Compositional and Functional Adaptations of Intestinal Microbiota and Related Metabolites in CKD Patients Receiving Dietary Protein Restriction
Abstract
1. Introduction
2. Materials and Methods
2.1. Subjects and Settings
2.2. Low Protein Diet and Compliance
2.3. Targeted Metabolomics Profiling of Gut-Producing Metabolites
2.4. Fecal 16S rRNA Gene Sequencing and Functional Prediction of Bacterial Gene
2.5. Statistics Analysis
3. Results
3.1. Subject Characteristics
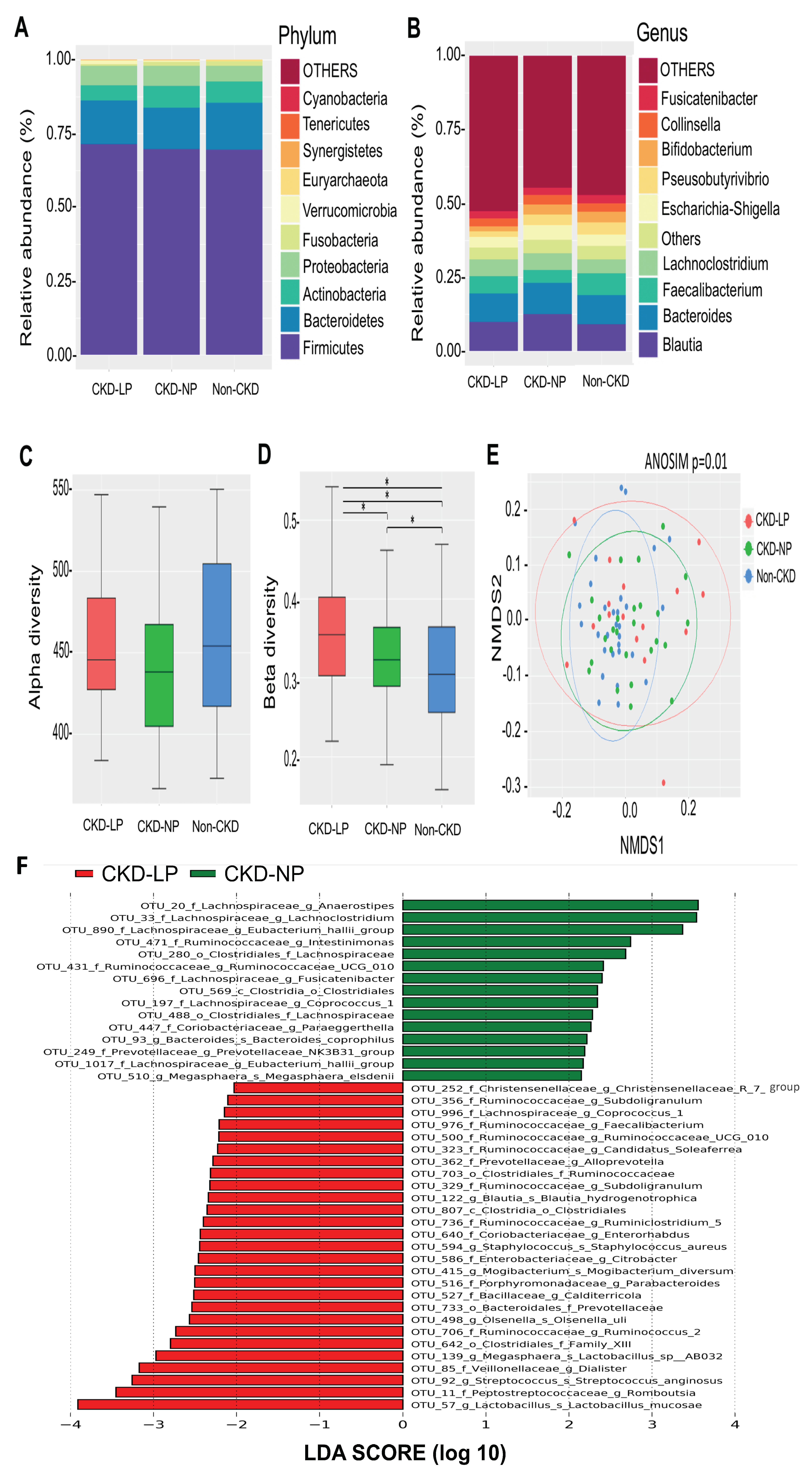
3.2. Changes of Microbial Composition and Diversity in CKD Patients Receiving LPD
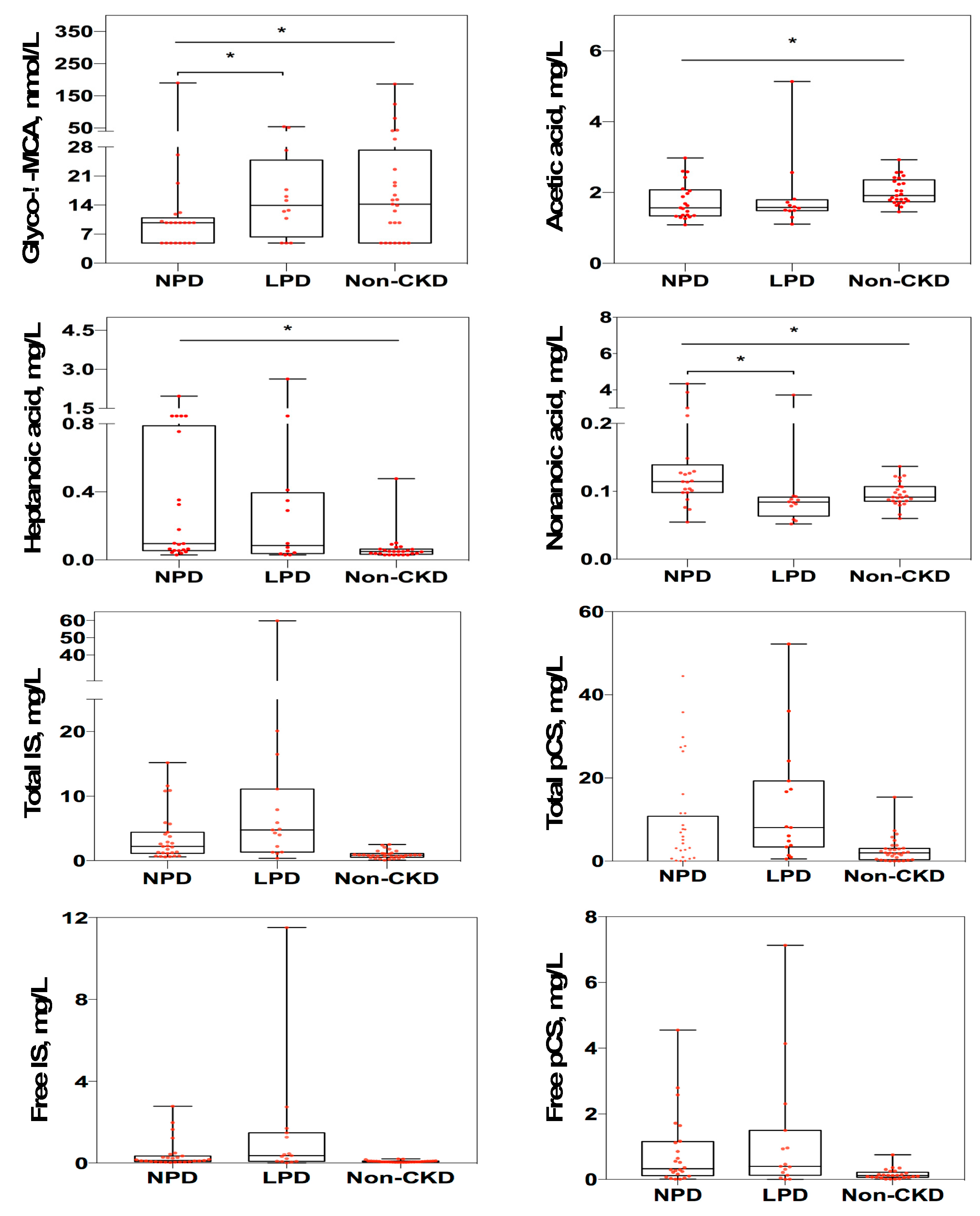
3.3. Changes of Targeted Metabolomics Profiling in CKD Patients Receiving LPD
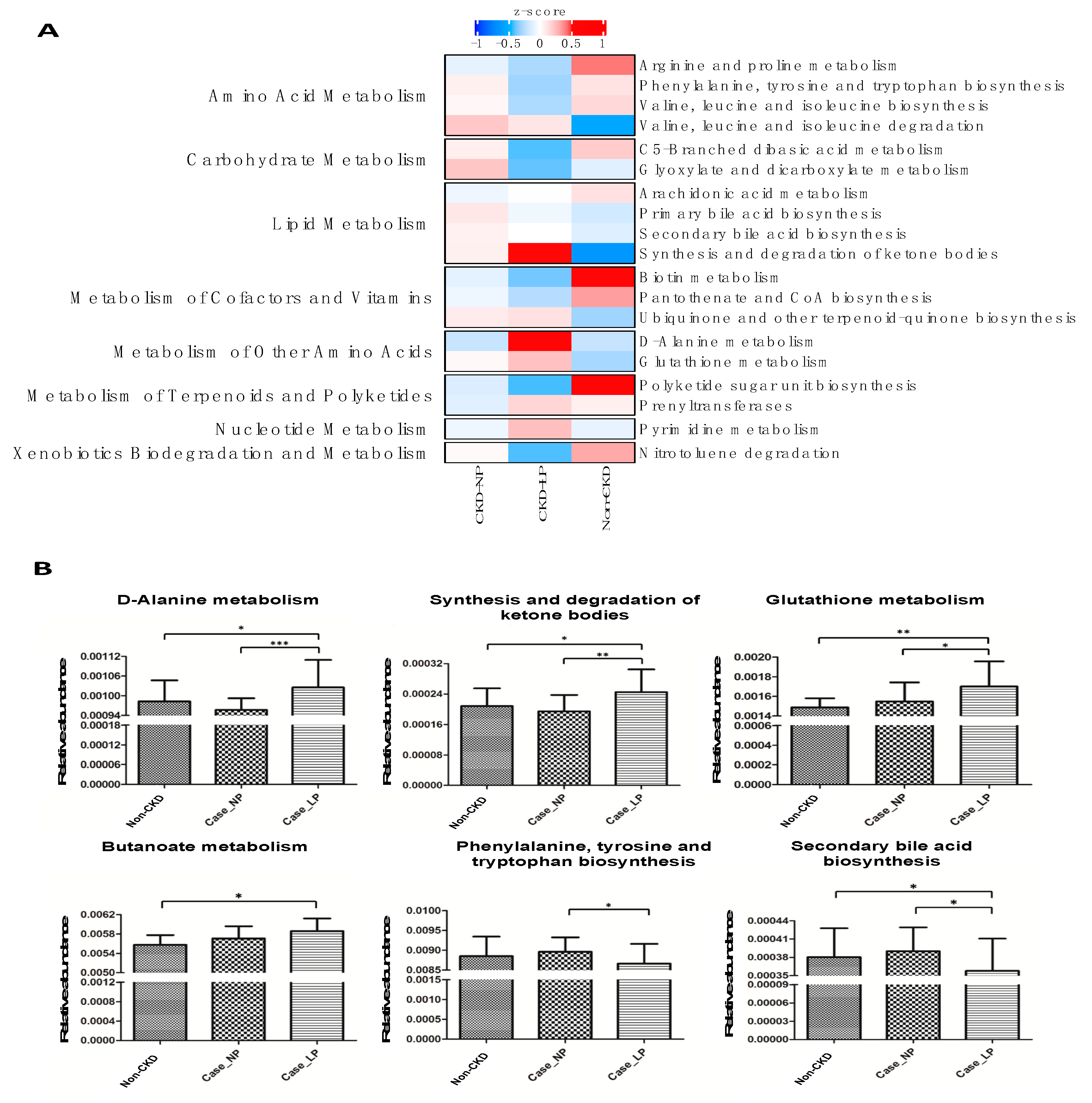
3.4. Functional Prediction of Change of Intestinal Microbiota Associated with LPD in CKD Patients
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Garneata, L.; Stancu, A.; Dragomir, D.; Stefan, G.; Mircescu, G. Ketoanalogue-supplemented vegetarian very low-protein diet and ckd progression. J. Am. Soc. Nephrol. 2016, 27, 2164–2176. [Google Scholar] [CrossRef]
- Li, A.; Lee, H.Y.; Lin, Y.C. The effect of ketoanalogues on chronic kidney disease deterioration: A meta-analysis. Nutrients 2019, 11, 957. [Google Scholar] [CrossRef] [PubMed]
- Klahr, S.; Levey, A.S.; Beck, G.J.; Caggiula, A.W.; Hunsicker, L.; Kusek, J.W.; Striker, G. The effects of dietary protein restriction and blood-pressure control on the progression of chronic renal disease. Modification of diet in renal disease study group. N. Engl. J. Med. 1994, 330, 877–884. [Google Scholar] [CrossRef] [PubMed]
- Milovanova, L.; Fomin, V.; Moiseev, S.; Taranova, M.; Milovanov, Y.; Lysenko Kozlovskaya, L.; Kozlov, V.; Kozevnikova, E.; Milovanova, S.; Lebedeva, M.; et al. Effect of essential amino acid кetoanalogues and protein restriction diet on morphogenetic proteins (FGF-23 and Кlotho) in 3b-4 stages chronic кidney disease patients: A randomized pilot study. Clin. Exp. Nephrol. 2018, 22, 1351–1359. [Google Scholar] [CrossRef]
- Kalantar-Zadeh, K.; Fouque, D. Nutritional management of chronic kidney disease. N. Engl. J. Med. 2017, 377, 1765–1776. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Tokumoto, M.; Tatsumoto, N.; Tsuruya, K.; Kitazono, T.; Ooboshi, H. Very low protein diet enhances inflammation, malnutrition, and vascular calcification in uremic rats. Life Sci. 2016, 146, 117–123. [Google Scholar] [CrossRef] [PubMed]
- Gentile, C.L.; Weir, T.L. The gut microbiota at the intersection of diet and human health. Science 2018, 362, 776–780. [Google Scholar] [CrossRef]
- Wu, G.D.; Chen, J.; Hoffmann, C.; Bittinger, K.; Chen, Y.Y.; Keilbaugh, S.A.; Bewtra, M.; Knights, D.; Walters, W.A.; Knight, R.; et al. Linking long-term dietary patterns with gut microbial enterotypes. Science 2011, 334, 105–108. [Google Scholar] [CrossRef]
- Black, A.P.; Anjos, J.S.; Cardozo, L.; Carmo, F.L.; Dolenga, C.J.; Nakao, L.S.; de Carvalho Ferreira, D.; Rosado, A.; Carraro Eduardo, J.C.; Mafra, D. Does low-protein diet influence the uremic toxin serum levels from the gut microbiota in nondialysis chronic kidney disease patients? J. Ren. Nutr. 2018, 28, 208–214. [Google Scholar] [CrossRef]
- Di Iorio, B.R.; Rocchetti, M.T.; De Angelis, M.; Cosola, C.; Marzocco, S.; Di Micco, L.; di Bari, I.; Accetturo, M.; Vacca, M.; Gobbetti, M.; et al. Nutritional therapy modulates intestinal microbiota and reduces serum levels of total and free indoxyl sulfate and p-cresyl sulfate in chronic kidney disease (medika study). J. Clin. Med. 2019, 8, 1424. [Google Scholar] [CrossRef]
- Wu, I.W.; Gao, S.S.; Chou, H.C.; Yang, H.Y.; Chang, L.C.; Kuo, Y.L.; Dinh, M.C.V.; Chung, W.H.; Yang, C.W.; Lai, H.C.; et al. Integrative metagenomic and metabolomic analyses reveal severity-specific signatures of gut microbiota in chronic kidney disease. Theranostics 2020, 10, 5398–5411. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.N.; Wu, I.W.; Huang, Y.F.; Peng, S.Y.; Huang, Y.C.; Ning, H.C. Measuring serum total and free indoxyl sulfate and p-cresyl sulfate in chronic kidney disease using uplc-ms/ms. J. Food Drug Anal. 2019, 27, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Wu, I.W.; Lin, C.Y.; Chang, L.C.; Lee, C.C.; Chiu, C.Y.; Hsu, H.J.; Sun, C.Y.; Chen, Y.C.; Kuo, Y.L.; Yang, C.W.; et al. Gut microbiota as diagnostic tools for mirroring disease progression and circulating nephrotoxin levels in chronic kidney disease: Discovery and validation study. Int. J. Biol. Sci. 2020, 16, 420–434. [Google Scholar] [CrossRef] [PubMed]
- Edgar, R.C. Uparse: Highly accurate otu sequences from microbial amplicon reads. Nat. Methods 2013, 10, 996–998. [Google Scholar] [CrossRef] [PubMed]
- Quast, C.; Pruesse, E.; Yilmaz, P.; Gerken, J.; Schweer, T.; Yarza, P.; Peplies, J.; Glockner, F.O. The silva ribosomal rna gene database project: Improved data processing and web-based tools. Nucleic Acids Res. 2013, 41, D590–D596. [Google Scholar] [CrossRef]
- Oksanen, J.; Blanchet, F.; Kindt, R.; Legendre, P.; Minchin, P.; O’Hara, R.; Simpson, G.; Solymos, P.; Stevens, M.; Wagner, H. Vegan: Community Ecology Package. R Package Version 2.1-1.; R Foundation for Statistical Computing: Vienna, Austria, 2015. [Google Scholar]
- Langille, M.G.; Zaneveld, J.; Caporaso, J.G.; McDonald, D.; Knights, D.; Reyes, J.A.; Clemente, J.C.; Burkepile, D.E.; Vega Thurber, R.L.; Knight, R.; et al. Predictive functional profiling of microbial communities using 16s rrna marker gene sequences. Nat. Biotechnol. 2013, 31, 814–821. [Google Scholar] [CrossRef]
- Markowitz, V.M.; Chen, I.M.; Palaniappan, K.; Chu, K.; Szeto, E.; Grechkin, Y.; Ratner, A.; Jacob, B.; Huang, J.; Williams, P.; et al. Img: The integrated microbial genomes database and comparative analysis system. Nucleic Acids Res. 2012, 40, D115–D122. [Google Scholar] [CrossRef]
- DeSantis, T.Z.; Hugenholtz, P.; Larsen, N.; Rojas, M.; Brodie, E.L.; Keller, K.; Huber, T.; Dalevi, D.; Hu, P.; Andersen, G.L. Greengenes, a chimera-checked 16s rrna gene database and workbench compatible with arb. Appl. Environ. Microbiol. 2006, 72, 5069–5072. [Google Scholar] [CrossRef]
- Kruskal, W.H.; Wallis, W.A. Use of ranks in one-criterion variance analysis. J. Am. Stat. Assoc. 1952, 47, 583–621. [Google Scholar] [CrossRef]
- Breiman, L. Random forests. Mach. Learn. 2001, 45, 5–32. [Google Scholar] [CrossRef]
- Feng, Y.L.; Cao, G.; Chen, D.Q.; Vaziri, N.D.; Chen, L.; Zhang, J.; Wang, M.; Guo, Y.; Zhao, Y.Y. Microbiome-metabolomics reveals gut microbiota associated with glycine-conjugated metabolites and polyamine metabolism in chronic kidney disease. Cell Mol. Life Sci. 2019, 76, 4961–4978. [Google Scholar] [CrossRef] [PubMed]
- Vaziri, N.D.; Wong, J.; Pahl, M.; Piceno, Y.M.; Yuan, J.; DeSantis, T.Z.; Ni, Z.; Nguyen, T.H.; Andersen, G.L. Chronic kidney disease alters intestinal microbial flora. Kidney Int. 2013, 83, 308–315. [Google Scholar] [CrossRef]
- Lai, S.; Molfino, A.; Testorio, M.; Perrotta, A.M.; Currado, A.; Pintus, G.; Pietrucci, D.; Unida, V.; La Rocca, D.; Biocca, S.; et al. Effect of low-protein diet and inulin on microbiota and clinical parameters in patients with chronic kidney disease. Nutrients 2019, 11, 3006. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Wu, W.; Chen, L.; Yang, W.; Huang, X.; Ma, C.; Chen, F.; Xiao, Y.; Zhao, Y.; Ma, C.; et al. Microbiota-derived short-chain fatty acids promote th1 cell il-10 production to maintain intestinal homeostasis. Nat. Commun. 2018, 9, 3555. [Google Scholar] [CrossRef] [PubMed]
- Pluznick, J.L. Gut microbiota in renal physiology: Focus on short-chain fatty acids and their receptors. Kidney Int. 2016, 90, 1191–1198. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Lv, D.; Jiang, S.; Jiang, J.; Liang, M.; Hou, F.; Chen, Y. Quantitative reduction in short-chain fatty acids, especially butyrate, contributes to the progression of chronic kidney disease. Clin. Sci. 2019, 133, 1857–1870. [Google Scholar] [CrossRef]
- Wong, J.; Piceno, Y.M.; DeSantis, T.Z.; Pahl, M.; Andersen, G.L.; Vaziri, N.D. Expansion of urease- and uricase-containing, indole- and p-cresol-forming and contraction of short-chain fatty acid-producing intestinal microbiota in esrd. Am. J. Nephrol. 2014, 39, 230–237. [Google Scholar] [CrossRef]
- Chu, L.; Zhang, K.; Zhang, Y.; Jin, X.; Jiang, H. Mechanism underlying an elevated serum bile acid level in chronic renal failure patients. Int. Urol. Nephrol. 2015, 47, 345–351. [Google Scholar] [CrossRef]
- Li, R.; Zeng, L.; Xie, S.; Chen, J.; Yu, Y.; Zhong, L. Targeted metabolomics study of serum bile acid profile in patients with end-stage renal disease undergoing hemodialysis. PeerJ 2019, 7, e7145. [Google Scholar] [CrossRef]
- Winston, J.A.; Theriot, C.M. Diversification of host bile acids by members of the gut microbiota. Gut Microbes 2019, 11, 158–171. [Google Scholar] [CrossRef]
- Wahlstrom, A.; Sayin, S.I.; Marschall, H.U.; Backhed, F. Intestinal crosstalk between bile acids and microbiota and its impact on host metabolism. Cell Metab. 2016, 24, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Jiang, C.; Krausz, K.W.; Li, Y.; Albert, I.; Hao, H.; Fabre, K.M.; Mitchell, J.B.; Patterson, A.D.; Gonzalez, F.J. Microbiome remodelling leads to inhibition of intestinal farnesoid x receptor signalling and decreased obesity. Nat. Commun. 2013, 4, 2384. [Google Scholar] [CrossRef] [PubMed]
- Sayin, S.I.; Wahlstrom, A.; Felin, J.; Jantti, S.; Marschall, H.U.; Bamberg, K.; Angelin, B.; Hyotylainen, T.; Oresic, M.; Backhed, F. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring fxr antagonist. Cell Metab. 2013, 17, 225–235. [Google Scholar] [CrossRef]
- Nazzal, L.; Roberts, J.; Singh, P.; Jhawar, S.; Matalon, A.; Gao, Z.; Holzman, R.; Liebes, L.; Blaser, M.J.; Lowenstein, J. Microbiome perturbation by oral vancomycin reduces plasma concentration of two gut-derived uremic solutes, indoxyl sulfate and p-cresyl sulfate, in end-stage renal disease. Nephrol. Dial. Transplant. 2017, 32, 1809–1817. [Google Scholar] [CrossRef] [PubMed]
- Koppe, L.; Mafra, D.; Fouque, D. Probiotics and chronic kidney disease. Kidney Int. 2015, 88, 958–966. [Google Scholar] [CrossRef]
- Gryp, T.; De Paepe, K.; Vanholder, R.; Kerckhof, F.M.; Van Biesen, W.; Van de Wiele, T.; Verbeke, F.; Speeckaert, M.; Joossens, M.; Couttenye, M.M.; et al. Gut microbiota generation of protein-bound uremic toxins and related metabolites is not altered at different stages of chronic kidney disease. Kidney Int. 2020, 97, 1230–1242. [Google Scholar] [CrossRef]
- Cabrera-Mulero, A.; Tinahones, A.; Bandera, B.; Moreno-Indias, I.; Macías-González, M.; Tinahones, F.J. Keto microbiota: A powerful contributor to host disease recovery. Rev. Endocr. Metab. Disord. 2019, 20, 415–425. [Google Scholar] [CrossRef]
| All Patients | Non-CKD | CKD-NPD | CKD-LPD | p | |
|---|---|---|---|---|---|
| n = 77 | n = 34 | n = 27 | n = 16 | ||
| Age, mean (SD) | 63.40 ± 6.49 | 62.15 ± 6.58 | 63.48 ± 6.12 | 65.94 ± 6.52 | 0.227 |
| Male, n (%) | 38 (49.40) | 14 (41.20) | 13 (48.10) | 11 (68.80) | 0.267 |
| Diabetes, n (%) | 41 (53.20) | 16 (47.10) | 15 (55.60) | 10 (62.50) | 0.477 |
| Hypertension, n (%) | 60 (77.90) | 21 (61.80) | 23 (85.20) | 16 (100.00) | 0.245 |
| Body mass index | 25.82 ± 3.47 | 25.92 ± 3.82 | 26.12 ± 3.04 | 25.10 ± 3.51 | 0.271 |
| Systolic pressure, mmHg | 133.26 ± 17.14 | 130.88 ± 18.10 | 135.15 ± 13.50 | 135.13 ± 20.71 | 0.891 |
| Dietary intake (serving/day) | |||||
| Vegetable | 1.7 ± 0.8 | 1.9 ± 0.8 | 1.5 ± 0.6 | 1.0 ± 0.5 | 0.182 |
| Meat | 1.6 ± 0.8 | 1.8 ± 0.9 | 1.6 ± 0.8 | 1.1 ± 0.6 | 0.274 |
| Fruit | 0.7 ± 0.6 | 0.7 ± 0.5 | 0.7 ± 0.6 | 0.6 ± 0.5 | 0.930 |
| Rice/noodle | 2.3 ± 0.7 | 2.3 ± 0.7 | 2.5 ± 0.8 | 2.3 ± 0.9 | 0.661 |
| Estimated protein intake, g/day | 1.00 ± 0.35 | 1.22 ± 0.41 | 1.16 ± 0.26 | 0.65 ± 0.12 | <0.001 |
| Blood urea nitrogen, mg/dL | 27.82 ± 25.74 | 14.35 ± 3.81 | 30.59 ± 23.19 | 51.75 ± 36.84 | 0.057 |
| Serum creatinine, mg/dL | 1.81 ± 2.00 | 0.78 ± 0.23 | 2.08 ± 1.83 | 3.52 ± 2.94 | 0.065 |
| Estimated GFR, mL/min/m2 | 69.24 ± 52.15 | 99.52 ± 56.04 | 53.22 ± 36.06 | 31.95 ± 24.31 | 0.047 |
| Hemoglobin, g/dL | 12.71 ± 2.07 | 13.59 ± 1.22 | 12.66 ± 1.81 | 10.94 ± 2.74 | 0.043 |
| Serum albumin, mg/dL | 4.43 ± 0.46 | 4.54 ± 0.24 | 4.53 ± 0.37 | 4.02 ± 0.68 | 0.012 |
| Serum potassium, mEq/L | 4.22 ± 0.47 | 4.06 ± 0.33 | 4.31 ± 0.37 | 4.43 ± 0.71 | 0.595 |
| Fasting sugar, mg/dL | 126.09 ± 45.79 | 119.88 ± 30.97 | 132.33 ± 52.62 | 128.75 ± 59.59 | 0.788 |
| Total cholesterol, mg/dL | 191.75 ± 37.57 | 197.35 ± 26.09 | 187.33 ± 46.46 | 187.31 ± 42.26 | 0.923 |
| hs-CRP, mg/L # | 1.62 (2.02) | 1.38 (1.82) | 1.35 (3.57) | 2.15 (1.55) | 0.479 |
| Urine protein-creatinine ratio, g/g # | 108.34 (375.72) | 77.24 (32.49) | 223.95 (819.14) | 411.85 (2639.28) | 0.471 |
| Urine output, mL/day | 2081.3 ± 668.3 | 2197.3 ± 513.9 | 2229.6 ± 685.0 | 1751.3 ± 659.2 | 0.029 |
| Gut Microbiota | RA (%) Non-CKD | RA (%) CKD-LPD | RA (%) CKD-NPD | P * | p (LPD vs. NPD) # | p (LPD vs. Non-CKD) # | p (NPD vs. Non-CKD) # | |
|---|---|---|---|---|---|---|---|---|
| Family | Genus | |||||||
| Bacillaceae | Calditerricola ↑ | 0.0000887 | 0.00117 | 0.0000985 | 0.003579 | 0.001398 | 0.001015 | 0.4818 |
| Desulfovibrionaceae | Desulfovibrio ↓ | 0.000177 | 0.0015 | 0.00405 | 0.01513 | 0.4994 | 0.01308 | 0.004705 |
| Lachnospiraceae | Pseudobutyrivibrio ↓ | 4.099 | 1.878 | 3.557 | 0.0007933 | 0.02095 | 0.0000888 | 0.02529 |
| Lachnospiraceae | Lachnospira ↓ | 0.06535 | 0.0258 | 0.07014 | 0.003283 | 0.04567 | 0.0004307 | 0.03001 |
| Lachnospiraceae | Eubacterium_hallii_group ↓ | 0.7137 | 0.3318 | 0.8227 | 0.007689 | 0.01179 | 0.0009717 | 0.1778 |
| Lachnospiraceae | Roseburia ↓ | 0.2302 | 0.08307 | 0.1382 | 0.007848 | 0.04956 | 0.001018 | 0.05064 |
| Lachnospiraceae | Coprococcus_1 ↑ | 0.00071 | 0.01715 | 0.00069 | 0.01597 | 0.01435 | 0.00229 | 0.2399 |
| Lachnospiraceae | Fusicatenibacter ↓ | 0.2073 | 0.1658 | 0.2177 | 0.01854 | 0.006801 | 0.004211 | 0.4444 |
| Lachnospiraceae | Anaerostipes ↓ | 1.653 | 0.8215 | 1.58 | 0.02399 | 0.01337 | 0.00408 | 0.3255 |
| Lachnospiraceae | Lachnoclostridium ↓ | 0.8864 | 0.2202 | 0.8797 | 0.0005679 | 0.002618 | 0.0000611 | 0.1225 |
| Peptostreptococcaceae | Romboutsia ↑ | 0.9836 | 1.486 | 0.9934 | 0.04816 | 0.007215 | 0.03711 | 0.2043 |
| Porphyromonadaceae | Parabacteroides ↑ | 0.000444 | 0.0015 | 0.000197 | 0.02739 | 0.004277 | 0.01831 | 0.2457 |
| Prevotellaceae | Alloprevotella ↑ | 0.00471 | 0.03646 | 0.00543 | 0.02063 | 0.005736 | 0.005909 | 0.4726 |
| Prevotellaceae | Prevotellaceae_NK3B31 ↓ | 0.02477 | 0.000499 | 0.015 | 0.02164 | 0.02963 | 0.00285 | 0.1629 |
| Ruminococcaceae | Subdoligranulum ↑ | 0.0087 | 0.02014 | 0.000395 | 0.0007012 | 0.000141 | 0.0757 | 0.004089 |
| Ruminococcaceae | Ruminococcaceae_UCG-010 ↑ | 0.000355 | 0.002 | 0.00178 | 0.008634 | 0.01071 | 0.001147 | 0.2054 |
| Ruminococcaceae | Faecalibacterium ↑ | 0.02628 | 0.02231 | 0.0146 | 0.01278 | 0.004034 | 0.2441 | 0.009593 |
| Ruminococcaceae | Subdoligranulum ↑ | 0.00648 | 0.01698 | 0.00128 | 0.02464 | 0.008032 | 0.2859 | 0.01378 |
| Synergistaceae | Cloacibacillus ↑ | 0.00311 | 0.01282 | 0.01154 | 0.02992 | 0.007619 | 0.008901 | 0.452 |
| Family/genus | Species | |||||||
| Bacteroidaceae/Bacteroides | Bacteroides_coprophilus ↓ | 0.4395 | 0.01698 | 0.04913 | 0.003919 | 0.02987 | 0.0004587 | 0.05165 |
| Bacteroidaceae/Bacteroides | Bacteroides_plebeius ↓ | 2.189 | 0.3233 | 0.6128 | 0.005041 | 0.1501 | 0.001213 | 0.01054 |
| Bacteroidaceae/Bacteroides | Bacteroides_eggerthii ↓ | 0.22 | 0.0283 | 0.2326 | 0.01589 | 0.03211 | 0.001999 | 0.1235 |
| Clostridiaceae/Clostridium_sensu_stricto_1 | Clostridium_paraputrificum ↑ | 0.01474 | 0.03363 | 0.01934 | 0.003622 | 0.03818 | 0.0004482 | 0.03853 |
| Clostridiaceae/Peptoclostridium | Clostridium_sordellii ↑ | 0.01385 | 0.05743 | 0.03325 | 0.0001,322 | 0.03922 | 0.0000,226 | 0.003827 |
| Coriobacteriaceae/Olsenella | Olsenella_uli ↑ | 0.00124 | 0.00183 | 0.000394 | 0.02391 | 0.004397 | 0.1401 | 0.03176 |
| Eubacteriaceae/Mogibacterium | Mogibacterium_diversum ↑ | 0.000621 | 0.00216 | 0.000789 | 0.01012 | 0.004809 | 0.002067 | 0.3947 |
| Lachnospiraceae/Blautia | Blautia_hydrogenotrophica ↑ | 0.08488 | 0.09689 | 0.0731 | 0.03329 | 0.009464 | 0.008729 | 0.493 |
| Lactobacillaceae/Lactobacillus | Lactobacillus_mucosae ↑ | 0.04377 | 1.765 | 0.07665 | 0.01466 | 0.01764 | 0.001946 | 0.1935 |
| Porphyromonadaceae/Porphyromonas | Porphyromonas_gingivalis ↑ | 0.000355 | 0.01731 | 0.000789 | 0.02485 | 0.04228 | 0.00328 | 0.1312 |
| Streptococcaceae/Streptococcus | Streptococcus_anginosus ↑ | 0.05727 | 0.4275 | 0.07586 | 0.009104 | 0.004643 | 0.001801 | 0.3808 |
| Veillonellaceae/Megasphaera | Lactobacillus_sp._AB032 ↑ | 0.01394 | 0.1806 | 0.00641 | 0.03676 | 0.01011 | 0.009835 | 0.4841 |
| Overall Patients | Only CKD Patients | |||
|---|---|---|---|---|
| r | p | r | p | |
| Genus | ||||
| Anaerostipes | 0.343 | 0.009 | 0.346 | 0.023 |
| Calditerricola | −0.278 | 0.036 | −0.336 | 0.027 |
| Eubacterium hallii group | 0.263 | 0.048 | 0.371 | 0.014 |
| Species | ||||
| Streptococcus anginosus | −0.466 | <0.001 | −0.442 | 0.003 |
| Lactobacillus mucosae | −0.4 | 0.002 | −0.409 | 0.006 |
| Clostridium paraputrificum | −309 | 0.019 | −0.446 | 0.003 |
| r = rho-based Spearman correlation coefficient | ||||
| rCKD-NPD | rCKD-LPD | p | |
|---|---|---|---|
| Age, mean (SD) | 65.14 ± 6.5 | 64.86 ± 5.3 | 0.890 |
| Male, n (%) | 5 (35.7%) | 9 (64.3%) | 0.131 |
| Diabetes, n (%) | 8 (57.1%) | 9 (64.3%) | 0.699 |
| Estimated GFR, mL/min/m2 | 40.58 ± 23.3 | 30.22 ± 22.8 | 0.240 |
| Serum creatinine, mg/dL | 2.42 ± 2.1 | 3.66 ± 3.1 | 0.220 |
| Estimated protein intake, g/day | 1.22 ± 0.3 | 0.67 ± 0.1 | <0.001 |
| Urine protein-creatinine ratio, g/g # | 465.85 (1952.44) | 264.41 (2241.62) | 0.950 |
| Urine output, mL/day | 2182.14 ± 721.5 | 1739.29 ± 697.6 | 0.110 |
| Genus, relative abundance (%) | |||
| Anaerostipes | 1.77 ± 0.0,142 | 0.9 ± 0.006 | 0.047 |
| Calditerricola | 0.0001 ± 0.0,001 | 0.001 ± 0.002 | 0.050 |
| Eubacterium hallii group # | 3.04 ± 0.0,238 | 1.81 ± 0.0105 | 0.050 |
| Species, relative abundance (%) | |||
| Streptococcus anginosus # | 0.06 ± 0.0,007 | 0.47 ± 0.0121 | 0.041 |
| Lactobacillus mucosae # | 0.1 ± 0.0,012 | 2.02 ± 0.0498 | 0.035 |
| Clostridium paraputrificum # | 0.01 ± 0.0,001 | 0.04 ± 0.0004 | 0.060 |
| Metabolites | |||
| Glyco-λ-muricholic acid, nmol/L # | 9.75 (5.04) | 13.93 (18.5) | 0.011 |
| Acetic acid, mg/L # | 1.69 (0.94) | 1.58 (0.31) | 0.860 |
| Heptanoic acid, mg/L # | 0.33 (1.04) | 0.09 (0.36) | 0.176 |
| Nonanoic acid, mg/L # | 0.12 (2.76) | 0.08 (0.03) | 0.005 |
| Total indoxyl sulfate, mg/L # | 2.7 (5.87) | 4.54 (13.84) | 0.257 |
| Total p-cresyl sulfate, mg/L # | 8.66 (23.76) | 5.45 (20.5) | 0.946 |
| Free indoxyl sulfate, mg/L # | 0.26 (0.94) | 0.34 (1.56) | 0.257 |
| Free p-cresyl sulfate, mg/L # | 0.64 (1.23) | 0.39 (1.30) | 0.796 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, I.-W.; Lee, C.-C.; Hsu, H.-J.; Sun, C.-Y.; Chen, Y.-C.; Yang, K.-J.; Yang, C.-W.; Chung, W.-H.; Lai, H.-C.; Chang, L.-C.; et al. Compositional and Functional Adaptations of Intestinal Microbiota and Related Metabolites in CKD Patients Receiving Dietary Protein Restriction. Nutrients 2020, 12, 2799. https://doi.org/10.3390/nu12092799
Wu I-W, Lee C-C, Hsu H-J, Sun C-Y, Chen Y-C, Yang K-J, Yang C-W, Chung W-H, Lai H-C, Chang L-C, et al. Compositional and Functional Adaptations of Intestinal Microbiota and Related Metabolites in CKD Patients Receiving Dietary Protein Restriction. Nutrients. 2020; 12(9):2799. https://doi.org/10.3390/nu12092799
Chicago/Turabian StyleWu, I-Wen, Chin-Chan Lee, Heng-Jung Hsu, Chiao-Yin Sun, Yuen-Chan Chen, Kai-Jie Yang, Chi-Wei Yang, Wen-Hun Chung, Hsin-Chih Lai, Lun-Ching Chang, and et al. 2020. "Compositional and Functional Adaptations of Intestinal Microbiota and Related Metabolites in CKD Patients Receiving Dietary Protein Restriction" Nutrients 12, no. 9: 2799. https://doi.org/10.3390/nu12092799
APA StyleWu, I.-W., Lee, C.-C., Hsu, H.-J., Sun, C.-Y., Chen, Y.-C., Yang, K.-J., Yang, C.-W., Chung, W.-H., Lai, H.-C., Chang, L.-C., & Su, S.-C. (2020). Compositional and Functional Adaptations of Intestinal Microbiota and Related Metabolites in CKD Patients Receiving Dietary Protein Restriction. Nutrients, 12(9), 2799. https://doi.org/10.3390/nu12092799






