The Effects of Cow-Milk Protein Supplementation in Elderly Population: Systematic Review and Narrative Synthesis
Abstract
1. Introduction
- average protein intake for older people should range from 1.0 to 1.2 g/kg of body weight per day (while in young adults, the recommended intake is about 0.7–0.8 g/kg/day) [12];
- it must be taken into account that the feeding-associated anabolic threshold for dietary protein is higher in the elderly than in younger subjects, with the amount of protein required to reach it from a variety of foods being in the order of 25–30 g of protein per meal;
- dietary recommendations for protein intake in the elderly should consider, beyond quantity, also quality, protein source and timing of intake;
- best protein sources are rich in leucine;
- oral supplementation should be considered when dietary protein intake does not reach recommended goals [11].
2. Materials and Methods
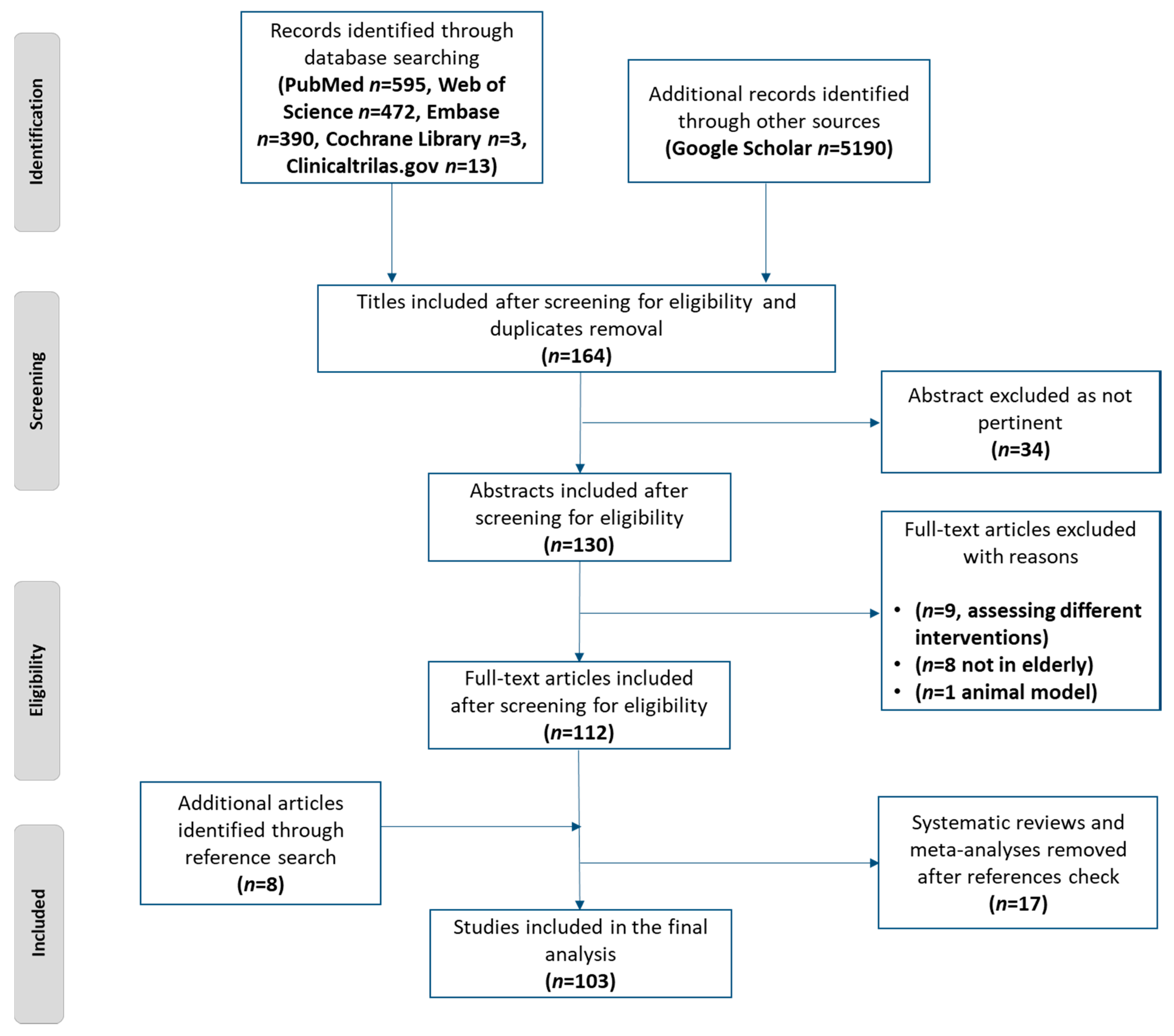
2.1. Literature Search Strategy
2.2. Study Selection
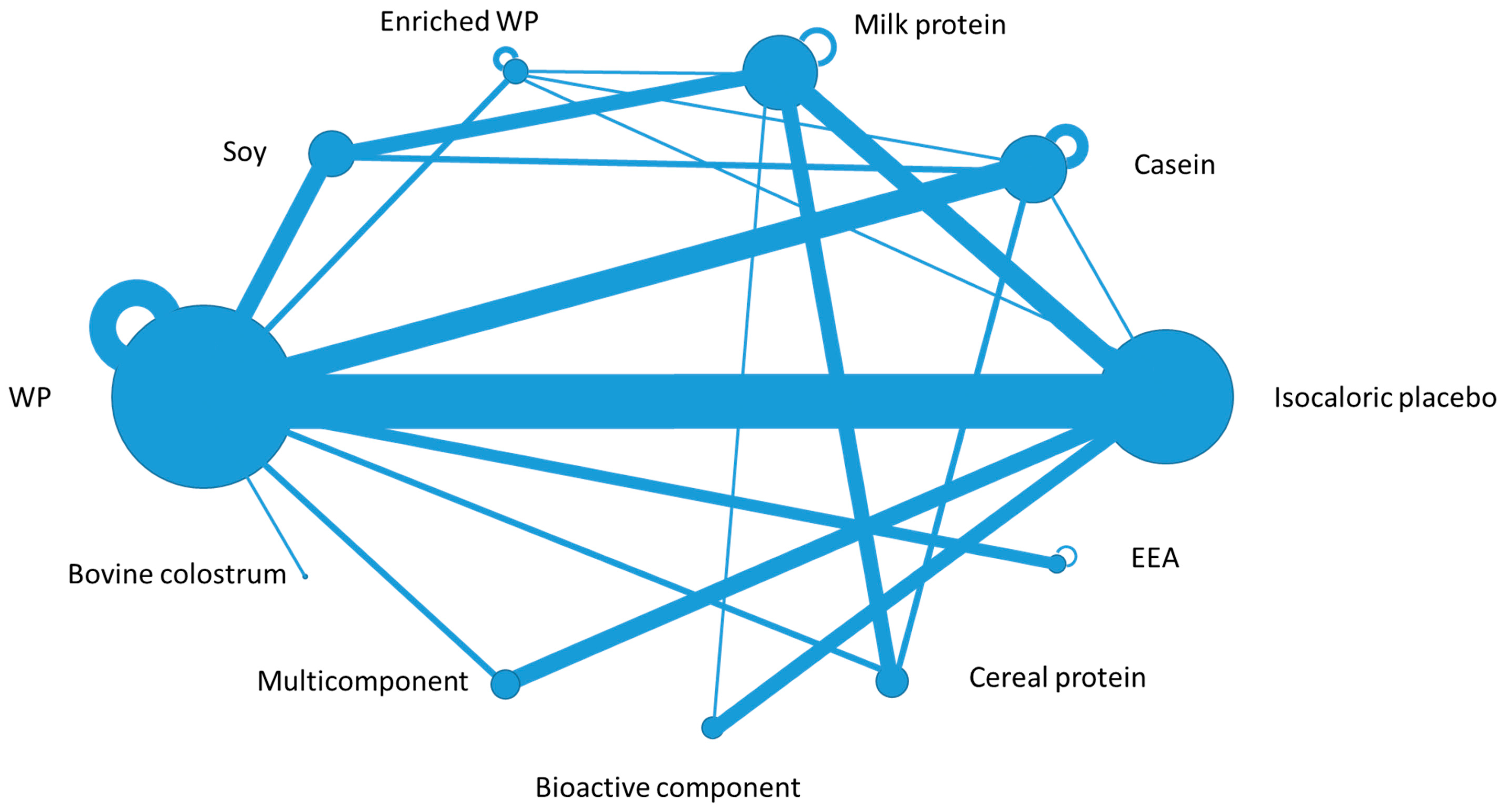
3. Results
3.1. Muscle Related Endpoints among Healthy Subjects (40 Studies)
- co-ingestion of carbohydrates and fats with 21 g of leucine enriched WP did not affect the improvement of MPS rates, among 45 nonsarcopenic older men [45];
- a high WP-leucine- and vitamin D-enriched supplement (21 g protein in 150 Kcal/serving, 10 servings per week) was effective in preserving muscle mass during intentional weight reduction in association with regular physical activity, among 80 obese older adults [46];
- leucine-enriched WP (21 g) and vitamin D (800 IU) daily supplementation before breakfast enhanced post prandial MPS (acute effect) and muscle mass (long term effect) in 24 healthy elderly men in a ‘proof of principle’ trial [47];
- a multi-ingredient supplementation consisting of 30 g WP, 2.5 g creatine, 500 IU vitamin D, 400 mg calcium and 1500 mg n-3 PUFA was tested with and without exercise versus placebo and was effective in increasing both muscle strength and mass among 49 older men [48];
- co-ingestion of milk fat (26.7 g) did not affect the raise in plasma amino-acids and MPS after the ingestion of 20 g of casein, among 24 healthy older males [49].
3.2. Muscle Related Endpoints among Patients (14 Studies)
- during a six-month resistance training (RT) intervention among 80 mobility-limited older adults, 40 g of daily WP supplementation did not add benefit to exercise in improving lean mass, muscle strength and physical function [66];
- a leucine-enriched WP supplement with vitamin D (20 g + 800 IU, twice a day for 13 weeks) was tested versus an iso-caloric dietary supplement, and was superior to placebo in improving muscle mass and lower-extremity function among a large cohort of 380 sarcopenic older adults, even in patients who were unable to exercise [67];
- the association of physical activity with a daily supplementation consisting of WP (22 g), essential amino acids (10.9 g including 4 g of leucine) and vitamin D (100 IU) was more effective than physical activity plus placebo in increasing fat free mass and muscle strength, in improving quality of life and in decreasing inflammation index in 130 sarcopenic elderly people [68];
- the combination of regular resistance muscle training with a nutrition therapy based on an oral supplement offered twice daily (containing 20 g WP, 9 g carbohydrates, 3 g fat, 800 IU vitamin D, and a mixture of vitamins, minerals, and fibers per serving) was superior to exercise alone in improving muscle mass and strength, in 34 elderly patients at high risk of sarcopenia [69];
- the combination of RE with different isocaloric shakes containing 12 g of milk protein or 12 g of soy proteins versus placebo (rice milk, considered as non-protein control) had a positive effect on muscle mass, independently from the type of protein source (milk or soy), among 26 sarcopenic men. The same intervention study among 26 overweight sarcopenic men resulted in a decrease in fat mass only in the dairy supplemented group [70,71].
3.3. Bones (12 Studies)
3.4. Cardiovascular Diseases (Eight Studies)
3.5. Protein Intake and Metabolism (Seven Studies)
3.6. Inflammation Markers (Seven Studies)
3.7. Chronic Obstructive Pulmonary Disease (Four Studies)
3.8. Neurocognitive Function (Four Studies)
3.9. Response to Vaccines (Two Studies)
3.10. Miscellanea (5 Studies)
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Decade of Healthy Ageing (2020–2030). Available online: https://www.who.int/ageing/decade-of-healthy-ageing (accessed on 13 May 2020).
- Cosco, T.D.; Howse, K.; Brayne, C. Healthy ageing, resilience and wellbeing. Epidemiol. Psychiatr. Sci. 2017, 26, 579–583. [Google Scholar] [CrossRef] [PubMed]
- Peel, N.; Bartlett, H.; McClure, R. Healthy ageing: How is it defined and measured? Australas. J. Ageing 2004, 23, 115–119. [Google Scholar] [CrossRef]
- Fielding, R.A.; Vellas, B.; Evans, W.J.; Bhasin, S.; Morley, J.E.; Newman, A.B.; van Kan, G.A.; Andrieu, S.; Bauer, J.; Breuille, D.; et al. Sarcopenia: An Undiagnosed Condition in Older Adults. Current Consensus Definition: Prevalence, Etiology, and Consequences. International Working Group on Sarcopenia. J. Am. Med. Dir. Assoc. 2011, 12, 249–256. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyère, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Sarcopenia: Revised European consensus on definition and diagnosis. Age Ageing 2019, 48, 16–31. [Google Scholar] [CrossRef] [PubMed]
- Beasley, J.M.; Shikany, J.M.; Thomson, C.A. The Role of Dietary Protein Intake in the Prevention of Sarcopenia of Aging. Nutr. Clin. Pr. 2013, 28, 684–690. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, R.R. Update on protein intake: Importance of milk proteins for health status of the elderly. Nutr. Rev. 2015, 73, 41–47. [Google Scholar] [CrossRef] [PubMed]
- Walrand, S.; Boline, Y. Optimizing protein intake in aging. Curr. Opin. Clin. Nutr. Metab. Care 2005, 8, 89–94. [Google Scholar] [CrossRef]
- Malcomson, F.C.; Mathers, J.C. Nutrition and Ageing. Subcell. Biochem. 2018, 90, 373–424. [Google Scholar]
- McGee, M.; Jensen, G.L. Nutrition in the Elderly. J. Clin. Gastroenterol. 2000, 30, 372–380. [Google Scholar] [CrossRef]
- Bauer, J.; Biolo, G.; Cederholm, T.; Cesari, M.; Cruz-Jentoft, A.J.; Morley, J.E.; Phillips, S.; Sieber, C.; Stehle, P.; Teta, D.; et al. Evidence-Based Recommendations for Optimal Dietary Protein Intake in Older People: A Position Paper From the PROT-AGE Study Group. J. Am. Med. Dir. Assoc. 2013, 14, 542–559. [Google Scholar] [CrossRef]
- Joint FAO/WHO/UNU Expert Consultation on Protein and Amino Acid Requirements in Human Nutrition (2002: Geneva, Switzerland); Food and Agriculture Organization of the United Nations; World Health Organization; United Nations University. Protein and Amino Acid Requirements in Human Nutrition: Report of a Joint FAO/WHO/UNU Expert Consultation; World Health Organization: Geneva, Switzerland, 2007; ISBN 978-92-4-120935-9. [Google Scholar]
- Hesselink, M.K.C.; Minnaard, R.; Schrauwen, P. Eat the meat or feed the meat: Protein turnover in remodeling muscle. Curr. Opin. Clin. Nutr. Metab. Care 2006, 9, 672–676. [Google Scholar] [CrossRef] [PubMed]
- Rémond, D.; Machebeuf, M.; Yven, C.; Buffière, C.; Mioche, L.; Mosoni, L.; Mirand, P.P. Postprandial whole-body protein metabolism after a meat meal is influenced by chewing efficiency in elderly subjects. Am. J. Clin. Nutr. 2007, 85, 1286–1292. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-Y.; Acosta, A.; Camilleri, M.; Burton, D.; Harmsen, W.S.; Fox, J.; Szarka, L.A. Gastric Motor Dysfunction in Patients With Functional Gastroduodenal Symptoms. Am. J. Gastroenterol. 2017, 112, 1689–1699. [Google Scholar] [CrossRef] [PubMed]
- Godos, J.; Tieri, M.; Ghelfi, F.; Titta, L.; Marventano, S.; Lafranconi, A.; Gambera, A.; Alonzo, E.; Sciacca, S.; Buscemi, S.; et al. Dairy foods and health: An umbrella review of observational studies. Int. J. Food Sci. Nutr. 2020, 71, 138–151. [Google Scholar] [CrossRef] [PubMed]
- Mah, J.Y.; Choy, S.W.; Roberts, M.A.; Desai, A.M.; Corken, M.; Gwini, S.M.; McMahon, L.P. Oral protein-based supplements versus placebo or no treatment for people with chronic kidney disease requiring dialysis. Cochrane Database Syst. Rev. 2020, 5, CD012616. [Google Scholar] [PubMed]
- Dawson, B.; Taylor, J.; Favaloro, E.J. Potential benefits of improved protein intake in older people. Nutr. Diet. 2008, 65, 151–156. [Google Scholar] [CrossRef]
- Gryson, C.; Walrand, S.; Giraudet, C.; Rousset, P.; Migne, C.; Bonhomme, C.; Le Ruyet, P.; Boirie, Y. “Fast proteins” with a unique essential amino acid content as an optimal nutrition in the elderly: Growing evidence. Clin. Nutr. 2014, 33, 642–648. [Google Scholar] [CrossRef]
- Moher, D.; Liberati, A.; Tetzlaff, J.; Altman, D.G.; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: The PRISMA statement. PLoS Med. 2009, 6, e1000097. [Google Scholar] [CrossRef]
- Van den Borne, J.J.; Kudla, U.; Geurts, J.M. Translating novel insights from age-related loss of skeletal muscle mass and phenotypic flexibility into diet and lifestyle recommendations for the elderly. Curr. Opin. Food Sci. 2016, 10, 60–67. [Google Scholar] [CrossRef]
- Tang, J.E.; Phillips, S.M. Maximizing muscle protein anabolism: The role of protein quality. Curr. Opin. Clin. Nutr. Metab. Care 2009, 12, 66–71. [Google Scholar] [CrossRef]
- Devries, M.C.; McGlory, C.; Bolster, D.R.; Kamil, A.; Rahn, M.; Harkness, L.; Baker, S.K.; Phillips, S.M. Protein leucine content is a determinant of shorter- and longer-term muscle protein synthetic responses at rest and following resistance exercise in healthy older women: A randomized, controlled trial. Am. J. Clin. Nutr. 2018, 107, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Bukhari, S.S.I.; Phillips, B.E.; Wilkinson, D.J.; Limb, M.C.; Rankin, D.; Mitchell, W.K.; Kobayashi, H.; Greenhaff, P.L.; Smith, K.; Atherton, P.J.; et al. Intake of low-dose leucine-rich essential amino acids stimulates muscle anabolism equivalently to bolus whey protein in older women at rest and after exercise. Am. J. Physiol.-Endocrinol. Metab. 2015, 308, E1056–E1065. [Google Scholar] [CrossRef] [PubMed]
- Dardevet, D.; Rémond, D.; Peyron, M.-A.; Papet, I.; Savary-Auzeloux, I.; Mosoni, L. Muscle wasting and resistance of muscle anabolism: The “anabolic threshold concept” for adapted nutritional strategies during sarcopenia. Sci. World J. 2012, 2012, 269531. [Google Scholar] [CrossRef] [PubMed]
- Devries, M.C.; McGlory, C.; Bolster, D.R.; Kamil, A.; Rahn, M.; Harkness, L.; Baker, S.K.; Phillips, S.M. Leucine, Not Total Protein, Content of a Supplement Is the Primary Determinant of Muscle Protein Anabolic Responses in Healthy Older Women. J. Nutr. 2018, 148, 1088–1095. [Google Scholar] [CrossRef] [PubMed]
- Schnebelen-Berthier, C.; Baudry, C.; Clerc, E.; Jaruga, A.; Le Ruyet, P.; Lecerf, J.-M. Effect of supplementing meals with soluble milk proteins on plasma leucine levels in healthy older people: A randomized pilot study. Nutr. Aging 2015, 3, 139–146. [Google Scholar] [CrossRef]
- Gorissen, S.H.; Horstman, A.M.; Franssen, R.; Kouw, I.W.; Wall, B.T.; Burd, N.A.; de Groot, L.C.; van Loon, L.J. Habituation to low or high protein intake does not modulate basal or postprandial muscle protein synthesis rates: A randomized trial. Am. J. Clin. Nutr. 2017, 105, 332–342. [Google Scholar] [CrossRef]
- Kiskini, A.; Hamer, H.M.; Wall, B.T.; Groen, B.B.L.; de Lange, A.; Bakker, J.A.; Senden, J.M.G.; Verdijk, L.B.; van Loon, L.J.C. The muscle protein synthetic response to the combined ingestion of protein and carbohydrate is not impaired in healthy older men. Age 2013, 35, 2389–2398. [Google Scholar] [CrossRef]
- Paddon-Jones, D.; Sheffield-Moore, M.; Katsanos, C.S.; Zhang, X.-J.; Wolfe, R.R. Differential stimulation of muscle protein synthesis in elderly humans following isocaloric ingestion of amino acids or whey protein. Exp. Gerontol. 2006, 41, 215–219. [Google Scholar] [CrossRef]
- Katsanos, C.S.; Chinkes, D.L.; Paddon-Jones, D.; Zhang, X.; Aarsland, A.; Wolfe, R.R. Whey protein ingestion in elderly persons results in greater muscle protein accrual than ingestion of its constituent essential amino acid content. Nutr. Res. 2008, 28, 651–658. [Google Scholar] [CrossRef]
- Koopman, R.; Crombach, N.; Gijsen, A.P.; Walrand, S.; Fauquant, J.; Kies, A.K.; Lemosquet, S.; Saris, W.H.M.; Boirie, Y.; van Loon, L.J.C. Ingestion of a protein hydrolysate is accompanied by an accelerated in vivo digestion and absorption rate when compared with its intact protein. Am. J. Clin. Nutr. 2009, 90, 106–115. [Google Scholar] [CrossRef]
- Pennings, B.; Boirie, Y.; Senden, J.M.G.; Gijsen, A.P.; Kuipers, H.; van Loon, L.J.C. Whey protein stimulates postprandial muscle protein accretion more effectively than do casein and casein hydrolysate in older men. Am. J. Clin. Nutr. 2011, 93, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Burd, N.A.; Yang, Y.; Moore, D.R.; Tang, J.E.; Tarnopolsky, M.A.; Phillips, S.M. Greater stimulation of myofibrillar protein synthesis with ingestion of whey protein isolate v. micellar casein at rest and after resistance exercise in elderly men. Br. J. Nutr. 2012, 108, 958–962. [Google Scholar] [CrossRef] [PubMed]
- Groen, B.B.L.; Res, P.T.; Pennings, B.; Hertle, E.; Senden, J.M.G.; Saris, W.H.M.; van Loon, L.J.C. Intragastric protein administration stimulates overnight muscle protein synthesis in elderly men. Am. J. Physiol.-Endocrinol. Metab. 2012, 302, E52–E60. [Google Scholar] [CrossRef] [PubMed]
- Pennings, B.; Groen, B.; de Lange, A.; Gijsen, A.P.; Zorenc, A.H.; Senden, J.M.G.; van Loon, L.J.C. Amino acid absorption and subsequent muscle protein accretion following graded intakes of whey protein in elderly men. Am. J. Physiol.-Endocrinol. Metab. 2012, 302, E992–E999. [Google Scholar] [CrossRef] [PubMed]
- Wall, B.T.; Hamer, H.M.; de Lange, A.; Kiskini, A.; Groen, B.B.L.; Senden, J.M.G.; Gijsen, A.P.; Verdijk, L.B.; van Loon, L.J.C. Leucine co-ingestion improves post-prandial muscle protein accretion in elderly men. Clin. Nutr. 2013, 32, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Luiking, Y.C.; Deutz, N.E.P.; Memelink, R.G.; Verlaan, S.; Wolfe, R.R. Postprandial muscle protein synthesis is higher after a high whey protein, leucine-enriched supplement than after a dairy-like product in healthy older people: A randomized controlled trial. Nutr. J. 2014, 13, 9. [Google Scholar] [CrossRef]
- Churchward-Venne, T.A.; Snijders, T.; Linkens, A.M.A.; Hamer, H.M.; van Kranenburg, J.; van Loon, L.J.C. Ingestion of Casein in a Milk Matrix Modulates Dietary Protein Digestion and Absorption Kinetics but Does Not Modulate Postprandial Muscle Protein Synthesis in Older Men. J. Nutr. 2015, 145, 1438–1445. [Google Scholar] [CrossRef]
- Borack, M.S.; Reidy, P.T.; Husaini, S.H.; Markofski, M.M.; Deer, R.R.; Richison, A.B.; Lambert, B.S.; Cope, M.B.; Mukherjea, R.; Jennings, K.; et al. Soy-Dairy Protein Blend or Whey Protein Isolate Ingestion Induces Similar Postexercise Muscle Mechanistic Target of Rapamycin Complex 1 Signaling and Protein Synthesis Responses in Older Men. J. Nutr. 2016, 146, 2468–2475. [Google Scholar] [CrossRef]
- Gorissen, S.H.; Horstman, A.M.; Franssen, R.; Crombag, J.J.; Langer, H.; Bierau, J.; Respondek, F.; van Loon, L.J. Ingestion of Wheat Protein Increases In Vivo Muscle Protein Synthesis Rates in Healthy Older Men in a Randomized Trial. J. Nutr. 2016, 146, 1651–1659. [Google Scholar] [CrossRef]
- Walrand, S.; Gryson, C.; Salles, J.; Giraudet, C.; Migné, C.; Bonhomme, C.; Le Ruyet, P.; Boirie, Y. Fast-digestive protein supplement for ten days overcomes muscle anabolic resistance in healthy elderly men. Clin. Nutr. 2016, 35, 660–668. [Google Scholar] [CrossRef]
- Kouw, I.W.K.; Holwerda, A.M.; Trommelen, J.; Kramer, I.F.; Bastiaanse, J.; Halson, S.L.; Wodzig, W.K.W.H.; Verdijk, L.B.; van Loon, L.J.C. Protein Ingestion before Sleep Increases Overnight Muscle Protein Synthesis Rates in Healthy Older Men: A Randomized Controlled Trial. J. Nutr. 2017, 147, 2252–2261. [Google Scholar] [CrossRef] [PubMed]
- Hamarsland, H.; Aas, S.N.; Nordengen, A.L.; Holte, K.; Garthe, I.; Paulsen, G.; Cotter, M.; Borsheim, E.; Benestad, H.B.; Raastad, T. Native Whey Induces Similar Post Exercise Muscle Anabolic Responses as Regular Whey, Despite Greater Leucinemia, in Elderly Individuals. J. Nutr. Health Aging 2019, 23, 42–50. [Google Scholar] [CrossRef]
- Kramer, I.F.; Verdijk, L.B.; Hamer, H.M.; Verlaan, S.; Luiking, Y.; Kouw, I.W.K.; Senden, J.M.; van Kranenburg, J.; Gijsen, A.P.; Poeze, M.; et al. Impact of the Macronutrient Composition of a Nutritional Supplement on Muscle Protein Synthesis Rates in Older Men: A Randomized, Double Blind, Controlled Trial. J. Clin. Endocrinol. Metab. 2015, 100, 4124–4132. [Google Scholar] [CrossRef]
- Verreijen, A.M.; Verlaan, S.; Engberink, M.F.; Swinkels, S.; de Vogel-van den Bosch, J.; Weijs, P.J.M. A high whey protein-, leucine-, and vitamin D-enriched supplement preserves muscle mass during intentional weight loss in obese older adults: A double-blind randomized controlled trial. Am. J. Clin. Nutr. 2015, 101, 279–286. [Google Scholar] [CrossRef] [PubMed]
- Chanet, A.; Verlaan, S.; Salles, J.; Giraudet, C.; Patrac, V.; Pidou, V.; Pouyet, C.; Hafnaoui, N.; Blot, A.; Cano, N.; et al. Supplementing Breakfast with a Vitamin D and Leucine—Enriched Whey Protein Medical Nutrition Drink Enhances Postprandial Muscle Protein Synthesis and Muscle Mass in Healthy Older Men. J. Nutr. 2017, 147, 2262–2271. [Google Scholar] [CrossRef] [PubMed]
- Bell, K.E.; Snijders, T.; Zulyniak, M.; Kumbhare, D.; Parise, G.; Chabowski, A.; Phillips, S.M. A whey protein-based multi-ingredient nutritional supplement stimulates gains in lean body mass and strength in healthy older men: A randomized controlled trial. PLoS ONE 2017, 12, e0181387. [Google Scholar] [CrossRef]
- Gorissen, S.H.M.; Burd, N.A.; Kramer, I.F.; van Kranenburg, J.; Gijsen, A.P.; Rooyackers, O.; van Loon, L.J.C. Co-ingesting milk fat with micellar casein does not affect postprandial protein handling in healthy older men. Clin. Nutr. 2017, 36, 429–437. [Google Scholar] [CrossRef]
- Hulmi, J.J.; Kovanen, V.; Lisko, I.; Selänne, H.; Mero, A.A. The effects of whey protein on myostatin and cell cycle-related gene expression responses to a single heavy resistance exercise bout in trained older men. Eur. J. Appl. Physiol. 2008, 102, 205–213. [Google Scholar] [CrossRef]
- Dirks, M.L.; Wall, B.T.; Kramer, I.F.; Zorenc, A.H.; Goessens, J.P.B.; Gijsen, A.P.; van Loon, L.J.C. A single session of neuromuscular electrical stimulation does not augment postprandial muscle protein accretion. Am. J. Physiol.-Endocrinol. Metab. 2016, 311, E278–E285. [Google Scholar] [CrossRef]
- Dideriksen, K.J.; Reitelseder, S.; Petersen, S.G.; Hjort, M.; Helmark, I.C.; Kjaer, M.; Holm, L. Stimulation of muscle protein synthesis by whey and caseinate ingestion after resistance exercise in elderly individuals. Scand. J. Med. Sci. Sports 2011, 21, e372–e383. [Google Scholar] [CrossRef]
- Yang, Y.; Breen, L.; Burd, N.A.; Hector, A.J.; Churchward-Venne, T.A.; Josse, A.R.; Tarnopolsky, M.A.; Phillips, S.M. Resistance exercise enhances myofibrillar protein synthesis with graded intakes of whey protein in older men. Br. J. Nutr. 2012, 108, 1780–1788. [Google Scholar] [CrossRef] [PubMed]
- Arnarson, A.; Geirsdottir, O.G.; Ramel, A.; Briem, K.; Jonsson, P.V.; Thorsdottir, I. Effects of whey proteins and carbohydrates on the efficacy of resistance training in elderly people: Double blind, randomised controlled trial. Eur. J. Clin. Nutr. 2013, 67, 821–826. [Google Scholar] [CrossRef] [PubMed]
- Gryson, C.; Ratel, S.; Rance, M.; Penando, S.; Bonhomme, C.; Le Ruyet, P.; Duclos, M.; Boirie, Y.; Walrand, S. Four-Month Course of Soluble Milk Proteins Interacts With Exercise to Improve Muscle Strength and Delay Fatigue in Elderly Participants. J. Am. Med. Dir. Assoc. 2014, 15, 958.e1–958.e9. [Google Scholar] [CrossRef] [PubMed]
- Karelis, A.D.; Messier, V.; Suppère, C.; Briand, P.; Rabasa-Lhoret, R. Effect of cysteine-rich whey protein (immunocal®) supplementation in combination with resistance training on muscle strength and lean body mass in non-frail elderly subjects: A randomized, double-blind controlled study. J. Nutr. Health Aging 2015, 19, 531–536. [Google Scholar] [CrossRef]
- Weisgarber, K.D.; Candow, D.G.; Farthing, J.P. Whey protein and high-volume resistance training in postmenopausal women. J. Nutr. Health Aging 2015, 19, 511–517. [Google Scholar] [CrossRef]
- Thomson, R.L.; Brinkworth, G.D.; Noakes, M.; Buckley, J.D. Muscle strength gains during resistance exercise training are attenuated with soy compared with dairy or usual protein intake in older adults: A randomized controlled trial. Clin. Nutr. 2016, 35, 27–33. [Google Scholar] [CrossRef]
- Mori, H.; Tokuda, Y. Effect of whey protein supplementation after resistance exercise on the muscle mass and physical function of healthy older women: A randomized controlled trial. Geriatr. Gerontol. Int. 2018, 18, 1398–1404. [Google Scholar] [CrossRef]
- Junior, P.S.; Ribeiro, A.S.; Nabuco, H.C.G.; Fernandes, R.R.; Tomeleri, C.M.; Cunha, P.M.; Venturini, D.; Barbosa, D.S.; Schoenfeld, B.J.; Cyrino, E.S. Effects of Whey Protein Supplementation Associated With Resistance Training on Muscular Strength, Hypertrophy, and Muscle Quality in Preconditioned Older Women. Int. J. Sport Nutr. Exerc. Metab. 2018, 28, 528–535. [Google Scholar] [CrossRef]
- Hays, N.P.; Kim, H.; Wells, A.M.; Kajkenova, O.; Evans, W.J. Effects of whey and fortified collagen hydrolysate protein supplements on nitrogen balance and body composition in older women. J. Am. Diet. Assoc. 2009, 109, 1082–1087. [Google Scholar] [CrossRef]
- Ottestad, I.; Løvstad, A.T.; Gjevestad, G.O.; Hamarsland, H.; Benth, J.Š.; Andersen, L.F.; Bye, A.; Biong, A.S.; Retterstøl, K.; Iversen, P.O.; et al. Intake of a Protein-Enriched Milk and Effects on Muscle Mass and Strength. A 12-Week Randomized Placebo Controlled Trial among Community-Dwelling Older Adults. J. Nutr. Health Aging 2017, 21, 1160–1169. [Google Scholar] [CrossRef]
- Liu, Z.M.; Ho, S.C.; Chen, Y.M.; Ho, Y.P. A mild favorable effect of soy protein with isoflavones on body composition--a 6-month double-blind randomized placebo-controlled trial among Chinese postmenopausal women. Int. J. Obes. 2010, 34, 309–318. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Kerr, D.A.; Meng, X.; Devine, A.; Solah, V.; Binns, C.W.; Prince, R.L. Two-Year Whey Protein Supplementation Did Not Enhance Muscle Mass and Physical Function in Well-Nourished Healthy Older Postmenopausal Women. J. Nutr. 2015, 145, 2520–2526. [Google Scholar] [CrossRef] [PubMed]
- Kramer, I.F.; Verdijk, L.B.; Hamer, H.M.; Verlaan, S.; Luiking, Y.C.; Kouw, I.W.K.; Senden, J.M.; van Kranenburg, J.; Gijsen, A.P.; Bierau, J.; et al. Both basal and post-prandial muscle protein synthesis rates, following the ingestion of a leucine-enriched whey protein supplement, are not impaired in sarcopenic older males. Clin. Nutr. 2017, 36, 1440–1449. [Google Scholar] [CrossRef] [PubMed]
- Chalé, A.; Cloutier, G.J.; Hau, C.; Phillips, E.M.; Dallal, G.E.; Fielding, R.A. Efficacy of whey protein supplementation on resistance exercise-induced changes in lean mass, muscle strength, and physical function in mobility-limited older adults. J. Gerontol. A Biol. Sci. Med. Sci. 2013, 68, 682–690. [Google Scholar] [CrossRef]
- Bauer, J.M.; Verlaan, S.; Bautmans, I.; Brandt, K.; Donini, L.M.; Maggio, M.; McMurdo, M.E.T.; Mets, T.; Seal, C.; Wijers, S.L.; et al. Effects of a vitamin D and leucine-enriched whey protein nutritional supplement on measures of sarcopenia in older adults, the PROVIDE study: A randomized, double-blind, placebo-controlled trial. J. Am. Med. Dir. Assoc. 2015, 16, 740–747. [Google Scholar] [CrossRef]
- Rondanelli, M.; Klersy, C.; Terracol, G.; Talluri, J.; Maugeri, R.; Guido, D.; Faliva, M.A.; Solerte, B.S.; Fioravanti, M.; Lukaski, H.; et al. Whey protein, amino acids, and vitamin D supplementation with physical activity increases fat-free mass and strength, functionality, and quality of life and decreases inflammation in sarcopenic elderly. Am. J. Clin. Nutr. 2016, 103, 830–840. [Google Scholar] [CrossRef]
- Molnár, A.; Sztruhár, I.J.; Csontos, Á.A.; Ferencz, C.; Várbíró, S.; Székács, B. Special nutrition intervention is required for muscle protective efficacy of physical exercise in elderly people at highest risk of sarcopenia. Physiol. Int. 2016, 103, 368–376. [Google Scholar] [CrossRef]
- Maltais, M.L.; Perreault, K.; Courchesne-Loyer, A.; Lagacé, J.C.; Barsalani, R.; Dionne, I.J. Effect of Resistance Training and Various Sources of Protein Supplementation on Body Fat Mass and Metabolic Profile in Sarcopenic Overweight Older Adult Men: A Pilot Study. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 71–77. [Google Scholar] [CrossRef]
- Maltais, M.L.; Ladouceur, J.P.; Dionne, I.J. The Effect of Resistance Training and Different Sources of Postexercise Protein Supplementation on Muscle Mass and Physical Capacity in Sarcopenic Elderly Men. J. Strength Cond. Res. 2016, 30, 1680–1687. [Google Scholar] [CrossRef]
- Gade, J.; Beck, A.M.; Bitz, C.; Christensen, B.; Klausen, T.W.; Vinther, A.; Astrup, A. Protein-enriched, milk-based supplement to counteract sarcopenia in acutely ill geriatric patients offered resistance exercise training during and after hospitalisation: Study protocol for a randomised, double-blind, multicentre trial. BMJ Open 2018, 8, e019210. [Google Scholar] [CrossRef]
- Dirks, M.L.; Tieland, M.; Verdijk, L.B.; Losen, M.; Nilwik, R.; Mensink, M.; de Groot, L.C.P.G.M.; van Loon, L.J.C. Protein Supplementation Augments Muscle Fiber Hypertrophy but Does Not Modulate Satellite Cell Content During Prolonged Resistance-Type Exercise Training in Frail Elderly. J. Am. Med. Dir. Assoc. 2017, 18, 608–615. [Google Scholar] [CrossRef] [PubMed]
- Niccoli, S.; Kolobov, A.; Bon, T.; Rafilovich, S.; Munro, H.; Tanner, K.; Pearson, T.; Lees, S.J. Whey Protein Supplementation Improves Rehabilitation Outcomes in Hospitalized Geriatric Patients: A Double Blinded, Randomized Controlled Trial. J. Nutr. Gerontol. Geriatr. 2017, 36, 149–165. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Choi, J.-E.; Hwang, H.-S. Protein supplementation improves muscle mass and physical performance in undernourished prefrail and frail elderly subjects: A randomized, double-blind, placebo-controlled trial. Am. J. Clin. Nutr. 2018, 108, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, A.L.; Hayashi, A.P.; Jambassi-Filho, J.C.; de Capitani, M.D.; de Santana, D.A.; Gualano, B.; Roschel, H. Different protein and derivatives supplementation strategies combined with resistance training in pre-frail and frail elderly: Rationale and protocol for the “Pro-Elderly” Study. Nutr. Health 2017, 23, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Collins, J.; Longhurst, G.; Roschel, H.; Gualano, B. Resistance Training and Co-supplementation with Creatine and Protein in Older Subjects with Frailty. J. Frailty Aging 2016, 5, 126–134. [Google Scholar] [PubMed]
- Bjorkman, M.P.; Finne-Soveri, H.; Tilvis, R.S. Whey protein supplementation in nursing home residents. A randomized controlled trial. Eur. Geriatr. Med. 2012, 3, 161–166. [Google Scholar] [CrossRef]
- Demontiero, O.; Vidal, C.; Duque, G. Aging and bone loss: New insights for the clinician. Ther. Adv. Musculoskelet. Dis. 2012, 4, 61–76. [Google Scholar] [CrossRef]
- Kerstetter, J.E.; O’Brien, K.O.; Caseria, D.M.; Wall, D.E.; Insogna, K.L. The impact of dietary protein on calcium absorption and kinetic measures of bone turnover in women. J. Clin. Endocrinol. Metab. 2005, 90, 26–31. [Google Scholar] [CrossRef]
- Durosier-Izart, C.; Biver, E.; Merminod, F.; van Rietbergen, B.; Chevalley, T.; Herrmann, F.R.; Ferrari, S.L.; Rizzoli, R. Peripheral skeleton bone strength is positively correlated with total and dairy protein intakes in healthy postmenopausal women. Am. J. Clin. Nutr. 2017, 105, 513–525. [Google Scholar] [CrossRef]
- Langsetmo, L.; Shikany, J.M.; Burghardt, A.J.; Cawthon, P.M.; Orwoll, E.S.; Cauley, J.A.; Taylor, B.C.; Schousboe, J.T.; Bauer, D.C.; Vo, T.N.; et al. High dairy protein intake is associated with greater bone strength parameters at the distal radius and tibia in older men: A cross-sectional study. Osteoporos. Int. 2018, 29, 69–77. [Google Scholar] [CrossRef]
- Khalil, D.A.; Lucas, E.A.; Juma, S.; Smith, B.J.; Payton, M.E.; Arjmandi, B.H. Soy protein supplementation increases serum insulin-like growth factor-I in young and old men but does not affect markers of bone metabolism. J. Nutr. 2002, 132, 2605–2608. [Google Scholar] [CrossRef] [PubMed]
- Holm, L.; Olesen, J.L.; Matsumoto, K.; Doi, T.; Mizuno, M.; Alsted, T.J.; Mackey, A.L.; Schwarz, P.; Kjaer, M. Protein-containing nutrient supplementation following strength training enhances the effect on muscle mass, strength, and bone formation in postmenopausal women. J. Appl. Physiol. 2008, 105, 274–281. [Google Scholar] [CrossRef] [PubMed]
- Adolphi, B.; Scholz-Ahrens, K.E.; de Vrese, M.; Acil, Y.; Laue, C.; Schrezenmeir, J. Short-term effect of bedtime consumption of fermented milk supplemented with calcium, inulin-type fructans and caseinphosphopeptides on bone metabolism in healthy, postmenopausal women. Eur. J. Nutr. 2009, 48, 45–53. [Google Scholar] [CrossRef] [PubMed]
- Chevalley, T.; Hoffmeyer, P.; Bonjour, J.-P.; Rizzoli, R. Early serum IGF-I response to oral protein supplements in elderly women with a recent hip fracture. Clin. Nutr. 2010, 29, 78–83. [Google Scholar] [CrossRef]
- Zhu, K.; Meng, X.; Kerr, D.A.; Devine, A.; Solah, V.; Binns, C.W.; Prince, R.L. The effects of a two-year randomized, controlled trial of whey protein supplementation on bone structure, IGF-1, and urinary calcium excretion in older postmenopausal women. J. Bone Miner. Res. 2011, 26, 2298–2306. [Google Scholar] [CrossRef]
- Kerstetter, J.E.; Bihuniak, J.D.; Brindisi, J.; Sullivan, R.R.; Mangano, K.M.; Larocque, S.; Kotler, B.M.; Simpson, C.A.; Cusano, A.M.; Gaffney-Stomberg, E.; et al. The Effect of a Whey Protein Supplement on Bone Mass in Older Caucasian Adults. J. Clin. Endocrinol. Metab. 2015, 100, 2214–2222. [Google Scholar] [CrossRef]
- Bharadwaj, S.; Naidu, A.G.T.; Betageri, G.V.; Prasadarao, N.V.; Naidu, A.S. Milk ribonuclease-enriched lactoferrin induces positive effects on bone turnover markers in postmenopausal women. Osteoporos. Int. 2009, 20, 1603–1611. [Google Scholar] [CrossRef]
- Aoe, S.; Koyama, T.; Toba, Y.; Itabashi, A.; Takada, Y. A controlled trial of the effect of milk basic protein (MBP) supplementation on bone metabolism in healthy menopausal women. Osteoporos. Int. 2005, 16, 2123–2128. [Google Scholar] [CrossRef]
- Narva, M.; Kärkkäinen, M.; Poussa, T.; Lamberg-Allardt, C.; Korpela, R. Caseinphosphopeptides in milk and fermented milk do not affect calcium metabolism acutely in postmenopausal women. J. Am. Coll. Nutr. 2003, 22, 88–93. [Google Scholar] [CrossRef]
- Jackson, C.F.; Wenger, N.K. Cardiovascular disease in the elderly. Rev. Esp. Cardiol. 2011, 64, 697–712. [Google Scholar] [CrossRef]
- Teede, H.J.; Dalais, F.S.; Kotsopoulos, D.; Liang, Y.L.; Davis, S.; McGrath, B.P. Dietary soy has both beneficial and potentially adverse cardiovascular effects: A placebo-controlled study in men and postmenopausal women. J. Clin. Endocrinol. Metab. 2001, 86, 3053–3060. [Google Scholar] [CrossRef]
- Kreijkamp-Kaspers, S.; Kok, L.; Bots, M.L.; Grobbee, D.E.; Lampe, J.W.; van der Schouw, Y.T. Randomized controlled trial of the effects of soy protein containing isoflavones on vascular function in postmenopausal women. Am. J. Clin. Nutr. 2005, 81, 189–195. [Google Scholar] [CrossRef][Green Version]
- Cuevas, A.M.; Irribarra, V.L.; Castillo, O.A.; Yañez, M.D.; Germain, A.M. Isolated soy protein improves endothelial function in postmenopausal hypercholesterolemic women. Eur. J. Clin. Nutr. 2003, 57, 889–894. [Google Scholar] [CrossRef]
- Yoshizawa, M.; Maeda, S.; Miyaki, A.; Misono, M.; Choi, Y.; Shimojo, N.; Ajisaka, R.; Tanaka, H. Additive beneficial effects of lactotripeptides and aerobic exercise on arterial compliance in postmenopausal women. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1899–H1903. [Google Scholar] [CrossRef]
- Yoshizawa, M.; Maeda, S.; Miyaki, A.; Misono, M.; Choi, Y.; Shimojo, N.; Ajisaka, R.; Tanaka, H. Additive beneficial effects of lactotripeptides intake with regular exercise on endothelium-dependent dilatation in postmenopausal women. Am. J. Hypertens. 2010, 23, 368–372. [Google Scholar] [CrossRef]
- Pal, S.; Ellis, V.; Ho, S. Acute effects of whey protein isolate on cardiovascular risk factors in overweight, post-menopausal women. Atherosclerosis 2010, 212, 339–344. [Google Scholar] [CrossRef]
- Pal, S.; Ellis, V. Acute effects of whey protein isolate on blood pressure, vascular function and inflammatory markers in overweight postmenopausal women. Br. J. Nutr. 2011, 105, 1512–1519. [Google Scholar] [CrossRef]
- Hodgson, J.M.; Zhu, K.; Lewis, J.R.; Kerr, D.; Meng, X.; Solah, V.; Devine, A.; Binns, C.W.; Woodman, R.J.; Prince, R.L. Long-term effects of a protein-enriched diet on blood pressure in older women. Br. J. Nutr. 2012, 107, 1664–1672. [Google Scholar] [CrossRef]
- Dangin, M.; Boirie, Y.; Guillet, C.; Beaufrère, B. Influence of the protein digestion rate on protein turnover in young and elderly subjects. J. Nutr. 2002, 132, 3228S–3233S. [Google Scholar] [CrossRef]
- Dangin, M.; Guillet, C.; Garcia-Rodenas, C.; Gachon, P.; Bouteloup-Demange, C.; Reiffers-Magnani, K.; Fauquant, J.; Ballèvre, O.; Beaufrère, B. The rate of protein digestion affects protein gain differently during aging in humans. J. Physiol. 2003, 549, 635–644. [Google Scholar] [CrossRef]
- Giezenaar, C.; Trahair, L.G.; Rigda, R.; Hutchison, A.T.; Feinle-Bisset, C.; Luscombe-Marsh, N.D.; Hausken, T.; Jones, K.L.; Horowitz, M.; Chapman, I.; et al. Lesser suppression of energy intake by orally ingested whey protein in healthy older men compared with young controls. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015, 309, R845–R854. [Google Scholar] [CrossRef]
- Giezenaar, C.; Hutchison, A.T.; Luscombe-Marsh, N.D.; Chapman, I.; Horowitz, M.; Soenen, S. Effect of Age on Blood Glucose and Plasma Insulin, Glucagon, Ghrelin, CCK, GIP, and GLP-1 Responses to Whey Protein Ingestion. Nutrients 2017, 10, 2. [Google Scholar] [CrossRef]
- Giezenaar, C.; Luscombe-Marsh, N.D.; Hutchison, A.T.; Standfield, S.; Feinle-Bisset, C.; Horowitz, M.; Chapman, I.; Soenen, S. Dose-Dependent Effects of Randomized Intraduodenal Whey-Protein Loads on Glucose, Gut Hormone, and Amino Acid Concentrations in Healthy Older and Younger Men. Nutrients 2018, 10, 78. [Google Scholar] [CrossRef]
- Giezenaar, C.; van der Burgh, Y.; Lange, K.; Hatzinikolas, S.; Hausken, T.; Jones, K.L.; Horowitz, M.; Chapman, I.; Soenen, S. Effects of Substitution, and Adding of Carbohydrate and Fat to Whey-Protein on Energy Intake, Appetite, Gastric Emptying, Glucose, Insulin, Ghrelin, CCK and GLP-1 in Healthy Older Men-A Randomized Controlled Trial. Nutrients 2018, 10, 113. [Google Scholar] [CrossRef]
- Xia, S.; Zhang, X.; Zheng, S.; Khanabdali, R.; Kalionis, B.; Wu, J.; Wan, W.; Tai, X. An Update on Inflamm-Aging: Mechanisms, Prevention, and Treatment. J. Immunol. Res. 2016, 2016, 8426874. [Google Scholar] [CrossRef]
- Power-Grant, O.; McCormack, W.G.; De Cap, M.R.; Amigo-Benavent, M.; Fitzgerald, R.J.; Jakeman, P. Evaluation of the antioxidant capacity of a milk protein matrix in vitro and in vivo in women aged 50–70 years. Int. J. Food Sci. Nutr. 2016, 67, 325–334. [Google Scholar] [CrossRef]
- Laviolette, L.; Lands, L.C.; Dauletbaev, N.; Saey, D.; Milot, J.; Provencher, S.; LeBlanc, P.; Maltais, F. Combined effect of dietary supplementation with pressurized whey and exercise training in chronic obstructive pulmonary disease: A randomized, controlled, double-blind pilot study. J. Med. Food 2010, 13, 589–598. [Google Scholar] [CrossRef]
- Sugawara, K.; Takahashi, H.; Kashiwagura, T.; Yamada, K.; Yanagida, S.; Homma, M.; Dairiki, K.; Sasaki, H.; Kawagoshi, A.; Satake, M.; et al. Effect of anti-inflammatory supplementation with whey peptide and exercise therapy in patients with COPD. Respir. Med. 2012, 106, 1526–1534. [Google Scholar] [CrossRef]
- De Aguilar-Nascimento, J.E.; Silveira, B.R.P.; Dock-Nascimento, D.B. Early enteral nutrition with whey protein or casein in elderly patients with acute ischemic stroke: A double-blind randomized trial. Nutrition 2011, 27, 440–444. [Google Scholar] [CrossRef]
- Duff, W.R.D.; Chilibeck, P.D.; Rooke, J.J.; Kaviani, M.; Krentz, J.R.; Haines, D.M. The effect of bovine colostrum supplementation in older adults during resistance training. Int. J. Sport Nutr. Exerc. Metab. 2014, 24, 276–285. [Google Scholar] [CrossRef]
- Stojkovic, V.; Simpson, C.A.; Sullivan, R.R.; Cusano, A.M.; Kerstetter, J.E.; Kenny, A.M.; Insogna, K.L.; Bihuniak, J.D. The Effect of Dietary Glycemic Properties on Markers of Inflammation, Insulin Resistance, and Body Composition in Postmenopausal American Women: An Ancillary Study from a Multicenter Protein Supplementation Trial. Nutrients 2017, 9, 484. [Google Scholar] [CrossRef]
- Bell, K.E.; Snijders, T.; Zulyniak, M.A.; Kumbhare, D.; Parise, G.; Chabowski, A.; Phillips, S.M. A multi-ingredient nutritional supplement enhances exercise training-related reductions in markers of systemic inflammation in healthy older men. Appl. Physiol. Nutr. Metab. 2018, 43, 299–302. [Google Scholar] [CrossRef] [PubMed]
- Vaz Fragoso, C.A.; Gill, T.M. Defining Chronic Obstructive Pulmonary Disease in an Aging Population. J. Am. Geriatr. Soc. 2010, 58, 2224–2226. [Google Scholar] [CrossRef] [PubMed]
- Albertson, T.E.; Louie, S.; Chan, A.L. The diagnosis and treatment of elderly patients with acute exacerbation of chronic obstructive pulmonary disease and chronic bronchitis. J. Am. Geriatr. Soc. 2010, 58, 570–579. [Google Scholar] [CrossRef] [PubMed]
- Engelen, M.P.K.J.; De Castro, C.L.N.; Rutten, E.P.A.; Wouters, E.F.M.; Schols, A.M.W.J.; Deutz, N.E.P. Enhanced anabolic response to milk protein sip feeding in elderly subjects with COPD is associated with a reduced splanchnic extraction of multiple amino acids. Clin. Nutr. 2012, 31, 616–624. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Jonker, R.; Deutz, N.E.P.; Harrykissoon, R.; Zachria, A.J.; Veley, E.A.; Engelen, M.P.K.J. A critical evaluation of the anabolic response after bolus or continuous feeding in COPD and healthy older adults. Clin. Sci. 2018, 132, 17–31. [Google Scholar] [CrossRef]
- Lum, C.; Lo, R.; Ng, K.; Woo, J.; Tang, N.; Fallows, S. A study on whey protein supplement on physical performance and quality of life among elderly patients with chronic obstructive pulmonary disease. Australas. J. Ageing 2007, 26, 168–172. [Google Scholar] [CrossRef]
- Jonker, R.; Deutz, N.E.; Erbland, M.L.; Anderson, P.J.; Engelen, M.P. Effectiveness of essential amino acid supplementation in stimulating whole body net protein anabolism is comparable between COPD patients and healthy older adults. Metab. Clin. Exp. 2017, 69, 120–129. [Google Scholar] [CrossRef]
- Konar, A.; Singh, P.; Thakur, M.K. Age-associated Cognitive Decline: Insights into Molecular Switches and Recovery Avenues. Aging Dis. 2016, 7, 121–129. [Google Scholar] [CrossRef]
- Van der Zwaluw, N.L.; van de Rest, O.; Tieland, M.; Adam, J.J.; Hiddink, G.J.; van Loon, L.J.C.; de Groot, L.C.P.G.M. The impact of protein supplementation on cognitive performance in frail elderly. Eur. J. Nutr. 2014, 53, 803–812. [Google Scholar] [CrossRef]
- Zajac, I.T.; Herreen, D.; Bastiaans, K.; Dhillon, V.S.; Fenech, M. The Effect of Whey and Soy Protein Isolates on Cognitive Function in Older Australians with Low Vitamin B12: A Randomised Controlled Crossover Trial. Nutrients 2018, 11, 19. [Google Scholar] [CrossRef] [PubMed]
- Akazawa, N.; Hamasaki, A.; Tanahashi, K.; Kosaki, K.; Yoshikawa, T.; Myoenzono, K.; Maeda, S. Lactotripeptide ingestion increases cerebral blood flow velocity in middle-aged and older adults. Nutr. Res. 2018, 53, 61–66. [Google Scholar] [CrossRef] [PubMed]
- Scholey, A.B.; Camfield, D.A.; Hughes, M.E.; Woods, W.K.; Stough, C.K.; White, D.J.; Gondalia, S.V.; Frederiksen, P.D. A randomized controlled trial investigating the neurocognitive effects of Lacprodan® PL-20, a phospholipid-rich milk protein concentrate, in elderly participants with age-associated memory impairment: The Phospholipid Intervention for Cognitive Ageing Reversal (PLICAR): Study protocol for a randomized controlled trial. Trials 2013, 14, 404. [Google Scholar] [PubMed]
- Ginaldi, L.; Loreto, M.F.; Corsi, M.P.; Modesti, M.; De Martinis, M. Immunosenescence and infectious diseases. Microbes Infect. 2001, 3, 851–857. [Google Scholar] [CrossRef]
- Akha, A.A.S. Aging and the immune system: An overview. J. Immunol. Methods 2018, 463, 21–26. [Google Scholar] [CrossRef] [PubMed]
- Oh, S.-J.; Lee, J.K.; Shin, O.S. Aging and the Immune System: The Impact of Immunosenescence on Viral Infection, Immunity and Vaccine Immunogenicity. Immune Netw. 2019, 19, e37. [Google Scholar] [CrossRef] [PubMed]
- Gill, H.S.; Rutherfurd, K.J.; Cross, M.L.; Gopal, P.K. Enhancement of immunity in the elderly by dietary supplementation with the probiotic Bifidobacterium lactis HN019. Am. J. Clin. Nutr. 2001, 74, 833–839. [Google Scholar] [CrossRef] [PubMed]
- Gavazzi, G.; Krause, K.-H. Ageing and infection. Lancet Infect. Dis. 2002, 2, 659–666. [Google Scholar] [CrossRef]
- Guillemard, E.; Tondu, F.; Lacoin, F.; Schrezenmeir, J. Consumption of a fermented dairy product containing the probiotic Lactobacillus casei DN-114001 reduces the duration of respiratory infections in the elderly in a randomised controlled trial. Br. J. Nutr. 2010, 103, 58–68. [Google Scholar] [CrossRef]
- Makino, S.; Ikegami, S.; Kume, A.; Horiuchi, H.; Sasaki, H.; Orii, N. Reducing the risk of infection in the elderly by dietary intake of yoghurt fermented with Lactobacillus delbrueckii ssp. bulgaricus OLL1073R-1. Br. J. Nutr. 2010, 104, 998–1006. [Google Scholar] [CrossRef]
- Pawelec, G. Age and immunity: What is “immunosenescence”? Exp. Gerontol. 2018, 105, 4–9. [Google Scholar] [CrossRef] [PubMed]
- Boge, T.; Rémigy, M.; Vaudaine, S.; Tanguy, J.; Bourdet-Sicard, R.; van der Werf, S. A probiotic fermented dairy drink improves antibody response to influenza vaccination in the elderly in two randomised controlled trials. Vaccine 2009, 27, 5677–5684. [Google Scholar] [CrossRef] [PubMed]
- Jackson, L.A.; Neuzil, K.M.; Yu, O.; Benson, P.; Barlow, W.E.; Adams, A.L.; Hanson, C.A.; Mahoney, L.D.; Shay, D.K.; Thompson, W.W. Effectiveness of pneumococcal polysaccharide vaccine in older adults. N. Engl. J. Med. 2003, 348, 1747–1755. [Google Scholar] [CrossRef] [PubMed]
- Freeman, S.L.; Fisher, L.; German, J.B.; Leung, P.S.; Prince, H.; Selmi, C.; Naguwa, S.M.; Gershwin, M.E. Dairy proteins and the response to pneumovax in senior citizens: A randomized, double-blind, placebo-controlled pilot study. Ann. N. Y. Acad. Sci. 2010, 1190, 97–103. [Google Scholar] [CrossRef]
- Schaefer, S.; Hettinga, K.A.; Cullor, J.; German, J.B.; Henrick, B.M. Use of UV Treated Milk Powder to Increase Vaccine Efficacy in the Elderly. Front. Immunol. 2018, 9, 2254. [Google Scholar] [CrossRef] [PubMed]
- Numan, S.C.; Veldkamp, P.; Kuijper, E.J.; van den Berg, R.J.; van Dissel, J.T. Clostridium difficile-associated diarrhoea: Bovine anti-Clostridium difficile whey protein to help aid the prevention of relapses. Gut 2007, 56, 888–889. [Google Scholar] [CrossRef]
- Coker, R.H.; Miller, S.; Schutzler, S.; Deutz, N.; Wolfe, R.R. Whey protein and essential amino acids promote the reduction of adipose tissue and increased muscle protein synthesis during caloric restriction-induced weight loss in elderly, obese individuals. Nutr. J. 2012, 11, 105. [Google Scholar] [CrossRef]
- Ooi, E.M.; Adams, L.A.; Zhu, K.; Lewis, J.R.; Kerr, D.A.; Meng, X.; Solah, V.; Devine, A.; Binns, C.W.; Prince, R.L. Consumption of a whey protein-enriched diet may prevent hepatic steatosis associated with weight gain in elderly women. Nutr. Metab. Cardiovasc. Dis. 2015, 25, 388–395. [Google Scholar] [CrossRef]
- Dhillon, V.S.; Zabaras, D.; Almond, T.; Cavuoto, P.; James-Martin, G.; Fenech, M. Whey protein isolate improves vitamin B12 and folate status in elderly Australians with subclinical deficiency of vitamin B12. Mol. Nutr. Food Res. 2017, 61, 1600915. [Google Scholar] [CrossRef]
- Song, X.; Perez-Cueto, F.J.A.; Bredie, W.L.P. Sensory-Driven Development of Protein-Enriched Rye Bread and Cream Cheese for the Nutritional Demands of Older Adults. Nutrients 2018, 10, 1006. [Google Scholar] [CrossRef]
- Volkert, D.; Beck, A.M.; Cederholm, T.; Cruz-Jentoft, A.; Goisser, S.; Hooper, L.; Kiesswetter, E.; Maggio, M.; Raynaud-Simon, A.; Sieber, C.C.; et al. ESPEN guideline on clinical nutrition and hydration in geriatrics. Clin. Nutr. 2019, 38, 10–47. [Google Scholar] [CrossRef] [PubMed]
- Van Staveren, W.A.; Steijns, J.M.; de Groot, L.C.P.G.M. Dairy products as essential contributors of (micro-) nutrients in reference food patterns: An outline for elderly people. J. Am. Coll. Nutr. 2008, 27, 747S–754S. [Google Scholar] [CrossRef] [PubMed]
- Pamplona, R.; Barja, G. Mitochondrial oxidative stress, aging and caloric restriction: The protein and methionine connection. Biochim. Biophys. Acta (BBA)-Bioenerg. 2006, 1757, 496–508. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Jentoft, A.J.; Baeyens, J.P.; Bauer, J.M.; Boirie, Y.; Cederholm, T.; Landi, F.; Martin, F.C.; Michel, J.-P.; Rolland, Y.; Schneider, S.M.; et al. Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Marzetti, E.; Calvani, R.; Tosato, M.; Cesari, M.; Di Bari, M.; Cherubini, A.; Collamati, A.; D’Angelo, E.; Pahor, M.; Bernabei, R.; et al. Sarcopenia: An overview. Aging Clin. Exp. Res. 2017, 29, 11–17. [Google Scholar] [CrossRef]
- Rodondi, A.; Ammann, P.; Ghilardi-Beuret, S.; Rizzoli, R. Zinc increases the effects of essential amino acids-whey protein supplements in frail elderly. J. Nutr. Health Aging 2009, 13, 491–497. [Google Scholar] [CrossRef]
- Ticinesi, A.; Meschi, T.; Lauretani, F.; Felis, G.; Franchi, F.; Pedrolli, C.; Barichella, M.; Benati, G.; Di Nuzzo, S.; Ceda, G.P.; et al. Nutrition and Inflammation in Older Individuals: Focus on Vitamin D, n-3 Polyunsaturated Fatty Acids and Whey Proteins. Nutrients 2016, 8, 186. [Google Scholar] [CrossRef]
- Van de Bool, C.; Rutten, E.P.A.; van Helvoort, A.; Franssen, F.M.E.; Wouters, E.F.M.; Schols, A.M.W.J. A randomized clinical trial investigating the efficacy of targeted nutrition as adjunct to exercise training in COPD. J. Cachexia Sarcopenia Muscle 2017, 8, 748–758. [Google Scholar] [CrossRef]
- Camfield, D.A.; Owen, L.; Scholey, A.B.; Pipingas, A.; Stough, C. Dairy constituents and neurocognitive health in ageing. Br. J. Nutr. 2011, 106, 159–174. [Google Scholar] [CrossRef]
| Author, Year | Number Participants, Gender | Age (Mean or Range) | Type of Study: Intervention Arms | Main Endpoints | Results |
|---|---|---|---|---|---|
| Paddon-Jones, 2006 [30] | 14, 7 ♀/7 ♂ | 68 | RCT: 15 g WP vs. 15 g EAA | Muscle FSR for 3.5 h after ingestion | Both supplementations stimulated FSR, with greater increase in EAA arm |
| Katsanos, 2008 [31] | 15, 6 ♀/9 ♂ | 60–85 | RCT: 15 g WP vs. 6.72 g WP’s EAA vs. 7.57 WP’s Non-EAA | blood phenylalanine, insulin, glucose concentration, muscle biopsy | WP improves MP accrual through mechanisms beyond its EAA content |
| Koopman, 2009 [32] | 10, ♂ (cross over) | 64 | Case-control study: 35 g intact casein vs. 35 g hydrolyzed casein | blood phenylalanine concentration, muscle biopsy (FSR) | Hydrolysate accelerates protein digestion and absorption, increase AA availability and FSR |
| Pennings, 2011 [33] | 48, ♂ | 74 | RCT: 20 g WP vs. 20 g casein vs. 20 g casein hydrolysate | Postprandial Muscle FSR | MP accretion more effective in WP arm |
| Burd, 2012 [34] | 14, ♂ | 71 | RCT: 20 g micellar casein vs. 20 g WP | Rate of MPS at rest and after exercise | Greater rates of MPS in WP arm |
| Groen, 2012 [35] | 16, ♂ | 74 | RCT: intra-gastric administration during sleep of 400 mL of water with vs. without 40 g casein | BPB, MPS | Casein administration during sleep improves BPB and stimulates MPS |
| Pennings, 2012 [36] | 33, ♂ | 73 | RCT: 10 g vs. 20 g vs. 35 g WP | AA absorption, BPB, MPA | 35 g WP reaches best values in all endpoints |
| Wall, 2013 [37] | 24, ♂ | 74 | RCT: 20 g casein vs. 20 g casein + 2.5 g leucine | MPA | Leucine co-ingestion improves MPA |
| Luiking, 2014 [38] | 19, 10 ♀/9 ♂ | 69 | RCT: 20 g WP vs. 6 g milk protein, both arms after unilateral resistance exercise | MPS | Higher MPS with WP, without further enhance with exercise |
| Churchward-Venne, 2015 [39] | 32, ♂ | 71 | Parallel group study: 25 g casein in milk matrix vs. 25 g casein in water | Post-prandial MPS | Milk matrix delays casein digestion and absorption without affecting MPS |
| Borack, 2016 [40] | 20, ♂ | 55–75 | RCT: 30 g WP isolate vs. 30 g soy-dairy protein blend (25% soy, 25% WP and 50% casein); both arms after resistance exercise | Blood and muscle AA concentration; FSR | No differences in endpoints among arms |
| Gorissen, 2016 [41] | 60, ♂ | 71 | RCT: 35 g WhP vs. 35 g WhPH, vs. 35 g micellar casein vs. 35 g WP vs. 35 g WPH vs. 60 g WhP | Post-prandial AA concentration and MPS | Greater AA concentration after WP, greater MPS after micellar casein |
| Walrand, 2016 [42] | 31, ♂ | 72 | RCT: 10-day period of AP or HP diet followed by ingestion of 15 g or 30 g casein vs. 15 g or 30 g of soluble milk proteins | FSR | Greater increase in FSR after ingestion of soluble milk proteins only in the AP group |
| Kouw, 2017 [43] | 48, ♂ | 72 | RCT: before sleep administration of 40 g casein vs. 20 g casein vs. 20 g casein + 1.5 g leucine vs. placebo | MPS | Ingestion of 40 g casein increases MPS better than other arms |
| Hamarsland, 2019 [44] | 21, 8 ♀/13 ♂ | 74 | RCT: 20 g WP vs. 20 g native WP vs. milk (ingested after 2 h of resistance training) | Serum leucine concentration; FSR | Greater increase in serum leucine in native WP arm, but no difference with WP in FSR (only superior to milk) |
| Author, Year | Number Participants, Gender | Age (Mean or Range) | Duration | Intervention Arms | Main Endpoints | Results |
|---|---|---|---|---|---|---|
| Dideriksen 2011 [52] | 24, 9 ♀/15 ♂ | 68 | acute supplementation | RE + WP (0.45 g/kg) vs. RE + caseinate (0.45 g/kg) | MPS | Increase in MPS, no difference between arms |
| Yang 2012 [53] | 37 ♂ | 71 | acute supplementation | WP 0 g, 10 g, 20 g or 40 g vs. WP same doses + RE | MPS | RE increases MPS at all WP doses with greater extent with 40 g WP |
| Arnarson 2013 [54] | 161, 94 ♀/67 ♂ | 65–91 | 12 weeks | RE + WP (20 g) vs. RE + isocaloric CHO | Lean body mass, strength, physical function | Increase in all endpoints, no difference between arms |
| Gryson 2014 [55] | 48, ♂ | 61 | 16 weeks (sedentary) | MET + total milk proteins (10 g) vs. MET + soluble milk proteins rich in leucine (10 g) | Muscle mass and strength, time to task failure, index of muscle fatigue | Better results in all endpoints with soluble milk proteins + after MET |
| Karelis 2015 [56] | 99, 76 ♀/23 ♂ | 65–88 | 135 days | 20 g of cysteine enrich-WP vs. 20 g casein (both arms in combination with RT) | Body composition (DXA), muscle strength | Muscle strength increases in both arms, additional increasing WP arm |
| Weisgarber 2015 [57] | 12, ♀ | 57 | 10 weeks | RE (high volume) + WP (40 g) vs. RE + placebo | Lean tissue mass, muscle thickness, muscle strength | Increase in muscle thickness and strength, but no difference between arms |
| Thomson 2016 [58] | 179, 99 ♀/80 ♂ | 62 | 12 weeks | RE + high dairy protein (1.2 g/kg) vs. RE +high soy protein (1.2 g/kg) vs. RE + usual protein intake (<1.2 g/kg) | Muscle strength, body composition, physical function, quality of life | Increase in lean mass, physical function and mental health in all arms, increase in strength attenuated in soy arm |
| Mori 2018 [59] | 81, ♀ | 65–80 | 24 weeks | RE + WP (22.3 g/day) vs. RE alone vs. WP alone | Muscle mass, physical function | Higher improvement in all endpoints in RE + WP arm |
| Sugihara 2018 [60] | 31, ♀ | 67 | 12 weeks | RE + WP (35 g) vs. RE + placebo | Muscle strength, hypertrophy, muscle quality | Higher increase in muscle strength and hypertrophy in RE + WP |
| Author, Year | Number Participants, Gender | Age Mean and/or Range | Duration | Intervention Arms | Main Endpoints | Results |
|---|---|---|---|---|---|---|
| Khalil 2002 [83] | 17, ♂ | 65–84 | 3 months | Daily supplementation of 40 g SP vs. 40 g MP | Bone specific ALP activity, urinary deoxypyridinoline excretion | No endpoint difference among arms |
| Holm 2008 [84] | 29, ♀ Postmenopausal | 55 | 24 weeks | 10 g WP + 31 g CHO + 1 g fat + 5 mcg vitamin D + 250 mg calcium vs. 6 g CHO + 12 mg calcium; both arms with ST | BMD with DXA, Osteocalcin, CTx | Increase in BMD and osteocalcin in WP multi-ingredient arm |
| Adolphi 2009 [85] | 85, ♀ postmenopausal | 59 (48–67) | 2 weeks | Bedtime consumption of 175 mL Fm vs. 175 mL Fm + 510 mg Calcium vs. Fm+ 510 mg calcium + 0.175 g CPP + 1.75 g ITF | Nocturnal bone resorption markers | Fm reduced bone resorption independently of further supplementation |
| Chevallley 2010 [86] | 45, ♀ (recent hip fracture) | 81 | 1 week | Daily supplementation of 20 g casein vs. 15 g WP vs. 5 g EAA | Elevation of circulating IGF-1 | Increase in IGF-1 in casein arm supplementation |
| Zhu 2011 [87] | 219, ♀ | 70–80 | 2 years | Daily supplementation of a drink with 30 g WP vs. placebo | BMD with DXA and QCT. IGF-1 level, urinary calcium excretion | Increase in IGF-1 at year 1 and 2 in WP arm, but no effect on bone mass or strength |
| Kerstetter 2015 [88] | 208, 178 ♀/30 ♂ | 70 | 18 months | Supplementation of 45 g WP vs. isocaloric placebo | BMD with DXA, fat free mass | No difference in BMD, better preservation of fat free mass in WP arm |
| Author, Year | Study Type | Intervention | Main Endpoints | Results | Brief Comment |
|---|---|---|---|---|---|
| Numan 2007 [138] | Pilot study | Anti-Clostridium difficile WP concentrate | Prevention of relapse of Clostridium difficile infection | 10% relapse rate in comparison to 20–25% relapse rate in a control contemporary cohort | Waiting for confirmation in RCT |
| Coker 2012 [139] | RCT during caloric restriction | Meal replacement with WP and EAA vs. a standard meal replacement | Weight loss preserving lean tissue (muscle mass) | WP + EAA was effective in weight reduction promoting preferential reduction of adipose tissue | Small sample size (12 subjects) |
| Ooi 2015 [140] | RCT | 30 g WP supplementation vs. a high CHO energy match supplementation | Weight reduction and reduction of hepatic steatosis in women | No difference in weight reduction or hepatic steatosis | WP supplementation may reduce hepatic steatosis despite weight gain |
| Dhillon 2017 [141] | RCT crossover design | WP isolate (50 g) vs. soy protein isolate (50 g) | Bioavailability of folates and Vitamin B12 in elderly with subclinical deficiencies | WP isolate was superior to soy in improving active B12 and folate status | - |
| Song 2018 [142] | Blind sensory analysis | Rye bread and cream cheese enriched with:
| Consumer acceptance | Better acceptance of WP hydrolysate in bread and of WP isolate in cheese | Developing protein enriched food may increase protein intake in elderly but innovation in protein enriched appealing food is challenging |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zanini, B.; Simonetto, A.; Zubani, M.; Castellano, M.; Gilioli, G. The Effects of Cow-Milk Protein Supplementation in Elderly Population: Systematic Review and Narrative Synthesis. Nutrients 2020, 12, 2548. https://doi.org/10.3390/nu12092548
Zanini B, Simonetto A, Zubani M, Castellano M, Gilioli G. The Effects of Cow-Milk Protein Supplementation in Elderly Population: Systematic Review and Narrative Synthesis. Nutrients. 2020; 12(9):2548. https://doi.org/10.3390/nu12092548
Chicago/Turabian StyleZanini, Barbara, Anna Simonetto, Matilde Zubani, Maurizio Castellano, and Gianni Gilioli. 2020. "The Effects of Cow-Milk Protein Supplementation in Elderly Population: Systematic Review and Narrative Synthesis" Nutrients 12, no. 9: 2548. https://doi.org/10.3390/nu12092548
APA StyleZanini, B., Simonetto, A., Zubani, M., Castellano, M., & Gilioli, G. (2020). The Effects of Cow-Milk Protein Supplementation in Elderly Population: Systematic Review and Narrative Synthesis. Nutrients, 12(9), 2548. https://doi.org/10.3390/nu12092548






