Effects of Caloric Restriction Diet on Arterial Hypertension and Endothelial Dysfunction
Abstract
1. Introduction
2. Definition, Classification, and Management of Arterial Hypertension
3. Arterial Hypertension and Endothelial Alterations
4. Methods
5. Caloric Restriction Diet
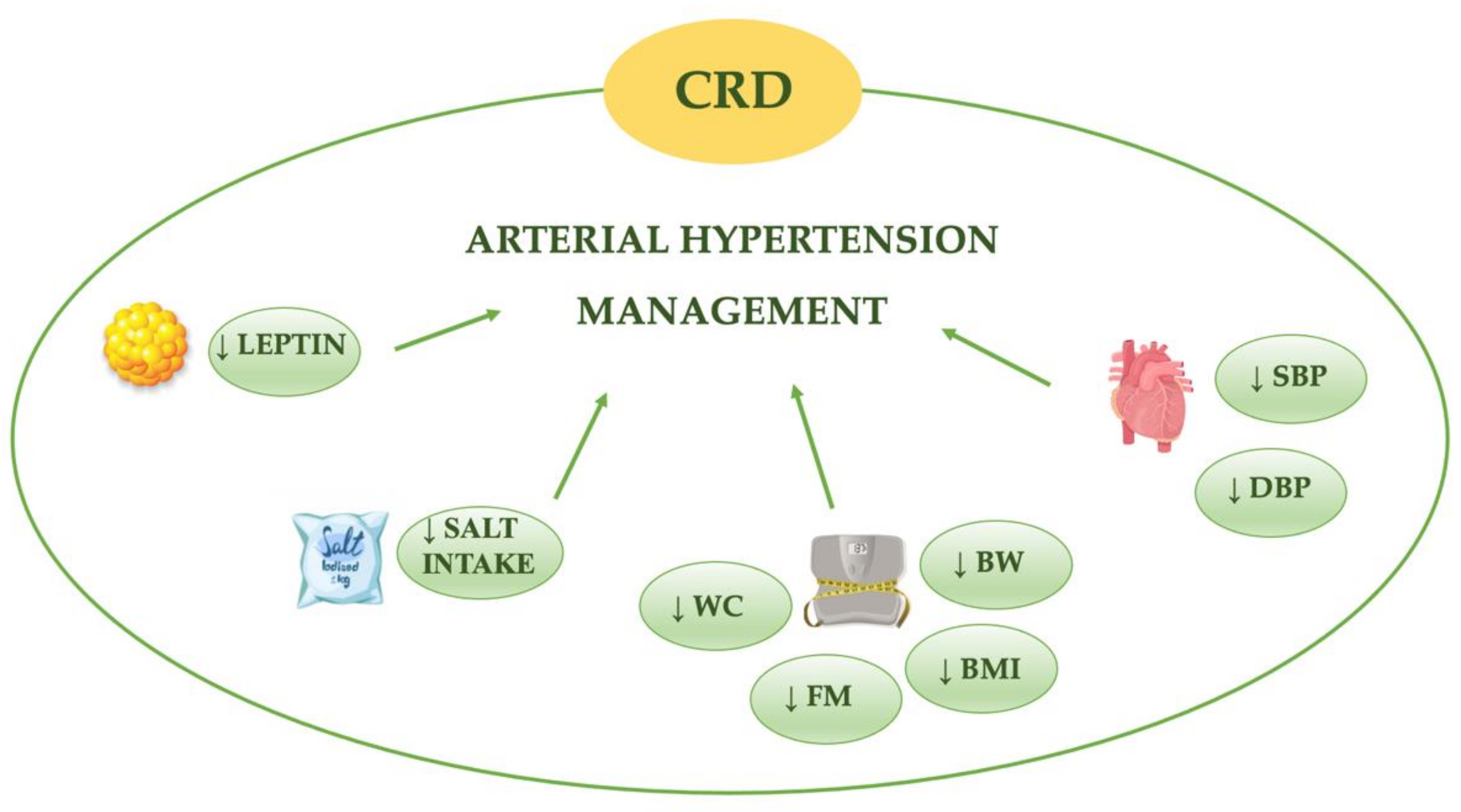
6. Caloric Restriction Diet and Arterial Hypertension
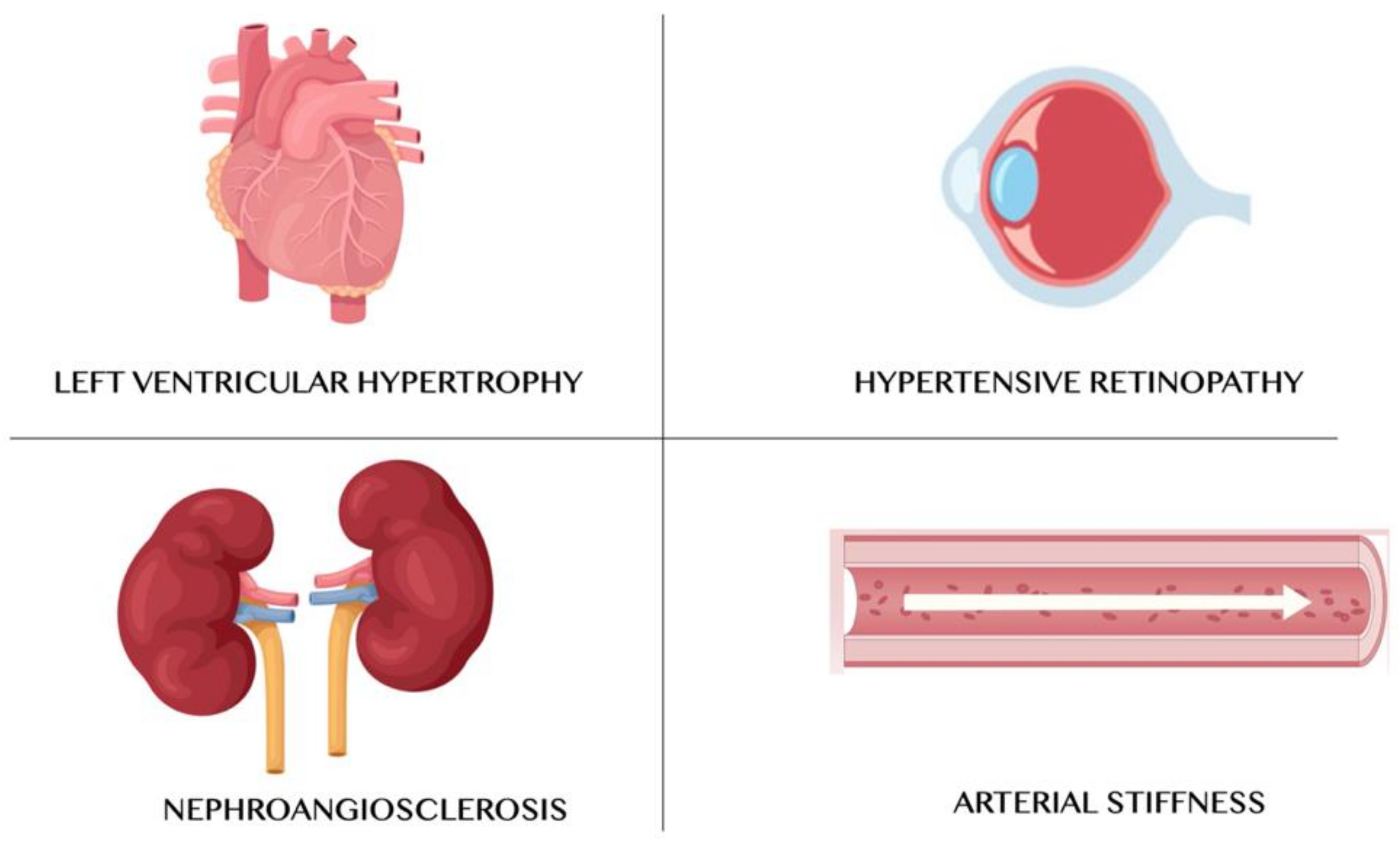
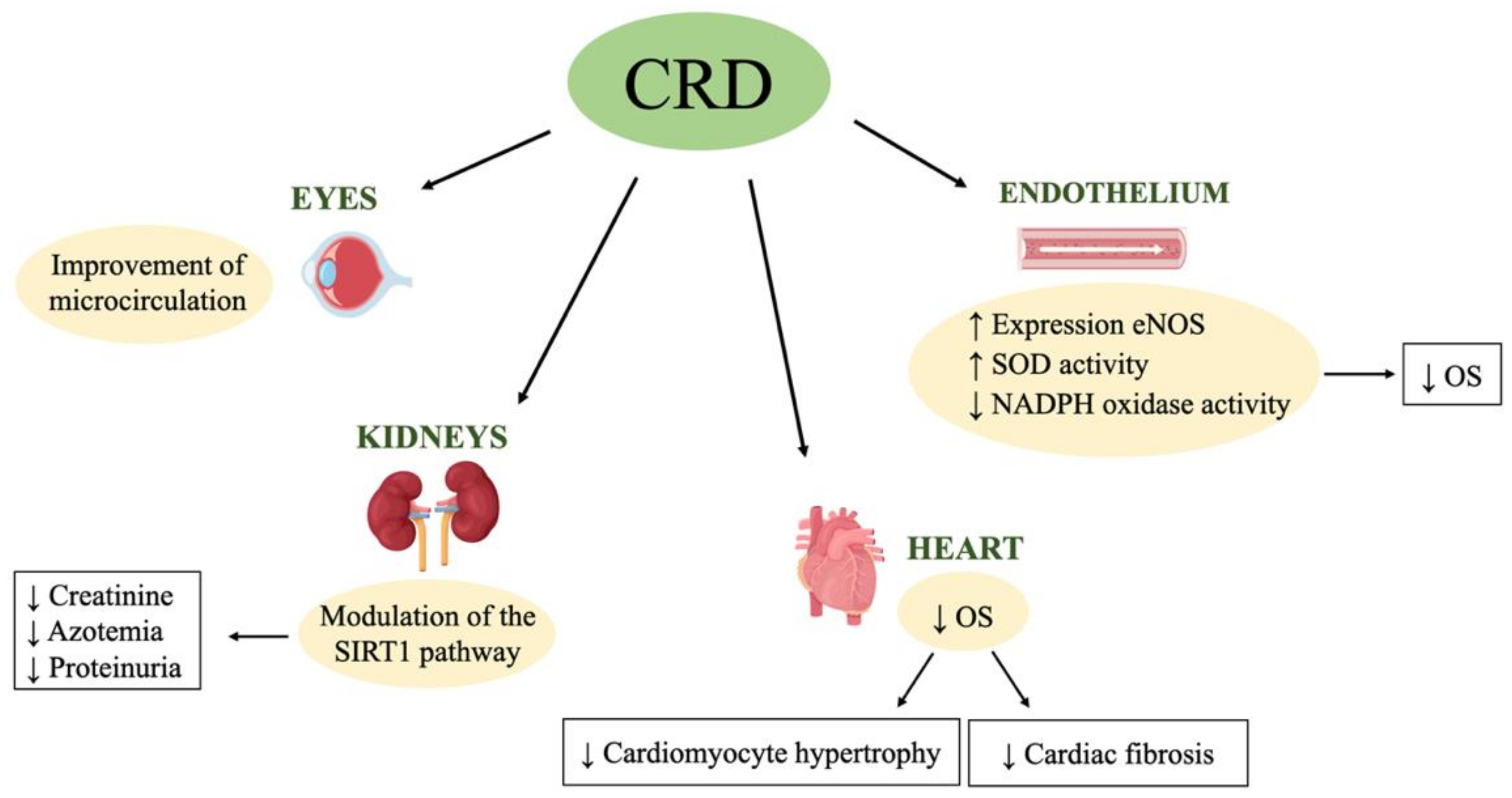
7. Caloric Restriction Diet and Hypertensive Organ Damages
7.1. Left Ventricular Hypertrophy
7.2. Kidney Damage—Nephroangiosclerosis
7.3. Arterial Stiffness
7.4. Hypertensive Retinopathy
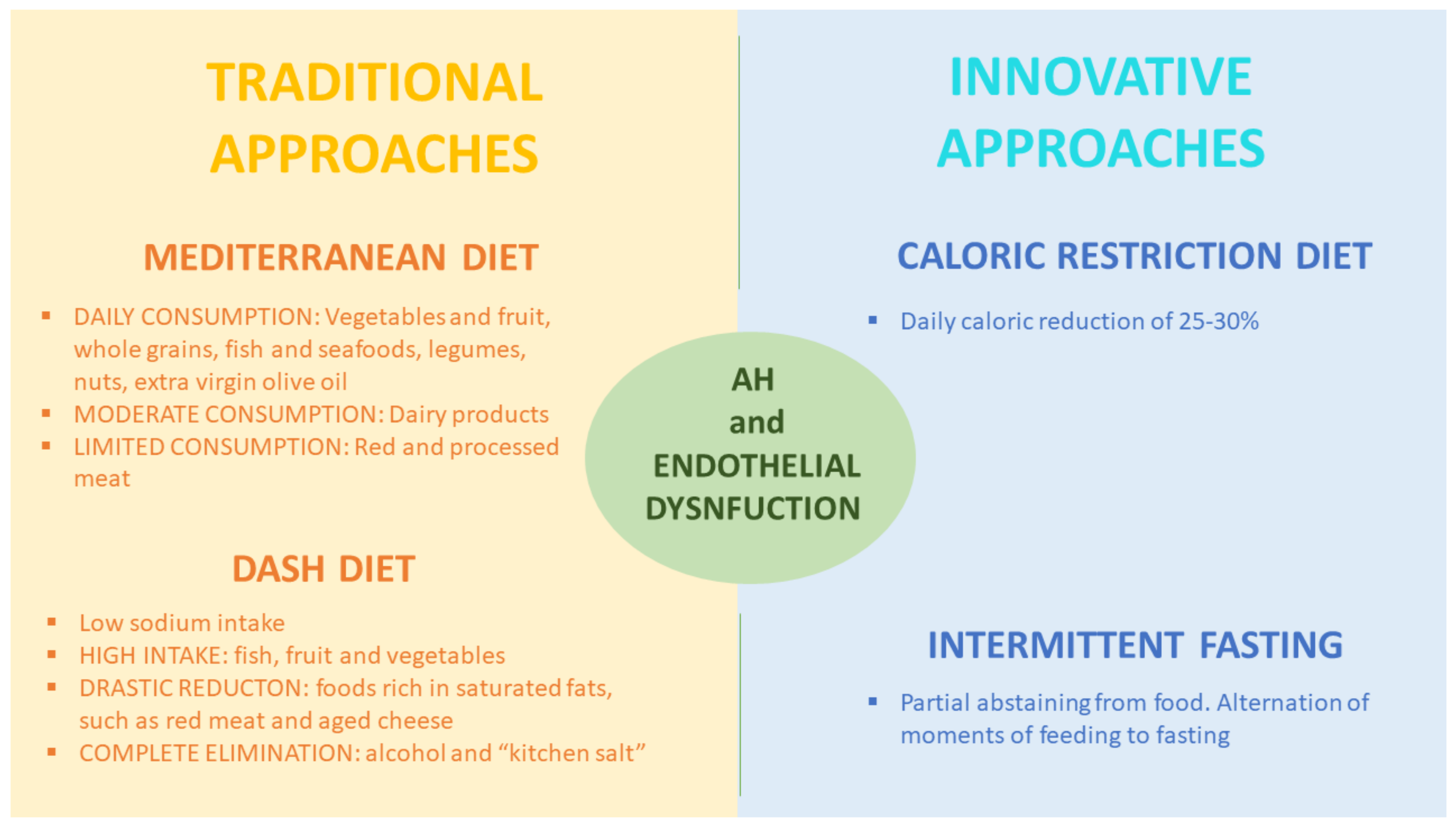
8. Secondary AH: Traditional Dietary Nutritional Protocols
9. Other Innovative Nutritional Approaches for Essential AH Treatment
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
List of Acronyms
| ACE | angiotensin conversion enzyme |
| AH | arterial hypertension |
| AMPK | AMP-activated protein kinase |
| ARB | angiotensin receptor blockers |
| AS | arterial stiffness |
| BMI | body mass index |
| BP | blood pressure |
| BW | body weight |
| CALERIE | Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy |
| cBP | central blood pressure |
| cGMP | cyclic guanosine monophosphate |
| CKD | chronic kidney disease |
| CRD | caloric restriction diet |
| CRP | c-reactive protein |
| CV | cardiovascular diseases |
| DBP | diastolic blood pressure |
| eNOS | endothelial nitric oxide synthase |
| ESC | European Society of Cardiology |
| ESH | European Society of Hypertension |
| HDL-c | high-density lipoprotein cholesterol |
| HRV | heart rate variability |
| IF | intermittent fasting |
| IGF-1 | insulin-like growth factor-1 |
| IL | interleukin |
| LDL-c | low-density lipoprotein cholesterol |
| LVH | left ventricular hypertrophy |
| m-TOR | mammalian target of rapamycin |
| MBIP | model-based image processing |
| MDRD | modification of diet in real disease study |
| NAD | nicotinamide dinucleotide |
| NAS | nephroangiosclerosis |
| NIH | National Institute of Health |
| NO | nitric oxide |
| NOS | nitric oxide-synthase |
| OS | oxidative stress |
| PGC1-α | peroxisome proliferator-activated receptor gamma coactivator 1-α |
| PWV | pulsating wave velocity |
| RAAS | renin–angiotensin–aldosterone system |
| ROS | reactive oxygen species |
| SBP | systolic bloody pressure |
| SIRT1 | sirtuin1 |
| SOD | superoxide dismutase |
| TGF- β1 | transforming growth factor-β1 |
| TNF | tumor necrosis factor |
| TRF | time-restricted feeding |
| UAE | urinary albumin excretion |
| WHO | World Health Organization |
References
- Kitt, J.; Fox, R.; Tucker, K.L.; McManus, R.J. New Approaches in Hypertension Management: A Review of Current and Developing Technologies and Their Potential Impact on Hypertension Care. Curr. Hypertens. Rep. 2019, 21, 44. [Google Scholar] [CrossRef] [PubMed]
- Jordan, J.; Kurschat, C.; Reuter, H. Arterial Hypertension. Dtsch. Arztebl. Int. 2018, 115, 557–568. [Google Scholar] [CrossRef]
- World Healt Organization (WHO). Hypertension. Available online: https://www.who.int/health-topics/hypertension/#tab=tab_1 (accessed on 13 November 2020).
- Lawes, C.M.; Vander Hoorn, S.; Rodgers, A.; International Society of Hypertension. Global burden of blood-pressure-related disease, 2001. Lancet 2008, 371, 1513–1518. [Google Scholar] [CrossRef]
- Yusuf, S.; Hawken, S.; Ounpuu, S.; Dans, T.; Avezum, A.; Lanas, F.; McQueen, M.; Budaj, A.; Pais, P.; Varigos, J.; et al. Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): Case-control study. Lancet 2004, 364, 937–952. [Google Scholar] [CrossRef]
- Volpe, M.; Rosei, E.A.; Ambrosioni, E.; Cottone, S.; Cuspidi, C.; Borghi, C.; de Luca, N.; Fallo, F.; Ferri, C.; Morganti, A.; et al. 2012 consensus document of the Italian Society of Hypertension (SIIA): Strategies to improve blood pressure control in Italy: From global cardiovascular risk stratification to combination therapy. High Blood Press Cardiovasc. Prev. 2013, 20, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Flack, J.M.; Adekola, B. Blood pressure and the new ACC/AHA hypertension guidelines. Trends Cardiovasc. Med. 2020, 30, 160–164. [Google Scholar] [CrossRef] [PubMed]
- Brandhorst, S.; Longo, V.D. Dietary Restrictions and Nutrition in the Prevention and Treatment of Cardiovascular Disease. Circ. Res. 2019, 124, 952–965. [Google Scholar] [CrossRef] [PubMed]
- Kord-Varkaneh, H.; Nazary-Vannani, A.; Mokhtari, Z.; Salehi-Sahlabadi, A.; Rahmani, J.; Clark, C.C.T.; Fatahi, S.; Zanghelini, F.; Hekmatdoost, A.; Okunade, K.; et al. The Influence of Fasting and Energy Restricting Diets on Blood Pressure in Humans: A Systematic Review and Meta-Analysis. High Blood Press Cardiovasc. Prev. 2020, 27, 271–280. [Google Scholar] [CrossRef]
- Ross, R.; Faggiotto, A.; Bowen-Pope, D.; Raines, E. The role of endothelial injury and platelet and macrophage interactions in atherosclerosis. Circulation 1984, 70, III77–III82. [Google Scholar]
- Lerman, A.; Zeiher, A.M. Endothelial function: Cardiac events. Circulation 2005, 111, 363–368. [Google Scholar] [CrossRef]
- Behrendt, D.; Ganz, P. Endothelial function. From vascular biology to clinical applications. Am. J. Cardiol. 2002, 90, 40L–48L. [Google Scholar] [CrossRef]
- Davis, N.; Katz, S.; Wylie-Rosett, J. The effect of diet on endothelial function. Cardiol. Rev. 2007, 15, 62–66. [Google Scholar] [CrossRef]
- Davignon, J.; Ganz, P. Role of endothelial dysfunction in atherosclerosis. Circulation 2004, 109, III27–III32. [Google Scholar] [CrossRef]
- Gonzalez, M.A.; Selwyn, A.P. Endothelial function, inflammation, and prognosis in cardiovascular disease. Am. J. Med. 2003, 115, 99S–106S. [Google Scholar] [CrossRef]
- Sullivan, D. The Effects of Hypertension on the Body. Available online: https://www.healthline.com/health/high-blood-pressure-hypertension/effect-on-body (accessed on 10 November 2020).
- Williams, B.; Mancia, G.; Spiering, W.; Agabiti Rosei, E.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; de Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104. [Google Scholar] [CrossRef]
- Frisoli, T.M.; Schmieder, R.E.; Grodzicki, T.; Messerli, F.H. Beyond salt: Lifestyle modifications and blood pressure. Eur. Heart J. 2011, 32, 3081–3087. [Google Scholar] [CrossRef] [PubMed]
- OsMed. Appropriatezza Prescrittiva e Aderenza alle Terapie. Il Quadro Emerso dagli Indicatori OsMed per Alcune Aree Terapeutiche. Available online: http://www.agenziafarmaco.gov.it/content/appropriatezza-prescrittiva-e-aderenza-alle-terapie-il-quadro-emerso-dagli-indicatori-osmed- (accessed on 15 October 2020).
- Scuteri, A.; Modestino, A.; Frattari, A.; di Daniele, N.; Tesauro, M. Occurrence of Hypotension in Older Participants. Which 24-hour ABPM Parameter Better Correlate With? J. Gerontol. A Biol. Sci. Med. Sci. 2012, 67, 804–810. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zheng, H.; Du, H.B.; Tian, X.P.; Jiang, Y.J.; Zhang, S.L.; Kang, Y.; Li, X.; Chen, J.; Lu, C.; et al. The multiple lifestyle modification for patients with prehypertension and hypertension patients: A systematic review protocol. BMJ Open 2014, 4, e004920. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, F.D.; Whelton, P.K. High Blood Pressure and Cardiovascular Disease. Hypertension 2020, 75, 285–292. [Google Scholar] [CrossRef] [PubMed]
- Versari, D.; Daghini, E.; Virdis, A.; Ghiadoni, L.; Taddei, S. Endothelial dysfunction as a target for prevention of cardiovascular disease. Diabetes Care 2009, 32, S314–S321. [Google Scholar] [CrossRef]
- Sandoo, A.; van Zanten, J.J.; Metsios, G.S.; Carroll, D.; Kitas, G.D. The endothelium and its role in regulating vascular tone. Open Cardiovasc. Med. J. 2010, 4, 302–312. [Google Scholar] [CrossRef] [PubMed]
- Van Hinsbergh, V.W. Endothelium--role in regulation of coagulation and inflammation. Semin. Immunopathol. 2012, 34, 93–106. [Google Scholar] [CrossRef]
- Godo, S.; Shimokawa, H. Endothelial Functions. Arterioscler. Thromb Vasc. Biol. 2017, 37, e108–e114. [Google Scholar] [CrossRef] [PubMed]
- Forstermann, U.; Sessa, W.C. Nitric oxide synthases: Regulation and function. Eur. Heart J. 2012, 33, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Sriram, K.; Laughlin, J.G.; Rangamani, P.; Tartakovsky, D.M. Shear-Induced Nitric Oxide Production by Endothelial Cells. Biophys. J. 2016, 111, 208–221. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.Y.; Griendling, K.K. Redox signaling, vascular function, and hypertension. Antioxid. Redox Signal. 2008, 10, 1045–1059. [Google Scholar] [CrossRef]
- Gimbrone, M.A., Jr.; Garcia-Cardena, G. Endothelial Cell Dysfunction and the Pathobiology of Atherosclerosis. Circ. Res. 2016, 118, 620–636. [Google Scholar] [CrossRef]
- Gryglewski, R.J.; Palmer, R.M.; Moncada, S. Superoxide anion is involved in the breakdown of endothelium-derived vascular relaxing factor. Nature 1986, 320, 454–456. [Google Scholar] [CrossRef]
- Luscher, T.F. Endothelial dysfunction: The role and impact of the renin-angiotensin system. Heart 2000, 84, i20–i22:discussion i50. [Google Scholar] [CrossRef][Green Version]
- Campia, U.; Tesauro, M.; di Daniele, N.; Cardillo, C. The vascular endothelin system in obesity and type 2 diabetes: Pathophysiology and therapeutic implications. Life Sci. 2014, 118, 149–155. [Google Scholar] [CrossRef]
- Davenport, A.P.; Hyndman, K.A.; Dhaun, N.; Southan, C.; Kohan, D.E.; Pollock, J.S.; Pollock, D.M.; Webb, D.J.; Maguire, J.J. Endothelin. Pharmacol. Rev. 2016, 68, 357–418. [Google Scholar] [CrossRef] [PubMed]
- Flammer, A.J.; Anderson, T.; Celermajer, D.S.; Creager, M.A.; Deanfield, J.; Ganz, P.; Hamburg, N.M.; Luscher, T.F.; Shechter, M.; Taddei, S.; et al. The assessment of endothelial function: From research into clinical practice. Circulation 2012, 126, 753–767. [Google Scholar] [CrossRef]
- Dharmashankar, K.; Widlansky, M.E. Vascular endothelial function and hypertension: Insights and directions. Curr. Hypertens. Rep. 2010, 12, 448–455. [Google Scholar] [CrossRef]
- Dinh, Q.N.; Drummond, G.R.; Sobey, C.G.; Chrissobolis, S. Roles of inflammation, oxidative stress, and vascular dysfunction in hypertension. Biomed. Res. Int. 2014, 2014, 406960. [Google Scholar] [CrossRef]
- Mordi, I.; Mordi, N.; Delles, C.; Tzemos, N. Endothelial dysfunction in human essential hypertension. J. Hypertens. 2016, 34, 1464–1472. [Google Scholar] [CrossRef]
- Yannoutsos, A.; Levy, B.I.; Safar, M.E.; Slama, G.; Blacher, J. Pathophysiology of hypertension: Interactions between macro and microvascular alterations through endothelial dysfunction. J. Hypertens. 2014, 32, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Most, J.; Tosti, V.; Redman, L.M.; Fontana, L. Calorie restriction in humans: An update. Ageing Res. Rev. 2017, 39, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Meyer, T.E.; Klein, S.; Holloszy, J.O. Long-term calorie restriction is highly effective in reducing the risk for atherosclerosis in humans. Proc. Natl. Acad. Sci. USA 2004, 101, 6659–6663. [Google Scholar] [CrossRef]
- Osborne, T.B.; Mendel, L.B.; Ferry, E.L. The Effect of Retardation of Growth Upon the Breeding Period and Duration of Life of Rats. Science 1917, 45, 294–295. [Google Scholar] [CrossRef]
- Weindruch, R.; Walford, R.L.; Fligiel, S.; Guthrie, D. The retardation of aging in mice by dietary restriction: Longevity, cancer, immunity and lifetime energy intake. J. Nutr. 1986, 116, 641–654. [Google Scholar] [CrossRef]
- Lee, S.H.; Min, K.J. Caloric restriction and its mimetics. BMB Rep. 2013, 46, 181–187. [Google Scholar] [CrossRef]
- Zhang, Y.; Ikeno, Y.; Qi, W.; Chaudhuri, A.; Li, Y.; Bokov, A.; Thorpe, S.R.; Baynes, J.W.; Epstein, C.; Richardson, A.; et al. Mice deficient in both Mn superoxide dismutase and glutathione peroxidase-1 have increased oxidative damage and a greater incidence of pathology but no reduction in longevity. J. Gerontol. A Biol. Sci. Med. Sci. 2009, 64, 1212–1220. [Google Scholar] [CrossRef]
- Smirnov, A.; Panatta, E.; Lena, A.; Castiglia, D.; di Daniele, N.; Melino, G.; Candi, E. FOXM1 regulates proliferation, senescence and oxidative stress in keratinocytes and cancer cells. Aging (Albany N. Y.) 2016, 8, 1384–1397. [Google Scholar] [CrossRef]
- Singh, R.; Lakhanpal, D.; Kumar, S.; Sharma, S.; Kataria, H.; Kaur, M.; Kaur, G. Late-onset intermittent fasting dietary restriction as a potential intervention to retard age-associated brain function impairments in male rats. Age 2012, 34, 917–933. [Google Scholar] [CrossRef] [PubMed]
- Anisimov, V.N.; Bartke, A. The key role of growth hormone-insulin-IGF-1 signaling in aging and cancer. Crit. Rev. Oncol. Hematol. 2013, 87, 201–223. [Google Scholar] [CrossRef]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Curb, J.D.; Suzuki, M. Caloric restriction and human longevity: What can we learn from the Okinawans? Biogerontology 2006, 7, 173–177. [Google Scholar] [CrossRef]
- Colman, R.J.; Anderson, R.M.; Johnson, S.C.; Kastman, E.K.; Kosmatka, K.J.; Beasley, T.M.; Allison, D.B.; Cruzen, C.; Simmons, H.A.; Kemnitz, J.W.; et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science 2009, 325, 201–204. [Google Scholar] [CrossRef]
- Mattison, J.A.; Roth, G.S.; Beasley, T.M.; Tilmont, E.M.; Handy, A.M.; Herbert, R.L.; Longo, D.L.; Allison, D.B.; Young, J.E.; Bryant, M.; et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 2012, 489, 318–321. [Google Scholar] [CrossRef]
- Guarente, L. Sir2 links chromatin silencing, metabolism, and aging. Genes Dev. 2000, 14, 1021–1026. [Google Scholar]
- Toiber, D.; Sebastian, C.; Mostoslavsky, R. Characterization of nuclear sirtuins: Molecular mechanisms and physiological relevance. Handb. Exp. Pharmacol. 2011, 206, 189–224. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Lee, J.H.; Lee, H.Y.; Min, K.J. Sirtuin signaling in cellular senescence and aging. BMB Rep. 2019, 52, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Ogawa, K.; Ikei, T.; Udono, M.; Fujiki, T.; Katakura, Y. SIRT1 prevents replicative senescence of normal human umbilical cord fibroblast through potentiating the transcription of human telomerase reverse transcriptase gene. Biochem. Biophys. Res. Commun. 2012, 417, 630–634. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.M.; Xiao, C.; Finley, L.W.; Lahusen, T.; Souza, A.L.; Pierce, K.; Li, Y.H.; Wang, X.; Laurent, G.; German, N.J.; et al. SIRT4 has tumor-suppressive activity and regulates the cellular metabolic response to DNA damage by inhibiting mitochondrial glutamine metabolism. Cancer Cell 2013, 23, 450–463. [Google Scholar] [CrossRef] [PubMed]
- Cohen, H.Y.; Miller, C.; Bitterman, K.J.; Wall, N.R.; Hekking, B.; Kessler, B.; Howitz, K.T.; Gorospe, M.; de Cabo, R.; Sinclair, D.A. Calorie restriction promotes mammalian cell survival by inducing the SIRT1 deacetylase. Science 2004, 305, 390–392. [Google Scholar] [CrossRef]
- Chen, D.; Bruno, J.; Easlon, E.; Lin, S.J.; Cheng, H.L.; Alt, F.W.; Guarente, L. Tissue-specific regulation of SIRT1 by calorie restriction. Genes Dev. 2008, 22, 1753–1757. [Google Scholar] [CrossRef]
- Deota, S.; Chattopadhyay, T.; Ramachandran, D.; Armstrong, E.; Camacho, B.; Maniyadath, B.; Fulzele, A.; Gonzalez-de-Peredo, A.; Denu, J.M.; Kolthur-Seetharam, U. Identification of a Tissue-Restricted Isoform of SIRT1 Defines a Regulatory Domain that Encodes Specificity. Cell Rep. 2017, 18, 3069–3077. [Google Scholar] [CrossRef]
- Meyer, T.E.; Kovacs, S.J.; Ehsani, A.A.; Klein, S.; Holloszy, J.O.; Fontana, L. Long-term caloric restriction ameliorates the decline in diastolic function in humans. J. Am. Coll. Cardiol. 2006, 47, 398–402. [Google Scholar] [CrossRef]
- Sohal, R.S.; Weindruch, R. Oxidative stress, caloric restriction, and aging. Science 1996, 273, 59–63. [Google Scholar] [CrossRef]
- Liguori, I.; Russo, G.; Curcio, F.; Bulli, G.; Aran, L.; Della-Morte, D.; Gargiulo, G.; Testa, G.; Cacciatore, F.; Bonaduce, D.; et al. Oxidative stress, aging, and diseases. Clin. Interv. Aging 2018, 13, 757–772. [Google Scholar] [CrossRef]
- Ingram, D.K.; Cutler, R.G.; Weindruch, R.; Renquist, D.M.; Knapka, J.J.; April, M.; Belcher, C.T.; Clark, M.A.; Hatcherson, C.D.; Marriott, B.M.; et al. Dietary restriction and aging: The initiation of a primate study. J. Gerontol. 1990, 45, B148–B163. [Google Scholar] [CrossRef]
- Zanetti, M.; Gortan Cappellari, G.; Burekovic, I.; Barazzoni, R.; Stebel, M.; Guarnieri, G. Caloric restriction improves endothelial dysfunction during vascular aging: Effects on nitric oxide synthase isoforms and oxidative stress in rat aorta. Exp. Gerontol. 2010, 45, 848–855. [Google Scholar] [CrossRef] [PubMed]
- Rippe, C.; Lesniewski, L.; Connell, M.; LaRocca, T.; Donato, A.; Seals, D. Short-term calorie restriction reverses vascular endothelial dysfunction in old mice by increasing nitric oxide and reducing oxidative stress. Aging Cell 2010, 9, 304–312. [Google Scholar] [CrossRef] [PubMed]
- Donato, A.J.; Walker, A.E.; Magerko, K.A.; Bramwell, R.C.; Black, A.D.; Henson, G.D.; Lawson, B.R.; Lesniewski, L.A.; Seals, D.R. Life-long caloric restriction reduces oxidative stress and preserves nitric oxide bioavailability and function in arteries of old mice. Aging Cell 2013, 12, 772–783. [Google Scholar] [CrossRef] [PubMed]
- Kobara, M.; Furumori-Yukiya, A.; Kitamura, M.; Matsumura, M.; Ohigashi, M.; Toba, H.; Nakata, T. Short-Term Caloric Restriction Suppresses Cardiac Oxidative Stress and Hypertrophy Caused by Chronic Pressure Overload. J. Card. Fail 2015, 21, 656–666. [Google Scholar] [CrossRef] [PubMed]
- Waldman, M.; Cohen, K.; Yadin, D.; Nudelman, V.; Gorfil, D.; Laniado-Schwartzman, M.; Kornwoski, R.; Aravot, D.; Abraham, N.G.; Arad, M.; et al. Regulation of diabetic cardiomyopathy by caloric restriction is mediated by intracellular signaling pathways involving ‘SIRT1 and PGC-1alpha’. Cardiovasc. Diabetol. 2018, 17, 111. [Google Scholar] [CrossRef]
- An, H.S.; Lee, J.Y.; Choi, E.B.; Jeong, E.A.; Shin, H.J.; Kim, K.E.; Park, K.A.; Jin, Z.; Lee, J.E.; Koh, J.S.; et al. Caloric restriction reverses left ventricular hypertrophy through the regulation of cardiac iron homeostasis in impaired leptin signaling mice. Sci. Rep. 2020, 10, 7176. [Google Scholar] [CrossRef]
- Wadden, T.A.; Considine, R.V.; Foster, G.D.; Anderson, D.A.; Sarwer, D.B.; Caro, J.S. Short- and long-term changes in serum leptin dieting obese women: Effects of caloric restriction and weight loss. J. Clin. Endocrinol. Metab. 1998, 83, 214–218. [Google Scholar] [CrossRef]
- Nakano, Y.; Oshima, T.; Sasaki, S.; Higashi, Y.; Ozono, R.; Takenaka, S.; Miura, F.; Hirao, H.; Matsuura, H.; Chayama, K.; et al. Calorie restriction reduced blood pressure in obesity hypertensives by improvement of autonomic nerve activity and insulin sensitivity. J. Cardiovasc. Pharmacol. 2001, 38, S69–S74. [Google Scholar] [CrossRef]
- Facchini, M.; Malfatto, G.; Sala, L.; Silvestri, G.; Fontana, P.; Lafortuna, C.; Sartorio, A. Changes of autonomic cardiac profile after a 3-week integrated body weight reduction program in severely obese patients. J. Endocrinol. Invest. 2003, 26, 138–142. [Google Scholar] [CrossRef]
- Das, S.K.; Gilhooly, C.H.; Golden, J.K.; Pittas, A.G.; Fuss, P.J.; Cheatham, R.A.; Tyler, S.; Tsay, M.; McCrory, M.A.; Lichtenstein, A.H.; et al. Long-term effects of 2 energy-restricted diets differing in glycemic load on dietary adherence, body composition, and metabolism in CALERIE: A 1-y randomized controlled trial. Am. J. Clin. Nutr. 2007, 85, 1023–1030. [Google Scholar] [CrossRef]
- Lecoultre, V.; Ravussin, E.; Redman, L.M. The fall in leptin concentration is a major determinant of the metabolic adaptation induced by caloric restriction independently of the changes in leptin circadian rhythms. J. Clin. Endocrinol. Metab. 2011, 96, E1512–E1516. [Google Scholar] [CrossRef] [PubMed]
- Stewart, T.M.; Bhapkar, M.; Das, S.; Galan, K.; Martin, C.K.; McAdams, L.; Pieper, C.; Redman, L.; Roberts, S.; Stein, R.I.; et al. Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy Phase 2 (CALERIE Phase 2) screening and recruitment: Methods and results. Contemp. Clin. Trials 2013, 34, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Ruggenenti, P.; Abbate, M.; Ruggiero, B.; Rota, S.; Trillini, M.; Aparicio, C.; Parvanova, A.; Petrov Iliev, I.; Pisanu, G.; Perna, A.; et al. Renal and Systemic Effects of Calorie Restriction in Patients With Type 2 Diabetes With Abdominal Obesity: A Randomized Controlled Trial. Diabetes 2017, 66, 75–86. [Google Scholar] [CrossRef] [PubMed]
- Most, J.; Gilmore, L.A.; Smith, S.R.; Han, H.; Ravussin, E.; Redman, L.M. Significant improvement in cardiometabolic health in healthy nonobese individuals during caloric restriction-induced weight loss and weight loss maintenance. Am. J. Physiol. Endocrinol. Metab. 2018, 314, E396–E405. [Google Scholar] [CrossRef]
- Kraus, W.E.; Bhapkar, M.; Huffman, K.M.; Pieper, C.F.; Krupa Das, S.; Redman, L.M.; Villareal, D.T.; Rochon, J.; Roberts, S.B.; Ravussin, E.; et al. 2 years of calorie restriction and cardiometabolic risk (CALERIE): Exploratory outcomes of a multicentre, phase 2, randomised controlled trial. Lancet Diabetes Endocrinol. 2019, 7, 673–683. [Google Scholar] [CrossRef]
- Harsha, D.W.; Lin, P.H.; Obarzanek, E.; Karanja, N.M.; Moore, T.J.; Caballero, B.; DASH Collaborative Research Group. Dietary Approaches to Stop Hypertension: A summary of study results. J. Am. Diet Assoc. 1999, 99, S35–S39. [Google Scholar] [CrossRef]
- Arnason, T.G.; Bowen, M.W.; Mansell, K.D. Effects of intermittent fasting on health markers in those with type 2 diabetes: A pilot study. World J. Diabetes 2017, 8, 154–164. [Google Scholar] [CrossRef]
- Erdem, Y.; Ozkan, G.; Ulusoy, S.; Arici, M.; Derici, U.; Sengul, S.; Sindel, S.; Erturk, S.; Turkish Society of, H.; Renal, D. The effect of intermittent fasting on blood pressure variability in patients with newly diagnosed hypertension or prehypertension. J. Am. Soc. Hypertens. 2018, 12, 42–49. [Google Scholar] [CrossRef]
- Furmli, S.; Elmasry, R.; Ramos, M.; Fung, J. Therapeutic use of intermittent fasting for people with type 2 diabetes as an alternative to insulin. BMJ Case Rep. 2018, 2018. [Google Scholar] [CrossRef]
- Wilhelmi de Toledo, F.; Grundler, F.; Bergouignan, A.; Drinda, S.; Michalsen, A. Safety, health improvement and well-being during a 4 to 21-day fasting period in an observational study including 1422 subjects. PLoS ONE 2019, 14, e0209353. [Google Scholar] [CrossRef]
- Mager, D.E.; Wan, R.; Brown, M.; Cheng, A.; Wareski, P.; Abernethy, D.R.; Mattson, M.P. Caloric restriction and intermittent fasting alter spectral measures of heart rate and blood pressure variability in rats. FASEB J. 2006, 20, 631–637. [Google Scholar] [CrossRef] [PubMed]
- Rickman, A.D.; Williamson, D.A.; Martin, C.K.; Gilhooly, C.H.; Stein, R.I.; Bales, C.W.; Roberts, S.; Das, S.K. The CALERIE Study: Design and methods of an innovative 25% caloric restriction intervention. Contemp. Clin. Trials 2011, 32, 874–881. [Google Scholar] [CrossRef] [PubMed]
- Di Renzo, L.; Gualtieri, P.; Romano, L.; Marrone, G.; Noce, A.; Pujia, A.; Perrone, M.A.; Aiello, V.; Colica, C.; de Lorenzo, A. Role of Personalized Nutrition in Chronic-Degenerative Diseases. Nutrients 2019, 11, 1707. [Google Scholar] [CrossRef] [PubMed]
- Di Daniele, N. The Role of Preventive Nutrition in Chronic Non-Communicable Diseases. Nutrients 2019, 11, 1074. [Google Scholar] [CrossRef] [PubMed]
- Bidani, A.K.; Griffin, K.A. Basic science: Hypertensive target organ damage. J. Am. Soc. Hypertens. 2015, 9, 235–237; quiz 238. [Google Scholar] [CrossRef] [PubMed]
- Mensah, G.A.; Croft, J.B.; Giles, W.H. The heart, kidney, and brain as target organs in hypertension. Cardiol. Clin. 2002, 20, 225–247. [Google Scholar] [CrossRef]
- Aronow, W.S. Hypertension and left ventricular hypertrophy. Ann. Transl. Med. 2017, 5, 310. [Google Scholar] [CrossRef]
- Gallu, M.; Marrone, G.; Legramante, J.M.; de Lorenzo, A.; Di Daniele, N.; Noce, A. Female Sex as a Thromboembolic Risk Factor in the Era of Nonvitamin K Antagonist Oral Anticoagulants. Cardiovasc. Ther. 2020, 2020, 1743927. [Google Scholar] [CrossRef]
- Landecho, M.F.; Tuero, C.; Valenti, V.; Bilbao, I.; de la Higuera, M.; Fruhbeck, G. Relevance of Leptin and Other Adipokines in Obesity-Associated Cardiovascular Risk. Nutrients 2019, 11, 2664. [Google Scholar] [CrossRef]
- Hall, M.E.; Harmancey, R.; Stec, D.E. Lean heart: Role of leptin in cardiac hypertrophy and metabolism. World J. Cardiol. 2015, 7, 511–524. [Google Scholar] [CrossRef]
- Raju, S.V.; Zheng, M.; Schuleri, K.H.; Phan, A.C.; Bedja, D.; Saraiva, R.M.; Yiginer, O.; Vandegaer, K.; Gabrielson, K.L.; O’Donnell, C.P.; et al. Activation of the cardiac ciliary neurotrophic factor receptor reverses left ventricular hypertrophy in leptin-deficient and leptin-resistant obesity. Proc. Natl. Acad. Sci. USA 2006, 103, 4222–4227. [Google Scholar] [CrossRef] [PubMed]
- Beltowski, J. Role of leptin in blood pressure regulation and arterial hypertension. J. Hypertens. 2006, 24, 789–801. [Google Scholar] [CrossRef] [PubMed]
- Bravo, P.E.; Morse, S.; Borne, D.M.; Aguilar, E.A.; Reisin, E. Leptin and hypertension in obesity. Vasc. Health Risk Manag. 2006, 2, 163–169. [Google Scholar] [CrossRef] [PubMed]
- Chiu, H.C.; Kovacs, A.; Ford, D.A.; Hsu, F.F.; Garcia, R.; Herrero, P.; Saffitz, J.E.; Schaffer, J.E. A novel mouse model of lipotoxic cardiomyopathy. J. Clin. Invest. 2001, 107, 813–822. [Google Scholar] [CrossRef]
- Kamimura, D.; Suzuki, T.; Wang, W.; deShazo, M.; Hall, J.E.; Winniford, M.D.; Kullo, I.J.; Mosley, T.H.; Butler, K.R.; Hall, M.E. Higher plasma leptin levels are associated with reduced left ventricular mass and left ventricular diastolic stiffness in black women: Insights from the Genetic Epidemiology Network of Arteriopathy (GENOA) study. Hypertens. Res. 2018, 41, 629–638. [Google Scholar] [CrossRef]
- Abel, E.D.; Litwin, S.E.; Sweeney, G. Cardiac remodeling in obesity. Physiol. Rev. 2008, 88, 389–419. [Google Scholar] [CrossRef]
- Maulik, S.K.; Kumar, S. Oxidative stress and cardiac hypertrophy: A review. Toxicol. Mech. Methods 2012, 22, 359–366. [Google Scholar] [CrossRef]
- Cianci, R.; Barbano, B.; Martina, P.; Gigante, A.; Polidori, L.; Lai, S.; Ascoli, G.; de Francesco, I.; di Donato, D.; Fuiano, G.; et al. Nephroangiosclerosis and its pharmacological approach. Curr. Vasc. Pharmacol. 2011, 9, 238–243. [Google Scholar] [CrossRef]
- Yamanouchi, M.; Hoshino, J.; Ubara, Y.; Takaichi, K.; Kinowaki, K.; Fujii, T.; Ohashi, K.; Mise, K.; Toyama, T.; Hara, A.; et al. Clinicopathological predictors for progression of chronic kidney disease in nephrosclerosis: A biopsy-based cohort study. Nephrol. Dial. Transplant. 2019, 34, 1182–1188. [Google Scholar] [CrossRef]
- O’Donnell, M.; Mente, A.; Yusuf, S. Sodium intake and cardiovascular health. Circ. Res. 2015, 116, 1046–1057. [Google Scholar] [CrossRef]
- Wade, B.; Petrova, G.; Mattson, D.L. Role of immune factors in angiotensin II-induced hypertension and renal damage in Dahl salt-sensitive rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018, 314, R323–R333. [Google Scholar] [CrossRef] [PubMed]
- Noce, A.; Marrone, G.; di Lauro, M.; Urciuoli, S.; Pietroboni Zaitseva, A.; Wilson Jones, G.; di Daniele, N.; Romani, A. Cardiovascular Protection of Nephropathic Male Patients by Oral Food Supplements. Cardiovasc. Ther. 2020, 2020, 1807941. [Google Scholar] [CrossRef]
- Mattson, D.L.; James, L.; Berdan, E.A.; Meister, C.J. Immune suppression attenuates hypertension and renal disease in the Dahl salt-sensitive rat. Hypertension 2006, 48, 149–156. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; Crowley, S.D. Inflammation in Salt-Sensitive Hypertension and Renal Damage. Curr. Hypertens. Rep. 2018, 20, 103. [Google Scholar] [CrossRef] [PubMed]
- Hildebrand, A.M.; Garg, A.X. Blood pressure targets in chronic kidney disease: Does proteinuria dictate how low we go? CMAJ 2013, 185, 941–942. [Google Scholar] [CrossRef][Green Version]
- Xu, X.M.; Cai, G.Y.; Bu, R.; Wang, W.J.; Bai, X.Y.; Sun, X.F.; Chen, X.M. Beneficial Effects of Caloric Restriction on Chronic Kidney Disease in Rodent Models: A Meta-Analysis and Systematic Review. PLoS ONE 2015, 10, e0144442. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.Y.; Cai, G.Y.; Chen, X.M. Energy restriction in renal protection. Br. J. Nutr. 2018, 120, 1149–1158. [Google Scholar] [CrossRef] [PubMed]
- Dong, D.; Cai, G.Y.; Ning, Y.C.; Wang, J.C.; Lv, Y.; Hong, Q.; Cui, S.Y.; Fu, B.; Guo, Y.N.; Chen, X.M. Alleviation of senescence and epithelial-mesenchymal transition in aging kidney by short-term caloric restriction and caloric restriction mimetics via modulation of AMPK/mTOR signaling. Oncotarget 2017, 8, 16109–16121. [Google Scholar] [CrossRef]
- D’Elia, L.; Rossi, G.; Schiano di Cola, M.; Savino, I.; Galletti, F.; Strazzullo, P. Meta-Analysis of the Effect of Dietary Sodium Restriction with or without Concomitant Renin-Angiotensin-Aldosterone System-Inhibiting Treatment on Albuminuria. Clin. J. Am. Soc. Nephrol. 2015, 10, 1542–1552. [Google Scholar] [CrossRef]
- Cheung, Y.F. Arterial stiffness in the young: Assessment, determinants, and implications. Korean Circ. J. 2010, 40, 153–162. [Google Scholar] [CrossRef]
- Schinzari, F.; Iantorno, M.; Campia, U.; Mores, N.; Rovella, V.; Tesauro, M.; di Daniele, N.; Cardillo, C. Vasodilator responses and endothelin-dependent vasoconstriction in metabolically healthy obesity and the metabolic syndrome. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E787–E792. [Google Scholar] [CrossRef] [PubMed]
- Noce, A.; Fabrini, R.; Dessi, M.; Bocedi, A.; Santini, S.; Rovella, V.; Pastore, A.; Tesauro, M.; Bernardini, S.; di Daniele, N.; et al. Erythrocyte glutathione transferase activity: A possible early biomarker for blood toxicity in uremic diabetic patients. Acta Diabetol. 2014, 51, 219–224. [Google Scholar] [CrossRef] [PubMed]
- Covic, A.; Gusbeth-Tatomir, P.; Goldsmith, D.J. Arterial stiffness in renal patients: An update. Am. J. Kidney Dis. 2005, 45, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z. Aging, arterial stiffness, and hypertension. Hypertension 2015, 65, 252–256. [Google Scholar] [CrossRef] [PubMed]
- Martens, C.R.; Seals, D.R. Practical alternatives to chronic caloric restriction for optimizing vascular function with ageing. J. Physiol. 2016, 594, 7177–7195. [Google Scholar] [CrossRef]
- D’Elia, L.; Galletti, F.; La Fata, E.; Sabino, P.; Strazzullo, P. Effect of dietary sodium restriction on arterial stiffness: Systematic review and meta-analysis of the randomized controlled trials. J. Hypertens. 2018, 36, 734–743. [Google Scholar] [CrossRef]
- Henderson, A.D.; Bruce, B.B.; Newman, N.J.; Biousse, V. Hypertension-related eye abnormalities and the risk of stroke. Rev. Neurol. Dis. 2011, 8, 1–9. [Google Scholar]
- Chatterjee, S.; Chattopadhyay, S.; Hope-Ross, M.; Lip, P.L. Hypertension and the eye: Changing perspectives. J. Hum. Hypertens. 2002, 16, 667–675. [Google Scholar] [CrossRef]
- Kawashima, M.; Ozawa, Y.; Shinmura, K.; Inaba, T.; Nakamura, S.; Kawakita, T.; Watanabe, M.; Tsubota, K. Calorie restriction (CR) and CR mimetics for the prevention and treatment of age-related eye disorders. Exp. Gerontol. 2013, 48, 1096–1100. [Google Scholar] [CrossRef]
- Katsi, V.; Marketou, M.; Vlachopoulos, C.; Tousoulis, D.; Souretis, G.; Papageorgiou, N.; Stefanadis, C.; Vardas, P.; Kallikazaros, I. Impact of arterial hypertension on the eye. Curr. Hypertens. Rep. 2012, 14, 581–590. [Google Scholar] [CrossRef]
- Viera, A.J.; Neutze, D.M. Diagnosis of secondary hypertension: An age-based approach. Am. Fam. Phys. 2010, 82, 1471–1478. [Google Scholar]
- Charles, L.; Triscott, J.; Dobbs, B. Secondary Hypertension: Discovering the Underlying Cause. Am. Fam. Phys. 2017, 96, 453–461. [Google Scholar]
- Calhoun, D.A.; Jones, D.; Textor, S.; Goff, D.C.; Murphy, T.P.; Toto, R.D.; White, A.; Cushman, W.C.; White, W.; Sica, D.; et al. Resistant hypertension: Diagnosis, evaluation, and treatment: A scientific statement from the American Heart Association Professional Education Committee of the Council for High Blood Pressure Research. Circulation 2008, 117, e510–e526. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.A.; Mittal, P.K.; Little, B.P.; Miller, F.H.; Akduman, E.I.; Ali, K.; Sartaj, S.; Moreno, C.C. Secondary Hypertension and Complications: Diagnosis and Role of Imaging. Radiographics 2019, 39, 1036–1055. [Google Scholar] [CrossRef] [PubMed]
- Acelajado, M.C.; Calhoun, D.A. Resistant hypertension, secondary hypertension, and hypertensive crises: Diagnostic evaluation and treatment. Cardiol. Clin. 2010, 28, 639–654. [Google Scholar] [CrossRef] [PubMed]
- Filippou, C.D.; Tsioufis, C.P.; Thomopoulos, C.G.; Mihas, C.C.; Dimitriadis, K.S.; Sotiropoulou, L.I.; Chrysochoou, C.A.; Nihoyannopoulos, P.I.; Tousoulis, D.M. Dietary Approaches to Stop Hypertension (DASH) Diet and Blood Pressure Reduction in Adults with and without Hypertension: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Adv. Nutr. 2020, 11, 1150–1160. [Google Scholar] [CrossRef]
- Troyer, J.L.; Racine, E.F.; Ngugi, G.W.; McAuley, W.J. The effect of home-delivered Dietary Approach to Stop Hypertension (DASH) meals on the diets of older adults with cardiovascular disease. Am. J. Clin. Nutr. 2010, 91, 1204–1212. [Google Scholar] [CrossRef]
- Fu, J.; Liu, Y.; Zhang, L.; Zhou, L.; Li, D.; Quan, H.; Zhu, L.; Hu, F.; Li, X.; Meng, S.; et al. Nonpharmacologic Interventions for Reducing Blood Pressure in Adults With Prehypertension to Established Hypertension. J. Am. Heart Assoc. 2020, 9, e016804. [Google Scholar] [CrossRef]
- The sixth report of the Joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure. Arch. Intern. Med. 1997, 157, 2413–2446. [CrossRef]
- D’Elia, L.; La Fata, E.; Giaquinto, A.; Strazzullo, P.; Galletti, F. Effect of dietary salt restriction on central blood pressure: A systematic review and meta-analysis of the intervention studies. J. Clin. Hypertens. 2020, 22, 814–825. [Google Scholar] [CrossRef]
- Malinowski, B.; Zalewska, K.; Wesierska, A.; Sokolowska, M.M.; Socha, M.; Liczner, G.; Pawlak-Osinska, K.; Wicinski, M. Intermittent Fasting in Cardiovascular Disorders-An Overview. Nutrients 2019, 11, 673. [Google Scholar] [CrossRef]
- Barnosky, A.R.; Hoddy, K.K.; Unterman, T.G.; Varady, K.A. Intermittent fasting vs daily calorie restriction for type 2 diabetes prevention: A review of human findings. Transl. Res. 2014, 164, 302–311. [Google Scholar] [CrossRef] [PubMed]
- Johnstone, A. Fasting for weight loss: An effective strategy or latest dieting trend? Int. J. Obes. 2015, 39, 727–733. [Google Scholar] [CrossRef] [PubMed]
- Harvie, M.; Howell, A. Potential Benefits and Harms of Intermittent Energy Restriction and Intermittent Fasting Amongst Obese, Overweight and Normal Weight Subjects-A Narrative Review of Human and Animal Evidence. Behav. Sci. 2017, 7, 4. [Google Scholar] [CrossRef] [PubMed]
- Aly, S.M. Role of intermittent fasting on improving health and reducing diseases. Int. J. Health Sci. 2014, 8, V–VI. [Google Scholar] [CrossRef]
- Patterson, R.E.; Sears, D.D. Metabolic Effects of Intermittent Fasting. Annu. Rev. Nutr. 2017, 37, 371–393. [Google Scholar] [CrossRef] [PubMed]
- Eckel-Mahan, K.L.; Patel, V.R.; de Mateo, S.; Orozco-Solis, R.; Ceglia, N.J.; Sahar, S.; Dilag-Penilla, S.A.; Dyar, K.A.; Baldi, P.; Sassone-Corsi, P. Reprogramming of the circadian clock by nutritional challenge. Cell 2013, 155, 1464–1478. [Google Scholar] [CrossRef] [PubMed]
- Turnbaugh, P.J.; Ley, R.E.; Mahowald, M.A.; Magrini, V.; Mardis, E.R.; Gordon, J.I. An obesity-associated gut microbiome with increased capacity for energy harvest. Nature 2006, 444, 1027–1031. [Google Scholar] [CrossRef] [PubMed]
- Noce, A.; Marrone, G.; Di Daniele, F.; Ottaviani, E.; Wilson Jones, G.; Bernini, R.; Romani, A.; Rovella, V. Impact of Gut Microbiota Composition on Onset and Progression of Chronic Non-Communicable Diseases. Nutrients 2019, 11, 1073. [Google Scholar] [CrossRef]
- Noce, A.; Tarantino, A.; Tsague Djoutsop, C.; Erald, V.; de Lorenzo, A.; di Daniele, N. Gut Microbioma Population: An Indicator Really Sensible to Any Change in Age, Diet, Metabolic Syndrome, and Life-Style. Mediat. Inflam. 2014, 2014. [Google Scholar] [CrossRef]
- Chowdhury, E.A.; Richardson, J.D.; Tsintzas, K.; Thompson, D.; Betts, J.A. Effect of extended morning fasting upon ad libitum lunch intake and associated metabolic and hormonal responses in obese adults. Int. J. Obes. 2016, 40, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Hatori, M.; Vollmers, C.; Zarrinpar, A.; DiTacchio, L.; Bushong, E.A.; Gill, S.; Leblanc, M.; Chaix, A.; Joens, M.; Fitzpatrick, J.A.; et al. Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice fed a high-fat diet. Cell Metab. 2012, 15, 848–860. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Uemura, H.; Katsuura-Kamano, S.; Nakamoto, M.; Hiyoshi, M.; Takami, H.; Sawachika, F.; Juta, T.; Arisawa, K. Relationship of dietary factors and habits with sleep-wake regularity. Asia Pac. J. Clin. Nutr. 2013, 22, 457–465. [Google Scholar] [CrossRef] [PubMed]
- Camandola, S.; Mattson, M.P. Brain metabolism in health, aging, and neurodegeneration. EMBO J. 2017, 36, 1474–1492. [Google Scholar] [CrossRef]
- Mattson, M.P.; Longo, V.D.; Harvie, M. Impact of intermittent fasting on health and disease processes. Ageing Res. Rev. 2017, 39, 46–58. [Google Scholar] [CrossRef]
| Nutritional Treatment | Type of the Study | Authors | Year | Organ/System/Metabolic Target | Duration of Intervention | Impact of Intervention |
|---|---|---|---|---|---|---|
| CRD | Animal study | Zanetti et al. [64] | 2010 |
| 3 weeks | ↓ Oxidative stress ↓ iNOS ↓ Total nitrite ↓ Calcium-independent NOS activity ↑ SOD activity |
| CRD | Animal study | Rippe et al. [65] | 2010 |
| 8 weeks | ↓ Oxidative stress ↓ Blood glucose ↓ TG ↑ NO bioavailability ↑ Arterial expression of SIRT1 |
| CRD | Animal study | Donato et al. [66] | 2013 |
| 30–31 months | ↓ NADPH oxidase activity ↓ Oxidative stress ↓ TG ↓ BW ↑ SOD activity ↑ Catalase |
| CRD | Animal study | Kobara et al. [67] | 2015 |
| 4 weeks | ↓ ROS ↓ Cardiac hypertrophy and fibrosis |
| CRD | Animal study | Waldman et al. [68] | 2018 |
| 4 weeks | ↓ Oxidative stress ↓ Inflammation ↑ SIRT1 |
| CRD | Animal study | An et al. [69] | 2020 |
| 12 weeks | ↓ Cardiac hypertrophy and fibrosis ↓ Cardiac inflammation ⇅ Cellular regulation of iron homeostasis |
| CRD | Human study | Wadden et al. [70] | 1998 |
| 40 weeks | ↓ BW ↓ FM ↓ Serum leptin |
| CRD | Human study | Nakano et al. [71] | 2001 |
| 2 weeks | ↓ BMI ↓ BW ↓ TG ↓ HOMA-index ↓ SBP ↓ DBP |
| CRD | Human study | Facchini et al. [72] | 2003 |
| 3 weeks | ↓ BMI ↓ Heart rate ↑ Parasympathetic activity |
| CRD | Human study | Das et al. [73] | 2007 |
| 12 months | ↓ FM ↓ BW ↓ TG ↓ insulin ↓ LDL-c ↓ TC ↑ HDL-c |
| CRD | Human study | Lecoultre et al. [74] | 2011 |
| 6 months | ↓ BW ↓ Mean 24 h circulating leptin ↓ Urinary norepinephrine |
| CRD | Human study | Stewart et al. [75] | 2013 |
| 24 months | ↓ LDL-c ↓ TC/HDL-c↓ TG ↓ DBP ↓ BW ↓ BMI ↓ FM ↓ MSS ↑ IS |
| CRD | Human study | Ruggenenti et al. [76] | 2017 |
| 6 months | ↓ WC ↓ BW ↓ BMI ↓ BG ↓ HbA1c |
| CRD | Human study | Most et al. [77] | 2018 |
| 24 months | ↓ VAT ↓ SAAT ↓ SBP ↓ DBP ↓ TC ↓ LDL-c ↓ IR |
| CRD | Human study | Kraus et al. [78] | 2019 |
| 24 months | ↓ LDL-c ↓ TC/HDL-c ↓ SBP ↓ DBP ↓ MSS ↑ IS |
| DASH DIET | Human study | Harsha et al. [79] | 1999 |
| 8 weeks | ↓ SBP ↓ DBP |
| IF | Human study | Arnason et al. [80] | 2017 |
| 6 weeks | ↓ BW ↓ BMI |
| IF | Human study | Erdem et al. [81] | 2018 |
| - | ↓ SBP ↓ Heart rate ↓ USE |
| IF | Human study | Furmli et al. [82] | 2018 |
| 12 weeks | ↓ HbA1c ↓ BW ↓ WC |
| IF | Human study | Wilhelmi de Toledo et al. [83] | 2019 |
| 24 months | ↓ SBP ↓ DBP ↓ BW ↓ Abdominal circumference ↓ Blood glucose ↓ TG ↓ LDL-c ↓ HDL-c ↓ TC ↑ Physical and emotional well-being |
| CRD vs IF | Animal study | Magger et al. [84] | 2006 |
| 16 weeks | ↓ BW ↓ Heart rate ↓ SBP ↓ DBP ↓ Blood glucose |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Daniele, N.; Marrone, G.; Di Lauro, M.; Di Daniele, F.; Palazzetti, D.; Guerriero, C.; Noce, A. Effects of Caloric Restriction Diet on Arterial Hypertension and Endothelial Dysfunction. Nutrients 2021, 13, 274. https://doi.org/10.3390/nu13010274
Di Daniele N, Marrone G, Di Lauro M, Di Daniele F, Palazzetti D, Guerriero C, Noce A. Effects of Caloric Restriction Diet on Arterial Hypertension and Endothelial Dysfunction. Nutrients. 2021; 13(1):274. https://doi.org/10.3390/nu13010274
Chicago/Turabian StyleDi Daniele, Nicola, Giulia Marrone, Manuela Di Lauro, Francesca Di Daniele, Daniela Palazzetti, Cristina Guerriero, and Annalisa Noce. 2021. "Effects of Caloric Restriction Diet on Arterial Hypertension and Endothelial Dysfunction" Nutrients 13, no. 1: 274. https://doi.org/10.3390/nu13010274
APA StyleDi Daniele, N., Marrone, G., Di Lauro, M., Di Daniele, F., Palazzetti, D., Guerriero, C., & Noce, A. (2021). Effects of Caloric Restriction Diet on Arterial Hypertension and Endothelial Dysfunction. Nutrients, 13(1), 274. https://doi.org/10.3390/nu13010274





