Intermittent Fasting Enhanced the Cognitive Function in Older Adults with Mild Cognitive Impairment by Inducing Biochemical and Metabolic changes: A 3-Year Progressive Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Size Calculation
2.2. Subject Recruitment
2.3. Intermittent Fasting Grouping
2.4. Socio-Demographic Data
2.5. Body Composition and Blood Pressure Measurement
2.6. Assessment of Cognitive Function
2.7. Cognitive Aging Grouping
2.8. Biochemical Analyses
2.9. DNA Damage Analysis
2.10. Malondialdehyde Analysis
2.11. Superoxide Dismutase Analysis
2.12. Inflammatory Marker and Insulin Analysis
2.13. 1H Nuclear Magnetic Resonance Spectroscopic Analysis
2.14. Metabolomics Profiling and Identification
2.15. Statistical Analysis
3. Results
3.1. Socio-Demographic Data
3.2. Anthropometry Analysis and Blood Pressure
3.3. Biochemical Profiles
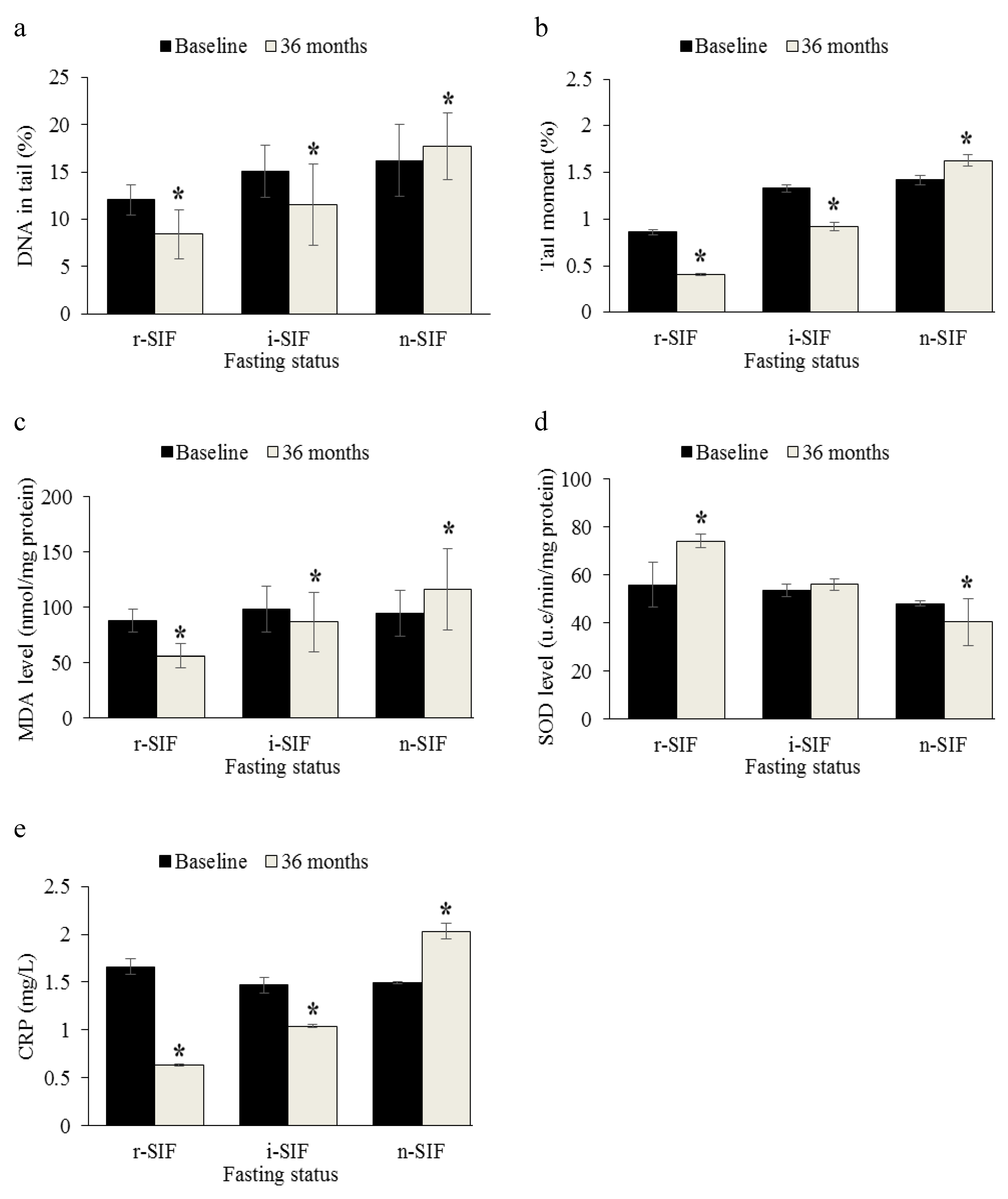
3.4. Analysis of DNA Damage
3.5. Analysis of Oxidative Stress Markers
3.6. Analysis of Inflammatory Marker
3.7. Cognitive Performance
3.8. Metabolite Identification
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gandhi, S.; Abramov, A.Y. Mechanism of Oxidative Stress in Neurodegeneration. Oxidative Med. Cell. Longev. 2012, 2012, 428010. [Google Scholar] [CrossRef]
- Nematy, M.; Alinezhad-Namaghi, M.; Rashed, M.M.; Mozhdehifard, M.; Sajjadi, S.S.; Akhlaghi, S.; Sabery, M.; Mohajeri, S.A.; Shalaey, N.; Moohebati, M.; et al. Effects of Ramadan fasting on cardiovascular risk factors: A prospective observational study. Nutr. J. 2012, 11, 69. [Google Scholar] [CrossRef]
- Reddy, P.H.; Beal, M.F. Amyloid beta, mitochondrial dysfunction and synaptic damage: Implications for cognitive decline in aging and Alzheimer’s disease. Trends Mol. Med. 2008, 14, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.H. Amyloid beta, mitochondrial structural and functional dynamics in Alzheimer’s disease. Exp. Neurol. 2009, 218, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.H.; Reddy, T.P. Mitochondria as a therapeutic target for aging and neurodegenerative diseases. Curr. Alzheimer Res. 2011, 8, 393–409. [Google Scholar] [CrossRef] [PubMed]
- Solfrizzi, V.; Scafato, E.; Capurso, C.; D’Introno, A.; Colacicco, A.M.; Frisardi, V.; Vendemiale, G.; Baldereschi, M.; Crepaldi, G.; Di Carlo, A.; et al. Metabolic syndrome, mild cognitive impairment, and progression to dementia. The Italian Longitudinal Study on Aging. Neurobiol. Aging 2011, 32, 1932–1941. [Google Scholar] [CrossRef] [PubMed]
- Sohal, R.S.; Weindruch, R. Oxidative stress, caloric restriction, and aging. Science 1996, 273, 59–63. [Google Scholar] [CrossRef]
- van Praag, H.; Fleshner, M.; Schwartz, M.W.; Mattson, M.P. Exercise, energy intake, glucose homeostasis, and the brain. J. Neurosci. Off. J. Soc. Neurosci. 2014, 34, 15139–15149. [Google Scholar] [CrossRef]
- Caramoci, A.; Mitoiu, B.; Pop, M.; Mazilu, V.; Vasilescu, M.; Ionescu, A.M.; Eugenia, R. Is intermittent fasting a scientifically-based dietary method? Med. Sport. J. Rom. Sports Med. Soc. 2016, 12, 2747. [Google Scholar]
- Anton, S.; Leeuwenburgh, C. Fasting or caloric restriction for healthy aging. Exp. Gerontol. 2013, 48, 1003–1005. [Google Scholar] [CrossRef]
- Harvie, M.; Wright, C.; Pegington, M.; McMullan, D.; Mitchell, E.; Martin, B.; Cutler, R.G.; Evans, G.; Whiteside, S.; Maudsley, S.; et al. The effect of intermittent energy and carbohydrate restriction v. daily energy restriction on weight loss and metabolic disease risk markers in overweight women. Br. J. Nutr. 2013, 110, 1534–1547. [Google Scholar] [CrossRef]
- Teng, N.I.; Shahar, S.; Rajab, N.F.; Manaf, Z.A.; Johari, M.H.; Ngah, W.Z. Improvement of metabolic parameters in healthy older adult men following a fasting calorie restriction intervention. Aging Male 2013, 16, 177–183. [Google Scholar] [CrossRef] [PubMed]
- Meramat, A.; Sharif, R.; Shahar, S.; Rajab, N.F. Relationship of Sunnah Fasting and DNA Damage Among Mild Cognitive Impairment (MCI) Older Adults. Malays. J. Health Sci. 2018, 16, 213. [Google Scholar] [CrossRef]
- Vanoh, D.; Shahar, S.; Din, N.C.; Omar, A.; Vyrn, C.A.; Razali, R.; Ibrahim, R.; Hamid, T.A. Predictors of poor cognitive status among older Malaysian adults: Baseline findings from the LRGS TUA cohort study. Aging Clin. Exp. Res. 2017, 29, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Lau, H.; Shahar, S.; Hussin, N.; Kamarudin, M.Z.; Hamid, T.A.; Mukari, S.Z.; Rajab, N.F.; Din, N.C.; Omar, A.; Singh, D.K.; et al. Methodology approaches and challenges in population-based longitudinal study of a neuroprotective model for healthy longevity. Geriatr. Gerontol. Int. 2019, 19, 233–239. [Google Scholar] [CrossRef]
- Petersen, R.C.; Caracciolo, B.; Brayne, C.; Gauthier, S.; Jelic, V.; Fratiglioni, L. Mild cognitive impairment: A concept in evolution. J. Intern. Med. 2014, 275, 214–228. [Google Scholar] [CrossRef]
- Shahar, S.; Omar, A.; Vanoh, D.; Hamid, T.A.; Mukari, S.Z.; Din, N.C.; Rajab, N.F.; Mohammed, Z.; Ibrahim, R.; Loo, W.H.; et al. Approaches in methodology for population-based longitudinal study on neuroprotective model for healthy longevity (TUA) among Malaysian Older Adults. Aging Clin. Exp. Res. 2016, 28, 1089–1104. [Google Scholar] [CrossRef]
- Hamid, T.A.; Momtaz, Y.A.; Ibrahim, R. Predictors and prevalence of successful aging among older Malaysians. Gerontology 2012, 58, 366–370. [Google Scholar] [CrossRef]
- Vanoh, D.; Shahar, S.; Rosdinom, R.; Din, N.C.; Yahya, H.M.; Omar, A. Development of TUA-WELLNESS screening tool for screening risk of mild cognitive impairment among community-dwelling older adults. Clin. Interv. Aging 2016, 11, 579–587. [Google Scholar] [CrossRef]
- Meramat, A.; Rajab, N.F.; Shahar, S.; Sharif, R.A. DNA Damage, Copper and Lead Associates with Cognitive Function among Older Adults. J. Nutr. Health Aging 2017, 21, 539–545. [Google Scholar] [CrossRef]
- Stocks, J.; Gutteridge, J.M.; Sharp, R.J.; Dormandy, T.L. Assay using brain homogenate for measuring the antioxidant activity of biological fluids. Clin. Sci. Mol. Med. 1974, 47, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Beckonert, O.; Keun, H.C.; Ebbels, T.M.; Bundy, J.; Holmes, E.; Lindon, J.C.; Nicholson, J.K. Metabolic profiling, metabolomic and metabonomic procedures for NMR spectroscopy of urine, plasma, serum and tissue extracts. Nat. Protoc. 2007, 2, 2692–2703. [Google Scholar] [CrossRef] [PubMed]
- Verheijden, M.W.; Bakx, J.C.; van Weel, C.; Koelen, M.A.; van Staveren, W.A. Role of social support in lifestyle-focused weight management interventions. Eur. J. Clin. Nutr. 2005, 59 (Suppl. 1), S179–S186. [Google Scholar] [CrossRef]
- Morabia, A.; Curtin, F.; Bernstein, M.S. Effects of smoking and smoking cessation on dietary habits of a Swiss urban population. Eur. J. Clin. Nutr. 1999, 53, 239–243. [Google Scholar] [CrossRef] [PubMed]
- Danielsson, T.; Rössner, S.; Westin, A. Open randomised trial of intermittent very low energy diet together with nicotine gum for stopping smoking in women who gained weight in previous attempts to quit. BMJ 1999, 319, 490–493, discussion 494. [Google Scholar] [CrossRef]
- Barnosky, A.R.; Hoddy, K.K.; Unterman, T.G.; Varady, K.A. Intermittent fasting vs. daily calorie restriction for type 2 diabetes prevention: A review of human findings. Transl. Res. J. Lab. Clin. Med. 2014, 164, 302–311. [Google Scholar] [CrossRef]
- Dye, L.; Boyle, N.B.; Champ, C.; Lawton, C. The relationship between obesity and cognitive health and decline. Proc. Nutr. Soc. 2017, 76, 443–454. [Google Scholar] [CrossRef]
- Nguyen, J.C.; Killcross, A.S.; Jenkins, T.A. Obesity and cognitive decline: Role of inflammation and vascular changes. Front Neurosci. 2014, 8, 375. [Google Scholar] [CrossRef]
- Gustafson, D.; Lissner, L.; Bengtsson, C.; Björkelund, C.; Skoog, I. A 24-year follow-up of body mass index and cerebral atrophy. Neurology 2004, 63, 1876–1881. [Google Scholar] [CrossRef]
- Volkow, N.D.; Wang, G.J.; Telang, F.; Fowler, J.S.; Goldstein, R.Z.; Alia-Klein, N.; Logan, J.; Wong, C.; Thanos, P.K.; Ma, Y.; et al. Inverse association between BMI and prefrontal metabolic activity in healthy adults. Obesity 2009, 17, 60–65. [Google Scholar] [CrossRef]
- Gunstad, J.; Paul, R.H.; Cohen, R.A.; Tate, D.F.; Spitznagel, M.B.; Grieve, S.; Gordon, E. Relationship between body mass index and brain volume in healthy adults. Int. J. Neurosci. 2008, 118, 1582–1593. [Google Scholar] [CrossRef] [PubMed]
- Mertens, I.L.; Van Gaal, L.F. Overweight, obesity, and blood pressure: The effects of modest weight reduction. Obes. Res. 2000, 8, 270–278. [Google Scholar] [CrossRef] [PubMed]
- Beleslin, B.; Cirić, J.; Zarković, M.; Vujović, S.; Trbojević, B.; Drezgić, M. The effects of three-week fasting diet on blood pressure, lipid profile and glucoregulation in extremely obese patients. Srp. Arh. Celok. Lek. 2007, 135, 440–446. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, S.; Higashi, Y.; Nakagawa, K.; Kimura, M.; Noma, K.; Sasaki, S.; Hara, K.; Matsuura, H.; Goto, C.; Oshima, T.; et al. A low-calorie diet improves endothelium-dependent vasodilation in obese patients with essential hypertension. Am. J. Hypertens. 2002, 15, 302–309. [Google Scholar] [CrossRef]
- Budge, M.M.; de Jager, C.; Hogervorst, E.; Smith, A.D. Total plasma homocysteine, age, systolic blood pressure, and cognitive performance in older people. J. Am. Geriatr. Soc. 2002, 50, 2014–2018. [Google Scholar] [CrossRef] [PubMed]
- Kuo, H.-K.; Sorond, F.; Iloputaife, I.; Gagnon, M.; Milberg, W.; Lipsitz, L.A. Effect of Blood Pressure on Cognitive Functions in Elderly Persons. J. Gerontol. Ser. A 2004, 59, 1191–1194. [Google Scholar] [CrossRef]
- Reitz, C.; Luchsinger, J.A. Relation of Blood Pressure to Cognitive Impairment and Dementia. Curr. Hypertens. Rev. 2007, 3, 166–176. [Google Scholar] [CrossRef]
- Faris, M.A.; Hussein, R.N.; Al-Kurd, R.A.; Al-Fararjeh, M.A.; Bustanji, Y.K.; Mohammad, M.K. Impact of ramadan intermittent fasting on oxidative stress measured by urinary 15-f(2t)-isoprostane. J. Nutr. Metab. 2012, 2012, 802924. [Google Scholar] [CrossRef]
- Mansi, K.M.S. Study the Effects of Ramadan Fasting on the Serum Glucose and Lipid Profile among Healthy Jordanian Students. Am. J. Appl. Sci. 2007, 4. [Google Scholar] [CrossRef]
- Al-Hourani, H.M.; Atoum, M.F. Body composition, nutrient intake and physical activity patterns in young women during Ramadan. Singap. Med. J. 2007, 48, 906–910. [Google Scholar]
- Barzilai, A.; Yamamoto, K. DNA damage responses to oxidative stress. DNA Repair 2004, 3, 1109–1115. [Google Scholar] [CrossRef]
- Chen, J.H.; Hales, C.N.; Ozanne, S.E. DNA damage, cellular senescence and organismal ageing: Causal or correlative? Nucleic Acids Res. 2007, 35, 7417–7428. [Google Scholar] [CrossRef] [PubMed]
- Soares, J.P.; Cortinhas, A.; Bento, T.; Leitão, J.C.; Collins, A.R.; Gaivão, I.; Mota, M.P. Aging and DNA damage in humans: A meta-analysis study. Aging 2014, 6, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Deary, I.J.; Corley, J.; Gow, A.J.; Harris, S.E.; Houlihan, L.M.; Marioni, R.E.; Penke, L.; Rafnsson, S.B.; Starr, J.M. Age-associated cognitive decline. Br. Med. Bull. 2009, 92, 135–152. [Google Scholar] [CrossRef] [PubMed]
- Heilbronn, L.K.; de Jonge, L.; Frisard, M.I.; DeLany, J.P.; Larson-Meyer, D.E.; Rood, J.; Nguyen, T.; Martin, C.K.; Volaufova, J.; Most, M.M.; et al. Effect of 6-month calorie restriction on biomarkers of longevity, metabolic adaptation, and oxidative stress in overweight individuals: A randomized controlled trial. JAMA 2006, 295, 1539–1548. [Google Scholar] [CrossRef] [PubMed]
- Sawa, K.; Uematsu, T.; Korenaga, Y.; Hirasawa, R.; Kikuchi, M.; Murata, K.; Zhang, J.; Gai, X.; Sakamoto, K.; Koyama, T.; et al. Krebs Cycle Intermediates Protective against Oxidative Stress by Modulating the Level of Reactive Oxygen Species in Neuronal HT22 Cells. Antioxidants 2017, 6, 21. [Google Scholar] [CrossRef]
- Vermeij, W.P.; Dollé, M.E.T.; Reiling, E.; Jaarsma, D.; Payan-Gomez, C.; Bombardieri, C.R.; Wu, H.; Roks, A.J.M.; Botter, S.M.; van der Eerden, B.C.; et al. Restricted diet delays accelerated ageing and genomic stress in DNA-repair-deficient mice. Nature 2016, 537, 427–431. [Google Scholar] [CrossRef]
- Ott, B.; Skurk, T.; Hastreiter, L.; Lagkouvardos, I.; Fischer, S.; Büttner, J.; Kellerer, T.; Clavel, T.; Rychlik, M.; Haller, D.; et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci. Rep. 2017, 7, 1–10. [Google Scholar] [CrossRef]
- Marinac, C.R.; Sears, D.D.; Natarajan, L.; Gallo, L.C.; Breen, C.I.; Patterson, R.E. Frequency and Circadian Timing of Eating May Influence Biomarkers of Inflammation and Insulin Resistance Associated with Breast Cancer Risk. PLoS ONE 2015, 10, e0136240. [Google Scholar] [CrossRef]
- Brooks, G.C.; Blaha, M.J.; Blumenthal, R.S. Relation of C-reactive protein to abdominal adiposity. Am. J. Cardiol. 2010, 106, 56–61. [Google Scholar] [CrossRef]
- Dionne, D.A.; Skovsø, S.; Templeman, N.M.; Clee, S.M.; Johnson, J.D. Caloric Restriction Paradoxically Increases Adiposity in Mice With Genetically Reduced Insulin. Endocrinology 2016, 157, 2724–2734. [Google Scholar] [CrossRef] [PubMed]
- Larson-Meyer, D.E.; Heilbronn, L.K.; Redman, L.M.; Newcomer, B.R.; Frisard, M.I.; Anton, S.; Smith, S.R.; Alfonso, A.; Ravussin, E. Effect of calorie restriction with or without exercise on insulin sensitivity, beta-cell function, fat cell size, and ectopic lipid in overweight subjects. Diabetes Care 2006, 29, 1337–1344. [Google Scholar] [CrossRef] [PubMed]
- Moloney, A.M.; Griffin, R.J.; Timmons, S.; O’Connor, R.; Ravid, R.; O’Neill, C. Defects in IGF-1 receptor, insulin receptor and IRS-1/2 in Alzheimer’s disease indicate possible resistance to IGF-1 and insulin signalling. Neurobiol. Aging 2010, 31, 224–243. [Google Scholar] [CrossRef] [PubMed]
- Burns, J.M.; Honea, R.A.; Vidoni, E.D.; Hutfles, L.J.; Brooks, W.M.; Swerdlow, R.H. Insulin is differentially related to cognitive decline and atrophy in Alzheimer’s disease and aging. Biochim. Et Biophys. Acta 2012, 1822, 333–339. [Google Scholar] [CrossRef] [PubMed]
- Kim, B.; Feldman, E.L. Insulin resistance in the nervous system. Trends Endocrinol. Metab. 2012, 23, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Blázquez, E.; Velázquez, E.; Hurtado-Carneiro, V.; Ruiz-Albusac, J.M. Insulin in the brain: Its pathophysiological implications for States related with central insulin resistance, type 2 diabetes and Alzheimer’s disease. Front Endocrinol. 2014, 5, 161. [Google Scholar] [CrossRef]
- Bloomgarden, Z.T. Insulin Resistance Concepts. Diabetes Care 2007, 30, 1320. [Google Scholar] [CrossRef]
- Grajower, M.M.; Horne, B.D. Clinical Management of Intermittent Fasting in Patients with Diabetes Mellitus. Nutrients 2019, 11, 873. [Google Scholar] [CrossRef]
- Witte, A.V.; Fobker, M.; Gellner, R.; Knecht, S.; Flöel, A. Caloric restriction improves memory in elderly humans. Proc. Natl. Acad. Sci. USA 2009, 106, 1255–1260. [Google Scholar] [CrossRef]
- Horie, N.C.; Serrao, V.T.; Simon, S.S.; Gascon, M.R.P.; dos Santos, A.X.; Zambone, M.A.; del Bigio de Freitas, M.M.; Cunha-Neto, E.; Marques, E.L.; Halpern, A.; et al. Cognitive effects of intentional weight loss in elderly obese individuals with mild cognitive impairment. J. Clin. Endocrinol. Metab. 2016, 101, 1104–1112. [Google Scholar] [CrossRef]
- Alsharidah, A.M.; Murtaza, G.; Alsharidah, M.M.; Bashir, S. Fasting in Ramadan affects cognitive and physiological function in normal subjects (pilot study). Neurosci. Med. 2016, 7, 60–65. [Google Scholar] [CrossRef]
- Tian, H.H.; Aziz, A.R.; Png, W.; Wahid, M.F.; Yeo, D.; Constance Png, A.L. Effects of fasting during ramadan month on cognitive function in muslim athletes. Asian J. Sports Med. 2011, 2, 145–153. [Google Scholar] [CrossRef] [PubMed]
- Pinu, F.R.; Goldansaz, S.A.; Jaine, J. Translational Metabolomics: Current Challenges and Future Opportunities. Metabolites 2019, 9, 108. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, M.; Suzuki, M.; Sato, K.; Dohi, S.; Sato, T.; Matsuura, A.; Hiraide, A. Effect of beta-hydroxybutyrate, a cerebral function improving agent, on cerebral hypoxia, anoxia and ischemia in mice and rats. Jpn. J. Pharmacol. 2001, 87, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Maalouf, M.; Rho, J.M.; Mattson, M.P. The neuroprotective properties of calorie restriction, the ketogenic diet, and ketone bodies. Brain Res. Rev. 2009, 59, 293–315. [Google Scholar] [CrossRef] [PubMed]
- Kashiwaya, Y.; Bergman, C.; Lee, J.H.; Wan, R.; King, M.T.; Mughal, M.R.; Okun, E.; Clarke, K.; Mattson, M.P.; Veech, R.L. A ketone ester diet exhibits anxiolytic and cognition-sparing properties, and lessens amyloid and tau pathologies in a mouse model of Alzheimer’s disease. Neurobiol. Aging 2013, 34, 1530–1539. [Google Scholar] [CrossRef]
- Newman, J.C.; Verdin, E. Ketone bodies as signaling metabolites. Trends Endocrinol. Metab. 2014, 25, 42–52. [Google Scholar] [CrossRef]
- Mattson, M.P. Lifelong brain health is a lifelong challenge: From evolutionary principles to empirical evidence. Ageing Res. Rev. 2015, 20, 37–45. [Google Scholar] [CrossRef]
- Rui, L. Energy metabolism in the liver. Compr. Physiol. 2014, 4, 177–197. [Google Scholar] [CrossRef]
- Contrusciere, V.; Paradisi, S.; Matteucci, A.; Malchiodi-Albedi, F. Branched-chain amino acids induce neurotoxicity in rat cortical cultures. Neurotox. Res. 2010, 17, 392–398. [Google Scholar] [CrossRef]
- Kaufman, S. An evaluation of the possible neurotoxicity of metabolites of phenylalanine. J. Pediatr. 1989, 114, 895–900. [Google Scholar] [CrossRef]
- Preissler, T.; Bristot, I.J.; Costa, B.M.; Fernandes, E.K.; Rieger, E.; Bortoluzzi, V.T.; de Franceschi, I.D.; Dutra-Filho, C.S.; Moreira, J.C.; Wannmacher, C.M. Phenylalanine induces oxidative stress and decreases the viability of rat astrocytes: Possible relevance for the pathophysiology of neurodegeneration in phenylketonuria. Metab. Brain Dis. 2016, 31, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Pocernich, C.B.; Butterfield, D.A. Elevation of glutathione as a therapeutic strategy in Alzheimer disease. Biochim. Et Biophys. Acta 2012, 1822, 625–630. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.M.; Murch, S.J.; Ball, R.O. Proline is synthesized from glutamate during intragastric infusion but not during intravenous infusion in neonatal piglets. J. Nutr. 1996, 126, 878–886. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, Y.; Iwashima, A.; Yamada, E.; Yamada, R. Enzymological evidence for the indispensability of small intestine in the synthesis of arginine from glutamate. II. N-acetylglutamate synthase. Arch. Biochem. Biophys. 1991, 291, 9–14. [Google Scholar] [CrossRef]
- Lewerenz, J.; Maher, P. Chronic Glutamate Toxicity in Neurodegenerative Diseases-What is the Evidence? Front Neurosci. 2015, 9, 469. [Google Scholar] [CrossRef]
- Gao, X.; Lai, C.Q.; Scott, T.; Shen, J.; Cai, T.; Ordovas, J.M.; Tucker, K.L. Urinary 8-hydroxy-2-deoxyguanosine and cognitive function in Puerto Rican adults. Am. J. Epidemiol. 2010, 172, 271–278. [Google Scholar] [CrossRef]
- Itoh, T.; Imano, M.; Nishida, S.; Tsubaki, M.; Mizuguchi, N.; Hashimoto, S.; Ito, A.; Satou, T. Increased apoptotic neuronal cell death and cognitive impairment at early phase after traumatic brain injury in aged rats. Brain Struct Funct. 2013, 218, 209–220. [Google Scholar] [CrossRef]
| Characteristics | r-IF (n = 37) Number (%) or Mean ± SD | i-IF (n = 35) Number (%) or Mean ± SD | n-IF (n = 27) Number (%) or Mean ± SD | p-Value | |
|---|---|---|---|---|---|
| Age (mean) | 68.7 ± 4.6 | 67.9 ± 5.6 | 69.1 ± 5.0 | 0.65 | |
| Gender | Male | 23 (62.2) | 15 (42.9) | 15 (55.6) | 0.25 |
| Female | 14 (37.8) | 20 (57.1) | 12 (44.4) | ||
| Marital status | Married | 29 (78.4) | 22 (62.9) | 11 (40.7) | 0.01 * |
| Single/divorced/widowed | 8 (21.6) | 13 (37.1) | 16 (59.3) | ||
| Years of education (year) | ≤6 years | 24 (64.9) | 30 (85.7) | 24 (88.9) | 0.03 * |
| >6 years | 13 (35.1) | 5 (14.3) | 3 (11.1) | ||
| Living status | Alone | 1 (2.7) | 4 (11.4) | 6 (22.2) | 0.05 |
| With others | 36 (97.3) | 31 (88.6) | 21 (77.8) | ||
| Smoking habits | Active smoker | 6 (16.2) | 15 (42.9) | 15 (55.6) | 0.01 * |
| Ex-smoker | 10 (27.0) | 5 (14.2) | 1 (3.7) | ||
| Non-smoker | 21 (56.8) | 15 (42.9) | 11 (40.7) |
| Parameter | Fasting Status | Baseline | 36 Months | p-Value (Partial Eta Square) | ||||
|---|---|---|---|---|---|---|---|---|
| Mean ± SD | 95% CI | Mean ± SD | 95% CI | Effect of Time | Effect of Group | Interaction Effect | ||
| Anthropometry | ||||||||
| Weight (kg) | r-IF (n = 37) | 52.84 ± 1.47 | 49.92, 55.75 | 49.19 ± 1.48 ###,a,b | 46.26, 52.12 | 0.12 (0.01) | <0.001 (0.34) * | 0.03 (0.47) * |
| i-IF (n = 35) | 61.47 ± 1.51 | 58.47, 64.47 | 61.11 ± 1.52 a | 58.09, 64.12 | ||||
| n-IF (n = 27) | 63.99 ± 1.72 | 60.57, 67.40 | 66.27 ± 1.73 ###,b | 62.84, 69.71 | ||||
| BMI (kg/m2) | r-IF (n = 37) | 24.17 ± 0.53 | 23.11, 25.24 | 22.64 ± 0.52 ###,a,b | 21.60, 23.68 | 0.02 (0.09) * | <0.001 (0.59) * | <0.001 (0.31) * |
| i-IF (n = 35) | 26.14 ± 0.50 | 25.15, 27.14 | 27.35 ± 0.48 ##,a,c | 26.39, 28.33 | ||||
| n-IF (n = 27) | 30.03 ± 0.63 | 28.78, 31.27 | 33.56 ± 0.61 ###,b,c | 32.34, 34.78 | ||||
| WC (cm) | r-IF (n = 37) | 86.63 ± 1.32 | 84.02, 89.24 | 81.53 ± 1.57 ###,b | 78.42, 84.65 | 0.02 (0.06) * | <0.001 (0.93) * | 0.03 (0.04) * |
| i-IF (n = 35) | 87.46 ± 1.35 | 84.78, 90.15 | 87.81 ± 1.61 c | 83.61, 90.02 | ||||
| n-IF (n = 27) | 92.85 ± 1.54 | 89.79, 95.91 | 95.80 ± 1.84 ###,b,c | 92.16, 99.45 | ||||
| HC (cm) | r-IF (n = 37) | 90.59 ± 1.09 | 88.43, 92.75 | 90.22 ± 0.97 b | 88.29, 92.15 | <0.001 (0.12) * | 0.06 (0.05) | <0.001 (0.17) * |
| i-IF (n = 35) | 90.87 ± 1.12 | 88.65, 93.09 | 92.47 ± 1.29 # | 89.17, 94.33 | ||||
| n-IF (n = 27) | 91.75 ± 1.29 | 89.17, 94.33 | 96.35 ± 1.16 ###,b | 94.05, 98.65 | ||||
| Blood pressure | ||||||||
| Systolic (mmHg) | r-IF (n = 37) | 139.35 ± 2.83 | 133.74, 144.96 | 132.31 ± 2.49 ###,a,b | 127.36, 137.27 | 0.74 (0.001) | 0.01 (0.09) * | <0.001 (0.21) * |
| i-IF (n = 35) | 142.65 ± 2.91 | 136.89, 148.43 | 145.46 ± 2.57 a | 140.36, 150.55 | ||||
| n-IF (n = 27) | 143.48 ± 2.57 | 136.91, 150.05 | 151.17 ± 2.92 ##,b | 145.37, 156.97 | ||||
| Diastolic (mmHg) | r-IF (n = 37) | 76.76 ± 1.75 | 73.29, 80.22 | 72.97 ± 1.70 ##,b | 69.59, 76.35 | 0.38 (0.01) | 0.13 (0.04) | 0.02 (0.08) * |
| i-IF (n = 35) | 78.06 ± 1.79 | 74.49, 81.62 | 76.98 ± 1.75 | 73.52, 80.46 | ||||
| n-IF (n = 27) | 77.93 ± 2.05 | 73.87, 81.99 | 81.24 ± 1.99 #,b | 77.29, 85.19 | ||||
| Biochemical profile | ||||||||
| FBS (mmol/L) | r-IF (n = 37) | 5.49 ± 0.72 | 5.21,5.77 | 5.12 ± 0.60 ## | 4.63, 5.62 | 0.15 (0.02) | <0.001 (0.17) * | 0.004 (0.11) * |
| i-IF (n = 35) | 5.97 ± 1.11 | 5.61, 6.34 | 6.12 ± 1.43 c | 5.56, 6.67 | ||||
| n-IF (n = 27) | 5.76 ± 0.94 | 5.44, 6.09 | 6.92 ± 2.40 #,c | 6.64, 7.56 | ||||
| HDL (mmol/L) | r-IF (n = 37) | 1.42 ± 0.27 | 1.33, 1.52 | 1.75 ± 0.39 ###,a | 1.65, 1.86 | 0.008 (0.07) * | <0.001 (0.27) * | <0.001 (0.24) * |
| i-IF (n = 35) | 1.25 ± 0.37 | 1.13, 1.38 | 1.39 ± 0.39 ###,a | 1.25, 1.54 | ||||
| n-IF (n = 27) | 1.24 ± 0.31 | 1.12, 1.35 | 1.05 ± 0.29 # | 0.92, 1.18 | ||||
| LDL (mmol/L) | r-IF (n = 37) | 3.20 ± 0.66 | 2.98, 3.43 | 2.77 ± 0.61 a,b | 2.51, 3.03 | 0.003 (0.08) * | 0.046 (0.06) * | 0.05 (0.06) |
| i-IF (n = 35) | 3.56 ± 0.74 | 3.26, 3.86 | 3.07 ± 0.84 ###,a | 2.73, 3.41 | ||||
| n-IF (n = 27) | 3.56 ± 0.89 | 3.29, 3.84 | 3.77 ± 1.13 b | 3.44, 4.07 | ||||
| TG (mmol/L) | r-IF (n = 37) | 1.66 ± 0.51 | 1.43, 1.89 | 1.46 ± 0.68 a,b | 1.13, 1.76 | 0.604 (0.03) | <0.001 (0.18) * | 0.02 (0.08) * |
| i-IF (n = 35) | 2.14 ± 0.98 | 1.84, 2.45 | 1.65 ± 0.84 ###,a,c | 1.23, 2.06 | ||||
| n-IF (n = 27) | 2.39 ± 0.88 | 2.11, 2.67 | 2.94 ± 1.51 ###,b,c | 2.57, 3.32 | ||||
| TC (mmol/L) | r-IF (n = 37) | 4.03 ± 0.82 | 3.73, 4.34 | 3.57 ± 1.09 a,b | 3.22, 3.93 | 0.002 (0.10) * | 0.06 (0.06) | 0.02 (0.08) * |
| i-IF (n = 35) | 4.41 ± 1.33 | 3.99, 4.79 | 3.68 ± 1.29 ##,a | 3.21, 4.14 | ||||
| n-IF (n = 27) | 4.30 ± 0.97 | 3.94, 4.68 | 4.53 ± 1.15 b | 4.11, 4.96 | ||||
| INS (pmol/L) | r-IF (n = 37) | 112.61 ± 0.96 | 110.70, 114.52 | 92.97 ± 0.88 ###,a,b | 91.23, 94.72 | 0.002 (0.10) * | <0.001 (0.94) * | <0.001 (0.76) * |
| i-IF (n = 35) | 129.00 ± 0.98 | 127.39, 131.32 | 131.97 ± 0.91 #,a,c | 130.17, 133.77 | ||||
| n-IF (n = 27) | 140.83 ± 0.24 | 138.59, 143.06 | 168.14 ± 0.31 ###,b,c | 146.09, 170.18 | ||||
| Cognitive test performance | ||||||||
| Digit Span (scale score) | r-IF (n = 37) | 7.84 ± 1.86 | 7.19, 8.48 | 8.88 ± 2.38 ##,a,b | 8.18, 9.59 | <0.001 (0.07) * | 0.004 (0.13) * | <0.001 (0.19) * |
| i-IF (n = 35) | 6.76 ± 2.49 | 5.94, 7.65 | 7.32 ± 2.21 ##,a | 7.42, 9.29 | ||||
| n-IF (n = 27) | 7.76 ± 2.28 | 6.96, 8.52 | 5.73 ± 2.44 ###,b | 4.86, 6.56 | ||||
| RAVLT (T5 score) | r-IF (n = 37) | 30.00 ± 0.66 | 29.23, 31.91 | 36.05 ± 0.61 ###,b | 38.85, 37.25 | <0.001 (0.28) * | <0.001 (0.62) * | <0.001 (0.66) * |
| i-IF (n = 35) | 30.21 ± 0.68 | 28.40, 32.02 | 32.14 ± 0.62 ###,c | 30.91, 33.37 | ||||
| n-IF (n = 27) | 24.70 ± 0.77 | 23.16, 26.24 | 20.41 ± 0.71 ###,b,c | 19.00, 21.81 | ||||
| MMSE (total score) | r-IF (n = 37) | 19.59 ± 2.05 | 18.99, 20.18 | 24.05 ± 3.25 ###,a,b | 22.98, 25.11 | <0.001 (0.16) * | <0.001 (0.32) * | <0.001 (0.29) * |
| i-IF (n = 35) | 18.60 ± 2.38 | 17.84, 19.41 | 22.40 ± 3.38 ###,a,c | 20.95, 23.78 | ||||
| n-IF (n = 27) | 18.73 ± 1.48 | 17.99, 19.43 | 16.33 ± 4.11 ##,b,c | 15.06, 17.65 | ||||
| MoCA (total score) | r-IF (n = 37) | 16.82 ± 4.10 | 15.70, 17.94 | 19.43 ± 4.45 ###,b | 18.26, 20.61 | <0.001 (0.18) * | 0.002 (0.12) * | 0.003 (0.11) * |
| i-IF (n = 35) | 15.84 ± 2.73 | 14.34, 17.32 | 19.00 ± 2.04 ###,c | 17.42, 20.55 | ||||
| n-IF (n = 27) | 14.77 ± 3.82 | 13.42, 16.13 | 14.37 ± 4.25 ##,b,c | 12.95, 15.81 | ||||
| Digit Symbol (scale score) | r-IF (n = 37) | 4.11 ± 1.33 | 3.69, 4.53 | 5.39 ± 2.78 ##,b | 4.70, 6.07 | 0.04 (0.64) * | 0.19 (0.03) | <0.001 (0.16) * |
| i-IF (n = 35) | 3.64 ± 1.55 | 3.09, 4.20 | 4.68 ± 1.73 # | 3.76, 5.57 | ||||
| n-IF (n = 27) | 4.43 ± 1.33 | 3.92, 4.94 | 3.33 ± 1.81 ###,b | 2.52, 4.17 | ||||
| Cognitive Group (36 Months of Follow-Up) | Fasting Status Group | p-Value | ||
|---|---|---|---|---|
| r-IF (n = 37) | i-IF (n = 35) | n-IF (n = 27) | ||
| Number (%) | Number (%) | Number (%) | ||
| Successful aging (SA) | 9 (24.3) | 5 (14.2) | 1 (3.7) | <0.001 * |
| Normal aging (UA) | 27 (73.0) | 22 (62.9) | 8 (29.6) | |
| MCI | 1 (2.7) | 8 (22.9) | 18 (66.7) | |
| Metabolite | δ1H (ppm) | Changes | ||
|---|---|---|---|---|
| r-IF vs. i-IF | r-IF vs. n-IF | n-IF vs. i-IF | ||
| Valine | 1.04 (d) | x | x | + * |
| 3-Hydroxybutyrate | 1.18 (m) | + * | x | x |
| Isoleucine | 1.24 (s) | ─ * | + * | x |
| 3-Hydroxy-3-methylglutaryl-CoA | 1.28 (s) | x | + * | x |
| Alanine | 1.48 (d) | x | ─ | + * |
| Acetate | 1.88 (s) | + * | x | x |
| 2-Aminoadipate | 1.96 (d) | x | + * | x |
| Glutamate | 2 (m) | x | ─ * | + * |
| Glutathione | 2.2 (s) | x | + * | x |
| 4-Hydroxybutyrate | 2.28 (s) | x | ─ | + * |
| Acetoacetate | 3.42 (m) | + * | x | x |
| S-sulfocysteine | 3.48 (m) | x | ─ * | + * |
| n-Acetylglucosamine | 3.68 (q) | + * | ─ * | ─ * |
| 2′-Deoxyguanosine | 3.72 (m) | x | ─ * | + * |
| 2-Hydroxybutyrate | 3.92 (d) | x | x | + * |
| Hippurate | 3.96 (s) | x | ─ * | + * |
| Proline | 4.16 (q) | x | x | + * |
| Guanosine | 4.44 (s) | + * | ─ * | x |
| Glucose | 5.24 (d) | + * | ─ * | x |
| n-Acetylserotonin | 7.04 (s) | + * | x | x |
| Phenylalanine | 7.32 (d) | x | x | + * |
| Metabolite Pathway | p-Value | Holm Adjust | False Discovery Rate | Impact |
|---|---|---|---|---|
| Synthesis and degradation of ketone bodies | 1.13 × 10−5 | 9.01 × 10−4 | 6.57 × 10−4 | 0.9 |
| Glutathione metabolism | 4.22 × 10−2 | 1.00 | 2.72 × 10−1 | 0.25 |
| Alanine, aspartate, and glutamate metabolism | 1.78 × 10−2 | 1.00 | 1.58 × 10−1 | 0.23 |
| Phenylalanine metabolism | 5.23 × 10−2 | 1.00 | 3.28 × 10−1 | 0.15 |
| Arginine and proline metabolism | 1.44 × 10−2 | 1.00 | 6.89 × 10−1 | 0.14 |
| D-glutamine and D-glutamate metabolism | 5.21 × 10−2 | 1.00 | 4.91 × 10−1 | 0.11 |
| Pyruvate metabolism | 2.46 × 10−2 | 1.00 | 8.08 × 10−1 | 0.1 |
| Butanoate metabolism | 1.64 × 10−5 | 1.30 × 10−3 | 6.57 × 10−4 | 0.09 |
| Lysine biosynthesis | 2.46 × 10−1 | 1.00 | 8.08 × 10−1 | 0.07 |
| Aminoacyl-tRNA biosynthesis | 2.80 × 10−5 | 2.19 × 10−3 | 7.47 × 10−4 | 0.06 |
| Valine, leucine, and isoleucine degradation | 3.19 × 10−4 | 2.46 × 10−2 | 6.39 × 10−3 | 0.06 |
| Propanoate metabolism | 3.13 × 10−3 | 2.38 × 10−1 | 5.01 × 10−2 | 0.03 |
| Taurine and hypotaurine metabolism | 1.25 × 10−2 | 9.27 × 10−1 | 1.43 × 10−1 | 0.03 |
| Valine, leucine, and isoleucine biosynthesis | 2.23 × 10−2 | 1.00 | 1.79 × 10−1 | 0.03 |
| Sulfur metabolism | 1.46 × 10−1 | 1.00 | 6.89 × 10−1 | 0.03 |
| Terpenoid backbone biosynthesis | 2.53 × 10−1 | 1.00 | 8.08 × 10−1 | 0.03 |
| Purine metabolism | 1.90 × 10−1 | 1.00 | 8.01 × 10−1 | 0.02 |
| Lysine degradation | 3.40 × 10−1 | 1.00 | 9.72 × 10−1 | 0.02 |
| Starch and sucrose metabolism | 3.58 × 10−1 | 1.00 | 9.87 × 10−1 | 0.02 |
| Tryptophan metabolism | 5.05 × 10−1 | 1.00 | 1.00 | 0.02 |
| Cysteine and methionine metabolism | 1.18 × 10−2 | 8.84 × 10−1 | 1.43 × 10−1 | 0.01 |
| Amino sugar and nucleotide sugar metabolism | 1.78 × 10−1 | 1.00 | 7.89 × 10−1 | 0.01 |
| Selenoamino acid metabolism | 1.51 × 10−2 | 1.00 | 1.51 × 10−1 | 0 |
| Glycolysis or gluconeogenesis | 2.90 × 10−2 | 1.00 | 2.11 × 10−1 | 0 |
| Nitrogen metabolism | 4.43 × 10−2 | 1.00 | 2.72 × 10−1 | 0 |
| Pantothenate and CoA biosynthesis | 2.12 × 10−1 | 1.00 | 8.07 × 10−1 | 0 |
| Phenylalanine, tyrosine, and tryptophan biosynthesis | 2.12 × 10−1 | 1.00 | 8.07 × 10−1 | 0 |
| Pentose phosphate pathway | 2.46 × 10−1 | 1.00 | 8.08 × 10−1 | 0 |
| Galactose metabolism | 3.04 × 10−1 | 1.00 | 9.35 × 10−1 | 0 |
| Histidine metabolism | 3.22 × 10−1 | 1.00 | 9.55 × 10−1 | 0 |
| Tyrosine metabolism | 4.92 × 10−1 | 1.00 | 1.00 | 0 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ooi, T.C.; Meramat, A.; Rajab, N.F.; Shahar, S.; Ismail, I.S.; Azam, A.A.; Sharif, R. Intermittent Fasting Enhanced the Cognitive Function in Older Adults with Mild Cognitive Impairment by Inducing Biochemical and Metabolic changes: A 3-Year Progressive Study. Nutrients 2020, 12, 2644. https://doi.org/10.3390/nu12092644
Ooi TC, Meramat A, Rajab NF, Shahar S, Ismail IS, Azam AA, Sharif R. Intermittent Fasting Enhanced the Cognitive Function in Older Adults with Mild Cognitive Impairment by Inducing Biochemical and Metabolic changes: A 3-Year Progressive Study. Nutrients. 2020; 12(9):2644. https://doi.org/10.3390/nu12092644
Chicago/Turabian StyleOoi, Theng Choon, Asheila Meramat, Nor Fadilah Rajab, Suzana Shahar, Intan Safinar Ismail, Amalina Ahmad Azam, and Razinah Sharif. 2020. "Intermittent Fasting Enhanced the Cognitive Function in Older Adults with Mild Cognitive Impairment by Inducing Biochemical and Metabolic changes: A 3-Year Progressive Study" Nutrients 12, no. 9: 2644. https://doi.org/10.3390/nu12092644
APA StyleOoi, T. C., Meramat, A., Rajab, N. F., Shahar, S., Ismail, I. S., Azam, A. A., & Sharif, R. (2020). Intermittent Fasting Enhanced the Cognitive Function in Older Adults with Mild Cognitive Impairment by Inducing Biochemical and Metabolic changes: A 3-Year Progressive Study. Nutrients, 12(9), 2644. https://doi.org/10.3390/nu12092644







