Could Vitamin D Be Effective in Prevention of Preeclampsia?
Abstract
1. Introduction
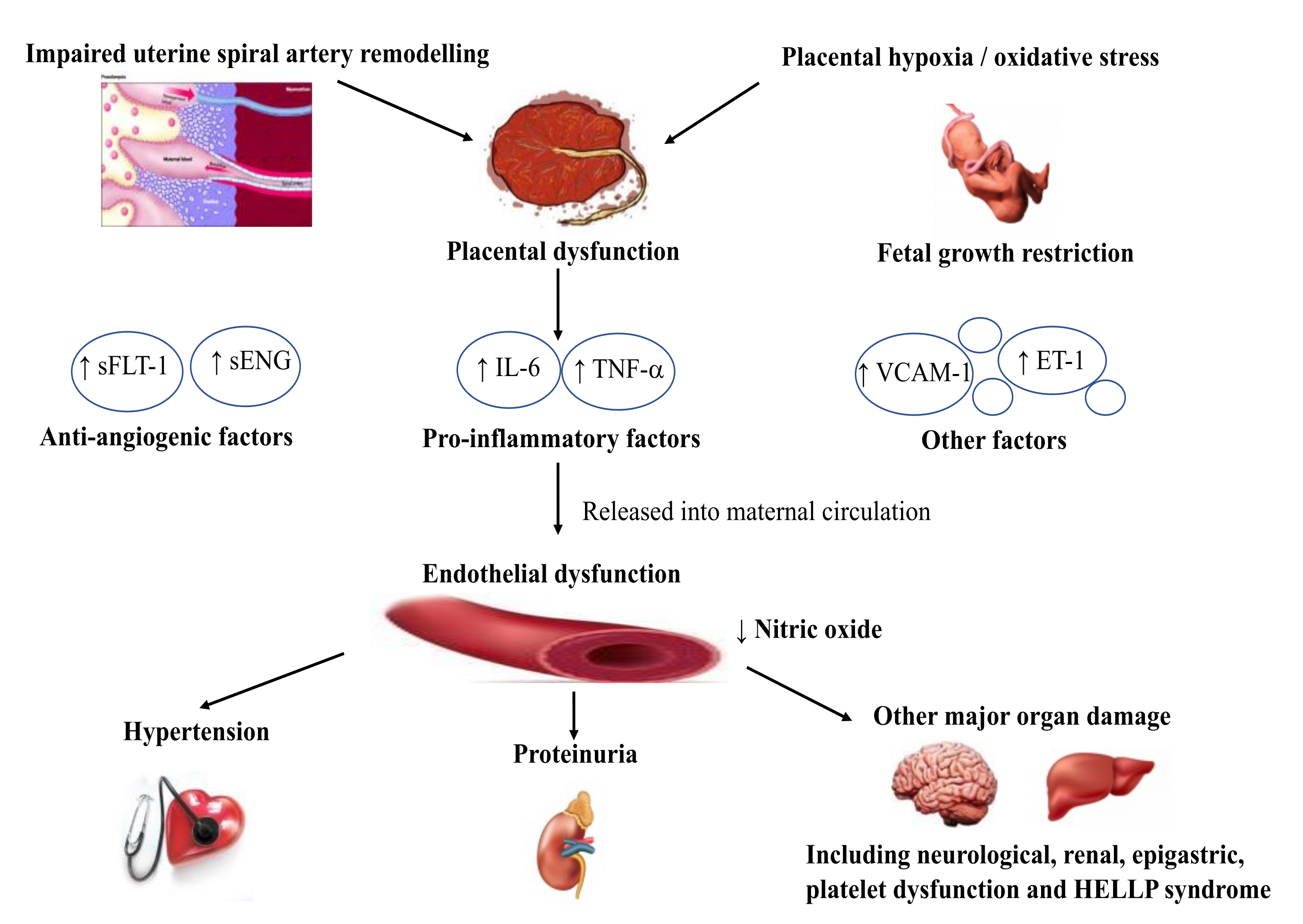
2. Pathogenesis of Preeclampsia
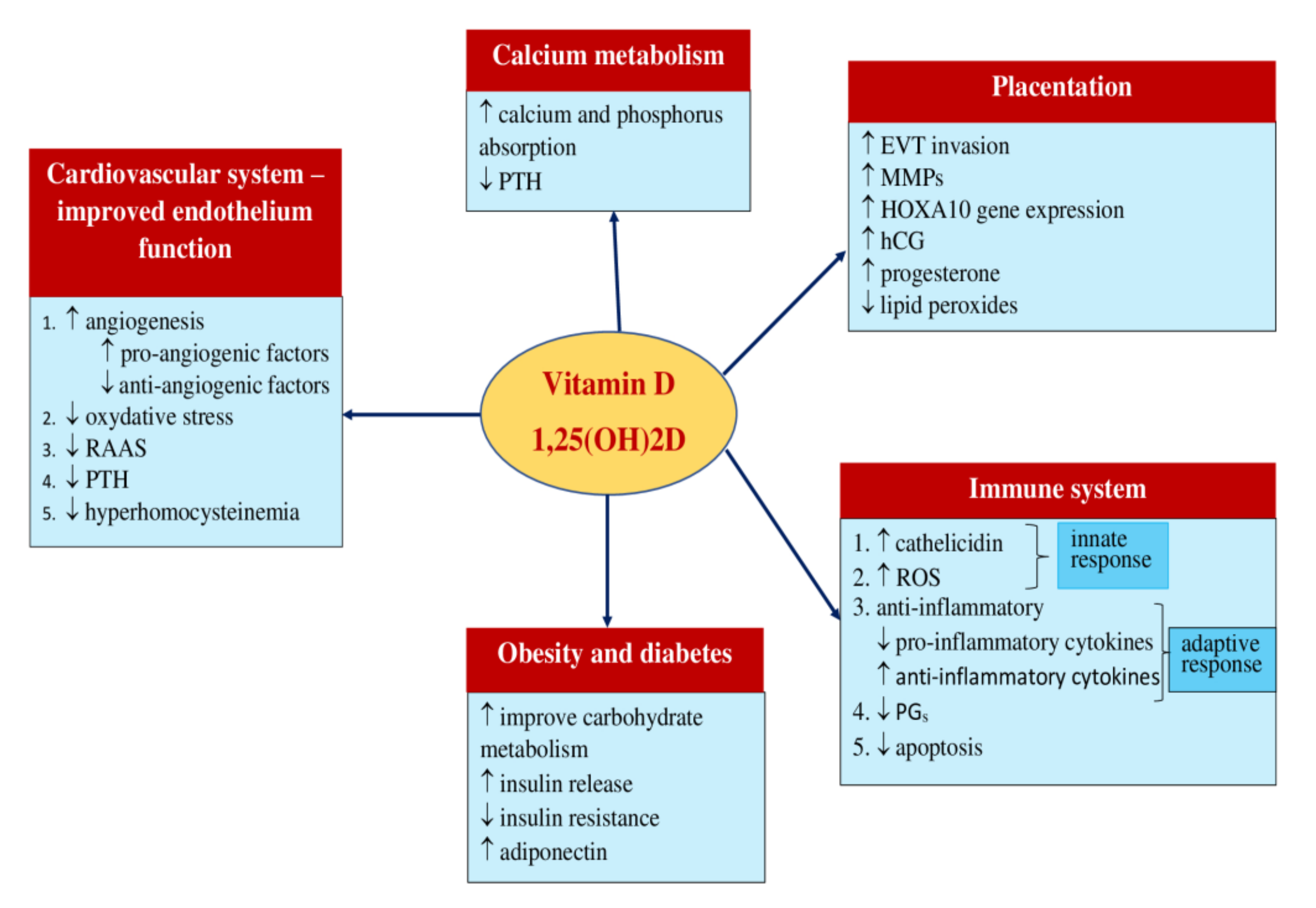
3. Vitamin D
3.1. Metabolism
3.2. Mechanism of Action
3.2.1. Calcium and Phosphorus Metabolism
3.2.2. Immune System
3.2.3. Cardiovascular System
4. Vitamin D in Pregnancy
Recommendations on Supplementation of Vitamin D
5. Vitamin D and Preeclampsia—Experimental Research
5.1. Trophoblast
5.2. Angiogenic Factors and Endothelium
5.3. Immune System
5.4. RAAS
6. Vitamin D and Preeclampsia Risk
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- ACOG. ACOG Practice Bulletin No. 203: Chronic Hypertension in Pregnancy. Obstet. Gynecol. 2019, 133, e26–e50. [Google Scholar] [CrossRef] [PubMed]
- ACOG. ACOG Practice Bulletin No. 202: Gestational Hypertension and Preeclampsia. Obstet. Gynecol. 2019, 133, e1–e25. [Google Scholar] [CrossRef]
- Duley, L. The Global Impact of Pre-eclampsia and Eclampsia. Semin. Perinatol. 2009, 33, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Ronsmans, C.; Graham, W.J. Maternal mortality: Who, when, where, and why. Lancet 2006, 368, 1189–1200. [Google Scholar] [CrossRef]
- Khan, K.S.; Wojdyla, D.; Say, L.; Gülmezoglu, A.M.; Van Look, P.F. WHO analysis of causes of maternal death: A systematic review. Lancet 2006, 367, 1066–1074. [Google Scholar] [CrossRef]
- McDonald, S.D.; Malinowski, A.; Zhou, Q.; Yusuf, S.; Devereaux, P. Cardiovascular sequelae of preeclampsia/eclampsia: A systematic review and meta-analyses. Am. Heart J. 2008, 156, 918–930. [Google Scholar] [CrossRef]
- Prejbisz, A.; Dobrowolski, P.; Kosiński, P.; Bomba-Opoń, D.; Adamczak, M.; Bekiesinska-Figatowska, M.; Kądziela, J.; Konopka, A.; Kostka-Jeziorny, K.; Kurnatowska, I.; et al. Management of hypertension in pregnancy: Prevention, diagnosis, treatment and long-term prognosis. Kardiol. Pol. 2019, 77, 757–806. [Google Scholar] [CrossRef]
- Brown, M.A.; Magee, L.A.; Kenny, L.C.; Karumanchi, S.A.; McCarthy, F.; Saito, S.; Hall, D.R.; Warren, C.E.; Adoyi, G.; Ishaku, S. The hypertensive disorders of pregnancy: ISSHP classification, diagnosis & management recommendations for international practice. Pregnancy Hypertens. 2018, 13, 291–310. [Google Scholar] [CrossRef]
- Magee, L.A.; Pels, A.; Helewa, M.; Rey, E.; von Dadelszen, P. In Response. J. Obstet. Gynaecol. Can. 2014, 36, 575–576. [Google Scholar] [CrossRef]
- North, R.A.; McCowan, L.; Dekker, G.A.; Poston, L.; Chan, E.H.Y.; Stewart, A.W.; Black, M.; Taylor, R.; Walker, J.; Baker, P.; et al. Clinical risk prediction for pre-eclampsia in nulliparous women: Development of model in international prospective cohort. BMJ 2011, 342, d1875. [Google Scholar] [CrossRef] [PubMed]
- Browne, J.L.; Klipstein-Grobusch, K.; Franx, A.; Grobbee, D.E. Prevention of Hypertensive Disorders of Pregnancy: A Novel Application of the Polypill Concept. Curr. Cardiol. Rep. 2016, 18, 59. [Google Scholar] [CrossRef] [PubMed]
- Costantine, M.M.; Cleary, K. Pravastatin for the Prevention of Preeclampsia in High-Risk Pregnant Women. Obstet. Gynecol. 2013, 121, 349–353. [Google Scholar] [CrossRef] [PubMed]
- Tong, S.; Kaitu’U-Lino, T.J.; Hastie, R.; Brownfoot, F.; Cluver, C.; Hannan, N. Pravastatin, proton-pump inhibitors, metformin, micronutrients, and biologics: New horizons for the prevention or treatment of preeclampsia. Am. J. Obstet. Gynecol. 2020, 16, S0002-9378(20)31071-1. [Google Scholar] [CrossRef] [PubMed]
- Poniedziałek-Czajkowska, E.; Mierzyński, R.; Dłuski, D.; Leszczyńska-Gorzelak, B. Prevention of Hypertensive Disorders of Pregnancy—Is There a Place for Metformin? J. Clin. Med. 2021, 10, 2805. [Google Scholar] [CrossRef]
- Karras, S.N.; Wagner, C.L.; Castracane, V.D. Understanding vitamin D metabolism in pregnancy: From physiology to pathophysiology and clinical outcomes. Metabolism 2018, 86, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Vitamin D Deficiency. N. Engl. J. Med. 2007, 357, 266–281. [Google Scholar] [CrossRef]
- Reid, I. What diseases are causally linked to vitamin D deficiency? Arch. Dis. Child. 2015, 101, 185–189. [Google Scholar] [CrossRef] [PubMed]
- Kaufmann, P.; Black, S.; Huppertz, B. Endovascular Trophoblast Invasion: Implications for the Pathogenesis of Intrauterine Growth Retardation and Preeclampsia. Biol. Reprod. 2003, 69, 1–7. [Google Scholar] [CrossRef]
- Ishihara, N.; Matsuo, H.; Murakoshi, H.; Laoag-Fernandez, J.B.; Samoto, T.; Maruo, T. Increased apoptosis in the syncytiotrophoblast in human term placentas complicated by either preeclampsia or intrauterine growth retardation. Am. J. Obstet. Gynecol. 2002, 186, 158–166. [Google Scholar] [CrossRef]
- Perez-Sepulveda, A.; Torres, M.J.; Khoury, M.; Illanes, S.E. Innate Immune System and Preeclampsia. Front. Immunol. 2014, 5, 244. [Google Scholar] [CrossRef]
- Redman, C.W.G.; Sargent, I.L. Immunology of Pre-Eclampsia. Am. J. Reprod. Immunol. 2010, 63, 534–543. [Google Scholar] [CrossRef]
- Borzychowski, A.; Sargent, I.; Redman, C. Inflammation and pre-eclampsia. Semin. Fetal Neonatal Med. 2006, 11, 309–316. [Google Scholar] [CrossRef] [PubMed]
- Luppi, P.; DeLoia, J.A. Monocytes of preeclamptic women spontaneously synthesize pro-inflammatory cytokines. Clin. Immunol. 2006, 118, 268–275. [Google Scholar] [CrossRef]
- Hsu, P.; Santner-Nanan, B.; Dahlstrom, J.; Fadia, M.; Chandra, A.; Peek, M.; Nanan, R. Altered Decidual DC-SIGN+ Antigen-Presenting Cells and Impaired Regulatory T-Cell Induction in Preeclampsia. Am. J. Pathol. 2012, 181, 2149–2160. [Google Scholar] [CrossRef] [PubMed]
- Cerdeira, A.S.; Rajakumar, A.; Royle, C.M.; Lo, A.; Husain, Z.; Thadhani, R.I.; Sukhatme, V.P.; Karumanchi, S.A.; Kopcow, H.D. Conversion of Peripheral Blood NK Cells to a Decidual NK-like Phenotype by a Cocktail of Defined Factors. J. Immunol. 2013, 190, 3939–3948. [Google Scholar] [CrossRef] [PubMed]
- Guimond, M.-J.; Wang, B.; Croy, B.A. Engraftment of Bone Marrow from Severe Combined Immunodeficient (SCID) Mice Reverses the Reproductive Deficits in Natural Killer Cell–deficient tgε26 Mice. J. Exp. Med. 1998, 187, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Fraser, R.; Whitley, G.S.; Johnstone, A.P.; Host, A.J.; Sebire, N.J.; Thilaganathan, B.; Cartwright, J.E. Impaired decidual natural killer cell regulation of vascular remodelling in early human pregnancies with high uterine artery resistance. J. Pathol. 2012, 228, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Redman, C. Pre-eclampsia and the placenta. Placenta 1991, 12, 301–308. [Google Scholar] [CrossRef]
- Brennan, L.J.; Morton, J.S.; Davidge, S.T. Vascular Dysfunction in Preeclampsia. Microcirculation 2014, 21, 4–14. [Google Scholar] [CrossRef]
- Poniedziałek–Czajkowska, E.; Mierzyński, R.; Dłuski, D.; Leszczyńska-Gorzelak, B. Adipokines and Endothelium Dysfunction Markers in Pregnant Women with Gestational Hypertension. Int. J. Hypertens. 2019, 2019, 7541846. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Zheng, D.-Y.; Yang, J.-M.; Wang, M.; Zhang, X.-T.; Sun, L.; Yun, X.-G. Maternal serum uric acid concentration is associated with the expression of tumour necrosis factor-α and intercellular adhesion molecule-1 in patients with preeclampsia. J. Hum. Hypertens. 2015, 30, 456–462. [Google Scholar] [CrossRef]
- Endemann, D.H. Endothelial Dysfunction. J. Am. Soc. Nephrol. 2004, 15, 1983–1992. [Google Scholar] [CrossRef] [PubMed]
- Mutter, W.P.; Karumanchi, S.A. Molecular mechanisms of preeclampsia. Microvasc. Res. 2008, 75, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Levine, R.J.; Maynard, S.E.; Qian, C.; Lim, K.-H.; England, L.J.; Yu, K.F.; Schisterman, E.; Thadhani, R.; Sachs, B.P.; Epstein, F.H.; et al. Circulating Angiogenic Factors and the Risk of Preeclampsia. N. Engl. J. Med. 2004, 350, 672–683. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Rana, S.; Karumanchi, S.A. Preeclampsia: The Role of Angiogenic Factors in Its Pathogenesis. Physiology 2009, 24, 147–158. [Google Scholar] [CrossRef] [PubMed]
- Bdolah, Y.; Sukhatme, V.P.; Karumanchi, S.A. Angiogenic imbalance in the pathophysiology of preeclampsia: Newer insights. Semin. Nephrol. 2004, 24, 548–556. [Google Scholar] [CrossRef] [PubMed]
- Millauer, B. High affinity VEGF binding and developmental expression suggest Flk-1 as a major regulator of vasculogenesis and angiogenesis. Cell 1993, 72, 835–846. [Google Scholar] [CrossRef]
- Mandriota, S.J.; Seghezzi, G.; Vassalli, J.-D.; Ferrara, N.; Wasi, S.; Mazzieri, R.; Mignatti, P.; Pepper, M. Vascular Endothelial Growth Factor Increases Urokinase Receptor Expression in Vascular Endothelial Cells. J. Biol. Chem. 1995, 270, 9709–9716. [Google Scholar] [CrossRef] [PubMed]
- Köroğlu, N.; Tola, E.; Yuksel, I.T.; Cetin, B.A.; Turhan, U.; Topcu, G.; Dag, I. Maternal serum AMP-activated protein kinase levels in mild and severe preeclampsia. J. Matern.-Fetal Neonatal Med. 2018, 32, 2735–2740. [Google Scholar] [CrossRef] [PubMed]
- Waker, C.; Albers, R.E.; Pye, R.L.; Doliboa, S.R.; Wyatt, C.N.; Brown, T.L.; Mayes, D.A. AMPK Knockdown in Placental Labyrinthine Progenitor Cells Results in Restriction of Critical Energy Resources and Terminal Differentiation Failure. Stem Cells Dev. 2017, 26, 808–817. [Google Scholar] [CrossRef] [PubMed]
- Xiang, L.; Varshney, R.; Rashdan, N.A.; Shaw, J.H.; Lloyd, P.G. Placenta Growth Factor and Vascular Endothelial Growth Factor A Have Differential, Cell-Type Specific Patterns of Expression in Vascular Cells. Microcirculation 2014, 21, 368–379. [Google Scholar] [CrossRef] [PubMed]
- Autiero, M.; Luttun, A.; Tjwa, M.; Carmeliet, P. Placental growth factor and its receptor, vascular endothelial growth factor receptor-1: Novel targets for stimulation of ischemic tissue revascularization and inhibition of angiogenic and inflammatory disorders. J. Thromb. Haemost. 2003, 1, 1356–1370. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Chen, L.; Liu, B.; Vialli, C.; Stone, P.; Ching, L.-M.; Chamley, L. The role of autocrine TGFβ1 in endothelial cell activation induced by phagocytosis of necrotic trophoblasts: A possible role in the pathogenesis of pre-eclampsia. J. Pathol. 2010, 221, 87–95. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Shen, L.; Tan, H. Polymorphisms and Plasma Level of Transforming Growth Factor-Beta 1 and Risk for Preeclampsia: A Systematic Review. PLoS ONE 2014, 9, e97230. [Google Scholar] [CrossRef] [PubMed]
- Neufeld, G.; Cohen, T.; Gengrinovitch, S.; Poltorak, Z. Vascular endothelial growth factor (VEGF) and its receptors. FASEB J. 1999, 13, 9–22. [Google Scholar] [CrossRef]
- Maynard, S.E.; Min, J.-Y.; Merchan, J.; Lim, K.-H.; Li, J.; Mondal, S.; Libermann, T.A.; Morgan, J.P.; Sellke, F.W.; Stillman, I.E.; et al. Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J. Clin. Investig. 2003, 111, 649–658. [Google Scholar] [CrossRef]
- Maynard, S.E.; Venkatesha, S.; Thadhani, R.; Karumanchi, S.A. Soluble Fms-like Tyrosine Kinase 1 and Endothelial Dysfunction in the Pathogenesis of Preeclampsia. Pediatr. Res. 2005, 57, 1R–7R. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira, L.; Peraçoli, J.C.; Peracoli, M.T.; Korkes, H.; Zampieri, G.; Moron, A.; Sass, N. sFlt-1/PlGF ratio as a prognostic marker of adverse outcomes in women with early-onset preeclampsia. Pregnancy Hypertens. 2013, 3, 191–195. [Google Scholar] [CrossRef]
- Chaiworapongsa, T.; Chaemsaithong, P.; Yeo, L.; Romero, R. Pre-eclampsia part 1: Current understanding of its pathophysiology. Nat. Rev. Nephrol. 2014, 10, 466–480. [Google Scholar] [CrossRef]
- Leaños-Miranda, A.; Navarro-Romero, C.S.; Sillas-Pardo, L.J.; Ramírez-Valenzuela, K.L.; Isordia-Salas, I.; Jiménez-Trejo, L.M. Soluble Endoglin as a Marker for Preeclampsia, Its Severity, and the Occurrence of Adverse Outcomes. Hypertension 2019, 74, 991–997. [Google Scholar] [CrossRef]
- Luft, F.C. Soluble endoglin (sEng) joins the soluble fms-like tyrosine kinase (sFlt) receptor as a pre-eclampsia molecule. Nephrol. Dial. Transplant. 2006, 21, 3052–3054. [Google Scholar] [CrossRef]
- Venkatesha, S.; Toporsian, M.; Lam, C.; Hanai, J.-I.; Mammoto, T.; Kim, Y.M.; Bdolah, Y.; Lim, K.-H.; Yuan, H.-T.; Libermann, T.; et al. Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat. Med. 2006, 12, 642–649. [Google Scholar] [CrossRef]
- Sutton, E.F.; Gemmel, M.; Powers, R.W. Nitric oxide signaling in pregnancy and preeclampsia. Nitric Oxide 2019, 95, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Nejabati, H.R.; Latifi, Z.; Ghasemnejad, T.; Fattahi, A.; Nouri, M. Placental growth factor (PlGF) as an angiogenic/inflammatory switcher: Lesson from early pregnancy losses. Gynecol. Endocrinol. 2017, 33, 668–674. [Google Scholar] [CrossRef] [PubMed]
- Zafer, E.; Yenisey, C.; Eken, M.K.; Ozdemir, E.; Omurlu, I.K.; Yuksel, H. Second trimester maternal serum–amniotic fluid nitric oxide and vascular endothelial growth factor levels in relation to uterine artery Doppler indices in pregnancies with normal outcome. J. Obstet. Gynaecol. 2018, 38, 1088–1092. [Google Scholar] [CrossRef]
- Taysi, S.; Tascan, A.S.; Ugur, M.G.; Demir, M.; Uuro, M.G. Radicals, Oxidative/Nitrosative Stress and Preeclampsia. Mini-Rev. Med. Chem. 2019, 19, 178–193. [Google Scholar] [CrossRef] [PubMed]
- Chaiworapongsa, T.; Romero, R.; Yoshimatsu, J.; Espinoza, J.; Kim, Y.M.; Park, K.; Kalache, K.; Edwin, S.; Bujold, E.; Gomez, R. Soluble adhesion molecule profile in normal pregnancy and pre-eclampsia. J. Matern.-Fetal Neonatal Med. 2002, 12, 19–27. [Google Scholar] [CrossRef]
- George, E.; Granger, J.P. Endothelin: Key Mediator of Hypertension in Preeclampsia. Am. J. Hypertens. 2011, 24, 964–969. [Google Scholar] [CrossRef]
- Austgulen, R.; Lien, E.; Vince, G.; Redman, C.W. Increased maternal plasma levels of soluble adhesion molecules (ICAM-1, VCAM-1, E-selectin) in preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 1997, 71, 53–58. [Google Scholar] [CrossRef]
- Rana, S.; Schnettler, W.T.; Powe, C.; Wenger, J.; Salahuddin, S.; Cerdeira, A.S.; Verlohren, S.; Perschel, F.H.; Arany, Z.; Lim, K.-H.; et al. Clinical characterization and outcomes of preeclampsia with normal angiogenic profile. Hypertens. Pregnancy 2013, 32, 189–201. [Google Scholar] [CrossRef] [PubMed]
- Verlohren, S.; Stepan, H.; Dechend, R. Angiogenic growth factors in the diagnosis and prediction of pre-eclampsia. Clin. Sci. 2011, 122, 43–52. [Google Scholar] [CrossRef]
- Tal, R. The Role of Hypoxia and Hypoxia-Inducible Factor-1Alpha in Preeclampsia Pathogenesis. Biol. Reprod. 2012, 87, 134. [Google Scholar] [CrossRef]
- Isaka, K.; Usuda, S.; Ito, H.; Sagawa, Y.; Nakamura, H.; Nishi, H.; Suzuki, Y.; Li, Y.; Takayama, M. Expression and Activity of Matrix Metalloproteinase 2 and 9 in Human Trophoblasts. Placenta 2003, 24, 53–64. [Google Scholar] [CrossRef]
- Merchant, S.J.; Narumiya, H.; Zhang, Y.; Guilbert, L.J.; Davidge, S.T. The Effects of Preeclampsia and Oxygen Environment on Endothelial Release of Matrix Metalloproteinase-2. Hypertens. Pregnancy 2004, 23, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Khalil, R.A. Matrix Metalloproteinases in Normal Pregnancy and Preeclampsia. In Progress in Molecular Biology and Translational Science; Elsevier: Amsterdam, The Netherlands, 2017; Volume 148, pp. 87–165. [Google Scholar] [CrossRef]
- Walsh, S.W. Eicosanoids in preeclampsia. Prostaglandins Leukot. Essent. Fat. Acids 2004, 70, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Walsh, S.W.; Coulter, S. Increased placental progesterone may cause decreased placental prostacyclin production in preeclampsia. Am. J. Obstet. Gynecol. 1989, 161, 1586–1592. [Google Scholar] [CrossRef]
- Irani, R.; Xia, Y. The Functional Role of the Renin–Angiotensin System in Pregnancy and Preeclampsia. Placenta 2008, 29, 763–771. [Google Scholar] [CrossRef]
- Langer, B. Plasma Active Renin, Angiotensin I, and Angiotensin II During Pregnancy and in Preeclampsia. Obstet. Gynecol. 1998, 91, 196–202. [Google Scholar] [CrossRef]
- Velloso, E.; Vieira, R.; Cabral, A.; Kalapothakis, E.; Santos, R. Reduced plasma levels of angiotensin-(1-7) and renin activity in preeclamptic patients are associated with the angiotensin I- converting enzyme deletion/deletion genotype. Braz. J. Med. Biol. Res. 2007, 40, 583–590. [Google Scholar] [CrossRef]
- Faulkner, J.L.; Amaral, L.M.; Cornelius, D.; Cunningham, M.W.; Ibrahim, T.; Heep, A.; Campbell, N.; Usry, N.; Wallace, K.; Herse, F.; et al. Vitamin D supplementation reduces some AT1-AA-induced downstream targets implicated in preeclampsia including hypertension. Am. J. Physiol. Integr. Comp. Physiol. 2017, 312, R125–R131. [Google Scholar] [CrossRef]
- Gathiram, P.; Moodley, J. The Role of the Renin-Angiotensin-Aldosterone System in Preeclampsia: A Review. Curr. Hypertens. Rep. 2020, 22, 1–9. [Google Scholar] [CrossRef]
- Pérez-Sepúlveda, A.; España-Perrot, P.P.; Fernández, B.X.; Ahumada, V.; Bustos, V.; Arraztoa, J.A.; Dobierzewska, A.; Figueroa-Diesel, H.; Rice, G.E.; Illanes, S.E. Levels of Key Enzymes of Methionine-Homocysteine Metabolism in Preeclampsia. BioMed Res. Int. 2013, 2013, 731962. [Google Scholar] [CrossRef] [PubMed]
- Rolfo, A.; Many, A.; Racano, A.; Tal, R.; Tagliaferro, A.; Ietta, F.; Wang, J.; Post, M.; Caniggia, I. Abnormalities in Oxygen Sensing Define Early and Late Onset Preeclampsia as Distinct Pathologies. PLoS ONE 2010, 5, e13288. [Google Scholar] [CrossRef]
- Herrmann, W.; Isber, S.; Obeid, R.; Herrmann, M.; Jouma, M. Concentrations of homocysteine, related metabolites and asymmetric dimethylarginine in preeclamptic women with poor nutritional status. Clin. Chem. Lab. Med. 2005, 43, 1139–1146. [Google Scholar] [CrossRef] [PubMed]
- Reilly, R.; McNulty, H.; Pentieva, K.; Strain, J.J.; Ward, M. MTHFR677TT genotype and disease risk: Is there a modulating role for B-vitamins? Proc. Nutr. Soc. 2013, 73, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.; MacLaughlin, J.A.; Clark, M.B.; Holick, S.A.; Potts, J.T.; Anderson, R.R.; Blank, I.H.; Parrish, J.A.; Elias, P. Photosynthesis of previtamin D3 in human skin and the physiologic consequences. Science 1980, 210, 203–205. [Google Scholar] [CrossRef] [PubMed]
- Webb, A.R.; DeCosta, B.R.; Holick, M. Sunlight Regulates the Cutaneous Production of Vitamin D3by Causing Its Photodegradation. J. Clin. Endocrinol. Metab. 1989, 68, 882–887. [Google Scholar] [CrossRef] [PubMed]
- Ross, A.C. The 2011 report on dietary reference intakes for calcium and vitamin D. Public Health Nutr. 2011, 14, 938–939. [Google Scholar] [CrossRef]
- Holick, M.F. Vitamin D: Photobiology, metabolism and clinical applications. In Endocrinology, 3rd ed.; Degroot, L., Ed.; Saunders: Philadelphia, PA, USA, 1995; Volume 2, pp. 990–1014. [Google Scholar]
- Andersson, S.; Davis, D.L.; Dahlbäck, H.; Jörnvall, H.; Russell, D.W. Cloning, structure, and expression of the mitochondrial cyto-chrome P-450 sterol 26-hydroxylase, a bile acid biosynthetic enzyme. J. Biol. Chem. 1989, 264, 8222–8229. [Google Scholar] [CrossRef]
- Heaney, R.P.; Leite, H.P.; De Lima, L.F.P.; Taddei, J.A.D.A. Serum 25-hydroxyvitamin D is a reliable indicator of vitamin D status. Am. J. Clin. Nutr. 2011, 94, 619–620. [Google Scholar] [CrossRef]
- Karras, S.N.; Koufakis, T.; Fakhoury, H.; Kotsa, K. Deconvoluting the Biological Roles of Vitamin D-Binding Protein During Pregnancy: A Both Clinical and Theoretical Challenge. Front. Endocrinol. 2018, 9, 259. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.-J.; Ji, M.; Song, J.; Moon, H.-W.; Hur, M.; Yun, Y.-M. Clinical Utility of Measurement of Vitamin D-Binding Protein and Calculation of Bioavailable Vitamin D in Assessment of Vitamin D Status. Ann. Lab. Med. 2017, 37, 34–38. [Google Scholar] [CrossRef]
- Chun, R.; Peercy, B.E.; Orwoll, E.S.; Nielson, C.M.; Adams, J.S.; Hewison, M. Vitamin D and DBP: The free hormone hypothesis revisited. J. Steroid Biochem. Mol. Biol. 2013, 144, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Nykjaer, A.; Dragun, D.; Walther, D.; Vorum, H.; Jacobsen, C.; Herz, J.; Melsen, F.; Christensen, E.I.; Willnow, T.E. An Endocytic Pathway Essential for Renal Uptake and Activation of the Steroid 25-(OH) Vitamin D3. Cell 1999, 96, 507–515. [Google Scholar] [CrossRef]
- Cheng, J.B.; Motola, D.L.; Mangelsdorf, D.; Russell, D. De-orphanization of Cytochrome P450 2R1. J. Biol. Chem. 2003, 278, 38084–38093. [Google Scholar] [CrossRef] [PubMed]
- Cheng, J.B.; Levine, M.; Bell, N.H.; Mangelsdorf, D.; Russell, D. Genetic evidence that the human CYP2R1 enzyme is a key vitamin D 25-hydroxylase. Proc. Natl. Acad. Sci. USA 2004, 101, 7711–7715. [Google Scholar] [CrossRef] [PubMed]
- Hollis, B.W.; Wagner, C.L. The Role of the Parent Compound Vitamin D with Respect to Metabolism and Function: Why Clinical Dose Intervals Can Affect Clinical Outcomes. J. Clin. Endocrinol. Metab. 2013, 98, 4619–4628. [Google Scholar] [CrossRef] [PubMed]
- Speeckaert, M.M.; Speeckaert, R.; van Geel, N.; Delanghe, J.R. Vitamin D Binding Protein. Adv. Clin. Chem. 2013, 63, 1–57. [Google Scholar] [CrossRef]
- Henry, H.L. Regulation of vitamin D metabolism. Best Pract. Res. Clin. Endocrinol. Metab. 2011, 25, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Shimada, T.; Hasegawa, H.; Yamazaki, Y.; Muto, T.; Hino, R.; Takeuchi, Y.; Fujita, T.; Nakahara, K.; Fukumoto, S.; Yamashita, T. FGF-23 Is a Potent Regulator of Vitamin D Metabolism and Phosphate Homeostasis. J. Bone Miner. Res. 2003, 19, 429–435. [Google Scholar] [CrossRef]
- Ohyama, Y.; Noshiro, M.; Okuda, K. Cloning and expression of cDNA encoding 25-hydroxyvitamin D3 24-hydroxylase. FEBS Lett. 1991, 278, 195–198. [Google Scholar] [CrossRef]
- Houghton, L.A.; Vieth, R. The case against ergocalciferol (vitamin D2) as a vitamin supplement. Am. J. Clin. Nutr. 2006, 84, 694–697. [Google Scholar] [CrossRef]
- Tripkovic, L.; Lambert, H.; Hart, K.; Smith, C.P.; Bucca, G.; Penson, S.; Chope, G.; Hypponen, E.; Berry, J.; Vieth, R.; et al. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2012, 95, 1357–1364. [Google Scholar] [CrossRef]
- Rosen, C.J.; Adams, J.; Bikle, D.D.; Black, D.M.; DeMay, M.B.; Manson, J.E.; Murad, M.H.; Kovacs, C. The Nonskeletal Effects of Vitamin D: An Endocrine Society Scientific Statement. Endocr. Rev. 2012, 33, 456–492. [Google Scholar] [CrossRef] [PubMed]
- Jin, C.H.; Pike, J.W. Human vitamin D receptor-dependent transactivation in Saccharomyces cerevisiae requires retinoid X receptor. Mol. Endocrinol. 1996, 10, 196–205. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bouillon, R.; Carmeliet, G.; Verlinden, L.; van Etten, E.; Verstuyf, A.; Luderer, H.F.; Lieben, L.; Mathieu, C.; DeMay, M. Vitamin D and Human Health: Lessons from Vitamin D Receptor Null Mice. Endocr. Rev. 2008, 29, 726–776. [Google Scholar] [CrossRef]
- Somjen, D.; Weisman, Y.; Kohen, F.; Gayer, B.; Limor, R.; Sharon, O.; Jaccard, N.; Knoll, E.; Stern, N. 25-Hydroxyvitamin D3-1α-Hydroxylase Is Expressed in Human Vascular Smooth Muscle Cells and Is Upregulated by Parathyroid Hormone and Estrogenic Compounds. Circulation 2005, 111, 1666–1671. [Google Scholar] [CrossRef] [PubMed]
- Merke, J.; Milde, P.; Lewicka, S.; Hügel, U.; Klaus, G.; Mangelsdorf, D.; Haussler, M.R.; Rauterberg, E.W.; Ritz, E. Identification and regulation of 1,25-dihydroxyvitamin D3 receptor activity and biosynthesis of 1,25-dihydroxyvitamin D3. Studies in cultured bovine aortic endothelial cells and human dermal capillaries. J. Clin. Investig. 1989, 83, 1903–1915. [Google Scholar] [CrossRef] [PubMed]
- Tishkoff, D.X.; Nibbelink, K.A.; Holmberg, K.H.; Dandu, L.; Simpson, R.U. Functional Vitamin D Receptor (VDR) in the T-Tubules of Cardiac Myocytes: VDR Knockout Cardiomyocyte Contractility. Endocrinology 2007, 149, 558–564. [Google Scholar] [CrossRef] [PubMed]
- Pike, J.W.; Meyer, M. The Vitamin D Receptor: New Paradigms for the Regulation of Gene Expression by 1,25-Dihydroxyvitamin D3. Endocrinol. Metab. Clin. N. Am. 2010, 39, 255–269. [Google Scholar] [CrossRef]
- Masuda, S.; Jones, G. Promise of vitamin D analogues in the treatment of hyperproliferative conditions. Mol. Cancer Ther. 2006, 5, 797–808. [Google Scholar] [CrossRef] [PubMed]
- Gröber, U.; Spitz, J.; Reichrath, J.; Kisters, K.; Holick, M.F. Vitamin D. Dermato-Endocrinology 2013, 5, 331–347. [Google Scholar] [CrossRef]
- Malloy, P.J.; Zhou, Y.; Wang, J.; Hiort, O.; Feldman, D. Hereditary vitamin D-resistant rickets (HVDRR) owing to a heterozygous mutation in the vitamin D receptor. J. Bone Miner. Res. 2011, 26, 2710–2718. [Google Scholar] [CrossRef] [PubMed]
- Strugnell, S.A.; DeLuca, H.F. The vitamin D receptor--structure and transcriptional activation. Proc. Soc. Exp. Boil. Med. 1997, 215, 223–228. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.; Ozono, K.; Sone, T.; McDonnell, D.P.; Pike, J.W. Vitamin D receptor interaction with specific DNA requires a nuclear protein and 1,25-dihydroxyvitamin D3. Proc. Natl. Acad. Sci. USA 1990, 87, 9751–9755. [Google Scholar] [CrossRef] [PubMed]
- Pike, J.W.; Kesterson, R.A.; Scott, R.A. Vitamin D3 receptors: Molecular structure of the protein and its chromosomal gene. In Vitamin D: Molecular, Cellular and Clinical Endocrinology; Norman, A.W., Schaefer, K., Grigoleit, H.G., Eds.; Walter de Gruyter & Co.: Berlin, Germany; New York, NY, USA, 1988. [Google Scholar]
- Zehnder, D.; Bland, R.; Williams, M.C.; McNinch, R.W.; Howie, A.J.; Stewart, P.M.; Hewison, M. Extrarenal Expression of 25-Hydroxyvitamin D3-1α-Hydroxylase1. J. Clin. Endocrinol. Metab. 2001, 86, 888–894. [Google Scholar] [CrossRef]
- Overbergh, L.; Stoffels, K.; Valckx, D.; Giulietti, A.; Bouillon, R.; Mathieu, C. Regulation of 25-hydroxyvitamin d-1α-hydroxylase by IFNγ in human monocytic THP1 cells. J. Steroid Biochem. Mol. Biol. 2004, 89–90, 453–455. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.S.; Singer, F.R.; Gacad, M.A.; Sharma, O.P.; Hayes, M.J.; Vouros, P.; Holick, M.F. Isolation and Structural Identification of 1,25- Dihydroxyvitamin D3Produced by Cultured Alveolar Macrophages in Sarcoidosis. J. Clin. Endocrinol. Metab. 1985, 60, 960–966. [Google Scholar] [CrossRef]
- Prentice, A. Vitamin D deficiency: A global perspective. Nutr. Rev. 2008, 66, S153–S164. [Google Scholar] [CrossRef]
- Daly, R.M.; Gagnon, C.; Lu, Z.X.; Magliano, D.; Dunstan, D.; Sikaris, K.; Zimmet, P.; Ebeling, P.R.; Shaw, J.E. Prevalence of vitamin D deficiency and its determinants in Australian adults aged 25 years and older: A national, population-based study. Clin. Endocrinol. 2011, 77, 26–35. [Google Scholar] [CrossRef]
- Gustafsson, M.K.; Romundstad, P.R.; Stafne, S.N.; Helvik, A.-S.; Stunes, A.K.; Mørkved, S.; Salvesen, K.; Thorsby, P.M.; Syversen, U. Alterations in the vitamin D endocrine system during pregnancy: A longitudinal study of 855 healthy Norwegian women. PLoS ONE 2018, 13, e0195041. [Google Scholar] [CrossRef]
- Holick, M.; Binkley, N.C.; Bischoff-Ferrari, H.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Gröber, U.; Kisters, K. Influence of drugs on vitamin D and calcium metabolism. Dermato-Endocrinology 2012, 4, 158–166. [Google Scholar] [CrossRef] [PubMed]
- Nowson, C.A.; McGrath, J.J.; Ebeling, P.R.; Haikerwal, A.; Daly, R.M.; Sanders, K.M.; Seibel, M.J.; Mason, R.S. Vitamin D and health in adults in Australia and New Zealand: A position statement. Med. J. Aust. 2012, 196, 686–687. [Google Scholar] [CrossRef] [PubMed]
- Food and Nutrition Board IoM. Dietary Reference Intakes for Calcium and Vitamin D.; The National Academic Press: Washington, DC, USA, 2011. [Google Scholar]
- Ross, A.C.; Manson, J.E.; Abrams, S.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.; Jones, G.; et al. The 2011 Report on Dietary Reference Intakes for Calcium and Vitamin D from the Institute of Medicine: What Clinicians Need to Know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Vogiatzi, M.G.; Jacobson-Dickman, E.; DeBoer, M.D. Vitamin D Supplementation and Risk of Toxicity in Pediatrics: A Review of Current Literature. J. Clin. Endocrinol. Metab. 2014, 99, 1132–1141. [Google Scholar] [CrossRef] [PubMed]
- Marcinowska-Suchowierska, E.; Kupisz-Urbańska, M.; Łukaszkiewicz, J.; Pludowski, P.; Jones, G. Vitamin D Toxicity–A Clinical Perspective. Front. Endocrinol. 2018, 9, 550. [Google Scholar] [CrossRef]
- Holick, M.F. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am. J. Clin. Nutr. 2004, 80, 1678S–1688S. [Google Scholar] [CrossRef] [PubMed]
- Adams, J.; Hewison, M. Unexpected actions of vitamin D: New perspectives on the regulation of innate and adaptive immunity. Nat. Clin. Pract. Endocrinol. Metab. 2008, 4, 80–90. [Google Scholar] [CrossRef]
- Kundu, R.; Chain, B.M.; Coussens, A.K.; Khoo, B.; Noursadeghi, M. Regulation of CYP27B1 and CYP24A1 hydroxylases limits cell-autonomous activation of vitamin D in dendritic cells. Eur. J. Immunol. 2014, 44, 1781–1790. [Google Scholar] [CrossRef]
- Gombart, A.F.; Borregaard, N.; Koeffler, H.P. Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D 3. FASEB J. 2005, 19, 1067–1077. [Google Scholar] [CrossRef] [PubMed]
- van Etten, E.; Mathieu, C. Immunoregulation by 1,25-dihydroxyvitamin D3: Basic concepts. J. Steroid Biochem. Mol. Biol. 2005, 97, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Bock, G.; Prietl, B.; Mader, J.K.; Höller, E.; Wolf, M.; Pilz, S.; Graninger, W.B.; Obermayer-Pietsch, B.M.; Pieber, T.R. The effect of vitamin D supplementation on peripheral regulatory T cells and β cell function in healthy humans: A randomized controlled trial. Diabetes/Metab. Res. Rev. 2011, 27, 942–945. [Google Scholar] [CrossRef] [PubMed]
- Cohen-Lahav, M.; Shany, S.; Tobvin, D.; Chaimovitz, C.; Douvdevani, A. Vitamin D decreases NFκB activity by increasing IκBα levels. Nephrol. Dial. Transplant. 2006, 21, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Bao, B.-Y.; Yao, J.; Lee, Y.-F. 1α, 25-dihydroxyvitamin D3 suppresses interleukin-8-mediated prostate cancer cell angiogenesis. Carcinogenesis 2006, 27, 1883–1893. [Google Scholar] [CrossRef] [PubMed]
- Helming, L.; Böse, J.; Ehrchen, J.; Schiebe, S.; Frahm, T.; Geffers, R.; Probst-Kepper, M.; Balling, R.; Lengeling, A. 1α,25-dihydroxyvitamin D3 is a potent suppressor of interferon -mediated macrophage activation. Blood 2005, 106, 4351–4358. [Google Scholar] [CrossRef] [PubMed]
- Noyola-Martínez, N.; Díaz, L.; Zaga-Clavellina, V.; Avila, E.; Halhali, A.; Larrea, F.; Barrera, D. Regulation of CYP27B1 and CYP24A1 gene expression by recombinant pro-inflammatory cytokines in cultured human trophoblasts. J. Steroid Biochem. Mol. Biol. 2014, 144, 106–109. [Google Scholar] [CrossRef]
- Dickie, L.J.; Church, L.D.; Coulthard, L.R.; Mathews, R.; Emery, P.; McDermott, M.F. Vitamin D3 down-regulates intracellular Toll-like receptor 9 expression and Toll-like receptor 9-induced IL-6 production in human monocytes. Rheumatology 2010, 49, 1466–1471. [Google Scholar] [CrossRef]
- Wu, Y.; Craig, T.A.; Lutz, W.H.; Kumar, R. Identification of 1α,25-Dihydroxyvitamin D3 Response Elements in the Human Transforming Growth Factor β2 Gene. Biochemistry 1999, 38, 2654–2660. [Google Scholar] [CrossRef]
- Chen, N.; Wan, Z.; Han, S.-F.; Li, B.-Y.; Zhang, Z.-L.; Qin, L.-Q. Effect of Vitamin D Supplementation on the Level of Circulating High-Sensitivity C-Reactive Protein: A Meta-Analysis of Randomized Controlled Trials. Nutrients 2014, 6, 2206–2216. [Google Scholar] [CrossRef]
- Jiang, W.-L.; Gu, H.-B.; Zhang, Y.-F.; Xia, Q.; Qi, J.; Chen, J.-C. Vitamin D Supplementation in the Treatment of Chronic Heart Failure: A Meta-analysis of Randomized Controlled Trials. Clin. Cardiol. 2015, 39, 56–61. [Google Scholar] [CrossRef]
- Jamka, M.; Wozniewicz, M.; Jeszka, J.; Mardas, M.; Bogdański, P.; Stelmach-Mardas, M. The effect of vitamin D supplementation on insulin and glucose metabolism in overweight and obese individuals: Systematic review with meta-analysis. Sci. Rep. 2015, 5, 16142. [Google Scholar] [CrossRef]
- Mora, J.R.; Iwata, M.; von Andrian, U.H. Vitamin effects on the immune system: Vitamins A and D take centre stage. Nat. Rev. Immunol. 2008, 8, 685–698. [Google Scholar] [CrossRef]
- van Etten, E.; Branisteanu, D.D.; Verstuyf, A.; Waer, M.; Bouillon, R.; Mathieu, C. Analogs of 1,25-dihydroxyvitamin d 3 as dose-reducing agents for classical immunosuppressants12. Transplantation 2000, 69, 1932–1942. [Google Scholar] [CrossRef] [PubMed]
- Krishnan, A.V.; Feldman, D. Mechanisms of the Anti-Cancer and Anti-Inflammatory Actions of Vitamin D. Annu. Rev. Pharmacol. Toxicol. 2011, 51, 311–336. [Google Scholar] [CrossRef]
- Moreno, J.; Krishnan, A.V.; Swami, S.; Nonn, L.; Peehl, D.M.; Feldman, D. Regulation of Prostaglandin Metabolism by Calcitriol Attenuates Growth Stimulation in Prostate Cancer Cells. Cancer Res. 2005, 65, 7917–7925. [Google Scholar] [CrossRef] [PubMed]
- De Haes, P.; Garmyn, M.; Degreef, H.; Vantieghem, K.; Bouillon, R.; Segaert, S. 1,25-dihydroxyvitamin D3 inhibits ultraviolet B-induced apoptosis, Jun kinase activation, and interleukin-6 production in primary human keratinocytes. J. Cell. Biochem. 2003, 89, 663–673. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.H.; Koenig, R.J.; Giordano, T.; Myc, A.; Thompson, N.W.; Baker, J.R. 1α,25-Dihydroxyvitamin D3 Up-Regulates Bcl-2 Expression and Protects Normal Human Thyrocytes from Programmed Cell Death. Endocrinology 1999, 140, 1649–1656. [Google Scholar] [CrossRef]
- Riachy, R.; Vandewalle, B.; Conte, J.K.; Moerman, E.; Sacchetti, P.; Lukowiak, B.; Gmyr, V.; Bouckenooghe, T.; Dubois, M.; Pattou, F. 1,25-Dihydroxyvitamin D3Protects RINm5F and Human Islet Cells against Cytokine-Induced Apoptosis: Implication of the Antiapoptotic Protein A20. Endocrinology 2002, 143, 4809–4819. [Google Scholar] [CrossRef]
- Riachy, R.; Vandewalle, B.; Moerman, E.; Belaich, S.; Lukowiak, B.; Gmyr, V.; Muharram, G.; Conte, J.K.; Pattou, F. 1,25-dihydroxyvitamin D3 protects human pancreatic islets against cytokine-induced apoptosis via down-regulation of the fas receptor. Apoptosis 2006, 11, 151–159. [Google Scholar] [CrossRef]
- Mandarino, N.R.; Júnior, F.D.C.M.; Salgado, J.V.L.; Lages, J.S.; Filho, N.S. Is Vitamin D Deficiency a New Risk Factor for Cardiovascular Disease? Open Cardiovasc. Med. J. 2015, 9, 40–49. [Google Scholar] [CrossRef]
- Anderson, J.L.; May, H.; Horne, B.D.; Bair, T.L.; Hall, N.L.; Carlquist, J.F.; Lappé, D.L.; Muhlestein, J.B. Relation of Vitamin D Deficiency to Cardiovascular Risk Factors, Disease Status, and Incident Events in a General Healthcare Population. Am. J. Cardiol. 2010, 106, 963–968. [Google Scholar] [CrossRef]
- Chen, S.; Sun, Y.; Agrawal, D.K. Vitamin D deficiency and essential hypertension. J. Am. Soc. Hypertens. 2015, 9, 885–901. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Fang, F.; Tang, J.; Jia, L.; Feng, Y.; Xu, P.; Faramand, A. Association between vitamin D supplementation and mortality: Systematic review and meta-analysis. BMJ 2019, 366, l4673. [Google Scholar] [CrossRef]
- Wang, T.J.; Pencina, M.J.; Booth, S.L.; Jacques, P.F.; Ingelsson, E.; Lanier, K.; Benjamin, E.; D’Agostino, R.B.; Wolf, M.; Vasan, R.S. Vitamin D Deficiency and Risk of Cardiovascular Disease. Circulation 2008, 117, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Parker, J.; Hashmi, O.; Dutton, D.; Mavrodaris, A.; Stranges, S.; Kandala, N.-B.; Clarke, A.; Franco, O. Levels of vitamin D and cardiometabolic disorders: Systematic review and meta-analysis. Maturitas 2010, 65, 225–236. [Google Scholar] [CrossRef] [PubMed]
- Liu, D.; Fernandez, B.; Hamilton, A.; Lang, N.; Gallagher, J.M.; Newby, D.E.; Feelisch, M.; Weller, R.B. UVA Irradiation of Human Skin Vasodilates Arterial Vasculature and Lowers Blood Pressure Independently of Nitric Oxide Synthase. J. Investig. Dermatol. 2014, 134, 1839–1846. [Google Scholar] [CrossRef]
- Margolis, K.L.; Ray, R.M.; Van Horn, L.; Manson, J.E.; Allison, M.A.; Black, H.R.; Beresford, S.A.; Connelly, S.A.; Curb, J.D.; Grimm, R.H.; et al. Effect of Calcium and Vitamin D Supplementation on Blood Pressure. Hypertension 2008, 52, 847–855. [Google Scholar] [CrossRef]
- Chen, Y.; Kong, J.; Sun, T.; Li, G.; Szeto, F.L.; Liu, W.; Deb, D.K.; Wang, Y.; Zhao, Q.; Thadhani, R.; et al. 1,25-Dihydroxyvitamin D3 suppresses inflammation-induced expression of plasminogen activator inhibitor-1 by blocking nuclear factor-κB activation. Arch. Biochem. Biophys. 2011, 507, 241–247. [Google Scholar] [CrossRef]
- Wimalawansa, S.J. Associations of vitamin D with insulin resistance, obesity, type 2 diabetes, and metabolic syndrome. J. Steroid Biochem. Mol. Biol. 2018, 175, 177–189. [Google Scholar] [CrossRef]
- Forman, J.P.; Giovannucci, E.; Holmes, M.D.; Bischoff-Ferrari, H.A.; Tworoger, S.S.; Willett, W.C.; Curhan, G.C. Plasma 25-Hydroxyvitamin D Levels and Risk of Incident Hypertension. Hypertension 2007, 49, 1063–1069. [Google Scholar] [CrossRef]
- Giovannucci, E. 25-Hydroxyvitamin D and Risk of Myocardial Infarction in MenA Prospective Study. Arch. Intern. Med. 2008, 168, 1174–1180. [Google Scholar] [CrossRef]
- Garland, C.F.; Kim, J.J.; Mohr, S.B.; Gorham, E.D.; Grant, W.; Giovannucci, E.L.; Baggerly, L.; Hofflich, H.; Ramsdell, J.W.; Zeng, K.; et al. Meta-analysis of All-Cause Mortality According to Serum 25-Hydroxyvitamin D. Am. J. Public Health 2014, 104, e43–e50. [Google Scholar] [CrossRef] [PubMed]
- Husemoen, L.L.N.; Skaaby, T.; Martinussen, T.; Jørgensen, T.; Thuesen, B.H.; Kistorp, C.; Jeppesen, J.; Thyssen, J.P.; Meldgaard, M.; Szecsi, P.B.; et al. Investigating the causal effect of vitamin D on serum adiponectin using a mendelian randomization approach. Eur. J. Clin. Nutr. 2013, 68, 189–195. [Google Scholar] [CrossRef]
- Theodoratou, E.; Tzoulaki, I.; Zgaga, L.; Ioannidis, J.P.A. Vitamin D and multiple health outcomes: Umbrella review of systematic reviews and meta-analyses of observational studies and randomised trials. BMJ 2014, 348, g2035. [Google Scholar] [CrossRef]
- Bjelakovic, G.; Gluud, L.L.; Nikolova, D.; Whitfield, K.; Wetterslev, J.; Simonetti, R.G.; Bjelakovic, M.; Gluud, C. Vitamin D supplementation for prevention of mortality in adults. Cochrane Database Syst. Rev. 2014, 10, CD007470. [Google Scholar] [CrossRef]
- Amrein, K.; Zajic, P.; Schnedl, C.; Waltensdorfer, A.; Fruhwald, S.; Holl, A.; Purkart, T.U.; Wünsch, G.; Valentin, T.; Grisold, A.; et al. Vitamin D status and its association with season, hospital and sepsis mortality in critical illness. Crit. Care 2014, 18, R47. [Google Scholar] [CrossRef]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Zehnder, D.; Bland, R.; Chana, R.S.; Wheeler, D.C.; Howie, A.J.; Williams, M.C.; Stewart, P.M.; Hewison, M. Synthesis of 1,25-Dihydroxyvitamin D3 by Human Endothelial Cells Is Regulated by Inflammatory Cytokines: A Novel Autocrine Determinant of Vascular Cell Adhesion. J. Am. Soc. Nephrol. 2002, 13, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Grundmann, M.; Haidar, M.; Placzko, S.; Niendorf, R.; Darashchonak, N.; Hubel, C.A.; Von Versen-Höynck, F. Vitamin D improves the angiogenic properties of endothelial progenitor cells. Am. J. Physiol. Physiol. 2012, 303, C954–C962. [Google Scholar] [CrossRef]
- Okuda, N.; Takeda, S.; Shinomiya, K.; Muneta, T.; Itoh, S.; Noda, M.; Asou, Y. ED-71, a novel vitamin D analog, promotes bone formation and angiogenesis and inhibits bone resorption after bone marrow ablation. Bone 2007, 40, 281–292. [Google Scholar] [CrossRef] [PubMed]
- Lin, R.; Amizuka, N.; Sasaki, T.; Aarts, M.M.; Ozawa, H.; Goltzman, D.; Henderson, J.E.; White, J.H. 1α,25-Dihydroxyvitamin D3 Promotes Vascularization of the Chondro-osseous Junction by Stimulating Expression of Vascular Endothelial Growth Factor and Matrix Metalloproteinase 9. J. Bone Miner. Res. 2002, 17, 1604–1612. [Google Scholar] [CrossRef] [PubMed]
- Mantell, D.J.; Owens, P.E.; Bundred, N.J.; Mawer, E.B.; Canfield, A.E. 1α,25-Dihydroxyvitamin D 3 Inhibits Angiogenesis In Vitro and In Vivo. Circ. Res. 2000, 87, 214–220. [Google Scholar] [CrossRef] [PubMed]
- Ota, K.; Takahashi, T.; Han, A.R.; Damvaeba, S.; Mizunuma, H.; Kwak-Kim, J. Effects of MTHFR C677T polymorphism on vitamin D, homocysteine and natural killer cell cytotoxicity in women with recurrent pregnancy losses. Hum. Reprod. 2020, 35, 1276–1287. [Google Scholar] [CrossRef]
- Asemi, Z.; Karamali, M.; Esmaillzadeh, A. Effects of calcium–vitamin D co-supplementation on glycaemic control, inflammation and oxidative stress in gestational diabetes: A randomised placebo-controlled trial. Diabetologia 2014, 57, 1798–1806. [Google Scholar] [CrossRef] [PubMed]
- Sharifi, N.; Amani, R.; Hajiani, E.; Cheraghian, B. Does vitamin D improve liver enzymes, oxidative stress, and inflammatory biomarkers in adults with non-alcoholic fatty liver disease? A randomized clinical trial. Endocrine 2014, 47, 70–80. [Google Scholar] [CrossRef]
- Li, Y.C.; Kong, J.; Wei, M.; Chen, Z.-F.; Liu, S.Q.; Cao, L.-P. 1,25-Dihydroxyvitamin D3 is a negative endocrine regulator of the renin-angiotensin system. J. Clin. Investig. 2002, 110, 229–238. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Lu, F.; Cao, K.; Xu, D.; Goltzman, D.; Miao, D. Calcium-independent and 1,25(OH)2D3-dependent regulation of the renin-angiotensin system in 1α-hydroxylase knockout mice. Kidney Int. 2008, 74, 170–179. [Google Scholar] [CrossRef]
- Xiang, W.; Kong, J.; Chen, S.; Cao, L.-P.; Qiao, G.; Zheng, W.; Liu, W.; Li, X.; Gardner, D.G.; Li, Y.C. Cardiac hypertrophy in vitamin D receptor knockout mice: Role of the systemic and cardiac renin-angiotensin systems. Am. J. Physiol. Metab. 2005, 288, E125–E132. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C. Molecular mechanism of vitamin D in the cardiovascular system. J. Investig. Med. 2011, 59, 868–871. [Google Scholar] [CrossRef] [PubMed]
- Yuan, W.; Pan, W.; Kong, J.; Zheng, W.; Szeto, F.L.; Wong, K.E.; Cohen, R.; Klopot, A.; Zhang, Z.; Li, Y.C. 1,25-Dihydroxyvitamin D3 Suppresses Renin Gene Transcription by Blocking the Activity of the Cyclic AMP Response Element in the Renin Gene Promoter. J. Biol. Chem. 2007, 282, 29821–29830. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Wong, S.L.; Lau, C.W.; Lee, H.K.; Ng, C.-F.; Zhang, L.; Yao, X.; Chen, Z.Y.; Vanhoutte, P.M.; Huang, Y. Calcitriol protects renovascular function in hypertension by down-regulating angiotensin II type 1 receptors and reducing oxidative stress. Eur. Heart J. 2012, 33, 2980–2990. [Google Scholar] [CrossRef] [PubMed]
- Christakos, S.; Dhawan, P.; Verstuyf, A.; Verlinden, L.; Carmeliet, G. Vitamin D: Metabolism, Molecular Mechanism of Action, and Pleiotropic Effects. Physiol. Rev. 2016, 96, 365–408. [Google Scholar] [CrossRef]
- Fitzpatrick, L.A.; Bilezikian, J.P.; Silverberg, S.J. Parathyroid hormone and the cardiovascular system. Curr. Osteoporos. Rep. 2008, 6, 77–83. [Google Scholar] [CrossRef] [PubMed]
- Rutledge, M.R.; Farah, V.; Adeboye, A.A.; Seawell, M.R.; Bhattacharya, S.K.; Weber, K.T. Parathyroid Hormone, A Crucial Mediator of Pathologic Cardiac Remodeling in Aldosteronism. Cardiovasc. Drugs Ther. 2012, 27, 161–170. [Google Scholar] [CrossRef]
- Tiosano, D.; Gepstein, V. Vitamin D action. Curr. Opin. Endocrinol. Diabetes Obes. 2012, 19, 452–459. [Google Scholar] [CrossRef]
- Best, C.; Pressman, E.K.; Queenan, R.A.; Cooper, E.; O’Brien, K.O. Longitudinal changes in serum vitamin D binding protein and free 25-hydroxyvitamin D in a multiracial cohort of pregnant adolescents. J. Steroid Biochem. Mol. Biol. 2018, 186, 79–88. [Google Scholar] [CrossRef]
- Bikle, D.D.; Gee, E.; Halloran, B.; Haddad, J.G. Free 1,25-dihydroxyvitamin D levels in serum from normal subjects, pregnant subjects, and subjects with liver disease. J. Clin. Investig. 1984, 74, 1966–1971. [Google Scholar] [CrossRef] [PubMed]
- Seely, E.W.; Brown, E.M.; DeMaggio, D.M.; Weldon, D.K.; Graves, S.W. A prospective study of calciotropic hormones in pregnancy and post partum: Reciprocal changes in serum intact parathyroid hormone and 1,25-dihydroxyvitamin D. Am. J. Obstet. Gynecol. 1997, 176, 214–217. [Google Scholar] [CrossRef]
- Ardawi, M.S.; Nasrat, H.A.; Ba’Aqueel, H.S. Calcium-regulating hormones and parathyroid hormone-related peptide in normal human pregnancy and postpartum: A longitudinal study. Eur. J. Endocrinol. 1997, 137, 402–409. [Google Scholar] [CrossRef]
- Olmos-Ortiz, A.; Avila, E.; Durand-Carbajal, M.; Díaz, L. Regulation of Calcitriol Biosynthesis and Activity: Focus on Gestational Vitamin D Deficiency and Adverse Pregnancy Outcomes. Nutrients 2015, 7, 443–480. [Google Scholar] [CrossRef]
- Haddow, J.E.; Neveux, L.M.; Palomaki, G.E.; Lambert-Messerlian, G.; Canick, J.A.; Grenache, D.G.; Lu, J. The relationship between PTH and 25-hydroxy vitamin D early in pregnancy. Clin. Endocrinol. 2011, 75, 309–314. [Google Scholar] [CrossRef]
- Knabl, J.; Vattai, A.; Ye, Y.; Jueckstock, J.; Hutter, S.; Kainer, F.; Mahner, S.; Jeschke, U. Role of Placental VDR Expression and Function in Common Late Pregnancy Disorders. Int. J. Mol. Sci. 2017, 18, 2340. [Google Scholar] [CrossRef]
- Chary, A.V.; Hemalatha, R.; Seshacharyulu, M.; Murali, M.V.; Jayaprakash, D.; Kumar, B.D. Vitamin D deficiency in pregnant women impairs regulatory T cell function. J. Steroid Biochem. Mol. Biol. 2015, 147, 48–55. [Google Scholar] [CrossRef]
- Zhang, J.Y.; Lucey, A.J.; Horgan, R.; Kenny, L.C.; Kiely, M. Impact of pregnancy on vitamin D status: A longitudinal study. Br. J. Nutr. 2014, 112, 1081–1087. [Google Scholar] [CrossRef] [PubMed]
- Heijboer, A.C.; Blankenstein, M.; Kema, I.P.; Buijs, M.M. Accuracy of 6 Routine 25-Hydroxyvitamin D Assays: Influence of Vitamin D Binding Protein Concentration. Clin. Chem. 2012, 58, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Karras, S.N.; Kotsa, K.; Angeloudi, E.; Zebekakis, P.; Naughton, D.P. The Road Not So Travelled: Should Measurement of Vitamin D Epimers during Pregnancy Affect Our Clinical Decisions? Nutrients 2017, 9, 90. [Google Scholar] [CrossRef] [PubMed]
- Liong, S.; Di Quinzio, M.; Fleming, G.; Permezel, M.; Rice, G.E.; Georgiou, H. New biomarkers for the prediction of spontaneous preterm labour in symptomatic pregnant women: A comparison with fetal fibronectin. BJOG Int. J. Obstet. Gynaecol. 2014, 122, 370–379. [Google Scholar] [CrossRef]
- Xia, J.; Song, Y.; Rawal, S.; Wu, J.; Hinkle, S.; Tsai, M.Y.; Zhang, C. Vitamin D status during pregnancy and the risk of gestational diabetes mellitus: A longitudinal study in a multiracial cohort. Diabetes Obes. Metab. 2019, 21, 1895–1905. [Google Scholar] [CrossRef]
- Kaludjerovic, J.; Vieth, R. Relationship Between Vitamin D During Perinatal Development and Health. J. Midwifery Women’s Health 2010, 55, 550–560. [Google Scholar] [CrossRef]
- Liu, N.Q.; Kaplan, A.; Lagishetty, V.; Ouyang, Y.B.; Ouyang, Y.; Simmons, C.F.; Equils, O.; Hewison, M. Vitamin D and the Regulation of Placental Inflammation. J. Immunol. 2011, 186, 5968–5974. [Google Scholar] [CrossRef] [PubMed]
- Tamblyn, J.A.; Hewison, M.; Wagner, C.L.; Bulmer, J.N.; Kilby, M.D. Immunological role of vitamin D at the maternal–fetal interface. J. Endocrinol. 2015, 224, R107–R121. [Google Scholar] [CrossRef]
- Zahran, A.M.; Zharan, K.M.; Hetta, H.F. Significant correlation between regulatory T cells and vitamin D status in term and preterm labor. J. Reprod. Immunol. 2018, 129, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Sharif, K.; Sharif, Y.; Watad, A.; Yavne, Y.; Lichtbroun, B.; Bragazzi, N.L.; Amital, H.; Shoenfeld, Y. Vitamin D, autoimmunity and recurrent pregnancy loss: More than an association. Am. J. Reprod. Immunol. 2018, 80, e12991. [Google Scholar] [CrossRef]
- van der Steeg, J.W.; Steures, P.; Eijkemans, M.J.; Habbema, J.D.F.; Hompes, P.G.; Burggraaff, J.M.; Oosterhuis, G.J.E.; Bossuyt, P.M.; van der Veen, F.; Mol, B.W. Obesity affects spontaneous pregnancy chances in subfertile, ovulatory women. Hum. Reprod. 2007, 23, 324–328. [Google Scholar] [CrossRef] [PubMed]
- Hollist, B.W.; Pittard, W.B. Evaluation of the Total Fetomaternal Vitamin D Relationships at Term: Evidence for Racial Differences. J. Clin. Endocrinol. Metab. 1984, 59, 652–657. [Google Scholar] [CrossRef]
- Kovacs, C. Bone Development and Mineral Homeostasis in the Fetus and Neonate: Roles of the Calciotropic and Phosphotropic Hormones. Physiol. Rev. 2014, 94, 1143–1218. [Google Scholar] [CrossRef]
- Dror, D.K.; Allen, L.H. Vitamin D inadequacy in pregnancy: Biology, outcomes, and interventions. Nutr. Rev. 2010, 68, 465–477. [Google Scholar] [CrossRef]
- Shand, A.; Nassar, N.; Von Dadelszen, P.; Innis, S.; Green, T. Maternal vitamin D status in pregnancy and adverse pregnancy outcomes in a group at high risk for pre-eclampsia. BJOG Int. J. Obstet. Gynaecol. 2010, 117, 1593–1598. [Google Scholar] [CrossRef]
- Finch, P.J.; Ang, L.; Colston, K.W.; Nisbet, J.; Maxwell, J.D. Blunted seasonal variation in serum 25-hydroxy vitamin D and in-creased risk of osteomalacia in vegetarian London Asians. Eur. J. Clin. Nutr. 1992, 46, 509–515. [Google Scholar]
- Nesby-O’Dell, S.; Scanlon, K.S.; Cogswell, M.E.; Gillespie, C.; Hollis, B.W.; Looker, A.C.; Allen, C.; Doughertly, C.; Gunter, E.W.; Bowman, B.A. Hypovitaminosis D prevalence and determinants among African American and white women of reproductive age: Third National Health and Nutrition Examination Survey, 1988–1994. Am. J. Clin. Nutr. 2002, 76, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Anastasiadis, P.; Tsikouras, P.; Galazios, G.; Liberis, V.; Grapsas, X.; Koutlaki, N.; Anastasiadis, A.F. Hypertensive disorders in pregnancy: Risk factors and epidemiologic analysis. Clin. Exp. Obstet. Gynecol. 2007, 34, 154–158. [Google Scholar] [PubMed]
- Zhang, M.; Wan, P.; Ng, K.; Singh, K.; Cheng, T.H.; Velickovic, I.; Dalloul, M.; Wlody, D. Preeclampsia Among African American Pregnant Women: An Update on Prevalence, Complications, Etiology, and Biomarkers. Obstet. Gynecol. Surv. 2020, 75, 111–120. [Google Scholar] [CrossRef] [PubMed]
- Nakimuli, A.; Chazara, O.; Byamugisha, J.; Elliott, A.M.; Kaleebu, P.; Mirembe, F.; Moffett, A. Pregnancy, parturition and preeclampsia in women of African ancestry. Am. J. Obstet. Gynecol. 2013, 210, 510–520. [Google Scholar] [CrossRef]
- Mousa, A.; Abell, S.; Scragg, R.; De Courten, B. Vitamin D in Reproductive Health and Pregnancy. Semin. Reprod. Med. 2016, 34, e1–e13. [Google Scholar] [CrossRef]
- Zimmer, M.; Sieroszewski, P.; Oszukowski, P.; Huras, H.; Fuchs, T.; Pawlosek, A. Polish Society of Gynecologists and Obstetricians recommendations on supplementation during pregnancy. Ginekol Pol. 2020, 91, 644–653. [Google Scholar] [CrossRef]
- Daniel, D.; Hardigan, P.; Bray, N.; Penzell, D.; Savu, C. The incidence of vitamin D deficiency in the obese: A retrospective chart review. J. Community Hosp. Intern. Med. Perspect. 2015, 5, 26069. [Google Scholar] [CrossRef]
- Parva, N.R.; Tadepalli, S.; Singh, P.; Qian, A.; Joshi, R.; Kandala, H.; Nookala, V.K.; Cheriyath, P. Prevalence of Vitamin D Deficiency and Associated Risk Factors in the US Population (2011–2012). Cureus 2018, 10, e2741. [Google Scholar] [CrossRef]
- Hollis, B.W.; Wagner, C.L. New insights into the vitamin D requirements during pregnancy. Bone Res. 2017, 5, 17030. [Google Scholar] [CrossRef]
- McDonnell, S.L.; Baggerly, K.A.; Baggerly, C.A.; Aliano, J.L.; French, C.B.; Baggerly, L.L.; Ebeling, M.D.; Rittenberg, C.S.; Goodier, C.G.; Niño, J.F.M.; et al. Maternal 25(OH)D concentrations ≥ 40 ng/mL associated with 60% lower preterm birth risk among general obstetrical patients at an urban medical center. PLoS ONE 2017, 12, e0180483. [Google Scholar] [CrossRef]
- Mumford, S.L.; Garbose, R.A.; Kim, K.; Kissell, K.; Kuhr, D.L.; Omosigho, U.R.; Perkins, N.; Galai, N.; Silver, R.M.; Sjaarda, L.A.; et al. Association of preconception serum 25-hydroxyvitamin D concentrations with livebirth and pregnancy loss: A prospective cohort study. Lancet Diabetes Endocrinol. 2018, 6, 725–732. [Google Scholar] [CrossRef]
- Weiss, S.T.; Litonjua, A.A. Vitamin D in Host Defense: Implications for Future Research. Am. J. Respir. Cell Mol. Biol. 2017, 56, 692–693. [Google Scholar] [CrossRef]
- Mirzakhani, H.; Litonjua, A.A.; McElrath, T.F.; O’Connor, G.; Lee-Parritz, A.; Iverson, R.; Macones, G.; Strunk, R.C.; Bacharier, L.B.; Zeiger, R.; et al. Early pregnancy vitamin D status and risk of preeclampsia. J. Clin. Investig. 2016, 126, 4702–4715. [Google Scholar] [CrossRef]
- Palacios, C.; Kostiuk, L.K.; Peña-Rosas, J.P. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst. Rev. 2019, 7, CD008873. [Google Scholar] [CrossRef]
- Harvey, N.; Holroyd, C.; Ntani, G.; Javaid, M.; Cooper, P.; Moon, R.; Cole, Z.; Tinati, T.; Godfrey, K.; Dennison, E.; et al. Vitamin D supplementation in pregnancy: A systematic review. Health Technol. Assess. 2014, 18, 1–190. [Google Scholar] [CrossRef] [PubMed]
- Khaing, W.; Vallibhakara, S.A.-O.; Tantrakul, V.; Vallibhakara, O.; Rattanasiri, S.; McEvoy, M.; Attia, J.; Thakkinstian, A. Calcium and Vitamin D Supplementation for Prevention of Preeclampsia: A Systematic Review and Network Meta-Analysis. Nutrients 2017, 9, 1141. [Google Scholar] [CrossRef]
- Munns, C.F.; Shaw, N.; Kiely, M.; Specker, B.L.; Thacher, T.; Ozono, K.; Michigami, T.; Tiosano, D.; Mughal, M.Z.; Mäkitie, O.; et al. Global Consensus Recommendations on Prevention and Management of Nutritional Rickets. J. Clin. Endocrinol. Metab. 2016, 101, 394–415. [Google Scholar] [CrossRef] [PubMed]
- Kiely, M.E.; Zhang, J.Y.; Kinsella, M.; Khashan, A.S.; Kenny, L.C. Vitamin D status is associated with uteroplacental dysfunction indicated by pre-eclampsia and small-for-gestational-age birth in a large prospective pregnancy cohort in Ireland with low vitamin D status. Am. J. Clin. Nutr. 2016, 104, 354–361. [Google Scholar] [CrossRef]
- O’Callaghan, K.M.; Hennessy, Á.; Hull, G.; Healy, K.; Ritz, C.; Kenny, L.C.; Cashman, K.D.; Kiely, M.E. Estimation of the maternal vitamin D intake that maintains circulating 25-hydroxyvitamin D in late gestation at a concentration sufficient to keep umbilical cord sera ≥ 25–30 nmol/L: A dose-response, double-blind, randomized placebo-controlled trial in pregnant women at northern latitude. Am. J. Clin. Nutr. 2018, 108, 77–91. [Google Scholar] [CrossRef] [PubMed]
- Hemmingway, A.; Kenny, L.C.; Malvisi, L.; Kiely, M.E. Exploring the concept of functional vitamin D deficiency in pregnancy: Impact of the interaction between 25-hydroxyvitamin D and parathyroid hormone on perinatal outcomes. Am. J. Clin. Nutr. 2018, 108, 821–829. [Google Scholar] [CrossRef] [PubMed]
- Bodnar, L.M.; Simhan, H.N.; Powers, R.W.; Frank, M.P.; Cooperstein, E.; Roberts, J.M. High Prevalence of Vitamin D Insufficiency in Black and White Pregnant Women Residing in the Northern United States and Their Neonates. J. Nutr. 2007, 137, 447–452. [Google Scholar] [CrossRef]
- Hollis, B.W.; Johnson, D.; Hulsey, T.C.; Ebeling, M.; Wagner, C.L. Vitamin D supplementation during pregnancy: Double-blind, randomized clinical trial of safety and effectiveness. J. Bone Miner. Res. 2011, 26, 2341–2357. [Google Scholar] [CrossRef] [PubMed]
- Roth, D.E.; Perumal, N.; Al Mahmud, A.; Baqui, A.H. Maternal Vitamin D3 Supplementation during the Third Trimester of Pregnancy: Effects on Infant Growth in a Longitudinal Follow-Up Study in Bangladesh. J. Pediatr. 2013, 163, 1605–1611. [Google Scholar] [CrossRef] [PubMed]
- Sablok, A.; Batra, A.; Thariani, K.; Batra, A.; Bharti, R.; Aggarwal, A.R.; Kabi, B.; Chellani, H. Supplementation of vitamin D in pregnancy and its correlation with feto-maternal outcome. Clin. Endocrinol. 2015, 83, 536–541. [Google Scholar] [CrossRef] [PubMed]
- Friedman, W.F.; Roberts, W.C. Vitamin D and the Supravalvar Aortic Stenosis Syndrome. Circulation 1966, 34, 77–86. [Google Scholar] [CrossRef]
- Akbari, S.; Khodadadi, B.; Ahmadi, S.A.Y.; Abbaszadeh, S.; Shahsavar, F. Association of vitamin D level and vitamin D deficiency with risk of preeclampsia: A systematic review and updated meta-analysis. Taiwan J. Obstet. Gynecol. 2018, 57, 241–247. [Google Scholar] [CrossRef]
- Baca, K.M.; Govil, M.; Zmuda, J.M.; Simhan, H.N.; Marazita, M.L.; Bodnar, L.M. Vitamin D metabolic loci and preeclampsia risk in multi-ethnic pregnant women. Physiol. Rep. 2018, 6, e13468. [Google Scholar] [CrossRef]
- Robinson, C.J.; Alanis, M.C.; Wagner, C.L.; Hollis, B.W.; Johnson, D.D. Plasma 25-hydroxyvitamin D levels in early-onset severe preeclampsia. Am. J. Obstet. Gynecol. 2010, 203, 366.e1–366.e6. [Google Scholar] [CrossRef] [PubMed]
- Rezavand, N.; Tabarok, S.; Rahimi, Z.; Vaisi-Raygani, A.; Mohammadi, E.; Rahimi, Z. The effect of VDR gene polymorphisms and vitamin D level on blood pressure, risk of preeclampsia, gestational age, and body mass index. J. Cell. Biochem. 2018, 120, 6441–6448. [Google Scholar] [CrossRef]
- Ganguly, A.; Tamblyn, J.A.; Finn-Sell, S.; Chan, S.; Westwood, M.; Gupta, J.; Kilby, M.D.; Gross, S.; Hewison, M. Vitamin D, the placenta and early pregnancy: Effects on trophoblast function. J. Endocrinol. 2018, 236, R93–R103. [Google Scholar] [CrossRef]
- Hollis, B.W. Vitamin D status during pregnancy: The importance of getting it right. EBioMedicine 2018, 39, 23–24. [Google Scholar] [CrossRef]
- Barrera, D.; Avila, E.; Hernández, G.; Méndez, I.; González, L.; Halhali, A.; Larrea, F.; Morales, A.; Díaz, L. Calcitriol affects hCG gene transcription in cultured human syncytiotrophoblasts. Reprod. Biol. Endocrinol. 2008, 6, 3. [Google Scholar] [CrossRef] [PubMed]
- Weisman, Y.; Harell, A.; Edelstein, S.; David, M.; Spirer, Z.; Golander, A. 1α, 25-Dihydroxyvitamin D3, and 24,25-dihydroxyvitamin D3 in vitro synthesis by human decidua and placenta. Nature 1979, 281, 317–319. [Google Scholar] [CrossRef]
- Díaz, L.; Arranz, C.; Avila, E.; Halhali, A.; Vilchis, F.; Larrea, F. Expression and Activity of 25-Hydroxyvitamin D-1?-Hydroxylase Are Restricted in Cultures of Human Syncytiotrophoblast Cells from Preeclamptic Pregnancies. J. Clin. Endocrinol. Metab. 2002, 87, 3876–3882. [Google Scholar] [CrossRef] [PubMed]
- Chan, S.; Susarla, R.; Canovas, D.; Vasilopoulou, E.; Ohizua, O.; McCabe, C.; Hewison, M.; Kilby, M. Vitamin D promotes human extravillous trophoblast invasion in vitro. Placenta 2015, 36, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.; Gu, Y.; Zhao, S.; Sun, J.; Groome, L.J.; Wang, Y. Expressions of vitamin D metabolic components VDBP, CYP2R1, CYP27B1, CYP24A1, and VDR in placentas from normal and preeclamptic pregnancies. Am. J. Physiol. Metab. 2012, 303, E928–E935. [Google Scholar] [CrossRef]
- Zabul, P.; Wozniak, M.; Slominski, A.T.; Preis, K.; Gorska, M.; Korozan, M.; Wieruszewski, J.; Zmijewski, M.A.; Zabul, E.; Tuckey, R.; et al. A Proposed Molecular Mechanism of High-Dose Vitamin D3 Supplementation in Prevention and Treatment of Preeclampsia. Int. J. Mol. Sci. 2015, 16, 13043–13064. [Google Scholar] [CrossRef]
- Sasan, S.B.; Zandvakili, F.; Soufizadeh, N.; Baybordi, E. The Effects of Vitamin D Supplement on Prevention of Recurrence of Preeclampsia in Pregnant Women with a History of Preeclampsia. Obstet. Gynecol. Int. 2017, 2017, 8249264. [Google Scholar] [CrossRef]
- Reynolds, J.; Ray, D.; Alexander, M.Y.; Bruce, I. Role of vitamin D in endothelial function and endothelial repair in clinically stable systemic lupus erythematosus. Lancet 2015, 385, S83. [Google Scholar] [CrossRef]
- Kanikarla, P.; Jain, S.K. 1,25(OH) 2 D 3 inhibits oxidative stress and monocyte adhesion by mediating the upregulation of GCLC and GSH in endothelial cells treated with acetoacetate (ketosis). J. Steroid Biochem. Mol. Biol. 2016, 159, 94–101. [Google Scholar] [CrossRef]
- Schulz, E.V.; Cruze, L.; Wei, W.; Gehris, J.; Wagner, C.L. Maternal vitamin D sufficiency and reduced placental gene expression in angiogenic biomarkers related to comorbidities of pregnancy. J. Steroid Biochem. Mol. Biol. 2017, 173, 273–279. [Google Scholar] [CrossRef]
- Cardus, A.; Panizo, S.; Encinas, M.; Dolcet, X.; Gallego, C.; Aldea, M.; Fernandez, E.; Valdivielso, J.M. 1,25-Dihydroxyvitamin D3 regulates VEGF production through a vitamin D response element in the VEGF promoter. Atherosclerosis 2009, 204, 85–89. [Google Scholar] [CrossRef]
- Jamali, N.; Song, Y.; Sorenson, C.M.; Sheibani, N. 1,25(OH)2D3 regulates the proangiogenic activity of pericyte through VDR-mediated modulation of VEGF production and signaling of VEGF and PDGF receptors. FASEB BioAdv. 2019, 1, 415–434. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.; Gu, B.; Gu, Y.; Groome, L.J.; Sun, J.; Wang, Y. Activation of vitamin D receptor promotes VEGF and CuZn-SOD expression in endothelial cells. J. Steroid Biochem. Mol. Biol. 2013, 140, 56–62. [Google Scholar] [CrossRef]
- von Versen-Höynck, F.; Brodowski, L.; Dechend, R.; Myerski, A.C.; Hubel, C.A. Vitamin D Antagonizes Negative Effects of Preeclampsia on Fetal Endothelial Colony Forming Cell Number and Function. PLoS ONE 2014, 9, e98990. [Google Scholar] [CrossRef] [PubMed]
- Brodowski, L.; Burlakov, J.; Myerski, A.C.; Von Kaisenberg, C.S.; Grundmann, M.; Hubel, C.A.; Von Versen-Höynck, F. Vitamin D Prevents Endothelial Progenitor Cell Dysfunction Induced by Sera from Women with Preeclampsia or Conditioned Media from Hypoxic Placenta. PLoS ONE 2014, 9, e98527. [Google Scholar] [CrossRef] [PubMed]
- Evans, K.N.; Bulmer, J.N.; Kilby, M.D.; Hewison, M. Vitamin D and Placental-Decidual Function. J. Soc. Gynecol. Investig. 2004, 11, 263–271. [Google Scholar] [CrossRef] [PubMed]
- Piccinni, M.P. Role of hormone-controlled Th1- and Th2-type cytokines in successful pregnancy. J. Neuroimmunol. 2000, 109, 30–33. [Google Scholar] [CrossRef]
- Noyola-Martínez, N.; Díaz, L.; Avila, E.; Halhali, A.; Larrea, F.; Barrera, D. Calcitriol downregulates TNF-α and IL-6 expression in cultured placental cells from preeclamptic women. Cytokine 2013, 61, 245–250. [Google Scholar] [CrossRef]
- Xu, L.; Lee, M.; Jeyabalan, A.; Roberts, J.M. The relationship of hypovitaminosis D and IL-6 in preeclampsia. Am. J. Obstet. Gynecol. 2013, 210, 149.e1–149.e7. [Google Scholar] [CrossRef]
- Thorne-Lyman, A.; Fawzi, W.W. Vitamin D During Pregnancy and Maternal, Neonatal and Infant Health Outcomes: A Systematic Review and Meta-analysis. Paediatr. Périnat. Epidemiol. 2012, 26, 75–90. [Google Scholar] [CrossRef] [PubMed]
- Rostami, M.; Tehrani, F.R.; Simbar, M.; Yarandi, R.B.; Minooee, S.; Hollis, B.W.; Hosseinpanah, F. Effectiveness of Prenatal Vitamin D Deficiency Screening and Treatment Program: A Stratified Randomized Field Trial. J. Clin. Endocrinol. Metab. 2018, 103, 2936–2948. [Google Scholar] [CrossRef] [PubMed]
- Karamali, M.; Beihaghi, E.; Mohammadi, A.A.; Asemi, Z. Effects of High-Dose Vitamin D Supplementation on Metabolic Status and Pregnancy Outcomes in Pregnant Women at Risk for Pre-Eclampsia. Horm. Metab. Res. 2015, 47, 867–872. [Google Scholar] [CrossRef] [PubMed]
- Ali, A.M.; Alobaid, A.; Malhis, T.N.; Khattab, A.F. Effect of vitamin D3 supplementation in pregnancy on risk of pre-eclampsia–Randomized controlled trial. Clin. Nutr. 2018, 38, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Palacios, C.; De-Regil, L.M.; Lombardo, L.K.; Peña-Rosas, J.P. Vitamin D supplementation during pregnancy: Updated meta-analysis on maternal outcomes. J. Steroid Biochem. Mol. Biol. 2016, 164, 148–155. [Google Scholar] [CrossRef]
- Fogacci, S.; Fogacci, F.; Banach, M.; Michos, E.D.; Hernandez, A.V.; Lip, G.Y.; Blaha, M.J.; Toth, P.P.; Borghi, C.; Cicero, A.F. Vitamin D supplementation and incident preeclampsia: A systematic review and meta-analysis of randomized clinical trials. Clin. Nutr. 2019, 39, 1742–1752. [Google Scholar] [CrossRef]
- Gallo, S.; McDermid, J.M.; Al-Nimr, R.I.; Hakeem, R.; Moreschi, J.M.; Pari-Keener, M.; Stahnke, B.; Papoutsakis, C.; Handu, D.; Cheng, F.W. Vitamin D Supplementation during Pregnancy: An Evidence Analysis Center Systematic Review and Meta-Analysis. J. Acad. Nutr. Diet. 2019, 120, 898–924. [Google Scholar] [CrossRef]
- Pérez-López, F.R.; Pasupuleti, V.; Mezones-Holguin, E.; Zapata, V.A.B.; Thota, P.; Deshpande, A.; Hernandez, A.V. Effect of vitamin D supplementation during pregnancy on maternal and neonatal outcomes: A systematic review and meta-analysis of randomized controlled trials. Fertil. Steril. 2015, 103, 1278–1288. [Google Scholar] [CrossRef] [PubMed]
- Roth, D.E.; Leung, M.; Mesfin, E.; Qamar, H.; Watterworth, J.; Papp, E. Vitamin D supplementation during pregnancy: State of the evidence from a systematic review of randomised trials. BMJ 2017, 359, j5237. [Google Scholar] [CrossRef]
- Aguilar-Cordero, M.; Lasserrot-Cuadrado, A.; Mur-Villar, N.; Rios, X.A.L.; Rivero-Blanco, T.; Pérez-Castillo, I.M. Vitamin D, preeclampsia and prematurity: A systematic review and meta-analysis of observational and interventional studies. Midwifery 2020, 87, 102707. [Google Scholar] [CrossRef]
- Fu, Z.-M.; Ma, Z.-Z.; Liu, G.-J.; Wang, L.-L.; Guo, Y. Vitamins supplementation affects the onset of preeclampsia. J. Formos. Med. Assoc. 2018, 117, 6–13. [Google Scholar] [CrossRef] [PubMed]
- Hyppönen, E.; Cavadino, A.; Williams, D.; Fraser, A.; Vereczkey, A.; Fraser, W.D.; Bánhidy, F.; Lawlor, D.; Czeizel, A.E. Vitamin D and Pre-Eclampsia: Original Data, Systematic Review and Meta-Analysis. Ann. Nutr. Metab. 2013, 63, 331–340. [Google Scholar] [CrossRef]
- Aghajafari, F.; Nagulesapillai, T.; Ronksley, P.; Tough, S.C.; O’Beirne, M.; Rabi, D.M. Association between maternal serum 25-hydroxyvitamin D level and pregnancy and neonatal outcomes: Systematic review and meta-analysis of observational studies. BMJ 2013, 346, f1169. [Google Scholar] [CrossRef]
- Tabesh, M.; Salehi-Abargouei, A.; Tabesh, M.; Esmaillzadeh, A. Maternal Vitamin D Status and Risk of Pre-Eclampsia: A Systematic Review and Meta-Analysis. J. Clin. Endocrinol. Metab. 2013, 98, 3165–3173. [Google Scholar] [CrossRef]
- Martínez-Domínguez, S.J.; Tajada, M.; Chedraui, P.; Pérez-López, F.R. Systematic review and meta-analysis of Spanish studies regarding the association between maternal 25-hydroxyvitamin D levels and perinatal outcomes. Gynecol. Endocrinol. 2018, 34, 987–994. [Google Scholar] [CrossRef] [PubMed]
- Kinshella, M.-L.; Omar, S.; Scherbinsky, K.; Vidler, M.; Magee, L.; von Dadelszen, P.; Moore, S.; Elango, R. The PRECISE Conceptual Framework Working Group Effects of Maternal Nutritional Supplements and Dietary Interventions on Placental Complications: An Umbrella Review, Meta-Analysis and Evidence Map. Nutrients 2021, 13, 472. [Google Scholar] [CrossRef] [PubMed]
- Yuan, Y.; Tai, W.; Xu, P.; Fu, Z.; Wang, X.; Long, W.; Guo, X.; Ji, C.; Zhang, L.; Zhang, Y.; et al. Association of maternal serum 25-hydroxyvitamin D concentrations with risk of preeclampsia: A nested case-control study and meta-analysis. J. Matern. Neonatal Med. 2019, 34, 1576–1585. [Google Scholar] [CrossRef]
| Appropriate Vit D Level | ≥50 nmol/L |
|---|---|
| Vit D deficiency—mild | 30–49 nmol/L |
| Vit D deficiency—moderate | 12.5–29 nmol/L |
| Vit D deficiency—severe | <12.5 nmol/L |
| Recommended Daily Dose of Vitamin D (IU) | Minimal Vit D (25(OH)D) Level (nmol/L) | |
|---|---|---|
| WHO | 200 | >50 |
| Institute of Medicine (USA) | 600–1000 | ≥30 |
| Endocrine Society (USA) | 1500–2000 | ≥75 |
| ACOG (USA) | 600 | ≥50 |
| NICE (UK) | 400–800 | >30 |
| RANZCOG | 400–2000 | >50 |
| PTGiP | 1500–2000 BMI > 30 kg/m2–up to 4000 | No data |
| Author | Aim of the Study | Size of Groups | Vit D Dose (IU) and Duration of Treatment | GA at the Entry to the Study | Main Outcome |
|---|---|---|---|---|---|
| Mirzakhani et al. 2016 [217] | PE risk | Vit D (SG) 408 CG 408 | 4400 daily 400 daily | 10–18th week | PE incidence SG 8.08% CG 8.33%, NS RR 0.97 95% CI: 0.61–1.53 |
| Rostami et al. 2018 [256] | Vit D status screening | Screened Vit D 800 Without Vit D 200 Non screened 900 | 50,000–300,000 weekly or monthly; 6–12 weeks | <14th week | Screening reduces PE risk by 60% RR 0.40 95% CI: 0.30–0.60 |
| Karamali et al. 2015 [257] | PE risk | Vit D (SG) 30 CG 30 patients with high PE risk | 50,000 every 2 weeks | 20–32nd week | PE incidence SG 3.3% CG 10% p = 0.3 |
| Sablok et al. 2015 [228] | Pregnancy complication risk | Vit D (SG) 120 CG 60 | 60,000–120,000 every 4 weeks | 20–32nd week | PE incidence SG 11.1% CG 21.1% p = 0.08 |
| Ali et al. 2019 [258] | PE risk | Vit D (SG) 83 CG 81 | 4000 daily | at 13th week up to 12th week after delivery | PE incidence SG 1.2% CG 7.4% p = 0.049 |
| Authors | Studied Group | Number of Participants | Impact on PE | Additional Information |
|---|---|---|---|---|
| Khaing et al. 2017 [220] | Vit D vs. placebo | 357 | RR 0.47 95% CI: 0.24–0.89 | NNT 17 |
| Palacios et al. 2016 [259] | Vit D vs. no treatment | 219 | RR 0.52 95% CI: 0.25–1.05 | PE occurrence 8.9% vs. 15.5% |
| Palacios et al. 2019 [218] | Vit D vs. no treatment | 499 | RR 0.48 95% CI: 0.30–0.79 | |
| Fogacci et al. 2020 [260] | Vit D vs. no treatment Vit D vs. no treatment < 20th week | 4777 | RR 0.37 95% CI: 0.26–0.52 RR 0.35 95% CI: 0.24–0.50, p < 0.001 | Increasing dose–decreasing PE risk RR −1.10 95% CI: −1.73–1.46, p < 0.001 |
| Gallo et al. 2020 [261] | Vit D vs. no treatment | 364 | PE RR 0.7 95% CI: 0.4–1.4, NS GH RR 0.8 95% CI: 0.3–2.2, NS | |
| Pérez-López et al. 2015 [262] | Vit D vs. placebo | 877 | RR 0.88 95% CI: 0.51–1.52, NS | |
| Roth et al. 2017 [263] | Vit D vs. no treatment | 3398 | RR 1.09 95% CI:0.43–2.76, NS | |
| Aguilar-Cordero et al. 2020 [264] | Random effects meta-analysis 25(OH)D < 75 nmol/L 25(OH)D < 50 nmol/L Fixed effect meta-analysis 25(OH)D < 75 nmol/L 25(OH)D < 50 nmol/L Interventional studies Vit D supplementation | 10,979 14,496 10,979 14,469 1660 | RR 1.26 95% CI: 0.87–1.82, NS RR 1.42 95% CI: 0.99–2.04, NS RR 1.44 95% CI: 1.26–1.64 p < 0.00001 RR 1.47 95% CI: 1.29–1.67 p < 0.00001 RR 0.68 95% CI: 0.49–0.95 | |
| Akbari et al. 2020 [230] | 25(OH)D < 20 ng/ml | 21,546 | Fixed RR 1.33; p < 0.0001; random RR 1.54 p = 0.0029 | |
| Fu et al. 2018 [265] | Vit D supplementation | 21,127 | RR = 0.41 95%CI = 0.22-0.78 | |
| Hyppönen et al. 2014 [266] | Vit D supplementation early in pregnancy higher serum 25(OH)D Vit D supplementation | 59,789 5058 5982 | RR 0.81 95% CI: 0.75–0.87 p < 0.000001 RR 0.52 95% CI: 0.30–0.89 p = 0.02 RR 0.66 95% CI: 0.52–0.83 p = 0.001 | |
| Aghajafari et al. 2013 [267] | Observational study Insufficient 25(OH)D levels 25(OH)D < 75 nmol/L 25(OH)D < 50 nmol/L | 3190 | RR 1.79 95% CI: 1.25–2.58 RR 2.11 95% CI: 1.36–3.27 RR 1.27 95% CI: 0.60–2.42 | |
| Tabesh et al. 2013 [268] | Vit D deficiency 25(OH)D ≤ 50 nmol/L (20 ng/mL), 25(OH)D <38 nmol/L (15.2 ng/mL) | 1775 931 | RR 2.78 95% CI: 1.45–5.33 NS | |
| Martínez-Domínguez et al. 2018 [269] | First half of pregnancy normal 25(OH)D (≥30.0 ng/mL) Insufficient (20.0–29.9 ng/mL) Deficient (<20.0 ng/mL) | 817 323 494 | RR 0.73 95% CI: 0.35–1.51, NS RR 0.79 95% CI: 0.28–2.21, NS RR 0.67 95% CI: 0.24–1.89, NS | |
| Kinshella et al. 2021 [270] | Vit D supplementation | 1353 | RR 0.62 95% CI: 0.43–0.91 NS | Decrease in PE risk by 38% |
| Yuan et al. 2021 [271] | Low 25(OH)D levels | 39,031 | RR 1.62 95% CI: 1.36–1.94 p < 0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poniedziałek-Czajkowska, E.; Mierzyński, R. Could Vitamin D Be Effective in Prevention of Preeclampsia? Nutrients 2021, 13, 3854. https://doi.org/10.3390/nu13113854
Poniedziałek-Czajkowska E, Mierzyński R. Could Vitamin D Be Effective in Prevention of Preeclampsia? Nutrients. 2021; 13(11):3854. https://doi.org/10.3390/nu13113854
Chicago/Turabian StylePoniedziałek-Czajkowska, Elżbieta, and Radzisław Mierzyński. 2021. "Could Vitamin D Be Effective in Prevention of Preeclampsia?" Nutrients 13, no. 11: 3854. https://doi.org/10.3390/nu13113854
APA StylePoniedziałek-Czajkowska, E., & Mierzyński, R. (2021). Could Vitamin D Be Effective in Prevention of Preeclampsia? Nutrients, 13(11), 3854. https://doi.org/10.3390/nu13113854






