Quality of Life: Psychological Symptoms—Effects of a 2-Month Healthy Diet and Nutraceutical Intervention; A Randomized, Open-Label Intervention Trial (RISTOMED)
Abstract
1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Study Design
2.3. Intervention
2.4. Descriptive Variables
2.5. Outcomes
2.6. Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dantzer, R. Cytokine, sickness behavior, and depression. Neurol. Clin. 2006, 24, 441–460. [Google Scholar] [CrossRef] [PubMed]
- Maes, M.; Berk, M.; Goehler, L.; Song, C.; Anderson, G.; Galecki, P.; Leonard, B. Depression and sickness behavior are Janus-faced responses to shared inflammatory pathways. BMC Med. 2012, 10, 66. [Google Scholar] [CrossRef]
- Garvin, P.; Nilsson, E.; Ernerudh, J.; Kristenson, M. The joint subclinical elevation of CRP and IL−6 is associated with lower health-related quality of life in comparison with no elevation or elevation of only one of the biomarkers. Qual. Life Res. 2015, 25, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Heinzelmann, M.; Nilsson, E.; Ernerudh, J.; Kristenson, M. Military personnel with chronic symptoms following blast traumatic brain injury have differential expression of neuronal recovery and epidermal growth factor receptor genes. Front. Neurol. 2014, 5, 198. [Google Scholar] [CrossRef] [PubMed]
- Castanon, N.; Luheshi, G.; Laye, S. Role of neuroinflammation in the emotional and cognitive alterations displayed by animal models of obesity. Front. Neurosci. 2015, 9, 229. [Google Scholar] [CrossRef] [PubMed]
- Tabatabaeizadeh, S.A.; Abdizadeh, M.F.; Meshkat, Z.; Khodashenas, E.; Darroudi, S.; Fazeli, M.; Ferns, G.A.; Avan, A.; Ghayour-Mobarhan, M. There is an association between serum high-sensitivity C-reactive protein (hs-CRP) concentrations and depression score in adolescent girls. Psychoneuroendocrinology 2018, 88, 102–104. [Google Scholar] [CrossRef]
- Tayefi, M.; Shafiee, M.; Kazemi-Bajestani, S.M.R.; Esmaeili, H.; Darroudi, S.; Khakpouri, S.; Mohammadi, M.; Ghaneifar, Z.; Azarpajouh, M.R.; Moohebati, M.; et al. Depression and anxiety both associate with serum level of hs-CRP: A gender-stratified analysis in a population-based study. Psychoneuroendocrinology 2017, 81, 63–69. [Google Scholar] [CrossRef]
- Faugere, M.; Micoulaud-Franchi, J.A.; Faget-Agius, C.; Lancon, C.; Cermolacce, M.; Richieri, R. High C-reactive protein levels are associated with depressive symptoms in schizophrenia. J. Affect. Disord. 2018, 225, 671–675. [Google Scholar] [CrossRef]
- Capuron, L.; Miller, A.H. Immune system to brain signaling: Neuropsychopharmacological implications. Pharmacol. Ther. 2011, 130, 226–238. [Google Scholar] [CrossRef]
- Gopinath, B.; Flood, V.M.; Burlutksy, G.; Louie, J.C.; Mitchell, P. Association between carbohydrate nutrition and prevalence of depressive symptoms in older adults. Br. J. Nutr. 2016, 116, 2109–2114. [Google Scholar] [CrossRef]
- Napoli, N.; Shah, K.; Waters, D.L.; Sinacore, D.R.; Qualls, C.; Villareal, D.T. Effect of weight loss, exercise, or both on cognition and quality of life in obese older adults. Am. J. Clin. Nutr. 2014, 100, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Campisi, J. Chronic inflammation (inflammaging) and its potential contribution to age-associated diseases. J. Gerontol. Biol. Sci. Med. Sci. 2014, 69, S4–S9. [Google Scholar] [CrossRef] [PubMed]
- Ostan, R.; Bene, M.C.; Spazzafumo, L.; Pinto, A.; Donini, L.M.; Pryen, F.; Charrouf, Z.; Valentini, L.; Lochs, H.; Bourdel-Marchasson, I.; et al. Impact of diet and nutraceutical supplementation on inflammation in elderly people. Results from the RISTOMED study, an open-label randomized control trial. Clin. Nutr. 2015, 35, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Valentini, L.; Pinto, A.; Bourdel-Marchasson, I.; Ostan, R.; Brigidi, P.; Turroni, S.; Hrelia, S.; Hrelia, P.; Bereswill, S.; Fischer, A.; et al. Impact of personalized diet and probiotic supplementation on inflammation, nutritional parameters and intestinal microbiota—The “RISTOMED project”: Randomized controlled trial in healthy older people. Clin. Nutr. 2015, 34, 593–602. [Google Scholar] [CrossRef]
- Dai, C.; Zheng, C.Q.; Meng, F.J.; Zhou, Z.; Sang, L.X.; Jiang, M. VSL#3 probiotics exerts the anti-inflammatory activity via PI3k/Akt and NF-kappaB pathway in rat model of DSS-induced colitis. Mol. Cell. Biochem. 2013, 374, 1–11. [Google Scholar] [PubMed]
- Yu, L.; Yan, J.; Sun, Z. D-limonene exhibits anti-inflammatory and antioxidant properties in an ulcerative colitis rat model via regulation of iNOS, COX−2, PGE2 and ERK signaling pathways. Mol. Med. Rep. 2017, 15, 2339–2346. [Google Scholar] [CrossRef]
- Menni, H.B.; Menni, H.B.; Belarbi, M.; Menni, D.B.; Bendiab, H.; Kherraf, Y.; Ksouri, R.; Djebli, N.; Visioli, F. Anti-inflammatory activity of argan oil and its minor components. Int. J. Food Sci. Nutr. 2019, 1–8. [Google Scholar] [CrossRef]
- Adolph, M.; Heller, A.R.; Koch, T.; Koletzko, B.; Kreymann, K.G.; Krohn, K.; Pscheidl, E.; Senkal, M.; Working group for developing the guidelines for parenteral nutrition of The German Association for Nutritional Medicine. Lipid emulsions—Guidelines on Parenteral Nutrition, Chapter 6. Ger. Med. Sci. 2009, 7, Doc22. [Google Scholar]
- Chambers, A.J.; Parise, E.; McCrory, J.L.; Cham, R. A comparison of prediction equations for the estimation of body fat percentage in non-obese and obese older Caucasian adults in the United States. J. Nutr. Health Aging 2014, 18, 586–590. [Google Scholar] [CrossRef]
- Durnin, J.V.; Womersley, J. Body fat assessed from total body density and its estimation from skinfold thickness: Measurements on 481 men and women aged from 16 to 72 years. Br. J. Nutr. 1974, 32, 77–97. [Google Scholar] [CrossRef]
- Lawton, M.P.; Brody, E.M. Assessment of older people: Self-maintaining and instrumental activities of daily living. Gerontologist 1969, 9, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Matthews, D.R.; Hosker, J.P.; Rudenski, A.S.; Naylor, B.A.; Treacher, D.F.; Turner, R.C. Homeostasis model assessment: Insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 1985, 28, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Ware, J.E., Jr.; Sherbourne, C.D. The MOS 36-item short-form health survey (SF−36). I. Conceptual framework and item selection. Med. Care 1992, 30, 473–483. [Google Scholar] [PubMed]
- Radloff, L.S. The CES-D scale: A self report depression scale for research in the general population. Appl. Psychol. Meas. 1977, 1, 385–401. [Google Scholar] [CrossRef]
- Spielberger, C.D. Manual for the State-Trait. Anxiety Inventory (form Y); Consulting Psychologist Press: Palo Alto, CA, USA, 1983. [Google Scholar]
- Cruz-Jentoft, A.J.; Bahat, G.; Bauer, J.; Boirie, Y.; Bruyere, O.; Cederholm, T.; Cooper, C.; Landi, F.; Rolland, Y.; Sayer, A.A.; et al. Writing Group for the European Working Group on Sarcopenia in Older People and the Extended Group for EWGSOP2 Sarcopenia: European consensus on definition and diagnosis: Report of the European Working Group on Sarcopenia in Older People. Age Ageing 2010, 39, 412–423. [Google Scholar] [CrossRef]
- Carroll, A.J.; Carnethon, M.R.; Liu, K.; Jacobs, D.R.; Colangelo, L.A.; Stewart, J.C.; Carr, J.J.; Widome, R.; Auer, R.; Hitsman, B. Interaction between smoking and depressive symptoms with subclinical heart disease in the Coronary Artery Risk Development in Young Adults (CARDIA) study. Health Psychol. 2017, 36, 101–111. [Google Scholar] [CrossRef]
- Sanchez-Villegas, A.; Perez-Cornago, A.; Zazpe, I.; Santiago, S.; Lahortiga, F.; Martinez-Gonzalez, M.A. Micronutrient intake adequacy and depression risk in the SUN cohort study. Eur. J. Nutr. 2018, 57, 2409–2419. [Google Scholar] [CrossRef]
- Opie, R.S.; Itsiopoulos, C.; Parletta, N.; Sanchez-Villegas, A.; Akbaraly, T.N.; Ruusunen, A.; Jacka, F.N. Dietary recommendations for the prevention of depression. Nutr. Neurosci. 2017, 20, 161–171. [Google Scholar] [CrossRef]
- Molendijk, M.; Molero, P.; Ortuno Sanchez-Pedreno, F.; Van der Does, W.; Angel Martinez-Gonzalez, M. Diet quality and depression risk: A systematic review and dose-response meta-analysis of prospective studies. J. Affect. Disord. 2018, 226, 346–354. [Google Scholar] [CrossRef]
- Jacka, F.N.; O’Neil, A.; Opie, R.; Itsiopoulos, C.; Cotton, S.; Mohebbi, M.; Castle, D.; Dash, S.; Mihalopoulos, C.; Chatterton, M.L.; et al. A randomised controlled trial of dietary improvement for adults with major depression (the ‘SMILES’ trial). BMC Med. 2017, 15, 23. [Google Scholar] [CrossRef]
- McCambridge, J.; Witton, J.; Elbourne, D.R. Systematic review of the Hawthorne effect: New concepts are needed to study research participation effects. J. Clin. Epidemiol. 2014, 67, 267–277. [Google Scholar] [CrossRef] [PubMed]
| Mean (SD) | Arm A Healthy Diet (HD) n = 31 | Arm B HD + De Simone Formulation (Probiotics) n = 31 | Arm C HD + Monoterpene AISA 5203-L n = 30 | Arm D HD + Native®Argan Oil n = 33 | ||||
|---|---|---|---|---|---|---|---|---|
| Age (years) | 70.5 | (4) | 69.7 | (3.9) | 70.0 | (3.8) | 71.4 | (3.9) |
| Gender: women, n (%) | 17 | (50) | 16 | (50) | 16 | (50) | 18 | (50) |
| BMI (kg/m2) | 26.9 | (3.4) | 26.7 | (3.8) | 26,8 | (2,9) | 26.7 | (3.5) |
| Waist circumference (cm) | 90.6 | (15.7) | 94.5 | (12.9) | 94.1 | (9.9) | 92.8 | (13.5) |
| Fat mass proportion (% body) | 32.4 | (7.1) | 32.4 | (6.1) | 33.7 | (6.4) | 32.9 | (6.3) |
| Function | ||||||||
| SPPB (0–12) | 11.2 | (1.8) | 11.3 | (1.2) | 11.7 | (0.7) | 11.3 | (1.2) |
| IADL (0–8) | 8.0 | (0) | 7.8 | (0.6) | 8.0 | (0) | 8.0 | (0) |
| Hand-grip strength (kg) | ||||||||
| Women | 20.4 | (4.9) | 21.3 | (3.7) | 21.3 | (4.9) | 22.2 | (7.5) |
| Men | 38.0 | (10.7) | 39.1 | (8.4) | 39.1 | (12.5) | 37.9 | (8.3) |
| MOS SF−36 (0–100) | ||||||||
| MCS | 53.5 | (7.6) | 53.6 | (6.6) | 49.4 | (8.3) | 52.4 | (6.5) |
| PCS | 54.6 | (9.9) | 55.0 | (8.4) | 56.4 | (6.7) | 54.8 | (6.8) |
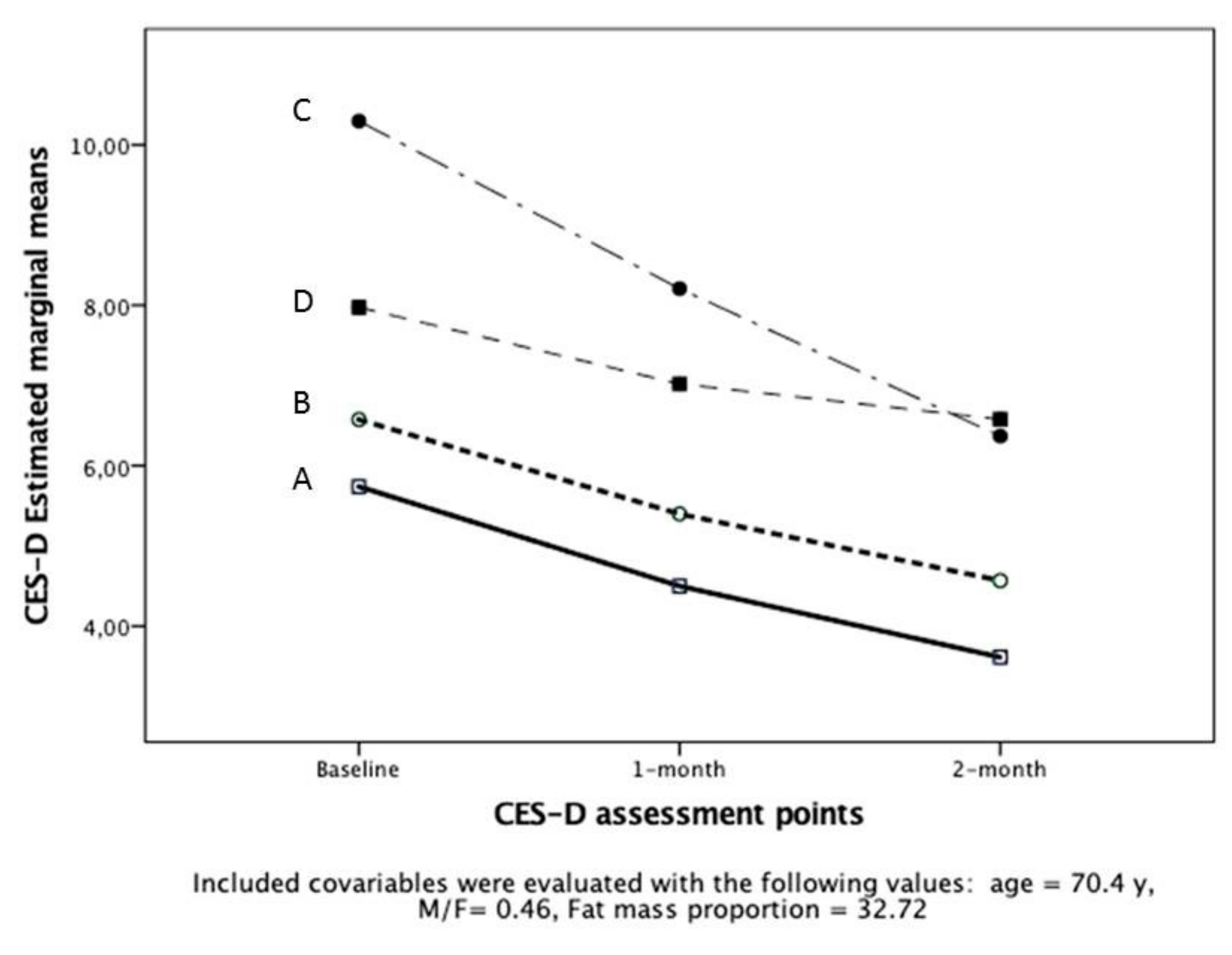
| CES-D (0–60) | 6.7 | (6.7) | 6.7 | (5.4) | 10.7 | (10.2) | 8.1 | (7.7) |
| STAI state (20–80) | 35.7 | (11.4) | 35.6 | (10.1) | 38.2 | (11.6) | 38.4 | (10.1) |
| Fasting glucose (mg/dL) | 94.5 | (23.0) | 92.5 | (12.6) | 95.2 | (13.2) | 93.7 | (17.3) |
| Insulin (µU/mL) | 9.0 | (6.2) | 9.0 | (7.2) | 10.6 | (9.4) | 10.2 | (6.8) |
| HOMA-IR index | 2.3 | (1.9) | 2.2 | (2.1) | 2.2 | (1.1) | 2.6 | (2.4) |
| Cholesterol (mg/dL) | 215 | (42) | 227 | (44) | 229 | (43) | 224 | (38) |
| Triglycerides (mg/dL) | 120 | (64) | 112 | (48) | 110 | (54) | 109 | (48) |
| WBC (103/mmc) | 6.07 | (1.41) | 5.97 | (1.47) | 5.92 | (1.14) | 6.21 | (1.48) |
| hsCRP (mg/L) | 3.6 | (3.6) | 2.9 | (3.8) | 3.6 | (4.9) | 2.5 | (2.6) |
| ESR (mm/h) | 24.4 | (18.2) | 23.5 | (21.2) | 20.9 | (13.6) | 21.2 | (15.7) |
| Fibrinogen (mm/dL) | 377 | (96) | 394 | (104) | 379 | (105) | 359 | (92) |
| TNF-a (pg/mL) | 60.4 | (151.7) | 7.2 | (17.9) | 8.3 | (27.7) | 15.3 | (53.6) |
| IL−6 (pg/mL) | 39.9 | (73.0) | 12.3 | (10.0) | 30.1 | (67.4) | 18.9 | (23.5) |
| Changes from Baseline | ARM A Healthy Diet (HD) n = 31 | p | ARM B HD + De Simone Formulation (Probiotics) n = 31 | p | ARM C HD + Monoterpene AISA 5203-L n = 30 | p | ARM D HD + Native® Argan Oil n = 33 | p |
|---|---|---|---|---|---|---|---|---|
| Anthropometric measures | ||||||||
| BMI (kg/m2) | −1.1% | 0.005 | −0.7% | 0.012 | −0.2% | 0.028 | 0.0% | 0.400 |
| Waist circumference (cm) | −0.9% | 0.112 | 0.0% | 0.190 | 0.0% | 0.449 | 0.0% | 0.602 |
| Body fat mass proportion (%) | +0.4% | 0.295 | +0.4% | 0.223 | −0.1% | 0.667 | +0.4% | 0.190 |
| Lipid profile | ||||||||
| Total cholesterol (mg/dL) | −2.7% | 0.002 | −3.4% | 0.120 | +1.7% | 0.936 | −1.75% | 0.401 |
| Triglycerides (mg/dL) | −5.6% | 0.383 | −2.7% | 0.739 | +10.0% | 0.218 | −4.6% | 0.372 |
| Insulin resistance markers | ||||||||
| Glucose (mg/dL) | −2.1% | 0.029 | 0.0% | 0.682 | −4.3% | 0.005 | −0.9% | 0.147 |
| Insulin (microU/mL) | −1.0% | 0.264 | −2.8% | 0.111 | −20.4% | 0.039 | −1.7% | 0.597 |
| HOMA-IR Index | −17.1% | 0.126 | −7.9% | 0.262 | −23.1% | 0.023 | −2.9% | 0.526 |
| Inflammatory profile | ||||||||
| WBC (103/mmc) | −2.5% | 0.347 | 0.5% | 0.313 | −1.9% | 0.210 | −1.8% | 0.191 |
| hsCRP (mg/L) | −7.7% | 0.688 | 0.0% | 0.712 | −7.1% | 0.313 | 0.0% | 0.757 |
| ESR (mm/hr) | −32.8% | 0.023 | −28.6% | 0.046 | −46.5% | 0.021 | −35.3% | 0.011 |
| Fibrinogen (mg/dL) | +6.4% | 0.456 | −1.4% | 0.532 | −7.0% | 0.082 | +2.9% | 0.137 |
| IL−6 (pg/mL) | +18.2% | 0.127 | +15.9% | 0.073 | −16.1% | 0.217 | −3.7% | 0.636 |
| TNF-α (pg/mL) | 0.0% | 0.322 | 0.0% | 0.225 | 0.0% | 0.765 | 0.0% | 0.614 |
| Changes from Baseline | ARM A Healthy Diet (HD) n = 31 | p | ARM B HD + De Simone Formulation (Probiotics) n = 31 | p | ARM C HD + monoterpene AISA 5203-L n = 30 | p | ARM D HD + Native® Argan oil n = 33 | p |
|---|---|---|---|---|---|---|---|---|
| SPPB (0–12) | 0.0% | 0.856 | 0.0% | 0.297 | 0.0% | 0.538 | 0.0% | 0.672 |
| Handgrip Strength Test (kg) | −0.7% | 0.289 | −1.7% | 0.175 | −3.6% | 0.104 | 0.0% | 0.999 |
| MOS SF36-PCS (0–100) | 1.6% | 0.277 | 1.6% | 0.524 | −1.1% | 0.905 | 0.0% | 0.727 |
| MOS SF36-MCS (0–100) | 2.0% | 0.020 | 0.8% | 0.938 | 2.1% | 0.025 | −0.3% | 0.446 |
| CES-D (0–60) | −40.0% | 0.001 | −32.5% | 0.023 | −42.8% | 0.004 | −33.3% | 0.021 |
| STAI state (20–80) | 0.0% | 0.675 | 0.0% | 0.798 | −2.3% | 0.375 | 0.0% | 0.468 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bourdel-Marchasson, I.; Ostan, R.; Regueme, S.C.; Pinto, A.; Pryen, F.; Charrouf, Z.; d’Alessio, P.A.; Roubaud Baudron, C.; Guerville, F.; Durrieu, J.; et al. Quality of Life: Psychological Symptoms—Effects of a 2-Month Healthy Diet and Nutraceutical Intervention; A Randomized, Open-Label Intervention Trial (RISTOMED). Nutrients 2020, 12, 800. https://doi.org/10.3390/nu12030800
Bourdel-Marchasson I, Ostan R, Regueme SC, Pinto A, Pryen F, Charrouf Z, d’Alessio PA, Roubaud Baudron C, Guerville F, Durrieu J, et al. Quality of Life: Psychological Symptoms—Effects of a 2-Month Healthy Diet and Nutraceutical Intervention; A Randomized, Open-Label Intervention Trial (RISTOMED). Nutrients. 2020; 12(3):800. https://doi.org/10.3390/nu12030800
Chicago/Turabian StyleBourdel-Marchasson, Isabelle, Rita Ostan, Sophie C Regueme, Alessandro Pinto, Florence Pryen, Zoubida Charrouf, Patrizia A d’Alessio, Claire Roubaud Baudron, Florent Guerville, Jessica Durrieu, and et al. 2020. "Quality of Life: Psychological Symptoms—Effects of a 2-Month Healthy Diet and Nutraceutical Intervention; A Randomized, Open-Label Intervention Trial (RISTOMED)" Nutrients 12, no. 3: 800. https://doi.org/10.3390/nu12030800
APA StyleBourdel-Marchasson, I., Ostan, R., Regueme, S. C., Pinto, A., Pryen, F., Charrouf, Z., d’Alessio, P. A., Roubaud Baudron, C., Guerville, F., Durrieu, J., Donini, L. M., Franceschi, C., & Valentini, L. (2020). Quality of Life: Psychological Symptoms—Effects of a 2-Month Healthy Diet and Nutraceutical Intervention; A Randomized, Open-Label Intervention Trial (RISTOMED). Nutrients, 12(3), 800. https://doi.org/10.3390/nu12030800






