Vitamin D Dosing: Basic Principles and a Brief Algorithm (2021 Update)
Abstract
1. Introduction
2. Is There a Need for New Guidelines/Algorithms?
- Despite the available evidence of vitamin’s D important role for the human organism, including extra-skeletal health and the high prevalence of low Vit. D status in different regions of the world [17,18,19,20,21], many countries still do not have national, up-to-date, approved Vit. D guidelines. The same applies also to Lithuania, which has only the Rickets’ diagnosis and treatment guidelines approved in 2015. Moreover, in most countries, the potential beneficial role of Vit. D for COVID-19 prevention and treatment (i.e., Vit. D as an adjuvant) is still not accepted; consequently, no specialized relevant recommendations are developed. Paradoxically, it is the COVID-19 pandemic that inspired the author of the present article to start developing national Vit. D guidelines for Lithuania. Hopefully, the basic principles of those guidelines presented in the current paper could be an additional source for more specialized future recommendations both for Lithuania and for other countries.
- Traditionally, any well-prepared Vit. D guidelines should reflect clinical practice and therefore must include the following domains: definition of risk groups for low vitamin D; principles of evaluation of Vit. D status by using laboratory measurements; and Vit. D dosing for prevention and treatment. However, the COVID-19 pandemic brought some challenges that aggravated our routine clinical practice. Firstly, due to reduced accessibility to health care facilities, mandatory isolation of some patients (due to diagnosed COVID-19 disease or due to close contact with a confirmed COVID-19 case), or a patient’s fear of getting SARS-CoV-2 during visits to a clinic or laboratory, it is not possible to perform the measurements of serum 25OH-D levels at the desired time. Therefore, the recent Vit. D status of many outpatients could remain unknown. Secondly, with the absence of data on recent 25OH-D level measurements, it might be difficult for physicians to make decisions regarding Vit. D dosing, particularly for low Vit. D risk group patients. We need an extended list of risk factors that might suggest the clinician to presume that certain patients could be put into a Vit. D risk group and, consequently, to suggest him/her higher Vit. D doses for supplementation. Finally, even disregarding the potentially beneficial direct Vit. D role on COVID-19 prevention and treatment, it is wise to remember that the problem of low Vit. D in society has not disappeared during the pandemic. Moreover, some people, due to various reasons during lockdowns, may have even higher risk to newly develop Vit. D insufficiency, leading to poorer skeletal and extra-skeletal health [10]. Patients having low 25OH-D levels might be considered as high-risk group for getting severe illness from COVID-19 [22].
- In older Vit. D guidelines, there is almost no talk about the causes that could result in failure to achieve the desired levels of 25OH-D by supplementing Vit. D, and the suggested actions for physicians. In the present article, the author also tried, in part, to fulfil those gaps.
- It is the Vit. D supplementation that modern guidelines should be mostly oriented to. Production of vitamin D3 in the skin is not a reliable source for repletion of low Vit. D status. Firstly, human skin is able to produce only limited amount of vitamin D3 that can enter the circulation [23,24]. Secondly, it is difficult to predict the effect of solar radiation in regard to vitamin D3 production and its influence on 25OH-D levels, since a large number of factors might affect vitamin D3 synthesis in the skin, e.g., skin type, patient age, time of the day, altitude, etc. [23,25,26]. Finally, in some countries, e.g., Lithuania, that are located at the middle latitudes, the intensity of solar radiation decreases significantly during the cold season, and the synthesis of vitamin D3 in the skin is almost absent during the period from October till March [27,28]. Food, unless fortified with Vit. D, usually cannot serve as a valuable source of this vitamin, too [27,29,30]. Therefore, this paper does not discuss recommendations on exposure to sunlight or influence of certain types of food for prevention or treatment of low Vit. D status.
3. Risk Factors for Low Vitamin D Status
4. Evaluation of Vitamin D Status
5. Vitamin D Dosing Principles
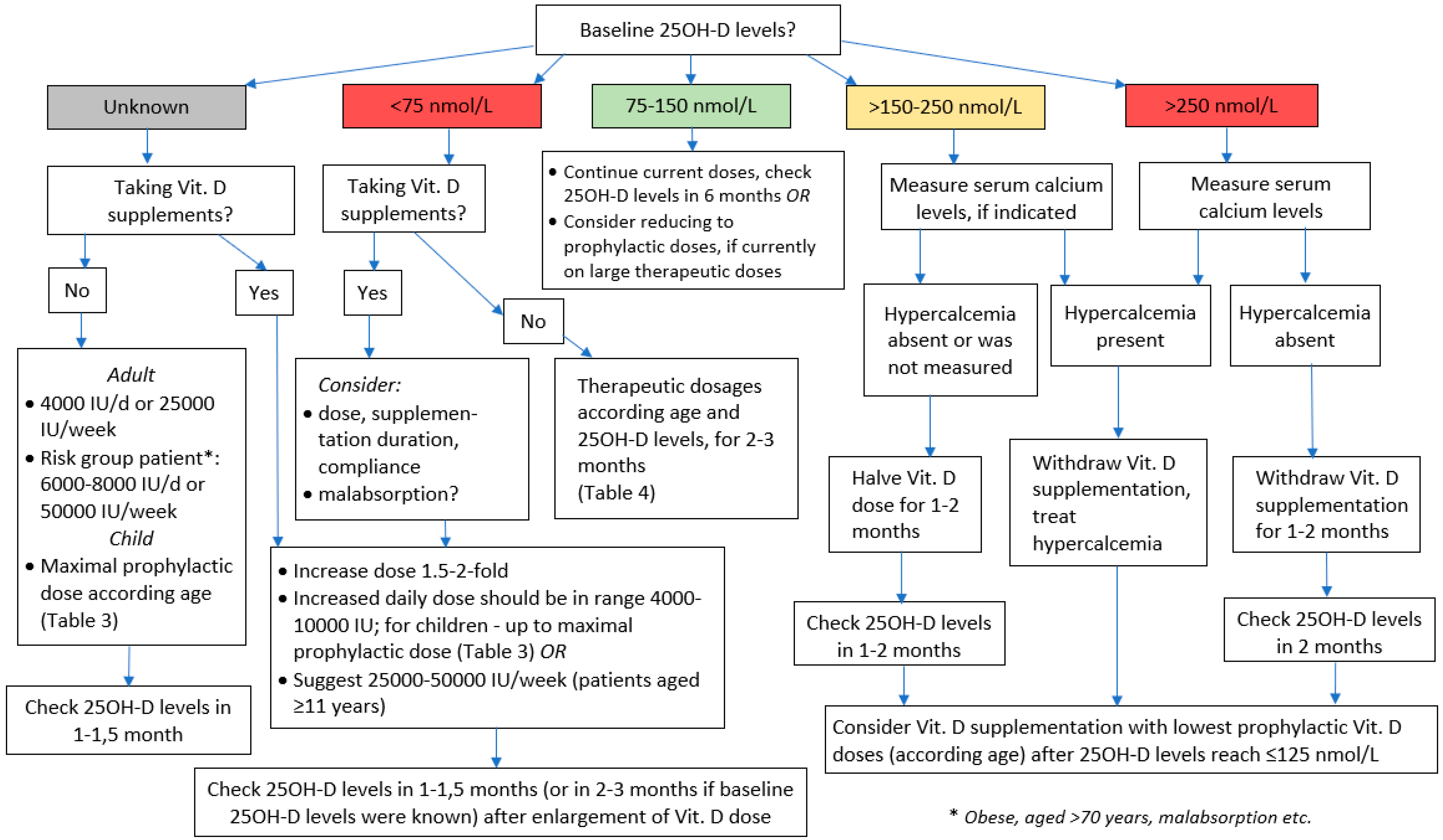
6. A Brief Algorithm for Vitamin D Dosing
7. Dealing with Failure to Increase 25-Hydroxy-Vitamin D Levels
- The dose that was prescribed and the duration of supplementation. If the Vit. D dose could have been too small, it can be increased two-fold, and the next check of 25OH-D levels can be performed at 1.5–2 months after dose correction.
- The compliance. Some patients prefer not to take large Vit. D doses even by physician prescription and, in fact, consume only small doses, for the fear of Vit. D overdose.
- Possibility of unreported chronic diseases or use of certain drugs that could impair Vit. D metabolism. Some patients might be candidates to be examined for possible malabsorption syndrome, particularly in cases when 25OH-D levels did not increase significantly even after the supplementation with doubled dose. In some cases, e.g., for those with celiac disease, severe liver disease, or after bariatric surgery, calcidiol might be suggested, since it has better intestinal absorption than Vit. D and appears to be two to three times more effective in increasing serum 25OH-D levels than vitamin D3 [20], and this feature of calcidiol might be very important also in early stages of the COVID-19 disease, when low serum 25OH-D levels need to be increased as soon as possible [8,61].
- Adequacy of calcium (Ca) and/or magnesium (Mg) intake. During the treatment of low Vit. D, supplementation with Mg (daily dose in the range 250–500 mg/d) is recommended, since Mg acts as a cofactor in many enzymes involved in Vit. D metabolism [44]. In addition, it is worth understanding that long-term decreased intake of Ca with food can, in turn, aggravate low Vit. D status because of compensatory hyperparathyroidism that increases the production of calcitriol in the kidney from 25OH-D, and this consequently contributes to diminishing serum 25OH-D levels. Therefore, with adequate Ca intake (including Ca from supplementation, if necessary), a better response to Vit. D preparations can be expected [39,62]. The recommended daily intake of Ca for adults is ~1000–1200 mg; more data about Ca inadequacy can be found elsewhere [63].
8. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Over 200 Scientists & Doctors Call for Increased Vitamin D Use to Combat COVID-19. Available online: https://vitamind4all.org/letter.html (accessed on 2 February 2021).
- Holick, M.F. Vitamin D: Extraskeletal health. Endocrinol. Metab. Clin. Am. 2010, 39, 381–400. [Google Scholar] [CrossRef]
- Entrenas Castillo, M.; Entrenas Costa, L.M.; Vaquero Barrios, J.M.; Alcala Diaz, J.F.; Lopez Miranda, J.; Bouillon, R.; Quesada Gomez, J.M. Effect of calcifediol treatment and best available therapy versus best available therapy on intensive care unit admission and mortality among patients hospitalized for COVID-19: A pilot randomized clinical study. J. Steroid. Biochem. Mol. Biol. 2020, 203, 105751. [Google Scholar] [CrossRef]
- Rastogi, A.; Bhansali, A.; Khare, N.; Suri, V.; Yaddanapudi, N.; Sachdeva, N.; Puri, G.D.; Malhotra, P. Short term, high-dose vitamin D supplementation for COVID-19 disease: A randomised, placebo-controlled, study (SHADE study). Postgrad. Med. J. 2020. [Google Scholar] [CrossRef]
- Tan, C.W.; Ho, L.P.; Kalimuddin, S.; Cherng, B.P.Z.; Teh, Y.E.; Thien, S.Y.; Wong, H.M.; Tern, P.J.W.; Chandran, M.; Chay, J.W.M.; et al. Cohort study to evaluate the effect of vitamin D, magnesium, and vitamin B12 in combination on progression to severe outcomes in older patients with coronavirus (COVID-19). Nutrition 2020, 79, 111017. [Google Scholar] [CrossRef]
- Annweiler, G.; Corvaisier, M.; Gautier, J.; Dubee, V.; Legrand, E.; Sacco, G.; Annweiler, C. Vitamin D Supplementation Associated to Better Survival in Hospitalized Frail Elderly COVID-19 Patients: The GERIA-COVID Quasi-Experimental Study. Nutrients 2020, 12, 3377. [Google Scholar] [CrossRef] [PubMed]
- Annweiler, C.; Hanotte, B.; Grandin de l’Eprevier, C.; Sabatier, J.M.; Lafaie, L.; Celarier, T. Vitamin D and survival in COVID-19 patients: A quasi-experimental study. J. Steroid. Biochem. Mol. Biol. 2020, 204, 105771. [Google Scholar] [CrossRef]
- Grant, W.B.; Lordan, R. Vitamin D for COVID-19 on trial: An update on prevention and therapeutic application. Endocr. Pract. 2021, 27, 1266–1268. [Google Scholar] [CrossRef]
- Yisak, H.; Ewunetei, A.; Kefale, B.; Mamuye, M.; Teshome, F.; Ambaw, B.; Yideg Yitbarek, G. Effects of Vitamin D on COVID-19 Infection and Prognosis: A Systematic Review. Risk Manag. Health Policy 2021, 14, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Bleizgys, A. Vitamin D and COVID-19: It is time to act. Int. J. Clin. Pract. 2020, 75, e13748. [Google Scholar] [CrossRef]
- Chiu, S.K.; Tsai, K.W.; Wu, C.C.; Zheng, C.M.; Yang, C.H.; Hu, W.C.; Hou, Y.C.; Lu, K.C.; Chao, Y.C. Putative Role of Vitamin D for COVID-19 Vaccination. Int. J. Mol. Sci. 2021, 22, 8988. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M.; Endocrine, S. Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Karczmarewicz, E.; Bayer, M.; Carter, G.; Chlebna-Sokol, D.; Czech-Kowalska, J.; Debski, R.; Decsi, T.; Dobrzanska, A.; Franek, E.; et al. Practical guidelines for the supplementation of vitamin D and the treatment of deficits in Central Europe—Recommended vitamin D intakes in the general population and groups at risk of vitamin D deficiency. Endokrynol. Pol. 2013, 64, 319–327. [Google Scholar] [CrossRef] [PubMed]
- Pludowski, P.; Holick, M.F.; Grant, W.B.; Konstantynowicz, J.; Mascarenhas, M.R.; Haq, A.; Povoroznyuk, V.; Balatska, N.; Barbosa, A.P.; Karonova, T.; et al. Vitamin D supplementation guidelines. J. Steroid Biochem. Mol. Biol. 2018, 175, 125–135. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 report on dietary reference intakes for calcium and vitamin D from the Institute of Medicine: What clinicians need to know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef]
- Kimball, S.M.; Holick, M.F. Official recommendations for vitamin D through the life stages in developed countries. Eur. J. Clin. Nutr. 2020, 74, 1514–1518. [Google Scholar] [CrossRef] [PubMed]
- van Schoor, N.M.; Lips, P. Worldwide vitamin D status. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 671–680. [Google Scholar] [CrossRef]
- Lips, P. Vitamin D status and nutrition in Europe and Asia. J. Steroid Biochem. Mol. Biol. 2007, 103, 620–625. [Google Scholar] [CrossRef]
- Pludowski, P.; Grant, W.B.; Bhattoa, H.P.; Bayer, M.; Povoroznyuk, V.; Rudenka, E.; Ramanau, H.; Varbiro, S.; Rudenka, A.; Karczmarewicz, E.; et al. Vitamin D status in central europe. Int. J. Endocrinol. 2014, 2014, 589587. [Google Scholar] [CrossRef]
- Lips, P.; Cashman, K.D.; Lamberg-Allardt, C.; Bischoff-Ferrari, H.A.; Obermayer-Pietsch, B.; Bianchi, M.L.; Stepan, J.; El-Hajj Fuleihan, G.; Bouillon, R. Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: A position statement of the European Calcified Tissue Society. Eur. J. Endocrinol. Eur. Fed. Endocr. Soc. 2019, 180, P23–P54. [Google Scholar] [CrossRef]
- Kara, M.; Ekiz, T.; Ricci, V.; Kara, O.; Chang, K.V.; Ozcakar, L. “Scientific Strabismus” or two related pandemics: Coronavirus disease and vitamin D deficiency. Br. J. Nutr. 2020, 124, 736–741. [Google Scholar] [CrossRef]
- Ebadi, M.; Montano-Loza, A.J. Perspective: Improving vitamin D status in the management of COVID-19. Eur. J. Clin. Nutr. 2020, 74, 856–859. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Rusinska, A.; Pludowski, P.; Walczak, M.; Borszewska-Kornacka, M.K.; Bossowski, A.; Chlebna-Sokol, D.; Czech-Kowalska, J.; Dobrzanska, A.; Franek, E.; Helwich, E.; et al. Vitamin D Supplementation Guidelines for General Population and Groups at Risk of Vitamin D Deficiency in Poland-Recommendations of the Polish Society of Pediatric Endocrinology and Diabetes and the Expert Panel with Participation of National Specialist Consultants and Representatives of Scientific Societies-2018 Update. Front. Endocrinol. 2018, 9, 246. [Google Scholar] [CrossRef]
- Giustina, A.; Adler, R.A.; Binkley, N.; Bollerslev, J.; Bouillon, R.; Dawson-Hughes, B.; Ebeling, P.R.; Feldman, D.; Formenti, A.M.; Lazaretti-Castro, M.; et al. Consensus statement from 2(nd) International Conference on Controversies in Vitamin D. Rev. Endocr. Metab. Disord. 2020, 21, 89–116. [Google Scholar] [CrossRef]
- Gueli, N.; Verrusio, W.; Linguanti, A.; Di Maio, F.; Martinez, A.; Marigliano, B.; Cacciafesta, M. Vitamin D: Drug of the future. A new therapeutic approach. Arch. Gerontol. Geriatr. 2012, 54, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Foss, Y.J. Vitamin D deficiency is the cause of common obesity. Med. Hypotheses 2009, 72, 314–321. [Google Scholar] [CrossRef]
- Diffey, B.L. Is casual exposure to summer sunlight effective at maintaining adequate vitamin D status? Photodermatol. Photoimmunol. Photomed. 2010, 26, 172–176. [Google Scholar] [CrossRef]
- Grant, W.B.; Boucher, B.J. Requirements for Vitamin D across the life span. Biol. Res. Nurs. 2011, 13, 120–133. [Google Scholar] [CrossRef]
- Strucinska, M.; Rowicka, G.; Dylag, H.; Riahi, A.; Bzikowska, A. Dietary intake of vitamin D in obese children aged 1–3 years. Rocz. Panstw. Zakl. Hig. 2015, 66, 353–360. [Google Scholar]
- Guessous, I. Role of Vitamin D deficiency in extraskeletal complications: Predictor of health outcome or marker of health status? Biomed. Res. Int. 2015, 2015, 563403. [Google Scholar] [CrossRef] [PubMed]
- Black, L.J.; Anderson, D.; Clarke, M.W.; Ponsonby, A.L.; Lucas, R.M.; Ausimmune Investigator Group. Analytical Bias in the Measurement of Serum 25-Hydroxyvitamin D Concentrations Impairs Assessment of Vitamin D Status in Clinical and Research Settings. PLoS ONE 2015, 10, e0135478. [Google Scholar] [CrossRef] [PubMed]
- Madsen, K.H.; Rasmussen, L.B.; Mejborn, H.; Andersen, E.W.; Molgaard, C.; Nissen, J.; Tetens, I.; Andersen, R. Vitamin D status and its determinants in children and adults among families in late summer in Denmark. Br. J. Nutr. 2014, 112, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Holick, M.F. The vitamin D deficiency pandemic: Approaches for diagnosis, treatment and prevention. Rev. Endocr. Metab. Disord. 2017, 18, 153–165. [Google Scholar] [CrossRef]
- Collins, A. Practice implications for preventing population vulnerability related to vitamin D status. J. Am. Assoc. Nurse Pract. 2013, 25, 109–118. [Google Scholar] [CrossRef]
- LeFevre, M.L. Screening for vitamin D deficiency in adults: U.S. Preventive Services Task Force recommendation statement. Ann. Intern. Med. 2015, 162, 133–140. [Google Scholar] [CrossRef]
- Cooper, I.D.; Crofts, C.A.P.; DiNicolantonio, J.J.; Malhotra, A.; Elliott, B.; Kyriakidou, Y.; Brookler, K.H. Relationships between hyperinsulinaemia, magnesium, vitamin D, thrombosis and COVID-19: Rationale for clinical management. Open Heart 2020, 7, e001356. [Google Scholar] [CrossRef]
- Mazahery, H.; von Hurst, P.R. Factors Affecting 25-Hydroxyvitamin D Concentration in Response to Vitamin D Supplementation. Nutrients 2015, 7, 5111–5142. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Zittermann, A.; Trummer, C.; Theiler-Schwetz, V.; Lerchbaum, E.; Keppel, M.H.; Grubler, M.R.; Marz, W.; Pandis, M. Vitamin D testing and treatment: A narrative review of current evidence. Endocr. Connect. 2019, 8, R27–R43. [Google Scholar] [CrossRef]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Kostenberger, M.; Tmava Berisha, A.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D deficiency 2.0: An update on the current status worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef]
- Bonham, M.P.; Lamberg-Allardt, C. Vitamin D in public health nutrition. Public Health Nutr. 2014, 17, 717–720. [Google Scholar] [CrossRef] [PubMed]
- Boyages, S.C. Vitamin D testing: New targeted guidelines stem the overtesting tide. Med. J. Aust. 2016, 204, 18. [Google Scholar] [CrossRef][Green Version]
- Grant, W.B.; Lahore, H.; McDonnell, S.L.; Baggerly, C.A.; French, C.B.; Aliano, J.L.; Bhattoa, H.P. Evidence that Vitamin D Supplementation Could Reduce Risk of Influenza and COVID-19 Infections and Deaths. Nutrients 2020, 12, 988. [Google Scholar] [CrossRef] [PubMed]
- Lopez, A.G.; Kerlan, V.; Desailloud, R. Non-classical effects of vitamin D: Non-bone effects of vitamin D. Ann. Endocrinol. 2021, 82, 43–51. [Google Scholar] [CrossRef]
- American Geriatrics Society Workgroup on Vitamin D Supplementation for Older Adults. Recommendations abstracted from the American Geriatrics Society Consensus Statement on vitamin D for Prevention of Falls and Their Consequences. J. Am. Geriatr. Soc. 2014, 62, 147–152. [Google Scholar] [CrossRef]
- Wimalawansa, S.J. Global epidemic of coronavirus—COVID-19: What can we do to minimize risks. Eur. J. Biomed. Pharm. Sci. 2020, 7, 432–438. [Google Scholar]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
- Grober, U.; Holick, M.F. The coronavirus disease (COVID-19)—A supportive approach with selected micronutrients. Int. J. Vitam. Nutr. Res. 2021, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Shirvani, A.; Holick, M.F. Vitamin D and Its Potential Benefit for the COVID-19 Pandemic. Endocr. Pract. 2021, 27, 484–493. [Google Scholar] [CrossRef]
- Rachito Diagnostika ir Gydymas (Protokolas). Available online: https://sam.lrv.lt/uploads/sam/documents/files/Veiklos_sritys/Programos_ir_projektai/Asmens_sveikatos_prieziuros_kokybes_grinimas/Vaiku%20ligu%20protokolai%20(papildomi%2023).zip (accessed on 7 February 2021).
- Munns, C.F.; Shaw, N.; Kiely, M.; Specker, B.L.; Thacher, T.D.; Ozono, K.; Michigami, T.; Tiosano, D.; Mughal, M.Z.; Makitie, O.; et al. Global Consensus Recommendations on Prevention and Management of Nutritional Rickets. J. Clin. Endocrinol. Metab. 2016, 101, 394–415. [Google Scholar] [CrossRef]
- Misra, M. Vitamin D Insufficiency and Deficiency in Children and Adolescents. 2020. Available online: https://www.uptodate.cn/contents/zh-Hans/vitamin-d-insufficiency-and-deficiency-in-children-and-adolescents?search (accessed on 21 November 2021).
- Carpenter, T. Etiology and Treatment of Calcipenic Rickets in Children. 2020. Available online: https://www.uptodate.cn/contents/zh-Hans/etiology-and-treatment-of-calcipenic-rickets-in-children?search (accessed on 21 November 2021).
- Dawson-Hughes, B. Vitamin D Deficiency in Adults: Definition, Clinical Manifestations, and Treatment. 2020. Available online: https://www.uptodate.cn/contents/zh-Hans/vitamin-d-deficiency-in-adults-definition-clinical-manifestations-and-treatment?search (accessed on 21 November 2021).
- Rhodes, J.M.; Subramanian, S.; Laird, E.; Griffin, G.; Kenny, R.A. Perspective: Vitamin D deficiency and COVID-19 severity—Plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis. J. Intern. Med. 2021, 289, 97–115. [Google Scholar] [CrossRef] [PubMed]
- Ferder, L.; Martin Gimenez, V.M.; Inserra, F.; Tajer, C.; Antonietti, L.; Mariani, J.; Manucha, W. Vitamin D Supplementation as a Rational Pharmacological Approach in the Covid-19 Pandemic. Am. J. Physiol. Lung Cell. Mol. Physiol. 2020, 319, L941–L948. [Google Scholar] [CrossRef]
- Marcinowska-Suchowierska, E.; Kupisz-Urbanska, M.; Lukaszkiewicz, J.; Pludowski, P.; Jones, G. Vitamin D Toxicity-A Clinical Perspective. Front. Endocrinol. 2018, 9, 550. [Google Scholar] [CrossRef] [PubMed]
- Girdžiūtė, M. Hipokalcemija ir hiperkalcemija: Diagnostikos ir gydymo aspektai. Internistas 2017, 2, 10–13. [Google Scholar]
- Ali, N. Role of vitamin D in preventing of COVID-19 infection, progression and severity. J. Infect. Public Health 2020, 13, 1373–1380. [Google Scholar] [CrossRef]
- Maghbooli, Z.; Sahraian, M.A.; Jamalimoghadamsiahkali, S.; Asadi, A.; Zarei, A.; Zendehdel, A.; Varzandi, T.; Mohammadnabi, S.; Alijani, N.; Karimi, M.; et al. Treatment With 25-Hydroxyvitamin D3 (Calcifediol) Is Associated with a Reduction in the Blood Neutrophil-to-Lymphocyte Ratio Marker of Disease Severity in Hospitalized Patients With COVID-19: A Pilot Multicenter, Randomized, Placebo-Controlled, Double-Blinded Clinical Trial. Endocr. Pract. 2021, 27, 1242–1251. [Google Scholar] [CrossRef]
- Christakos, S.; Li, S.; De La Cruz, J.; Bikle, D.D. New developments in our understanding of vitamin metabolism, action and treatment. Metabolism 2019, 98, 112–120. [Google Scholar] [CrossRef]
- Calcium. Fact Sheet for Health Professionals. Available online: https://ods.od.nih.gov/factsheets/Calcium-HealthProfessional (accessed on 21 November 2021).
| Groups of Risk Factors | Examples: Diseases, Conditions, Lifestyle Features |
|---|---|
| Musculoskeletal disorders | Rickets, osteoporosis, osteopenia, “bone pains”, muscle pain, myopathy, myodystrophy, recurrent (“low energy”) bone fractures, recurrent falls, bone deformities |
| Endocrine and metabolic diseases/conditions | Diabetes mellitus (type I and II), metabolic syndrome, obesity, overweight, hypo- and hyperparathyroidism, hypo- and hyperthyroidism, hypocalcemia, calciuria, phosphatemia, hypo- and hyperphosphatasia, phosphaturia, dyslipidemias |
| Increased demand for physiological reasons | Childhood, adolescence, pregnancy, breastfeeding |
| Malabsorption syndromes | Pancreatic exocrine insufficiency (old age, pancreatitis, type II diabetes, etc.), inflammatory bowel disease (Crohn’s disease, ulcerative colitis), cystic fibrosis, lactose intolerance, celiac disease, bariatric surgery |
| Diseases of the liver and bile ducts | Hepatic insufficiency, cirrhosis of the liver, cholestasis, hepatosteatosis |
| Kidney diseases | Renal insufficiency, chronic kidney disease (especially stages III–V), nephrotic syndrome |
| Respiratory diseases | Bronchial asthma, chronic obstructive pulmonary disease |
| Infectious diseases | Tuberculosis, recurrent respiratory infections |
| Systemic connective tissue diseases | Rheumatoid arthritis, systemic lupus erythematosus, dermatomyositis, fibromyalgia |
| Skin diseases | Atopic dermatitis, psoriasis |
| Diseases of the nervous system | Multiple sclerosis, Parkinson’s disease, dementia, cerebral palsy, autism |
| Decreased production of vitamin D3 in the skin | Older age (especially >70 years) Active protection against sun exposure (sunscreens, etc.) Cultural features (usual full-body clothing) Rare outdoor activities (work and leisure predominantly indoors; living in a care home) Increased air pollution (living in a city) Winter season (at medium latitudes) Dark-skinned (especially Africans) |
| Nutritional features | Veganism and other types of vegetarianism Allergy to cow’s milk Low-fat diet Insufficient magnesium intake Insufficient calcium intake |
| Long-term use of drugs | Antiepileptic drugs (e.g., valproate, phenytoin); antiretroviral drugs; glucocorticoids; systemic antifungal drugs; rifampin; bile acid sequestrants (cholestyramine); lipase inhibitors (orlistat) |
| Malignant neoplasms | Colon cancer, lymphatic system and blood cancers, breast cancer, ovarian cancer, prostate cancer |
| Granulomatous diseases | Sarcoidosis, histoplasmosis, coccidiomycosis, berylliosis |
| Mental illnesses | Depression, schizophrenia, anorexia nervosa |
| Cardiovascular diseases | Arterial hypertension, ischemic heart disease, heart failure |
| Others | Chronic fatigue syndrome Inpatient treatment (especially in the resuscitation and intensive care unit) Awaiting organ transplantation and post-transplant |
| Category | 25OH-D Levels, nmol/L |
|---|---|
| Severe deficiency | <25 |
| Moderate deficiency | 25–<50 |
| Insufficiency | 50–<75 |
| Sufficiency | 75–<100 |
| Optimal levels (optimal levels in tissues/cells) | 100–<150 |
| Increased levels | 150–<250 |
| Overdose | ≥250 |
| Intoxication * | ≥375 |
| Patient Age | Recommended Daily Dose (IU/d) | Recommended Intermittent Dose | Upper Tolerable Daily Dose (IU) |
|---|---|---|---|
| Infants < 6 months | 400–600 | – | 1000 |
| Infants 6–<12 months | 600–800 | – | 1000 |
| Children 1–10 yrs. | 600–1000 | – | 2000 |
| Teens 11–<18 yrs. | 800–2000 | 25,000 IU in 5–2 weeks | 4000 |
| Adults 18–<75 yrs. | 1000–2000 | 25,000 IU in 4–2 weeks | 4000 |
| Adults ≥ 75 yrs. | 2000–4000 | 25,000 IU in 2–1 weeks | 4000 |
| Patient Age | Recommended Daily Dose and Duration | Recommended Intermittent Dose and Duration |
|---|---|---|
| 25OH-D Levels < 25 nmol/L | ||
| Infants < 1 month | 1000 IU/d 3 months | – |
| Infants 1–<12 months | 2000 IU/d 3 months | – |
| Children 1–<11 yrs. | 3000–6000 IU/d 3 months | – |
| Children 11–<18 yrs. | 6000 IU/d 3 months | 50,000 IU/week 1.5–2 months |
| Adults | 6000 IU/d 3 months | 50,000 IU/week 2 months |
| 25OH-D Levels 25–<75 nmol/L | ||
| Infants < 1 month |
| – |
| Children 1–10 yrs. | – | |
| Children 11–<18 yrs. | 25,000 IU/week 2 months | |
| Adults |
| 50,000 IU/week 2 months |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bleizgys, A. Vitamin D Dosing: Basic Principles and a Brief Algorithm (2021 Update). Nutrients 2021, 13, 4415. https://doi.org/10.3390/nu13124415
Bleizgys A. Vitamin D Dosing: Basic Principles and a Brief Algorithm (2021 Update). Nutrients. 2021; 13(12):4415. https://doi.org/10.3390/nu13124415
Chicago/Turabian StyleBleizgys, Andrius. 2021. "Vitamin D Dosing: Basic Principles and a Brief Algorithm (2021 Update)" Nutrients 13, no. 12: 4415. https://doi.org/10.3390/nu13124415
APA StyleBleizgys, A. (2021). Vitamin D Dosing: Basic Principles and a Brief Algorithm (2021 Update). Nutrients, 13(12), 4415. https://doi.org/10.3390/nu13124415





