Supper Timing and Cardiovascular Mortality: The Japan Collaborative Cohort Study
Abstract
:1. Introduction
2. Materials and Methods
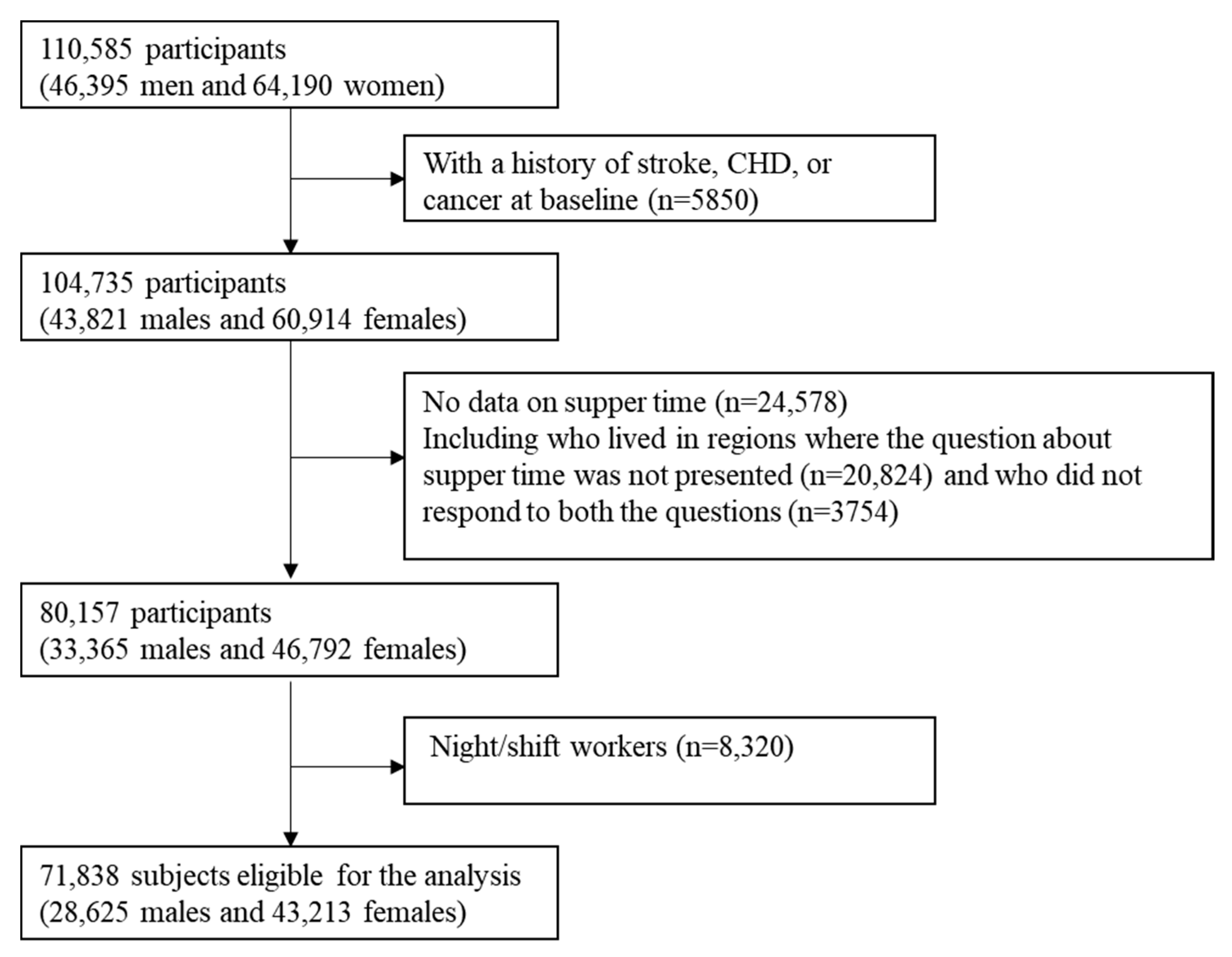
2.1. Study Population
2.2. Ethical Approval
2.3. Dietary Assessment
2.4. Mortality Surveillance
2.5. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- GBD 2015 Mortality and Causes of Death Collaborators. Global, Regional, and National Life Expectancy, All-Cause Mortality, and Cause-Specific Mortality for 249 Causes of Death, 1980–2015: A Systematic Analysis for the Global Burden of Disease Study 2015. Lancet 2016, 388, 1459–1544. [Google Scholar] [CrossRef] [Green Version]
- Nomura, S.; Sakamoto, H.; Glenn, S.; Tsugawa, Y.; Abe, S.K.; Rahman, M.M.; Brown, J.C.; Ezoe, S.; Fitzmaurice, C.; Inokuchi, T.; et al. Population Health and Regional Variations of Disease Burden in Japan, 1990–2015: A Systematic Subnational Analysis for the Global Burden of Disease Study 2015. Lancet 2017, 390, 1521–1538. [Google Scholar] [CrossRef] [Green Version]
- St-Onge, M.P.; Ard, J.; Baskin, M.L.; Chiuve, S.E.; Johnson, H.M.; Kris-Etherton, P.; Varady, K. Meal Timing and Frequency: Implications for Cardiovascular Disease Prevention: A Scientific Statement From the American Heart Association. Circulation 2017, 135, e96–e121. [Google Scholar] [CrossRef]
- Guinter, M.A.; Campbell, P.T.; Patel, A.V.; McCullough, M.L. Irregularity in Breakfast Consumption and Daily Meal Timing Patterns in Association with Body Weight Status and Inflammation. Br. J. Nutr. 2019, 122, 1192–1200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zerón-Rugerio, M.F.; Hernáez, Á.; Porras-Loaiza, A.P.; Cambras, T.; Izquierdo-Pulido, M. Eating Jet Lag: A Marker of the Variability in Meal Timing and Its Association with Body Mass Index. Nutrients 2019, 11, 2980. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cahill Leah, E.; Chiuve Stephanie, E.; Mekary Rania, A.; Jensen Majken, K.; Flint Alan, J.; Hu Frank, B.; Rimm Eric, B. Prospective Study of Breakfast Eating and Incident Coronary Heart Disease in a Cohort of Male US Health Professionals. Circulation 2013, 128, 337–343. [Google Scholar] [CrossRef]
- Tamakoshi, A.; Ozasa, K.; Fujino, Y.; Suzuki, K.; Sakata, K.; Mori, M.; Kikuchi, S.; Iso, H.; JACC Study Group; Sakauchi, F.; et al. Cohort Profile of the Japan Collaborative Cohort Study at Final Follow-Up. J. Epidemiol. 2013, 23, 227–232. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Date, C.; Fukui, M.; Yamamoto, A.; Wakai, K.; Ozeki, A.; Motohashi, Y.; Adachi, C.; Okamoto, N.; Kurosawa, M.; Tokudome, Y.; et al. Reproducibility and Validity of a Self-Administered Food Frequency Questionnaire Used in the JACC Study. J. Epidemiol. 2005, 15, S9–S23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taira, H. Standard Tables of Food Composition in Japan, Fifth Revised Edition. Nippon Shokuhin Kagaku Kogaku Kaishi 2001, 48, 549–553. [Google Scholar] [CrossRef]
- Ohno, Y.; Tamakoshi, A.; JACC Study Group. Japan Collaborative Cohort Study for Evaluation of Cancer Risk Sponsored by Monbusho (JACC Study). J. Epidemiol. 2001, 11, 144–150. [Google Scholar] [CrossRef] [Green Version]
- Oosterman, J.E.; Kalsbeek, A.; la Fleur, S.E.; Belsham, D.D. Impact of Nutrients on Circadian Rhythmicity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 308, R337–R350. [Google Scholar] [CrossRef] [Green Version]
- Albrecht, U. Timing to Perfection: The Biology of Central and Peripheral Circadian Clocks. Neuron 2012, 74, 246–260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McHill, A.W.; Phillips, A.J.; Czeisler, C.A.; Keating, L.; Yee, K.; Barger, L.K.; Garaulet, M.; Scheer, F.A.; Klerman, E.B. Later Circadian Timing of Food Intake Is Associated with Increased Body Fat. Am. J. Clin. Nutr. 2017, 106, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Nagata, C.; Tamura, T.; Wada, K.; Konishi, K.; Goto, Y.; Nagao, Y.; Ishihara, K.; Yamamoto, S. Sleep Duration, Nightshift Work, and the Timing of Meals and Urinary Levels of 8-Isoprostane and 6-Sulfatoxymelatonin in Japanese Women. Chronobiol. Int. 2017, 34, 1187–1196. [Google Scholar] [CrossRef] [PubMed]
- Pickel, L.; Sung, H.-K. Feeding Rhythms and the Circadian Regulation of Metabolism. Front. Nutr. 2020, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Garaulet, M.; Gómez-Abellán, P. Timing of Food Intake and Obesity: A Novel Association. Physiol. Behav. 2014, 134, 44–50. [Google Scholar] [CrossRef]
- Shin, A.; Lim, S.-Y.; Sung, J.; Shin, H.-R.; Kim, J. Dietary Intake, Eating Habits, and Metabolic Syndrome in Korean Men. J. Am. Diet. Assoc. 2009, 109, 633–640. [Google Scholar] [CrossRef]
- Xiao, Q.; Garaulet, M.; Scheer, F.A.J.L. Meal Timing and Obesity: Interactions with Macronutrient Intake and Chronotype. Int. J. Obes. 2019, 43, 1701–1711. [Google Scholar] [CrossRef]
- Mota, M.C.; Silva, C.M.; Balieiro, L.C.T.; Gonçalves, B.F.; Fahmy, W.M.; Crispim, C.A. Association between Social Jetlag Food Consumption and Meal Times in Patients with Obesity-Related Chronic Diseases. PLoS ONE 2019, 14, e0212126. [Google Scholar] [CrossRef] [Green Version]
- Chellappa, S.L.; Vujovic, N.; Williams, J.S.; Scheer, F.A.J.L. Impact of Circadian Disruption on Cardiovascular Function and Disease. Trends Endocrinol. Metab. TEM 2019, 30, 767–779. [Google Scholar] [CrossRef]
- Parsons, M.J.; Moffitt, T.E.; Gregory, A.M.; Goldman-Mellor, S.; Nolan, P.M.; Poulton, R.; Caspi, A. Social Jetlag, Obesity and Metabolic Disorder: Investigation in a Cohort Study. Int. J. Obes. 2015, 39, 842–848. [Google Scholar] [CrossRef] [Green Version]
- Cui, R.; Iso, H.; Toyoshima, H.; Date, C.; Yamamoto, A.; Kikuchi, S.; Kondo, T.; Watanabe, Y.; Koizumi, A.; Wada, Y.; et al. Body mass index and mortality from cardiovascular disease among Japanese men and women: The JACC study. Stroke 2005, 36, 1377–1382. [Google Scholar] [CrossRef] [Green Version]
- Kaneko, H.; Itoh, H.; Kiriyama, H.; Kamon, T.; Fujiu, K.; Morita, K.; Michihata, N.; Jo, T.; Takeda, N.; Morita, H.; et al. Possible Association between Eating Behaviors and Cardiovascular Disease in the General Population: Analysis of a Nationwide Epidemiological Database. Atherosclerosis 2021, 320, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Ofori-Asenso, R.; Owen, A.J.; Liew, D. Skipping Breakfast and the Risk of Cardiovascular Disease and Death: A Systematic Review of Prospective Cohort Studies in Primary Prevention Settings. J. Cardiovasc. Dev. Dis. 2019, 6, 30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yasuhiko, K.; Hiroyasu, I.; Norie, S.; Shoichiro, T. Association of Breakfast Intake with Incident Stroke and Coronary Heart Disease. Stroke 2016, 47, 477–481. [Google Scholar] [CrossRef] [Green Version]
- Yoshida, J.; Eguchi, E.; Nagaoka, K.; Ito, T.; Ogino, K. Association of Night Eating Habits with Metabolic Syndrome and Its Components: A Longitudinal Study. BMC Public Health 2018, 18, 1366. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Brereton, N.; Schweitzer, A.; Cotter, M.; Duan, D.; Børsheim, E.; Wolfe, R.R.; Pham, L.V.; Polotsky, V.Y.; Jun, J.C. Metabolic Effects of Late Dinner in Healthy Volunteers-A Randomized Crossover Clinical Trial. J. Clin. Endocrinol. Metab. 2020, 105, 2789–2802. [Google Scholar] [CrossRef]
- Kutsuma, A.; Nakajima, K.; Suwa, K. Potential Association between Breakfast Skipping and Concomitant Late-Night-Dinner Eating with Metabolic Syndrome and Proteinuria in the Japanese Population. Scientifica 2014, 2014, e253581. [Google Scholar] [CrossRef]
- Okada, C.; Imano, H.; Muraki, I.; Yamada, K.; Iso, H. The Association of Having a Late Dinner or Bedtime Snack and Skipping Breakfast with Overweight in Japanese Women. J. Obes. 2019, 2019, 2439571. [Google Scholar] [CrossRef]
- Kito, K.; Kuriyama, A.; Takahashi, Y.; Nakayama, T. Impacts of Skipping Breakfast and Late Dinner on the Incidence of Being Overweight: A 3-Year Retrospective Cohort Study of Men Aged 20–49 Years. J. Hum. Nutr. Diet. Off. J. Br. Diet. Assoc. 2019, 32, 349–355. [Google Scholar] [CrossRef]
- Sandhu, S.K.; Tang, T.S. When’s Dinner? Does Timing of Dinner Affect the Cardiometabolic Risk Profiles of South-Asian Canadians at Risk for Diabetes. Diabet. Med. J. Br. Diabet. Assoc. 2017, 34, 539–542. [Google Scholar] [CrossRef]
- Baron, K.G.; Reid, K.J.; Horn, L.V.; Zee, P.C. Contribution of Evening Macronutrient Intake to Total Caloric Intake and Body Mass Index. Appetite 2013, 60, 246–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Coulthard, J.D.; Pot, G.K. The timing of the evening meal: How is this associated with weight status in UK children? Br. J. Nutr. 2016, 115, 1616–1622. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baron, K.G.; Reid, K.J.; Kern, A.S.; Zee, P.C. Role of sleep timing in caloric intake and BMI. Obesity 2011, 19, 1374–1381. [Google Scholar] [CrossRef] [PubMed]
| Supper Time, p.m. | |||
|---|---|---|---|
| Always ≤8 | Irregular | Always >8 | |
| No. of subjects | 66198 | 3875 | 1765 |
| Age (years) | 58 ± 10 | 52 ± 9 ‡ | 52 ± 9 ‡ |
| Sex (male %) | 39 | 55 ‡ | 54 ‡ |
| Body mass index (kg/m2) | 22.7 ± 3.0 | 22.9 ± 3.0 | 22.8 ± 2.9 |
| Overweight-BMI ≥ 25 kg/m2 (%) | 20 | 23 ‡ | 20 |
| BMI < 18.5 kg/m2 (%) | 6 | 5 | 5 |
| Current smokers (%) | 24 | 39 ‡ | 36 ‡ |
| Alcohol intake (g/day of ethanol) | 28 ± 22 | 34 ± 26 ‡ | 30 ± 24 * |
| History of hypertension (%) | 22 | 16 | 14 |
| History of diabetes mellitus (%) | 5 | 5 ‡ | 4 |
| Sports time ≥ 5 h/week (%) | 6 | 4 | 4 |
| Walking time ≥ 1 h/day (%) | 51 | 45 ‡ | 48 * |
| Education level ≥ college (%) | 13 | 17 ‡ | 21 ‡ |
| High perceived mental stress (%) | 19 | 39 ‡ | 37 ‡ |
| Sleeping time (hours/day) | 7.3 ± 1.1 | 6.9 ± 1.0 ‡ | 6.8 ± 1.0 ‡ |
| Energy intake (kcal/day) | 1558 ± 456 | 1547 ± 463 † | 1538 ± 455 † |
| Sodium intake (mg/day) | 2017 ± 908 | 1772 ± 869 ‡ | 1772 ± 854 ‡ |
| Potassium intake (mg/day) | 2137 ± 614 | 1971 ± 611 ‡ | 1992 ± 617 ‡ |
| Calcium intake (mg/day) | 465 ± 156 | 433 ± 162 ‡ | 444 ± 161 ‡ |
| Cholesterol intake (mg/day) | 229 ± 89 | 215 ± 88 ‡ | 218 ± 89 ‡ |
| Saturated fatty acids intake(g/day) | 9.9 ± 3.7 | 9.8 ± 3.9 ‡ | 10.2 ± 4.0 |
| N-3 fatty acids intake (g/day) | 1.5 ± 0.6 | 1.3 ± 0.6 ‡ | 1.3 ± 0.6 ‡ |
| Vitamin D intake (µg/day) | 6.7 ± 3.2 | 6.1 ± 3.1 ‡ | 6.1 ± 3.1 ‡ |
| Fat intake (g/day) | 32.4 ± 10.9 | 30.8 ± 11 ‡ | 31.8 ± 11.2 † |
| Protein intake (g/day) | 54 ± 15 | 50 ± 15 ‡ | 50 ± 15 ‡ |
| Carbohydrate intake (g/day) | 238 ± 75 | 225 ± 75 ‡ | 225 ± 76 ‡ |
| Total dietary fiber intake (g/day) | 12.4 ± 3.8 | 11.1 ± 3.7 ‡ | 11.1 ± 3.7 ‡ |
| Vegetable intake (g/day) | 276 ± 322 | 226 ± 298 ‡ | 269 ± 321 |
| Fruit intake (g/day) | 88 ± 52 | 79 ± 53 ‡ | 78 ± 53 ‡ |
| Miso soup every day (%) | 71 | 60 ‡ | 61 ‡ |
| Other soy products intake (g/day) | 40 ± 24 | 33 ± 22 ‡ | 35 ± 23 ‡ |
| Seaweed intake (g/day) | 3.8 ± 1.0 | 3.6 ± 1.0 ‡ | 3.6 ± 1.1 ‡ |
| Milk and dairy products intake (g/day) | 92 ± 72 | 85 ± 71 ‡ | 87 ± 72 |
| Meat intake (g/day) | 29 ± 20 | 29 ± 21 ‡ | 29 ± 21 |
| Total seafood intake (g/day) | 50 ± 28 | 45 ± 27 ‡ | 45 ± 27 ‡ |
| Fresh fish intake (g/day) | 31 ± 21 | 29 ± 21 ‡ | 28 ± 20 ‡ |
| Egg intake (mean times/week) | 4.3 ± 2.5 | 4.1 ± 2.5 ‡ | 4.2 ± 2.6 † |
| Coffee intake every day (%) | 50 | 68 ‡ | 69 ‡ |
| Green tea intake every day (%) | 84 | 80 ‡ | 81 * |
| Marital status (married %) | 88 | 88 ‡ | 90 † |
| Skipping breakfast (%) | 2 | 8 ‡ | 8 ‡ |
| Supper Time, p.m. | |||
|---|---|---|---|
| Always ≤8 | Irregular | Always >8 | |
| No. of subjects | 66,198 | 3875 | 1765 |
| Person-years | 1,072,692 | 63,451 | 29,514 |
| Total stroke, n | 1952 | 69 | 23 |
| Age and sex-adjusted HR (95% CI) | 1 | 1.23 (0.97–1.57) | 0.87 (0.58–1.32) |
| Multivariable HR (95% CI) * | 1 | 1.16 (0.91–1.48) | 0.82 (0.54–1.24) |
| Multivariable HR (95% CI) † | 1 | 1.16 (0.91–1.48) | 0.81 (0.53–1.22) |
| Cerebral infarction, n | 1095 | 23 | 10 |
| Age and sex-adjusted HR (95% CI) | 1 | 0.86 (0.57–1.30) | 0.78 (0.42–1.46) |
| Multivariable HR (95% CI) * | 1 | 0.80 (0.53–1.21) | 0.73 (0.39–1.37) |
| Multivariable HR (95% CI) † | 1 | 0.81 (0.53–1.23) | 0.71 (0.38–1.32) |
| Hemorrhagic stroke, n | 790 | 43 | 13 |
| Age and sex--adjusted HR (95% CI) | 1 | 1.54 (1.13–2.09) | 1.00 (0.58–1.73) |
| Multivariable HR (95% CI) * | 1 | 1.43 (1.05–1.96) | 0.94 (0.54–1.63) |
| Multivariable HR (95% CI) † | 1 | 1.44 (1.05–1.97) | 0.94 (0.54–1.63) |
| Coronary heart disease, n | 908 | 32 | 14 |
| Age and sex-adjusted HR (95% CI) | 1 | 1.19 (0.83–1.69) | 1.10 (0.65–1.87) |
| Multivariable HR (95% CI) * | 1 | 1.05 (0.74–1.50) | 1.02 (0.60–1.73) |
| Multivariable HR (95% CI) † | 1 | 1.04 (0.72–1.48) | 0.99 (0.58–1.69) |
| Total cardiovascular disease, n | 4492 | 154 | 60 |
| Age and sex-adjusted HR (95% CI) | 1 | 1.20 (1.02–1.41) | 1.00 (0.77–1.29) |
| Multivariable HR (95% CI) * | 1 | 1.13 (0.96–1.33) | 0.94 (0.73–1.22) |
| Multivariable HR (95% CI) † | 1 | 1.12 (0.95–1.32) | 0.92 (0.71–1.19) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tang, J.; Dong, J.-Y.; Eshak, E.S.; Cui, R.; Shirai, K.; Liu, K.; Sakaniwa, R.; Tamakoshi, A.; Iso, H.; on behalf of the JACC Study Group. Supper Timing and Cardiovascular Mortality: The Japan Collaborative Cohort Study. Nutrients 2021, 13, 3389. https://doi.org/10.3390/nu13103389
Tang J, Dong J-Y, Eshak ES, Cui R, Shirai K, Liu K, Sakaniwa R, Tamakoshi A, Iso H, on behalf of the JACC Study Group. Supper Timing and Cardiovascular Mortality: The Japan Collaborative Cohort Study. Nutrients. 2021; 13(10):3389. https://doi.org/10.3390/nu13103389
Chicago/Turabian StyleTang, Jingyun, Jia-Yi Dong, Ehab S. Eshak, Renzhe Cui, Kokoro Shirai, Keyang Liu, Ryoto Sakaniwa, Akiko Tamakoshi, Hiroyasu Iso, and on behalf of the JACC Study Group. 2021. "Supper Timing and Cardiovascular Mortality: The Japan Collaborative Cohort Study" Nutrients 13, no. 10: 3389. https://doi.org/10.3390/nu13103389
APA StyleTang, J., Dong, J.-Y., Eshak, E. S., Cui, R., Shirai, K., Liu, K., Sakaniwa, R., Tamakoshi, A., Iso, H., & on behalf of the JACC Study Group. (2021). Supper Timing and Cardiovascular Mortality: The Japan Collaborative Cohort Study. Nutrients, 13(10), 3389. https://doi.org/10.3390/nu13103389






