A.; Grinspoon, S.K. Assessment of macronutrient and micronutrient intake in women with anorexia nervosa. Int. J. Eat. Disord. 2000, 28, 284–292. [Google Scholar] [CrossRef]
- Garner, D.; Olmstead, M.P.; Polivy, J. The Eating Disorders Inventory: A measure of the cognitive behavior dimensions of anorexia and bulimia. In Anorexia Nervosa: Recent Developments in Research; Alan R. Liss, Inc.: New York, NY, USA, 1983; pp. 173–184. [Google Scholar]
- Wade, T. Eating Attitudes Test. In Encyclopedia of Feeding and Eating Disorders; Wade, T., Ed.; Springer: Singapore, 2016; pp. 1–4. [Google Scholar] [CrossRef]
- Upton, J. Beck Depression Inventory (BDI). In Encyclopedia of Behavioral Medicine; Gellman, M.D., Turner, J.R., Eds.; Springer: New York, NY, USA, 2013; pp. 178–179. [Google Scholar] [CrossRef]
- Lovibond, S.H.; Lovibond, P.F. Manual for the Depression Anxiety Stress Scales; Psychology Foundation of Australia: Sydney, Australia, 1995. [Google Scholar]
- Burckhardt, C.S.; Anderson, K.L. The Quality of Life Scale (QOLS): Reliability, validity, and utilization. Health Qual. Life Outcomes 2003, 1, 60. [Google Scholar] [CrossRef] [Green Version]
- International Wellbeing Group. Personal Wellbeing Index, 5th ed.; Centre on Quality of Life, Deakin University: Melbourne, Australia, 2013. [Google Scholar]
| Participant Characteristics | Dietetic Intervention Group | Comparator Intervention Group(s) | ||||||||
| Study | Study Design, Country | Total Sample Size, Final Sample Size (% Drop Out), % Female | ED Diagnosis (n), Mean Age (SD) | No. (No. of Drop-Outs) | Mean Baseline BMI | Group/Individual, Duration | Intervention Type | No. (No. of Drop-Outs) | Mean Baseline BMI | Group/ Individual, Duration |
| Bachar 1999 [29] | RCT, Israel | 44, 33 (25), 100 | BN (25), 24.1 (SD = 3.3) AN (8), 18.1 (SD = 2.4) | 10 (3) | NR | Individual, 6 months | SPT + NC COT + NC | 17 (3) 17 (5) | NR | Individual, 12 months |
| Brambilla 2009 [30] | RCT, Italy | 30, 30 (0), 100 Inconsistent reporting of sample size and drop-out. | BED (30), 42.9 (SD = 9) | 10 (0) | 34 (SD = 5) | Group CBT + assume diet component individual but NR, 6 months | Group 1: 1700 calorie macronutrient-controlled diet + CBT + sertraline (50–150 mg/d) + topiramate (25–150 mg/d) Group 2: 1700 calorie, macronutrient-controlled diet + CBT + sertraline (50–150 mg/d) | 10 (0) 10 (0) | 39 (SD = 6) 34 (SD = 6) | Group CBT, assume diet component individual but NR, 6 months Group CBT, assume diet component individual but NR, 6 months |
| Compare 2013 & 2016 [31,32] | Controlled observational study, Italy | 189, 164 (13), 50 | BED (189), EFT group: 50.8 (SD = 6.0); Combined therapy group: 51.1 (SD = 4.1); DT group: 50.4 (SD = 4.7) | 63 (17) | 32.3 (SD = 1.3) | Individual and group, 20 weeks | Emotion-focused therapy Combined therapy | 63 (8) 63 (0) | 33.0 (SD = 1.6) 33.6 (SD = 2.6) | Group, 5 months Group EFT, 5 months + Individual DT, 3 months |
| Hall 1987 [33] | RCT, UK | 30, 25 (17), 100 | AN (30), 19.56 (range 13–27) | 15 (4) | Weight 39.54 kg | Individual, 12–24 weeks | Individual and family psychotherapy | 15 (1) | Weight 41.0 kg | Individual, 12–24 weeks |
| Hsu 2001 [34] | RCT, USA | 100, 73 (27), 100 | BN (100), 24.2 (SD = 5.6) | 23 (9) | NR | Individual, 14 weeks | Cognitive therapy CNT Support group | 26 (4) 27 (3) 24 (11) | NR | Individual, 14 weeks |
| Laessle 1991 [35] | RCT, Australia and Germany | 55, 48 (13), 100 | BN (55), 23.8 (SD = 3.8) | 27 (5) | 21.2 (SD = 1.8) | Group, 3 months | Stress management | 28 (2) | 20.6 (SD = 1.9) | Group, 3 months |
| Ruggiero 2003 [36] | Non-randomised controlled trial, Italy | 95, 95 (0), 96 | AN (95), 23.47 (SD = 4.93) | 74 (0) | 14.29 (SD = 2.18) | Individual, 12 months+ | Nutritional management + fluoxetine | 21 (0) | 14.83 (SD = 1.53) | Individual, 12 months+ |
| Serfaty 1999 [37] | RCT, UK | 35, 23 (34), 94 | AN (35), 20.9 (SD = 6.3) | 10 (10) | 17.0 (SD = 4.0) | Individual, 20 weeks | Cognitive therapy | 25 (2) | 16.2 (SD = 1.6) | Individual, 20 weeks |
| Sundgot-Borgen 2002 [38] | RCT, Norway | 64, 58 (9), 100 | BN (64), 22.5 (SD = 2.8) | 17 (0) | 21.0 (SD = 2.1) | Group, 16 weeks | Cognitive behavioural therapy Physical exercise | 16 (2) 15 (3) | 20.0 (SD = 1.9) 21.0 (SD = 2.0) | Group, 16 weeks Group, 16 weeks |
| Ventura 1999 [39] | RCT, Italy | 40, 36 (10), 100 | BN (40) PNR group: 24.1 (SD = 6) TNR group: 24.0 (SD = 5.6) | 20 (3) | 20.6 (SD = 1.5) | Individual, 24 weeks | Psychobiological nutritional rehabilitation | 20 (1) | 21 (SD = 1.6) | Individual, 24 weeks |
| Study | Specific Dietetic Intervention Components | General/Non-Dietetic-Specific Component |
| Bachar 1999 [29] |
| |
| Brambilla 2009 [30] |
| |
| Compare 2013 and 2016 [31,32] |
|
|
| Hall 1987 [33] |
|
|
| Hsu 2001 [34] |
| |
| Laessle 1991 [35] |
|
|
| Ruggiero 2003 [36] |
|
|
| Serfaty 1999 [37] |
|
|
| Sundgot-Borgen 2002 [38] |
| |
| Ventura 1999 [39] |
|
|
| Study | Timepoints | ED Psychopathology | Other Psychopathology | Level of Function/ Quality of Life | ED Behaviours/Weight | Diet Adequacy/ Diet Quality |
| Bachar 1999 [29] | Baseline, EoT | EAT 26: NSig within or between gps. | BSI: NSig within or between gps. | NR | NR | NR |
| Brambilla 2009 [30] | Baseline, EoT | EDI-2: Sig decrease within gp at EoT for Gp 1 but not Gp 2 or 3. Between gps NR. | SCL-90-R: Sig decrease within gp at EoT for Gp 1 total scores. Sig within gp at EoT for Gp 2 in subitems “depression” and “interpersonal relationships”. NSig within gp in Gp 3. Between gps NR. | NR | Binge frequency: Sig decrease within gp at EoT for Gp 1 patients but not Gp 2 or 3. Between gps NR. | NR |
| Compare 2013 and 2016 [31,32] | Baseline, EoT, 6-month FUp | BES: Sig decrease within gp at EoT and FUp in CT and EFT gps but not in DC gp. Between gps NR. | BUT: Sig decrease within gp at EoT and FUp for CT and EFT gps but not in DC gp. Between gps NR. | ORWELL-97: Sig decrease within gp at EoT and FUp in all gps. Between gps NR. | BES < 16: Sig within gp at EoT and FUp for CT, EFT but not DC. Between gps NR. Binge frequency: Sig decrease within gp at EoT and FUp for CT and EFT. NR in DC. Between gps NR. Weight: Sig decrease within gp at EoT and FUp for all gps. Between gps NR. | NR |
| Hall 1987 [33] | CCEI at baseline, EoT, 1-year FUp. Weight taken at baseline, 4× during treatment, 1-year FUp | CCEI (eating pattern score): Within gp sig NR. At FUp, PG > DAG in reducing symptoms of food avoidance and anxiety about eating with other people (sig NR). At FUp, DAG > PG in improving bulimia, vomiting, and purgation (sig NR). | CCEI (mental state score): Sig decrease within gp at FUp for dietary advice gp in somatic, phobic, and depression scales. Sig decrease within gp at FUp for psychotherapy gp in obsessional and depression scores. No between-gp differences. | CCEI (social adjustment score): Between-gp difference in social and sexual adjustment scores: PG > DAG. | Weight: Sig increase within gp at FUp for DAG only. No between-gp differences. | NR |
| Hsu 2001 [34] | Full assessment using all instruments assessed at baseline, week 6 of treatment, week 10 of treatment, and EoT Self-report measures and HDRS: week 6 of treatment and week 10 of treatment | EDI: Within gps sig NR. CNT > SG in reducing EDI subscales “drive for thinness”, “bulimia”, “ineffectiveness”, “perfectionism”, “interpersonal distrust”, and “interoceptive awareness”. CT > SG in EDI subscales “drive for thinness” and “ineffectiveness”. CNT > NT in reducing “drive for thinness”, “bulimia” and “ineffectiveness”. CT > NT only on the “bulimia” subscale. No between-gp differences between NT and SG, or between CT and CNT. | DAS: Sig within-gps decrease in DAS at EoT for all gps. Sig between-gp differences CT, CNT > SG. CNT > NT in decreasing DAS scores. No between-gp differences for CT and CNT. | SCS: Sig within-gp increases in SCS at EoT for all gps. Sig between-gp differences for self-control: CT, CNT, NT > SG. Sig between-gp differences in SCS: CNT > NT. | Binge frequency and vomit frequency: Sig within-gp decreases in binge and vomit frequency for all gps. No between-gp differences. | Meal pattern: Sig within-gp increase in number of meals eaten per day for all gps. No between-gp differences. |
| Laessle 1991 [35] | Baseline, week 3 of treatment, EoT, 6-month FUp, 12-month FUp | EDI: Sig decrease within gp at FUp for both gps. No between-gp differences. | BDI: Sig decrease within gp at FUp for BDI depression scores in both gps. Between groups NR. | NR | Binge frequency: Sig decrease within gp at EoT and FUp for both gps. No between-gp differences. Vomiting frequency: Sig decrease within gp at EoT for both gps. No between-gp differences. | Caloric intake: Sig increase in average amount of calories consumed in a day (outside of binges and not vomited) within gp at EoT and FUp in both treatments. No between-gp differences. |
| Ruggiero 2003 [36] | Baseline, 3 months into treatment, 6 months into treatment, 12 months into treatment | EDI: Sig within-gp decrease in “fear of fatness” in nutritional gp. | NR | NR | BMI: Sig within-gp increases in both gps. Pharmacological gp > nutritional treatment-only gp. | NR |
| Serfaty 1999 [37] | Baseline, 6 months into treatment, 6-month FUp | EDI: Sig within-gp decrease in CBT gp. Between groups N/A (100% DT attrition). | BDI: Sig within-gp decrease in CBT gp. Between groups N/A (100% DT attrition). | LCB: Sig within-gp decrease in CBT gp. Between groups N/A (100% DT attrition). | BMI: Sig within-gp increase in BMI in CBT gp. Between groups N/A (100% DT attrition). | NR |
| Sundgot-Borgen 2002 [38] | Baseline, EoT, 6-month FUp, 18-month FUp | EDI: No between-gp differences in “drive for thinness” or “body dissatisfaction” subscales at 18-month FUp. For “bulimia” subscale, CBT > NC at FUp. | NR | NR | Binge frequency: Within gps NR. Sig between-gp differences in reducing binge eating at FUp. Exercise gp > CBT Vomiting frequency: Within gps NR. Sig between-gp differences in reducing vomiting frequency, CBT > NC. Sig within-gp decrease in exercise gp. | NR |
| Ventura 1999 [39] | Monthly during treatment, 3-month FUp, 6-month FUp | NR | NR | NR | Binge frequency: Sig within-gp reduction in binge frequency for both gps. Between-gps PNR > TNR. Vomiting frequency: Sig within-gp reduction in vomiting frequency for both gps. Between-gps PNR > TNR. | Intake of carbohydrate servings: No between-gp differences. Intake of lipid servings (measured by serves of olive oil added): between gps PNR > TNR. |
| Bachar [29] | Brambilla [30] | Compare [31,32] | Hall [33] | Hsu [34] | Laessle [35] | Ruggiero [36] | Serfaty [37] | Sundgot-Borgen [38] | Ventura [39] | |
|---|---|---|---|---|---|---|---|---|---|---|
| Screening Questions | ||||||||||
| S1. Are there clear research questions? | Y | Y | Y | Y | Y | Y | Y | Y | Y | Y |
| S2. Do the collected data allow to address the research questions? | Y | Y | Y | Y | Y | Y | Y | N | N | Y |
| Quantitative randomised controlled trials | ||||||||||
| 2.1. Is randomisation appropriately performed? | CT | CT | NR | CT | CT | CT | NR | Y | CT | CT |
| 2.2. Are the groups comparable at baseline? | Y | Y | NR | Y | Y | Y | NR | N | Y | Y |
| 2.3. Are there complete outcome data? | Y | Y | NR | Y | Y | Y | NR | N | Y | Y |
| 2.4. Are (participants, researchers and) outcome assessors blinded to the intervention provided? | CT | Y | NR | CT | N | N | NR | CT | CT | CT |
| 2.5. Did the participants adhere to the assigned intervention? | CT | CT | NR | CT | Y | CT | NR | N | CT | Y |
| Quantitative non-randomised | ||||||||||
| 3.1. Are the participants representative of the target population? | NR | NR | CT | NR | NR | NR | Y | NR | NR | NR |
| 3.2. Are measurements appropriate regarding both the outcome and intervention (or exposure)? | NR | NR | Y | NR | NR | NR | Y | NR | NR | NR |
| 3.3. Are there complete outcome data? | NR | NR | Y | NR | NR | NR | CT | NR | NR | NR |
| 3.4. Are the confounders accounted for in the design and analysis? | NR | NR | Y | NR | NR | NR | N | NR | NR | NR |
| 3.5. During the study period, is the intervention administered (or exposure occurred) as intended? | NR | NR | CT | NR | NR | NR | CT | NR | NR | NR |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, Y.; Conti, J.; McMaster, C.M.; Hay, P. Beyond Refeeding: The Effect of Including a Dietitian in Eating Disorder Treatment. A Systematic Review. Nutrients 2021, 13, 4490. https://doi.org/10.3390/nu13124490
Yang Y, Conti J, McMaster CM, Hay P. Beyond Refeeding: The Effect of Including a Dietitian in Eating Disorder Treatment. A Systematic Review. Nutrients. 2021; 13(12):4490. https://doi.org/10.3390/nu13124490
Chicago/Turabian StyleYang, Yive, Janet Conti, Caitlin M. McMaster, and Phillipa Hay. 2021. "Beyond Refeeding: The Effect of Including a Dietitian in Eating Disorder Treatment. A Systematic Review" Nutrients 13, no. 12: 4490. https://doi.org/10.3390/nu13124490
APA StyleYang, Y., Conti, J., McMaster, C. M., & Hay, P. (2021). Beyond Refeeding: The Effect of Including a Dietitian in Eating Disorder Treatment. A Systematic Review. Nutrients, 13(12), 4490. https://doi.org/10.3390/nu13124490






