High Adherence to the Food Pyramid’s Recommendations Avoids the Risk of Insufficient Nutrient Intake among Farmers in Peri-Urban Kenya
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Demographic Survey
2.4. Physical Measurements
2.5. Dietary Survey
2.6. Adherence to the Kenyan Food Pyramid’s Recommendations
10 × (number of SVs/lower limit of the recommended range).
10 − 10 × (number of SVs − upper limit)/upper limit.
2.7. Statistical Analyses
3. Results
3.1. Characteristics of Participants
3.2. Energy and Nutrient Intake, and SV Counts
3.3. Risk of Excess or Insufficient Nutrient Intake against Recommended Nutrient Intakes
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization. The Double Burden of Malnutrition; WHO: Geneva, Switzerland, 2017. [Google Scholar]
- Vos, T.; Lim, S.S.; Abbafati, C.; Abbas, K.M.; Abbasi, M.; Abbasifard, M.; Abbasi-Kangevari, M.; Abbastabar, H.; Abd-Allah, F.; Abdelalim, A.; et al. Global burden of 369 diseases and injuries in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1204–1222. [Google Scholar] [CrossRef]
- Murray, C.J.L.; Aravkin, A.Y.; Zheng, P.; Abbafati, C.; Abbas, K.M.; Abbasi-Kangevari, M.; Abd-Allah, F.; Abdelalim, A.; Abdollahi, M.; Abdollahpour, I.; et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: A systematic analysis for the Global Burden of Disease Study 2019. Lancet 2020, 396, 1223–1249. [Google Scholar] [CrossRef]
- Ministry of Health Kenya; World Health Organization; Kenya National Bureau of Statistics. KENYA STEPwise Survey for Non Communicable Diseases Risk Factors 2015 Report; Division of Non Communicable Diseases: Nairobi, Kenya, 2016.
- Kenya National Bureau of Statistics; Ministry of Health Kenya; Kenya Medical Research Institute; National Council for Population and Development; ORC Macro; Center for Disease Control and Prevention. Kenya Demographic and Health Survey 2003. 2004. Available online: https://dhsprogram.com/pubs/pdf/FR151/FR151.pdf (accessed on 12 October 2021).
- Kenya National Bureau of Statistics; Ministry of Health Kenya; National AIDS Control Council; Kenya Medical Research Institute; National Council for Population and Development; The DHS program. Kenya Demographic and Health Survey 2014. 2015. Available online: https://dhsprogram.com/pubs/pdf/fr308/fr308.pdf (accessed on 12 October 2021).
- World Health Organization. Prevalence of anaemia in women of reproductive age. Available online: https://www.who.int/data/gho/data/indicators/indicator-details/GHO/prevalence-of-anaemia-in-women-of-reproductive-age-(-) (accessed on 29 November 2021).
- World Health Organization. Global Nutrition Targets 2025: Anaemia Policy Brief; WHO: Geneva, Switzerland, 2014. [Google Scholar]
- Steyn, N.P.; McHiza, Z.J. Obesity and the nutrition transition in Sub-Saharan Africa. Ann. N. Y. Acad. Sci. 2014, 1311, 88–101. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Healthy Diet. Available online: https://www.who.int/news-room/fact-sheets/detail/healthy-diet (accessed on 29 November 2021).
- Ministry of Health Kenya. National Guidelines for Healthy Diets and Physical Activity; Government of Kenya: Nairobi, Kenya, 2017.
- World Health Organization; Food and Agriculture Organization of the United Nations. Preparation and Use of Food Based Dietary Guidelines; WHO: Geneva, Switzerland, 1998; 107p. [Google Scholar]
- Kurotani, K.; Akter, S.; Kashino, I.; Goto, A.; Mizoue, T.; Noda, M.; Sasazuki, S.; Sawada, N.; Tsugane, S. Quality of diet and mortality among Japanese men and women: Japan Public Health Center based prospective study. BMJ 2016, 352. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwingshackl, L.; Bogensberger, B.; Hoffmann, G. Diet Quality as Assessed by the Healthy Eating Index, Alternate Healthy Eating Index, Dietary Approaches to Stop Hypertension Score, and Health Outcomes: An Updated Systematic Review and Meta-Analysis of Cohort Studies. J. Acad. Nutr. Diet. 2018, 118, 74–100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.; Heianza, Y.; Sun, D.; Huang, T.; Ma, W.; Rimm, E.B.; Manson, J.E.; Hu, F.B.; Willett, W.C.; Qi, L. Improving adherence to healthy dietary patterns, genetic risk, and long term weight gain: Gene-diet interaction analysis in two prospective cohort studies. BMJ 2018. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ministry of Medical services Kenya. Kenya National Clinical Nutrition and Dietetics Reference Manual, 1st ed.; Government of Kenya: Nairobi, Kenya, 2010.
- Kenya National Bureau of Statistics. Kenya Population and Housing Census Volume I: Population by County and Sub-County; Kenya National Bureau of Statistics: Nairobi, Kenia, 2019.
- Kiambu county government official site. Available online: https://kiambu.go.ke/ (accessed on 29 November 2021).
- Harvard Humanitarian Initiative. KoBoToolbox/Data Collection Tools for Challenging Environments. Available online: https://kobotoolbox.org/ (accessed on 30 November 2021).
- Hjelm, L.; Mathiassen, A.; Miller, D.; Wadhwa, A. VAM Guidance Paper: Creation of a Wealth Index; World Food Program: Roma, Italy, 2017. [Google Scholar]
- World Health Organization. Body Mass Index (BMI). Available online: https://www.euro.who.int/en/health-topics/disease-prevention/nutrition/a-healthy-lifestyle/body-mass-index-bmi (accessed on 30 November 2021).
- Anono, E.; Walsh, H.; Kanerva, N.; Mubasu, D.; Okoth, V.; Clinton, B.; Hyvönen, A.; Ochola, S.; Erkkola, M.; Onywera, V.; et al. Photographic Food Atlas for Kenyan Adolescents (9–14 Years); Kanerva, F.N., Ed.; Kenya-Finland Education and Research Alliance: Nairobi, Kenya, 2018. [Google Scholar]
- Food and Agriculture Organization of the United Nations & Government of Kenya. Kenya Food Composition Tables; FAO: Nairobi, Kenya, 2018; p. 254. [Google Scholar]
- Ministry of Education, Culture, Sports, Science and Technology Japan. MEXT: Standard Tables of Food Composition in Japan—2015 (Seventh Revised Version). Available online: https://www.mext.go.jp/en/policy/science_technology/policy/title01/detail01/1374030.htm (accessed on 30 November 2021).
- Institute of Medicine. Dietary Reference Intakes: The Essential Guide to Nutrient Requirements; National Academies Press: Washington, DC, USA, 2006. [Google Scholar]
- World Health Organization. Guideline: Sodium Intake for Adults and Children; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- World Health Organization. Guideline: Potassium Intake for Adults and Children; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Food and Agriculture Organization of the United Nations & FHI. Minimum Dietary Diversity for Women—A Guide to Measurement; FAO: Rome, Italy, 2016; Volume 82. [Google Scholar]
- Food and Agriculture Organization of the United Nations & Government of Kenya. Kenyan Food Recipes: A Recipe Book of Common Mixed Dishes with Nutrient Values; As Prepared by Communities; FAO: Nairobi, Kenya, 2018; p. 349. [Google Scholar]
- Oba, S.; Nagata, C.; Nakamura, K.; Fujii, K.; Kawachi, T.; Takatsuka, N.; Shimizu, H. Diet Based on the Japanese Food Guide Spinning Top and Subsequent Mortality among Men and Women in a General Japanese Population. J. Am. Diet. Assoc. 2009, 109, 1540–1547. [Google Scholar] [CrossRef] [PubMed]
- Willett, W. Nutritional Epidemiology, 3rd ed.; Oxford University Press: Oxford, UK, 2013; pp. 273–301. [Google Scholar]
- Amoutzopoulos, B.; Page, P.; Roberts, C.; Roe, M.; Cade, J.; Steer, T.; Baker, R.; Hawes, T.; Galloway, C.; Yu, D.; et al. Portion size estimation in dietary assessment: A systematic review of existing tools, their strengths and limitations. Nutr. Rev. 2020, 78, 885–900. [Google Scholar] [CrossRef] [PubMed]
- Arimond, M.; Wiesmann, D.; Becquey, E.; Carriquiry, A.; Daniels, M.C.; Deitchler, M.; Fanou-Fogny, N.; Joseph, M.L.; Kennedy, G.; Martin-Prevel, Y.; et al. Simple Food Group Diversity Indicators Predict Micronutrient Adequacy of Women’s Diets in 5 Diverse, Resource-Poor Settings. J. Nutr. 2010, 140, 2059S–2069S. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ministry of Health Kenya. Overview of food fortification program. Available online: https://www.nutritionhealth.or.ke/programmes/micronutrient-deficiency-control/food-fortification/ (accessed on 30 November 2021).
- Khamila, S.; Sila, D.N.; Makokha, A. Compliance status and stability of vitamins and minerals in Fortified Maize Flour in Kenya. Sci. Afr. 2020, 8, e00384. [Google Scholar] [CrossRef]
- Savy, M.; Martin-Prével, Y.; Danel, P.; Traissac, P.; Dabiré, H.; Delpeuch, F. Are dietary diversity scores related to the socioeconomic and anthropometric status of women living in an urban area in Burkina Faso? Public Health Nutr. 2008, 11, 132–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darmon, N.; Drewnowski, A. Does social class predict diet quality? Am. J. Clin. Nutr. 2008, 87, 1107–1117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarfo, J.; Keding, G.B.; Boedecker, J.; Pawelzik, E.; Termote, C. The Impact of Local Agrobiodiversity and Food Interventions on Cost, Nutritional Adequacy, and Affordability of Women and Children’s Diet in Northern Kenya: A Modeling Exercise. Front. Nutr. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
| Food Group (1) | Definition of 1 SV (2) | Recommended SVs (3) (SVs/Day) | FP Score |
|---|---|---|---|
| General starches | 20 g carbohydrate | 6–11 | 0–10 |
| Cereals and grains | |||
| Roots and tubers | |||
| Milk products | 300 mg Ca | 2–3 | 0–10 |
| Protein-rich foods | 6 g protein | 5–7 | 0–10 |
| Plant-based foods | |||
| Animal-based foods | |||
| Vegetables | 80 g edible weight | ≧3 | 0–10 |
| Green leafy vegetables | |||
| Other vegetables | |||
| Fruits | 100 g edible weight | 2–3 | 0–10 |
| Total | 0–50 |
| FP Score Tertiles | p * | |||
|---|---|---|---|---|
| Low (n = 39) | Middle (n = 41) | High (n = 40) | ||
| FP score | 19.4 ± 2.2 | 24.4 ± 1.3 | 31.0 ± 3.1 | - |
| Age (years old) | 42.5 ± 10.2 | 41.8 ± 9.2 | 43.1 ± 9.4 | 0.830 |
| Gender (women %) | 19 (48.7) | 23 (56.1) | 22 (55.0) | 0.580 |
| Household size (person) | 4.6 ± 1.3 | 4.9 ± 1.5 | 4.5 ± 1.4 | 0.548 |
| Full-time farmer | 31 (79.5) | 32 (78.0) | 38 (95.0) | 0.059 |
| Education level | ||||
| Primary school | 14 (35.9) | 12 (29.3) | 13 (32.5) | 0.987 |
| Secondary school | 15 (38.5) | 22 (53.7) | 18 (45.0) | |
| University | 10 (25.6) | 7 (17.1) | 9 (22.5) | |
| Socioeconomic status | ||||
| Low | 15 (38.5) | 15 (36.6) | 9 (22.5) | 0.660 |
| Middle | 11 (28.2) | 13 (31.7) | 21 (52.5) | |
| High | 12 (30.8) | 13 (31.7) | 9 (22.5) | |
| BMI (kg/m2) | 25.8 ± 5.2 | 25.5 ± 5.2 | 26.7 ± 5.1 | 0.284 |
| Underweight | 2 (5.1) | 2 (4.9) | 3 (7.5) | 0.344 |
| Normal weight | 18 (46.2) | 18 (43.9) | 13 (32.5) | |
| Overweight | 12 (30.8) | 14 (34.1) | 13 (32.5) | |
| Obesity | 7 (17.9) | 7 (17.1) | 11 (27.5) | |
| Waist circumference (cm) | 92.4 ± 11.9 | 90.8 ± 16.2 | 94.8 ± 13.9 | 0.426 |
| Hip circumference (cm) | 105.8 ± 13.0 | 103.8 ± 12.3 | 105.9 ± 9.7 | 0.745 |
| W/H ratio | 0.88 ± 0.07 | 0.87 ± 0.08 | 0.89 ± 0.09 | 0.182 |
| Body fat (%) | 28.4 ± 8.0 | 28.1 ± 7.1 | 29.3 ± 8.7 | 0.641 |
| SBP(mmHg) | 129.6 ± 20.9 | 129.9 ± 13.1 | 131.1 ± 19.3 | 0.392 |
| DBP(mmHg) | 83.6 ± 11.5 | 83.9 ± 13.5 | 83.7 ± 11.2 | 0.881 |
| Hypertension | 12 (30.8) | 13 (31.7) | 16 (40.0) | 0.387 |
| Severe hypertension | 5 (12.8) | 4 (9.8) | 3 (7.5) | 0.433 |
| Step counts (steps/day) | 13,580 ± 9258 | 14,747 ± 8539 | 14,955 ± 8011 | 0.291 |
| Meal frequency (times/day) | 3.4 ± 0.9 | 3.6 ± 0.8 | 4.1 ± 1.2 | 0.010 |
| Energy and Nutrient Intakes | FP Score Tertiles | p for Trend * | ||
|---|---|---|---|---|
| Low (n = 39) | Middle (n = 41) | High (n = 40) | ||
| Energy (kcal) | 1721 ± 817 | 1793 ± 665 | 1976 ± 514 | 0.002 |
| Protein (%E) | 11.5 ± 2.8 | 11.7 ± 1.8 | 12.4 ± 2.0 | 0.006 |
| Fat (%E) | 26.5 ± 4.1 | 26.5 ± 6.3 | 27.3 ± 5.1 | 0.584 |
| Carbohydrate (%E) | 62.0 ± 4.6 | 61.8 ± 6.1 | 60.3 ± 4.9 | 0.152 |
| Protein (g) | 50.2 ± 11.1 | 52.2 ± 10.1 | 56.1 ± 9.7 | 0.001 |
| Fat (g) | 56.4 ± 8.0 | 55.7 ± 11.7 | 55.5 ± 13.6 | 0.260 |
| Carbohydrate (g) | 263.3 ± 19.3 | 260.6 ± 26.7 | 256.6 ± 28.2 | 0.361 |
| Fibre (g) | 26.7 ± 9.3 | 31.4 ± 9.2 | 33.8 ± 8.9 | <0.001 |
| Sodium (mg) | 1804 ± 757 | 1921 ± 584 | 2136 ± 871 | 0.028 |
| Potassium (mg) | 1913 ± 803 | 2146 ± 589 | 2266 ± 599 | 0.002 |
| Calcium (mg) | 641 ± 165 | 788 ± 273 | 869 ± 274 | <0.001 |
| Magnesium (mg) | 257 ± 62 | 296 ± 62 | 328 ± 64 | <0.001 |
| Phosphorous (mg) | 1415 ± 262 | 1491 ± 281 | 1556 ± 305 | 0.018 |
| Iron (mg) | 18.0 ± 8.3 | 19.6 ± 7.2 | 19.8 ± 5.3 | 0.031 |
| Zinc (mg) | 8.0 ± 2.5 | 8.3 ± 2.1 | 9.2 ± 1.9 | <0.001 |
| Selenium (µg) | 34.3 ± 17.7 | 40.5 ± 18.7 | 46.0 ± 19.7 | 0.002 |
| Vitamin A (µgRAE) | 280 ± 101 | 370 ± 191 | 426 ± 160 | <0.001 |
| Vitamin B1 (mg) | 1.09 ± 0.37 | 1.09 ± 0.28 | 0.98 ± 0.31 | 0.117 |
| Vitamin B2 (mg) | 0.93 ± 0.23 | 1.03 ± 0.37 | 0.95 ± 0.29 | 0.825 |
| Niacin (mg) | 11.4 ± 3.1 | 11.5 ± 1.7 | 11.8 ± 2.8 | 0.355 |
| Vitamin B12 (µg) | 2.2 ± 0.8 | 2.2 ± 1.0 | 2.5 ± 1.1 | 0.171 |
| Folic acid (µg) | 487 ± 204 | 529 ± 141 | 451 ± 166 | 0.227 |
| Vitamin C (mg) | 71 ± 49 | 94 ± 59 | 118 ± 62 | <0.001 |
| Food Group Intakes (g) | FP Score Tertiles | p for Trend * | ||
|---|---|---|---|---|
| Low (n = 39) | Middle (n = 41) | High (n = 40) | ||
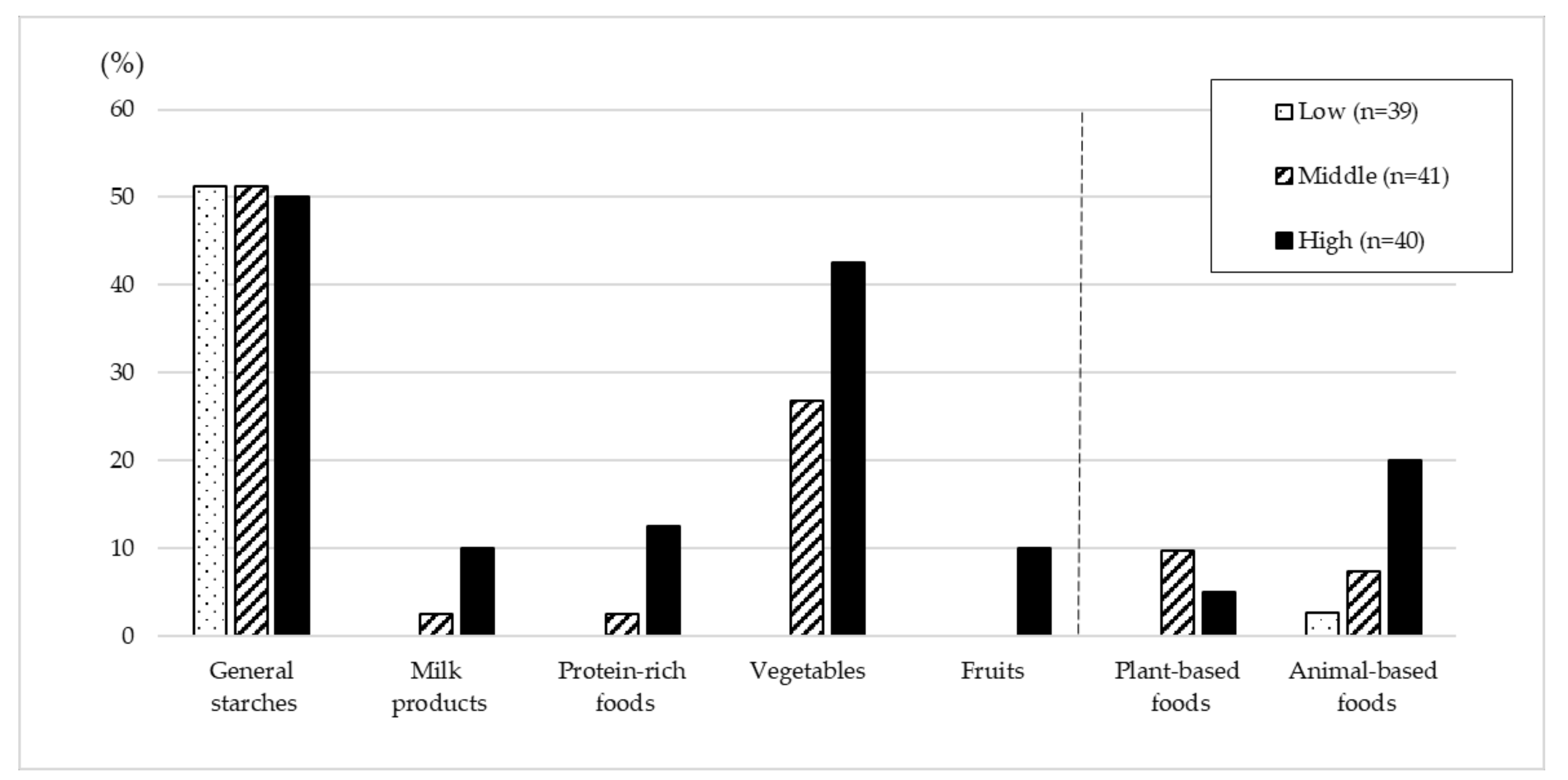
| General starches | 10.73 ± 1.81 | 10.16 ± 1.47 | 9.10 ± 1.60 | <0.001 |
| Cereal and grains | 9.28 ± 1.76 | 9.18 ± 1.56 | 8.23 ± 1.60 | 0.007 |
| Roots and tubers | 1.44 ± 2.36 | 0.98 ± 1.22 | 0.87 ± 1.41 | 0.084 |
| Milk products | 0.92 ± 0.32 | 0.91 ± 0.36 | 1.28 ± 0.49 | 0.001 |
| Protein-rich foods | 1.62 ± 1.95 | 2.08 ± 1.95 | 2.95 ± 2.03 | 0.001 |
| Plant-based foods | 0.82 ± 0.96 | 1.05 ± 1.05 | 1.19 ± 1.52 | 0.380 |
| Animal-based foods | 0.80 ± 1.77 | 1.03 ± 1.83 | 1.76 ± 1.99 | 0.022 |
| Meats | 0.68 ± 1.64 | 0.78 ± 1.79 | 1.46 ± 1.84 | 0.126 |
| Fishes | 0.12 ± 0.54 | 0.01 ± 0.03 | 0.01 ± 0.02 | 0.263 |
| Eggs | 0.06 ± 0.24 | 0.25 ± 0.61 | 0.30 ± 0.65 | 0.339 |
| Vegetables | 1.37 ± 0.91 | 2.67 ± 1.88 | 2.58 ± 1.27 | <0.001 |
| Green leafy vegetables | 0.95 ± 0.75 | 1.86 ± 1.79 | 1.97 ± 1.26 | <0.001 |
| Other vegetables | 0.42 ± 0.51 | 0.81 ± 0.73 | 0.61 ± 0.83 | 0.694 |
| Fruits | 0.05 ± 0.12 | 0.05 ± 0.17 | 0.45 ± 0.97 | 0.725 |
| Oils | 5.78 ± 5.35 | 4.63 ± 4.33 | 2.65 ± 3.75 | <0.001 |
| Sugars | 3.80 ± 1.34 | 3.66 ± 1.45 | 5.08 ± 1.98 | 0.003 |
| Criteria for Risk of Insufficient or Excess (1) | FP Score Tertiles | p for Trend * | |||
|---|---|---|---|---|---|
| Low (n = 39) | Middle (n = 41) | High (n = 40) | |||
| Energy (insufficient) | Underweight | 1 (2.6) | 2 (4.9) | 3 (7.5) | 0.316 |
| Energy (excess) | Overweight and obese | 19 (48.7) | 21 (51.2) | 24 (60.0) | 0.316 |
| Protein per kg BM (insufficient) | EAR | 21 (53.8) | 17 (41.5) | 11 (27.5) | 0.018 |
| Protein %E (insufficient) | LL of AMDR | 15 (38.5) | 5 (12.2) | 4 (10.0) | 0.002 |
| Fat %E (insufficient) | LL of AMDR | 2 (5.1) | 6 (14.6) | 3 (7.5) | 0.724 |
| Fat %E (excess) | UL of AMDR | 1 (2.6) | 6 (14.6) | 3 (7.5) | 0.437 |
| Carbohydrate %E (excess) | UL of AMDR | 11 (28.2) | 12 (29.3) | 6 (15.0) | 0.170 |
| Total fibre (insufficient) | AI | 17 (43.6) | 10 (24.4) | 8 (20.0) | 0.022 |
| Sodium (excess) | WHO recommendation | 10 (25.6) | 16 (39.0) | 22 (55.0) | 0.008 |
| Potassium (insufficient) | WHO recommendation | 36 (92.3) | 37 (90.2) | 37 (92.5) | 0.973 |
| Calcium (insufficient) | AI | 35 (89.7) | 33 (80.5) | 26 (65.0) | 0.008 |
| Magnesium (insufficient) | EAR | 29 (74.4) | 24 (58.5) | 15 (37.5) | 0.001 |
| Iron (insufficient) | EAR | 3 (7.7) | 1 (2.4) | 0 (0.0) | 0.058 |
| Zinc (insufficient) | EAR | 28 (71.8) | 21 (51.2) | 8 (20.0) | <0.001 |
| Selenium (insufficient) | EAR | 32 (82.1) | 30 (73.2) | 19 (47.5) | 0.001 |
| Vitamin A (insufficient) | EAR | 38 (97.4) | 36 (87.8) | 28 (70.0) | 0.001 |
| Vitamin B1 (insufficient) | EAR | 24 (61.5) | 23 (56.1) | 19 (47.5) | 0.211 |
| Vitamin B2 (insufficient) | EAR | 29 (74.4) | 25 (61.0) | 19 (47.5) | 0.015 |
| Niacin (insufficient) | EAR | 17 (43.6) | 18 (43.9) | 18 (45.0) | 0.900 |
| Vitamin B12 (insufficient) | EAR | 22 (56.4) | 24 (58.5) | 10 (25.0) | 0.005 |
| Folic acid (insufficient) | EAR | 23 (59.0) | 13 (31.7) | 14 (35.0) | 0.032 |
| Vitamin C (insufficient) | EAR | 22 (56.4) | 19 (46.3) | 10 (25.0) | 0.005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kishino, M.; Hida, A.; Hara, K.; Mungai, D.N.; Opiyo, R.O.; Matsuda, H.; Tada, Y.; Ishikawa-Takata, K.; Irie, K.; Morimoto, Y. High Adherence to the Food Pyramid’s Recommendations Avoids the Risk of Insufficient Nutrient Intake among Farmers in Peri-Urban Kenya. Nutrients 2021, 13, 4470. https://doi.org/10.3390/nu13124470
Kishino M, Hida A, Hara K, Mungai DN, Opiyo RO, Matsuda H, Tada Y, Ishikawa-Takata K, Irie K, Morimoto Y. High Adherence to the Food Pyramid’s Recommendations Avoids the Risk of Insufficient Nutrient Intake among Farmers in Peri-Urban Kenya. Nutrients. 2021; 13(12):4470. https://doi.org/10.3390/nu13124470
Chicago/Turabian StyleKishino, Madoka, Azumi Hida, Kenta Hara, David Nguatha Mungai, Rose Okoyo Opiyo, Hirotaka Matsuda, Yuki Tada, Kazuko Ishikawa-Takata, Kenji Irie, and Yasuyuki Morimoto. 2021. "High Adherence to the Food Pyramid’s Recommendations Avoids the Risk of Insufficient Nutrient Intake among Farmers in Peri-Urban Kenya" Nutrients 13, no. 12: 4470. https://doi.org/10.3390/nu13124470
APA StyleKishino, M., Hida, A., Hara, K., Mungai, D. N., Opiyo, R. O., Matsuda, H., Tada, Y., Ishikawa-Takata, K., Irie, K., & Morimoto, Y. (2021). High Adherence to the Food Pyramid’s Recommendations Avoids the Risk of Insufficient Nutrient Intake among Farmers in Peri-Urban Kenya. Nutrients, 13(12), 4470. https://doi.org/10.3390/nu13124470







