Insufficient Vitamin C Levels among Adults in the United States: Results from the NHANES Surveys, 2003–2006
Abstract
:1. Introduction
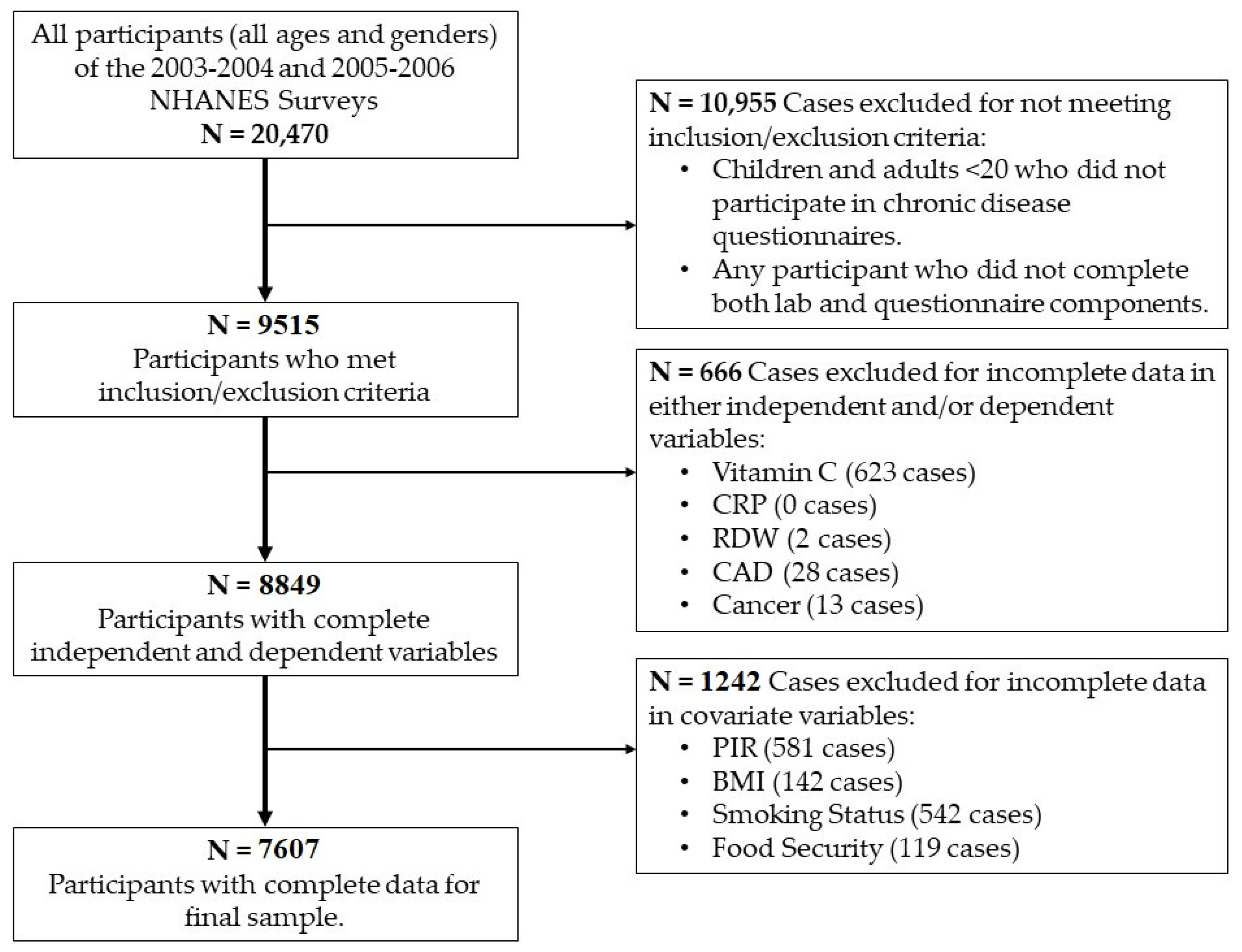
2. Materials and Methods
3. Results
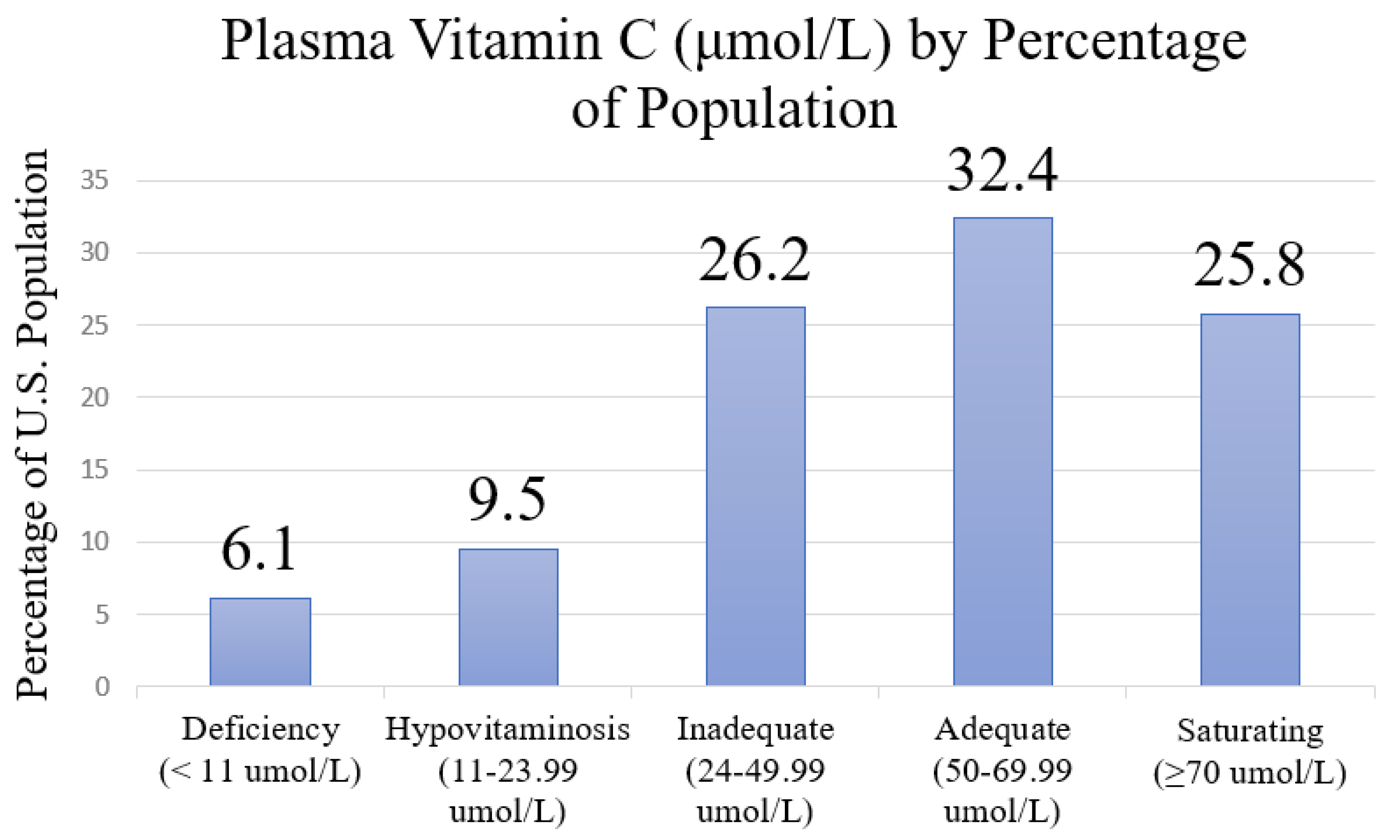
3.1. Prevalence of Vitamin C Levels
3.2. Associations between Sample Characteristics and Vitamin C Levels
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Beaulac, J.K.; Cummins, S.E. A systematic review of food deserts, 1966–2007. Prev. Chronic Dis. 2009, 6, 105. [Google Scholar]
- Carr, A.C.; Frei, B. Toward a new recommended dietary allowance for vitamin C based on antioxidant and health effects in humans. Am. J. Clin. Nutr. 1999, 69, 1086–1107. [Google Scholar] [CrossRef] [Green Version]
- Ver Ploeg, M.; Breneman, V.; Farrigan, T.; Hamrick, K.; Hopkins; Kaufman, P.; Lin, B.; Nord, M.; Smith, T.A.; Williams, R.; et al. Access to Affordable and Nutritious Food-Measuring and Under-Standing Food Deserts and Their Consequences: Report to Congress; United States Department of Agriculture: Washington, DC, USA, 2009; p. 160. [Google Scholar]
- Carr, A.C.; Rowe, S. Factors Affecting Vitamin C Status and Prevalence of Deficiency: A Global Health Perspective. Nutrients 2020, 12, 1963. [Google Scholar] [CrossRef]
- Rowe, S.; Carr, A.C. Global Vitamin C Status and Prevalence of Deficiency: A Cause for Concern? Nutrients 2020, 12, 2008. [Google Scholar] [CrossRef]
- Khan, R.M.; Iqbal, M.P. Deficiency of vitamin C in South Asia. Pak. J. Med. Sci. 2006, 22, 347. [Google Scholar]
- Canoy, D.; Wareham, N.; Welch, A.; Bingham, S.; Luben, R.; Day, N.; Khaw, K.-T. Plasma ascorbic acid concentrations and fat distribution in 19 068 British men and women in the European Prospective Investigation into Cancer and Nutrition Norfolk cohort study. Am. J. Clin. Nutr. 2005, 82, 1203–1209. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carr, A.C. A new clinical trial to test high-dose vitamin C in patients with COVID-19. Crit. Care 2020, 24, 1–2. [Google Scholar] [CrossRef] [Green Version]
- Hemila, H.; Chalker, E. Vitamin C as a possible therapy for COVID-19. Infect. Chemother. 2020, 52, 222–223. [Google Scholar] [CrossRef] [PubMed]
- Carr, A.C.; Rowe, S. The Emerging Role of Vitamin C in the Prevention and Treatment of COVID-19. Nutrients 2020, 12, 3286. [Google Scholar] [CrossRef]
- Carr, A.C.; Pullar, J.M.; Bozonet, S.M.; Vissers, M.C. Marginal ascorbate status (hypovitaminosis C) results in an attenuated response to vitamin C supplementation. Nutrients 2016, 8, 341. [Google Scholar] [CrossRef] [Green Version]
- Frei, B.; Birlouez-Aragon, I.; Lykkesfeldt, J. Authors’ perspective: What is the optimum intake of vitamin C in humans? Crit. Rev. Food Sci. Nutr. 2012, 52, 815–829. [Google Scholar] [CrossRef]
- Bengmark, S. Gut microbiota, immune development and function. Pharmacol. Res. 2013, 69, 87–113. [Google Scholar] [CrossRef] [PubMed]
- Elste, V.; Troesch, B.; Eggersdorfer, M.; Weber, P. Emerging evidence on neutrophil motility supporting its usefulness to define vitamin C intake requirements. Nutrients 2017, 9, 503. [Google Scholar] [CrossRef] [Green Version]
- Gorkom, G.W.; Van Elssen, C.R.; Wieten, L.; Germeraad, W.; Bos, G. Influence of vitamin C on lymphocytes: An overview. Antioxidants 2018, 7, 41. [Google Scholar] [CrossRef] [Green Version]
- Huijskens, M.; Walczak, M.; Sarkar, S.; Atrafi, F.; Senden-Gijsbers, B.; Tilanus, M.; Bos, G.M.J.; Wieten, L.; Germeraad, W.T.V. Ascorbic acid promotes proliferation of natural killer cell populations in culture systems applicable for natural killer cell therapy. Crytotherapy 2015, 17, 613–620. [Google Scholar] [CrossRef]
- Levine, M.; Padayatty, S.J.; Espey, M.G. Vitamin C: A concentration-function approach yields pharmacology and therapeutic discoveries. Adv. Nutr. 2011, 2, 78–88. [Google Scholar] [CrossRef] [Green Version]
- Pehlivan, F.E. Vitamin C: An antioxidant agent. Vitam. C 2017, 2, 23–35. [Google Scholar]
- Bakalova, R.; Zhelev, Z.; Miller, T.; Aoki, I.; Higashi, T. Vitamin C versus Cancer: Ascorbic Acid Radical and Impairment of Mitochondrial Respiration? Oxid. Med. Cell. Longev. 2020, 2020, 1504048. [Google Scholar] [CrossRef]
- Cimmino, L.; Neel, B.G.; Aifantis, I. Vitamin C in Stem Cell Reprogramming and Cancer. Trends Cell Biol. 2018, 28, 698–708. [Google Scholar] [CrossRef]
- Bruno, R.S.; Traber, M.G. Vitamin E biokinetics, oxidative stress and cigarette smoking. Pathophysiology 2006, 13, 143–149. [Google Scholar] [CrossRef]
- Packer, J.E.; Slater, T.F.; Willson, R.L. Direct observation of a free radical interaction between vitamin E and vitamin C. Nature 1979, 278, 737–738. [Google Scholar] [CrossRef]
- Heffernan, A.; Evans, C.; Holmes, M.; Moore, J.B. The regulation of dietary iron bioavailability by vitamin C: A systematic review and meta-analysis. Proc. Nutr. Soc. 2017, 76, OCE4. [Google Scholar] [CrossRef] [Green Version]
- Young, J.I.; Züchner, S.; Wang, G. Regulation of the epigenome by vitamin C. Annu. Rev. Nutr. 2015, 35, 545–564. [Google Scholar] [CrossRef] [Green Version]
- CCamarena, V.; Wang, G. The epigenetic role of vitamin C in health and disease. Cell. Mol. Life Sci. 2016, 73, 1645–1658. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klose, R.J.; Kallin, E.M.; Zhang, Y. JmjC-domain-containing proteins and histone demethylation. Nat. Rev. Genet. 2006, 7, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.-W.; Friso, S. Epigenetics: A New Bridge between Nutrition and Health. Adv. Nutr. 2010, 1, 8–16. [Google Scholar] [CrossRef]
- Schleicher, R.L.; Carroll, M.D.; Ford, E.S.; Lacher, D.A. Serum vitamin C and the prevalence of vitamin C deficiency in the United States: 2003–2004 National Health and Nutrition Examination Survey (NHANES). Am. J. Clin. Nutr. 2009, 90, 1252–1263. [Google Scholar] [CrossRef]
- Carr, A.C.; Lykkesfeldt, J. Discrepancies in global vitamin C recommendations: A review of RDA criteria and underlying health perspectives. Crit. Rev. Food Sci. Nutr. 2021, 61, 742–755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Health and Nutrition Examination Survey Data 2005–2006; US Department of Health and Human Services, Centers for Disease Control and Prevention: Hyattsville, MD, USA, 2008.
- Plotnick, M.D.; D’Urzo, K.A.; Gurd, B.J.; Pyke, K.E. The influence of vitamin C on the interaction between acute mental stress and endothelial function. Graefe’s Arch. Clin. Exp. Ophthalmol. 2017, 117, 1657–1668. [Google Scholar] [CrossRef]
- Moritz, B.; Schmitz, A.E.; Rodrigues, A.L.S.; Dafre, A.; Cunha, M.P. The role of vitamin C in stress-related disorders. J. Nutr. Biochem. 2020, 85, 108459. [Google Scholar] [CrossRef] [PubMed]
- Bailey, R.L.; Akabas, S.R.; Paxson, E.E.; Thuppal, S.V.; Saklani, S.; Tucker, K. Total Usual Intake of Shortfall Nutrients Varies with Poverty Among US Adults. J. Nutr. Educ. Behav. 2017, 49, 639–646.e3. [Google Scholar] [CrossRef] [Green Version]
- Ellulu, M.S. Obesity, cardiovascular disease, and role of vitamin C on inflammation: A review of facts and underlying mechanisms. Inflammopharmacology 2017, 25, 313–328. [Google Scholar] [CrossRef]
- Thomas-Valdés, S.; Tostes, M.D.G.V.; Anunciação, P.C.; Silva, B.; Sant’Ana, H.M.P. Association between vitamin deficiency and metabolic disorders related to obesity. Crit. Rev. Food Sci. Nutr. 2017, 57, 3332–3343. [Google Scholar] [CrossRef]
- Schectman, G.; Byrd, J.C.; Gruchow, H.W. The influence of smoking on vitamin C status in adults. Am. J. Public Health 1989, 79, 158–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pelletier, O. Smoking and Vitamin C Levels in Humans. Am. J. Clin. Nutr. 1968, 21, 1259–1267. [Google Scholar] [CrossRef] [PubMed]
- Karademirci, M.; Kutlu, R.; Kilinc, I. Relationship between smoking and total antioxidant status, total oxidant status, oxidative stress index, vit C, vit E. Clin. Respir. J. 2018, 12, 2006–2012. [Google Scholar] [CrossRef]
- Carr, A.C.; Maggini, S. Vitamin C and Immune Function. Nutrients 2017, 9, 1211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Healthy People 2030; United States Department of Agriculture: Washington, DC, USA, 2021.
- Schmidt, A.F.; Finan, C. Linear regression and the normality assumption. J. Clin. Epidemiol. 2018, 98, 146–151. [Google Scholar] [CrossRef] [Green Version]
- Wilson, R.W.J.; Gearry, R.B.; Hughes, A.; Lawley, B.; Skidmore, P.; Frampton, C.; Fleming, E.; Anderson, A.; Jones, L.; Tannock, G.W.; et al. SunGold kiwifruit supplementation of individuals with prediabetes alters gut microbiota and improves Vitamin C status, anthropometric and clinical markers. Nutrients 2018, 10, 895. [Google Scholar] [CrossRef] [Green Version]
- Alishahi, A.; Azizbeigi, K.; Salamat, K.M.; Yektayar, M. The Effect of Aerobic Training with Vitamin C Supplementation on Myeloperoxidase, Asymmetric Dimethyl Arginine and Blood Pressure in Middle-Age Hypertensive Overweight Men. J. Clin. Res. Paramedical Sci. 2019, 8, e86843. [Google Scholar] [CrossRef] [Green Version]
- Paalanen, L.; Prättälä, R.; Alfthan, G.; Salminen, I.; Laatikainen, T. Vegetable and fruit consumption, education and plasma vitamin C concentration in Russian and Finnish Karelia, 1992–2002. Public Health Nutr. 2013, 17, 2278–2286. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jungert, A.; Neuhäuser-Berthold, M. The lower vitamin C plasma concentrations in elderly men compared with elderly women can partly be attributed to a volumetric dilution effect due to differences in fat-free mass. Br. J. Nutr. 2015, 113, 859–864. [Google Scholar] [CrossRef] [Green Version]
- Ness, A.R.; Cappuccio, F.P.; Atkinson, R.W.; Khaw, K.T.; Cook, D.G. Plasma vitamin C levels in men and women from different ethnic backgrounds living in England. Int. J. Epidemiol. 1999, 28, 450–455. [Google Scholar] [CrossRef] [Green Version]
- Koh, E.T.; Chi, M.S.; Lowenstein, F.W. Comparison of selected blood components by race, sex, and age. Am. J. Clin. Nutr. 1980, 33, 1828–1835. [Google Scholar] [CrossRef] [PubMed]
- Brown, L.L.; Mitchell, U.A.; Ailshire, J.A. Disentangling the Stress Process: Race/Ethnic Differences in the Exposure and Appraisal of Chronic Stressors Among Older Adults. J. Gerontol. Ser. B 2020, 75, 650–660. [Google Scholar] [CrossRef]
- Vaccaro, J.A.; Huffman, F.G. Sex and race/ethnic disparities in food security and chronic diseases in US older adults. Gerontol. Geriatr. Med. 2017, 3, 2333721417718344. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Thornton, P.L.; Kumanyika, S.K.; Gregg, E.W.; Araneta, M.R.; Baskin, M.L.; Chin, M.H.; Crespo, C.J.; de Groot, M.; Garcia, D.O.; Haire-Joshu, D.; et al. New research directions on disparities in obesity and type 2 diabetes. Ann. N. Y. Acad. Sci. 2020, 1461, 5–24. [Google Scholar] [CrossRef]
- Lykkesfeldt, J.; Viscovich, M.; Poulsen, H.E. Plasma malondialdehyde is induced by smoking: A study with balanced antioxidant profiles. Br. J. Nutr. 2004, 92, 203–206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McCall, S.J.; Clark, A.B.; Luben, R.N.; Wareham, N.J.; Khaw, K.T.; Myint, P.K. Plasma Vitamin C levels: Risk factors for deficiency and association with self-reported functional health in the European Prospective Investigation into Cancer-Norfolk. Nutrients 2019, 11, 1552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cowan, A.E.; Jun, S.; Tooze, J.A.; Eicher-Miller, H.A.; Dodd, K.W.; Gahche, J.J.; Guenther, P.M.; Dwyer, J.T.; Potischman, N.; Bhadra, A.; et al. Total usual micronutrient intakes compared to the dietary reference intakes among US adults by food security status. Nutrients 2020, 12, 38. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelli, H.M.; Hammadah, M.; Ahmed, H.; Ko, Y.-A.; Topel, M.; Samman-Tahhan, A.; Awad, M.; Patel, K.; Mohammed, K.; Sperling, L.S.; et al. Association Between Living in Food Deserts and Cardiovascular Risk. Circ. Cardiovasc. Qual. Outcomes 2017, 10, 003532. [Google Scholar] [CrossRef] [PubMed]
- Karczewski, J.; Śledzińska, E.; Baturo, A.; Jończyk, I.; Maleszko, A.; Samborski, P.; Dobrowolska, A. Obesity and inflammation. Eur. Cytokine Netw. 2018, 29, 83–94. [Google Scholar] [CrossRef]
- Kwaifa, I.K.; Bahari, H.; Yong, Y.K.; Noor, S.M. Endothelial Dysfunction in Obesity-Induced Inflammation: Molecular Mechanisms and Clinical Implications. Biomolecules 2020, 10, 291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laborde, D.; Martin, W.; Swinnen, J.; Vos, R. COVID-19 risks to global food security. Science 2020, 369, 500–502. [Google Scholar] [CrossRef] [PubMed]
- Definitions of Food Insecurity: Ranges of Food Security and Insecurity; United States Department of Agriculture: Washington, DC, USA, 2020.
- Darma, I.Y.; Arni, A.; Basyir, V. The Relationship Vitamin C Level and MMP-2 Serum with Premature Rupture of Membranes and Normal Pregnancy. World J. Res. Rev. 2018, 7, 9–12. [Google Scholar] [CrossRef]
- Juhl, B.; Lauszus, F.F.; Lykkesfeldt, J. Poor Vitamin C Status Late in Pregnancy Is Associated with Increased Risk of Complications in Type 1 Diabetic Women: A Cross-Sectional Study. Nutrients 2017, 9, 186. [Google Scholar] [CrossRef]
- Gupta, S.; Gaikwad, H.S.; Nath, B.; Batra, A. Can vitamin C and interleukin 6 levels predict preterm premature rupture of membranes: Evaluating possibilities in North Indian population. Obstet. Gynecol. Sci. 2020, 63, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Abbasi, A.A.; Rudman, D. Observations on the Prevalence of Protein-Calorie Undernutrition in VA Nursing Homes. J. Am. Geriatr. Soc. 1993, 41, 117–121. [Google Scholar] [CrossRef]
- Assis, B.S.; Jairza, J.M.B.-M.; Lopes, J.A.; Roriz, A.K.C.; Melo, A.L.; Previdell, A.; Aquino, R.D.C.; Ramos, L.B. Micronutrient intake in elderly living in nursing homes. Nutr. Hosp. 2018, 35, 59–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keller, H.H.; Lengyel, C.; Carrier, N.; Slaughter, S.E.; Morrison, J.; Duncan, A.M.; Steele, C.M.; Duizer, L.; Brown, K.S.; Chaudhury, H.; et al. Prevalence of inadequate micronutrient intakes of Canadian long-term care residents. Br. J. Nutr. 2018, 119, 1047–1056. [Google Scholar] [CrossRef] [Green Version]
| Characteristics | n | Weighted n (%) | Mean (S.D.) | Range |
|---|---|---|---|---|
| Gender | ||||
| Male | 3699 | 48.7% ± 0.7% | ||
| Female | 3908 | 51.3% ± 0.7% | ||
| Age | ||||
| Young Adult 20–39 | 2751 | 37.5% ± 0.7% | ||
| Middle Adult 40–59 | 2295 | 40.1% ± 0.7% | ||
| Late Adult ≥60 | 2561 | 22.4% ± 0.5% | ||
| Race/Ethnicity | ||||
| Mexican American | 1516 | 7.6% ± 0.2% | ||
| Other Hispanic | 230 | 3.4% ± 0.3% | ||
| Non-Hispanic White | 4035 | 73.6% ± 0.5% | ||
| Non-Hispanic Black | 1536 | 10.5% ± 0.3% | ||
| Other | 290 | 4.9% ± 0.3% | ||
| Family PIR 1 | ||||
| High (0–1.5) | 5206 | 63.9% ± 0.5% | ||
| Medium (1.51–4.5) | 1614 | 22.6% ± 0.5% | ||
| Low (>4.51) | 787 | 13.5% ± 0.5% | ||
| Smoking Status | ||||
| Yes | 1997 | 29.4% ± 0.6% | ||
| No | 5610 | 70.6% ± 0.6% | ||
| Food Insecure | ||||
| Yes | 1449 | 14.1% ± 0.4% | ||
| No | 6158 | 85.9% ± 0.4% | ||
| BMI 2 | 7607 | 28.68 (6.44) | 13.36–76.07 | |
| Plasma Vitamin C 3 | 7607 | 54.37 (28.62) | 0.6–274.20 | |
| Vitamin C Intake Day 1 4 | 7468 | 91.91 (104.14) | 0.0–2261.00 | |
| Vitamin C Intake Day 2 4 | 6886 | 95.30 (99.31) | 0.0–1308.40 |
| Characteristics | n (Weighted %) | Deficiency | Hypo-Vitaminosis | Inadequate | Adequate | Saturating | p-Value | |
|---|---|---|---|---|---|---|---|---|
| n = 467, | n = 722 | n = 1991, 26.17% | n = 2467, 32.43% | n = 1960, 25.77% | Effect Size 5 | |||
| 6.14% | 9.49% | |||||||
| Gender | ||||||||
| Male | 3699 (48.7% ± 0.5) | 8.6% (±0.8) | 12.4% (±0.8) | 28.2% (±1.2) | 31.6% (±1.1) | 19.3% (±0.8) | <0.001 | 0.17 |
| Female | 3908 (51.3% ± 0.5) | 4.9% (±0.6) | 8.2% (±0.7) | 23.0% (±1.0) | 31.7% (±1.3) | 32.3% (±1.3) | ||
| Adulthood Stage | ||||||||
| Young (20–39) | 2751 (36.5% ± 0.8) | 6.6% (±0.8) | 11.0% (±0.9) | 28.9% (±1.1) | 31.8% (±1.3) | 21.7% (±1.4) | <0.001 | 0.11 |
| Middle (40–59) | 2295 (40.1% ± 0.8) | 7.9% (±1.0) | 11.0% (±0.7) | 26.3% (±1.0) | 32.8% (±1.2) | 22.1% (±1.2) | ||
| Late (≥60) | 2561 (22.3% ± 0.5) | 4.6% (±0.6) | 7.6% (±0.8) | 18.5% (±0.8) | 29.4% (±1.5) | 40.0% (±1.1) | ||
| Race/Ethnicity | <0.001 | |||||||
| Mexican American | 1516 (7.6% ± 1.1) | 3.8% (±0.8) | 7.7% (±1.3) | 32.3% (±1.5) | 37.6% (±1.8) | 18.7% (±1.5) | ||
| Other Hispanic | 230 (3.4% ± 0.5) | 1.3% (±0.8) | 10.5% (±3.1) | 29.0% (±3.1) | 38.6% (±4.3) | 20.6% (±3.7) | 0.08 | |
| Non-Hispanic White | 4305 (73.6% ± 2.1) | 7.6% (±0.7) | 10.6% (±0.7) | 23.2% (±0.9) | 30.5% (±1.1) | 28.2% (±1.1) | ||
| Non-Hispanic Black | 1536 (10.5% ± 1.2) | 5.5% (±0.8) | 8.3% (±1.0) | 34.7% (±1.6) | 32.3% (±1.7) | 19.1% (±1.2) | ||
| Other | 290 (4.9% ± 0.4) | 3.8% (±1.5) | 12.6% (±2.5) | 28.3% (±2.2) | 33.5% (±2.6) | 21.8% (±2.0) | ||
| Family PIR 1 | 0.002 | |||||||
| High (0–1.5) | 5206 (63.8% ± 1.1) | 6.3% (±0.6) | 11.1% (±0.7) | 26.5% (±0.7) | 31.5% (±1.2) | |||
| Medium (1.51–4.5) | 1614 (22.7% ± 0.5) | 9.5% (±1.6) | 9.8% (±1.1) | 24.2% (±1.3) | 31.0% (±1.6) | 24.5% (±0.9) | 0.06 | |
| Low (>4.51) | 787 (13.5% ± 0.6) | 3.7% (±1.1) | 7.0% (±1.0) | 22.9% (±2.4) | 33.4% (±1.6) | 25.5% (±1.8) | ||
| 33.1% (±2.1) | ||||||||
| Smoking Status | 0.001 | |||||||
| Yes | 1997 (29.4% ± 1.0) | 14.8% (±1.0) | 17.0% (±1.2) | 29.0% (±1.3) | 24.6% (±1.5) | 14.7% (±1.0) | 0.28 | |
| No | 5610 (70.6% ± 1.0) | 3.3% (±0.4) | 7.4% (±0.5) | 24.1% (±0.8) | 34.6% (±0.8) | 30.7% (±0.9) | ||
| Food Insecure | <0.001 | |||||||
| Yes | 1449 (14.1% ± 0.8) | 9.7% (±1.2) | 13.9% (±1.5) | 32.9% (±1.1) | 27.3% (±1.7) | 16.2% (±1.2) | 0.12 | |
| No | 6158 (85.9% ± 0.8) | 6.2% (±0.6) | 9.6% (±0.6) | 24.3% (±0.8) | 32.3% (±1.0) | 27.6% (±0.9) | ||
| BMI 2 | 7607 (100%) | 29.0 (±7.3) | 29.8 (±7.4) | 29.8 (±6.8) | 28.6 (±6.1) | 27.1 (±5.5) | <0.001 4 | 0.02 |
| Vitamin C Intake 3 | <0.001 4 | |||||||
| Day One | 7468 (98.1%) | 39.4 (±55.0) | 50.9 (±61.7) | 73.5 (±94.1) | 102.2 (±104.8) | 124.9 (±119.5) | 0.06 | |
| Day Two | 6886 (90.5%) | 41.6 (±68.7) | 60.2 (±74.2) | 84.2 (±89.1) | 104.7 (±106.8) | 118.8 (±103.4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Crook, J.; Horgas, A.; Yoon, S.-J.; Grundmann, O.; Johnson-Mallard, V. Insufficient Vitamin C Levels among Adults in the United States: Results from the NHANES Surveys, 2003–2006. Nutrients 2021, 13, 3910. https://doi.org/10.3390/nu13113910
Crook J, Horgas A, Yoon S-J, Grundmann O, Johnson-Mallard V. Insufficient Vitamin C Levels among Adults in the United States: Results from the NHANES Surveys, 2003–2006. Nutrients. 2021; 13(11):3910. https://doi.org/10.3390/nu13113910
Chicago/Turabian StyleCrook, Jennifer, Ann Horgas, Saun-Joo Yoon, Oliver Grundmann, and Versie Johnson-Mallard. 2021. "Insufficient Vitamin C Levels among Adults in the United States: Results from the NHANES Surveys, 2003–2006" Nutrients 13, no. 11: 3910. https://doi.org/10.3390/nu13113910
APA StyleCrook, J., Horgas, A., Yoon, S.-J., Grundmann, O., & Johnson-Mallard, V. (2021). Insufficient Vitamin C Levels among Adults in the United States: Results from the NHANES Surveys, 2003–2006. Nutrients, 13(11), 3910. https://doi.org/10.3390/nu13113910







