Logistic LASSO Regression for Dietary Intakes and Breast Cancer
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
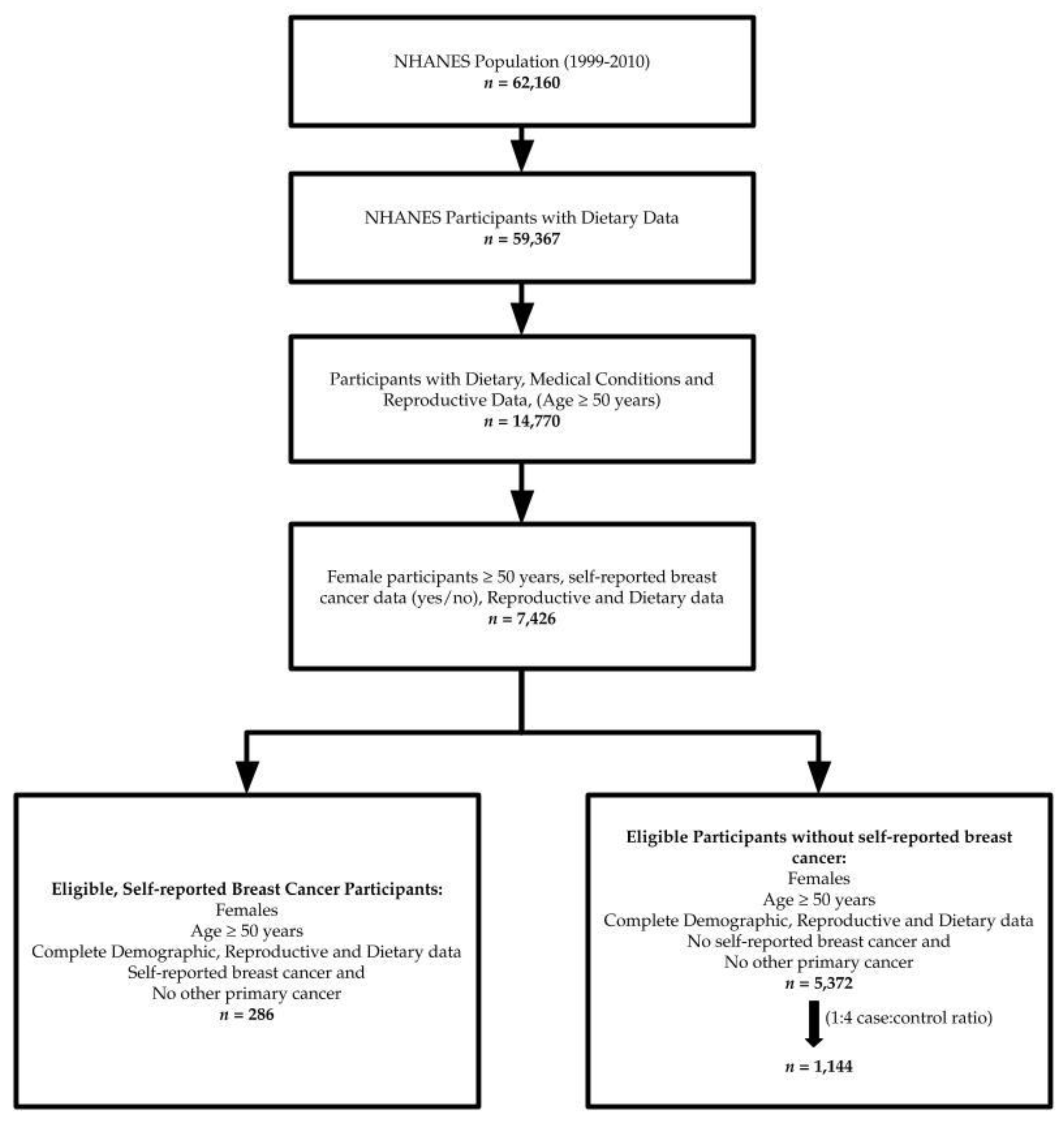
2.2. Sample
2.3. Breast Cancer Data
2.4. Dietary Intake
2.5. Other Measures
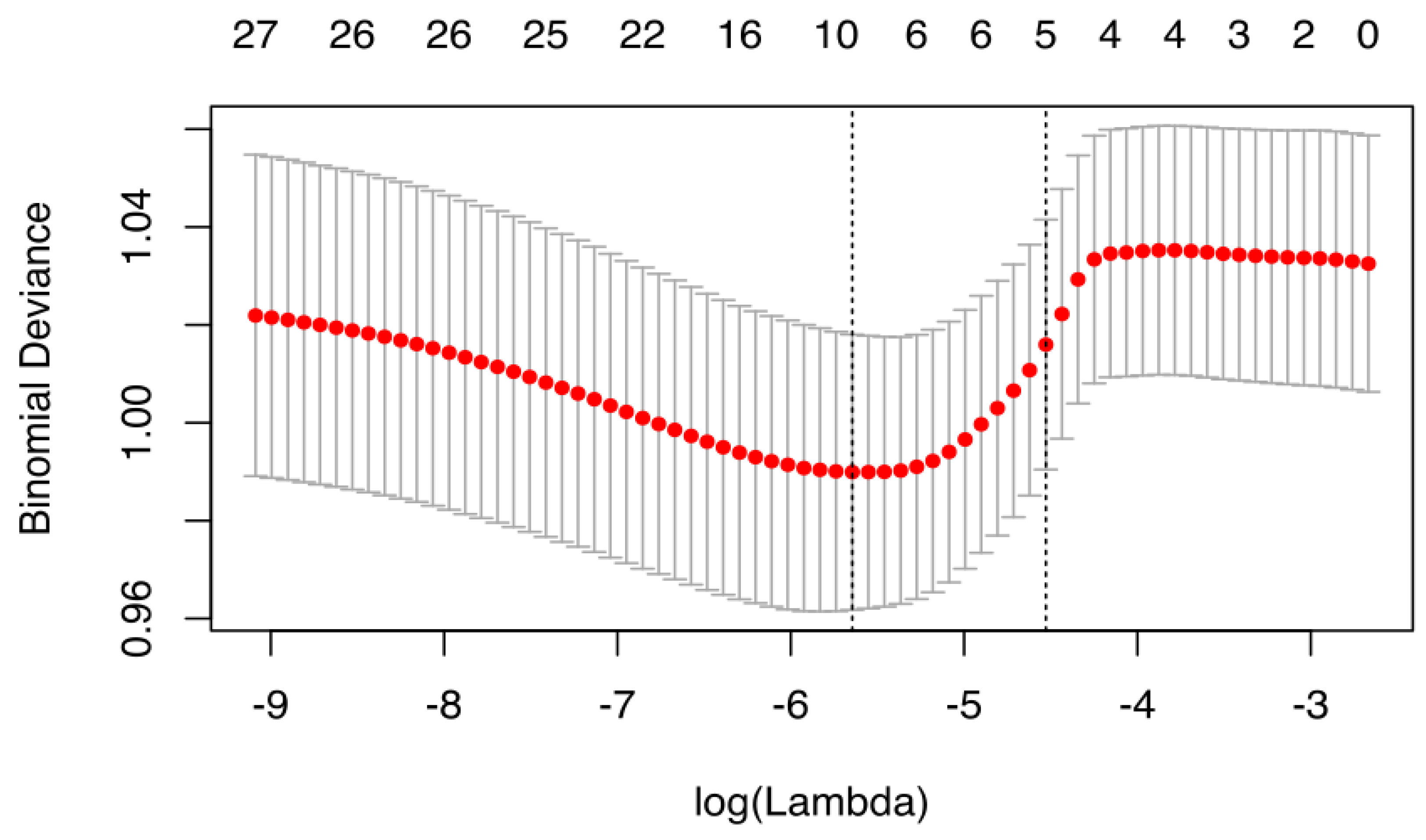
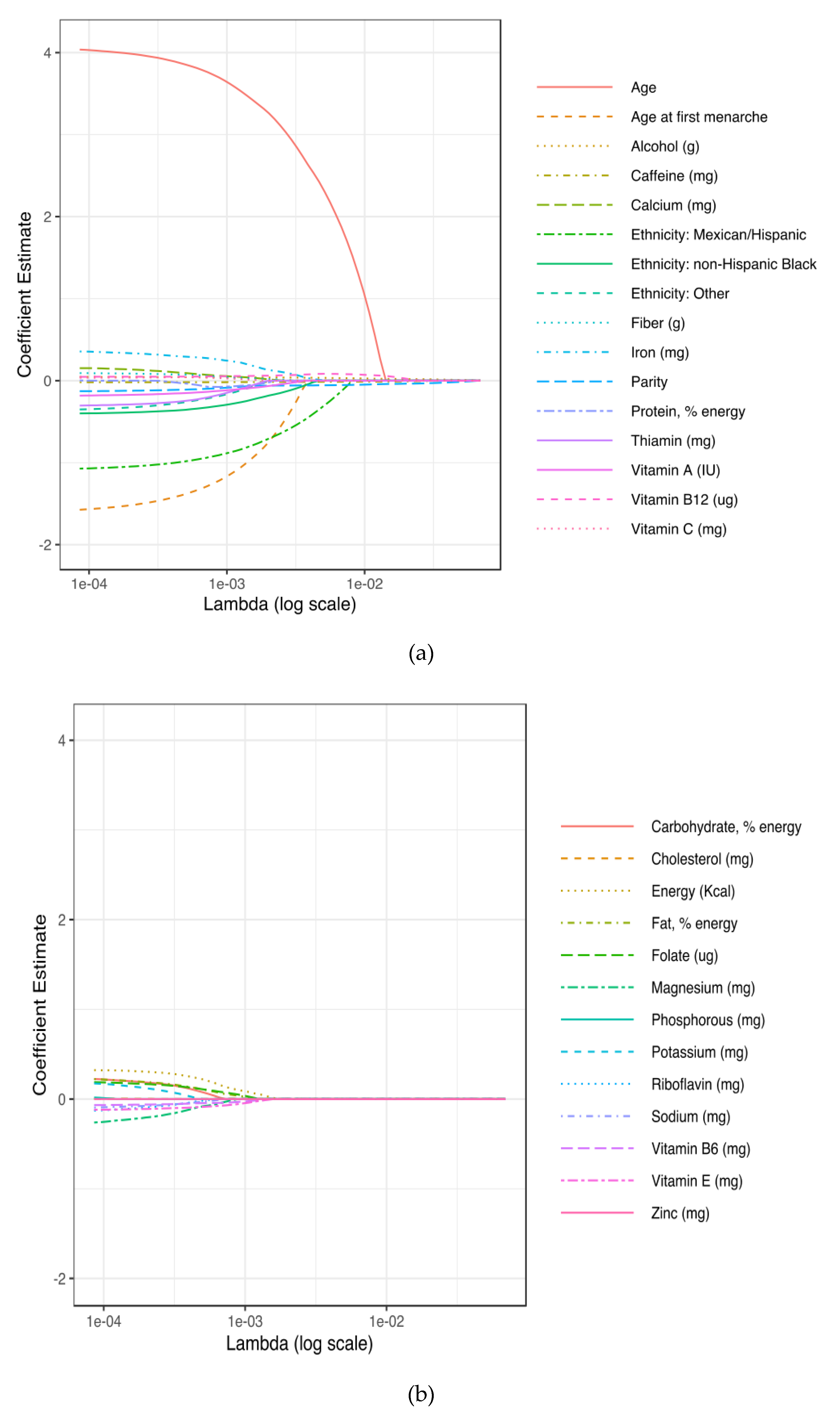
2.6. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Cancer Society. Breast Cancer Facts & Figures 2019–2020; American Cancer Society, Inc.: Atlanta, GA, USA, 2019. [Google Scholar]
- Patterson, R.E.; Rock, C.L.; Kerr, J.; Natarajan, L.; Marshall, S.J.; Pakiz, B.; Cadmus-Bertram, L.A. Metabolism and breast cancer risk: Frontiers in research and practice. J. Acad. Nutr. Diet. 2013, 113, 288–296. [Google Scholar] [CrossRef]
- World Cancer Research Fund and American Institute for Cancer Research. Continuous Update Project Report Expert Report 2018. Diet, Nutrition, Physical Activity, and Breast Cancer. Available online: dietandcancerreport.org (accessed on 24 July 2020).
- Farvid, M.S.; Chen, W.Y.; Rosner, B.A.; Tamimi, R.M.; Willett, W.C.; Eliassen, A.H. Fruit and vegetable consumption and breast cancer incidence: Repeated measures over 30 years of follow-up. Int J. Cancer 2019, 144, 1496–1510. [Google Scholar] [CrossRef] [PubMed]
- Kushi, L.H.; Doyle, C.; McCullough, M.; Rock, C.L.; Demark-Wahnefried, W.; Bandera, E.V.; Gapstur, S.; Patel, A.V.; Andrews, K.; Gansler, T. American Cancer Society 2010 Nutrition and Physical Activity Guidelines Advisory Committee. American Cancer Society Guidelines on nutrition and physical activity for cancer prevention: Reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J. Clin. 2012, 62, 30–67. [Google Scholar] [CrossRef] [PubMed]
- Chlebowski, R.T.; Luo, J.; Anderson, G.L.; Barrington, W.; Reding, K.; Simon, M.S.; Manson, J.E.; Rohan, T.E.; Wactawski-Wende, J.; Lane, D.; et al. Weight loss and breast cancer incidence in postmenopausal women. Cancer 2019, 125, 205–212. [Google Scholar] [CrossRef]
- Hruby, A.; Manson, J.E.; Qi, L.; Malik, V.S.; Rimm, E.B.; Sun, Q.; Willett, W.C.; Hu, F.B. Determinants and Consequences of Obesity. Am. J. Public Health. 2016, 106, 1656–1662. [Google Scholar] [CrossRef]
- Willett, W.C. Diet and breast cancer. J. Intern. Med. 2001, 249, 395–411. [Google Scholar] [CrossRef] [PubMed]
- Hunter, D.J.; Spiegelman, D.; Adami, H.O.; Beeson, L.; van den Brandt, P.A.; Folsom, A.R.; Fraser, G.E.; Goldbohm, R.A.; Graham, S.; Howe, G.R. Cohort studies of fat intake and the risk of breast cancer-a pooled analysis. N. Engl. J. Med. 1996, 334, 356–361. [Google Scholar] [CrossRef]
- Dolley, S. Big Data’s Role in Precision Public Health. Front. Public Health 2018, 6, 68. [Google Scholar] [CrossRef]
- Zipf, G.; Chiappa, M.; Porter, K.S.; Ostchega, Y.; Lewis, B.G.; Dostal, J. National health and nutrition examination survey: Plan and operations, 1999–2010. Vital Health Stat. 2013, 56, 1–37. [Google Scholar]
- Tibshirani, R. Regression shrinkage and selection via the Lasso. J. R Stat. Soc. Ser. B Methodol. 1996, 58, 267–288. [Google Scholar] [CrossRef]
- Bickel, P.J.; Ritov, Y.; Tsybakov, A.B. Simultaneous analysis of Lasso and dantzig selector. Ann. Stat. 2009, 37, 1705–1732. [Google Scholar] [CrossRef]
- Efron, B.; Hastie, T.; Johnstone, I.; Tibshirani, R. Least angle regression. Ann. Stat. 2004, 32, 407–451. [Google Scholar]
- Lee, S.; Kwon, S.; Kim, Y. A modified local quadratic approximation algorithm for penalized optimization problems. Comput Stat. Data Anal. 2016, 94, 275–286. [Google Scholar] [CrossRef]
- Hennessy, S.; Bilker, W.B.; Berlin, J.A.; Strom, B.L. Factors influencing the optimal control-to-case ratio in matched case-control studies. Am. J. Epidemiol. 1999, 149, 195–197. [Google Scholar] [CrossRef] [PubMed]
- United States Department of Agriculture. Agriculture Research Service. Available online: http://www.ars.usda.gov/ba/bhnrc/fsrg (accessed on 7 February 2017).
- United States Department of Agriculture. Available online: https://fdc.nal.usda.gov/ (accessed on 7 February 2017).
- McConville, K.S. Improved Estimation for Complex Surveys Using Modern Regression Techniques. Ph.D. Thesis, Colorado State University, Fort Collins, CO, USA, 2011. [Google Scholar]
- McConville, K.S.; Breidt, F.J.; Lee, T.C.; Moisen, G.G. Model-assisted survey regression estimation with the lasso. J. Surv Stat. Method. 2017, 5, 131–158. [Google Scholar] [CrossRef]
- Efron, B.; Hastie, T. Computer Age Statistical Inference: Algorithms, Evidence, and Data Science; Cambridge University Press: Cambridge, UK, 2016. [Google Scholar]
- Coughlin, S.S. Epidemiology of Breast Cancer in Women. Adv. Exp. Med. Biol. 2019, 1152, 9–29. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef]
- Tamimi, R.M.; Spiegelman, D.; Smith-Warner, S.A.; Wang, M.; Pazaris, M.; Willett, W.C.; Eliassen, A.H.; Hunter, D.J. Population Attributable Risk of Modifiable and Nonmodifiable Breast Cancer Risk Factors in Postmenopausal Breast Cancer. Am. J. Epidemiol. 2016, 184, 884–893. [Google Scholar] [CrossRef]
- Dall, G.V.; Britt, K.L. Estrogen Effects on the Mammary Gland in Early and Late Life and Breast Cancer Risk. Front. Oncol. 2017, 7, 110. [Google Scholar] [CrossRef]
- Gaudet, M.M.; Gierach, G.L.; Carter, B.D.; Luo, J.; Milne, R.L.; Weiderpass, E.; Giles, G.G.; Tamimi, R.M.; Eliassen, A.H.; Rosner, B.; et al. Pooled Analysis of Nine Cohorts Reveals Breast Cancer Risk Factors by Tumor Molecular Subtype. Cancer Res. 2018, 78, 6011–6021. [Google Scholar] [CrossRef]
- Kelsey, J.L.; Gammon, M.D.; John, E.M. Reproductive factors and breast cancer. Epidemiol. Rev. 1993, 15, 36–47. [Google Scholar] [CrossRef] [PubMed]
- Sineshaw, H.M.; Gaudet, M.; Ward, E.M.; Flanders, W.D.; Desantis, C.; Lin, C.C.; Jemal, A. Association of race/ethnicity, socioeconomic status, and breast cancer subtypes in the National Cancer Data Base (2010–2011). Breast Cancer Res. Treat. 2014, 145, 753–763. [Google Scholar] [CrossRef] [PubMed]
- IARC working group on the evaluation of carcinogenic risks to humans. Alcohol Consumption and Ethyl Carbamate. IARC Monogr. Eval. Carcinog. Risks Hum. 2010, 96, 3. [Google Scholar]
- Seitz, H.K.; Maurer, B. The relationship between alcohol metabolism, estrogen levels, and breast cancer risk. Alcohol Res. 2007, 30, 42–43. [Google Scholar]
- Rosenberg, L.; Shapiro, S.; Slone, D.; Kaufman, D.; Helmrich, S.; Miettinen, O.; Engle JR, R. Breast cancer and alcoholic-beverage consumption. Lancet 1982, 319, 267–271. [Google Scholar] [CrossRef]
- Talamini, R.; La Vecchia, C.; Decarli, A.; Franceschi, S.; Grattoni, E.; Grigoletto, E.; Tognoni, G. Social factors, diet and breast cancer in a northern Italian population. Br. J. Cancer 1984, 49, 723–729. [Google Scholar] [CrossRef]
- Coronado, G.D.; Beasley, J.; Livaudais, J. Alcohol consumption and the risk of breast cancer. Salud Publica Mex. 2011, 53, 440–447. [Google Scholar]
- Pelucchi, C.; Tramacere, I.; Boffetta, P.; Negri, E.; La Vecchia, C. Alcohol consumption and cancer risk. Nutr. Cancer 2011, 63, 983–990. [Google Scholar] [CrossRef]
- Chen, W.Y.; Rosner, B.; Hankinson, S.E.; Colditz, G.A.; Willett, W.C. Moderate alcohol consumption during adult life, drinking patterns, and breast cancer risk. JAMA. 2011, 306, 1884–1890. [Google Scholar] [CrossRef]
- Secretan, B.; Straif, K.; Baan, R.; Grosse, Y.; El Ghissassi, F.; Bouvard, V.; Benbrahim-Tallaa, L.; Guha, N.; Freeman, C.; Galichet, L.; et al. A review of human carcinogens—Part E: Tobacco, areca nut, alcohol, coal smoke, and salted fish. Lancet Oncol. 2009, 10, 1033–1034. [Google Scholar] [CrossRef]
- Baker, J.A.; Beehler, G.P.; Sawant, A.C.; Jayaprakash, V.; McCann, S.E.; Moysich, K.B. Consumption of coffee, but not black tea, is associated with decreased risk of premenopausal breast cancer. J. Nutr. 2006, 136, 166–171. [Google Scholar] [CrossRef]
- Lowcock, E.C.; Cotterchio, M.; Anderson, L.N.; Boucher, B.A.; El-Sohemy, A. High coffee intake, but not caffeine, is associated with reduced estrogen receptor negative and postmenopausal breast cancer risk with no effect modification by CYP1A2 genotype. Nutr. Cancer 2013, 65, 398–409. [Google Scholar] [CrossRef] [PubMed]
- Oh, J.K.; Sandin, S.; Ström, P.; Löf, M.; Adami, H.O.; Weiderpass, E. Prospective study of breast cancer in relation to coffee, tea and caffeine in Sweden. Int. J. Cancer 2015, 137, 1979–1989. [Google Scholar] [CrossRef] [PubMed]
- Bhoo-Pathy, N.; Peeters, P.H.M.; Uiterwaal, C.S.P.M.; Bueno-de-Mesquita, H.B.; Bulgiba, A.M.; Bech, B.H.; Overvad, K.; Tjønneland, A.; Olsen, A.; Clavel-Chapelon, F.; et al. Coffee and tea consumption and risk of pre- and postmenopausal breast cancer in the European Prospective Investigation into Cancer and Nutrition (EPIC) cohort study. Breast Cancer Res. 2015, 17, 15. [Google Scholar] [CrossRef] [PubMed]
- Fagherazzi, G.; Touillaud, M.S.; Boutron-Ruault, M.C.; Clavel-Chapelon, F.; Romieu, I. No association between coffee, tea or caffeine consumption and breast cancer risk in a prospective cohort study. Public Health Nutr. 2011, 14, 1315–1320. [Google Scholar] [CrossRef]
- Ishitani, K.; Lin, J.; Manson, J.E.; Buring, J.E.; Zhang, S.M. Caffeine consumption and the risk of breast cancer in a large prospective cohort of women. Arch. Intern. Med. 2008, 168, 2022–2031. [Google Scholar] [CrossRef]
- Ganmaa, D.; Willett, W.C.; Li, T.Y.; Feskanich, D.; van Dam, R.M.; Lopez-Garcia, E.; Hunter, D.J.; Holmes, M.D. Coffee, tea, caffeine and risk of breast cancer: A 22-year follow-up. Int. J. Cancer 2008, 122, 2071–2076. [Google Scholar] [CrossRef]
- Sisti, J.S.; Hankinson, S.E.; Caporaso, N.E.; Gu, F.; Tamimi, R.M.; Rosner, B.; Xu, X.; Ziegler, R.; Eliassen, A.H. Caffeine, coffee, and tea intake and urinary estrogens and estrogen metabolites in premenopausal women. Cancer Epidemiol. Biomark. Prev. 2015, 24, 1174–1183. [Google Scholar] [CrossRef]
- Kotsopoulos, J.; Eliassen, A.H.; Missmer, S.A.; Hankinson, S.E.; Tworoger, S.S. Relationship between caffeine intake and plasma sex hormone concentrations in premenopausal and postmenopausal women. Cancer 2009, 115, 2765–2774. [Google Scholar] [CrossRef]
- Halvorsen, B.L.; Carlsen, M.H.; Phillips, K.M.; Bøhn, S.K.; Holte, K.; Jacobs, D.R., Jr.; Blomhoff, R. Content of redox-active compounds (ie, antioxidants) in foods consumed in the United States. Am. J. Clin. Nutr. 2006, 84, 95–135. [Google Scholar] [CrossRef]
- Rahmioglu, N.; Heaton, J.; Clement, G.; Gill, R.; Surdulescu, G.; Zlobecka, K.; Hodgkiss, D.; Ma, Y.; Hider, R.C.; Smith, N.W.; et al. Genetic epidemiology of induced CYP3A4 activity. Pharmacogenet. Genom. 2011, 21, 642–651. [Google Scholar] [CrossRef] [PubMed]
- Gierach, G.L.; Freedman, N.D.; Andaya, A.; Hollenbeck, A.R.; Park, Y.; Schatzkin, A.; Brinton, L.A. Coffee intake and breast cancer risk in the NIH-AARP diet and health study cohort. Int. J. Cancer 2011, 131, 452–460. [Google Scholar] [CrossRef] [PubMed]
- Boggs, D.A.; Palmer, J.R.; Stampfer, M.J.; Spiegelman, D.; Adams-Campbell, L.L.; Rosenberg, L. Tea and coffee intake in relation to risk of breast cancer in the Black Women’s Health Study. Cancer Causes Control 2010, 21, 1941–1948. [Google Scholar] [CrossRef]
- Kotsopoulos, J.; Ghadirian, P.; El-Sohemy, A.; Lynch, H.T.; Snyder, C.; Daly, M.; Domchek, S.; Randall, S.; Karlan, B.; Zhang, P.; et al. The CYP1A2 genotype modifies the association between coffee consumption and breast cancer risk among BRCA1 mutation carriers. Cancer Epidemiol. Biomark. Prev. 2007, 16, 912–916. [Google Scholar] [CrossRef]
- Hunter, D.J.; Manson, J.E.; Stampfer, M.J.; Colditz, G.A.; Rosner, B.; Hennekens, C.H.; Speizer, F.E.; Willett, W.C. A prospective study of caffeine, coffee, tea, and breast cancer. Am. J. Epidemiol. 1992, 136, 1000–1001. [Google Scholar]
- Franceschi, S.; Favero, A.; La Vecchia, C.; Negri, E.; Dal Maso, L.; Salvini, S.; Decarli, A.; Giacosa, A. Influence of food groups and food diversity on breast cancer risk in Italy. Int. J. Cancer 1995, 63, 785–789. [Google Scholar] [CrossRef] [PubMed]
- Lubin, F.; Ron, E.; Wax, Y.; Modan, B. Coffee and methylxanthines and breast cancer: A case-control study. J. Natl. Cancer Inst. 1985, 74, 569–573. [Google Scholar]
- Matejcic, M.; de Batlle, J.; Ricci, C. Biomarkers of folate and vitamin B12 and breast cancer risk: Report from the EPIC cohort. Int. J. Cancer 2017, 140, 1246–1259. [Google Scholar] [CrossRef]
- Bassett, J.K.; Baglietto, L.; Hodge, A.M. Dietary intake of B vitamins and methionine and breast cancer risk. Cancer Causes Control 2013, 24, 1555–1563. [Google Scholar] [CrossRef]
- Lin, J.; Lee, I.M.; Cook, N.R. Plasma folate, vitamin B-6, vitamin B-12, and risk of breast cancer in women. Am. J. Clin. Nutr. 2008, 87, 734–743. [Google Scholar] [CrossRef]
- Wu, K.; Helzlsouer, K.J.; Comstock, G.W.; Hoffman, S.C.; Nadeau, M.R.; Selhub, J. A prospective study on folate, B12, and pyridoxal 5′-phosphate (B6) and breast cancer. Cancer Epidemiol. Biomark. Prev. 1999, 8, 209–217. [Google Scholar]
- Essén, A.; Santaolalla, A.; Garmo, H. Baseline serum folate, vitamin B12 and the risk of prostate and breast cancer using data from the Swedish AMORIS cohort. Cancer Causes Control 2019, 30, 603–615. [Google Scholar]
- Zhang, S.M.; Willett, W.C.; Selhub, J. Plasma folate, vitamin B6, vitamin B12, homocysteine, and risk of breast cancer. J. Natl. Cancer Inst. 2003, 95, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Chen, J. One-carbon metabolism and breast cancer: An epidemiological perspective. J. Genet. Genom. 2009, 36, 203–214. [Google Scholar] [CrossRef]
- Choi, S.W.; Mason, J.B. Folate status: Effects on pathways of colorectal carcinogenesis. J. Nutr. 2002, 132, 2413S–2418S. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.B. Biomarkers of nutrient exposure and status in one-carbon (methyl) metabolism. J. Nutr. 2003, 133, 941S–947S. [Google Scholar] [CrossRef]
- Davis, C.D.; Uthus, E.O. DNA methylation, cancer susceptibility, and nutrient interactions. Exp. Biol. Med. 2004, 229, 988–995. [Google Scholar] [CrossRef]
- Widschwendter, M.; Jones, P.A. DNA methylation and breast carcinogenesis. Oncogene. 2002, 21, 5462–5482. [Google Scholar] [CrossRef]
- Gonzalgo, M.L.; Jones, P.A. Mutagenic and epigenetic effects of DNA methylation. Mutat. Res. 1997, 386, 107–118. [Google Scholar] [CrossRef]
| Descriptive Variable | Women with Self-Reported Breast Cancer (n = 286) | Women without Self-Reported Breast Cancer (n = 1144) | p-Value |
|---|---|---|---|
| Mean age (± SD) | 68.46, (0.74) | 63.19, (0.36) | <0.001 |
| Parity, mean (± SD) | 2.49, (0.17) | 2.70, (0.07) | 0.15 |
| Age at first menarche, mean (± SD) | 12.62, (0.13) | 12.89, (0.06) | 0.06 |
| Ethnicity, n, (%) | |||
| Non-Hispanic White | 203, (88%) | 595, (77%) | <0.001 |
| Non-Hispanic Black | 44, (7.2%) | 219, (10.9%) | |
| Hispanic | 32, (2.6%) | 294, (8.1%) | |
| Unknown/Other | 5, (2.3%) | 36, (4.5%) | |
| BMI (kg/m2) 1, mean (± SD) | 28.89, (0.55) | 29.38, (0.33) | 0.43 |
| Descriptive Variable Mean (SD) 1 | Women with Self-Reported Breast Cancer (n = 286) | Women without Self-Reported Breast Cancer (n = 1144) | 95% CI (Difference of Means) 1 | p-Value |
|---|---|---|---|---|
| Energy (Kcal) | 1638 (43.60) | 1648 (28.23) | (−109.59, 91.01) | 0.46 |
| Carbohydrate (g) | 205.38 (6.46) | 204.75 (3.77) | (−14.34, 15.57) | 0.36 |
| Carbohydrate, % energy | 50.43 (0.86) | 50.44 (0.48) | (−2.00, 1.98) | 0.80 |
| Protein (g) | 64.20 (2.38) | 65.45 (1.31) | (−6.46, 3.97) | 0.68 |
| Protein, % energy | 15.89 (0.30) | 16.10 (0.18) | (−0.944, 0.520) | 0.72 |
| Total Fat (g) | 61.86 (2.05) | 63.99 (1.47) | (−6.83, 2.64) | 0.50 |
| Fat, % energy | 33.57 (0.60) | 33.97 (0.39) | (−1.88, 1.06) | 0.78 |
| Cholesterol (mg) | 213.80 (10.56) | 226.50 (7.70) | (−40.46, 15.05) | 0.40 |
| Fiber (g) | 15.23 (0.72) | 14.85 (0.34) | (−1.25, 2.02) | 0.38 |
| Folate (μg) | 353.49 (13.81) | 347.07 (8.40) | (−26.49, 39.34) | 0.38 |
| Vitamin B12 (μg) | 5.02 (0.75) | 4.17 (0.15) | (−0.632, 2.34) | 0.08 |
| Vitamin B6 (mg) | 1.61 (0.07) | 1.60 (0.04) | (−0.16, 0.19) | 0.45 |
| Thiamin (mg) | 1.36 (0.07) | 1.39 (0.04) | (−0.18, 0.15) | 0.73 |
| Riboflavin (mg) | 1.89 (0.07) | 1.88 (0.04) | (−0.164, 0.16) | 0.45 |
| Calcium (mg) | 772.74 (27.24) | 780.44 (20.44) | (−67.42, 52.03) | 0.21 |
| Phosphorous (mg) | 1082 (35.87) | 1096 (20.13) | (−91.45, 63.47) | 0.49 |
| Magnesium (mg) | 253.27 (9.13) | 256.33 (4.79) | (−24.00, 17.89) | 0.65 |
| Iron (mg) | 13.24 (0.56) | 12.84 (0.29) | (−0.93, 1.72) | 0.25 |
| Vitamin A (IU) | 685.55 (75.15) | 648.52 (18.85) | (−116.40, 190.45) | 0.19 |
| Vitamin C (mg) | 87.68 (4.14) | 92.04 (4.81) | (−16.66, 7.94) | 0.28 |
| Vitamin E (mg) | 6.66 (0.40) | 6.67 (0.19) | (−0.93, 0.91) | 0.52 |
| Zinc (mg) | 9.76 (0.35) | 9.61 (0.24) | (−0.61, 0.92) | 0.24 |
| Sodium (mg) | 2665 (84.90) | 2768 (59.30) | (−289.68, 81.87) | 0.80 |
| Potassium (mg) | 2452 (61.65) | 2476 (39.86) | (−158.56, 109.05) | 0.38 |
| Caffeine (mg) | 154.56 (14.42) | 174.94 (11.62) | (−57.97, 17.21) | 0.38 |
| Alcohol (g) | 5.31 (1.01) | 3.17 (0.49) | (0.08, 4.21) 2 | 0.19 |
| Variables | Coefficients (Bootstrap SE) |
|---|---|
| Well-established Variables | |
| Age (years) | 0.83 (0.41) |
| Parity (# live births) | −0.05 (0.03) |
| Age at first menstrual cycle | 0 |
| Alcohol (g) | 0.03 (0.02) |
| Other Variables | |
| Caffeine (mg) | −0.01 (0.02) |
| Mexican/Hispanic | 0 |
| Non-Hispanic Black | 0 |
| Other | 0 |
| Dietary Variables | |
| Energy (Kcal) | 0 |
| Carbohydrate, % energy | 0 |
| Protein, % energy | 0 |
| Fat, % energy | 0 |
| Cholesterol (mg) | 0 |
| Fiber (g) | 0 |
| Folate (μg) | 0 |
| Vitamin B12 (μg) | 0.07 (0.05) |
| Vitamin B6 (mg) | 0 |
| Thiamin (Vitamin B1) (mg) | 0 |
| Riboflavin (Vitamin B2) (mg) | 0 |
| Calcium (mg) | 0 |
| Phosphorous (mg) | 0 |
| Magnesium (mg) | 0 |
| Iron (mg) | 0 |
| Vitamin A (RE) | 0 |
| Vitamin C (mg) | 0 |
| Vitamin E (mg) | 0 |
| Zinc (mg) | 0 |
| Sodium (mg) | 0 |
| Potassium (mg) | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
McEligot, A.J.; Poynor, V.; Sharma, R.; Panangadan, A. Logistic LASSO Regression for Dietary Intakes and Breast Cancer. Nutrients 2020, 12, 2652. https://doi.org/10.3390/nu12092652
McEligot AJ, Poynor V, Sharma R, Panangadan A. Logistic LASSO Regression for Dietary Intakes and Breast Cancer. Nutrients. 2020; 12(9):2652. https://doi.org/10.3390/nu12092652
Chicago/Turabian StyleMcEligot, Archana J., Valerie Poynor, Rishabh Sharma, and Anand Panangadan. 2020. "Logistic LASSO Regression for Dietary Intakes and Breast Cancer" Nutrients 12, no. 9: 2652. https://doi.org/10.3390/nu12092652
APA StyleMcEligot, A. J., Poynor, V., Sharma, R., & Panangadan, A. (2020). Logistic LASSO Regression for Dietary Intakes and Breast Cancer. Nutrients, 12(9), 2652. https://doi.org/10.3390/nu12092652





