Wholegrain Consumption and Risk Factors for Cardiorenal Metabolic Diseases in Chile: A Cross-Sectional Analysis of 2016–2017 Health National Survey
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Sample
2.2. Dietary Exposure
2.3. Assessment of Risk Factors for Cardiorenal Metabolic Disease
2.4. Outcomes
2.5. Socio-Demographic and Clinical Covariates
2.6. Statistical Analyses
3. Results
3.1. General Characteristics of the Population According to the Frequency of WG Consumption
3.2. Risk Factors for CRMD According to the Frequency of WG Consumption
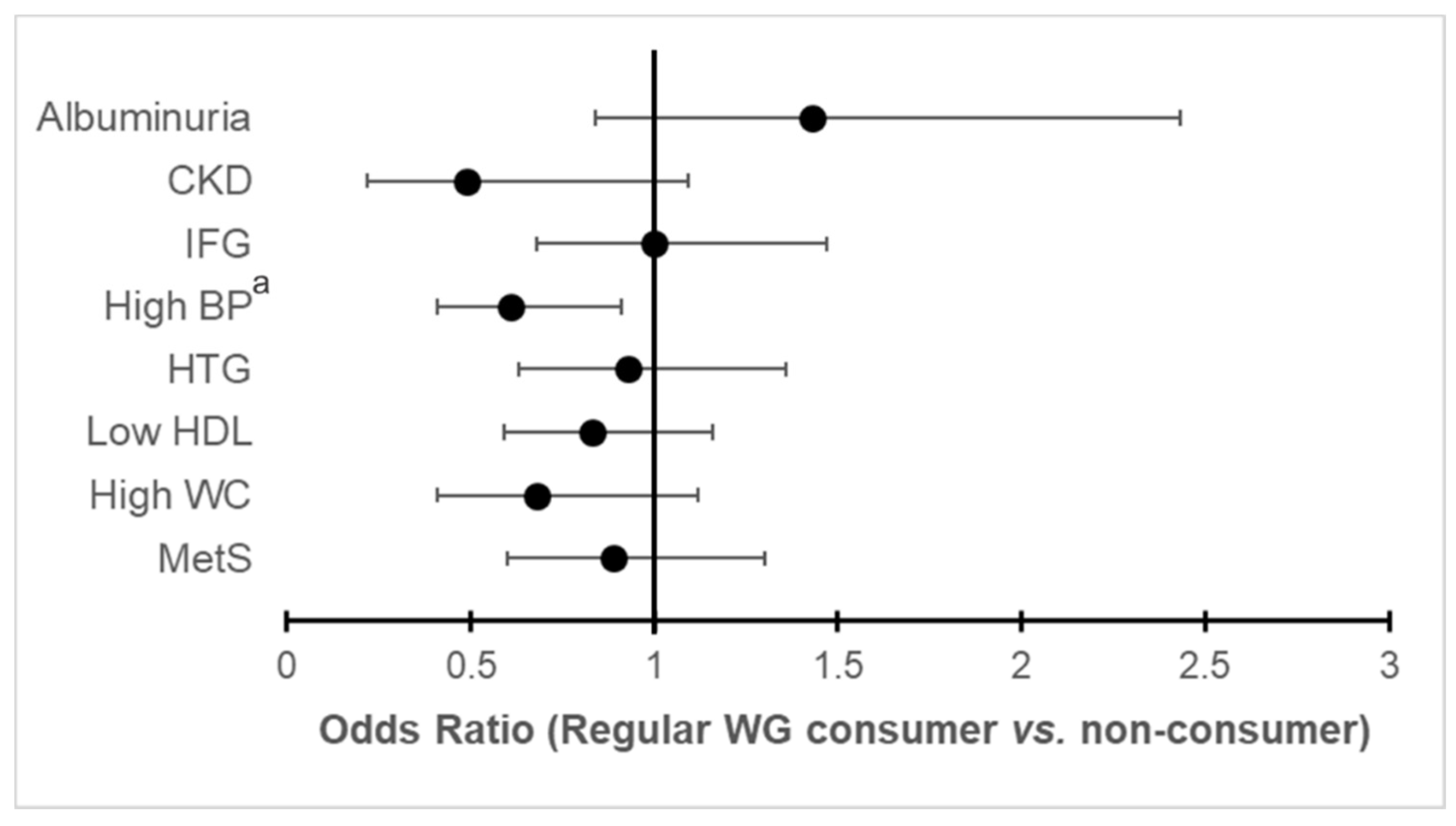
3.3. Association of WG Consumption with Cardiorenal Metabolic Outcomes
3.4. Sensitivity Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BP | Blood pressure |
| CKD | Chronic kidney disease |
| CRMD | Cardiorenal metabolic diseases |
| ENCA | Encuesta Nacional de Consumo Alimentario |
| ELANS | Latin American Study of Nutrition and Health |
| GFR | Glomerular filtration rate |
| HbA1c | Hemoglobin A1c |
| HDL | High-density lipoprotein |
| IFG | Impaired fasting glucose |
| LDL | Low-density lipoprotein |
| VLDL | Very low-density lipoprotein |
| WG | Wholegrains |
References
- Roth, G.A.; Abate, D.; Abate, K.H.; Abay, S.M.; Abbafati, C.; Abbasi, N.; Abbastabar, H.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet 2018, 392, 1736–1788. [Google Scholar] [CrossRef]
- Devaux, M.; Lerouge, A.; Giuffre, G.; Giesecke, S.; Baiocco, S.; Ricci, A.; Reyes, F.; Cantarero, D.; Ventelou, B.; Cecchini, M. How will the main risk factors contribute to the burden of non-communicable diseases under different scenarios by 2050? A modelling study. PLoS ONE 2020, 15, e231725. [Google Scholar] [CrossRef]
- Reynolds, A.; Mann, J.I.; Cummings, J.; Winter, N.; Mete, E.; Morenga, L.T. Carbohydrate quality and human health: A series of systematic reviews and meta-analyses. Lancet 2019, 393, 434–445. [Google Scholar] [CrossRef]
- Kovalskys, I.; Fisberg, M.; Gomez, G.; Pareja, R.G.; García, M.C.Y.; Sanabria, L.Y.C.; Herrera-Cuenca, M.; Rigotti, A.; Guajardo, V.; Zimberg, I.; et al. Energy intake and food sources of eight Latin American countries: Results from the Latin American Study of Nutrition and Health (ELANS). Public Health Nutr. 2018, 21, 2535–2547. [Google Scholar] [CrossRef] [PubMed]
- Kovalskys, I.; Rigotti, A.; Koletzko, B.; Fisberg, M.; Gómez, G.; Herrera-Cuenca, M.; Sanabria, L.Y.C.; García, M.C.Y.; Pareja, R.G.; Zimberg, I.; et al. Latin American consumption of major food groups: Results from the ELANS study. PLoS ONE 2019, 14, e225101. [Google Scholar] [CrossRef]
- Ministerio de Salud, Gobierno de Chile. Encuesta Nacional de Salud 2009–2010. 2011. Available online: http://epi.minsal.cl/wp-content/uploads/2016/05/presentacio%C3%8C_nENS2010final-20-de-enero.pdf (accessed on 21 May 2020).
- Olivares, C.S.; Zacarías, H.I.; González, G.C.G.; Villalobos, V.E. Proceso de formulación y validación de las guías alimentarias para la población chilena. Rev. Chil. Nutr. 2013, 40, 262–268. [Google Scholar] [CrossRef]
- Ministerio de Salud, Gobierno de Chile. Encuesta Nacional de Consumo Alimentario en Chile (ENCA). 2010. Available online: https://www.minsal.cl/enca/ (accessed on 15 May 2020).
- Fernández-Verdejo, R.; Moya-Osorio, J.L.; Fuentes-López, E.; Galgani, J.E. Metabolic health and its association with lifestyle habits according to nutritional status in Chile: A cross-sectional study from the National Health Survey 2016–2017. PLoS ONE 2020, 15, e236451. [Google Scholar] [CrossRef]
- Ministerio de Salud, Gobierno de Chile. Encuesta Nacional de Salud 2016–2017. 2018. Available online: http://epi.minsal.cl/encuesta-nacional-de-salud-2015-2016/ (accessed on 21 May 2020).
- Levey, A.S.; Stevens, L.A.; Schmid, C.H.; Zhang, Y.; Castro, A.F.; Feldman, H.I.; Kusek, J.W.; Eggers, P.; Van Lente, F.; Greene, T.; et al. A New Equation to Estimate Glomerular Filtration Rate. Ann. Intern. Med. 2009, 150, 604–612. [Google Scholar] [CrossRef]
- Stevens, P.E.; Levin, A. Evaluation and Management of Chronic Kidney Disease: Synopsis of the Kidney Disease: Improving Global Outcomes 2012 Clinical Practice Guideline. Ann. Intern. Med. 2013, 158, 825–830. [Google Scholar] [CrossRef]
- Chen, J.; Chen, G.; Wang, X.P.; Qin, L.Q.; Bai, Y. Dietary Fiber and Metabolic Syndrome: A Meta-Analysis and Review of Related Mechanisms. Nutrients 2018, 10, 24. [Google Scholar] [CrossRef]
- Esmaillzadeh, A.; Mirmiran, P.; Azizi, F. Whole-grain consumption and the metabolic syndrome: A favorable association in Tehranian adults. Eur. J. Clin. Nutr. 2005, 59, 353–362. [Google Scholar] [CrossRef] [PubMed]
- Ettehad, D.; Emdin, C.A.; Kiran, A.; Anderson, S.G.; Callender, T.; Emberson, J.R.; Chalmers, J.P.; Rodgers, A.; Rahimi, K. Blood pressure lowering for prevention of cardiovascular disease and death: A systematic review and meta-analysis. Lancet Lond. Engl. 2016, 387, 957–967. [Google Scholar] [CrossRef]
- Vasan, R.S.; Larson, M.G.; Leip, E.P.; Evans, J.C.; O’Donnell, C.J.; Kannel, W.B.; Levy, D. Impact of High-Normal Blood Pressure on the Risk of Cardiovascular Disease. N. Eng. J. Med. 2001, 345, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Karmali, K.N.; Lloyd-Jones, D.M. Global Risk Assessment to Guide Blood Pressure Management in Cardiovascular Disease Prevention. Hypertension 2017, 69, e2–e9. [Google Scholar] [CrossRef]
- Schwingshackl, L.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Iqbal, K.; Andriolo, V.; Bechthold, A.; Schlesinger, S.; Boeing, H. Food Groups and Risk of Hypertension: A Systematic Review and Dose-Response Meta-Analysis of Prospective Studies. Adv. Nutr. 2017, 8, 793–803. [Google Scholar] [CrossRef] [PubMed]
- Kashino, I.; Eguchi, M.; Miki, T.; Kochi, T.; Nanri, A.; Kabe, I.; Mizoue, T. Prospective Association between Whole Grain Consumption and Hypertension: The Furukawa Nutrition and Health Study. Nutrients 2020, 12, 902. [Google Scholar] [CrossRef]
- Hajihashemi, P.; Haghighatdoost, F. Effects of Whole-Grain Consumption on Selected Biomarkers of Systematic Inflammation: A Systematic Review and Meta-analysis of Randomized Controlled Trials. J. Am. Coll. Nutr. 2019, 38, 275–285. [Google Scholar] [CrossRef]
- Kopf, J.C.; Suhr, M.J.; Clarke, J.; Eyun, S.I.; Riethoven, J.J.M.; Ramer-Tait, A.E.; Rose, D.J. Role of whole grains versus fruits and vegetables in reducing subclinical inflammation and promoting gastrointestinal health in individuals affected by overweight and obesity: A randomized controlled trial. Nutr. J. 2018, 17, 72. [Google Scholar] [CrossRef]
- Knudsen, K.E.B. Microbial degradation of whole-grain complex carbohydrates and impact on short-chain fatty acids and health. Adv. Nutr. 2015, 6, 206–213. [Google Scholar] [CrossRef]
- Martínez, I.; Lattimer, J.M.; Hubach, K.L.; Case, A.J.; Yang, J.; Weber, C.G.; Louk, A.J.; Rose, D.J.; Kyureghian, G.; Peterson, A.D.; et al. Gut microbiome composition is linked to whole grain-induced immunological improvements. ISME J. 2013, 7, 269–280. [Google Scholar] [CrossRef] [PubMed]
- Oishi, Y.; Manabe, I. Organ System Crosstalk in Cardiometabolic Disease in the Age of Multimorbidity. Front. Cardiovasc. Med. 2020, 7. [Google Scholar] [CrossRef] [PubMed]
- Grooms, K.N.; Ommerborn, M.J.; Pham, D.Q.; Djoussé, L.; Clark, C.R. Dietary Fiber Intake and Cardiometabolic Risks among US Adults, NHANES 1999–2010. Am. J. Med. 2013, 126, 1059–1067. [Google Scholar] [CrossRef]
- Micha, R.; Shulkin, M.L.; Peñalvo, J.L.; Khatibzadeh, S.; Singh, G.M.; Rao, M.; Fahimi, S.; Powles, J.; Mozaffarian, D. Etiologic effects and optimal intakes of foods and nutrients for risk of cardiovascular diseases and diabetes: Systematic reviews and meta-analyses from the Nutrition and Chronic Diseases Expert Group (NutriCoDE). PLoS ONE 2017, 12, e175149. [Google Scholar] [CrossRef]
- Bechthold, A.; Boeing, H.; Schwedhelm, C.; Hoffmann, G.; Knüppel, S.; Iqbal, K.; De Henauw, S.; Michels, N.; Devleesschauwer, B.; Schlesinger, S.; et al. Food groups and risk of coronary heart disease, stroke and heart failure: A systematic review and dose-response meta-analysis of prospective studies. Crit. Rev. Food Sci. Nutr. 2019, 59, 1071–1090. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Whole grain consumption and risk of cardiovascular disease, cancer, and all cause and cause specific mortality: Systematic review and dose-response meta-analysis of prospective studies. BMJ 2016, 353, 1–14. [Google Scholar] [CrossRef]
- Johnsen, N.F.; Frederiksen, K.; Christensen, J.; Skeie, G.; Lund, E.; Landberg, R.; Johansson, I.; Nilsson, L.M.; Halkjær, J.; Olsen, A.; et al. Whole-grain products and whole-grain types are associated with lower all-cause and cause-specific mortality in the Scandinavian HELGA cohort. Br. J. Nutr. 2015, 114, 608–623. [Google Scholar] [CrossRef]
- Wu, H.; Flint, A.J.; Qi, Q.; Van Dam, R.M.; Sampson, L.A.; Rimm, E.B.; Holmes, M.D.; Willett, W.C.; Hu, F.B.; Sun, Q. Association Between Dietary Whole Grain Intake and Risk of Mortality: Two large prospective studies in US Men and Women. JAMA Intern. Med. 2015, 175, 373–384. [Google Scholar] [CrossRef] [PubMed]
- Pinto, V.; Landaeta-Díaz, L.; Castillo, O.; Villarroel, L.; Rigotti, A.; Echeverría, G. Assessment of Diet Quality in Chilean Urban Population through the Alternate Healthy Eating Index 2010: A Cross-Sectional Study. Nutrients 2019, 11, 891. [Google Scholar] [CrossRef]
- Hui, S.; Liu, K.; Lang, H.; Liu, Y.; Wang, X.; Zhu, X.; Doucette, S.; Yi, L.; Mi, M.T. Comparative effects of different whole grains and brans on blood lipid: A network meta-analysis. Eur. J. Nutr. 2019, 58, 2779–2787. [Google Scholar] [CrossRef]
- Carlos, Z.S.; Hans, M.O.; Maritza, F.O. Prevalencia de enfermedad renal crónica en centros urbanos de atención primaria. Rev. Med. Chil. 2011, 139, 1176–1184. [Google Scholar] [CrossRef][Green Version]
- World Health Organization (WHO). Global Health Observatory Data Repository (Region of the Americas). Available online: https://www.who.int/data/gho/data/indicators/indic (accessed on 21 May 2020).
- Barrett, E.M.; Batterham, M.J.; Ray, S.; Beck, E.J. Whole grain, bran and cereal fibre consumption and CVD: A systematic review. Br. J. Nutr. 2019, 8, 914–937. [Google Scholar] [CrossRef]
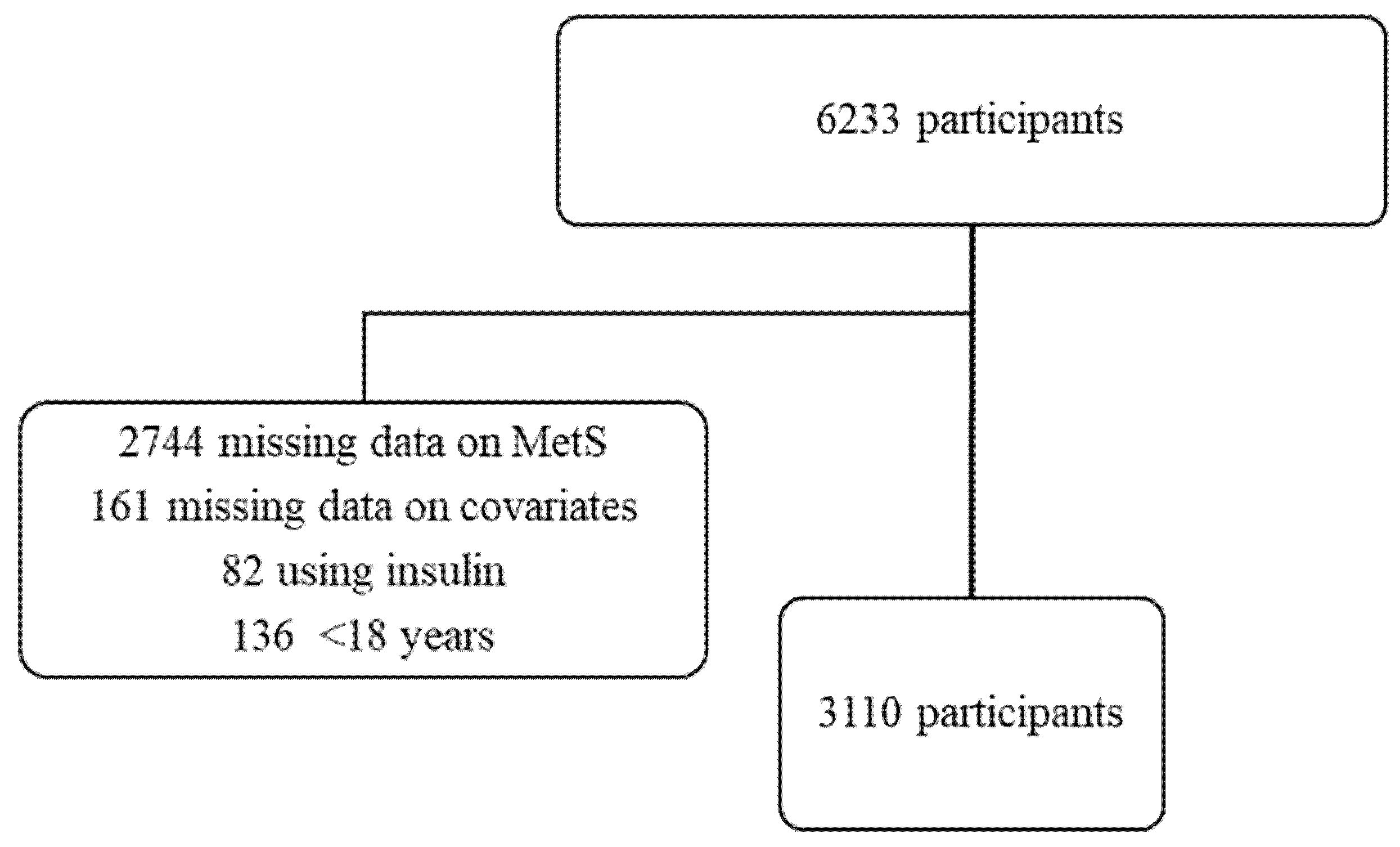
| All | Non–Consumers | Sporadic WG Consumers | Regular WG Consumers | p for Trend * | |
|---|---|---|---|---|---|
| Interviewed participants (n) | 3110 | 1739 | 613 | 758 | |
| Weighted population (n,%) | 11,810,647 (100) | 6,369,362 (54) | 2,316,451 (20) | 3,124,834 (26) | |
| Age (years) | 44.2 (0.5) | 47.1 (0.8) | 42.2 (1.1) | 40.0 (0.9) | <0.001 |
| Urban residents (%) | 88.9 (0.7) | 86.0 (1.1) | 90.1 (1.5) | 94.1 (1.1) | <0.001 |
| Female sex (%) | 51.5 (1.4) | 48.3 (1.8) | 54.9 (3.3) | 55.5 (3.5) | 0.025 |
| Education level (%) | <0.001 | ||||
| <8 years | 17.1 (1.1) | 22.6 (1.7) | 10.3 (1.5) | 11.0 (2.0) | |
| 8–12 years | 54.5 (1.9) | 58.9 (2.3) | 54.8 (3.3) | 45.4 (3.4) | |
| >12 years | 28.4 (1.8) | 18.5 (1.9) | 35.0 (3.4) | 43.6 (3.6) | |
| BMI category (%) | 0.320 | ||||
| <25 kg/m2 | 22.5 (1.4) | 21.2 (1.7) | 19.8 (2.7) | 27.0 (3.0) | |
| 25–30 kg/m2 | 42.7 (1.8) | 42.5 (2.3) | 46.2 (3.5) | 40.5 (3.2) | |
| >30 kg/m2 | 34.8 (1.5) | 36.3 (2.0) | 33.9 (3.1) | 32.5 (3.1) | |
| PA (%) | 0.044 | ||||
| Low | 34.7 (1.5) | 37.3 (2.1) | 31.4 (3.2) | 32.0 (3.0) | |
| Moderate | 23.8 (1.4) | 24.5 (1.8) | 23.8 (2.8) | 22.2 (2.8) | |
| High | 41.5 (1.8) | 38.2 (2.3) | 44.8 (3.5) | 45.7 (3.4) | |
| Tobacco use (%) | 36.1 (1.5) | 38.3 (2.0) | 39.6 (3.3) | 28.9 (3.2) | 0.002 |
| Frequency of alcohol | 0.125 | ||||
| consumption (%) | |||||
| Never | 27.1 (1.3) | 25.4 (1.5) | 25.4 (2.7) | 31.6 (3.0) | |
| ≤1 time/month | 37.6 (1.5) | 39.0 (2.0) | 34.1 (3.2) | 37.1 (3.0) | |
| 2–4 times/month | 24.8 (1.4) | 25.2 (2.0) | 29.6 (3.4) | 20.4 (2.3) | |
| 2–3 times/week | 7.9 (1.1) | 7.5 (1.4) | 9.1 (2.2) | 8.0 (2.0) | |
| >3 times/week | 2.6 (0.5) | 2.9 (0.7) | 1.8 (0.8) | 2.8 (1.1) | |
| Alcohol consumption (glasses) | 2.2 (0.1) | 2.4 (0.1) | 2.2 (0.1) | 1.9 (0.1) | <0.001 |
| Fruits (times/week) | 4.3 (0.1) | 4..0 (0.1) | 4.4 (0.1) | 5.1 (0.2) | <0.001 |
| Vegetables (times/week) | 5.5 (0.1) | 5.4 (0.1) | 5.6 (0.1) | 5.8 (0.1) | 0.008 |
| Legumes (%) | 0.503 | ||||
| ≤1 time/month | 12.1 (1.1) | 11.7 (1.3) | 12.4 (2.4) | 12.5 (2.4) | |
| >1 time/month | 62.8 (1.8) | 62.8 (2.3) | 65.9 (3.3) | 60.7 (3.1) | |
| ≥2 times/week | 25.1 (1.4) | 25.5 (1.8) | 21.7 (2.8) | 26.8 (2.9) | |
| HT (%) | 27.1 (1.3) | 31.5 (1.9) | 27.9 (3.1) | 17.7 (2.1) | 0.164 |
| Diabetes (%) | 12.8 (1.0) | 13.0 (1.4) | 12.4 (1.8) | 12.6 (2.1) | 0.368 |
| CVD (%) | 9.6 (0.9) | 11.0 (1.3) | 9.4 (1.8) | 6.8 (1.2) | 0.396 |
| MetS (%) | 41.7 (1.6) | 45.2 (2.1) | 42.3 (3.3) | 34.1 (3.3) | 0.323 |
| CKD (%) | 2.9 (0.5) | 3.6 (0.7) | 3.5 (1.1) | 1.1 (0.3) | 0.181 |
| uACR (%) | 0.962 | ||||
| 0.34–3.39 mg/mmol | 8.3 (0.8) | 7.4 (0.9) | 10.1 (2.0) | 8.7 (2.1) | |
| >3.39 mg/mmol | 1.4 (0.3) | 1.7 (0.5) | 1.5 (0.7) | 0.9 (0.4) |
| Non–Consumers (n: 6,369,362) † | Sporadic WG Consumers (n: 2,316,451) † | Regular WG Consumers (n: 3,124,834) † | p for Trend * | |
|---|---|---|---|---|
| WC (cm) | 94.5 (0.5) | 93.6 (0.9) | 91.7 (0.8) | 0.156 |
| SBP (mmHg) | 126.1 (0.8) | 124.1 (1.4) | 118.7 (0.9) | 0.006 |
| DBP (mmHg) | 75.6 (0.4) | 74.9 (0.7) | 72.2 (0.6) | 0.007 |
| Glucose (mmol/L) | 5.36 (0.04) | 5.31 (0.07) | 5.11 (0.05) | 0.389 |
| HbA1c (mmol/mol) | 43.2 (0.8) | 45.9 (1.8) | 41.6 (1.2) | 0.993 |
| TG (mmol/L) | 1.72 (0.05) | 1.60 (0.09) | 1.48 (0.07) | 0.007 |
| TC (mmol/L) | 4.67 (0.04) | 4.66 (0.07) | 4.50 (0.06) | 0.319 |
| HDL-C (mmol/L) | 1.18 (0.01) | 1.24 (0.02) | 1.25 (0.02) | 0.005 |
| LDL-C (mmol/L) | 2.70 (0.03) | 2.69 (0.06) | 2.59 (0.05) | 0.459 |
| VLDL-C (mmol/L) | 0.78 (0.02) | 0.71 (0.04) | 0.66 (0.25) | 0.008 |
| Non-HDL-C (mmol/L) | 3.50 (0.04) | 3.42 (0.07) | 3.25 (0.06) | 0.071 |
| TG/HDL-C | 1.7 (0.07) | 1.5 (0.1) | 1.4 (0.1) | 0.003 |
| Creatinine (μmol/L) | 70.7 (0.8) | 69.5 (1.1) | 68.4 (1.1) | 0.251 |
| GFR (mL/min.1.73 m2) | 101 (1) | 104 (1) | 106 (1) | 0.343 |
| uACR (mg/mmol) | 0.28 (0.05) | 0.26 (0.04) | 0.21 (0.04) | 0.990 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lanuza, F.; Zamora-Ros, R.; Hidalgo-Liberona, N.; Andrés-Lacueva, C.; Meroño, T. Wholegrain Consumption and Risk Factors for Cardiorenal Metabolic Diseases in Chile: A Cross-Sectional Analysis of 2016–2017 Health National Survey. Nutrients 2020, 12, 2815. https://doi.org/10.3390/nu12092815
Lanuza F, Zamora-Ros R, Hidalgo-Liberona N, Andrés-Lacueva C, Meroño T. Wholegrain Consumption and Risk Factors for Cardiorenal Metabolic Diseases in Chile: A Cross-Sectional Analysis of 2016–2017 Health National Survey. Nutrients. 2020; 12(9):2815. https://doi.org/10.3390/nu12092815
Chicago/Turabian StyleLanuza, Fabian, Raul Zamora-Ros, Nicole Hidalgo-Liberona, Cristina Andrés-Lacueva, and Tomás Meroño. 2020. "Wholegrain Consumption and Risk Factors for Cardiorenal Metabolic Diseases in Chile: A Cross-Sectional Analysis of 2016–2017 Health National Survey" Nutrients 12, no. 9: 2815. https://doi.org/10.3390/nu12092815
APA StyleLanuza, F., Zamora-Ros, R., Hidalgo-Liberona, N., Andrés-Lacueva, C., & Meroño, T. (2020). Wholegrain Consumption and Risk Factors for Cardiorenal Metabolic Diseases in Chile: A Cross-Sectional Analysis of 2016–2017 Health National Survey. Nutrients, 12(9), 2815. https://doi.org/10.3390/nu12092815






