Use of Plastics with Hot Food among Saudi Pregnant Women Is Associated with Increased Concentrations of A1C, Thyroid-Stimulating Hormone, and Homocysteine and Decreased Concentrations of Vitamins and Minerals
Abstract
1. Introduction
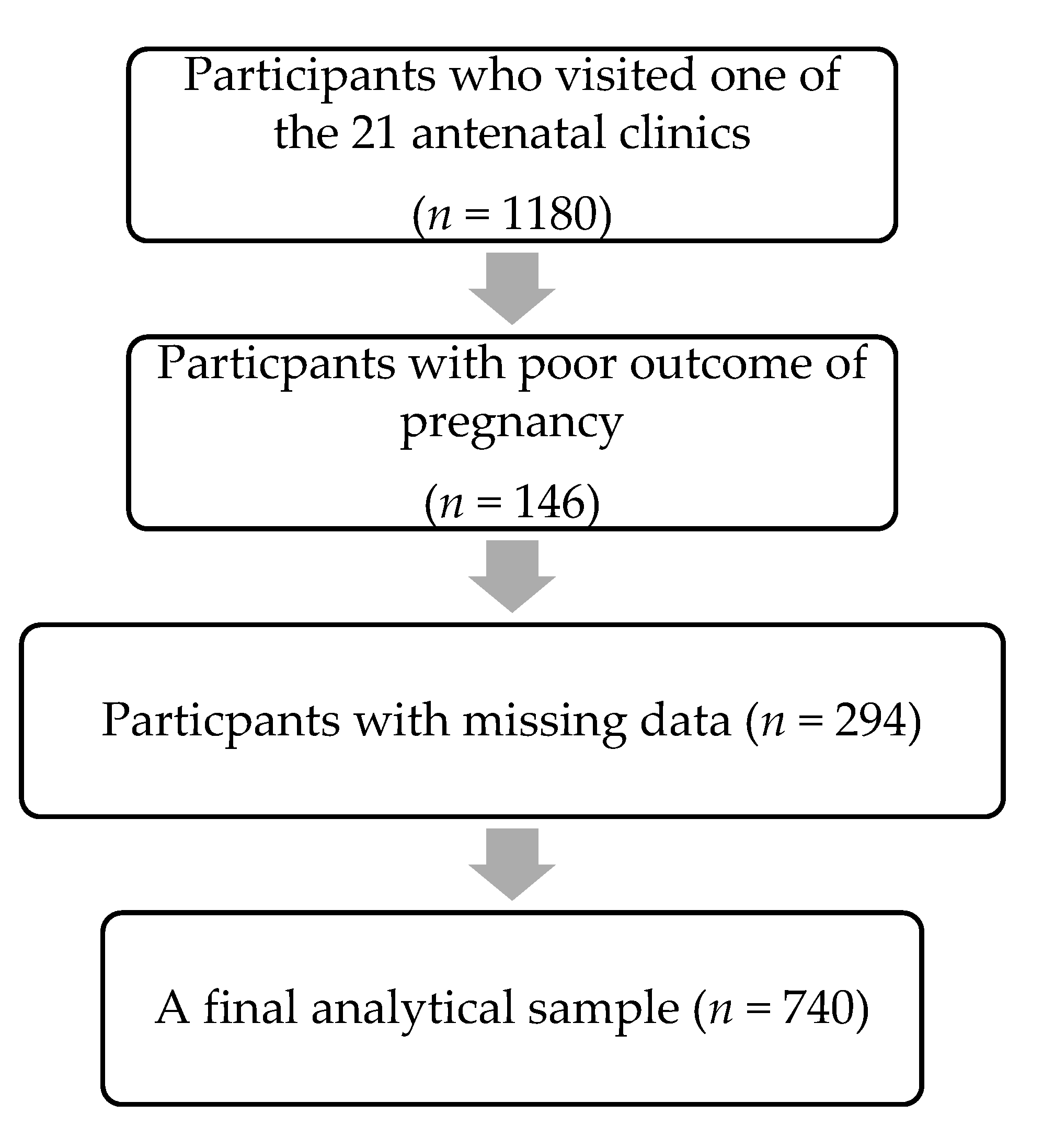
2. Materials and Methods
2.1. Biochemical Parameters
2.2. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rather, I.A.; Koh, W.Y.; Paek, W.K.; Lim, J. The sources of chemical contaminants in food and their health implications. Front. Pharmacol. 2017, 8, 830. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.K.; Singh, R.P.; Singh, P.; Singh, R.L. Chapter 2-Food Hazards: Physical, Chemical, and Biological. In Food Safety and Human Health, 1st ed.; Elsevier: Amsterdam, The Netherland, 2019; pp. 15–65. ISBN 978-0-12-816333-7. [Google Scholar]
- MALEK, C. Arab News. 2018. Available online: https://www.arabnews.com/node/1330096/middle-east (accessed on 20 August 2020).
- Whitehead, R. Foodnavigator-Asia. July 2017. Available online: https://www.foodnavigator-asia.com/Article/2017/07/12/Saudiexe...n?utm_source=copyright&utm_medium=OnSite&utm_campaign=copyright (accessed on 20 August 2020).
- Żwierełło, W.; Maruszewska, A.; Skórka-Majewicz, M.; Goschorska, M.; Baranowska-Bosiacka, I.; Dec, K.; Styburski, D.; Nowakowska, A.; Gutowska, I. The influence of polyphenols on metabolic disorders caused by compounds released from plastics-Review. Chemosphere 2020, 240, 124901. [Google Scholar] [CrossRef] [PubMed]
- Groh, K.J.; Backhaus, T.; Carney-Almroth, B.; Geueke, B.; Inostroza, P.A.; Lennquist, A.; Leslie, H.A.; Maffini, M.; Slunge, D.; Trasande, L.; et al. Overview of known plastic packaging-associated chemicals and their hazards. Sci. Total Environ. 2019, 651, 3253–3268. [Google Scholar] [CrossRef] [PubMed]
- Campanale, C.; Massarelli, C.; Savino, I.; Locaputo, V.; Uricchio, V.F. A Detailed review study on potential effects of microplastics and additives of concern on human health. Int. J. Environ. Res. Public Health 2020, 17, 1212. [Google Scholar] [CrossRef]
- Ehlert, K.A.; Beumer, C.W.E.; Groot, M.C.E. Migration of bisphenol A into water from polycarbonate baby bottles during microwave heating. Food Addit. Contam. Part A Chem. Anal. Control Expo. Risk Assess 2008, 25, 904–910. [Google Scholar] [CrossRef]
- Harkin, C. A review on plastic bioaccumulation, potential health effects and the potential to enhance biotransformation using herbal medicine and nutritional supplements. Int. J. Complement Altern. Med. 2020, 13, 18–26. [Google Scholar] [CrossRef]
- Bommarito, P.A.; Martin, E.; Fry, R.C. Effects of prenatal exposure to endocrine disruptors and toxic metals on the fetal epigenome. Epigenomics 2017, 9, 333–350. [Google Scholar] [CrossRef]
- Hahladakis, J.N.; Velis, C.A.; Weber, R.; Iacovidou, E.; Purnell, P. An overview of chemical additives present in plastics: Migration, release, fate and environmental impact during their use, disposal and recycling. J. Hazard. Mater. 2018, 344, 179–199. [Google Scholar] [CrossRef]
- Pacyga, D.C.; Sathyanarayana, S.; Strakovsky, R.S. Dietary predictors of phthalate and bisphenol exposures in pregnant women. Adv. Nutr. 2019, 10, 803–815. [Google Scholar] [CrossRef]
- Rafati, R.M.; Kazemi, S.; Moghadamnia, A.A. Cadmium toxicity and treatment: An update. Casp. J. Intern. Med. 2017, 8, 135–145. [Google Scholar] [CrossRef]
- Xu, J.; Huang, G.; Guo, T.L. Developmental bisphenol A exposure modulates immune-related diseases. Toxics 2016, 4, 23. [Google Scholar] [CrossRef] [PubMed]
- Rowdhwal, S.S.; Chen, J. Toxic effects of di-2-ethylhexyl phthalate: An overview. BioMed Res. Int. 2018, 2018, 1750368. [Google Scholar] [CrossRef] [PubMed]
- Tran, N.Q.V.; Miyake, K. Neurodevelopmental disorders and environmental toxicants: Epigenetics as an underlying mechanism. Int. J. Genom. 2017, 2017, 7526592. [Google Scholar] [CrossRef] [PubMed]
- Ferguson, K.K.; McElrath, T.F.; Meeker, J.D. Environmental phthalate exposure and preterm birth. JAMA Pediatr. 2014, 168, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Mustieles, V.; Yland, J.; Braun, J.M.; Williams, P.L.; Attaman, J.A.; Ford, J.B.; Calafat, A.M.; Hauser, R.; Messerlian, C. Association of parental preconception exposure to phthalates and phthalate substitutes with preterm birth. JAMA Netw. Open 2020, 3, e202159. [Google Scholar] [CrossRef]
- Filardi, T.; Panimolle, F.; Lenzi, A.; Morano, S. Bisphenol A and phthalates in diet: An emerging link with pregnancy complications. Nutrients 2020, 12, 525. [Google Scholar] [CrossRef]
- Hammouda, S.; Abd Al-Halim, O. Serum levels of some micronutrients and congenital malformations: A prospective cohort study in healthy Saudi Arabian first-trimester pregnant women. Int. J. Vitam. Nutr. Res. 2013, 83, 346–354. [Google Scholar] [CrossRef]
- Kasemsup, R.; Neesanan, N. Knowledge, attitudes and practices relating to plastic containers for food and drinks. J. Med. Assoc. Thai. 2011, 94 (Suppl. 3), S121–S125. [Google Scholar]
- Callan, A.C.; Hinwood, A.L.; Heffernan, A.; Eaglesham, G.; Mueller, J.; Odland, J.Ø. Urinary bisphenol A concentrations in pregnant women. Int. J. Hyg. Environ. Health 2013, 216, 641–644. [Google Scholar] [CrossRef]
- Mariscal-Arcas, M.; Rivas, A.; Granada, A.; Monteagudo, C.; Murcia, M.A.; Olea-Serrano, F. Dietary exposure assessment of pregnant women to bisphenol-A from cans and microwave containers in Southern Spain. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2009, 47, 506–510. [Google Scholar] [CrossRef]
- Valvi, D.; Monfort, N.; Ventura, R.; Casas, M.; Casas, L.; Sunyer, J.; Vrijheid, M. Variability and predictors of urinary phthalate metabolites in Spanish pregnant women. Int. J. Hyg. Environ. Health 2015, 218, 220–231. [Google Scholar] [CrossRef] [PubMed]
- Casas, M.; Valvi, D.; Luque, N.; Ballesteros-Gomez, A.; Carsin, A.-E.; Fernandez, M.F.; Koch, H.M.; Mendez, M.A.; Sunyer, J.; Rubio, S.; et al. Dietary and sociodemographic determinants of bisphenol A urine concentrations in pregnant women and children. Environ. Int. 2013, 56, 10–18. [Google Scholar] [CrossRef] [PubMed]
- SASO. Polypropylene or Polyethylene Food Packaging Containers for Multi-Use. 2020. Available online: https://saso.gov.sa/ar/mediacenter/public_multimedia/Documents/food packaging containers00.pdf (accessed on 2 July 2020).
- Lee, I.; Alakeel, R.; Kim, S.; Al-Sheikh, Y.; Al-mandeel, H.; Alyousef, A.; Kho, Y.; Choi, K. Urinary phthalate metabolites among children in Saudi Arabia: Occurrences, risks, and their association with oxidative stress markers. Sci. Total Environ. 2018, 654, 1350–1357. [Google Scholar] [CrossRef] [PubMed]
- Steegers-Theunissen, R.P.; Wathen, N.C.; Eskes, T.K.; van Raaij-Selten, B.; Chard, T. Maternal and fetal levels of methionine and homocysteine in early human pregnancy. Br. J. Obstet. Gynaecol. 1997, 104, 20–24. [Google Scholar] [CrossRef] [PubMed]
- Şanlıkan, F.; Altuncu, F.; Özbay, K.; Avcım, M.; Göçmen, A. Does serum homocysteine level have a role in the early pregnancy loss? Perinat. J. 2019, 27, 189–193. [Google Scholar] [CrossRef][Green Version]
- Aung, M.T.; Johns, L.E.; Ferguson, K.K.; Mukherjee, B.; McElrath, T.F.; Meeker, J.D. Thyroid hormone parameters during pregnancy in relation to urinary bisphenol A concentrations: A repeated measures study. Environ. Int. 2017, 104, 33–40. [Google Scholar] [CrossRef]
- Acconcia, F.; Pallottini, V.; Marino, M. Molecular Mechanisms of Action of BPA. Dose Response 2015, 13. [Google Scholar] [CrossRef]
- Ahmed, R.G. Maternal bisphenol A alters fetal endocrine system: Thyroid adipokine dysfunction. Food Chem. Toxicol. 2016, 95, 168–174. [Google Scholar] [CrossRef]
- Da Silva, M.M.; Xavier, L.L.; Gonçalves, C.F.; Santos-Silva, A.P.; Paiva-Melo, F.D.; De Freitas, M.L.; Fortunato, R.S.; Miranda-Alves, L.; Ferreira, A.C. Bisphenol A increases hydrogen peroxide generation by thyrocytes both in vivo and in vitro. Endocr. Connect. 2018, 7, 1196–1207. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Wade, M.G. Application of a nonradioactive assay for high throughput screening for inhibition of thyroid hormone uptake via the transmembrane transporter MCT8. Toxicol. In Vitro 2017, 40, 234–242. [Google Scholar] [CrossRef]
- Alonso-Magdalena, P.; Ropero, A.B.; Carrera, M.P.; Cederroth, C.R.; Baquié, M.; Gauthier, B.R.; Nef, S.; Stefani, E.; Nadal, A. Pancreatic insulin content regulation by the estrogen receptor ER alpha. PLoS ONE 2008, 3, e2069. [Google Scholar] [CrossRef] [PubMed]
- Alonso-Magdalena, P.; Morimoto, S.; Ripoll, C.; Fuentes, E.; Nadal, A. The estrogenic effect of bisphenol A disrupts pancreatic beta-cell function in vivo and induces insulin resistance. Environ. Health Perspect. 2006, 114, 106–112. [Google Scholar] [CrossRef] [PubMed]
- Hammouda, S.A.I.; Mumena, W.A. Reduced serum concentrations of vitamin B(12) and folate and elevated thyroid-stimulating hormone and homocysteine levels in first-trimester pregnant Saudi women with high A1C concentrations. Nutr. Res. 2019, 72, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Kutbi, H.A.; Hammouda, S.A. Plasma concentrations of vitamin A and E and risk of dysglycemia in first-trimester pregnant Saudi women. Diabetol. Metab. Syndr. 2020, 12, 17. [Google Scholar] [CrossRef] [PubMed]
- Hammouda, S.; Mohamadin, A. Serum levels of zinc, magnesium and selenium among first trimester pregnant Saudi women with pre-diabetes and diabetes. Endocrinol. Metab. Syndr. 2017, 6. [Google Scholar] [CrossRef]
| Daily Use (n = 78) | Weekly Use (n = 289) | Monthly Use (n = 145) | Never Use (n = 228) | p | |
|---|---|---|---|---|---|
| Age, years, mean ± SD | |||||
| Pregnant women | 26.6 ± 5.66 | 27.0 ± 5.94 | 29.0 ± 6.00 | 28.2 ± 6.52 | 0.002 1 |
| Education level of pregnant women, n (%) | |||||
| ≤Primary education | 16 (9.82) | 61 (37.4) | 29 (17.8) | 57 (35.0) | 0.321 |
| <University degree | 40 (11.0) | 141 (38.6) | 65 (17.8) | 119 (32.6) | |
| ≥University degree | 22 (10.4) | 86 (40.6) | 51 (24.1) | 53 (25.0) | |
| Smoking, spouse, n (%) | 30 (12.5) | 93 (38.8) | 44 (18.3) | 73 (30.4) | 0.654 |
| Pre-pregnancy BMI, kg/m2, mean ± SD | 25.1 ± 4.80 | 24.8 ± 5.53 | 25.9 ± 5.88 | 25.3 ± 5.43 | 0.284 |
| Daily Use (n = 78) | Weekly Use (n = 289) | Monthly Use (n = 145) | Never Use (n = 228) | p | |
|---|---|---|---|---|---|
| TSH, mIU/L | 2.01 ± 0.62 | 1.82 ± 0.58 | 1.82 ± 0.54 | 1.75 ± 0.57 | 0.019 1 |
| HCY, μmol/L | 7.57 ± 1.01 | 7.22 ± 0.93 | 7.21 ± 0.84 | 7.10 ± 0.88 | 0.005 1 |
| A1C, % | 5.07 ± 0.33 | 4.97 ± 0.32 | 4.98 ± 0.31 | 4.93 ± 0.32 | 0.020 1 |
| Magnesium, mg/dL | 4.26 ± 0.67 | 4.34 ± 0.67 | 4.31 ± 0.66 | 4.38 ± 0.64 | 0.455 |
| Vitamin B12, pmol/L | 539 ± 62.4 | 550 ± 64.5 | 553 ± 49.7 | 556 ± 56.2 | 0.084 |
| Folate, nmol/L | 53.3 ± 7.77 | 54.4 ± 7.71 | 55.0 ± 6.18 | 55.3 ± 7.08 | 0.139 |
| Vitamin E, mg/L | 29.9 ± 3.91 | 30.7 ± 3.91 | 30.8 ± 3.65 | 31.1 ± 3.80 | 0.097 |
| Vitamin A, µg/L | 676 ± 103 | 702 ± 101 | 701 ± 97.4 | 709 ± 100 | 0.102 |
| Zinc, μg/dL | 101 ± 13.3 | 104 ± 14.3 | 103 ± 13.2 | 106 ± 14.7 | 0.020 1 |
| Selenium, ng/mL | 11.4 ± 2.56 | 12.0 ± 2.54 | 12.0 ± 2.29 | 12.4 ± 2.50 | 0.019 1 |
| B | SE | p | 95% Confidence Interval | R-Square | |
|---|---|---|---|---|---|
| TSH, mIU/L | 0.06 | 0.02 | 0.004 1 | 0.02 to 0.10 | 0.01 |
| HCY, μmol/L | 0.11 | 0.03 | 0.001 1 | 0.04 to 0.17 | 0.02 |
| A1C, % | 0.03 | 0.01 | 0.005 1 | 0.01 to 0.06 | 0.01 |
| Magnesium, mg/dL | −0.03 | 0.02 | 0.216 | −0.08 to 0.02 | <0.01 |
| Vitamin B12, pmol/L | −4.22 | 2.15 | 0.051 | −8.44 to 0.01 | 0.01 |
| Folate, nmol/L | −0.52 | 0.26 | 0.051 | −1.04 to 0.00 | 0.01 |
| Vitamin E, mg/L | −0.31 | 0.14 | 0.027 1 | −0.58 to −0.03 | 0.01 |
| Vitamin A,µg/L | −6.89 | 3.65 | 0.060 | −14.1 to 0.28 | 0.01 |
| Zinc, μg/dL | −1.33 | 0.51 | 0.010 1 | −2.34 to −0.32 | 0.01 |
| Selenium, ng/mL | −0.26 | 0.09 | 0.005 1 | −0.04 to −0.02 | 0.01 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alharbi, M.H.; Mumena, W.A.; Hammouda, S.A. Use of Plastics with Hot Food among Saudi Pregnant Women Is Associated with Increased Concentrations of A1C, Thyroid-Stimulating Hormone, and Homocysteine and Decreased Concentrations of Vitamins and Minerals. Nutrients 2020, 12, 2609. https://doi.org/10.3390/nu12092609
Alharbi MH, Mumena WA, Hammouda SA. Use of Plastics with Hot Food among Saudi Pregnant Women Is Associated with Increased Concentrations of A1C, Thyroid-Stimulating Hormone, and Homocysteine and Decreased Concentrations of Vitamins and Minerals. Nutrients. 2020; 12(9):2609. https://doi.org/10.3390/nu12092609
Chicago/Turabian StyleAlharbi, Mudi H., Walaa A. Mumena, and Sahar A. Hammouda. 2020. "Use of Plastics with Hot Food among Saudi Pregnant Women Is Associated with Increased Concentrations of A1C, Thyroid-Stimulating Hormone, and Homocysteine and Decreased Concentrations of Vitamins and Minerals" Nutrients 12, no. 9: 2609. https://doi.org/10.3390/nu12092609
APA StyleAlharbi, M. H., Mumena, W. A., & Hammouda, S. A. (2020). Use of Plastics with Hot Food among Saudi Pregnant Women Is Associated with Increased Concentrations of A1C, Thyroid-Stimulating Hormone, and Homocysteine and Decreased Concentrations of Vitamins and Minerals. Nutrients, 12(9), 2609. https://doi.org/10.3390/nu12092609





