Effects of Nutrition on Cognitive Function in Adults with or without Cognitive Impairment: A Systematic Review of Randomized Controlled Clinical Trials
Abstract
:1. Introduction
2. Materials and Methods
2.1. Eligibility Criteria
2.2. Literature Search
2.3. Screening and Data Extraction
2.4. Quality Assessment
2.5. Data Synthesis
3. Results
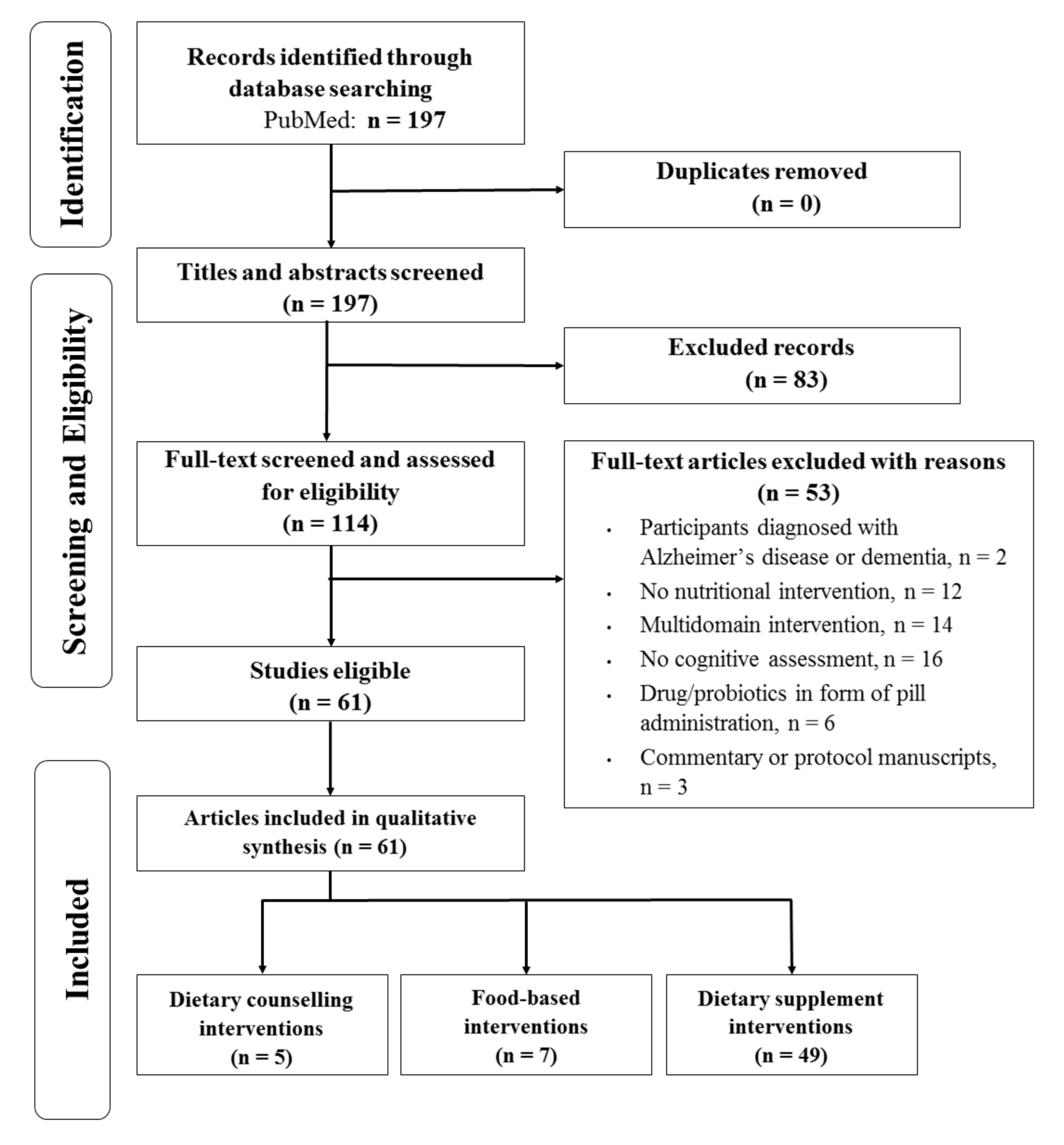
3.1. Search and Selection of Studies
3.2. Characteristics of Included Studies
3.3. Diet Counselling Interventions (n = 5)
3.4. Food-Based Interventions (n = 7)
3.5. Dietary Supplement Interventions (n = 49)
3.5.1. Protein and Amino Acid Supplements (n = 7)
3.5.2. Vitamin Supplements (n = 4)
3.5.3. Polyphenols Supplements (n = 10)
3.5.4. PUFAs Supplements (n = 5)
3.5.5. Combination of PUFAs, Polyphenols and Vitamins (n = 8)
3.5.6. Other Types of Dietary Supplements (n = 15)
| Author, Year | Design | Country | Study Population | n | Age, y (Mean ± sd or Range) | Intervention | Control | Duration | Outcomes 1 | Main Results |
|---|---|---|---|---|---|---|---|---|---|---|
| Dietary counselling interventions | ||||||||||
| Blumenthal et al., 2019 [30] | Double-blind, parallel | USA | SCD | 79 | 65.4 ± 6.8 | DASH diet | Usual dietary and exercise habits. | 6 m | TMT, the Stroop Test, the Digit Span Forward and Backward subtest and the Digit Symbol Substitution Test from the WAIS, the Ruff 2 and 7 Test, Animal Naming, HVLT-R, Medical College of Georgia Complex Figure Test and COWAT and Animal Naming test. | Non-significant |
| Komulainen et al., 2021 [31] | Single-blind, parallel | Finland | Apparently cognitively healthy | 469 | 57–78 | Finnish Nutrition Recommendations | General recommendations | 4 y | CERAD neuropsychological battery, MMSE | Non-significant |
| Tsolaki et al., 2020 [29] | Double-blind, parallel | Greece | MCI | 54 | 69.8 ± 6.9 | G1: 50 mL/d extra virgin olive oil + Mediterranean diet G2: 50 mL/d high phenolic early harvest extra virgin olive oil + Mediterranean diet | Mediterranean diet without olive oil | 12 m | ADAS-Cog, DST, Letter fluency, MMSE, Rivermead Behavioral Memory Test-Story Recall, ROCF, TMT-A and TMT-B, CDT | ADAS-cog, Digit Span and Letter fluency. MMSE |
| Wade et al., 2019 [27] | Blinding information not available, crossover | Australia | Cognitively healthy | 35 | 61.0 ± 7.1 | Mediterranean diet with 2–3 weekly servings of fresh, lean pork | Low fat diet | 8 w | Primary: Blood pressureSecondary: Cantab, ACE-R | Processing speed |
| Wade et al., 2020 [28] | Single-blind, crossover | Australia | Cognitively healthy | 43 | 60.2 ± 6.9 | Mediterranean diet: 3–4 daily servings of dairy foods | Low fat diet | 8 w | Primary: Blood pressure Secondary: Cantab, ACE-R | Processing speed. |
| Food-based interventions | ||||||||||
| Edwards et al., 2019 [33] | Single-blind, parallel | USA | Cognitively healthy | 163 | 25–45 | Fresh Hass Avocado: male (175 g/d containing 701 µg lutein and zeaxanthin); female (140 g/d containing 561 µg lutein and zeaxanthin) | Isocaloric meal without avocado (lutein/zeaxanthin content was 164/205 µg/d) | 12 w | Flanker, Nogo tasks, Oddball Task, Kaufman Brief Intelligence Test | Accuracy in the Flanker task (attentional inhibition) |
| Kuroda et al., 2019 [34] | Blinding information not available, parallel | Japan | Cognitively healthy | 52 | 72.9 ± 0.8 | 100 g/d of ultra-high hydrostatic pressurizing brown rice | 100 g/d of white rice | 24 m | HDS-R, MMSE, FAB, Cognitive CADi | HDS-R |
| Sakurai et al., 2020 [32] | Double-blind, parallel | Japan | Cognitively healthy | 61 | 60–84 | 3.0 g/d Matcha new green tea powder | Black tea flavored powder | 12 w | MoCa, MMSE, WMS-DR | MoCA score in the active group of women |
| Sala-Vila et al., 2020 [37] | Single-blind, parallel | Spain, USA | Cognitively healthy | 708 | 63–79 | Diet with walnuts (15% of energy) | Regular diet free of nuts | 2 y | RAVLT, ROCF, SVF, BNT, VOSP, WAIS-III, TMT A and B, Phonemic Fluency, SCWT, SDMT, DST, CPT-II, fMRI | Perception score |
| Sandberg et al., 2018 [36] | Blinding information not available, crossover | Sweden | Apparently cognitively healthy | 43 | 63.6 ± 5.3 | Ased bread consisted of a whole grain rye kernel/flour mixture (1:1 ratio) supplemented with resistant starch type 2 (239.2 g/d) | White wheat flour bread (170.9 g/d) | 3 d | VWM test, selective attention test. | Non-significant |
| Suzuki et al., 2019 [38] | Blinding information not available, crossover | Japan | MCI | 71 (F) | ≥70 | 33.4 g/d Camembert cheese | 33.4 g/d of processed cheese made from mozzarella cheese and cream cheese | 3 m | Primary: Serum brain-derived neurotrophic factor concentration Secondary: MMSE | Non-significant |
| Uenobe et al., 2019 [35] | Crossover, blinding information not available | Japan | Apparently cognitively healthy | 31 | IG 84.3 ± 0.3 CG 83.8 ± 9.1 | Dewaxed brown rice (3 meals/d, daily dose not available) | white rice (3 meals/d, daily dose not available) | 6 m | HDS-R | HDS-R |
| Dietary supplement interventions | ||||||||||
| Abe et al., 2020 [81] | Single-blind, parallel | Japan | Apparently cognitively healthy | 64 | 85.5 ± 6.8 | G1: 1.2 g/d L-leucine and 20 μg/d cholecalciferol and 6 g/d medium-chain triglycerides G2: 6 g/d medium-chain triglycerides | 6 g/d of long-chain triglycerides | 3 m | Primary: 10-s leg open and close test (muscle function) Secondary: MMSE, NM scale | MMSE |
| Ahles et al., 2020 [51] | Double-blind, parallel | Netherlands | Apparently cognitively healthy | 101 | 40–60 | Aronia melanocarpa extract: 90 and 150 mg/d (16 mg and 27 mg anthocyanins respectively) | Placebo (maltodextrin 150 mg/d) | 24 w | SCWT, grooved pegboard test, number cross-out test | Psychomotor speed. |
| Arellanes et al., 2020 [71] | Double-blind, parallel | USA | Cognitively healthy | 33 | 58–90 | 2152 mg/d of DHA and vitamin B complex(1 mg vitamin B12, 100 mg of vitamin B6 and 800 µg of folic acid) | Placebo(corn/soy oil and vitamin B complex (1 mg/d vitamin B12, 100 mg/d of vitamin B6 and 800 µg/d of folic acid)) | 6 m | Primary: DHA cerebral fluid contendingSecondary: CVLT-II, MoCa, TMT-A, TMT-B, CDR, MRI | Non-significant |
| Baleztena et al., 2018 [86] | Double-blind, parallel | Spain | Apparently cognitively healthy | 99 | 86.9 ± 5.9 | DHA 750 mg, EPA 120 mg, vitamin E 15 mg, Ginkgo biloba 180 mg, phosphatidylserine 45 mg, tryptophan 285 mg, vitamin B12 E 15 mg, folate 750 mg, daily | Placebo (gelatin capsule) | 1 y | MMSE, GDS, SPMSQ, SVF, CDT | Non-significant |
| Ban et al., 2018 [84] | Double-blind, parallel | South Korea | SCD | 75 | 40–65 | Tremella fuciformis capsules (Low-dose 600 mg/d, high-dose 1200 mg/d) | Placebo | 4, 8 w | SMCQ, WCST, Self-reported cognitive impairment, CANTAB | Short-term memory, executive functions, Gray matter volumes of brain regions Subjective memory |
| Bensalem et al., 2019 [53] | Double-blind, parallel | France and Canada | Cognitively healthy | 215 | 64.66 ± 2.91 | 600 mg/d of polyphenol-rich extract from grape and blueberry (258 mg flavonoids) | 600 mg/d of pure maltodextrin | 6 m | CANTAB (PALTEA section), CANTAB (SSP, VRM sections) | Verbal episodic memory in subjects with the highest cognitive impairments |
| Boyle et al., 2019 [80] | Double-blind, parallel | UK | Apparently cognitively healthy | 54 (M) | 18–55 | 250 mL/d water-based drink produced with milk protein concentrates (2.7 g of phospholipids and 300 mg phosphatidylserine) | Placebo (drink matched by adding butteroil) | 6 w | 2-back and attention switch task | Attention switch task |
| Carmichael et al., 2018 [65] | Double-blind, crossover | USA | Cognitively healthy | 11 | 67.3 ± 2.01 | Liquid emulsification (16 mg/d total ginsenosides: 960 mg EPA, 624 mg DHA, and 26 mg of green tea catechins) | Corn oil using non-essential fatty acid without ginsenosides nor catechins | 26 d | MMSE, DSST, Stroop test and LM (I & II), MRI | MMSE, Stroop test, DSST; brain activation (anterior and posterior cingulate cortex), functional connectivity (middle frontal gyrus and anterior cingulate cortex) |
| Chai et al., 2019 [54] | Blinding information not available, parallel | USA | Cognitively healthy | 37 | 65–80 | 480 mL tart cherry juice | Unsweetened black cherry flavored | 12 w | CANTAB (PAL, RVP, RTI, SWM and DST sections) | Memory, learning tasks, sustained attention, spatial working memory |
| Chhetri et al., 2018 [62] | Double-blind, parallel | France | Cognitively healthy | 637 | 75.3 ± 4.4 | PUFA ω-3 supplementation (800 mg/d of DHA and ≤225 mg/d of EPA) | Placebo (paraffin oil) | 3 y | Composite score (FCSRT, MMSE, DSST, CNT), TMT, WAIS-R and COWAT. | Non-significant |
| Chupel et al., 2018 [42] | Parallel, blinding information not available | Portugal | Apparently cognitively healthy | 24 (F) | 83.5 ± 6.9 | 150 mL/d water with 1.5 g taurine | Usual care | 14 w | MMSE | Non-significant |
| Cox et al., 2020 [52] | Double-blind, parallel | Australia | Cognitively healthy | 89 | 50–85 | 80 mg/d of curcumin extract (Longvida©) | Placebo | 12 w | DATT, vMWM, Serials Subtractions, Arrow Flankers Task | Working memory |
| Danthiir et al., 2018 [66] | Double-blind, parallel | Australia | Cognitively healthy | 403 | 65–90 | 1720 mg/d DHA and 600 mg/d EPA (plus mixed tocopherols added as antioxidant, 2.8–4.5 mg/g) | Placebo (3960 mg/d of low polyphenol olive oil + 40 mg/d of fish oil (7.2 mg EPA and 4.8 mg DHA)) | 18 m | Fluency, working memory, reasoning, and short-term memory (word memory—immediate recall), speed of memory scanning (Sternberg’s number and letter memory scanning), odd-man-out reaction time, perceptual speed, inhibition, simple and choice reaction time, psychomotor speed, MMSE score | Perceived cognitive mistakes Psychomotor speed only in men |
| Decroix et al., 2019 [50] | Double-blind, crossover | Belgium | Cognitively healthy | 22 | 41.2 ± 15.8 | 900 mg Cocoa Flavanols dissolved in 300 mL of skimmed milk. | 15 mg Cocoa Flavanols dissolved in 300 mL of skimmed milk. | 1 d | Flanker test and fMRI | Reaction time (flanker test) The BOLD response |
| Delrieu et al., 2020 [64] | Double-blind, parallel | France | Apparently cognitively healthy | 67 | 76.4 ± 4.2 | 800 mg/d of V0137 (DHA + EPA) | Placebo | 12 m | (18F) FDG PET imaging, FCRST, COWAT, Category Naming Test, DSST of WAIS-R, TMT, MMSE, CDR and Z-score, MRI | Non-significant |
| Fukuda et al., 2020 [82] | Double-blind, parallel | Japan | SCD | 60 | 45–64 | 35 mg/d of matured hop bitter acids (MHBAs) | Placebo (dextrin capsules) | 12 w | Word recall test; story recall test, VFT; semantic and phonemic fluency task; WMS-R; SWM; Stroop test, subjective memory performance, TMT-A, TMT-B | Verbal fluency and Stroop test |
| Fukuda, et al., 2020 [83] | Double-blind, parallel | Japan | SCD | 100 | 45–69 | 35 mg/d of MHBAs | Placebo (dextrin) | 12 w | CAT, SDMT, memory updating test; Position response test; Memory (RAVLT, S-PA, WMS-R) | Memory retrieval and attention |
| Giudici et al., 2020 [61] | Double-blind, parallel | France | Cognitively healthy | 715 | 75.3 ± 4.4 | PUFA ω-3 (800 mg/d of DHA and ≤ 225 mg/d of EPA) | Placebo (paraffin oil) | 3 y | Z-score (MMSE, Digit Symbol Substitution Test, free and total recall of the Free and Cued Selective Reminding test, and Category Naming Test) | Non-significant |
| Hamasaki et al., 2019 [41] | Double-blind, parallel | Japan | Apparently cognitively healthy | 35 | ≥50 | 3 tablets/d of lactotripeptide (casein hydrolysate with 1.4 mg valine-proline-proline and 2.0 mg isoleucine-proline-proline) | Placebo (sodium caseinate) | 8 w | Primary: Oxygenated hemoglobin (oxy-Hb) concentration (oxy-Hb change) in the prefrontal cortex during the Stroop task Secondary: Stroop task and stroop interference time | Non-significant |
| Herrlinger et al., 2018 [55] | Double-blind, parallel | USA | SCD | 90 | 59.4 ± 0.6 | 600 mg/d or 900 mg/d spearmint extract | Placebo | 90 d | Cognitive Drug Research System | Quality of working memory, spatial working memory accuracy, vigor-activity, alertness and behavior following wakefulness after the highest dose |
| Hu et al., 2018 [47] | Double-blind, parallel | China | MCI | 181 | 65–75 | 400 IU/d vitamin D | Placebo (starch granules) | 1 y | Chinese version of the WAIS-R; MMSE | Scores of information, digit span, vocabulary, block design and picture arrangement |
| Hwang et al., 2019 [56] | Double-blind, parallel | South Korea | MCI | 100 | 55–85 | 800 mg/d of Lactobacillus plantarum C29-fermented soybean enriched with isoflavones and saponins. | Placebo (cellulose) | 12 w | Computerized neurocognitive function tests, Verbal learning Test, ACPT, DST | cognitive functions and attention |
| Igase et al., 2017 [74] | Double-blind, parallel | Japan | Apparently cognitively healthy | 84 | 71 ± 9 | 125 mL juice enriched with 6 mg/d auraptene | 125 mL juice enriched with 0.1 mg auraptene/day | 24 w | MCI Screen using 10-word immediate recall test, MMSE | 10-word immediate recall test |
| Ito et al., 2018 [73] | Double-blind, parallel | Japan | MCI | 21 | 57–78 | 6 mg/d astaxanthin and 10 mg/d sesamin | Placebo (filling agent such as safflower oil, starch and water, dispersants and artificial colorants) | 12 w | CNSVS (SDC, Stroop test, shifting attention test and continuous performance test domains), Japanese version of the ADAS-cog | Psychomotor speed and processing speed |
| Kita et al., 2018 [44] | Double-blind, parallel | Japan | SCD | 101 | 45–64 | 1 g/d whey peptide which included 1.6 mg of glycine–threonine–tryptophan–tyrosine peptide | Placebo (maltodextrin) | 12 w | Story recall tests, VFT, Hamamatsu Higher Brain Function Scale, Japanese version of the RBMT, Stroop test, DST, and paced auditory serial addition test | VFT, Stroop, subjective memory function in subjects with high-level fatigue |
| Kuszewski et al., 2020 [68] | Double-blind, parallel | Australia | Cognitively healthy | 152 | 50–80 | G1: Fish oil (2000 mg/d DHA + 400 mg/d EPA); G2: curcumin (160 mg/d); G3: Fish oil (2000 mg/d DHA + 400 mg/d EPA) and curcumin (160 mg/d) | Placebo (mix of corn and olive oil with 20 mg fish oil) | 16 w | Primary: Cerebrovascular function Secondary: 7 tests from the NIH toolbox, RAVLT, Forward Spatial Span Test, and TMT parts A and B. | Processing speed (males in G1) verbal memory (males in G2) |
| Lau et al., 2020 [59] | Double-blind, parallel | Malaysia | MCI | 36 | 66.42 ± 0.63 | 500 mg/d Biokesum (extract of Persicaria minor which contains quercetin-3-glucuronide (not less than 0.45%), quercitrin (not less than 0.15%)) and total phenolic content (not less than 100 mg GAE/g dE). | Placebo (560 mg/d maltodextrin) | 6 m | MMSE, DST, RAVLT, VR I-II, Digit symbol substitution, POMS, fMRI, N-Back. | VR II, bilateral dorsolateral prefrontal cortex activation |
| Li et al., 2018 [70] | Double-blind, parallel | China | MCI | 240 | ≥60 | FA + DHA (FA 800 μg/d + DHA 800 mg/d) FA (FA 800 μg/d); DHA (DHA 800 mg/d) | Placebo tablets (corn starch) and placebo capsules (soybean oil) | 6 m | FSIQ and Chinese version of WAIS-R which included three verbal subtests (information, arithmetic and digit span) and three performance subtests (block design, picture completion and picture arrangement) | FSIQ, arithmetic, digit span, picture completion and block design (FA +DHA group) digit span and block design (FA group) information, arithmetic and digit span (DHA group) FSIQ scores, arithmetic and picture completion (FA+ DHA vs. FA) picture completion and block design (FA+ DHA vs. DHA) |
| Ma et al., 2019 [49] | Single-blind, parallel | China | MCI | 240 | ≥ 65 | Folic acid alone (two tablets of 400 μg/d of folic acid) Vitamin B12 alone (one tablet with 25 μg/d vitamin B12) Folic acid plus vitamin B12 (two tablets of 400 μg/d folic acid plus one tablet with 25 μg/d vitamin B12) | Without treatment | 6 m | Chinese version of the WAIS-RC (Information, Similarities, Vocabulary, Comprehension, Arithmetic, DST, Block Design, Picture Completion, Digit Symbol-Coding, Object Assembly, and Picture Arrangement domains), IQ index, MMSE. | Full Scale IQ, verbal IQ, Information and Digit Span |
| Marriott et al., 2021 [60] | Double-blind, parallel | USA | Apparently cognitively healthy | 555 | 20–35 | 8 dietary supplements daily of krill oil (2.3 g ω-3) | Placebo capsules (macadamia nut oil) | 20 w | Stroop Color-Word Inhibition Test, SDMT, Figural Continuous Paired Associates Test | Non-significant |
| Masuoka et al., 2019 [39] | Double-blind, parallel | Japan | MCI | 54 | 49–86 | 750 mg/d anserine plus 250 mg/d carnosine | Placebo | 12 w | CDR, ADAS-cog test, WMS-1, WMS-2, MMSE, gloCDR | gloCDR score in APOE4(+) |
| McNamara et al., 2018 [69] | Double-blind, parallel | USA | SCD | 94 | 62–80 | Fish oil group (FO): 1.6 g/d EPA and 0.8 g/d DHA Blueberry group (BB): 25 g/d blueberry powder rich in anthocyanins FO + BB group: 1.6 g/d EPA, 0.8 g/d DHA, 25 g/d blueberry powder | Placebo (corn oil powder) | 24 w | TMT A and B, Controlled Oral Word Production, Alternate forms of HVLT-R, Dysexecutive Questionnaire. | Memory discrimination (BB) |
| Cognitive symptoms (BB and FO) | ||||||||||
| Ochiai et al., 2019 [58] | Double-blind, crossover | Japan | MCI | 34 | 73.7 ± 6.0 | 1.107 g/d of chlorogenic acid (CGA) (553.6 mg in 100 mL of water) | Placebo | 12 w | MMSE; ADAS-cog test; TMT-A, TMT-B); cognitive function tests. | Number of errors in the TMT-B test |
| Owusu et al., 2019 [46] | Double-blind, parallel | USA | SCD | 260 (F) | 68.2 | D3 vitamin daily dose: 2400 IU, 3600 IU, or 4800 IU. | Placebo (1200 mg/d calcium carbonate) | 3 y | MMSE | Non-significant |
| Perry et al., 2018 [78] | Double-blind, parallel | United Kingdom | Cognitively healthy | 44 | 61 ± 9.26 | 10 mL/d SRM ethanol extract (SRM: Salvia officinalis L., Rosmarinus officinalis L. and Melissa officinalis L., collected and individually extracted 0.5 g/mL in 45% EtOH) | Placebo (50%º fresh sweet cicely) | 2 w | Immediate and delayed word recall to assess verbal working and episodic memory, VWM, Verbal Episodic Memory. | Delayed word recalls (subjects under 63 years) |
| Reid et al., 2018 [77] | Double-blind, parallel | South Korea | Cognitively healthy | 60 | IG 72.35 ± 5.54 CG 74.57 ± 5.69 | 1.5 g/day of fermented Laminaria japonica | Placebo (1.5 g/d of sucrose pill) | 6 w | MMSE, Numerical Memory Test, Raven’s Standard Progressive Matrices, Flanker Test, Iconic Memory Test and TMT. | K-MMSE, flanker test scores, working memory, visual and spatial reasoning |
| Saitsu et al., 2019 [76] | Double-blind, parallel | Japan | Cognitively healthy | 34 | >50 | 3.2 g/d Fruiting body of Hericium erinaceus | Placebo | 12 w | MMSE, Benton visual retention test, and S-PA. | MMSE |
| Schönhoff et al., 2018 [43] | Double-blind, crossover | Germany | Cognitively healthy | 20 | 35 ± 14 | 125 mg/d L-homoarginine supplement | Placebo | 4 w | VLMT, TAP, TMT, FWIT, Regensburg verbal fluency test | Non-significant |
| Schwarz et al., 2018 [72] | Double-blind, parallel | Germany | MCI | 49 | 50–80 | 1320 mg EPA, 880 mg DHA, 15 mg vitamin E, daily | Sunflower oil | 26 w | MRI | Cerebral perfusion |
| Stavrinou et al., 2020 [67] | Double-blind, parallel | Cyprus | MCI | 46 | ≥65 | 20 mL/d (4.14 g DHA, 0.810 mg EPA, 1.8 g GLA, 3.15 g LA, 0.6 mg vitamin A, 22 mg α-tocopherol, 760 mg ɣ-tocopherol) | Placebo (20 mL pure virgin olive oil) | 6 m | ACE-R, MMSE, TMT, Stroop Color and Word Test, symbol cancellation test. | Functional capacity, physical Health |
| Time to complete ACE-R, MMSE and STROOP | ||||||||||
| Tabue-Teguo et al., 2018 [63] | Double-blind, parallel | France | Cognitively healthy | 843 | 75.3 ± 4.4 | 800 mg/d of V0137 (DHA + EPA) | Placebo (paraffin oil) | 3 y | COWAT, Category Naming Test, DSST of the WAIS-R, TMT, MMSE | Non-significant |
| Tadokoro et al., 2019 [87] | Double-blind, parallel | Japan | MCI | 78 | 65–85 | Tablet daily dose: coenzyme Q10 (10 mg), niacin amid (2 mg), L-cystine (50 mg), ascorbic acid (94 mg), succinic acid (10 mg), fumaric acid (10 mg), L-glutamine (85 mg), and riboflavin (4 mg). | Placebo | 6 m | MMSE and HDS-R | MMSE, HDS-R |
| Tohda et al., 2020 [75] | Double-blind, parallel | Japan | Cognitively healthy | 31 | 40–80 | 203.01 mg/d of Eleutherococcus senticosus leaf extract, 20.01 mg/d of Rhizome of Drynaria fortunei extract, 133.38 mg/d crystal cellulose and 3.60 mg/d calcium stearate | Placebo (356.4 mg crystal cellulose and 3.60 mg calcium stearate) | 12 w | RBANS, Japanese versions of MMSE | Figure recall sub score of RBANS (language domain, semantic fluency and figure recall) |
| Varanoske et al., 2018 [45] | Double-blind, parallel | USA | Apparently cognitively healthy | 19 (M) | 18–35 | 12 g/day β-Alanine | Placebo | 14 d | Visual analog scale (Mood), Serial Subtraction Test (Mathematical processing), reaction time (by using Dynavision D2 Visuomotor Training Device), visual tracking speed (by using Neurotracker multiple object tracking device) | Errors on reaction time testing |
| Wirth et al., 2018 [85] | Double-blind, parallel | Germany | SCD | 30 | 65.6 ± 6.2 | 750 mg/d of spermidine-rich plant (1.2 mg of the natural polyamine plus 510 mg cellulose) | Placebo (750 mg/d of potato starch and 510 mg/d cellulose) | 3 m | Memory performance (Behavioral non-verbal MST), AVLT, digit symbol substitution test | Non-significant |
| You et al., 2021 [57] | Double-blind, parallel | Malaysia | MCI | 48 | 65.11 ± 4.05 | 500 mg/d of Cosmos caudatus powder | Placebo (1000 mg/d of maltodextrin) | 12 w | MMSE, DS, RAVLT, Digit Symbol substitution, Visual Reproduction and POMS | MMSE |
| Yuda et al., 2020 [40] | Double-blind, parallel | Japan | Cognitively healthy | 268 | ≥40 | 200 μg/d Met-Lys-Pro peptide in 1 g casein hydrolysate | Placebo (1 g dextrin with no detectable MKP) | 24 w | ADAS-cog, HDS-R, MoCa Japanese version, and Short-Form Health Survey (SF-8). | Orientation in ADAS-cog |
| Zajac et al., 2020 [48] | Double-blind, parallel | Australia | Cognitively healthy | 436 | 60–90 | Vitamin D2-enrich Mushroom (600 µg/d D2 vitamin); Vitamin D3 (600 µg/d D3 vitamin), standard mushroom capsule (NA) | Placebo | 6 m | CSIRO Cognitive Assessment Battery (C-CAB) | Non-significant |
| Zhu et al., 2020 [79] | Double-blind, parallel | USA | Apparently cognitively healthy | 250 | 40–85 | Magnesium glycinate capsules | Placebo (microcrystalline cellulose) | 12 w | MoCa score | MoCa |
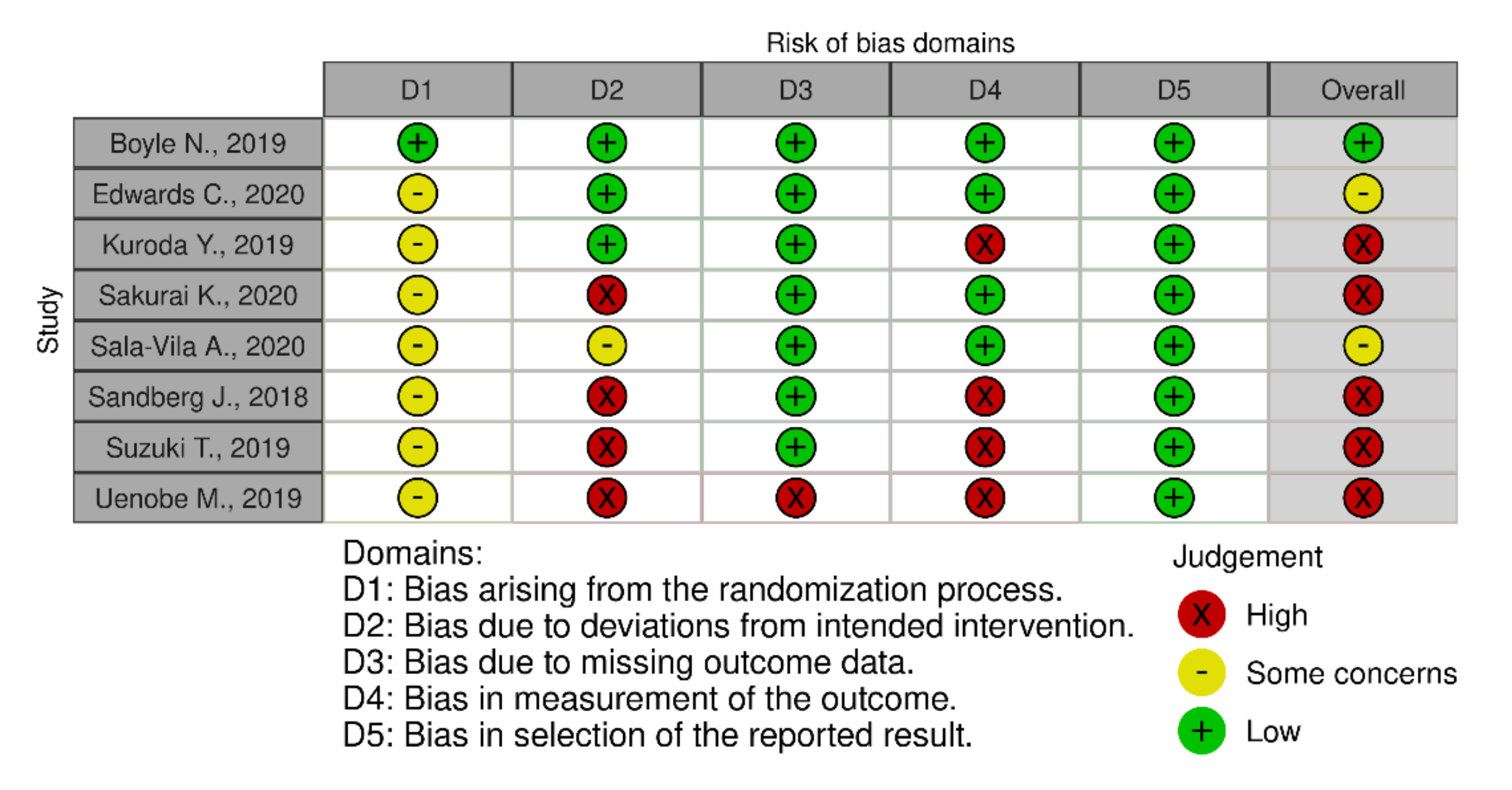
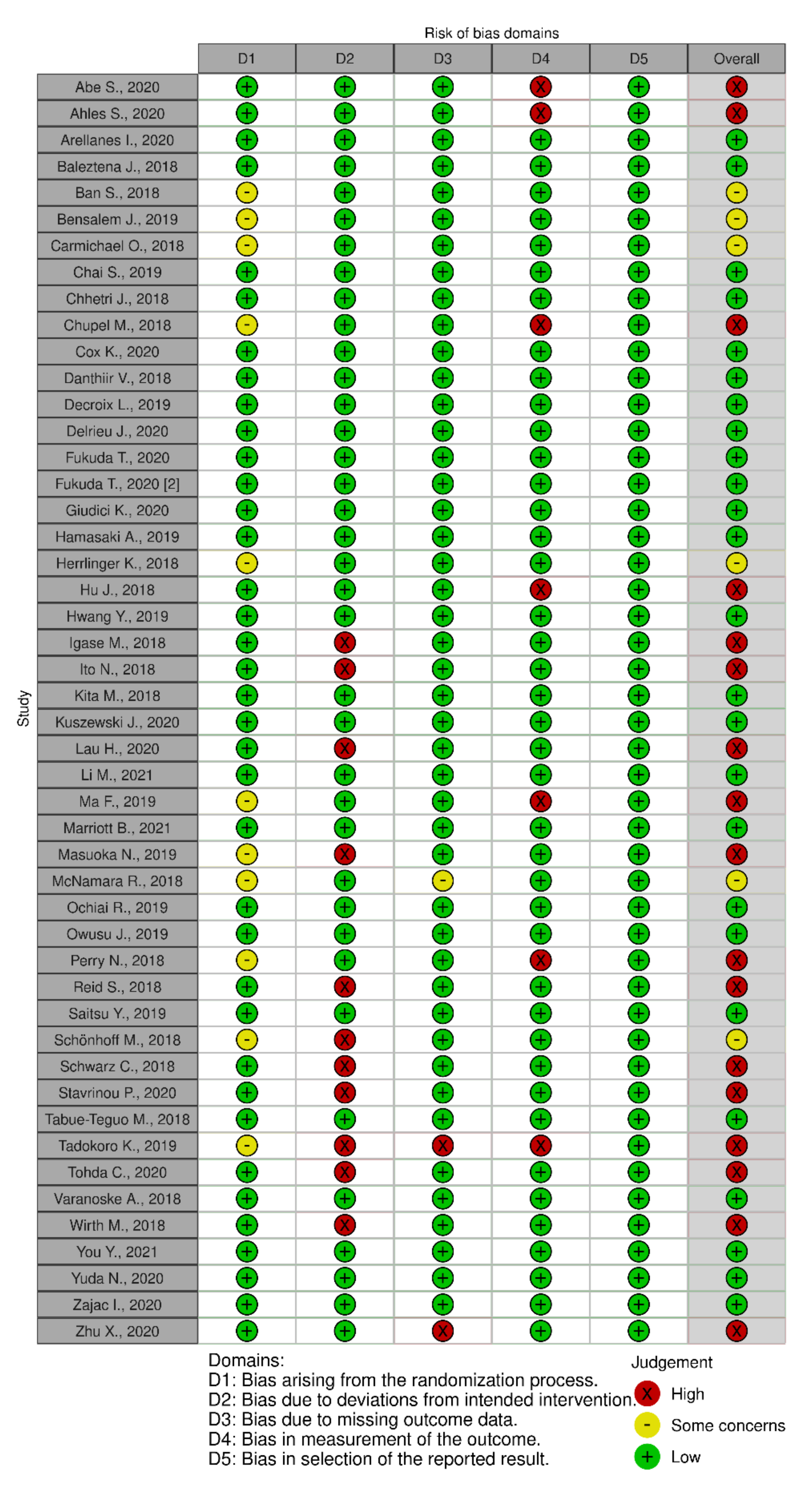
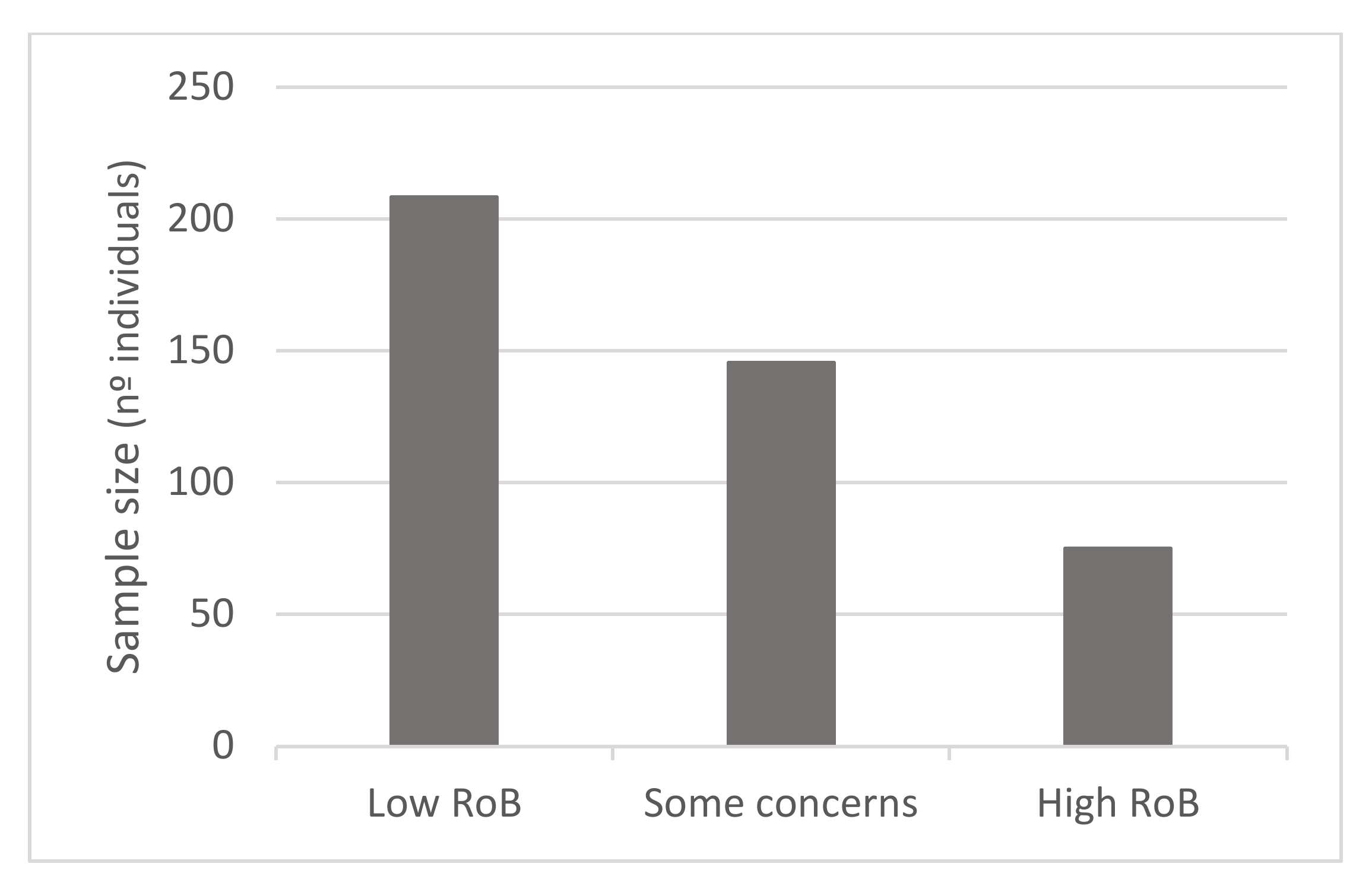
3.6. Risk of Bias Assessment
4. Discussion
4.1. Protein and Amino Acid Supplements
4.2. Vitamin Supplements
4.3. Polyphenol Supplements
4.4. PUFAs Supplements
4.5. Combination of PUFAs, Polyphenols and Vitamins
4.6. Other Types of Dietary Supplements
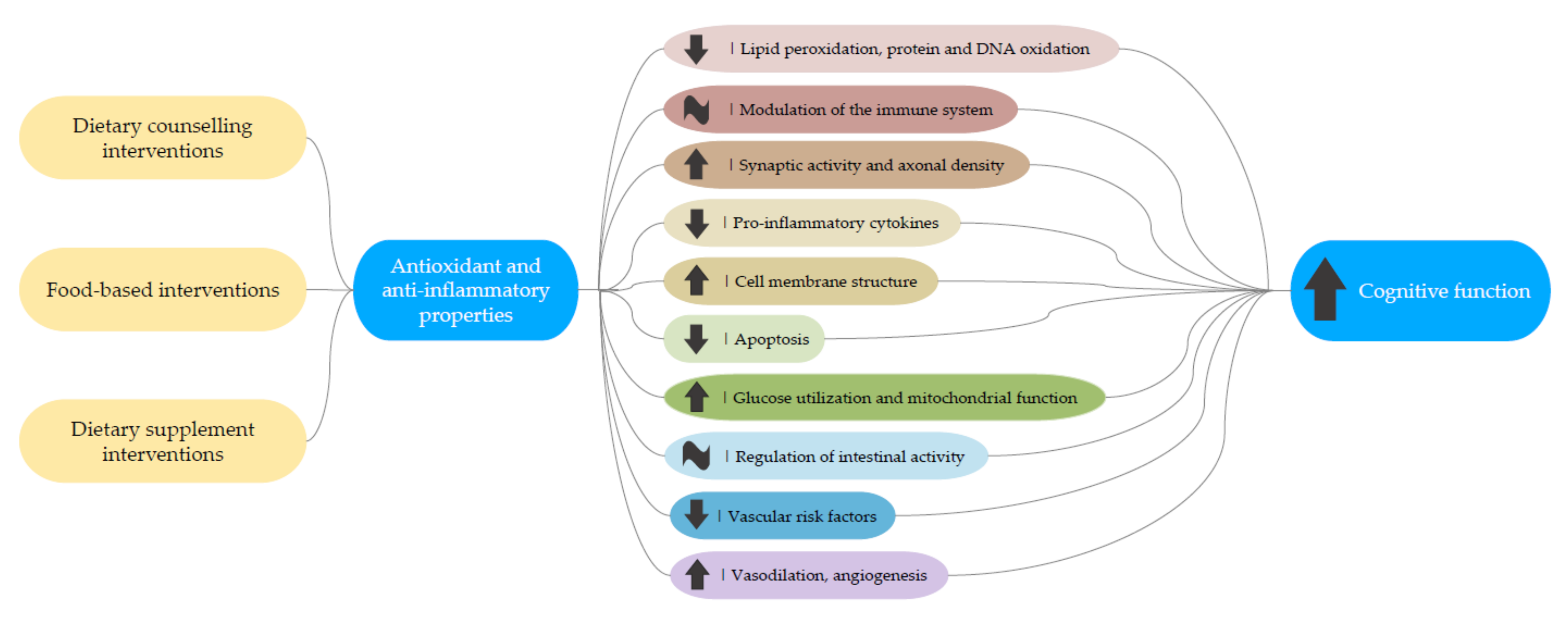
4.7. Potential Mechanisms
4.8. Limitations and Strengths
4.9. Further Study Directions
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dementia. Available online: https://www.who.int/news-room/fact-sheets/detail/dementia (accessed on 6 September 2021).
- Daviglus, M.L.; Bell, C.C.; Berrettini, W.; Bowen, P.E.; Connolly, E.S.; Cox, N.J.; Dunbar-Jacob, J.M.; Granieri, E.C.; Hunt, G.; McGarry, K.; et al. NIH State-of-the-Science Conference Statement: Preventing Alzheimer’s Disease and Cognitive Decline. NIH Consens. State Sci. Statements 2010, 27, 1–30. [Google Scholar] [PubMed]
- Panza, F.; Seripa, D.; Solfrizzi, V.; Imbimbo, B.P.; Lozupone, M.; Leo, A.; Sardone, R.; Gagliardi, G.; Lofano, L.; Creanza, B.C.; et al. Emerging Drugs to Reduce Abnormal β-Amyloid Protein in Alzheimer’s Disease Patients. Expert Opin. Emerg. Drugs 2016, 21, 377–391. [Google Scholar] [CrossRef]
- Petersen, R.C.; Caracciolo, B.; Brayne, C.; Gauthier, S.; Jelic, V.; Fratiglioni, L. Mild Cognitive Impairment: A Concept in Evolution. J. Intern. Med. 2014, 275, 214–228. [Google Scholar] [CrossRef] [PubMed]
- Parnetti, L.; Chipi, E.; Salvadori, N.; D’Andrea, K.; Eusebi, P. Prevalence and Risk of Progression of Preclinical Alzheimer’s Disease Stages: A Systematic Review and Meta-Analysis. Alzheimers Res. Ther. 2019, 11, 7. [Google Scholar] [CrossRef]
- Jessen, F.; Amariglio, R.E.; Van Boxtel, M.; Breteler, M.; Ceccaldi, M.; Chételat, G.; Dubois, B.; Dufouil, C.; Ellis, K.A.; Van der Flier, W.M.; et al. A Conceptual Framework for Research on Subjective Cognitive Decline in Preclinical Alzheimer’s Disease. Alzheimers Dement. J. Alzheimers Assoc. 2014, 10, 844–852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdulrab, K.; Heun, R. Subjective Memory Impairment. A Review of Its Definitions Indicates the Need for a Comprehensive Set of Standardised and Validated Criteria. Eur. Psychiatry J. Assoc. Eur. Psychiatr. 2008, 23, 321–330. [Google Scholar] [CrossRef]
- Mitchell, A.J. Is It Time to Separate Subjective Cognitive Complaints from the Diagnosis of Mild Cognitive Impairment? Age Ageing 2008, 37, 497–499. [Google Scholar] [CrossRef] [Green Version]
- Mitchell, A.J. The Clinical Significance of Subjective Memory Complaints in the Diagnosis of Mild Cognitive Impairment and Dementia: A Meta-Analysis. Int. J. Geriatr. Psychiatry 2008, 23, 1191–1202. [Google Scholar] [CrossRef]
- Darweesh, S.K.L.; Wolters, F.J.; Ikram, M.A.; De Wolf, F.; Bos, D.; Hofman, A. Inflammatory Markers and the Risk of Dementia and Alzheimer’s Disease: A Meta-Analysis. Alzheimers Dement. J. Alzheimers Assoc. 2018, 14, 1450–1459. [Google Scholar] [CrossRef]
- Féart, C.; Samieri, C.; Rondeau, V.; Amieva, H.; Portet, F.; Dartigues, J.-F.; Scarmeas, N.; Barberger-Gateau, P. Adherence to a Mediterranean Diet, Cognitive Decline, and Risk of Dementia. JAMA 2009, 302, 638–648. [Google Scholar] [CrossRef] [Green Version]
- Morris, M.C.; Tangney, C.C.; Wang, Y.; Sacks, F.M.; Bennett, D.A.; Aggarwal, N.T. MIND Diet Associated with Reduced Incidence of Alzheimer’s Disease. Alzheimers Dement. J. Alzheimers Assoc. 2015, 11, 1007–1014. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.; Maguire, B.; Brodaty, H.; O’Leary, F. Dietary Patterns and Cognitive Health in Older Adults: A Systematic Review. J. Alzheimers Dis. 2019, 67, 583–619. [Google Scholar] [CrossRef]
- Duplantier, S.C.; Gardner, C.D. A Critical Review of the Study of Neuroprotective Diets to Reduce Cognitive Decline. Nutrients 2021, 13, 2264. [Google Scholar] [CrossRef]
- Mottaghi, T.; Amirabdollahian, F.; Haghighatdoost, F. Fruit and Vegetable Intake and Cognitive Impairment: A Systematic Review and Meta-Analysis of Observational Studies. Eur. J. Clin. Nutr. 2018, 72, 1336–1344. [Google Scholar] [CrossRef]
- Theodore, L.E.; Kellow, N.J.; McNeil, E.A.; Close, E.O.; Coad, E.G.; Cardoso, B.R. Nut Consumption for Cognitive Performance: A Systematic Review. Adv. Nutr. 2021, 12, 777–792. [Google Scholar] [CrossRef]
- Román, G.C.; Jackson, R.E.; Reis, J.; Román, A.N.; Toledo, J.B.; Toledo, E. Extra-Virgin Olive Oil for Potential Prevention of Alzheimer Disease. Rev. Neurol. 2019, 175, 705–723. [Google Scholar] [CrossRef] [PubMed]
- Solfrizzi, V.; Colacicco, A.M.; D’Introno, A.; Capurso, C.; Torres, F.; Rizzo, C.; Capurso, A.; Panza, F. Dietary Intake of Unsaturated Fatty Acids and Age-Related Cognitive Decline: A 8.5-Year Follow-up of the Italian Longitudinal Study on Aging. Neurobiol. Aging 2006, 27, 1694–1704. [Google Scholar] [CrossRef] [PubMed]
- Letenneur, L.; Proust-Lima, C.; Le Gouge, A.; Dartigues, J.F.; Barberger-Gateau, P. Flavonoid Intake and Cognitive Decline over a 10-Year Period. Am. J. Epidemiol. 2007, 165, 1364–1371. [Google Scholar] [CrossRef] [PubMed]
- Goodwill, A.M.; Szoeke, C. A Systematic Review and Meta-Analysis of The Effect of Low Vitamin D on Cognition. J. Am. Geriatr. Soc. 2017, 65, 2161–2168. [Google Scholar] [CrossRef]
- McGrattan, A.M.; McEvoy, C.T.; McGuinness, B.; McKinley, M.C.; Woodside, J.V. Effect of Dietary Interventions in Mild Cognitive Impairment: A Systematic Review. Br. J. Nutr. 2018, 120, 1388–1405. [Google Scholar] [CrossRef] [Green Version]
- Solfrizzi, V.; Agosti, P.; Lozupone, M.; Custodero, C.; Schilardi, A.; Valiani, V.; Sardone, R.; Dibello, V.; Di Lena, L.; Lamanna, A.; et al. Nutritional Intervention as a Preventive Approach for Cognitive-Related Outcomes in Cognitively Healthy Older Adults: A Systematic Review. J. Alzheimers Dis. 2018, 64, S229–S254. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 Explanation and Elaboration: Updated Guidance and Exemplars for Reporting Systematic Reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef] [PubMed]
- McGuinness, L.A.; Higgins, J.P.T. Risk-of-Bias VISualization (Robvis): An R Package and Shiny Web App for Visualizing Risk-of-Bias Assessments. Res. Synth. Methods 2021, 12, 55–61. [Google Scholar] [CrossRef] [PubMed]
- Risk of Bias 2 (RoB 2) Tool. Available online: https://methods.cochrane.org/risk-bias-2 (accessed on 25 August 2021).
- Wade, A.T.; Davis, C.R.; Dyer, K.A.; Hodgson, J.M.; Woodman, R.J.; Keage, H.A.D.; Murphy, K.J. A Mediterranean Diet with Fresh, Lean Pork Improves Processing Speed and Mood: Cognitive Findings from the MedPork Randomised Controlled Trial. Nutrients 2019, 11, 1521. [Google Scholar] [CrossRef] [Green Version]
- Wade, A.T.; Davis, C.R.; Dyer, K.A.; Hodgson, J.M.; Woodman, R.J.; Keage, H.A.D.; Murphy, K.J. A Mediterranean Diet Supplemented with Dairy Foods Improves Mood and Processing Speed in an Australian Sample: Results from the MedDairy Randomized Controlled Trial. Nutr. Neurosci. 2020, 23, 646–658. [Google Scholar] [CrossRef]
- Tsolaki, M.; Lazarou, E.; Kozori, M.; Petridou, N.; Tabakis, I.; Lazarou, I.; Karakota, M.; Saoulidis, I.; Melliou, E.; Magiatis, P. A Randomized Clinical Trial of Greek High Phenolic Early Harvest Extra Virgin Olive Oil in Mild Cognitive Impairment: The MICOIL Pilot Study. J. Alzheimers Dis. 2020, 78, 801–817. [Google Scholar] [CrossRef]
- Blumenthal, J.A.; Smith, P.J.; Mabe, S.; Hinderliter, A.; Lin, P.-H.; Liao, L.; Welsh-Bohmer, K.A.; Browndyke, J.N.; Kraus, W.E.; Doraiswamy, P.M.; et al. Lifestyle and Neurocognition in Older Adults with Cognitive Impairments. Neurology 2019, 92, e212–e223. [Google Scholar] [CrossRef] [Green Version]
- Komulainen, P.; Tuomilehto, J.; Savonen, K.; Männikkö, R.; Hassinen, M.; Lakka, T.A.; Hänninen, T.; Kiviniemi, V.; Jacobs, D.R.; Kivipelto, M.; et al. Exercise, Diet, and Cognition in a 4-Year Randomized Controlled Trial: Dose-Responses to Exercise Training (DR’s EXTRA). Am. J. Clin. Nutr. 2021, 113, 1428–1439. [Google Scholar] [CrossRef]
- Sakurai, K.; Shen, C.; Ezaki, Y.; Inamura, N.; Fukushima, Y.; Masuoka, N.; Hisatsune, T. Effects of Matcha Green Tea Powder on Cognitive Functions of Community-Dwelling Elderly Individuals. Nutrients 2020, 12, 3639. [Google Scholar] [CrossRef]
- Edwards, C.; Walk, A.; Thompson, S.; Reeser, G.; Erdman, J., Jr.; Burd, N.; Holscher, H.; Khan, N. Effects of 12-Week Avocado Consumption on Cognitive Function among Adults with Overweight and Obesity. Int. J. Psychophysiol. 2019, 148, 13–24. [Google Scholar] [CrossRef]
- Kuroda, Y.; Matsuzaki, K.; Wakatsuki, H.; Shido, O.; Harauma, A.; Moriguchi, T.; Sugimoto, H.; Yamaguchi, S.; Yoshino, K.; Hashimoto, M. Influence of Ultra-High Hydrostatic Pressurizing Brown Rice on Cognitive Functions and Mental Health of Elderly Japanese Individuals: A 2-Year Randomized and Controlled Trial. J. Nutr. Sci. Vitaminol. 2019, 65, S80–S87. [Google Scholar] [CrossRef] [Green Version]
- Uenobe, M.; Saika, T.; Waku, N.; Ohno, M.; Inagawa, H. Effect of Continuous Dewaxed Brown Rice Ingestion on the Cognitive Function of Elderly Individuals. J. Nutr. Sci. Vitaminol. 2019, 65, S122–S124. [Google Scholar] [CrossRef] [Green Version]
- Sandberg, J.C.; Björck, I.M.E.; Nilsson, A.C. Impact of Rye-Based Evening Meals on Cognitive Functions, Mood and Cardiometabolic Risk Factors: A Randomized Controlled Study in Healthy Middle-Aged Subjects. Nutr. J. 2018, 17, 102. [Google Scholar] [CrossRef]
- Sala-Vila, A.; Valls-Pedret, C.; Rajaram, S.; Coll-Padrós, N.; Cofán, M.; Serra-Mir, M.; Pérez-Heras, A.M.; Roth, I.; Freitas-Simoes, T.M.; Doménech, M.; et al. Effect of a 2-Year Diet Intervention with Walnuts on Cognitive Decline. The Walnuts and Healthy Aging (WAHA) Study: A Randomized Controlled Trial. Am. J. Clin. Nutr. 2020, 111, 590–600. [Google Scholar] [CrossRef]
- Suzuki, T.; Kojima, N.; Osuka, Y.; Tokui, Y.; Takasugi, S.; Kawashima, A.; Yamaji, T.; Hosoi, E.; Won, C.W.; Kim, H. The Effects of Mold-Fermented Cheese on Brain-Derived Neurotrophic Factor in Community-Dwelling Older Japanese Women with Mild Cognitive Impairment: A Randomized, Controlled, Crossover Trial. J. Am. Med. Dir. Assoc. 2019, 20, 1509–1514.e2. [Google Scholar] [CrossRef] [PubMed]
- Masuoka, N.; Yoshimine, C.; Hori, M.; Tanaka, M.; Asada, T.; Abe, K.; Hisatsune, T. Effects of Anserine/Carnosine Supplementation on Mild Cognitive Impairment with APOE4. Nutrients 2019, 11, 1626. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yuda, N.; Tanaka, M.; Yamauchi, K.; Abe, F.; Kakiuchi, I.; Kiyosawa, K.; Miyasaka, M.; Sakane, N.; Nakamura, M. Effect of the Casein-Derived Peptide Met-Lys-Pro on Cognitive Function in Community-Dwelling Adults Without Dementia: A Randomized, Double-Blind, Placebo-Controlled Trial. Clin. Interv. Aging 2020, 15, 743–754. [Google Scholar] [CrossRef]
- Hamasaki, A.; Akazawa, N.; Yoshikawa, T.; Myoenzono, K.; Tanahashi, K.; Sawano, Y.; Nakata, Y.; Maeda, S. Combined Effects of Lactotripeptide and Aerobic Exercise on Cognitive Function and Cerebral Oxygenation in Middle-Aged and Older Adults. Am. J. Clin. Nutr. 2019, 109, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Chupel, M.U.; Minuzzi, L.G.; Furtado, G.; Santos, M.L.; Hogervorst, E.; Filaire, E.; Teixeira, A.M. Exercise and Taurine in Inflammation, Cognition, and Peripheral Markers of Blood-Brain Barrier Integrity in Older Women. Appl. Physiol. Nutr. Metab. 2018, 43, 733–741. [Google Scholar] [CrossRef] [Green Version]
- Schönhoff, M.; Weineck, G.; Hoppe, J.; Hornig, S.; Cordts, K.; Atzler, D.; Gerloff, C.; Böger, R.; Neu, A.; Schwedhelm, E.; et al. Cognitive Performance of 20 Healthy Humans Supplemented with L-Homoarginine for 4 Weeks. J. Clin. Neurosci. 2018, 50, 237–241. [Google Scholar] [CrossRef]
- Kita, M.; Obara, K.; Kondo, S.; Umeda, S.; Ano, Y. Effect of Supplementation of a Whey Peptide Rich in Tryptophan-Tyrosine-Related Peptides on Cognitive Performance in Healthy Adults: A Randomized, Double-Blind, Placebo-Controlled Study. Nutrients 2018, 10, 899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Varanoske, A.N.; Wells, A.J.; Kozlowski, G.J.; Gepner, Y.; Frosti, C.L.; Boffey, D.; Coker, N.A.; Harat, I.; Hoffman, J.R. Effects of β-Alanine Supplementation on Physical Performance, Cognition, Endocrine Function, and Inflammation during a 24 h Simulated Military Operation. Physiol. Rep. 2018, 6, e13938. [Google Scholar] [CrossRef] [Green Version]
- Owusu, J.E.; Islam, S.; Katumuluwa, S.S.; Stolberg, A.R.; Usera, G.L.; Anwarullah, A.A.; Shieh, A.; Dhaliwal, R.; Ragolia, L.; Mikhail, M.B.; et al. Cognition and Vitamin D in Older African-American Women—Physical Performance and Osteoporosis Prevention with Vitamin D in Older African Americans Trial and Dementia: Cognition And Vitamin D in African-American Women. J. Am. Geriatr. Soc. 2019, 67, 81–86. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hu, J.; Jia, J.; Zhang, Y.; Miao, R.; Huo, X.; Ma, F. Effects of Vitamin D3 Supplementation on Cognition and Blood Lipids: A 12-Month Randomised, Double-Blind, Placebo-Controlled Trial. J. Neurol. Neurosurg. Psychiatry 2018, 89, 1341–1347. [Google Scholar] [CrossRef] [PubMed]
- Zajac, I.T.; Barnes, M.; Cavuoto, P.; Wittert, G.; Noakes, M. The Effects of Vitamin D-Enriched Mushrooms and Vitamin D3 on Cognitive Performance and Mood in Healthy Elderly Adults: A Randomised, Double-Blinded, Placebo-Controlled Trial. Nutrients 2020, 12, 3847. [Google Scholar] [CrossRef] [PubMed]
- Ma, F.; Zhou, X.; Li, Q.; Zhao, J.; Song, A.; An, P.; Du, Y.; Xu, W.; Huang, G. Effects of Folic Acid and Vitamin B12, Alone and in Combination on Cognitive Function and Inflammatory Factors in the Elderly with Mild Cognitive Impairment: A Single-Blind Experimental Design. Curr. Alzheimer Res. 2019, 16, 622–632. [Google Scholar] [CrossRef]
- Decroix, L.; Van Schuerbeek, P.; Tonoli, C.; Van Cutsem, J.; Soares, D.D.; Heyman, E.; Vanderhasselt, T.; Verrelst, R.; Raeymaekers, H.; De Mey, J.; et al. The Effect of Acute Cocoa Flavanol Intake on the BOLD Response and Cognitive Function in Type 1 Diabetes: A Randomized, Placebo-Controlled, Double-Blinded Cross-over Pilot Study. Psychopharmacology 2019, 236, 3421–3428. [Google Scholar] [CrossRef] [PubMed]
- Ahles, S.; Stevens, Y.R.; Joris, P.J.; Vauzour, D.; Adam, J.; De Groot, E.; Plat, J. The Effect of Long-Term Aronia Melanocarpa Extract Supplementation on Cognitive Performance, Mood, and Vascular Function: A Randomized Controlled Trial in Healthy, Middle-Aged Individuals. Nutrients 2020, 12, E2475. [Google Scholar] [CrossRef]
- Cox, K.H.M.; White, D.J.; Pipingas, A.; Poorun, K.; Scholey, A. Further Evidence of Benefits to Mood and Working Memory from Lipidated Curcumin in Healthy Older People: A 12-Week, Double-Blind, Placebo-Controlled, Partial Replication Study. Nutrients 2020, 12, 1678. [Google Scholar] [CrossRef]
- Bensalem, J.; Dudonné, S.; Etchamendy, N.; Pellay, H.; Amadieu, C.; Gaudout, D.; Dubreuil, S.; Paradis, M.-E.; Pomerleau, S.; Capuron, L.; et al. Polyphenols from Grape and Blueberry Improve Episodic Memory in Healthy Elderly with Lower Level of Memory Performance: A Bicentric Double-Blind, Randomized, Placebo-Controlled Clinical Study. J. Gerontol. Ser. A 2019, 74, 996–1007. [Google Scholar] [CrossRef]
- Chai, S.C.; Jerusik, J.; Davis, K.; Wright, R.S.; Zhang, Z. Effect of Montmorency Tart Cherry Juice on Cognitive Performance in Older Adults: A Randomized Controlled Trial. Food Funct. 2019, 10, 4423–4431. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Herrlinger, K.A.; Nieman, K.M.; Sanoshy, K.D.; Fonseca, B.A.; Lasrado, J.A.; Schild, A.L.; Maki, K.C.; Wesnes, K.A.; Ceddia, M.A. Spearmint Extract Improves Working Memory in Men and Women with Age-Associated Memory Impairment. J. Altern. Complement. Med. 2018, 24, 37–47. [Google Scholar] [CrossRef] [PubMed]
- Hwang, Y.-H.; Park, S.; Paik, J.-W.; Chae, S.-W.; Kim, D.-H.; Jeong, D.-G.; Ha, E.; Kim, M.; Hong, G.; Park, S.-H.; et al. Efficacy and Safety of Lactobacillus Plantarum C29-Fermented Soybean (DW2009) in Individuals with Mild Cognitive Impairment: A 12-Week, Multi-Center, Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Nutrients 2019, 11, 305. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- You, Y.X.; Shahar, S.; Rajab, N.F.; Haron, H.; Yahya, H.M.; Mohamad, M.; Din, N.C.; Maskat, M.Y. Effects of 12 Weeks Cosmos Caudatus Supplement among Older Adults with Mild Cognitive Impairment: A Randomized, Double-Blind and Placebo-Controlled Trial. Nutrients 2021, 13, 434. [Google Scholar] [CrossRef]
- Ochiai, R.; Saitou, K.; Suzukamo, C.; Osaki, N.; Asada, T. Effect of Chlorogenic Acids on Cognitive Function in Mild Cognitive Impairment: A Randomized Controlled Crossover Trial. J. Alzheimers Dis. 2019, 72, 1209–1216. [Google Scholar] [CrossRef] [Green Version]
- Lau, H.; Shahar, S.; Mohamad, M.; Rajab, N.F.; Yahya, H.M.; Din, N.C.; Hamid, H.A. The Effects of Six Months Persicaria Minor Extract Supplement among Older Adults with Mild Cognitive Impairment: A Double-Blinded, Randomized, and Placebo-Controlled Trial. BMC Complement. Med. Ther. 2020, 20, 315. [Google Scholar] [CrossRef]
- Marriott, B.P.; Turner, T.H.; Hibbeln, J.R.; Newman, J.C.; Pregulman, M.; Malek, A.M.; Malcolm, R.J.; Burbelo, G.A.; Wismann, J.W. Impact of Fatty Acid Supplementation on Cognitive Performance among United States (US) Military Officers: The Ranger Resilience and Improved Performance on Phospholipid-Bound Omega-3’s (RRIPP-3) Study. Nutrients 2021, 13, 1854. [Google Scholar] [CrossRef]
- Giudici, K.V.; De Souto Barreto, P.; Beard, J.; Cantet, C.; Araujo de Carvalho, I.; Rolland, Y.; Vellas, B. MAPT DSA group Effect of Long-Term Omega-3 Supplementation and a Lifestyle Multidomain Intervention on Intrinsic Capacity among Community-Dwelling Older Adults: Secondary Analysis of a Randomized, Placebo-Controlled Trial (MAPT Study). Maturitas 2020, 141, 39–45. [Google Scholar] [CrossRef]
- Chhetri, J.K.; De Souto Barreto, P.; Cantet, C.; Pothier, K.; Cesari, M.; Andrieu, S.; Coley, N.; Vellas, B. Effects of a 3-Year Multi-Domain Intervention with or without Omega-3 Supplementation on Cognitive Functions in Older Subjects with Increased CAIDE Dementia Scores. J. Alzheimers Dis. 2018, 64, 71–78. [Google Scholar] [CrossRef]
- Tabue-Teguo, M.; Barreto de Souza, P.; Cantet, C.; Andrieu, S.; Simo, N.; Fougère, B.; Dartigues, J.F.; Vellas, B. Effect of Multidomain Intervention, Omega-3 Polyunsaturated Fatty Acids Supplementation or Their Combinaison on Cognitive Function in Non-Demented Older Adults According to Frail Status: Results from the MAPT Study. J. Nutr. Health Aging 2018, 22, 923–927. [Google Scholar] [CrossRef] [PubMed]
- Delrieu, J.; Voisin, T.; Saint-Aubert, L.; Carrie, I.; Cantet, C.; Vellas, B.; Payoux, P.; Andrieu, S. The Impact of a Multi-Domain Intervention on Cerebral Glucose Metabolism: Analysis from the Randomized Ancillary FDG PET MAPT Trial. Alzheimers Res. Ther. 2020, 12, 134. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, O.T.; Pillai, S.; Shankapal, P.; McLellan, A.; Kay, D.G.; Gold, B.T.; Keller, J.N. A Combination of Essential Fatty Acids, Panax Ginseng Extract, and Green Tea Catechins Modifies Brain FMRI Signals in Healthy Older Adults. J. Nutr. Health Aging 2018, 22, 837–846. [Google Scholar] [CrossRef] [PubMed]
- Danthiir, V.; Hosking, D.E.; Nettelbeck, T.; Vincent, A.D.; Wilson, C.; O’Callaghan, N.; Calvaresi, E.; Clifton, P.; Wittert, G.A. An 18-Mo Randomized, Double-Blind, Placebo-Controlled Trial of DHA-Rich Fish Oil to Prevent Age-Related Cognitive Decline in Cognitively Normal Older Adults. Am. J. Clin. Nutr. 2018, 107, 754–762. [Google Scholar] [CrossRef] [Green Version]
- Stavrinou, S.P.; Andreou, E.; Aphamis, G.; Pantzaris, M.; Ioannou, M.; Patrikios, S.I.; Giannaki, D.C. The Effects of a 6-Month High Dose Omega-3 and Omega-6 Polyunsaturated Fatty Acids and Antioxidant Vitamins Supplementation on Cognitive Function and Functional Capacity in Older Adults with Mild Cognitive Impairment. Nutrients 2020, 12, 325. [Google Scholar] [CrossRef] [Green Version]
- Kuszewski, J.C.; Howe, P.R.C.; Wong, R.H.X. Evaluation of Cognitive Performance Following Fish-Oil and Curcumin Supplementation in Middle-Aged and Older Adults with Overweight or Obesity. J. Nutr. 2020, 150, 3190–3199. [Google Scholar] [CrossRef]
- McNamara, R.K.; Kalt, W.; Shidler, M.D.; McDonald, J.; Summer, S.S.; Stein, A.L.; Stover, A.N.; Krikorian, R. Cognitive Response to Fish Oil, Blueberry, and Combined Supplementation in Older Adults with Subjective Cognitive Impairment. Neurobiol. Aging 2018, 64, 147–156. [Google Scholar] [CrossRef]
- Li, M.; Li, W.; Gao, Y.; Chen, Y.; Bai, D.; Weng, J.; Du, Y.; Ma, F.; Wang, X.; Liu, H.; et al. Effect of Folic Acid Combined with Docosahexaenoic Acid Intervention on Mild Cognitive Impairment in Elderly: A Randomized Double-Blind, Placebo-Controlled Trial. Eur. J. Nutr. 2021, 60, 1795–1808. [Google Scholar] [CrossRef]
- Arellanes, I.C.; Choe, N.; Solomon, V.; He, X.; Kavin, B.; Martinez, A.E.; Kono, N.; Buennagel, D.P.; Hazra, N.; Kim, G.; et al. Brain Delivery of Supplemental Docosahexaenoic Acid (DHA): A Randomized Placebo-Controlled Clinical Trial. EBioMedicine 2020, 59, 102883. [Google Scholar] [CrossRef]
- Schwarz, C.; Wirth, M.; Gerischer, L.; Grittner, U.; Witte, A.V.; Köbe, T.; Flöel, A. Effects of Omega-3 Fatty Acids on Resting Cerebral Perfusion in Patients with Mild Cognitive Impairment: A Randomized Controlled Trial. J. Prev. Alzheimers Dis. 2018, 5, 26–30. [Google Scholar] [CrossRef]
- Ito, N.; Saito, H.; Seki, S.; Ueda, F.; Asada, T. Effects of Composite Supplement Containing Astaxanthin and Sesamin on Cognitive Functions in People with Mild Cognitive Impairment: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Alzheimers Dis. 2018, 62, 1767–1775. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Igase, M.; Okada, Y.; Ochi, M.; Igase, K.; Ochi, H.; Okuyama, S.; Furukawa, Y.; Ohyagi, Y. Auraptene in The Peels of Citrus Kawachiensis (Kawachibankan) Contributes to the Preservation of Cognitive Function: A Randomized, Placebo-Controlled, Double-Blind Study in Healthy Volunteers. J. Prev. Alzheimers Dis. 2017, 5, 197–201. [Google Scholar] [CrossRef]
- Tohda, C.; Matsui, M.; Inada, Y.; Yang, X.; Kuboyama, T.; Kimbara, Y.; Watari, H. Combined Treatment with Two Water Extracts of Eleutherococcus Senticosus Leaf and Rhizome of Drynaria Fortunei Enhances Cognitive Function: A Placebo-Controlled, Randomized, Double-Blind Study in Healthy Adults. Nutrients 2020, 12, 303. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saitsu, Y.; Nishide, A.; Kikushima, K.; Shimizu, K.; Ohnuki, K. Improvement of Cognitive Functions by Oral Intake of Hericium Erinaceus. Biomed. Res. 2019, 40, 125–131. [Google Scholar] [CrossRef] [Green Version]
- Reid, S.N.S.; Ryu, J.; Kim, Y.; Jeon, B.H. The Effects of Fermented Laminaria Japonica on Short-Term Working Memory and Physical Fitness in the Elderly. Evid. Based Complement. Alternat. Med. 2018, 2018, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Perry, N.S.L.; Menzies, R.; Hodgson, F.; Wedgewood, P.; Howes, M.-J.R.; Brooker, H.J.; Wesnes, K.A.; Perry, E.K. A Randomised Double-Blind Placebo-Controlled Pilot Trial of a Combined Extract of Sage, Rosemary and Melissa, Traditional Herbal Medicines, on the Enhancement of Memory in Normal Healthy Subjects, Including Influence of Age. Phytomedicine 2018, 39, 42–48. [Google Scholar] [CrossRef]
- Zhu, X.; Borenstein, A.R.; Zheng, Y.; Zhang, W.; Seidner, D.L.; Ness, R.; Murff, H.J.; Li, B.; Shrubsole, M.J.; Yu, C.; et al. Ca:Mg Ratio, APOE Cytosine Modifications, and Cognitive Function: Results from a Randomized Trial. J. Alzheimers Dis. 2020, 75, 85–98. [Google Scholar] [CrossRef]
- Boyle, N.B.; Dye, L.; Arkbåge, K.; Thorell, L.; Frederiksen, P.; Croden, F.; Lawton, C. Effects of Milk-Based Phospholipids on Cognitive Performance and Subjective Responses to Psychosocial Stress: A Randomized, Double-Blind, Placebo-Controlled Trial in High-Perfectionist Men. Nutrition 2019, 57, 183–193. [Google Scholar] [CrossRef]
- Abe, S.; Ezaki, O.; Suzuki, M. Medium-Chain Triglycerides (8:0 and 10:0) Increase Mini-Mental State Examination (MMSE) Score in Frail Elderly Adults in a Randomized Controlled Trial. J. Nutr. 2020, 150, 2383–2390. [Google Scholar] [CrossRef]
- Fukuda, T.; Obara, K.; Saito, J.; Umeda, S.; Ano, Y. Effects of Hop Bitter Acids, Bitter Components in Beer, on Cognition in Healthy Adults: A Randomized Controlled Trial. J. Agric. Food Chem. 2020, 68, 206–212. [Google Scholar] [CrossRef]
- Fukuda, T.; Ohnuma, T.; Obara, K.; Kondo, S.; Arai, H.; Ano, Y. Supplementation with Matured Hop Bitter Acids Improves Cognitive Performance and Mood State in Healthy Older Adults with Subjective Cognitive Decline. J. Alzheimers Dis. 2020, 76, 387–398. [Google Scholar] [CrossRef]
- Ban, S.; Lee, S.L.; Jeong, H.S.; Lim, S.M.; Park, S.; Hong, Y.S.; Kim, J.E. Efficacy and Safety of Tremella Fuciformis in Individuals with Subjective Cognitive Impairment: A Randomized Controlled Trial. J. Med. Food 2018, 21, 400–407. [Google Scholar] [CrossRef]
- Wirth, M.; Benson, G.; Schwarz, C.; Köbe, T.; Grittner, U.; Schmitz, D.; Sigrist, S.J.; Bohlken, J.; Stekovic, S.; Madeo, F.; et al. The Effect of Spermidine on Memory Performance in Older Adults at Risk for Dementia: A Randomized Controlled Trial. Cortex 2018, 109, 181–188. [Google Scholar] [CrossRef]
- Baleztena, J.; Ruiz-Canela, M.; Sayon-Orea, C.; Pardo, M.; Añorbe, T.; Gost, J.I.; Gomez, C.; Ilarregui, B.; Bes-Rastrollo, M. Association between Cognitive Function and Supplementation with Omega-3 PUFAs and Other Nutrients in ≥ 75 Years Old Patients: A Randomized Multicenter Study. PLoS ONE 2018, 13, e0193568. [Google Scholar] [CrossRef] [Green Version]
- Tadokoro, K.; Morihara, R.; Ohta, Y.; Hishikawa, N.; Kawano, S.; Sasaki, R.; Matsumoto, N.; Nomura, E.; Nakano, Y.; Takahashi, Y.; et al. Clinical Benefits of Antioxidative Supplement Twendee X for Mild Cognitive Impairment: A Multicenter, Randomized, Double-Blind, and Placebo-Controlled Prospective Interventional Study. J. Alzheimers Dis. 2019, 71, 1063–1069. [Google Scholar] [CrossRef]
- Szczechowiak, K.; Diniz, B.S.; Leszek, J. Diet and Alzheimer’s Dementia—Nutritional Approach to Modulate Inflammation. Pharmacol. Biochem. Behav. 2019, 184, 172743. [Google Scholar] [CrossRef]
- Coelho-Júnior, H.J.; Trichopoulou, A.; Panza, F. Cross-Sectional and Longitudinal Associations between Adherence to Mediterranean Diet with Physical Performance and Cognitive Function in Older Adults: A Systematic Review and Meta-Analysis. Ageing Res. Rev. 2021, 70, 101395. [Google Scholar] [CrossRef] [PubMed]
- Valls-Pedret, C.; Sala-Vila, A.; Serra-Mir, M.; Corella, D.; De la Torre, R.; Martínez-González, M.Á.; Martínez-Lapiscina, E.H.; Fitó, M.; Pérez-Heras, A.; Salas-Salvadó, J.; et al. Mediterranean Diet and Age-Related Cognitive Decline: A Randomized Clinical Trial. JAMA Intern. Med. 2015, 175, 1094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wengreen, H.; Munger, R.G.; Cutler, A.; Quach, A.; Bowles, A.; Corcoran, C.; Tschanz, J.T.; Norton, M.C.; Welsh-Bohmer, K.A. Prospective Study of Dietary Approaches to Stop Hypertension- and Mediterranean-Style Dietary Patterns and Age-Related Cognitive Change: The Cache County Study on Memory, Health and Aging123. Am. J. Clin. Nutr. 2013, 98, 1263–1271. [Google Scholar] [CrossRef] [PubMed]
- Fiocco, A.J.; Shatenstein, B.; Ferland, G.; Payette, H.; Belleville, S.; Kergoat, M.-J.; Morais, J.A.; Greenwood, C.E. Sodium Intake and Physical Activity Impact Cognitive Maintenance in Older Adults: The NuAge Study. Neurobiol. Aging 2012, 33, 829.e21–829.e28. [Google Scholar] [CrossRef]
- Heye, A.K.; Thrippleton, M.J.; Chappell, F.M.; Valdés Hernández, M.; Del C. Valdés Hernández, M.; Armitage, P.A.; Makin, S.D.; Muñoz Maniega, S.; Sakka, E.; Flatman, P.W.; et al. Blood Pressure and Sodium: Association with MRI Markers in Cerebral Small Vessel Disease. J. Cereb. Blood Flow Metab. 2016, 36, 264–274. [Google Scholar] [CrossRef] [Green Version]
- Jang, H.; Lee, S.; Choi, S.L.; Kim, H.Y.; Baek, S.; Kim, Y. Taurine Directly Binds to Oligomeric Amyloid-β and Recovers Cognitive Deficits in Alzheimer Model Mice. Adv. Exp. Med. Biol. 2017, 975 Pt 1, 233–241. [Google Scholar] [CrossRef]
- Sultan, S.; Taimuri, U.; Basnan, S.A.; Ai-Orabi, W.K.; Awadallah, A.; Almowald, F.; Hazazi, A. Low Vitamin D and Its Association with Cognitive Impairment and Dementia. J. Aging Res. 2020, 2020, 6097820. [Google Scholar] [CrossRef]
- Kwok, T.; Lee, J.; Ma, R.C.; Wong, S.Y.; Kung, K.; Lam, A.; Ho, C.S.; Lee, V.; Harrison, J.; Lam, L. A Randomized Placebo Controlled Trial of Vitamin B12 Supplementation to Prevent Cognitive Decline in Older Diabetic People with Borderline Low Serum Vitamin B12. Clin. Nutr. 2017, 36, 1509–1515. [Google Scholar] [CrossRef]
- Keane, K.M.; Haskell-Ramsay, C.F.; Veasey, R.C.; Howatson, G. Montmorency Tart Cherries (Prunus cerasus L.) Modulate Vascular Function Acutely, in the Absence of Improvement in Cognitive Performance. Br. J. Nutr. 2016, 116, 1935–1944. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zehr, K.R.; Walker, M.K. Omega-3 Polyunsaturated Fatty Acids Improve Endothelial Function in Humans at Risk for Atherosclerosis: A Review. Prostaglandins Other Lipid Mediat. 2018, 134, 131–140. [Google Scholar] [CrossRef]
- Joris, P.J.; Mensink, R.P.; Adam, T.C.; Liu, T.T. Cerebral Blood Flow Measurements in Adults: A Review on the Effects of Dietary Factors and Exercise. Nutrients 2018, 10, 530. [Google Scholar] [CrossRef] [Green Version]
- Derbyshire, E. Brain Health across the Lifespan: A Systematic Review on the Role of Omega-3 Fatty Acid Supplements. Nutrients 2018, 10, 1094. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Konagai, C.; Yanagimoto, K.; Hayamizu, K.; Han, L.; Tsuji, T.; Koga, Y. Effects of Krill Oil Containing N-3 Polyunsaturated Fatty Acids in Phospholipid Form on Human Brain Function: A Randomized Controlled Trial in Healthy Elderly Volunteers. Clin. Interv. Aging 2013, 8, 1247–1257. [Google Scholar] [CrossRef] [Green Version]
- Cook, R.L.; Parker, H.M.; Donges, C.E.; O’Dwyer, N.J.; Cheng, H.L.; Steinbeck, K.S.; Cox, E.P.; Franklin, J.L.; Garg, M.L.; O’Connor, H.T. Omega-3 Polyunsaturated Fatty Acids Status and Cognitive Function in Young Women. Lipids Health Dis. 2019, 18, 194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Pi, J.; Woods, C.G.; Andersen, M.E. A Systems Biology Perspective on Nrf2-Mediated Antioxidant Response. Toxicol. Appl. Pharmacol. 2010, 244, 84–97. [Google Scholar] [CrossRef] [Green Version]
- Restrepo, M.S.L.; Morales, G.R.M.; Ramírez, G.M.C.; López, L.M.V.; Varela, L.L.E. Los Hábitos Alimentarios En El Adulto Mayor Y Su Relación Con Los Procesos Protectores Y Deteriorantes En Salud. Rev. Chil. Nutr. 2006, 33, 500–510. [Google Scholar] [CrossRef]
- Jia, X.; McNeill, G.; Avenell, A. Does Taking Vitamin, Mineral and Fatty Acid Supplements Prevent Cognitive Decline? A Systematic Review of Randomized Controlled Trials. J. Hum. Nutr. Diet. 2008, 21, 317–336. [Google Scholar] [CrossRef] [PubMed]
- Forbes, S.C.; Holroyd-Leduc, J.M.; Poulin, M.J.; Hogan, D.B. Effect of Nutrients, Dietary Supplements and Vitamins on Cognition: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Can. Geriatr. J. 2015, 18, 231–245. [Google Scholar] [CrossRef] [Green Version]
- Pugazhenthi, S.; Qin, L.; Reddy, P.H. Common Neurodegenerative Pathways in Obesity, Diabetes, and Alzheimer’s Disease. Biochim. Biophys. Acta Mol. Basis Dis. 2017, 1863, 1037–1045. [Google Scholar] [CrossRef]
- Palomar-Bonet, M.; Atienza, M.; Cantero, J.L. Blood Total Antioxidant Status Is Associated with Cortical Glucose Uptake and Factors Related to Accelerated Aging. Brain Struct. Funct. 2020, 225, 841–851. [Google Scholar] [CrossRef]
- Palomar-Bonet, M.; Atienza, M.; Hernández-Ledesma, B.; Cantero, J.L. Associations of Salivary Total Antioxidant Capacity with Cortical Amyloid-Beta Burden, Cortical Glucose Uptake, and Cognitive Function in Normal Aging. J. Gerontol. Ser. A 2021, 76, 1839–1845. [Google Scholar] [CrossRef]
- McGrattan, A.M.; McGuinness, B.; McKinley, M.C.; Kee, F.; Passmore, P.; Woodside, J.V.; McEvoy, C.T. Diet and Inflammation in Cognitive Ageing and Alzheimer’s Disease. Curr. Nutr. Rep. 2019, 8, 53–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Román, G.C.; Jackson, R.E.; Gadhia, R.; Román, A.N.; Reis, J. Mediterranean Diet: The Role of Long-Chain ω-3 Fatty Acids in Fish; Polyphenols in Fruits, Vegetables, Cereals, Coffee, Tea, Cacao and Wine; Probiotics and Vitamins in Prevention of Stroke, Age-Related Cognitive Decline, and Alzheimer Disease. Rev. Neurol. 2019, 175, 724–741. [Google Scholar] [CrossRef] [PubMed]
- Dyall, S.C. Long-Chain Omega-3 Fatty Acids and the Brain: A Review of the Independent and Shared Effects of EPA, DPA and DHA. Front. Aging Neurosci. 2015, 7, 52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Díaz, M.; Mesa-Herrera, F.; Marín, R. DHA and Its Elaborated Modulation of Antioxidant Defenses of the Brain: Implications in Aging and AD Neurodegeneration. Antioxidants 2021, 10, 907. [Google Scholar] [CrossRef]
- Romanenko, M.; Kholin, V.; Koliada, A.; Vaiserman, A. Nutrition, Gut Microbiota, and Alzheimer’s Disease. Front. Psychiatry 2021, 12, 712673. [Google Scholar] [CrossRef]
- Annunziata, G.; Sureda, A.; Orhan, I.E.; Battino, M.; Arnone, A.; Jiménez-García, M.; Capó, X.; Cabot, J.; Sanadgol, N.; Giampieri, F.; et al. The Neuroprotective Effects of Polyphenols, Their Role in Innate Immunity and the Interplay with the Microbiota. Neurosci. Biobehav. Rev. 2021, 128, 437–453. [Google Scholar] [CrossRef] [PubMed]
- Sun, G.Y.; Simonyi, A.; Fritsche, K.L.; Chuang, D.Y.; Hannink, M.; Gu, Z.; Greenlief, C.M.; Yao, J.K.; Lee, J.C.; Beversdorf, D.Q. Docosahexaenoic Acid (DHA): An Essential Nutrient and a Nutraceutical for Brain Health and Diseases. Prostaglandins Leukot. Essent. Fat. Acids 2018, 136, 3–13. [Google Scholar] [CrossRef]
- Marosi, K.; Mattson, M.P. BDNF Mediates Adaptive Brain and Body Responses to Energetic Challenges. Trends Endocrinol. Metab. 2014, 25, 89–98. [Google Scholar] [CrossRef] [Green Version]
- Gómez-Pinilla, F. Brain Foods: The Effects of Nutrients on Brain Function. Nat. Rev. Neurosci. 2008, 9, 568–578. [Google Scholar] [CrossRef] [Green Version]
- Wu, A.; Ying, Z.; Gomez-Pinilla, F. Dietary Omega-3 Fatty Acids Normalize BDNF Levels, Reduce Oxidative Damage, and Counteract Learning Disability after Traumatic Brain Injury in Rats. J. Neurotrauma 2004, 21, 1457–1467. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pifferi, F.; Roux, F.; Langelier, B.; Alessandri, J.-M.; Vancassel, S.; Jouin, M.; Lavialle, M.; Guesnet, P. (N-3) Polyunsaturated Fatty Acid Deficiency Reduces the Expression of Both Isoforms of the Brain Glucose Transporter GLUT1 in Rats. J. Nutr. 2005, 135, 2241–2246. [Google Scholar] [CrossRef] [Green Version]
- Flachs, P.; Horakova, O.; Brauner, P.; Rossmeisl, M.; Pecina, P.; Franssen-van Hal, N.; Ruzickova, J.; Sponarova, J.; Drahota, Z.; Vlcek, C.; et al. Polyunsaturated Fatty Acids of Marine Origin Upregulate Mitochondrial Biogenesis and Induce Beta-Oxidation in White Fat. Diabetologia 2005, 48, 2365–2375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petersson, S.D.; Philippou, E. Mediterranean Diet, Cognitive Function, and Dementia: A Systematic Review of the Evidence123. Adv. Nutr. 2016, 7, 889–904. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Rejón, A.I.; Castro-Quezada, I.; Ruano-Rodríguez, C.; Ruiz-López, M.D.; Sánchez-Villegas, A.; Toledo, E.; Artacho, R.; Estruch, R.; Salas-Salvadó, J.; Covas, M.I.; et al. Effect of a Mediterranean Diet Intervention on Dietary Glycemic Load and Dietary Glycemic Index: The predimed Study. J. Nutr. Metab. 2014, 2014, 985373. [Google Scholar] [CrossRef] [Green Version]
- Medawar, E.; Huhn, S.; Villringer, A.; Veronica Witte, A. The Effects of Plant-Based Diets on the Body and the Brain: A Systematic Review. Transl. Psychiatry 2019, 9, 226. [Google Scholar] [CrossRef] [PubMed]
- Spencer, S.J.; Korosi, A.; Layé, S.; Shukitt-Hale, B.; Barrientos, R.M. Food for Thought: How Nutrition Impacts Cognition and Emotion. Npj Sci. Food 2017, 1, 7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vellas, B.; Carrie, I.; Gillette-Guyonnet, S.; Touchon, J.; Dantoine, T.; Dartigues, J.F.; Cuffi, M.N.; Bordes, S.; Gasnier, Y.; Robert, P.; et al. Mapt Study: A Multidomain Approach for Preventing Alzheimer’s Disease: Design And Baseline Data. J. Prev. Alzheimers Dis. 2014, 1, 13–22. [Google Scholar]
- Kivipelto, M.; Solomon, A.; Ahtiluoto, S.; Ngandu, T.; Lehtisalo, J.; Antikainen, R.; Bäckman, L.; Hänninen, T.; Jula, A.; Laatikainen, T.; et al. The Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER): Study Design and Progress. Alzheimers Dement. 2013, 9, 657–665. [Google Scholar] [CrossRef] [PubMed]
- Tooley, K.L. Effects of the Human Gut Microbiota on Cognitive Performance, Brain Structure and Function: A Narrative Review. Nutrients 2020, 12, 3009. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gutierrez, L.; Folch, A.; Rojas, M.; Cantero, J.L.; Atienza, M.; Folch, J.; Camins, A.; Ruiz, A.; Papandreou, C.; Bulló, M. Effects of Nutrition on Cognitive Function in Adults with or without Cognitive Impairment: A Systematic Review of Randomized Controlled Clinical Trials. Nutrients 2021, 13, 3728. https://doi.org/10.3390/nu13113728
Gutierrez L, Folch A, Rojas M, Cantero JL, Atienza M, Folch J, Camins A, Ruiz A, Papandreou C, Bulló M. Effects of Nutrition on Cognitive Function in Adults with or without Cognitive Impairment: A Systematic Review of Randomized Controlled Clinical Trials. Nutrients. 2021; 13(11):3728. https://doi.org/10.3390/nu13113728
Chicago/Turabian StyleGutierrez, Laia, Alexandre Folch, Melina Rojas, José Luis Cantero, Mercedes Atienza, Jaume Folch, Antoni Camins, Agustín Ruiz, Christopher Papandreou, and Mònica Bulló. 2021. "Effects of Nutrition on Cognitive Function in Adults with or without Cognitive Impairment: A Systematic Review of Randomized Controlled Clinical Trials" Nutrients 13, no. 11: 3728. https://doi.org/10.3390/nu13113728
APA StyleGutierrez, L., Folch, A., Rojas, M., Cantero, J. L., Atienza, M., Folch, J., Camins, A., Ruiz, A., Papandreou, C., & Bulló, M. (2021). Effects of Nutrition on Cognitive Function in Adults with or without Cognitive Impairment: A Systematic Review of Randomized Controlled Clinical Trials. Nutrients, 13(11), 3728. https://doi.org/10.3390/nu13113728








