Ketogenic Diet for Cancer: Critical Assessment and Research Recommendations
Abstract
1. Introduction
2. Ketosis and Spectrum of Ketogenic Diets (KD)
3. KD as a Therapeutic for Cancer: Hypothesized Mechanisms
4. Preclinical Studies of KD for Cancer
5. Clinical Studies of KD and Cancer
6. Limitations of Current Literature
7. Conclusions and Future Directions
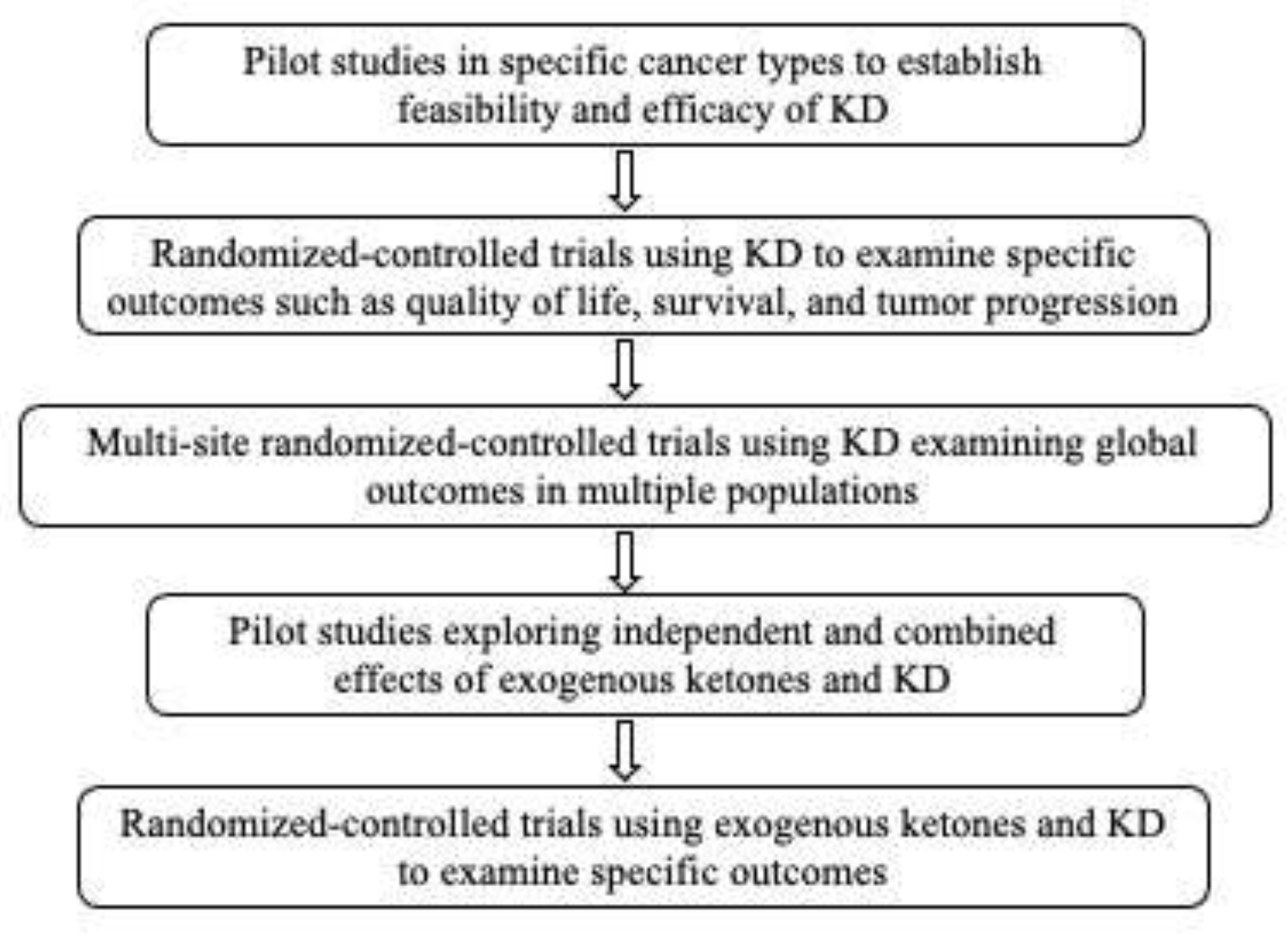
8. Research Recommendations for Moving the Field Forward
- Conduct small, rigorous non-randomized trials with homogeneous patient groups and common cancer sites to assess whether KD produces a “signal” on selected outcomes (particularly those related to response to standard care (e.g., effectiveness, side effects)) that would justify the conduct of larger, randomized-controlled trials.
- In randomized-controlled trials, provide sufficient detail of the KD and control diets (ensuring that they are comparable on vitamins, mineral and other nutrients) so they could be replicated by other investigators.
- Develop a standardized method to monitor and quantify adherence and tolerance to the KD (e.g., [93]).
- Develop a set of standardized assessments and outcome measures that include the full array of relevant variables (e.g., imaging of tumor characteristics, body composition, quality of life, and survival).
- Distinguish trials based on whether they attempt to isolate the unique effects of KD versus those which seek to estimate its effects as an adjunctive therapy.
- Examine the effects of exogenous ketones, alone and in conjunction with a KD, to determine whether they have synergistic or additive effects.
- Because it is unlikely that KD will cure cancer, trials should focus on whether KD reduces cancer progression or recurrence in those who experience remission through standard care.
Author Contributions
Funding
Conflicts of Interest
References
- Mukherjee, S.; Ghoshal, S. The Emperor of All Maladies: A Biography of Cancer. J. Postgrad. Med. Educ. Res. 2012, 46, 112. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer Statistics, 2021. CA Cancer J. Clin. 2021, 71, 7–33. [Google Scholar] [CrossRef]
- Rahib, L.; Wehner, M.R.; Matrisian, L.M.; Nead, K.T. Estimated Projection of US Cancer Incidence and Death to 2040. JAMA Netw. Open 2021, 4, e214708. [Google Scholar] [CrossRef]
- Doll, R.; Peto, R. The causes of cancer: Quantitative estimates of avoidable risks of cancer in the United States today. J. Natl. Cancer Inst. 1981, 66, 1192–1308. [Google Scholar] [CrossRef]
- Blot, W.J.; Tarone, R.E. Doll and Peto’s quantitative estimates of cancer risks: Holding generally true for 35 years. J. Natl. Cancer Inst. 2015, 107, djv044. [Google Scholar] [CrossRef]
- Steele, C.B.; el Huda, N.; Abdullah, I. Vital Signs: Trends in Incidence of Cancers Associated with Overweight and Obesity—United States, 2005–2014. MMWR Morb. Mortal. Wkly. Rep. 2017, 66, 1052–1058. [Google Scholar] [CrossRef]
- Seyfried, T.N.; Shelton, L.M. Cancer as a metabolic disease. Nutr. Metab. 2010, 7, 7. [Google Scholar] [CrossRef]
- Steck, S.E.; Murphy, E.A. Dietary patterns and cancer risk. Nat. Rev. Cancer 2019, 20, 125–138. [Google Scholar] [CrossRef] [PubMed]
- Kant, A.K. Dietary patterns and health outcomes. J. Am. Diet. Assoc. 2004, 104, 615–635. [Google Scholar] [CrossRef] [PubMed]
- Fabiani, R.; Minelli, L.; Bertarelli, G.; Bacci, S. A Western Dietary Pattern Increases Prostate Cancer Risk: A Systematic Review and Meta-Analysis. Nutrients 2016, 8, 626. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-F.; Yao, A.-L.; Sun, Y.-Y.; Zhang, A.-H. Empirically derived dietary patterns and ovarian cancer risk: A meta-analysis. Eur. J. Cancer Prev. 2018, 27, 493–501. [Google Scholar] [CrossRef]
- Xiao, Y.; Xia, J.; Li, L.; Ke, Y.; Cheng, J.; Xie, Y.; Chu, W.; Cheung, P.; Kim, J.H.; Colditz, G.A.; et al. Associations between dietary patterns and the risk of breast cancer: A systematic review and meta-analysis of observational studies. Breast Cancer Res. 2019, 21, 16. [Google Scholar] [CrossRef] [PubMed]
- Gray, A.; Dang, B.N.; Moore, T.B.; Clemens, R.; Pressman, P. A review of nutrition and dietary interventions in oncology. SAGE Open Med. 2020, 8. [Google Scholar] [CrossRef] [PubMed]
- Weber, D.D.; Aminzadeh-Gohari, S.; Tulipan, J. Ketogenic diet in the treatment of cancer—Where do we stand? Mol Metab. 2020, 33, 102–121. [Google Scholar] [CrossRef]
- Yang, Y.-F.; Mattamel, P.; Joseph, T.; Huang, J.; Chen, Q.; Akinwunmi, B.; Zhang, C.; Ming, W.-K. Efficacy of Low-Carbohydrate Ketogenic Diet as an Adjuvant Cancer Therapy: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2021, 13, 1388. [Google Scholar] [CrossRef]
- Römer, M.; Dörfler, J.; Huebner, J. The use of ketogenic diets in cancer patients: A systematic review. Clin. Exp. Med. 2021, 21, 1–36. [Google Scholar]
- Oliveira, C.L.; Mattingly, S.; Schirrmacher, R.; Sawyer, M.B.; Fine, E.J.; Prado, C.M. A Nutritional Perspective of Ketogenic Diet in Cancer: A Narrative Review. J. Acad. Nutr. Diet. 2017, 118, 668–688. [Google Scholar] [CrossRef] [PubMed]
- Sremanakova, J.; Sowerbutts, A.M.; Burden, S. A systematic review of the use of ketogenic diets in adult patients with cancer. J. Hum. Nutr. Diet. 2018, 31, 793–802. [Google Scholar] [CrossRef]
- Gershuni, V.M.; Yan, S.L.; Medici, V. Nutritional Ketosis for Weight Management and Reversal of Metabolic Syndrome. Curr. Nutr. Rep. 2018, 7, 97–106. [Google Scholar] [CrossRef]
- Allen, B.G.; Bhatia, S.K.; Anderson, C.; Eichenberger-Gilmore, J.M.; Sibenaller, Z.A.; Mapuskar, K.A.; Schoenfeld, J.D.; Buatti, J.; Spitz, D.; Fath, M. Ketogenic diets as an adjuvant cancer therapy: History and potential mechanism. Redox Biol. 2014, 2, 963–970. [Google Scholar] [CrossRef]
- Paoli, A.; Rubini, A.; Volek, J.S.; Grimaldi, K.A. Beyond weight loss: A review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur. J. Clin. Nutr. 2013, 67, 789–796. [Google Scholar] [CrossRef]
- Lane J, F.K. Ketogenic Diet and Health. OBM Integr. Complementary Med. 2021, 6. [Google Scholar] [CrossRef]
- Messer, R.D.K.; Eric, H. Ketogenic Diets for the Treatment of Epilepsy. Bioactive Nutraceuticals and Dietary Supplements in Neurological and Brain Disease: Prevention and Therapy; Academic Press: Cambridge, MA, USA, 2015; pp. 441–448. [Google Scholar]
- Di Lorenzo, C.; Ballerini, G.; Barbanti, P.; Bernardini, A.; D’Arrigo, G.; Egeo, G.; Frediani, F.; Garbo, R.; Pierangeli, G.; Prudenzano, M.; et al. Applications of Ketogenic Diets in Patients with Headache: Clinical Recommendations. Nutrients 2021, 13, 2307. [Google Scholar] [CrossRef] [PubMed]
- Martuscello, R.T.; Vedam-Mai, V.; McCarthy, D.J.; Schmoll, M.E.; Jundi, M.A.; Louviere, C.D.; Griffith, B.G.; Skinner, C.L.; Suslov, O.; Deleyrolle, L.P.; et al. A Supplemented High-Fat Low-Carbohydrate Diet for the Treatment of Glioblastoma. Clin. Cancer Res. 2015, 22, 2482–2495. [Google Scholar] [CrossRef] [PubMed]
- Prince, A.; Zhang, Y.; Croniger, C.; Puchowicz, M. Oxidative Metabolism: Glucose Versus Ketones. Adv. Exp. Med. Biol. 2013, 789, 323–328. [Google Scholar] [PubMed]
- Fine, E.J.; Segal-Isaacson, C.; Feinman, R.D.; Herszkopf, S.; Romano, M.C.; Tomuta, N.; Bontempo, A.F.; Negassa, A.; Sparano, J.A. Targeting insulin inhibition as a metabolic therapy in advanced cancer: A pilot safety and feasibility dietary trial in 10 patients. Nutrition 2012, 28, 1028–1035. [Google Scholar] [CrossRef]
- Youm, Y.-H.; Nguyen, K.Y.; Grant, R.; Goldberg, E.L.; Bodogai, M.; Kim, D.; D’Agostino, D.; Planavsky, N.J.; Lupfer, C.; Kanneganti, T.-D.; et al. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome–mediated inflammatory disease. Nat. Med. 2015, 21, 263–269. [Google Scholar] [CrossRef] [PubMed]
- Otto, A.M. Warburg effect(s)—A biographical sketch of Otto Warburg and his impacts on tumor metabolism. Cancer Metab. 2016, 4, 1–8. [Google Scholar] [CrossRef]
- Warburg, O. On the Origin of Cancer Cells. Science 1956, 123, 309–314. [Google Scholar] [CrossRef] [PubMed]
- Trabold, O.; Wagner, S.; Wicke, C.; Bs, H.S.; Hussain, M.Z.; Rosen, N.; Bs, A.S.; Becker, H.D.; Hunt, T.K. Lactate and oxygen constitute a fundamental regulatory mechanism in wound healing. Wound Repair Regen. 2003, 11, 504–509. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Lien, E.C.; Lyssiotis, A.; Cantley, L.C. Metabolic Reprogramming by the PI3K-Akt-mTOR Pathway in Cancer. Recent Results Cancer Res. 2016, 207, 39–72. [Google Scholar]
- Heiden, M.G.V.; Cantley, L.C.; Thompson, C.B. Understanding the Warburg Effect: The Metabolic Requirements of Cell Proliferation. Science 2009, 324, 1029–1033. [Google Scholar] [CrossRef]
- DeNicola, G.; Cantley, L.C. Cancer’s Fuel Choice: New Flavors for a Picky Eater. Mol. Cell 2015, 60, 514–523. [Google Scholar] [CrossRef]
- Wu, N.; Zheng, B.; Shaywitz, A.; Dagon, Y.; Tower, C.; Bellinger, G.; Shen, C.-H.; Wen, J.; Asara, J.; McGraw, T.E.; et al. AMPK-Dependent Degradation of TXNIP upon Energy Stress Leads to Enhanced Glucose Uptake via GLUT1. Mol. Cell 2013, 49, 1167–1175. [Google Scholar] [CrossRef]
- Carracedo, A.; Cantley, L.; Pandolfi, P.P. Cancer metabolism: Fatty acid oxidation in the limelight. Nat. Rev. Cancer 2013, 13, 227–232. [Google Scholar] [CrossRef]
- Vidali, S.; Aminzadeh, S.; Lambert, B.; Rutherford, T.; Sperl, W.; Kofler, B.; Feichtinger, R.G. Mitochondria: The ketogenic diet—A metabolism-based therapy. Int. J. Biochem. Cell Biol. 2015, 63, 55–59. [Google Scholar] [CrossRef]
- Branco, A.F.; Ferreira, A.; Simões, R.; Magalhaes-Novais, S.; Zehowski, C.; Cope, E.; Silva, A.M.; Pereira, D.; Sardao, V.; Cunha-Oliveira, T. Ketogenic diets: From cancer to mitochondrial diseases and beyond. Eur. J. Clin. Investig. 2016, 46, 285–298. [Google Scholar] [CrossRef]
- Otto, C.; Kaemmerer, U.; Illert, B.; Muehling, B.; Pfetzer, N.; Wittig, R.; Voelker, H.U.; Thiede, A.; Coy, J.F. Growth of human gastric cancer cells in nude mice is delayed by a ketogenic diet supplemented with omega-3 fatty acids and medium-chain triglycerides. BMC Cancer 2008, 8, 122. [Google Scholar] [CrossRef]
- Lussier, D.M.; Woolf, E.C.; Johnson, J.L.; Brooks, K.S.; Blattman, J.N.; Scheck, A.C. Enhanced immunity in a mouse model of malignant glioma is mediated by a therapeutic ketogenic diet. BMC Cancer 2016, 16, 310. [Google Scholar] [CrossRef]
- Hopkins, B.D.; Pauli, C.; Du, X.; Wang, D.G.; Li, X.; Wu, D.; Amadiume, S.C.; Goncalves, M.; Hodakoski, C.; Lundquist, M.R.; et al. Suppression of insulin feedback enhances the efficacy of PI3K inhibitors. Nature 2018, 560, 499–503. [Google Scholar] [CrossRef]
- Liśkiewicz, A.; Kasprowska, D.; Wojakowska, A.; Polański, K.; Lewin–Kowalik, J.; Kotulska-Jozwiak, K.; Jędrzejowska–Szypułka, H. Long-term High Fat Ketogenic Diet Promotes Renal Tumor Growth in a Rat Model of Tuberous Sclerosis. Sci. Rep. 2016, 6, 21807. [Google Scholar] [CrossRef]
- Xia, S.; Lin, R.; Jin, L.; Zhao, L.; Kang, H.-B.; Pan, Y.; Liu, S.; Qian, G.; Qian, Z.; Konstantakou, E.; et al. Prevention of Dietary-Fat-Fueled Ketogenesis Attenuates BRAF V600E Tumor Growth. Cell Metab. 2017, 25, 358–373. [Google Scholar] [CrossRef]
- Li, J.; Zhang, H.; Dai, Z. Cancer Treatment With the Ketogenic Diet: A Systematic Review and Meta-analysis of Animal Studies. Front. Nutr. 2021, 8, 315. [Google Scholar] [CrossRef]
- Klement, R.J.; Champ, C.; Otto, C.; Kämmerer, U. Anti-Tumor Effects of Ketogenic Diets in Mice: A Meta-Analysis. PLoS ONE 2016, 11, e0155050. [Google Scholar] [CrossRef]
- Lv, M.; Zhu, X.; Wang, H.; Wang, F.; Guan, W. Roles of Caloric Restriction, Ketogenic Diet and Intermittent Fasting during Initiation, Progression and Metastasis of Cancer in Animal Models: A Systematic Review and Meta-Analysis. PLoS ONE 2014, 9, e115147. [Google Scholar] [CrossRef]
- Talib, W.; Mahmod, A.; Kamal, A.; Rashid, H.; Alashqar, A.; Khater, S.; Jamal, D.; Waly, M. Ketogenic Diet in Cancer Prevention and Therapy: Molecular Targets and Therapeutic Opportunities. Curr. Issues Mol. Biol. 2021, 43, 42. [Google Scholar] [CrossRef]
- Plotti, F.; Terranova, C.; Luvero, D.; Bartolone, M.; Messina, G.; Feole, L.; Cianci, S.; Scaletta, G.; Marchetti, C.; Di Donato, V.; et al. Diet and Chemotherapy: The Effects of Fasting and Ketogenic Diet on Cancer Treatment. Chemotherapy 2020, 65, 77–84. [Google Scholar] [CrossRef]
- Mittelman, S.D. The Role of Diet in Cancer Prevention and Chemotherapy Efficacy. Annu. Rev. Nutr. 2020, 40, 273–297. [Google Scholar] [CrossRef]
- Freedland, S.J.; Allen, J.; Jarman, A.; Oyekunle, T.; Armstrong, A.J.; Moul, J.W.; Sandler, H.M.; Posadas, E.M.; Levin, D.; Wiggins, E.; et al. A Randomized Controlled Trial of a 6-Month Low-Carbohydrate Intervention on Disease Progression in Men with Recurrent Prostate Cancer: Carbohydrate and Prostate Study 2 (CAPS2). Clin. Cancer Res. 2020, 26, 3035–3043. [Google Scholar] [CrossRef]
- Khodabakhshi, A.; Akbari, M.E.; Mirzaei, H.R.; Mehrad-Majd, H.; Kalamian, M.; Davoodi, S.H. Feasibility, Safety, and Beneficial Effects of MCT-Based Ketogenic Diet for Breast Cancer Treatment: A Randomized Controlled Trial Study. Nutr. Cancer 2019, 72, 627–634. [Google Scholar] [CrossRef]
- Cohen, C.W.; Fontaine, K.R.; Arend, R.C.; Soleymani, T.; Gower, B.A. Favorable Effects of a Ketogenic Diet on Physical Function, Perceived Energy, and Food Cravings in Women with Ovarian or Endometrial Cancer: A Randomized, Controlled Trial. Nutrients 2018, 10, 1187. [Google Scholar] [CrossRef]
- Cohen, C.W.; Fontaine, K.R.; Arend, R.C.; Alvarez, R.D.; Iii, C.A.L.; Huh, W.K.; Bevis, K.S.; Kim, K.H.; Straughn, J.M.; A Gower, B. A Ketogenic Diet Reduces Central Obesity and Serum Insulin in Women with Ovarian or Endometrial Cancer. J. Nutr. 2018, 148, 1253–1260. [Google Scholar] [CrossRef] [PubMed]
- Klement, R.J.; Schäfer, G.; Sweeney, R.A. A ketogenic diet exerts beneficial effects on body composition of cancer patients during radiotherapy: An interim analysis of the KETOCOMP study. J. Tradit. Complement. Med. 2019, 10, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Ok, J.H.; Lee, H.; Chung, H.-Y.; Lee, S.H.; Choi, E.J.; Kang, C.M.; Lee, S.M. The Potential Use of a Ketogenic Diet in Pancreatobiliary Cancer Patients After Pancreatectomy. Anticancer. Res. 2018, 38, 6519–6527. [Google Scholar] [CrossRef]
- Champ, C.E.; Palmer, J.; Volek, J.S.; Werner-Wasik, M.; Andrews, D.W.; Evans, J.J.; Glass, J.; Kim, L.; Shi, W. Targeting metabolism with a ketogenic diet during the treatment of glioblastoma multiforme. J. Neuro-Oncology 2014, 117, 125–131. [Google Scholar] [CrossRef]
- Fearon, K.C.; Borland, W.; Preston, T.; Tisdale, M.J.; Shenkin, A.; Calman, K.C. Cancer cachexia: Influence of systemic ketosis on substrate levels and nitrogen metabolism. Am. J. Clin. Nutr. 1988, 47, 42–48. [Google Scholar] [CrossRef]
- Jansen, N.; Walach, H. The development of tumours under a ketogenic diet in association with the novel tumour marker TKTL1: A case series in general practice. Oncol. Lett. 2015, 11, 584–592. [Google Scholar] [CrossRef]
- Klement, R.J.; Sweeney, R.A. Impact of a ketogenic diet intervention during radiotherapy on body composition: I. Initial clinical experience with six prospectively studied patients. BMC Res. Notes 2016, 9, 143. [Google Scholar] [CrossRef]
- Martin-McGill, K.; Cherry, G.; Marson, A.; Smith, C.T.; Jenkinson, M. ACTR-29. KETOGENIC DIETS AS AN ADJUVANT THERAPY IN GLIOBLASTOMA (KEATING): A MIXED METHOD APPROACH TO ASSESSING TRIAL FEASIBILITY. Neuro-Oncology 2018, 20, vi17. [Google Scholar] [CrossRef][Green Version]
- Martin-McGill, K.J.; Marson, A.; Smith, C.T.; Jenkinson, M.D. The Modified Ketogenic Diet in Adults with Glioblastoma: An Evaluation of Feasibility and Deliverability within the National Health Service. Nutr. Cancer 2018, 70, 643–649. [Google Scholar] [CrossRef]
- Rieger, J.; Bähr, O.; Maurer, G.D.; Hattingen, E.; Franz, K.; Brucker, D.; Walenta, S.; Kämmerer, U.; Coy, J.F.; Weller, M.; et al. ERGO: A pilot study of ketogenic diet in recurrent glioblastoma Erratum in/ijo/45/6/2605. Int. J. Oncol. 2014, 44, 1843–1852. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, M.; Pfetzer, N.; Schwab, M.; Strauss, I.; Kämmerer, U. Effects of a ketogenic diet on the quality of life in 16 patients with advanced cancer: A pilot trial. Nutr. Metab. 2011, 8, 54. [Google Scholar] [CrossRef] [PubMed]
- Tan-Shalaby, J.L.; Carrick, J.; Edinger, K.; Genovese, D.; Liman, A.D.; Passero, V.A.; Shah, R.B. Modified Atkins diet in advanced malignancies—Final results of a safety and feasibility trial within the Veterans Affairs Pittsburgh Healthcare System. Nutr. Metab. 2016, 13, 1–12. [Google Scholar] [CrossRef]
- Van der Louw, E.J. Ketogenic diet treatment as adjuvant to standard treatment of glioblastoma multiforme: A feasibility and safety study. Therapeutic Adv. Med. Oncol. 2019, 11, 1758835919853958. [Google Scholar] [CrossRef]
- Woodhouse, C.; Ward, T.; Gaskill-Shipley, M.; Chaudhary, R. Feasibility of a Modified Atkins Diet in Glioma Patients During Radiation and Its Effect on Radiation Sensitization. Curr. Oncol. 2019, 26, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Zahra, A.; Fath, M.; Opat, E.; Mapuskar, K.A.; Bhatia, S.K.; Rodman, S.; Iii, S.N.R.; Snyders, T.P.; Chenard, C.A.; Eichenberger-Gilmore, J.M.; et al. Consuming a Ketogenic Diet while Receiving Radiation and Chemotherapy for Locally Advanced Lung Cancer and Pancreatic Cancer: The University of Iowa Experience of Two Phase 1 Clinical Trials. Radiat. Res. 2017, 187, 743–754. [Google Scholar] [CrossRef] [PubMed]
- Bozzetti, F.; Cozzaglio, L.; Gavazzi, C.; Brandi, S.; Bonfanti, G.; Lattarulo, M.; Gennari, L. Total nutritional manipulation in humans: Report of acancer patient. Clin. Nutr. 1996, 15, 207–209. [Google Scholar] [CrossRef]
- Schwartz, K.; Chang, H.T.; Nikolai, M.; Pernicone, J.; Rhee, S.; Olson, K.; Kurniali, P.C.; Hord, N.G.; Noel, M. Treatment of glioma patients with ketogenic diets: Report of two cases treated with an IRB-approved energy-restricted ketogenic diet protocol and review of the literature. Cancer Metab. 2015, 3, 1–10. [Google Scholar] [CrossRef]
- Zuccoli, G.; Marcello, N.; Pisanello, A.; Servadei, F.; Vaccaro, S.; Mukherjee, P.; Seyfried, T.N. Metabolic management of glioblastoma multiforme using standard therapy together with a restricted ketogenic diet: Case Report. Nutr. Metab. 2010, 7, 33. [Google Scholar] [CrossRef]
- Tóth, C.; Clemens, Z. Treatment of Rectal Cancer with the Paleolithic Ketogenic Diet: A 24-months Follow-up. Am. J. Med Case Rep. 2017, 5, 205–216. [Google Scholar] [CrossRef]
- Bozzetti, F.; Gavazzi, C.; Mariani, L.; Crippa, F. Glucose-based total parenteral nutrition does not stimulate glucose uptake by humans tumours. Clin. Nutr. 2004, 23, 417–421. [Google Scholar] [CrossRef]
- Branca, J.J.V.; Pacini, S.; Ruggiero, M. Effects of Pre-surgical Vitamin D Supplementation and Ketogenic Diet in a Patient with Recurrent Breast Cancer. Anticancer. Res. 2015, 35, 5525–5532. [Google Scholar]
- Nebeling, L.C.; Miraldi, F.; Shurin, S.B.; Lerner, E. Effects of a ketogenic diet on tumor metabolism and nutritional status in pediatric oncology patients: Two case reports. J. Am. Coll. Nutr. 1995, 14, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Fanelli, F.R.; Franchi, F.; Mulieri, M.; Cangiano, C.; Cascino, A.; Ceci, F.; Muscaritoli, M.; Seminara, P.; Bonomo, L. Effect of energy substrate manipulation on tumour cell proliferation in parenterally fed cancer patients. Clin. Nutr. 1991, 10, 228–232. [Google Scholar] [CrossRef]
- Schroeder, U.; Himpe, B.; Pries, R.; Vonthein, R.; Nitsch, S.; Wollenberg, B. Decline of Lactate in Tumor Tissue After Ketogenic Diet: In Vivo Microdialysis Study in Patients with Head and Neck Cancer. Nutr. Cancer 2013, 65, 843–849. [Google Scholar] [CrossRef] [PubMed]
- Artzi, M.; Liberman, G.; Vaisman, N.; Bokstein, F.; Vitinshtein, F.; Aizenstein, O.; Ben Bashat, D. Changes in cerebral metabolism during ketogenic diet in patients with primary brain tumors: 1H-MRS study. J. Neuro-Oncology 2017, 132, 267–275. [Google Scholar] [CrossRef]
- Iyikesici, M.S. Feasibility study of metabolically supported chemotherapy with weekly carboplatin/paclitaxel combined with ketogenic diet, hyperthermia and hyperbaric oxygen therapy in metastatic non-small cell lung cancer. Int. J. Hyperth. 2019, 36, 445–454. [Google Scholar] [CrossRef]
- Iyikesici, M.S. Long-Term Survival Outcomes of Metabolically Supported Chemotherapy with Gemcitabine-Based or FOLFIRINOX Regimen Combined with Ketogenic Diet, Hyperthermia, and Hyperbaric Oxygen Therapy in Metastatic Pancreatic Cancer. Complement. Med. Res. 2019, 27, 31–39. [Google Scholar] [CrossRef]
- Strowd, R.E.; Cervenka, M.C.; Henry, B.J.; Kossoff, E.H.; Hartman, A.L.; Blakeley, J.O. Glycemic modulation in neuro-oncology: Experience and future directions using a modified Atkins diet for high-grade brain tumors. Neuro-Oncology Pr. 2015, 2, 127–136. [Google Scholar] [CrossRef]
- Moore, K. Using the restricted ketogenic diet for brain cancer management: Comments from neuro-oncologist. In Cancer as a Metabolic Disease: Management and Prevention of Cancer, 1st ed.; John Wiley & Sons Inc.: New Jersey, NJ, USA, 2012; pp. 397–400. [Google Scholar]
- Elsakka, A.M.A.; Bary, M.A.; Abdelzaher, E.; Elnaggar, M.; Kalamian, M.; Mukherjee, P.; Seyfried, T.N. Management of Glioblastoma Multiforme in a Patient Treated With Ketogenic Metabolic Therapy and Modified Standard of Care: A 24-Month Follow-Up. Front. Nutr. 2018, 5, 20. [Google Scholar] [CrossRef]
- Schwalb, M.; Taubmann, M.; Hines, S.; Reinwald, H.; Ruggiero, M. Clinical Observation of a Novel, Complementary, Immunotherapeutic Approach based on Ketogenic Diet, Chondroitin Sulfate, Vitamin D3, Oleic Acid and a Fermented Milk and Colostrum Product. Am. J. Immunol. 2016, 12, 91–98. [Google Scholar] [CrossRef]
- Brünings, W. Beitraege zum Krebsproblem. Mitteilung: Ueber eine diaetetisch-hormonale Beeinflussung des Krebses. Münch. Med. Wschr. 1941, 88, 117–123. [Google Scholar]
- Brünings, W. Beitraege zum Krebsproblem. Mitteilung: Klinische Anwendungen der diaetetisch-hormonalen Krebsbeeinflussung (“Entzuckerungsmethode”). Münchener Med. Wochenschr. 1942, 89, 71–76. [Google Scholar]
- Schulte, G.; Schütz, H. Insulin in der Krebsbehandlung. Münch. Med. Wschr. 1942, 89, 648–650. [Google Scholar]
- Kang, C.M.; Yun, B.; Kim, M.; Song, M.; Kim, Y.-H.; Lee, S.H.; Lee, H.; Lee, S.M.; Lee, S.-M. Postoperative serum metabolites of patients on a low carbohydrate ketogenic diet after pancreatectomy for pancreatobiliary cancer: A nontargeted metabolomics pilot study. Sci. Rep. 2019, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Porper, K.; Shpatz, Y.; Plotkin, L.; Pechthold, R.G.; Talianski, A.; Hemi, R.; Mardor, Y.; Jan, E.; Genssin, H.; Symon, Z.; et al. A Phase I clinical trial of dose-escalated metabolic therapy combined with concomitant radiation therapy in high-grade glioma. Neuro-Oncology Adv. 2021, 3, i10. [Google Scholar] [CrossRef]
- Kato, I.; Dyson, G.; Snyder, M.; Kim, H.-R.; Severson, R.K. Differential effects of patient-related factors on the outcome of radiation therapy for rectal cancer. J. Radiat. Oncol. 2016, 5, 279–286. [Google Scholar] [CrossRef]
- Santos, J.G.; Da Cruz, W.M.S.; Sch�Nthal, A.; Salazar, M.D.; Fontes, C.A.P.; Quirico-Santos, T.; Da Fonseca, C.O. Efficacy of a ketogenic diet with concomitant intranasal perillyl alcohol as a novel strategy for the therapy of recurrent glioblastoma. Oncol. Lett. 2017, 15, 1263–1270. [Google Scholar] [CrossRef]
- Davis, R.A.H.; Deemer, S.E.; Bergeron, J.M.; Little, J.T.; Warren, J.L.; Fisher, G.; Smith, D.L.; Fontaine, K.R.; Dickinson, S.L.; Allison, D.B.; et al. Dietary R, S-1,3-butanediol diacetoacetate reduces body weight and adiposity in obese mice fed a high-fat diet. FASEB J. 2019, 33, 2409–2421. [Google Scholar] [CrossRef]
- Meidenbauer, J.J.; Mukherjee, P.; Seyfried, T.N. The glucose ketone index calculator: A simple tool to monitor therapeutic efficacy for metabolic management of brain cancer. Nutr. Metab. 2015, 12, 12. [Google Scholar] [CrossRef] [PubMed]
| Cancer Type(s) | Sample Size | Dietary Intervention | Study Duration | Results/Outcomes | References |
|---|---|---|---|---|---|
| Prostate | N = 45 Arm A: N = 27 Arm B: N = 18 | Arm A: A low-carbohydrate diet, goal: (≤20 g per day), estimated actual carbohydrate intake: 37 g/day; Arm B: Control group (no dietary intervention) | 6 months | -Weight loss -BMI reduction -Waist circumference reduction | [51] |
| Breast cancer | N = 60 Arm A: N = 30 Arm B: N = 30 | Arm A: Medium-chain triglycerides (MCT) based ketogenic diet (6% calories from Carbohydrates [CHO], 19% protein, 20% MCT, 55% fat); Patients received 500 mL of MCT oil from the Nutricia Company every 2 weeks Arm B: Standard Diet (55% CHO, 15% protein, and 30% fat) | 3 months | -Weight loss -BMI reduction -Reduction in body fat | [52] |
| Ovarian/endometrial cancer | N = 45 Arm A: N = 25 Arm B: N = 20 | Arm A: Ketogenic diet (70% (≥125 g): 25% (≤100 g): 5% (<20 g) energy per day from fat, protein, and carbohydrates) Arm B: American Cancer Society diet (ACS: high in fiber, low in fat) Individual diet advice from certified dietitians. Weekly emails or phone calls. One face-to-face meeting after baseline assessment | 3 months | -Self-reported improvement in energy levels (intervention group) -Fewer cravings for starchy foods and fast-food fats -Reduction in total body | [53,54] |
| Rectal cancer, head and neck cancer Breast cancer | N = 81 Arm A: N = 20 Arm B: N = 61 | Arm A: ketogenic diet with additional consumption of non-glucogenic amino acids Arm B: no dietary intervention | 30–40 days | -Decreased fat mass | [55] |
| Pancreatic cancer Duodenal cancer Common bile duct cancer Ampulla of Vater cancer Cholangiocarcinoma Neuroendocrine tumor | N = 19 Arm A: N = 10 Arm B: N = 9 | Arm A: Ketogenic diet (3–6%, 14–27%; 70–80% energy per day from carbohydrates, protein, and fat) served as three meals and three snacks per day Arm B: usual Korean diet (55–65%, 7–20%, 15–30% energy per day from carbohydrates, protein and fat) served as three meals per day | 12 days | -Decreased body cell mass higher in General Diet arm | [56] |
| Glioblastoma multiforme | N = 53 Arm A: N = 6 Arm B: N = 47 | Arm A: self-administered KD Arm B: unspecified standard American diet | Duration: 3–12 months | - Two patients with grade 1 constipation, 4 patients with grade 1 fatigue, 1 patient with grade 2 fatigue, 1 patient with deep venous thrombosis during treatment, 1 patient with asymptomatic hypoglycemia, 1 patient with nephrolithiasis no grade 3 and higher toxicities or symptomatic hypoglycemia -Weight loss on non-calorie-restricted KD: 1 to 27 Ibs -Weight loss on calorie-restricted KD: 46 Ibs | [57] |
| Fearon et al. [44] Ovarian, Lung, Gastric | N = 5 | Crossover study: Nasogastric tube feeding: normal, balanced regimen on days 1–6 KD containing same total calorie and protein on days 7–13 | 13 days | -Increase in body weight | [58] |
| Diverse | Recruited patients N = 12 Analyzed patients N = 10 | KD with targeted CHO intake below 5% of total energy intake, written menus and samples of CHO-restriction products were provided | 28 days | -Five patients with grade 2 fatigue, 5 patients with grade 1 constipation, 1 patient with grade 1 leg cramps -Weight loss - Decreased caloric intake -Adherence: 5 of 12 patients completed all 28 days of the diet | [27] |
| Diverse | Analyzed patients N = 78 Arm A: N = 7 Arm B: N = 6 Arm C: N = 65 | Arm A: full adoption of a non-specified KD, patients informed about a single company producing KD-related food Arm B: partial adoption of a non-specified KD, patients informed about a single company producing KD related food Arm C: patients who did not adopt a KD | Not specified | 1. Reduction in TKTL 1 was associated with adopting a KD; 2. Correlation between improvement in cancer status category and full adoption of a KD (χ2 = 33.26; df = 4; p = 0.00001 | [59] |
| Diverse | Analyzed patients N = 6 | Self-administered KD (recommended CHO intake < 50 g/day) during the course of RT/RCT; patients received basic information on KD; counseling at least once per week | Patient-dependent from 32 to 73 days | -Decreased fat mass | [60] |
| Glioblastoma | Assessed for eligibility: N = 57 Randomized: N = 12 Arm A: N = 6 Arm B: N = 6 Retention at 12 weeks. N = 4 Arm A: N = 3 Arm B: N = 1 | Arm A: MCTKD (75%; 15%; 10% of energy per day from fat, protein and carbohydrates, with 30% of fat from MCT nutritional products) Arm B: MKD (80%; 15%; 5% of energy per day from fat, protein and carbohydrates) | 12 weeks | 1. Arm A: Three patients retained for 3 months (drop-out = 50%) Arm B: One patient retained for 3 months (drop-out = 83%) 2. GHS at baseline: Arm A: patients who later withdrew: 72.2 ± 20.7; patients who retained: 75 ± 6.8 Arm B: patients who later withdrew: 70 ± 13.8; patients who retained: 80 ± 0 GHS: at week 6: Arm A: patients who withdrew at week 6: 41.7 ± 0; patients who retained: 66.7 ± 0 Arm B: patients who withdrew at week 6: 50 ± 0; patients who retained: 100 ± 0 3. Adverse events during the first 6 weeks: Arm A: diarrhea (n = 1, CTCAE grade 1), nausea (n = 1, CTCAE grade 1), vomiting (n = 1, CTCAE grade 2), dyspepsia (n = 1, CTCAE grade 1) Arm B: vomiting (n = 1, CTCAE grade 1), dry mouth (n = 1 MKD, CTCAE grade 1) | [61] |
| Glioblastoma | Enrolled: N = 6 Completed intervention: N = 4 | MKD (70%: 3–5% (≤20 g) energy per day from fat and carbohydrates; protein consumption was not restricted | 12 weeks | -Constipation in two patients, resolved with dietary modification | [62] |
| Glioblastoma | Included patients N = 20 Evaluable for efficiency N = 17 | KD with CO intake < 60 g/day, additionally highly fermented yoghurt drinks and two different plant oils were provided to be consumed at will. No calorie restriction, patients were instructed to always eat to satiety | Until progression of the disease | -Three out of 20 patients discontinued the diet after 2–3 weeks without progression, due to reduced QoL - Body weight reduction -Diarrhea, constipation, hunger and/or demand for glucose were present in some patients during the diet | [63] |
| Diverse | Enrolled: N = 16 Completed intervention: N = 5 | KD with CHO limited to 70 g per day and 20 g per meal Two oil–protein shakes consumed in the morning and in the afternoon | 12 weeks | -11/16 Patients discontinued the diet - 3/11 were unable to adhere to the diet, -6/11 discontinued due to progressive disease -2/11 died from progressive disease - reported side effects included increase in appetite loss, constipation, diarrhea and fatigue during the diet - QoL was low at baseline and stayed relatively stable during the intervention; worsening of fatigue, pain, dyspnea and role function but emotional functioning and insomnia improved slightly | [64] |
| Diverse | Enrolled: N = 17 Drop-out before first analysis: N = 6 Completed intervention: N = 4 | Modified Atkins Diet with 20 to 40 g of CHO and restricted consumption of high CHO foods no restrictions for calories, protein or fats | 16 weeks | -13/17 patients discontinued the diet before 16 weeks -weight loss -Reported adverse effects included: hyperuricemia (N = 7), hyperlipidemia (N = 2), pedal edema (N = 2), anemia (N = 2), halitosis (N = 2), pruritus (N = 2), hypoglycemia (N = 2), hyperkalemia (N = 2), hypokalemia (N = 2), hypomagnesemia (N = 2), flulike symptoms/fatigue (N = 2) | [65] |
| Glioblastoma multiforme | Phase A: N = 9 Phase B: N = 8 Completed intervention N = 6 | Phase A: Fluid KD with a 4:1 ratio (4 g fat versus 1 g protein plus carbohydrates, 90% energy from fat) Patients were allowed a snack with the same 4:1 diet ratio once a day Phase B: Solid-food KD (diet ratio 1.5–2.0:1) with MCT; (70% energy from fat with the consistency of an emulsion) | 14 weeks | -6/9 patients included in phase A completed the 14 weeks KD - Reported adverse effects included: constipation (n = 7), nausea/vomiting (n = 2), hypercholesterolemia (n = 1), hypoglycemia (n = 1), low carnitine (n = 1) and diarrhea (n = 1). CTCAE grade 2: hallucinations (n = 1), allergic reaction (n = 1) and wound infection (n = 1) | [66] |
| Glioma | N = 29 | MAD with a 0.8–1:1 ratio (0.8-1 g fat to 1 g carbohydrate plus protein Duration: 6 weeks | 6 weeks | -28/29 patients completed the 6-week diet - Reported adverse events: Grade 2 constipation (n = 1), grade 1 fatigue and nausea were present in the patients -Decreased BMI for all patients | [67] |
| Lung | Enrolled patients: N = 7 Completed intervention: N = 2 | KD with 90%; 8%; 2% of energy per day from fat, protein and carbohydrates. All meals prepared for the patients | 42 days | -Weight loss - Reported adverse events included: constipation, diarrhea, nausea, vomiting and fatigue; hyperuricemia | [68] |
| Pancreas | N = 2 | KD with 90%; 8%; 2% of energy per day from fat, protein and carbohydrates. All meals readily prepared for the patients | 34 days | -1/2 patients completed the intervention 2. Reported adverse events included: Constipation, diarrhea, nausea and vomiting, 1 patient experienced dehydration -Weight loss | [68] |
| Desmoid tumor | N = 1 | TPN consisting of 28 kcal fat/kg body weight/day, 1.5 g protein/kg body weight/day; 40 g glucose/day | Desmoid tumor | -Body weight increased | [69] |
| Glioma | N = 2 | ERKD: with a 3:1 ratio of ingested nutrients (3 g fat versus 1 g protein plus carbohydrates) 20% restriction of calories per day | 12 months | -Adherence: 1/2 patients completed the intervention -Reported headaches -Initial body weight decrease in both patients and remained stable afterward | [70] |
| Glioblastoma multiforme | N = 1 | ERKD delivering 600 kcal per day, consisting of 42 g fat, 32 g protein and 10 g CHO per day | 56 days | -Bodyweight decreased in the first 14 days of the diet - Grade 4 hyperuricemia reported, resulted in diet change to calorie restricted non-ketogenic diet | [71] |
| Rectal | N = 1 | Paleolithic KD, nutrients consumed in a fat:protein ratio of 2:1 animal fat, red meats and organ meats were encouraged, root vegetables were allowed, all other foods were prohibited | 24 months | -Decreased bodyweight -Initial decrease in volume after concomitant radiotherapy -Tumor volume remained stable but four hepatic metastases were detected at the end of the diet | [72] |
| Diverse | N = 12 | Single 3 h infusion of glucose-based (GTPN) or a lipid-based TPN (LTPN) containing 4 mg glucose/kg/min or 2 mg lipid/kg/min, respectively | 3 h | -No statistically significant stimulation or suppression of FDG uptake | [73] |
| Recurrent Breast | N = 1 | Self-administered high doses of oral vitamin D3 (10,000 IU/day), and KD rich in oleic acid. Duration: 3 weeks | 3 weeks | -Progesterone receptor status positivity increased -HER2 positivity decreased | [74] |
| Astrocytoma | N = 2 | KD with 60%; 20%; 10%, 10% of energy per day from MCT oil, protein, carbohydrates and dietary fat plus additional supplements | 8 weeks | -Dose uptake ratio tumor: decreased normal cortex decreased -Adherence: 100% patients were able to complete the dietary intervention | [75] |
| Esophagus Stomach Colon-rectum | N = 27 Arm A: N = 9 Arm B: N = 9 Arm C: N = 9 | Arm A: glucose-based TPN (100% of the calorie from dextrose); Arm B: lipid-based TPN (80% of the calorie from fat, 20% from dextrose); Arm C: oral diet All diets were iso-caloric and isonitrogenous. Duration: 2 weeks | 2 weeks | No statistically significant changes | [76] |
| Head and neck | N = 12 | Unspecified Western diet followed by unspecified KD | Variable, up to 4 days | Decline of mean lactate concentration in the tumor tissue during the KD | [77] |
| Brain | Included: N = 9 intervention: N = 5 retrospectively added control N = 4 | KD based on ready-made formula, with a 4:1 ratio of ingested nutrients (4 g fat versus 1 g protein plus carbohydrates) | variable from 2 to 31 months | -Diet tolerated by 4/5 patients,(strict adherence only in 2 patients) -Four out of 50 MRI spectroscopy scans detected ketone bodies in the brains of the patients following the KD | [78] |
| Lung | N = 44 | Mild KD (patients were encouraged to avoid high CHO food) in combination with HBO, hyperthermia and polychemotherapy administered during induced hypoglycemia | 24 weeks | -Adverse events reported—during treatment period: grade 5 neutropenia (N = 1), grade 3 neutropenia (N = 3), grade 3 anemia (N = 10), grade 4 thrombocytopenia (N = 3), grade 3 fatigue (N = 5), grade 3 diarrhea (N = 8), grade 3 neuropathy (N = 1), all of which were attributed to chemotherapy | [79] |
| Pancreas | N = 25 | Mild KD (patients were encouraged to avoid high CHO food) in combination with HBO, hyperthermia and polychemotherapy administered during induced hypoglycemia | Duration: mean follow-up: 25 months | -Adverse events reported: during treatment period: grade 3/4 neutropenia (N = 9), febrile neutropenia (N = 1), grade 3 anemia (N = 7), grade 4 thrombocytopenia (N = 4), grade 3 diarrhea (N = 2), all of which were attributed to chemotherapy | [80] |
| Brain | N = 8 | MAD with20g CHO/day restriction | 2-24 months: mean- 13 months | -7/8 completed intervention -Decreased body weight -Reduction in seizure frequency per week | [81] |
| Glioblastoma multiforme | N = 1 | Energy-restricted KD with a 4:1 ratio of calorie intake (fat versus protein plus carbohydrates) Total calories calculated 25% below BMR | 4 months | -No metabolically active tumor detected | [82] |
| Glioblastoma multiforme | N = 1 | KD with a 4:1 ratio of calorie intake (fat versus protein plus carbohydrates), delivered as calorie-restricted diet, combined with intermittent fasting, HBOT, other novel therapies and SOC treatment | 20 months | -Good surgical outcome and regressive changes in histopathology -Decreased body weight | [83] |
| Diverse | N = 6 | Very low CHO diet (not further specified) with a multitude of supplements, including amino acids and Vitamin D3 combined with SOC therapy | Varied | -Shrinkage of tumor or stable disease was reported during the intervention -Subjective improvement reported in some cases | [84] |
| Head and neck | N = 14 | KD with as little CHO as possible (estimated < 50 g per day), combined with insulin administration 3 × per day | Not specified | Visible remission after 2–3 weeks, but rebound effect after 2–3 months on the diet | [85] |
| Extra-cranial | N = 30 | KD with as little CHO as possible (estimated < 50 g per day), combined with insulin administration 3 × per day | Not specified | Tumor shrinkage in some cases Improvement in general condition and positive effects on clinical symptoms | [86] |
| Exra-cranial | N = 23 | KD with as little CHO as possible (estimated < 50 g per day), combined with insulin administration 3 × per day | Not specified | -Reduced pain severity, fatigue but deteriorated orientation | [87] |
| Pancreatic cancer Duodenal cancer Common bile duct cancer Ampulla of Vater cancer Neuroendocrine tumor | N = 18 | LCKD: Energy content: 1500 kcal/d, provided 4% from carbohydrate, 16% from protein and 80% from fat. Ketogenic ratio of 1.75:1 (F: C + P w/w) | 4 weeks | -Patients were in a poorer nutrition state after surgery, but this was alleviated at week 4; - LCKD induced ketone body production -Week 4, there were no significant differences in ketone levels | [88] |
| Glioma | N = 13 newly diagnosed= 6 recurrent=7 | KD + MCT + Metformin 850 | 6 weeks (recurrent) 2 weeks (newly diagnosed) | Increase in survival rate. Synergistic interaction between radiation therapy and KD. | [89] |
| Invasive Rectal | N = 359 | KD ≥ 40% kcal fat and <100 g/day glycemic load (48) | Not specified | Reduced risk of cancer-specific deaths | [90] |
| Glioblastoma | N = 32 | KD 50% kcal fat, 25% kcal CHO, 1.5 g/kg protein (17), CD (15) | 3 months | No change in glucose increased ketosis No change in body weight | [91] |
| Strength of Evidence | ||||
|---|---|---|---|---|
| Strong | Moderate | Weak | Unknown | |
| Pre-Clinical Studies | ||||
| Tumor weight | X | |||
| Antitumor effect/Tumor growth | X | |||
| Progression-free survival | X | |||
| Tumor volume | X | |||
| Overall survival time | X | |||
| Cells’ responsiveness to therapy | X | |||
| Body composition | X | |||
| Clinical Studies as an Adjunctive Therapy | ||||
| Tumor weight | X | |||
| Antitumor effect/Tumor growth | X | |||
| Progression-free survival | X | |||
| Tumor volume | X | |||
| Overall survival time | X | |||
| Cells’ responsiveness to therapy | X | |||
| Quality of life | X | |||
| Body composition | X | |||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lane, J.; Brown, N.I.; Williams, S.; Plaisance, E.P.; Fontaine, K.R. Ketogenic Diet for Cancer: Critical Assessment and Research Recommendations. Nutrients 2021, 13, 3562. https://doi.org/10.3390/nu13103562
Lane J, Brown NI, Williams S, Plaisance EP, Fontaine KR. Ketogenic Diet for Cancer: Critical Assessment and Research Recommendations. Nutrients. 2021; 13(10):3562. https://doi.org/10.3390/nu13103562
Chicago/Turabian StyleLane, Jordin, Nashira I. Brown, Shanquela Williams, Eric P. Plaisance, and Kevin R. Fontaine. 2021. "Ketogenic Diet for Cancer: Critical Assessment and Research Recommendations" Nutrients 13, no. 10: 3562. https://doi.org/10.3390/nu13103562
APA StyleLane, J., Brown, N. I., Williams, S., Plaisance, E. P., & Fontaine, K. R. (2021). Ketogenic Diet for Cancer: Critical Assessment and Research Recommendations. Nutrients, 13(10), 3562. https://doi.org/10.3390/nu13103562







