Anti-Inflammatory Effect of 4,5-Dicaffeoylquinic Acid on RAW264.7 Cells and a Rat Model of Inflammation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents
2.2. Cell Culture
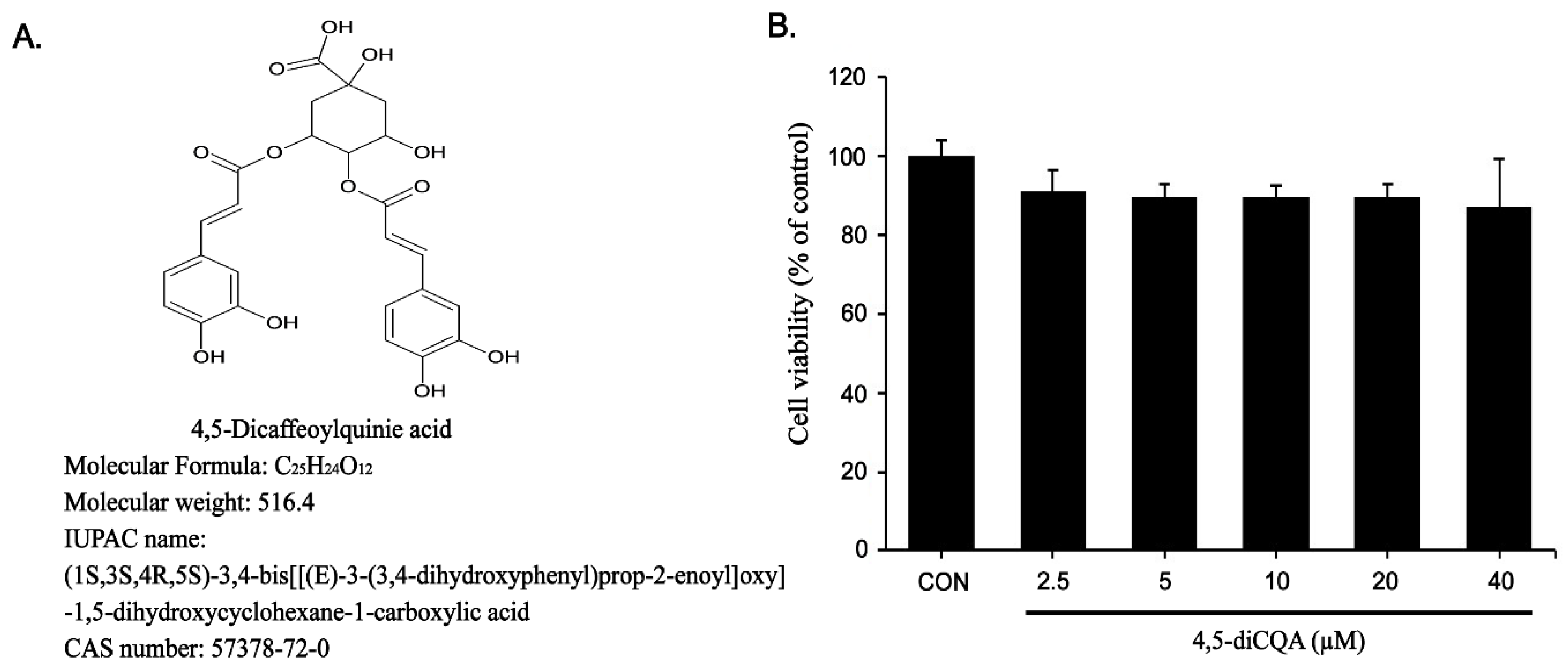
2.3. Cell Viability
2.4. Measurement of Nitrite and PGE2
2.5. Western Blot Analysis
2.6. Animals
2.7. Experimental Design and Drug Treatment
2.8. Statistical Analyses
3. Results
3.1. Effect of 4,5-diCQA on RAW264.7 Cell Viability
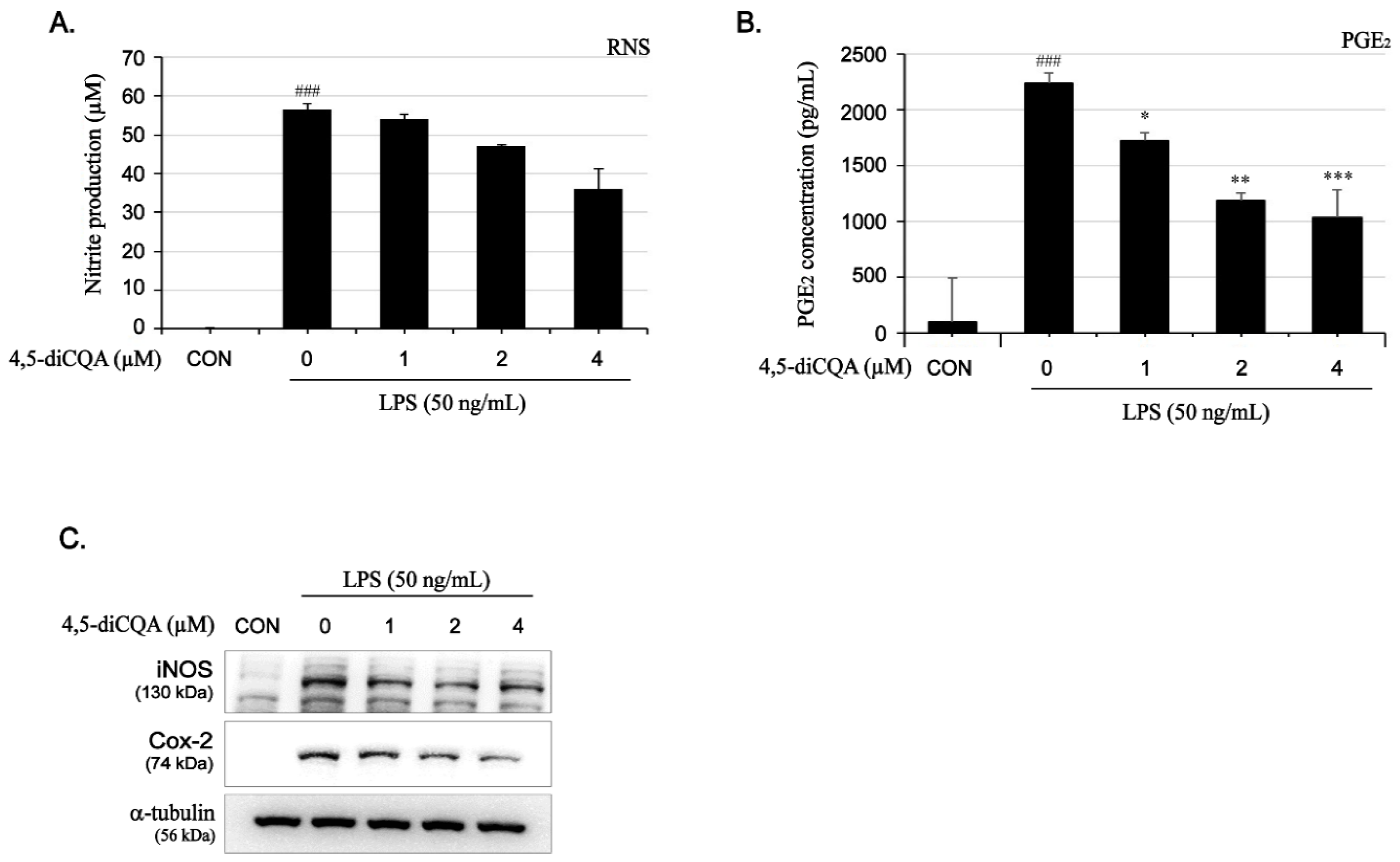
3.2. Inhibitory Effects of 4,5-diCQA on Nitrite Production and Anti-Inducible NO Synthase (iNOS) Protein Expression in RAW264.7 Cells Stimulated with Lipopolysaccharide (LPS)
3.3. Inhibitory Effects of 4,5-diCQA on PGE2 Production and COX-2 Protein Expression in RAW264.7 Cells Stimulated with LPS
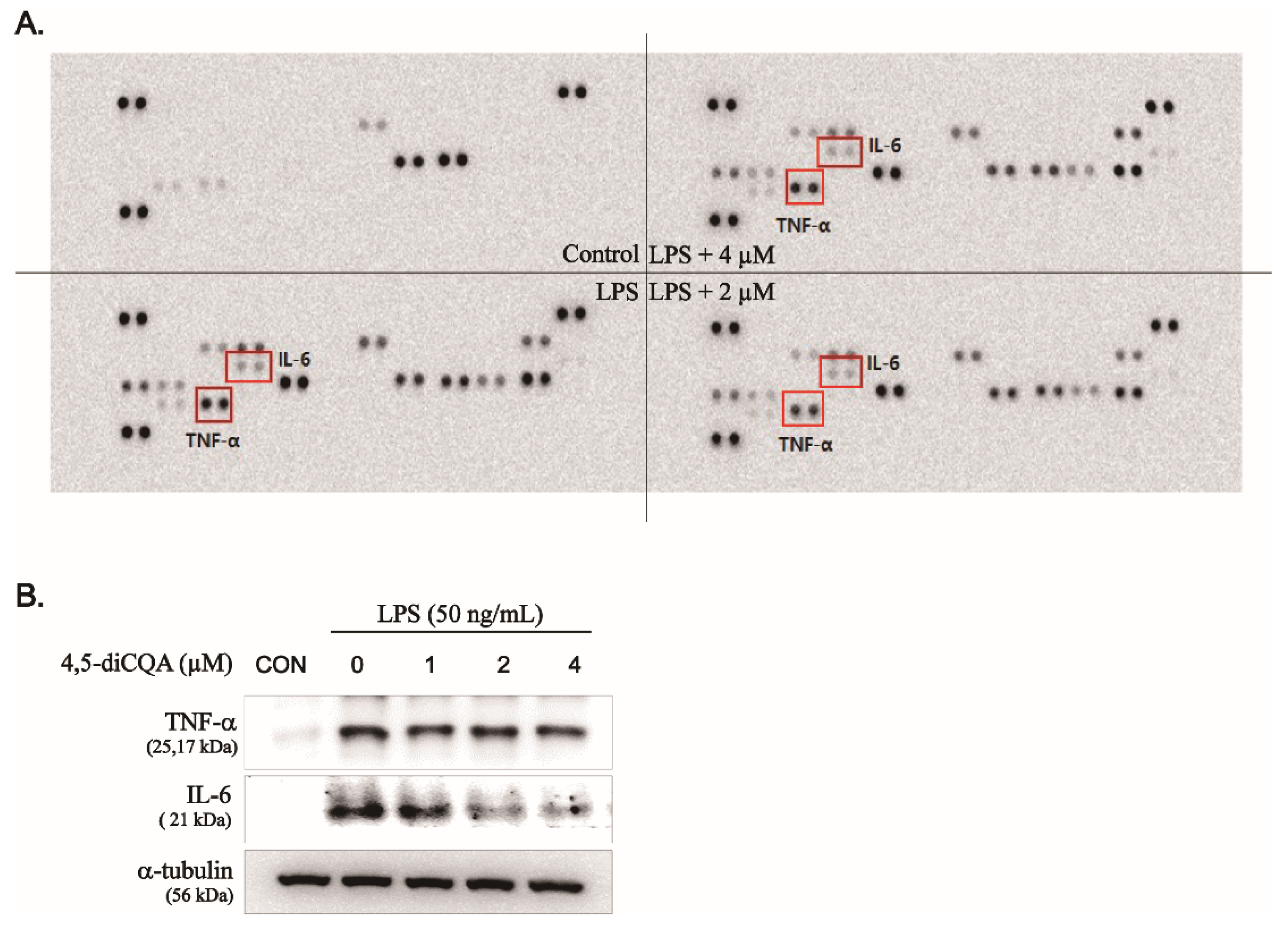
3.4. Inhibitory Effects of 4,5-diCQA on Pro-Inflammatory Cytokines in RAW264.7 Cells Stimulated with LPS
3.5. Suppression of NF-κB and MAPK Signaling Pathways by 4,5-diCQA in RAW264.7 Cells Stimulated with LPS
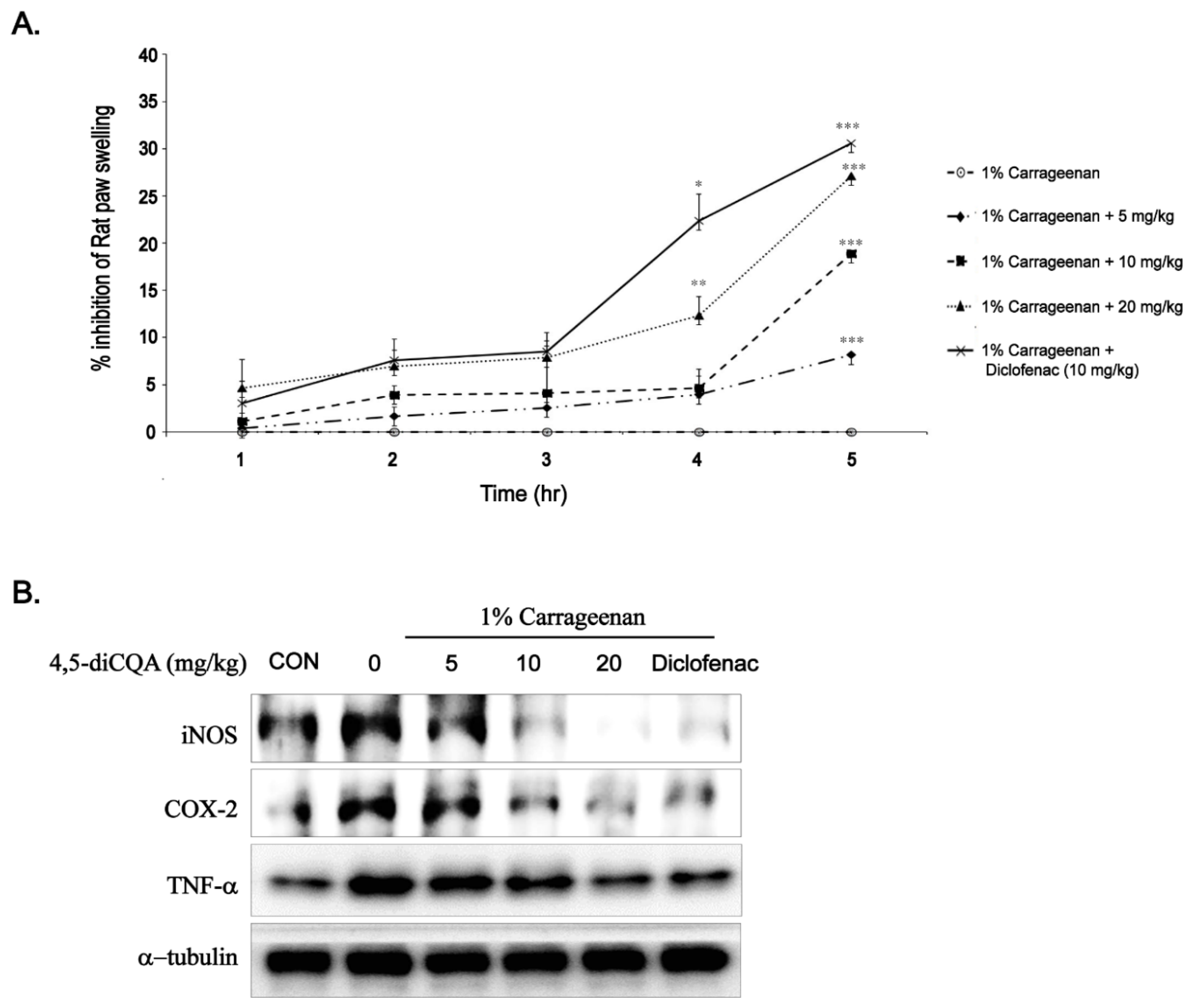
3.6. Anti-Inflammatory Effects of 4,5-diCQA In Vivo
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Germolec, D.R.; Shipkowski, K.A.; Frawley, R.P.; Evans, E. Markers of Inflammation. Meth. Mol. Biol. 2018, 1803, 57–79. [Google Scholar]
- Rankin, J.A. Biological mediators of acute inflammation. AACN Adv. Crit. Care 2004, 15, 3–17. [Google Scholar] [CrossRef]
- Libby, P. Inflammatory mechanisms: The molecular basis of inflammation and disease. Nutr. Rev. 2007, 5, S140–S146. [Google Scholar] [CrossRef]
- Medzhitov, R. Origin and physiological roles of inflammation. Nature 2007, 454, 428–435. [Google Scholar] [CrossRef]
- Branco, A.C.C.C.; Yoshikawa, F.S.Y.; Pietrobon, A.J.; Sato, M.N. Role of histamine in modulating the immune response and inflammation. Mediat. Inflamm. 2018, 2018, 9524075. [Google Scholar] [CrossRef]
- Viola, J.; Soehnlein, O. Atherosclerosis-a matter of unresolved inflammation. Semin. Immunol. 2015, 27, 184–193. [Google Scholar] [CrossRef]
- Hunter, D.J.; Bierma-Zeinstra, S. Osteoarthritis. Lancet 2019, 393, 1745–1759. [Google Scholar] [CrossRef]
- Danforth, D.N. The Role of Chronic Inflammation in the Development of Breast Cancer. Cancers 2021, 13, 3918. [Google Scholar] [CrossRef] [PubMed]
- Funk, C.D.; FitzGerald, G.A. COX-2 inhibitors and cardiovascular risk. J. Cardiovasc. Pharmacol. 2007, 50, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, G.H.; Ali, H.I. Design, synthesis, biological evaluation, and comparative Cox1 and Cox2 docking of p-substituted benzylidenamino phenyl esters of ibuprofenic and mefenamic acids. Bioorg. Med. Chem. 2012, 20, 1259–1270. [Google Scholar] [CrossRef] [PubMed]
- Cherniack, E.P. Polyphenols: Planting the seeds of treatment for the metabolic syndrome. Nutrition 2011, 27, 617–623. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.Y.; Park, S.K.; Kim, J.M.; Park, S.K.; Yoo, S.K.; Han, H.J.; Kim, D.O.; Heo, H.J. 4,5-dicaffeyolquinic acid improves high-fat diet-induced cognitive dysfunction through the regulation of insulin degrading enzyme. J. Food Biochem. 2019, 43, e12855. [Google Scholar] [CrossRef]
- Naveed, M.; Hejazi, V.; Abbas, M.; Kamboh, A.A.; Khan, G.J.; Shumzaid, M.; Ahmad, F.; Babazadeh, D.; FangFang, X.; Modearresi-Ghazani, F.; et al. Chlorogenic acid (CGA): A pharmacological review and call for further research. Biomed. Pharmacother. 2018, 97, 67–74. [Google Scholar] [CrossRef]
- Wianowska, D.; Gil, M. Recent advances in extraction and analysis procedures of natural chlorogenic acids. Phytochem. Rev. 2019, 18, 273–302. [Google Scholar] [CrossRef] [Green Version]
- Trendafilova, A.; Ivanova, V.; Rangelov, M.; Todorova, M.; Ozek, G.; Yur, S.; Ozek, T.; Aneva, I.; Veleva, R.; Moskova-Doumanova, V.; et al. Caffeoylquinic Acids, Cytotoxic, Antioxidant, Acetylcholinesterase and Tyrosinase Enzyme Inhibitory Activities of Six Inula Species from Bulgaria. Chem. Biodivers. 2020, 17, e2000051. [Google Scholar] [CrossRef] [PubMed]
- Kang, J.Y.; Park, S.K.; Guo, T.J.; Ha, J.S.; Lee, D.S.; Kim, J.M.; Lee, U.; Kim, D.O.; Heo, H.J. Reversal of trimethyltin-induced learning and memory deficits by 3,5-dicaffeoylquinic acid. Oxid. Med. Cell. Longev. 2016, 2016, 6981595. [Google Scholar] [CrossRef]
- Xu, J.; Hu, Q.; Liu, Y. Antioxidant and DNA-protective activities of chlorogenic acid isomers. J. Agric. Food Chem. 2012, 60, 11625–11630. [Google Scholar] [CrossRef]
- Lodise, O.; Patil, K.; Karshenboym, I.; Prombo, S.; Chukwueke, C.; Pai, S.B. Inhibition of Prostate Cancer Cells by 4,5-Dicaffeoylquinic Acid through Cell Cycle Arrest. Prostate Cancer 2019, 23, 4520645. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- National Research Council. Guide for the Care and Use of Laboratory Animals, 8th ed.; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- Zammel, N.; Saeed, M.; Bouali, N.; Elkahoui, S.; Alam, J.M.; Rebai, T.; Kausar, M.A.; Adnan, M.; Siddiqui, A.; Badraoui, R. Antioxidant and Anti-Inflammatory Effects of Zingiber officinale roscoe and Allium subhirsutum: In Silico, Biochemical and Histological Study. Foods 2021, 10, 1383. [Google Scholar] [CrossRef] [PubMed]
- Lii, J.A.S.; Ren, S.; Lang, R.A.; Duffield, J.S. Macrophages in development, homeostasis and regeneration. Trends Mol. Med. 2011, 17, 743–752. [Google Scholar]
- Liu, Y.; Fang, S.; Li, X.; Feng, J.; Du, J.; Gou, L.; Su, Y.; Zhou, J.; Ding, G.; Bai, Y.; et al. Aspirin inhibits LPS-induced macrophage activation via the NF-κB pathway. Sci. Rep. 2017, 7, 11549. [Google Scholar] [CrossRef] [Green Version]
- Neacsu, P.; Mazare, A.; Schmuki, P.; Cimpean, A. Attenuation of the macrophage inflammatory activity by TiO2 nanotubes via inhibition of MAPK and NF-κB pathways. Int. J. Nanomed. 2015, 10, 6455–6467. [Google Scholar]
- Lin, T.H.; Tamaki, Y.; Pajarinen, J.; Waters, H.A.; Woo, D.K.; Yao, Z.; Goodman, S.B. Chronic inflammation in biomaterial induced periprosthetic osteolysis: NF-κB as a therapeutic target. Acta Biomater. 2014, 10, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hommes, D.W.; Peppelenbosch, M.P.; van Deventer, S.J. Mitogen activated protein (MAP) kinase signal transduction pathways. Gut 2003, 52, 144–154. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, C.J.; Lin, J.Y. Anti-inflammatory and anti-apoptotic effects of strawberry and mulberry fruit polysaccharides on lipopolysaccharide-stimulated macrophages through modulating pro-/anti-inflammatory cytokines secretion and Bcl-2/Bak protein ratio. Food Chem. Toxicol. 2012, 50, 3032–3039. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.A.; Moon, S.M.; Choi, Y.H.; Han, S.H.; Park, B.R.; Choi, M.S.; Kim, J.S.; Kim, Y.H.; Kim, D.K.; Kim, C.S. Aqueous extract of Codium fragile suppressed inflammatory responses in lipopolysaccharide-stimulated RAW264.7 cells and carrageenan-induced rats. Biomed. Pharmacoth. 2017, 93, 1055–1064. [Google Scholar] [CrossRef] [PubMed]
- Winter, C.A.; Risley, E.A.; Nuss, G.W. Carrageenin-Induced Edema in Hind Paw of the Rat as an Assay for Antiiflammatory Drugs. Proc. Soc. Exp. Biol. Med. 1962, 111, 544–547. [Google Scholar] [CrossRef]
- Crunkhorn, P.; Meacock, S.C. Mediators of the inflammation induced in the rat paw by carrageenan. Br. J. Pharmacol. 1971, 42, 392–402. [Google Scholar] [CrossRef] [Green Version]
- Mansouri, M.T.; Hemmati, A.A.; Naghizadeh, B.; Mard, S.A.; Rezaie, A.; Ghorbanzadeh, B.A. Study of the mechanisms underlying the anti-inflammatory effect of ellagic acid in carrageenan-induced paw edema in rats. Indian J. Pharmacol. 2015, 47, 292–298. [Google Scholar] [PubMed] [Green Version]
- Li, Z.; Zhang, L.; Zhao, Z.; Malyngamide, F. Possesses Anti-Inflammatory and Antinociceptive Activity in Rat Models of Inflammation. Pain Res. Manag. 2021, 2021, 4919391. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jang, G.; Lee, S.; Hong, J.; Park, B.; Kim, D.; Kim, C. Anti-Inflammatory Effect of 4,5-Dicaffeoylquinic Acid on RAW264.7 Cells and a Rat Model of Inflammation. Nutrients 2021, 13, 3537. https://doi.org/10.3390/nu13103537
Jang G, Lee S, Hong J, Park B, Kim D, Kim C. Anti-Inflammatory Effect of 4,5-Dicaffeoylquinic Acid on RAW264.7 Cells and a Rat Model of Inflammation. Nutrients. 2021; 13(10):3537. https://doi.org/10.3390/nu13103537
Chicago/Turabian StyleJang, Goeun, Seulah Lee, Joonho Hong, Boram Park, Dokyung Kim, and Chunsung Kim. 2021. "Anti-Inflammatory Effect of 4,5-Dicaffeoylquinic Acid on RAW264.7 Cells and a Rat Model of Inflammation" Nutrients 13, no. 10: 3537. https://doi.org/10.3390/nu13103537
APA StyleJang, G., Lee, S., Hong, J., Park, B., Kim, D., & Kim, C. (2021). Anti-Inflammatory Effect of 4,5-Dicaffeoylquinic Acid on RAW264.7 Cells and a Rat Model of Inflammation. Nutrients, 13(10), 3537. https://doi.org/10.3390/nu13103537




